Abstract
Introduction
N
The nitrate-nitrite-nitric oxide (NO) pathway represents an alternative NO synthase (NOS)-independent pathway for NO formation. This is the first study demonstrating evidence for a cross-talk between the nitrate-nitrite-NO pathway and the classical L-Arginine-NOS-dependent system in control of vascular NO homeostasis and cardiovascular function. Our findings indicate that individual responses to dietary nitrate or nitrite will be dependent on the basal endothelial NOS (eNOS) activity. Thus, individuals with already compromised eNOS activity, for instance elderly and patients with cardiovascular disease, should have an augmented response to these inorganic anions. This finding could have important nutritional as well as therapeutic implications in patients with endothelial dysfunction and vascular NO deficiency.
NO is a powerful biological messenger whose production, metabolism, and overall bioactivity need to be tightly controlled (38). Negative feedback loops where the bioactive messenger controls its own formation are ubiquitous in biological signaling, and the NO system is not an exception. A number of feedback mechanisms exist for the control of NO formation, including direct inhibition of NOS by NO (22). In addition, post-translational modifications, including phosphorylation, are essential for the regulation of endothelial NOS (eNOS or NOS3) activity (13, 33). In light of this, if the nitrate-nitrite-NO pathway significantly contributes to NO homeostasis, one would expect a cross-talk between this and the classic NOS pathway.
In this study, we aimed at exploring a potential cross-talk between the systems, by investigating the hypothesis that boosting the nitrate-nitrite-NO pathway via dietary intervention with nitrate, or by acute nitrite treatment, modulates the activity of eNOS in the cardiovascular system.
Results
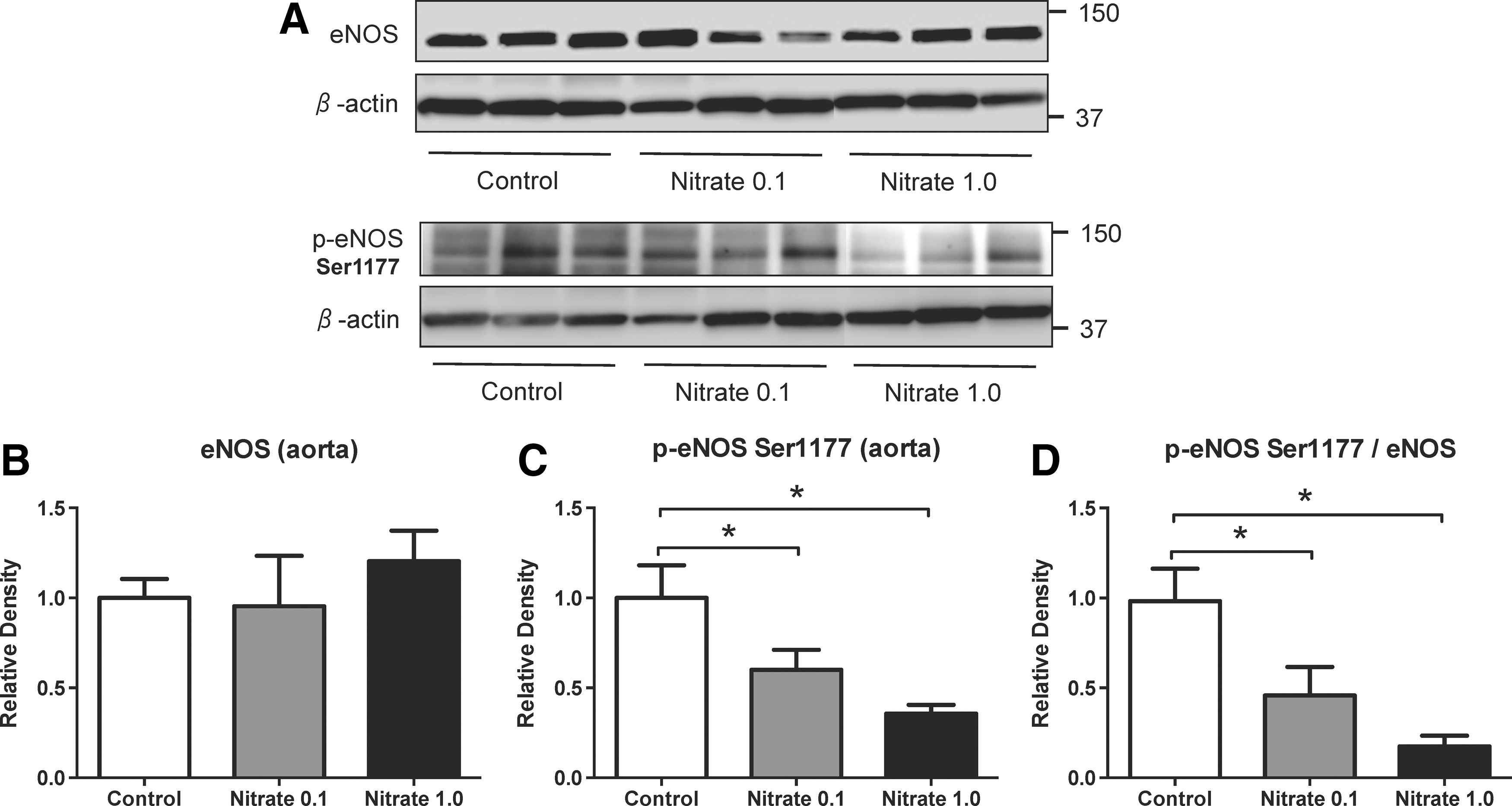
Dietary nitrate modulates phosphorylation of aortic eNOS
