Abstract
Introduction
R
The study describes a novel lipophilic reactive oxygen species (ROS) probe, which performed better than popular small molecule probes for ROS measurements in primary isolated cells (pancreatic acinar cells). The probe was particularly effective for resolving near-membrane ROS responses and was used to reveal dynamic changes of ROS in response to inducers of acute pancreatitis (including novel effects of pyocyanin and L-ornithine).
We selected pancreatic acinar cells to test the performance of the new ROS probe in resolving ROS responses triggered by inducers of acute pancreatitis (AP). One common property of the inducers of AP is that they trigger Ca2+ signals and prolonged Ca2+ elevation (Ca2+ overload), which is of paramount importance for the damage of acinar cells in conditions of AP (reviewed in Refs. 39, 50, 51, 62, 63). The relationship between Ca2+ signaling and ROS in acinar cells is complicated. Moderate mitochondrial ROS production seems to be important for the initiation of physiological Ca2+ signals and regulation of secretion in these cells (18). Ca2+ increase is required for taurolithocholic acid 3-sulfate (TLC-S)-induced ROS response (15). Oxidation inhibits the function of important Ca2+ influx channels (Orai channels) (12) and therefore could be protective against Ca2+ overload. Conversely, ROS inhibits plasma membrane Ca2+ ATPases and could therefore facilitate Ca2+ overload and cell damage (1). The amplitude and duration of ROS responses are probably critical in determining if these responses are going to be physiologically beneficial, protective, or damaging.
Mitochondria are important sources of ROS in many cell types, including pancreatic acinar cells (15, 18, 48). In our previous studies, we demonstrated the effect of inducers of pancreatitis on the mitochondria of these cells (15, 69, 70). Monitoring mitochondrial properties [e.g., mitochondrial membrane potential (ΔΨ) and mitochondrial NAD(P)H concentration] and application of mitochondrial inhibitors could therefore help to reveal the mitochondrial component of ROS responses.
Although the generation of ROS after the induction of AP is firmly established in whole pancreas (the evidence for this is based on detecting lipid peroxidation products and protein carbonyls, on monitoring decreases in the antioxidant capacity, and on measurements employing spin-resonance spectroscopy (40, 68), sensing ROS in individual acinar cells upon physiological or pathological stimulation has been more challenging [reviewed in Ref. (20)].
Oxidative stress is considered to be a contributing factor in the pathogenesis of acute and chronic pancreatitis (reviewed in Refs. 14, 52, 56); however, the evidence for the significance of ROS in the initiation and development of pancreatitis is controversial. Strong depletion of glutathione in pancreatic tissue has been reported in a cerulein-induced model of AP and interpreted as a reflection of ROS contribution to the pathogenesis of this disease (44, 47); however, another study concluded that the depletion of glutathione is “neither early nor critical” for the development of cerulein-induced AP (27). Moreover, the usage of antioxidants has produced disappointing results in some clinical trials (5, 59). ROS have been shown to potentiate the release of cytochrome c from the mitochondria of pancreatic acinar cells and by this mechanism influence apoptosis in these cells (48). This is important since in conditions of AP, apoptosis is considered to be protective (28, 33, 57); reviewed in Ref. (6). Recently, it has been shown that ROS production in pancreatic acinar cells can reduce the pancreatic acinar cell necrosis and increase the apoptosis caused by bile acids, also suggesting a protective action of ROS (15). In this previous study, we used commercially available ROS probes and successfully resolved ROS responses induced by TLC-S (15), but we had difficulties in resolving and/or characterizing ROS changes upon application of other inducers of AP. This was an important motivation for the development of a new probe with improved sensitivity. The uncertainty of the role of ROS in the etiology of AP and the recent focus on ROS in acinar cells made this cell type (in conjunction with chemical inducers of AP) a promising platform for testing the new probe.
Results
H2RB-C18–lipophilic ROS-sensitive probe
Dihydrorhodamine B octadecyl ester (H2RB-C18) was prepared from the parent compound rhodamine B octadecyl ester (RB-C18) by a standard sodium borohydride (NaBH4) reduction procedure (10, 65) (Fig. 1A). Within seconds of the addition of NaBH4, the dimethyl sulfoxide (DMSO) solution of RB-C18 lost its red color, indicating the conversion of RB-C18 into H2RB-C18. ROS, produced as a result of the Fenton reaction, oxidized H2RB-C18 back into RB-C18 (this is revealed by the increase of fluorescence). ROS-induced oxidation of dihydrorhodamine123 (H2R123) showed a similar response in a cell-free solution, confirming that H2RB-C18 is a ROS-sensitive probe (Fig. 1A, right panel). The new probe was not sensitive to hydrogen peroxide (H2O2) per se (Fig. 1A and Supplementary Fig. S1A; Supplementary Data are available online at
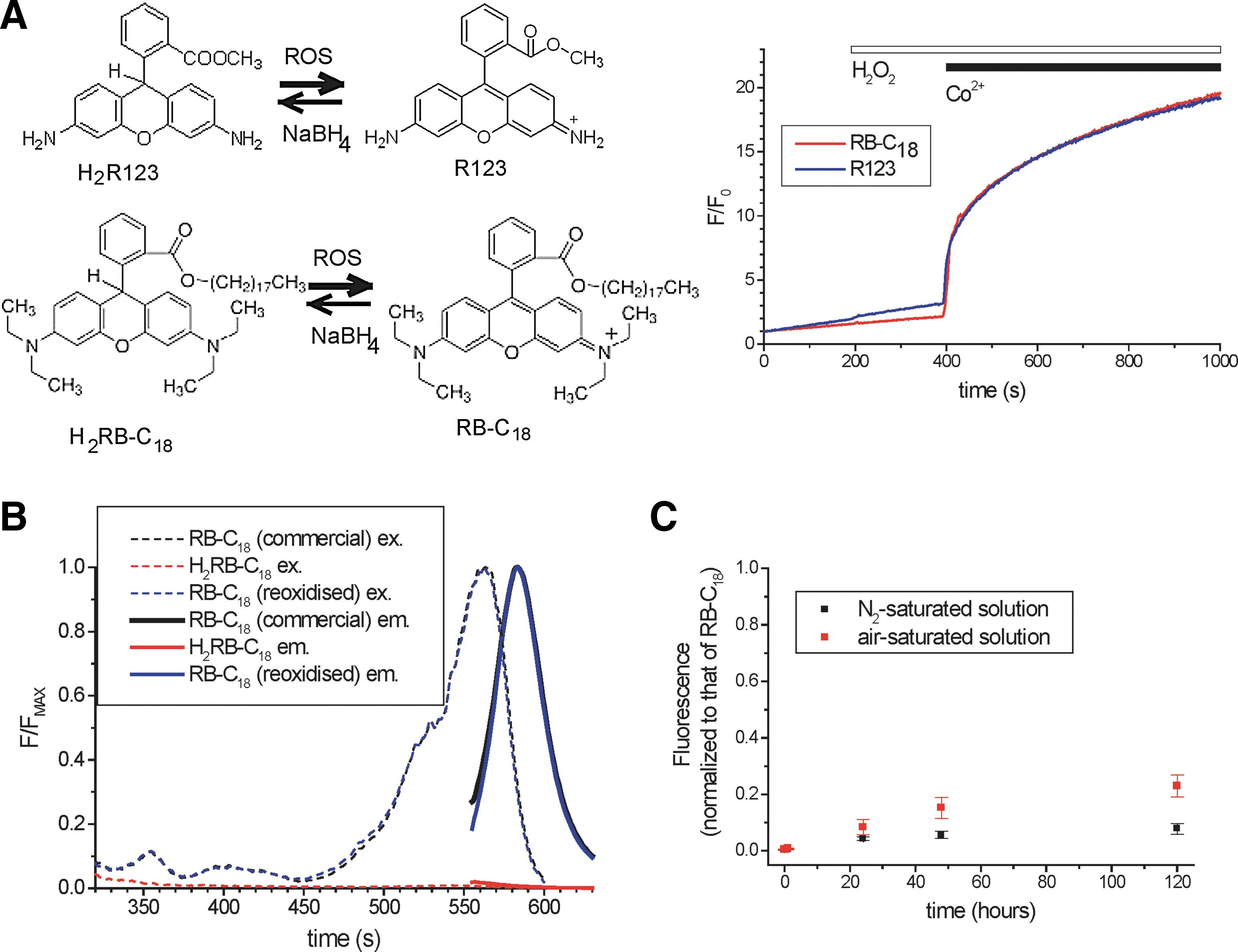
Importantly, the fluorescence of H2RB-C18 in a cell-free system was not changed by the addition of substances that caused significant fluorescence changes in cells loaded with the probe: 500 μM TLC-S, 20 mM L-ornithine, or 50 μM pyocyanin (data not shown).