Phosphorylation of eNOS (p-eNOS) occurs at different sites and results in either activation (Ser1177) or inhibition (Thr495) of the enzyme (14). In rats, dietary supplementation with sodium nitrate for 8–10 weeks had no significant effect on total eNOS expression in the aorta (Figs. 1B and 2B). A dose-dependent reduction in p-eNOS (Ser1177) and p-eNOS-to-total eNOS ratio was found in nitrate-treated rats in comparison to the placebo group (Fig. 1C, D). Moreover, a dose-dependent increase in p-eNOS (Thr495 site) and p-eNOS-to-total eNOS ratio was found (Fig. 2C, D). We also measured eNOS phosphorylation 2 days after dietary nitrate supplementation had been terminated. Both p-eNOS (Ser1177) and p-eNOS (Thr495) had returned to control levels, demonstrating the reversibility of this effect (Supplementary Fig. S1; Supplementary Data are available online at
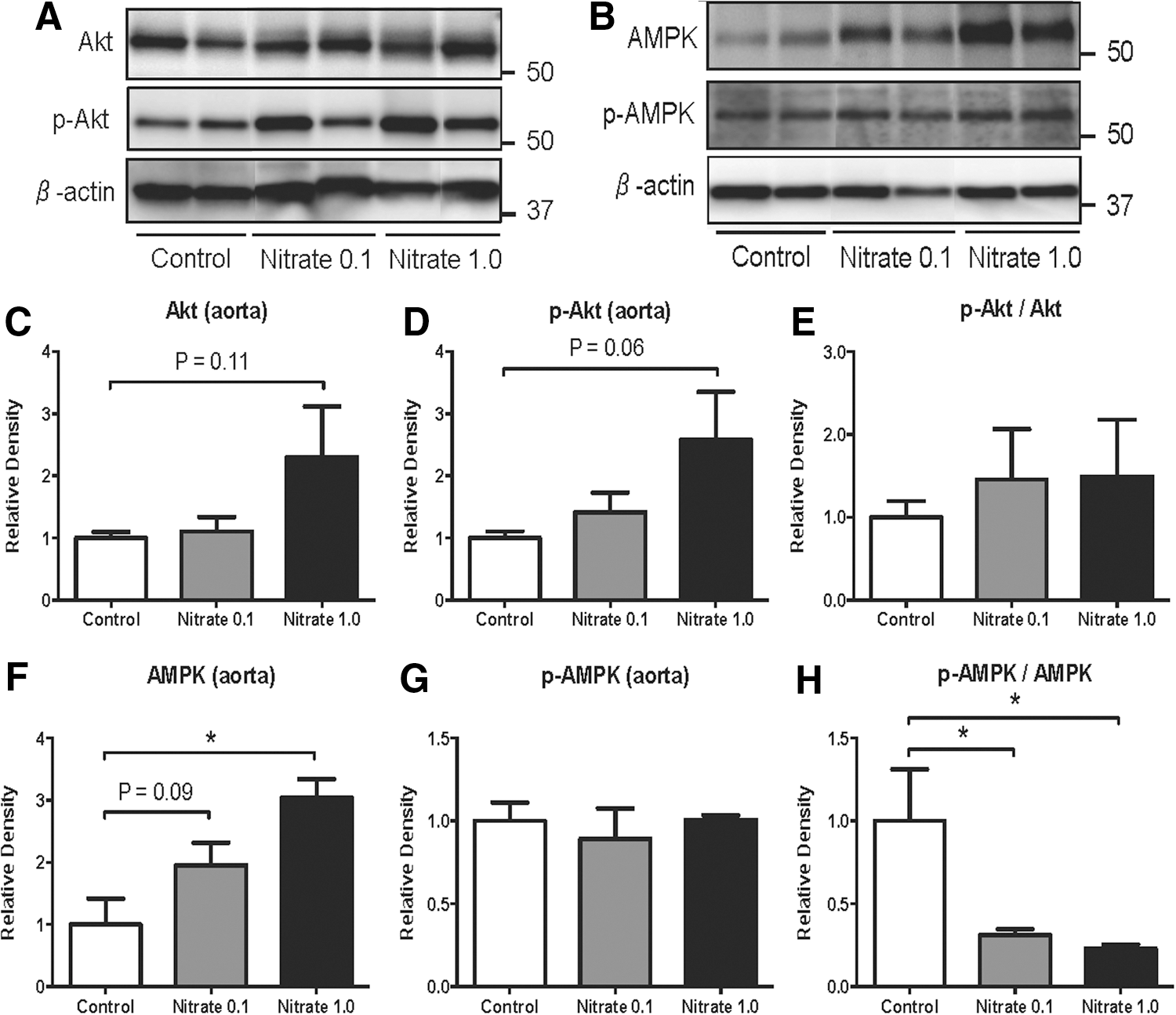
Dietary nitrate modulates phosphorylation of protein kinases in aorta
Both protein kinase B (Akt or PKB) and AMP-activated protein kinase (AMPK) are known to phosphorylate eNOS at Ser1177. Chronic supplementation with nitrate for 8–10 weeks did not significantly change the p-Akt-to-total Akt ratio in rat aorta (Fig. 3E). However, a significant reduction in p-AMPK-to-total AMPK ratio was observed in nitrate-treated rats (Fig. 3H). This modulatory effect on AMPK regulation was absent 2 days after dietary nitrate supplementation had been terminated, as evident by normal p-AMPK-to-total AMPK ratio (Supplementary Fig. S2).
Tissue NOS activity is decreased by nitrate and nitrite
A citrulline assay was used to study the effects of nitrate and nitrite on tissue NOS activity. In rats chronically treated with a high dose of nitrate, gastric ventricle Ca2+-dependent NOS activity had decreased to 10% of basal levels (Fig. 4A). When nitrite was acutely added to gastric tissue homogenates, we did not observe a statistically significant reduction in NOS activity.
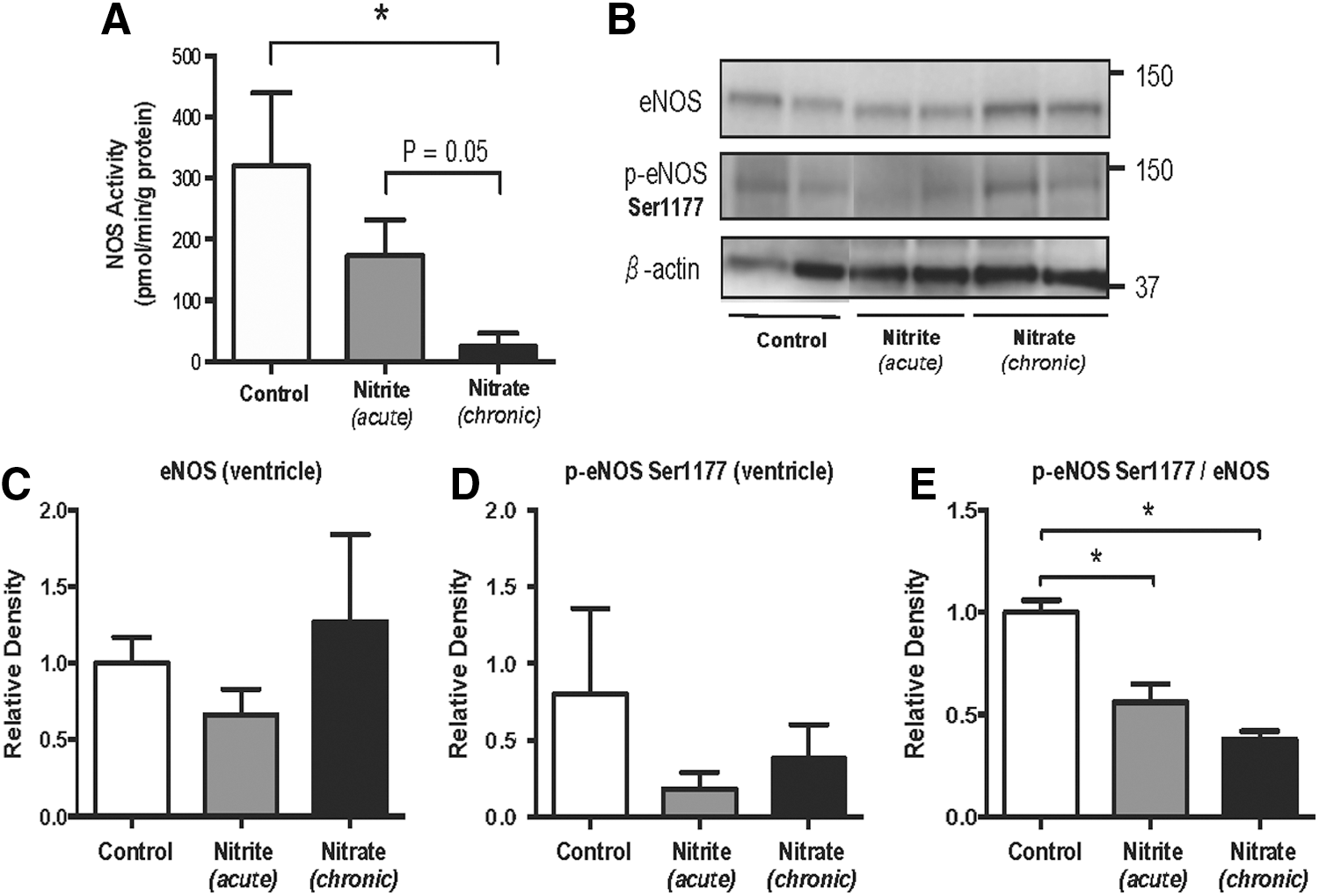
To confirm the involvement of eNOS, we performed a new series of western blots (Fig. 4). Again, a reduction in p-eNOS-to-total eNOS ratio was found in nitrate-treated rats in comparison to the placebo group (Fig. 4E). A similar reduction was also seen in gastric tissue homogenates that were acutely treated with nitrite.
Dietary nitrate decreases the citrulline–arginine ratio in plasma
Next, we wanted to explore whether the reduced eNOS activity found in the studied tissues was also reflected at a global level. The citrulline–arginine ratio in the blood may be used as a quantitative indicator of NOS activity reflecting the consumption of arginine and the concomitant production of citrulline by NOS. We measured citrulline–arginine ratios in rat plasma after nitrate supplementation (Table 1). This ratio was decreased in the plasma of rats treated with nitrate, indicating depression of endogenous NOS activity. The ornithine–citrulline ratio, however, was unaffected by nitrate supplementation, suggesting that the decreased citrulline–arginine ratio was not due to enhanced arginase activity.
n=8 in each experimental group, values are means±SEM.
p<0.05 versus controls.
Nitrate and nitrite levels
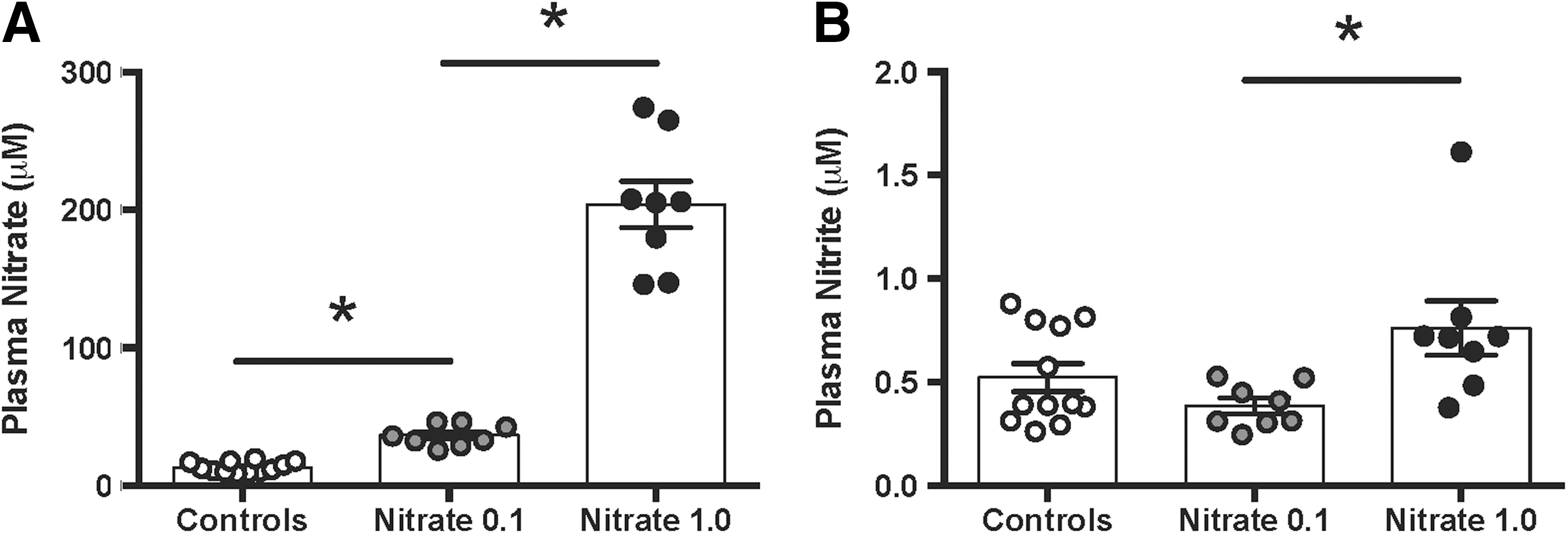
Plasma nitrate levels were dose dependently increased in animals receiving dietary nitrate. In accordance with our previous studies with chronic nitrate supplementation (3, 4), plasma nitrite levels were not significantly increased with either of the two nitrate doses compared with control rats. However, when comparing the low and high nitrate groups, the levels were slightly higher in animals receiving the high dose (Fig. 5A, B).
Effects on blood pressure
An effect of nitrate on vascular eNOS activity and NO homeostasis would likely be also reflected at a functional level in vivo. To study the effects of dietary nitrate supplementation on blood pressure, we used telemetric measurements (Figs. 6 and 7). The experiment started after 8–10 weeks of nitrate supplementation. Blood pressure was first monitored for 3 days in rats with chronic nitrate supplementation, and then continuously for two additional days after abrupt termination of nitrate supplementation. The mean arterial pressure was 91 mmHg in rats receiving no nitrate supplementation and 5 mmHg lower in animals treated with the low dose of nitrate (p<0.05). In the high-dose nitrate group, blood pressure was 15 mmHg higher compared with control animals (Fig. 6A) (p<0.05). Heart rate was not influenced by chronic nitrate supplementation (Fig. 6B).