The excitation and emission fluorescence spectra of RB-C18 and H2RB-C18 are shown in Figure 1B. The spectra of the commercial RB-C18 (purchased from Sigma-Aldrich and used to produce H2RB-C18 in our experiments) largely overlap with the spectra of the product of oxidation of H2RB-C18 (Fig. 1B). H2RB-C18 is reasonably stable and can be used for a few hours after formation by NaBH4 reduction; the stability of H2RB-C18 in deoxygenated solution and in solution exposed to atmospheric oxygen is illustrated in Figure 1C.
Fluorescence of the cells loaded with the parent (oxidized) compound RB-C18 and with rhodamine B (RB: the same fluorophore but without the lipophilic tail) is shown in Figure 2A and B (see also Supplementary Fig. S3 for RB-C18 fluorescence distribution in an acinar cell cluster). Local photobleaching of RB-C18 by UV light leads to a much slower recovery of fluorescence than the recovery following the local photobleaching of RB (Fig. 2A, B). The slow diffusion of RB-C18 can be explained by the lipophilic nature of this compound. This property is probably responsible for the increased sensitivity of the new probe in the cellular environment (see next section).
H2RB-C18 is a highly efficient probe for detecting cellular ROS responses
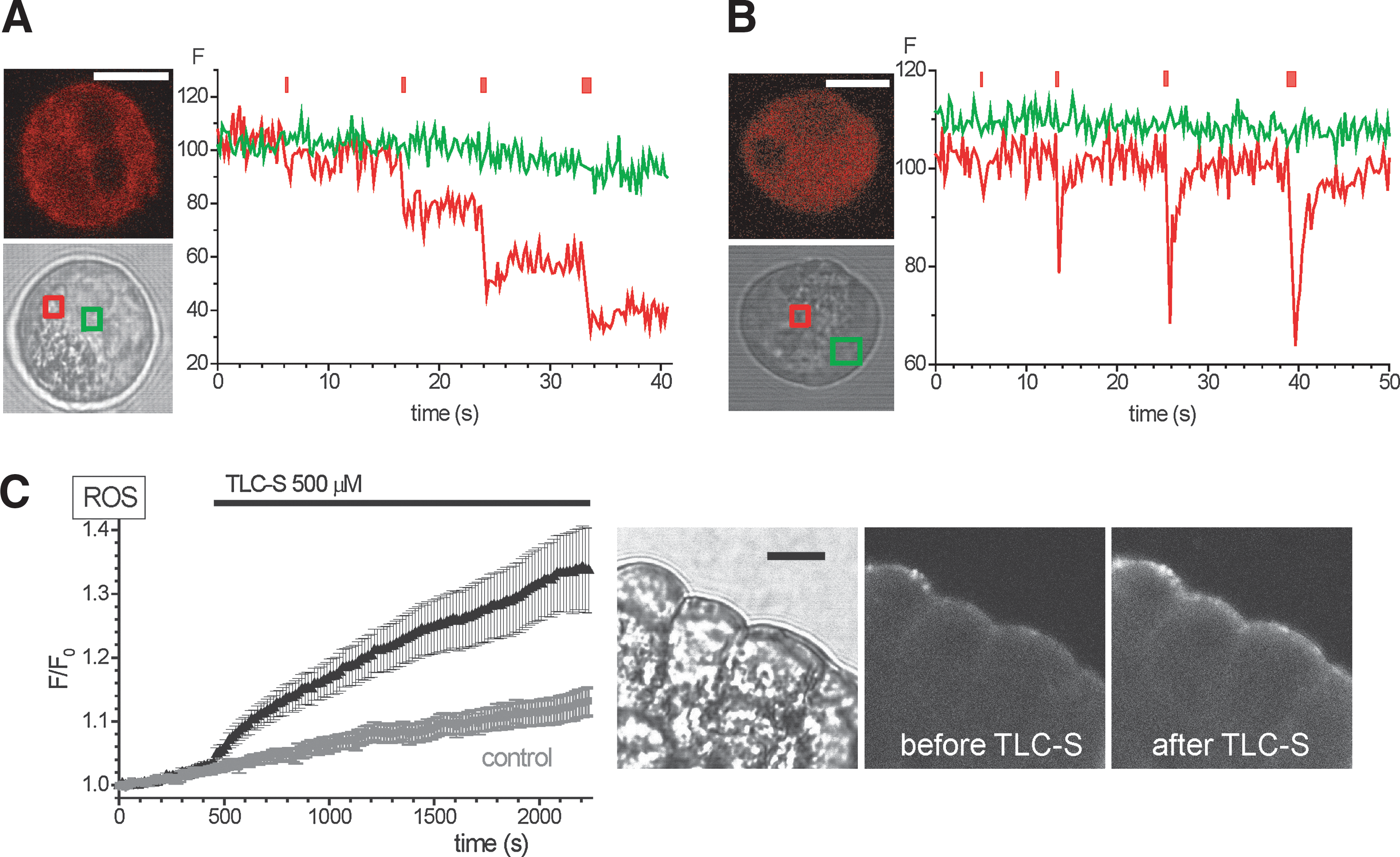
Using 5-(and-6)-chloromethyl-2′,7′-dichlorodihydrofluorescein diacetate [CM-H2DCF(DA)], we have recently reported ROS responses induced by the bile acid TLC-S in pancreatic acinar cells (15). The new probe H2RB-C18, loaded into acinar cells, also produced clearly resolvable response to 500 μM TLC-S (Fig. 2C). Furthermore, the amplitude of this response was considerably larger than that recorded with CM-H2DCF [see Ref. (15)], suggesting that H2RB-C18 is a more sensitive indicator. In unstimulated cells, the fluorescence of H2RB-C18 was nonuniform with a stronger peripheral (plasma membrane) component and weaker fluorescence in the central areas of the cell. The preferential peripheral localization of fluorescence was also observed after stimulation of H2RB-C18-loaded cells with TLC-S (Fig. 2C).