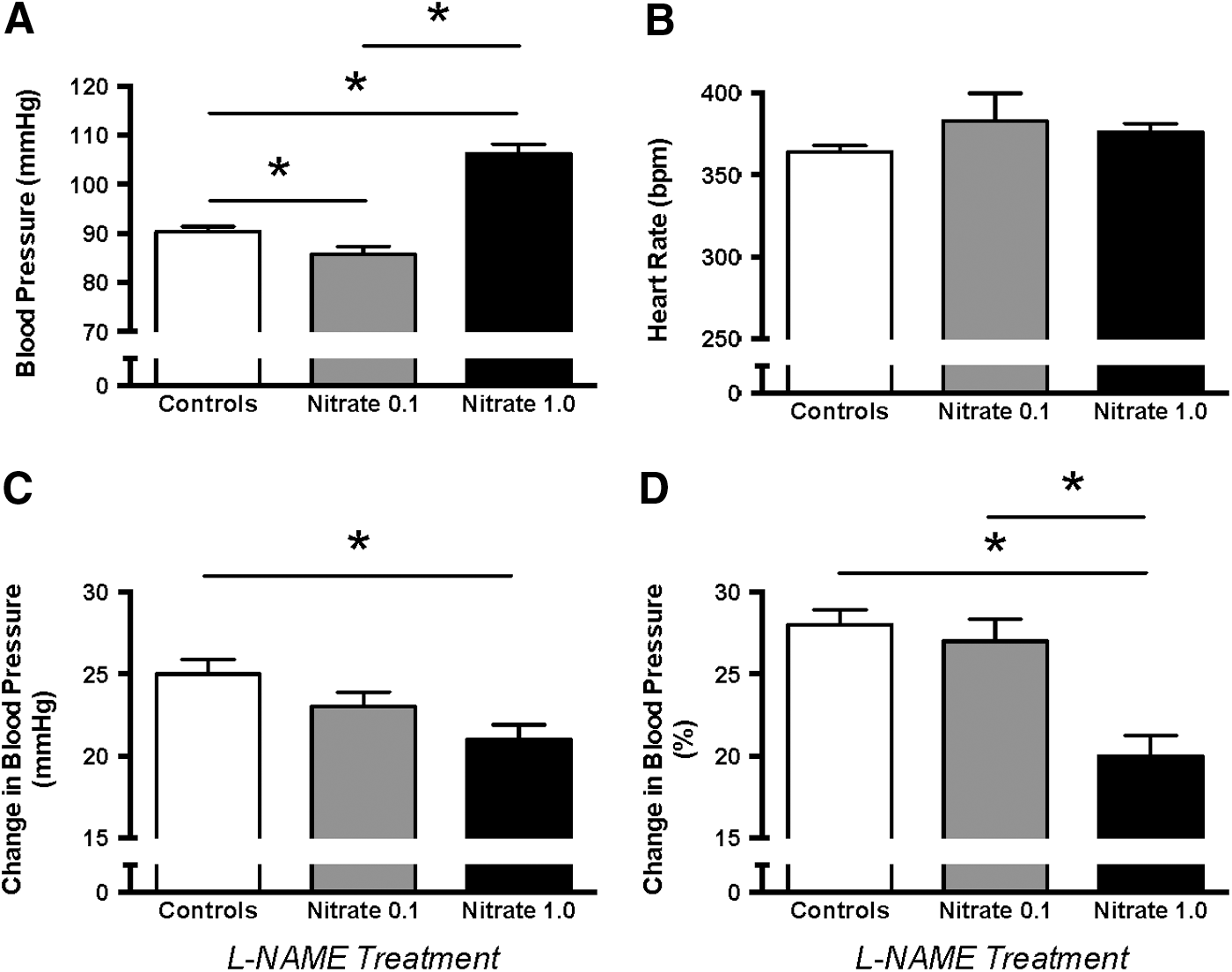
Next, we determined the effect of dietary nitrate on blood pressure responses to an NOS inhibitior. The NG-nitro-L-arginine methyl ester (L-NAME)-induced blood pressure elevation was attenuated in rats with high nitrate compared with those receiving placebo (Fig. 6C, D).
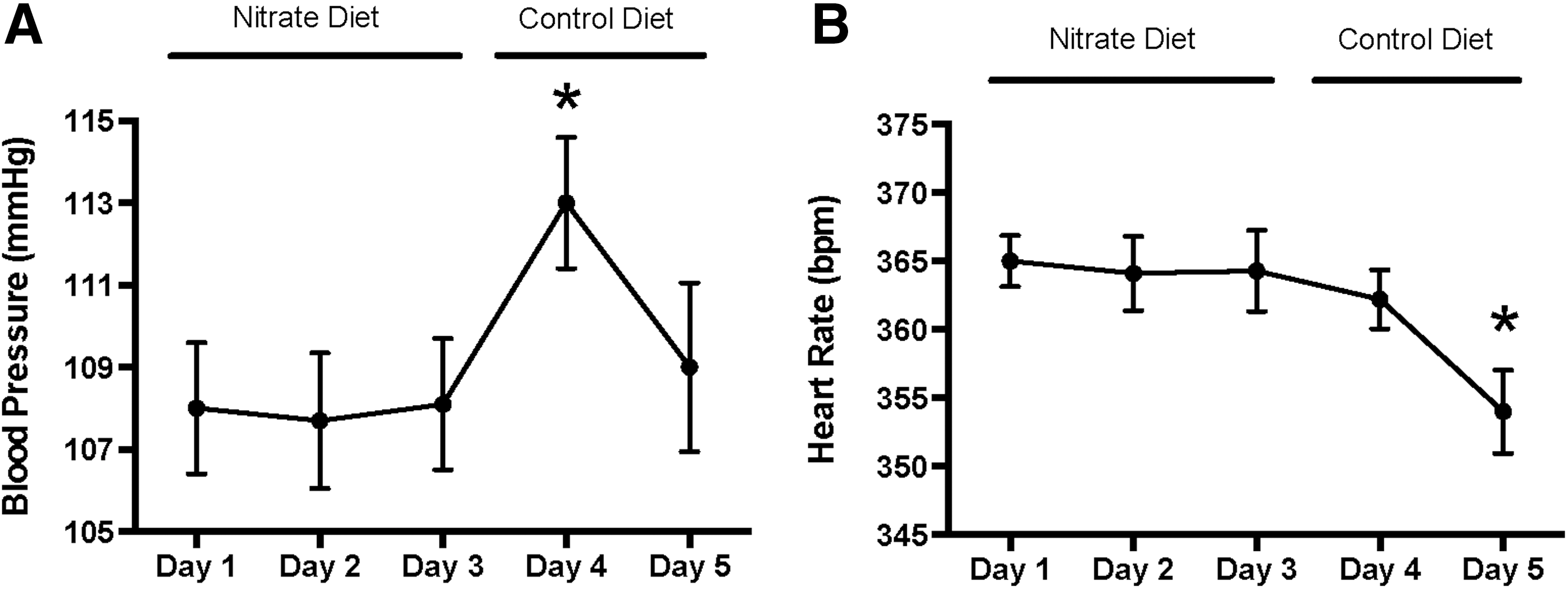
In rats with high nitrate supplementation, blood pressure increased further by 5 mmHg at day 1 after removing nitrate from the diet (p<0.05). At the 2nd day after nitrate removal, blood pressure returned to a similar level as it was with the nitrate diet (Fig. 7A), and heart rate was slightly lower (Fig. 7B). Collectively, the blood pressure data support the notion that long-term dietary nitrate supplementation down-regulates vascular eNOS activity.
Effects on cyclic guanosine monophosphate
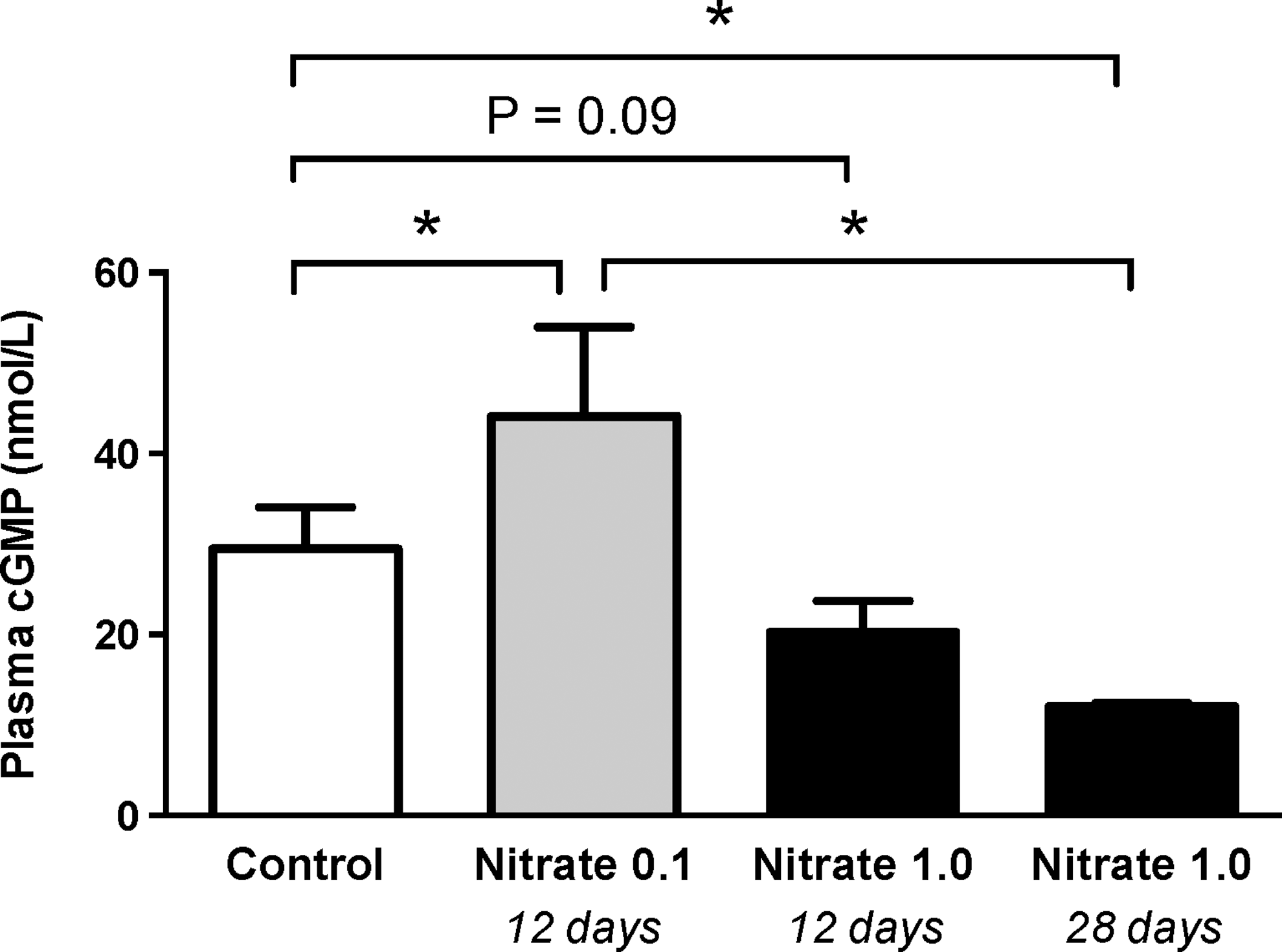
The vasoactivity of eNOS-derived NO is mainly mediated through the activation of soluble guanylyl cyclase with formation of the second messenger cyclic guanosine monophosphate (cGMP). We measured the levels of cGMP in mice given either a low or a high dose of nitrate for 2–4 weeks. In mice supplemented with the low dose of nitrate, cGMP levels were higher than in control animals. However, cGMP levels were significantly reduced in mice treated with the high dose of nitrate (Fig. 8).