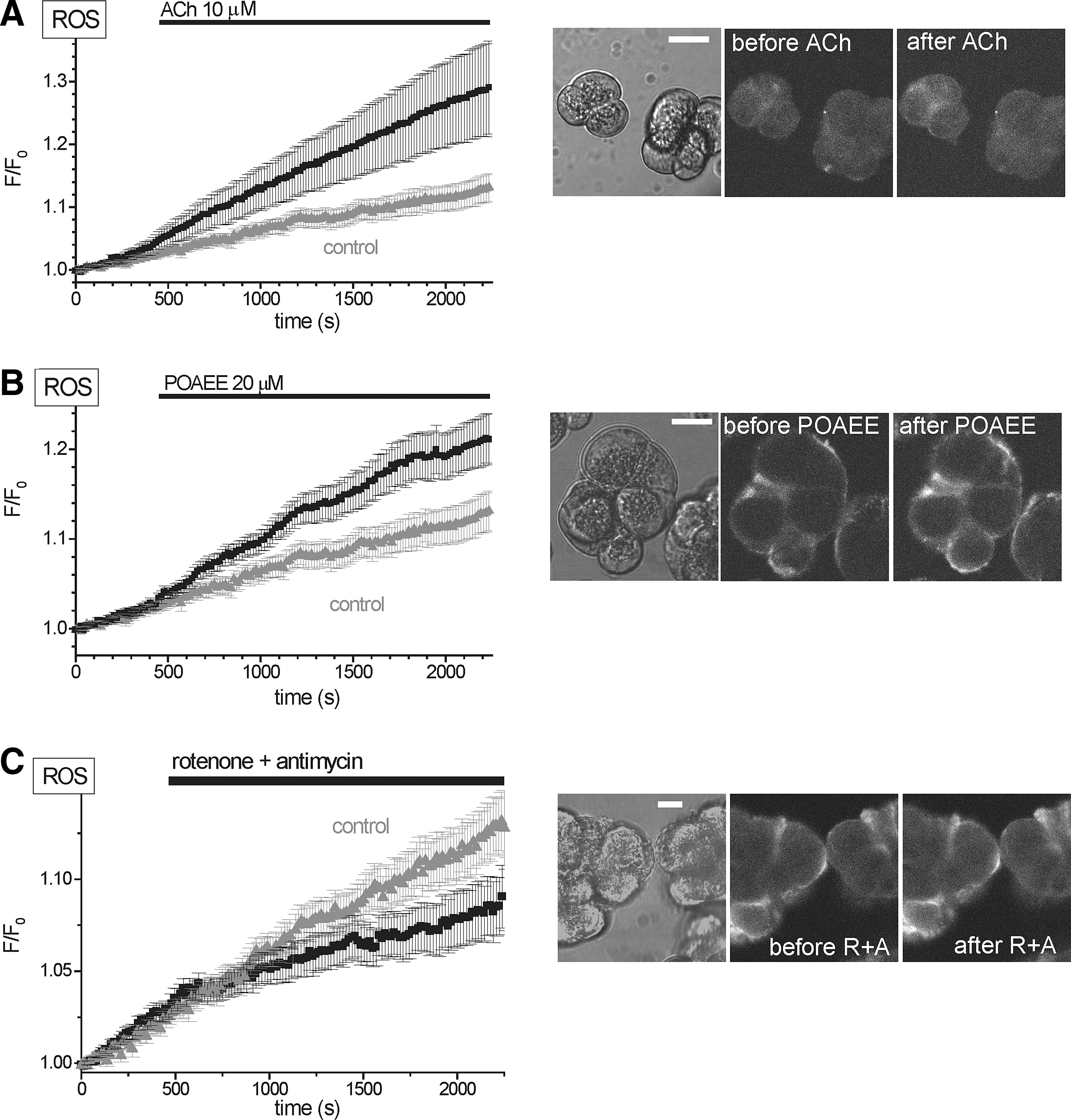
Increased H2RB-C18 fluorescence was also detected in acinar cells stimulated by acetylcholine (ACh) (Fig. 3A) and in cells treated with palmitoleic acid ethyl ester (POAEE) (Fig. 3B). These experiments further confirm the usability of the new probe for ROS measurements in acinar cells. We also tested the effect of mitochondrial inhibitors on ROS production in unstimulated acinar cells and observed that the treatment with rotenone plus antimycin showed a tendency to reduce the rate of the basal rise of fluorescence (Fig. 3C); however, the basal rise of fluorescence was slow, the difference was not statistically significant, and this phenomenon was not investigated further.
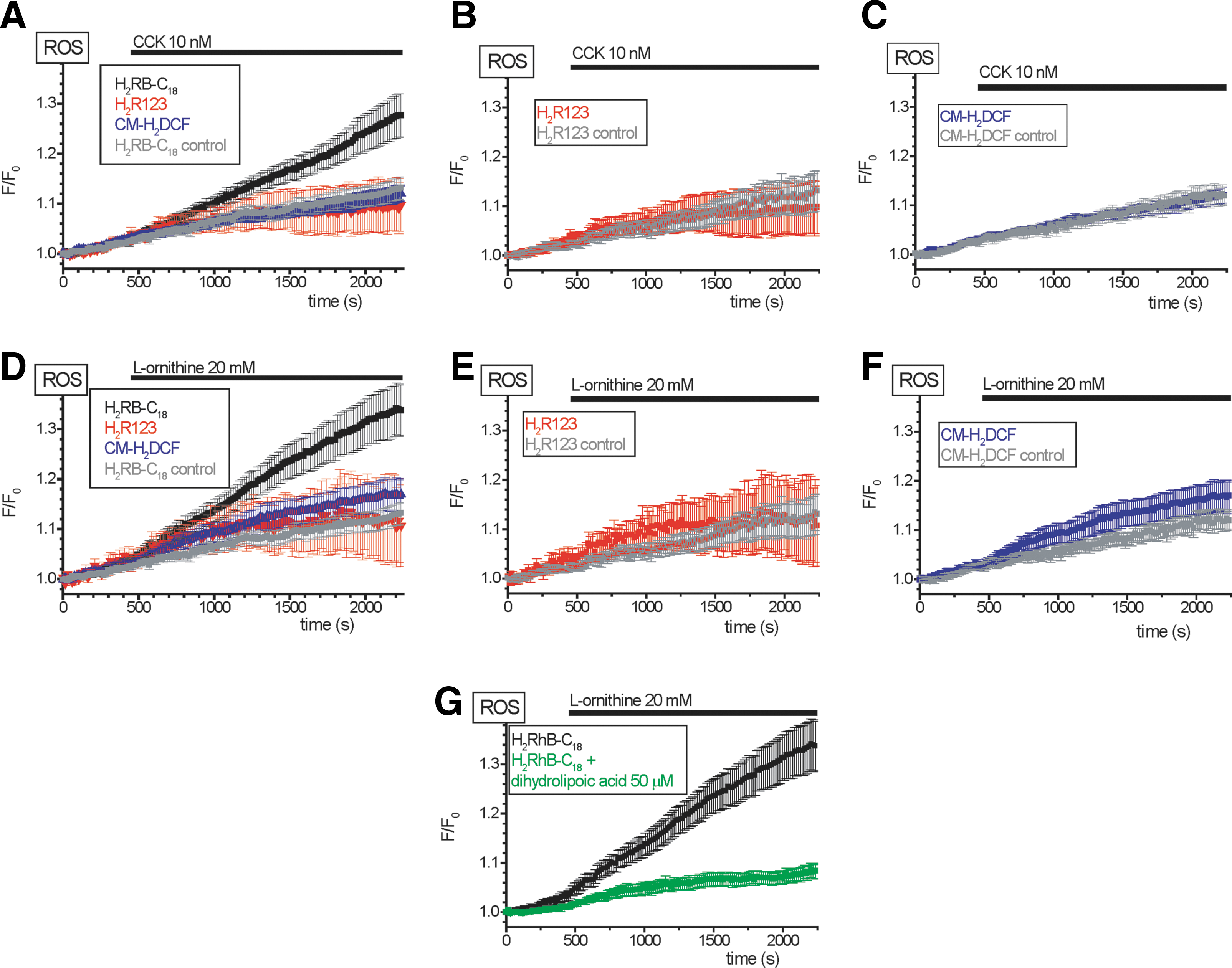
The increased sensitivity of H2RB-C18 allowed us to detect the ROS changes induced by 10 nM cholecystokinin (CCK), which were not resolvable with CM-H2DCF or H2R123 (Fig. 4A–C). The high sensitivity of H2RB-C18 for detection of ROS changes was also obvious from experiments with L-ornithine; H2RB-C18 showed clearly resolvable responses to the application of this amino acid (Fig. 4D; see also Fig. 6D), while the changes of fluorescence of CM-H2DCF and H2R123 were much less pronounced (Fig. 4E, F). The strong response to L-ornithine recorded in cells loaded with H2RB-C18 was almost completely eliminated by an antioxidant dihydrolipoic acid (compare black and green traces in Fig. 4G) confirming that the H2RB-C18 response to L-ornithine develops as the result of ROS generation. Notably, cells loaded with H2RB-C18 did not display obvious indications of cellular damage (no blebbing and no vacuolization). Basal oxygen consumption rate (OCR) and the rates in the presence of Oligomycin and FCCP were also unchanged (Supplementary Fig. S4). We concluded from these data that H2RB-C18 is well tolerated by the acinar cells and, under our experimental conditions, provides improved sensitivity for the detection of cellular ROS changes. We therefore decided to employ this new probe to characterize the effects of the putative ROS-producing damaging agents (pyocyanin and L-ornithine) on pancreatic acinar cells.
Pyocyanin-induced ROS responses measured with H2RB-C18
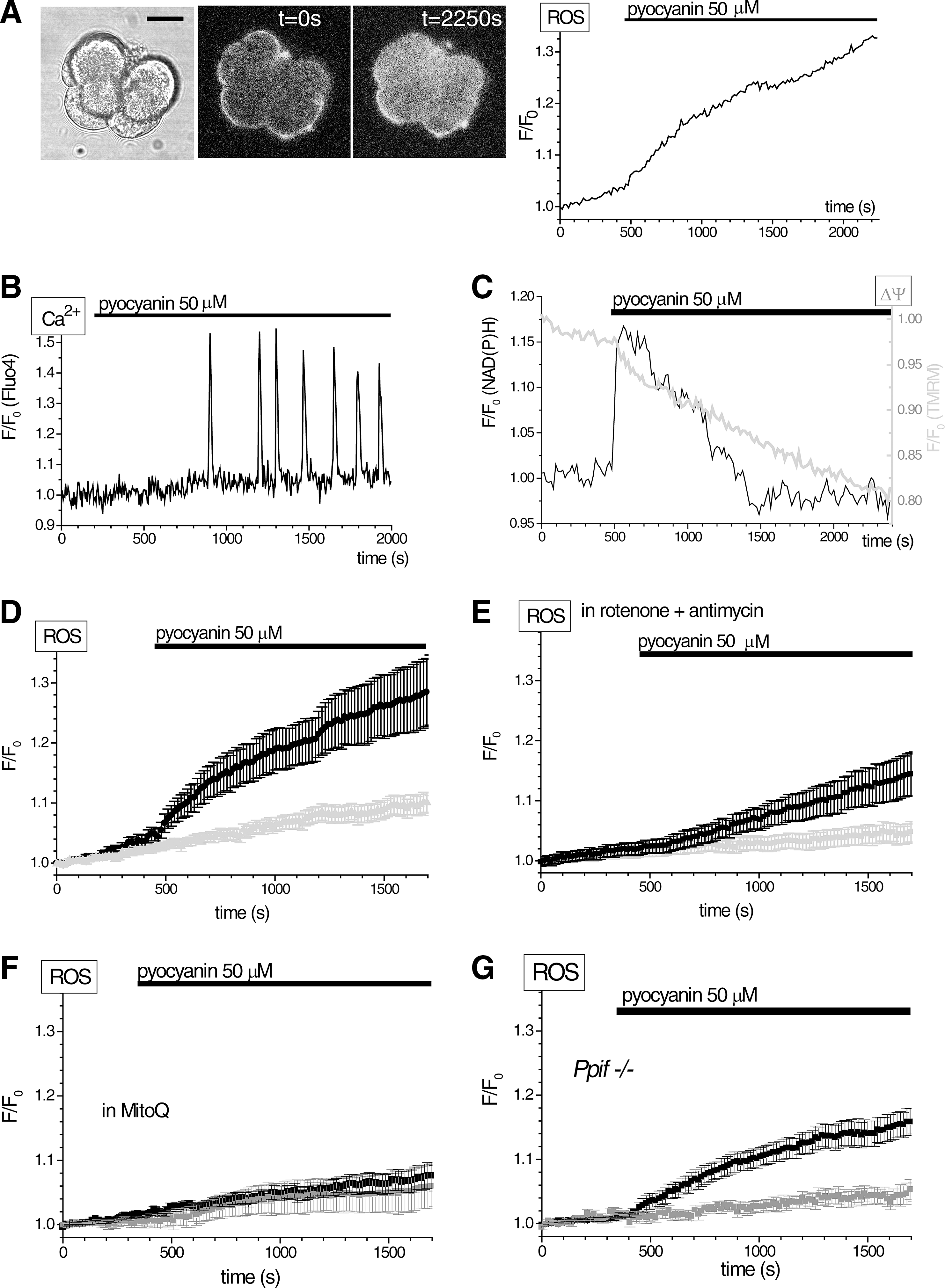
Pseudomonas aeruginosa, which has been shown to infect the pancreas in AP, cystic fibrosis, pancreatic transplantation, and endoscopic retrograde cholangiopancreatography receiving patients (9, 13, 42, 67), can produce copious amounts of pyocyanin (17, 73). Local concentrations of pyocyanin can reach 100 μM or even higher (17, 73). Unlike the action of other cytotoxins from P. aeruginosa (37), the effects of pyocyanin on pancreatic acinar cells have not been described. Using our H2RB-C18 dye, we observed substantial generation of ROS in response to 50 μM of