Effects of nitrate and nitrite on vascular reactivity
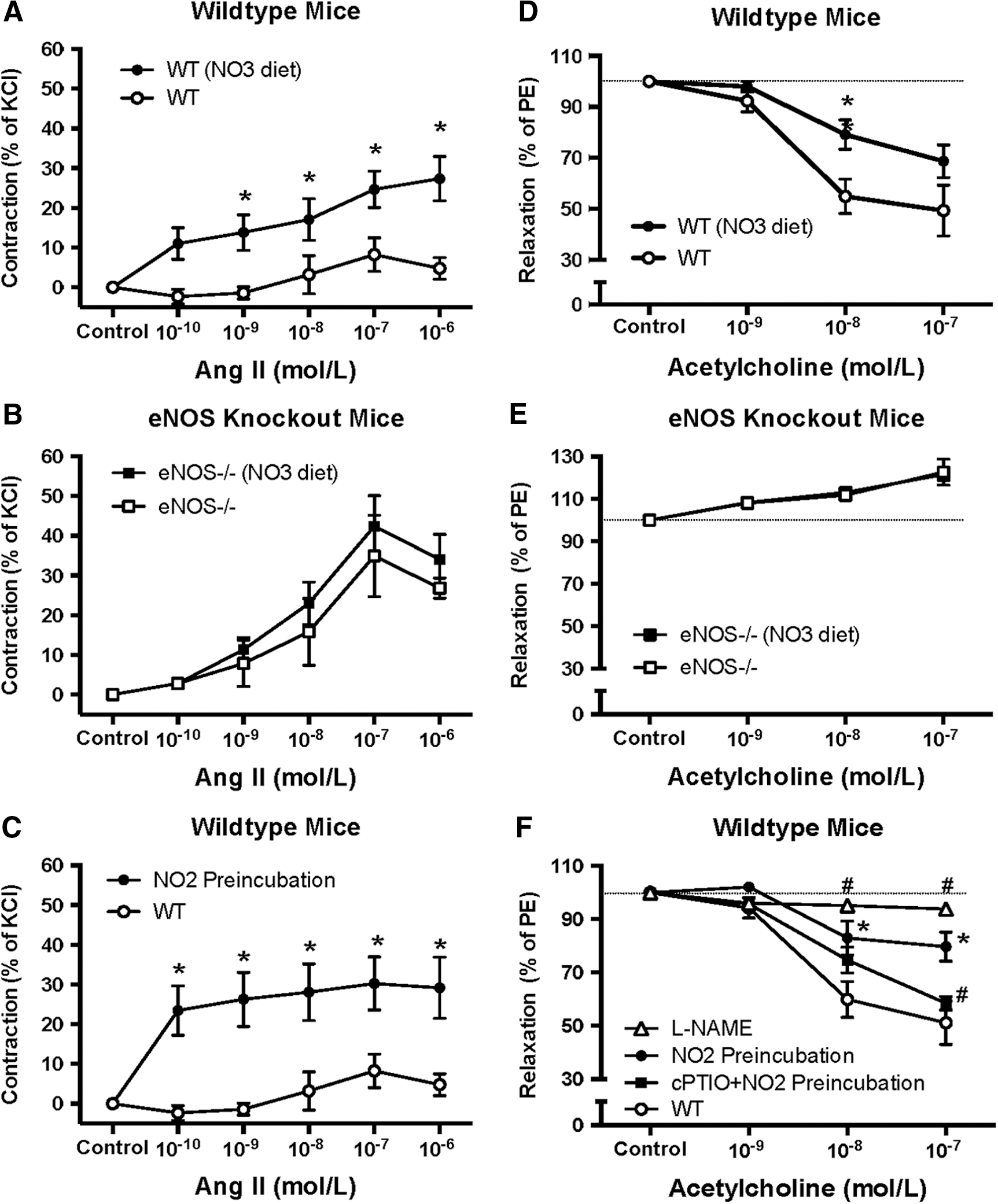
To further examine the effects of nitrate and nitrite on vascular eNOS function, we studied the reactivity of carotid arteries from mice (Fig. 9). Dietary supplementation with a high dose of nitrate (4 weeks) was associated with stronger contractile responses to angiotensin II (Fig. 9A), and attenuated acetylcholine-mediated vasorelaxation (Fig. 9D). These effects of nitrate were completely absent in eNOS null mice (Fig. 9B, E). Similar results, as observed with chronic dietary nitrate, were found in control mice if the vessels had been incubated with nitrite (2 h) before the assessment of vascular responses (Fig. 9C, F). Finally, the attenuated vasorelaxation to acetylcholine with acute nitrite was normalized after simultaneous incubation with the NO scavenger cPTIO (Fig. 9F).
Discussion
Under physiological conditions, NO generation from eNOS is tightly regulated to maintain balance in the cardiovascular system (14, 32, 33). In addition to the classical L-arginine-NOS-pathway, NO can also be formed by a nitrate-nitrite-NO pathway. In this study, we demonstrate that dietary supplementation with nitrate, or acute treatment with nitrite, is associated with a reversible down-regulation of eNOS activity, suggesting a negative cross-talk between the NOS pathway and the nitrate-nitrite-NO pathway. This conclusion is supported by in vitro studies of eNOS phosphorylation and activity, ex vivo studies of vessel reactivity, and in vivo functional assessment of eNOS activity.
We demonstrate that blood pressure was decreased with a low dose of nitrate, whereas chronic supplementation with a higher pharmacological dose was associated with an increased blood pressure. This might be a reflection of the overall NO production from the nitrate-nitrite pathway and eNOS. Thus, with a limited eNOS inhibition by the low nitrate dose, net formation of NO is still increased. Along the same lines of reasoning, as eNOS inhibition becomes more pronounced with the high dose of nitrate, a net decrease in the amounts of NO reaching guanylyl cyclase in the vascular smooth muscle cells is seen. The effects of dietary nitrate supplementation on plasma cGMP, which entirely mirrored the effects on blood pressure, support this idea. In addition to this effect, high pharmacological doses of nitrate might promote the formation of other reactive nitrogen oxides, including peroxynitrite, that could negatively influence NO signaling pathways; for example, via an interaction with the catalytic activity of guanylyl cyclase (15, 42, 44).
Studies have demonstrated that plasma nitrite levels reflect overall NO production. It is obvious from the current data that despite a robust dose-dependent increase in plasma nitrate after dietary nitrate supplementation, the levels of nitrite were not higher compared with animals without nitrate supplementation. This again might reflect the existence of a cross-talk. Nitrite derived from the dietary nitrate load is increased, whereas eNOS-derived nitrite may be decreased, leaving overall plasma nitrite levels unchanged. One should note, however, that nitrite by no means can replace eNOS as an endothelial NO source in the regulation of blood pressure. This is very clear from experiments in rats treated with a pharmacological NOS inhibitor. In these animals, blood pressure increased markedly and supplementation with dietary nitrate did not fully reverse this effect (3).
We demonstrate that dietary nitrate, in a dose-dependent manner, reduced eNOS phosphorylation at Ser1177 and increased phosphorylation at Thr495 in the aorta, both of which would contribute to a reduced eNOS activity. It is suggested that NO or related reactive nitrogen species, generated from dietary nitrate or nitrite, modulates the activity of kinases and phosphatases that regulate eNOS phosphorylation; for example, via nitrosation of critical thiols or via other redox-dependent post-translational modifications (30, 40). In support of this, it was recently demonstrated that nitrite formed in vivo from dietary nitrate facilitated nitrosation reactions (3, 4). It is also known that S-nitrosation of eNOS results in a decrease in catalytic activity which is inversely related to enzyme phosphorylation at Ser1179, that is, a similar site was found to be down-regulated by dietary nitrate (12). The underlying mechanism(s) for the observed alterations in eNOS phosphorylation by nitrate awaits further investigations. Nevertheless, the higher dose of nitrate was associated with reduced phosphorylated AMPK-to-total AMPK ratio, whereas no significant change was observed with the lower dose. This is interesting, as AMPK is thought to activate eNOS by phosphorylation at Ser1177 (8), which we found to be inversely related to nitrate intake.