pyocyanin (Fig. 5A). Pyocyanin also triggered [Ca2+]i oscillations (Fig. 5B), mitochondrial depolarization, and transient increases in mitochondrial NAD(P)H (Fig. 5C). The pyocyanin-induced ROS production had two components—an initial rapid rise followed by a slow increase (Fig. 5D). The initial rapid rise was largely suppressed by blocking the mitochondrial electron transport chain (ETC) by rotenone (5 μM) plus antimycin (10 μM) (compare Fig. 5D, E). The slow rise, however, was observed in the presence of the ETC inhibitors (Fig. 5E). Therefore, the experiments with rotenone and antimycin revealed ETC-dependent (the major contributor to the initial rapid rise) and ETC-independent (the slow but resolvable increase of fluorescence) components of the response to pyocyanin. Mitochondria-targeted antioxidant MitoQ (34) effectively suppressed fluorescence changes induced by pyocyanin (Fig. 5F). The pyocyanin response was clearly observed in pancreatic acinar cells from Ppif−/− animals, suggesting that mitochondrial permeability transition (MPTP) is not essential for the pyocyanin response (Fig. 5G). The Ppif gene codes for cyclophilin D (peptidyl-prolyl isomerize known to regulate opening of the MPTP). These data indicate that pyocyanin induces significant ROS responses accompanied by prominent changes in NAD(P)H and ΔΨ.
Characterizing ROS responses induced by L-ornithine
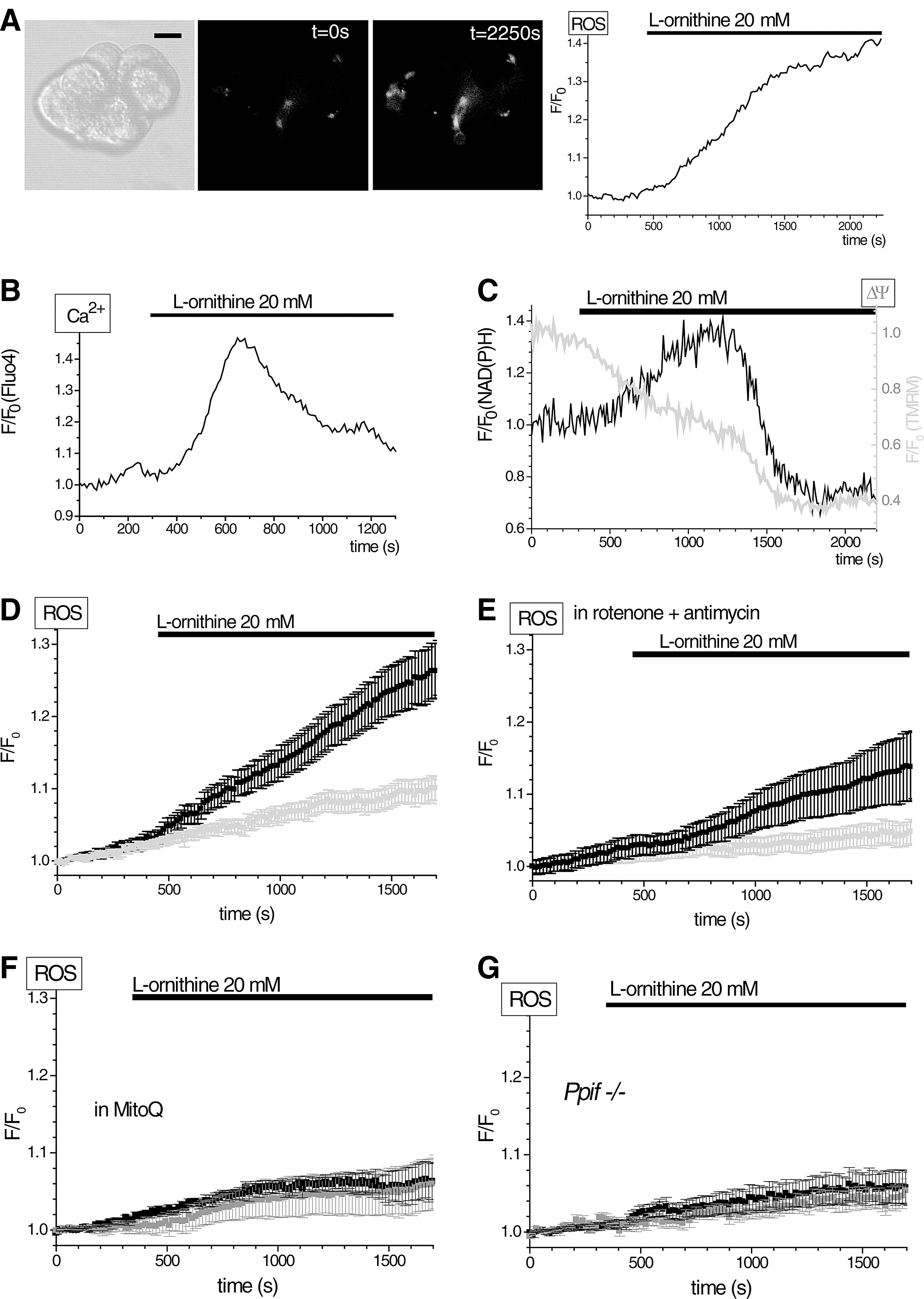
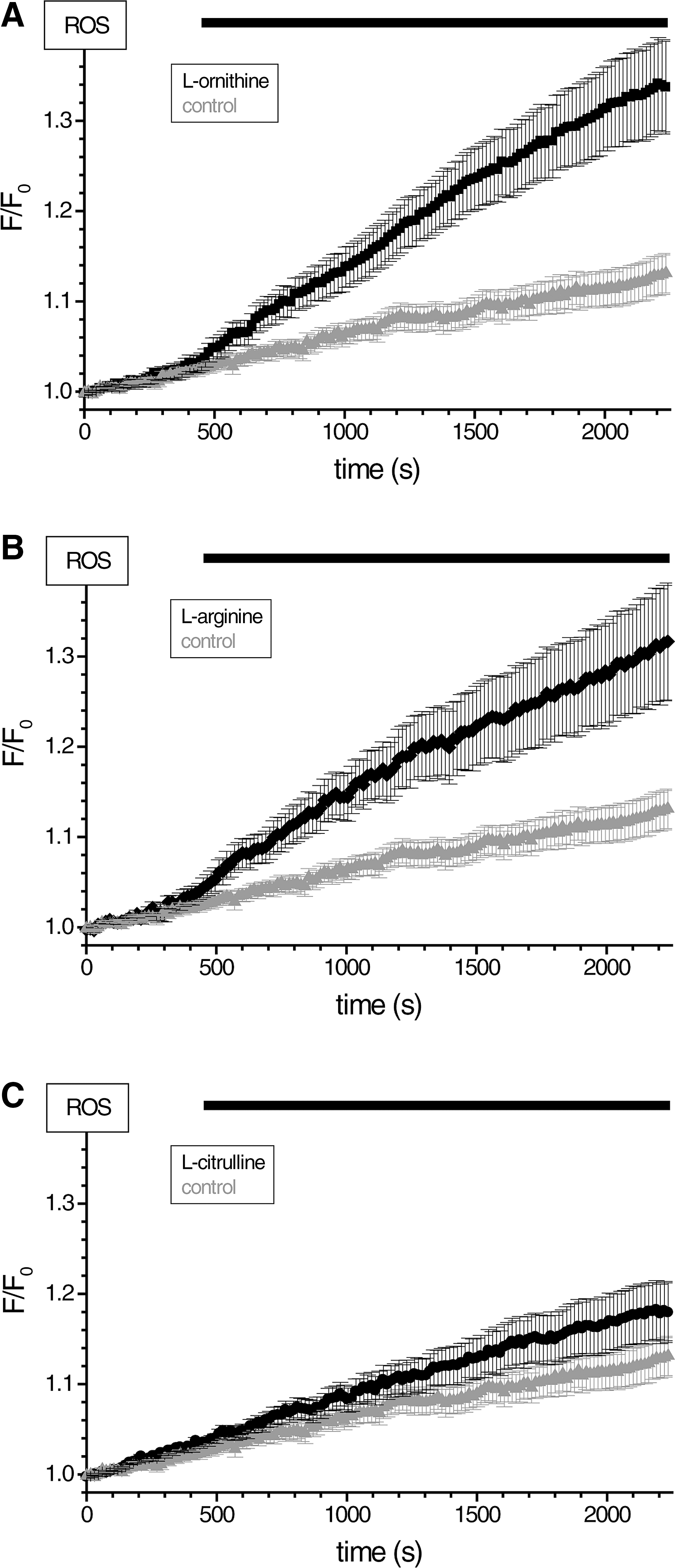
In this part of the study, we used the H2RB-C18 probe to detect the ROS changes induced by basic amino acids. Basic amino acids, at doses of a few grams per kilogram body weight, trigger AP in rodents (8, 23, 46, 53). Recently, it was suggested that L-arginine, which is most often used in these animal models of AP, may act through another basic amino acid L-ornithine (53). Using H2RB-C18 dye, we detected ROS production induced by 20 mM L-ornithine (Fig. 6A, D), which is close to the dose of this amino acid used in the rat model of AP (53). L-ornithine also caused transient elevation of [Ca2+]i (Fig. 6B) in a substantial proportion of tested cells (26/74). Other types of [Ca2+]i responses to L-ornithine included (Supplementary Fig. S5) short spikes (5/74) and sustained elevations (35/74). Finally, 8 of the 74 cells did not display any [Ca2+]i changes upon L-ornithine application. L-ornithine also caused mitochondrial depolarization (Fig. 6C) and a transient increase in NAD(P)H levels (Fig. 6C). A significant proportion of the ROS response to L-ornithine was eliminated when cells were treated with rotenone and antimycin, suggesting that the ETC is important for the L-ornithine response (Fig. 6D, E). MitoQ effectively suppressed the H2RB-C18 fluorescence changes induced by L-ornithine (Fig. 6F). The response to L-ornithine was also strongly inhibited in pancreatic acinar cells from Ppif−/− animals (Fig. 6G). These data show that L-ornithine can produce potentially harmful calcium and mitochondrial responses and that the L-ornithine-induced ROS response has significant mitochondria-dependent component. L-arginine [another inducer of AP (23, 46, 53)] produced equipotent ROS responses to those of L-ornithine, whereas L-citrulline was less active (compare Fig. 7A with B and C).
Calcium dependency of responses to pyocyanin and L-ornithine
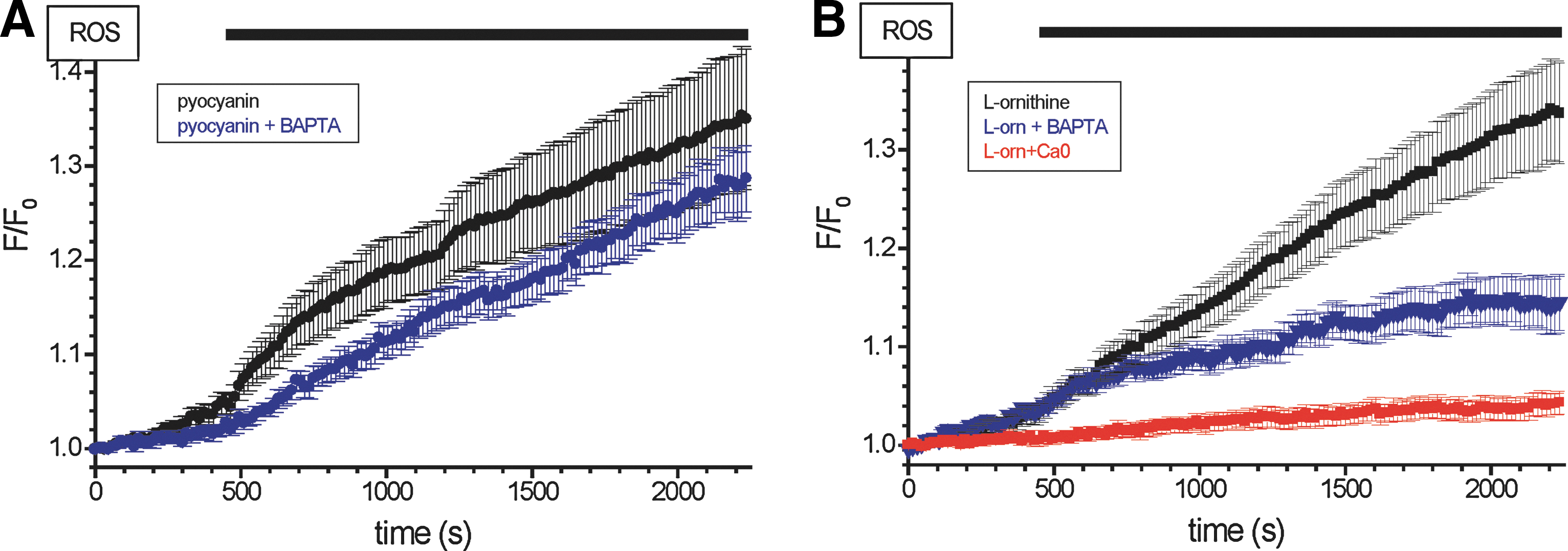
Pretreatment of the acinar cells with 30 μM 1,2-Bis(2-aminophenoxy)ethane-N,N,N′,N′-tetraacetic acid tetrakis (acetoxymethyl ester) (BAPTA-AM) (for 30 min) had a mild effect on the H2RB-C18 fluorescence response to pyocyanin (not statistically significant for the majority of time points, see Fig. 8A), indicating that the rise in [Ca2+]i is unlikely to be essential for the ROS response induced by pyocyanin. However, the pretreatment of the acinar cells with 30 μM BAPTA-AM (for 30 min) significantly suppressed the L-ornithine-induced ROS response (Fig. 8B), suggesting that in this case the ROS production requires [Ca2+]i increase. Interestingly, the removal of Ca2+ from the extracellular solution also suppressed the H2RB-C18 fluorescence response to L-ornithine (Fig. 8B), confirming the notion that Ca2+ signaling is important for this type of ROS response.