Lang et al. previously reported down-regulation of eNOS in humans treated with inhaled NO. In these subjects, the expression of eNOS in the liver was profoundly decreased after a short period of NO inhalation (22). This effect was associated with increases in plasma nitrite, and the authors speculated that nitrite was the effector, as NO itself unlikely survives transport from the lung to the liver. Moreover, in a model of chronic hind-limb ischemia, Kumar and colleagues showed that long-term nitrite therapy significantly decreased eNOS expression in the ischemic limb (21). In agreement with our findings, signs of a negative feedback with nitrate and nitrite on eNOS function have been suggested in studies by Bryan et al. (2) and Zeballos et al. (45), who reported acute increases in blood pressure after infusion with a low dose of nitrite and a very high dose of nitrate, respectively.
In this study, decreased citrulline-to-arginine ratio was observed in the plasma after long-term dietary nitrate supplementation. This would also support a cross-talk between the two pathways, with less L-arginine being utilized by the NOS to generate NO and citrulline. Other mechanisms such as increased arginase activity may have contributed to the observed changes in citrulline-to-arginine ratio. (19). However, the unaffected ornithine-to-citrulline ratio suggests that nitrate supplementation did not influence arginase activity in our study.
Ca2+-dependent NOS activity in gastric tissues was profoundly inhibited in rats fed a high nitrate diet along with a reduced phosphorylation at Ser1177. This is interesting, considering that the gastric compartment is the dominant site for nitrite reduction to NO in mammals because of the very high nitrite levels in swallowed saliva and the low pH which greatly enhances nonenzymatic NO formation (29). Similar results were obtained when nitrite was acutely administered ex vivo to gastric tissue homogenates, indicating a rapid-onset effect.
We also studied the functional responses in blood pressure observed after acute cessation of high dietary nitrate. If nitrate would down-regulate eNOS activity, one would expect a rebound in blood pressure, that is, a transient increase above basal values after cessation. Indeed, blood pressure increased significantly after abrupt termination of nitrate supplementation and returned to normal levels 2 days later, indicating a reversible and dynamic cross-talk between the systems. Moreover, if nitrate supplementation were associated with reduced eNOS activity, the acute blood pressure elevation in response to an NOS inhibitor would be attenuated. In our study, stimulation of the nitrate-nitrite-NO pathway was actually associated with reduced L-NAME responses.
To further investigate nitrate- and nitrite-dependent effects on the NOS pathway, ex vivo vascular experiments were conducted. Both chronic dietary supplementation with a high dose of nitrate and acute incubation with nitrite ex vivo attenuated acetylcholine-mediated vasorelaxation, which was indicative of reduced eNOS function. The effect of acute nitrite could be abolished by simultaneous preincubation with an NO scavenger, suggesting that nitrite-derived NO was inhibiting eNOS. Moreover, Angiotensin II-mediated contraction, which is usually moderated by eNOS-derived NO production (24, 35, 41), was enhanced after chronic supplementation with a high dose of nitrate. These effects were not observed in eNOS knockout mice, supporting the notion that stimulation of the nitrate-nitrite pathway may down-regulate vascular eNOS activity.
The physiological significance of nitrate-induced down-regulation of eNOS activity requires further investigations. The findings should be verified in humans to determine whether amounts of nitrate achieved via a normal diet are sufficient to significantly influence the eNOS system. For comparison, the low dose of nitrate in the present study is similar to what has been used in previous human studies, and it can be easily achieved via our everyday diet. One could speculate that the recently described reduction in blood pressure in humans after short-term dietary nitrate supplementation (23) may be attenuated after prolonged intake. Such effect would likely be most pronounced in young and healthy subjects in whom eNOS is already operating at its maximal capacity. In older subjects (7, 11) and in patients with cardiovascular disease (10), however, vascular eNOS activity has been reported to be diminished, and the net effect of chronic dietary nitrate supplementation is likely to increase NO bioavailability. Thus, one might speculate that any salutary effects of long-term dietary nitrate on the cardiovascular system would be most prominent in patients with endothelial dysfunction. In fact, this notion is supported by our recent study using a rodent model for hypertension and cardiovascular disease (4). Dietary nitrate, in an identical dose and a similar protocol as used in the current study, caused the lowering of robust blood pressure and prevented adverse cardiovascular outcomes. A recent study in humans lends further support to this idea. These authors demonstrated greater reductions in blood pressure by dietary nitrate in individuals with lower basal levels of nitrite in plasma (20).
In conclusion, this study shows that long-term dietary nitrate supplementation is associated with down-regulation of vascular eNOS activity in rodents. These results suggest the existence of a cross-talk between NOS-dependent and NOS-independent pathways in control of vascular NO homeostasis.
Materials and Methods
Animal and tissue preparation
All experiments were approved by the ethics committee in Stockholm and Uppsala (Sweden) for animal experiments, and they were conducted in accordance with the National Institutes of Health Guide for Care and Use of Laboratory Animals. Male Sprague–Dawley rats (250–350 g), or C57BL/6NCrl mice (Charles River Laboratories) and eNOS knockout mice were maintained under standard conditions of temperature (21°C–22°C) and illumination (12 h light/12 h darkness). The animals were kept in wide mesh-bottomed cages with free access to pelleted chow and tap water. The placebo animals were given a standard diet (R36; Lactamin). The treatment group was supplemented with two doses of sodium nitrate (0.14 or 1.4 g NaNO3 kg−1) added to the chow, to achieve a daily intake of 0.1 mmol (Nitrate 0.1) and 1 mmol (Nitrate 1.0) nitrate kg−1 day−1, respectively. The low dose resembles an intake of nitrate-rich vegetables in humans. The experimental protocols were carried out after 8–10 weeks (rats) or 2–4 weeks (mice) with sodium nitrate treatment. Following completed functional studies, animals were anesthetized and tissue and blood were collected for a later analysis.
Telemetric blood pressure measurements
Animals were anesthetized with inhalation of isoflurane (Forene®; Abbott Scandinavia AB) and continued throughout surgery by the inhalation of 2.2% isoflurane. The telemetric device (PA-C40) (DSI™; Transoma Medical) was implanted into the aortic lumen as previously described (6). After surgery, all animals were allowed to recover for at least 10 days before any cardiovascular measurements were conducted. Telemetric measurements of blood pressure and heart rate in all groups were performed continuously during a control period (72 h), followed by an additional 72 h period with the NOS inhibitor L-NAME, administered via the drinking water (1 g L−1). In a separate experimental series, telemetric blood pressure recordings were made during baseline and continuously for 48 h after abrupt termination of long-term dietary nitrate supplementation.
Plasma collection
At the end of the study period, rats were anesthetized by an i.v. injection of thiobutabarbital sodium (Inactin®, 120 mg/kg body weight), and a catheter was placed in the left carotid artery for blood sampling. Mice were anesthetized by spontaneous inhalation of isoflurane (2% in air), and blood was collected from vena cava inferior. Blood samples were collected into tubes containing a solution with NEM (final concentration 5 mM) and EDTA (final concentration 2 mM). The blood was centrifuged immediately at 730 g for 5 min at 4°C and stored at −80°C until analysis.