Discussion
The main technical advance in this study was the introduction of a new lipophilic ROS indicator, which preferentially reports ROS at membrane regions (and particularly the plasma membrane). The main findings were demonstration of pyocyanin-induced ROS production and characterization of ROS responses induced by the basic amino acids L-ornithine and L-arginine.
Difficulties in measuring ROS and reactive nitrogen species production in live cells using fluorescent probes are compounded by water-soluble antioxidants, such as glutathione and ascorbate, present in the cytoplasm in millimolar quantities. The membrane fraction contains alternative antioxidant system (2, 58). The relative efficiency of the cytosolic and membranous antioxidant systems is difficult to assess, but we hypothesize that the slower diffusion of antioxidants in membranes could make the integrative ROS probes (like reduced fluorescein and rhodamine derivatives) more sensitive if these could be targeted to the membrane compartments. We also considered that the membrane-localized probe will diffuse slower than water-soluble ROS sensors (and therefore could highlight the specific regions of ROS production). The indicator H2RB-C18, introduced in this study, is targeted to membrane compartments by its lipophilic C18 tail. The ability of molecules containing long lipophilic tails to flip in the plasma membrane and to translocate into internal membranes is determined by the properties of the part attached to the tail. For example, FIP-18 (lipophilic analogue of Ca2+ indicator Indo 1) effectively stains intracellular membranes, while Calcium Green C-18 stains plasma membrane and does not flip (11). The H2RB is expected to be electrically neutral and therefore should not impede flipping and intracellular localization of the sensory part of the probe. Unlike other known lipophilic redox probes [such as BODIPY-based lipid peroxidation sensors (2)], it also benefits from the superior dynamic range common to ROS probes from the dihydrorhodamine family (which increase the quantum yield upon oxidation by two orders of magnitude). In conditions of our experiments, H2RB-C18 showed better sensitivity to ROS in a cellular environment than the popular cytosolic ROS indicator H2CMDCF, in response to at least two stimuli: 10 nM CCK and 20 mM L-ornithine. Furthermore, it also produced larger responses to stimulation with TLC-S. We assume that this increased sensitivity is due to reduced competition of the probe with antioxidants in the membrane compartments. Importantly, the C18 tail slows the diffusion of the probe and enables region-specific detection of ROS, which so far can only be achieved with targeted fluorescent proteins. Similar to CM-H2DCF and H2R123 (71), H2RB-C18 does not react directly with superoxide radical and H2O2 but is effectively oxidized by products of the Fenton reaction (i.e., probably to hydroxyl radical). It is likely that, similar to other dihydrorhodamines, H2RB-C18 cannot specifically distinguish different types of ROS (22), as some proteins are able to do (4). However, the dynamic range of H2RB-C18 is much greater than that seen in redox-sensitive proteins (54). Thus, we considered that H2RB-C18 could be a useful tool in ROS research and proceeded to test this notion using primary isolated cells.
We applied H2RB-C18 to detect ROS production in pancreatic acinar cells challenged by high doses of secretagogues and by other molecules implicated in pancreatic pathology (all these substances have been shown to trigger cytosolic Ca2+ responses). We resolved ROS responses to all tested substances. Most of the stimuli generated region-specific patterns of ROS changes. The highest signals were seen in the areas that had already shown the brightest fluorescence at the time the cells were first imaged (i.e., before stimulation). These areas were usually located at the cell periphery in both apical and basal regions. However, some fluorescence changes were also recorded in the internal regions. The distribution of the fluorescence changes probably reflects both accumulation of the probe in the different cellular membranes (e.g., plasma membrane and membranes of endoplasmic reticulum (ER) and the distribution of ROS sources. Among these putative ROS sources are subplasmalemmal mitochondria (49). This is a prominent group of mitochondria in pancreatic acinar cells, which are located in the immediate proximity of the plasma membrane (25, 49, 70), are juxtaposed in the junctions between the ER and the plasma membrane (43), and preferentially respond to the Ca2+ influx (49). We cannot exclude a possible contribution of NADPH oxidases, reported to be expressed in a pancreatic cell line (74) to the observed ROS responses; however, this enzyme could not be detected by immunohistochemistry (28) and did not participate in bile acid-induced ROS generation (15) in primary pancreatic acinar cells.
Large and fast fluorescence changes of our new probe were observed following the application of the P. aeruginosa toxin pyocyanin. This was the first study testing the effect of pyocyanin on ROS production, cell signaling, and mitochondria status of pancreatic acinar cells. Pancreatic infection with P. aeruginosa is a problem for AP, cystic fibrosis, and pancreatic transplantation or endoscopic retrograde cholangiopancreatography receiving patients (9, 13, 26, 42). The ability of pyocyanin to trigger substantial ROS changes, induce [Ca2+]i and NAD(P)H responses, as well as its ability to reduce ΔΨ (i.e., to depolarize mitochondria and reduce the proton motive force vital for ATP production) suggests that this molecule can play an important role in the damage that P. aeruginosa causes in the pancreas and potentially in other organs. It is important to note that the decrease of tetramethylrhodamine, methyl ester (TMRM) fluorescence (i.e., dissipation of ΔΨ) in experiments with pyocyanin was slow and NAD(P)H concentration during the initial fast phase of ROS production actually increased. These changes are clearly permissive for accelerated ROS production (Fig. 4) (61). The later decrease in the rate of ROS production could be partially attributed to further loss of ΔΨ and a delayed reduction in NAD(P)H. The effect of pyocyanin was observed in Ppif−/− mice, was not inhibited by buffering of cytosolic Ca2+ with BAPTA, but was strongly suppressed by MitoQ. These results suggest a possible direct (i.e., not Ca2+- or MPTP-mediated) interaction of pyocyanin with the mitochondria of the acinar cells and consequent ROS production and detection by H2RB-C18. The observed distribution of the H2RB-C18 fluorescence during pyocyanin application (increase in the plasma membrane region) was clearly different from the distribution of mitochondria in pancreatic acinar cells, reported by a number of laboratories (25, 36, 49, 60, 64, 70). However, one group of mitochondria–subplasmalemmal mitochondria seems to be strategically positioned to mediate the observed response to pyocyanin. The role of these mitochondria will be investigated in a separate study.