Nitrate and nitrite analysis
Plasma content of nitrate and nitrite was measured with a dedicated high-performance liquid chromatography (HPLC) system (ENO-20; Eicom) as previously described (18). The method is based on the separation of nitrate by reverse-phase/ion exchange chromatography, followed by online reduction of nitrate to nitrite with cadmium and reduced copper. Derivatization of reduced nitrite was performed with Griess reagent, and the level of diazo compounds was measured by a visible detector at 540 nm.
Amino acid assay
Plasma levels of arginine, citrulline, and ornithine were measured by an HPLC method as previously described (4).
Cyclic guanosine monophosphate
Plasma content of cGMP was analyzed with ELISA method (Biotrak EIA System). An inhibitor of cAMP/cGMP phosphodiesterases (IBMX) was added to plasma samples (final conc. 10 μM) before freezing and later processing (3).
Western blot analyses
Frozen aortic tissues were weighed and homogenized (Bullet Blender™; Next Advance, Inc.) using 0.5 mm stainless steel silicate beads (Next Advance, Inc.) in 0.5 ml lysis buffer as previously described (17). Ventricle protein extracts were obtained as described for NOS activity assay (see the following section). After centrifugation, protein concentrations in the supernatants were determined by Bradford protein assay (Bio-Rad). Equal protein amounts were separated by 4%–20% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (Bio-Rad), followed by transfer to a polyvinylidene difluoride membrane (Bio-Rad). After blocking with 5% nonfat dry milk in Tween-containing Tris-buffered saline, membranes were incubated with specific primary antibodies (p-eNOS [Ser1177] rabbit monoclonal antibody [mAb], p-eNOS [Thr495] rabbit Ab, p-Akt [Ser473] rabbit Ab, p-AMPKα [Thr172] rabbit Ab, Akt rabbit Ab, and AMPKα rabbit Ab [all Cell Signaling], eNOS mouse mAb [BD Transduction Lab.], β-actin mouse mAb [Santa Cruz Biotechnology]), and respective secondary antibodies (horseradish peroxidase-conjugated goat antibodies to rabbit or mouse IgG; DAKO). Restore™ PLUS Western Blot Stripping Buffer (Thermo Scientific) was used to remove bound antibodies from the membranes, followed by blocking and re-probing the membranes with primary and secondary Ab. Bands were detected by a SuperSignal West Pico chemiluminescence substrate (Thermo Scientific), and results were normalized with β-actin. Images were analyzed by a luminescent image analysis system LAS 1000+ (Fujifilm). The results were quantified by densitometry and reported as relative optical density of the specific proteins.
NOS activity assay
NOS activity was measured by the conversion of L-[U-14C]arginine to [U-14C]citrulline as previously described (37). In brief, the stomach was excised and the nNOS-rich pyloric region was discarded while the rest of the ventricle was homogenized and used for analysis. Frozen ventricle tissues were homogenized (Bullet Blender) using 0.5 mm stainless steel silicate beads in 3 ml lysis buffer per g tissue, and 0.1 mM NaNO2 was present for the NO2 (acute) group (total incubation with NO2 for 3 h before the activity assay was performed). Twenty microliter of tissue extract (soluble fraction, all samples run in duplicate) was added to 15 ml plastic tubes containing 100 μl of a buffer, prewarmed to 37°C, consisting of 50 mM potassium phosphate, pH7, 60 mM L-valine, 1.2 mM MgCl2, 0.25 mM CaCl2, 120 μM NADPH, 1.2 mM L-citrulline, 22.4 μM L-arginine and 320 pmol L-[U-14C]arginine (274 mCi/mmol), and with 0.1 mM NaNO2 for the NO2 (acute) group. Parallel reactions were run containing an additional 1.6 mM EDTA and 1.6 mM L-NAME to determine the background. After incubation for 10 min at 37°C, the reaction was terminated by the addition of 1.5 ml of 1:1 (v/v) H2O/Dowex-50W. One milliliter of H2O was added and the mixture was allowed to settle for 10 min before 1 ml of the supernatant was examined by liquid-scintillation counting. NOS activities were adjusted by the amount of total proteins (determined by Bradford protein assay; Bio-Rad) and were reported as the formation of pmol L-citrulline min−1 g protein−1.
Vascular reactivity studies
Carotid arteries were isolated from anesthetized mice, removed after euthanasia by cervical dislocation, and placed in ice-cold Krebs solution (composition in mM: NaCl 119; KCl 4.7; CaCl2 1.6; KH2PO4 1.2; MgSO4·7H2O 1.2; NaHCO3 25.1; glucose 5.5; EDTA 0.026). Arterial rings (2 mm) were mounted on 40 μm stainless steel wires in a small vessel myograph (Model 620M; Danish Myo Technology) for recording isometric force by transducers (PowerLab 4/30; AD Instruments). The chambers were filled with Krebs solution (37°C, pH 7.4) that was aerated with Carbogen (95% O2, 5% CO2). Resting tension of arteries was set according to Mulvany's normalization procedure (34).
Vascular protocols
First, vessel viability was assessed by replacing Krebs solution in chambers twice with 0.1 M KCl. After washout, a cumulative concentration response for angiotensin II (Ang II; 10−12–10−6 M) was obtained, with or without 2 h preincubation with nitrite (10−4 M). Contractile responses were expressed as a percentage of constriction to 0.1 M KCl (% of KCl). Endothelium-derived relaxation was assessed by cumulative applications of acetylcholine (ACh) (Ach 10−9–10−7 M) after preconstriction with phenylephrine (PE; 10−6 M). The relaxation induced by ACh was expressed as percentage change from the phenylephrine response (% of PE). The contractile responses to Ang II and the relaxation to ACh were assessed in carotids from control and eNOS knockout mice, with or without chronic treatment with nitrate. Moreover, the effects of simultaneous incubation with L-NAME or preincubation with nitrite (10−4 M) for 2 h were assessed.
Statistical analysis
Data processing was performed with GraphPad Prism software 6. All data are shown as means±SEM. For multiple comparisons, ANOVA with the Bonferroni or Newman–Keuls multiple-comparison test was used. *p<0.05 was considered significant.
Footnotes
Acknowledgments
The authors thank Carina Nihlén, Margaretha Stensdotter, and Annika Olsson for their excellent technical assistance and helpful discussions, and Professor Paul L. Huang (Massachusetts General Hospital and Harvard Medical School) for the eNOS mice. The study was supported by the Swedish Research Council (K2012-99X-21971-01-3), Vinnova (CIDaT), the Swedish Heart and Lung Foundation (20110589), Jeanssons Foundation (JS2011-0212), Torsten Söderbergs Foundation, EUs 7th Framework program (Flaviola), The Wenner–Gren Foundation, and The Swedish Society for Medical Research (SSMF).
Author Disclosure Statement
The authors declare that they have no conflict of interest.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