We next applied H2RB-C18 to investigate the responses induced by basic amino acids. L-ornithine generated clearly resolvable ROS responses (in addition to triggering [Ca2+]i, NADH, and ΔΨ changes). L-arginine produced similar ROS responses, whereas L-citrulline was less effective. Interestingly, this correlates well with the reported potencies of AP induction by these amino acids in rats (53). The ROS response to L-ornithine appears to be dependent on [Ca2+]i changes (or at least require some “permissive” [Ca2+]i), as the pretreatment of cells with the Ca2+chelator BAPTA abolished the increase in H2RB-C18 fluorescence. Strong inhibition of ROS response to L-ornithine by the removal of the extracellular Ca2+ is consistent with this conclusion. The importance of mitochondria for the L-ornithine-induced ROS response was highlighted by the inhibitory effects of MitoQ and by the significant decrease of L-ornithine response in Ppif−/− mice. A possible mechanism of the L-ornithine response could involve Ca2+ release from the internal stores, activation of the Ca2+ influx through the store-operated Ca2+ influx channels and Ca2+-dependent (and possibly MPTP-dependent) acceleration of the mitochondrial ROS production. The importance of mitochondria for the ROS response observed in our study is consistent with a direct action of basic amino acids on the mitochondria of pancreatic acinar cells described recently (8).
Materials and Methods
Cell preparation, solutions, and chemicals
Pancreata were obtained from adult male mice (CD1), humanely killed in accordance with the Animals (Scientific Procedures) Act, 1986. The pancreatic acinar cells were isolated by digestion with purified collagenase (200 U/ml) as described previously (19) and used within 4 h.
Cyclophilin D-deficient mice generated by targeted disruption of the Ppif gene (3) were kindly provided by Dr. Derek Yellon (University College, London, United Kingdom).
The standard (for our laboratory) extracellular solution contained (in mM): NaCl, 140; KCl, 4.7; MgCl2, 1.13; CaCl2, 1.2; glucose, 10; HEPES-NaOH, 10 (pH 7.3). All the experiments were performed under continuous superfusion by the standard solution (controls) or by solutions based on this standard solution with the specified changes/additions. In amino acid-containing solutions, the NaCl concentration was decreased appropriately to maintain the osmolarity.
RB-C18 and all other chemicals, unless stated otherwise, were from Sigma-Aldrich. Fluo-4 acetoxymethyl ester (AM) and MitoTrackerRed were from Invitrogen, and collagenase was from Worthington Biomedical Corporation.
H2RB-C18 preparation and dye loading
To produce the fluorogenic ROS probe, we added 10 μl of NaBH4-containing solution (which was prepared as 1 mg/ml stock solution in DMSO) to 10 μl of DMSO-based solution containing 10 mM of RB-C18. Within seconds, the mixture lost its red color, indicating the conversion of RB-C18 into H2RB-C18. Four microliters of glycerol and 16 μl of ethanol were then added to the mixture for 5 min to quench NaBH4. H2RB-C18 concentration in the stock solution (produced by the above-described procedure) was 2.5 mM. To load the pancreatic acinar cells, 40 μl of this H2RB-C18-containing stock solution was added immediately to a 10-ml suspension of freshly isolated acinar cells (the final concentration of H2RB-C18 in the cell-containing solution was 10 μM) and incubated for 30 min at room temperature.
To study the responses of ROS sensors in cell-free system, 10 μM of the dyes dissolved in the extracellular buffer were tested by the addition of H2O2 (10 mM) followed by the addition of CoCl2 (10 μM). The fluorescence and fluorescence spectra were recorded using the LS 50B spectrofluorometer (Perkin Elmer).
Cells were loaded with Fluo-4 by incubation in a solution containing 5 μM Fluo-4-AM for 30 min at room temperature. Cells were loaded with CM-H2DCF by incubation in a solution containing 5 μM CM-H2DCF(DA) for 30 min at 37°C. Cells were loaded with H2R123 by incubation in a solution containing 5 μM of the probe for 30 min at 37°C. Cells were loaded with TMRM by incubation in a solution containing 50 nM TMRM for 30 min at 37°C.
Before the experiments, cells loaded with the fluorescent or fluorogenic probes were washed by centrifugation in a standard extracellular solution at room temperature.
Measurements of cell respiration
OCR of nonpermeabilized pancreatic acinar cells were assayed under basal and stress-test conditions using a Seahorse Bioscience XF24 Extracellular Flux Analyzer according to the manufacturer's specification (
Imaging experiments and data analysis
Imaging experiments were performed at 28°C–30°C. Fluorescence images were obtained using Leica SP2 inverted confocal microscopes (Leica Microsystems). Cells loaded with H2RB-C18 and TMRM were illuminated by 543 nm light using a×63 1.2 NA objective lens. Low illumination laser power of 2%–3% and a fixed zoom factor (3.3) were used in all experiments with H2RB-C18. Fluorescence emission was recorded at 560–620 nm. The recordings started after a 5-min baseline settling period. Recordings of H2RB-C18 fluorescence in unstimulated cells (control experiments) obtained throughout the duration of the project were pooled together, processed, and shown for comparison on appropriate figures. Cells loaded with Fluo-4, CM-H2DCF, and H2R123 were illuminated by 496 nm light, and fluorescence of these probes was recorded at 520–560 nm. The fluorescence of NAD(P)H was excited using 365 nm laser line and recorded at 450–490 nm. The results of fluorescence measurements were presented as normalized data by dividing the raw data by the average value of the fluorescence recorded for the first five time points. Results are reported as mean±SEM. Statistical comparisons were performed using the Student's t-test, and statistical significance was assumed at p<0.05.
Footnotes
Acknowledgments
The work was supported by the Medical Research Council (UK) grants G0700167 and MR/K012967/1 and by the National Institute for Health Research (UK) grant to the NIHR Liverpool Pancreas Biomedical Research Unit. The assistance of Hayley Dingsdale, David Collier, and Svetlana Voronina is gratefully acknowledged.
Author Disclosure Statement
The authors declare that no competing financial interests exist.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
