Abstract
Introduction
C
Heme oxygenase-1 (HO-1) haploinsufficiency impairs angiogenic potential of bone marrow-derived cells (BMDCs), but does not affect their proliferation, migration, and survival under oxidative stress. In diabetic animals, HO-1 haploinsufficiency leads to down-regulated expression of proangiogenic genes and to impaired revascularization of ischemic tissue, despite a potent mobilization of bone marrow-derived progenitor cells into peripheral blood. This indicates that angiogenic response in situ, but not a mobilization of progenitor cells, is crucial for blood flow recovery in diabetic ischemic muscles. Application of conditioned media from BMDCs, but not the cultured cells, efficiently augments muscle reperfusion. In addition, a local injection of BMDCs does not provide an improvement in the wound-healing model, possibly due to a very fast elution of the cells.
An important proangiogenic gene is Hmox1, coding for heme oxygenase-1 (HO-1), a stress-inducible cytoprotective enzyme (17). HO-1 degrades heme to carbon monoxide (CO), ferrous ion, and biliverdin, which is subsequently reduced to bilirubin by biliverdin reductase (44). HO-1 can be induced on injury (22), augmenting the tissue regeneration (54). Proangiogenic properties of HO-1 were confirmed in various research models, both in vitro and in vivo (40). Importantly, HO-1 was shown to be an upstream and downstream mediator of vascular endothelial growth factor (VEGF) and stromal cell-derived factor 1 (SDF-1)-induced angiogenesis [reviewed in Dulak et al. (17)].
Although homozygous HO-1 deficiency is extremely rare in humans, with only two cases described so far (55, 71), there is a considerable variability in HO-1 expression in human populations, which is caused by a polymorphism of Hmox1 promoter (61). Moreover, although large-scale analysis did not confirm a meaningful effect of Hmox1 promoter polymorphism on coronary artery disease or myocardial infarction (43), there are many clinical data indicating its influence on cardiovascular complications, at least in some groups of patients (18, 20). Thus, the presence of less active alleles was associated with an elevated rate of restenosis after balloon angioplasty (23) and with a higher incidence of coronary artery disease in type 2 diabetes (11). Moreover, among patients with peripheral artery disease (PAD), those with less active Hmox1 promoter had higher rates of myocardial infarction, percutaneous coronary interventions, and coronary bypass operations (16).
Noteworthy, the expression of HO-1 is down-regulated in some pathological conditions. We and others have demonstrated the diminished level of HO-1 in diabetic mice and rats (14, 22) and in leukocytes of type 2 diabetic patients (1, 50). This could contribute to cardiovascular complications common in diabetes, as the adenoviral Hmox1 gene transfer to diabetic mice improved angiogenesis and fastened wound healing (22). It is also known that the function of proangiogenic precursor cells is impaired in patients with cardiovascular disorders (42, 66, 67). Therefore, an inquiry into the role of HO-1 in the activity of proangiogenic BMDCs may provide new strategies for progenitor cell modifications that are aimed at the improvement of their regenerative potency.
A few studies examining the significance of HO-1 in proangiogenic progenitors have been published to date, indicating that this enzyme plays an important role in progenitor cell mobilization, homing, and endothelialization of blood vessels (38, 57, 65, 70). However, the function of HO-1-deficient proangiogenic precursors has not been sufficiently investigated, and nothing is known about the significance of HO-1 down-regulation in a more clinically relevant model, namely in proangiogenic progenitors of haplodeficient HO-1+/− mice. There are also no data concerning the potential effect of diabetes on tissue revascularization in such animals. Therefore, our aim was to investigate how HO-1 influences proangiogenic activities of BMDCs in vitro and in vivo under normoglycemic and hyperglycemic conditions, and to check whether the application of BMDCs or BMDC-derived conditioned media can improve angiogenesis in diabetic HO-1+/− mice.
Results
EPCs are less frequent in bone marrow of HO-1 deficient mice
A comparison of the bone marrow harvested from HO-1−/− mice with that of HO-1+/− and HO-1+/+ littermates demonstrated the decreased frequency of cells with phenotype ascribed to subpopulation of EPCs: Hoe+Lin−CD45−cKit−Sca-1+VEGFR-2+ in mice lacking HO-1 (Supplementary Fig. S1A; Supplementary Data are available online at
Cultured BMDCs are a heterogeneous population, with 80%–90% of cells incorporating acetylated low-density lipoprotein (acLDL) and binding Bandeiraea simplicifolia isolectin (BS1) (Supplementary Fig. S1B, C), which are capable of forming capillary-like structures after seeding on Matrigel (Supplementary Fig. S1D). Flow cytometry phenotyping revealed that, regardless of HO-1 genotype, the BMDC populations contained a similar proportion of cultured cells expressing endothelial or hematopoietic markers, such as CD31, VEGFR-2 (KDR), Tie2, CD105, Sca-1, and CD45, with a slightly lower fraction of CD34+ subset in HO-1+/− and HO-1−/− cultures (Supplementary Fig. S1E). During the 9-days of culture, the population became enriched in the CD45− cells co-expressing Sca-1 and VEGFR-2 by 400–500-fold (data not shown).
HO-1 deficiency affects apoptosis, proliferation, and migration of BMDCs
According to expectations, no HO-1 protein was detected in HO-1−/− BMDCs. The concentration of HO-1 in HO-1+/− cells was 11%–15% lower than in wild-type counterparts and was not modified by hyperglycemia (Fig. 1A). To evaluate the potential role of HO-1 in the resistance of BMDCs to oxidative stress, HO-1+/+, HO-1+/−, and HO-1−/− cultures were exposed for 6 h to H2O2 (100 μM) or hemin (50 μM) or both under normoglycemic (5 mM) and hyperglycemic (33 mM) conditions. The toxic effect of H2O2 was weak (Fig. 1B), but treatment with hemin significantly increased the number of apoptotic cells in HO-1-deficient BMDCs (Fig. 1C), which indicates that HO-1 acts as a cytoprotective agent, especially against hemin-induced injury. Hyperglycemia did not affect cell viability (Fig. 1B, C).
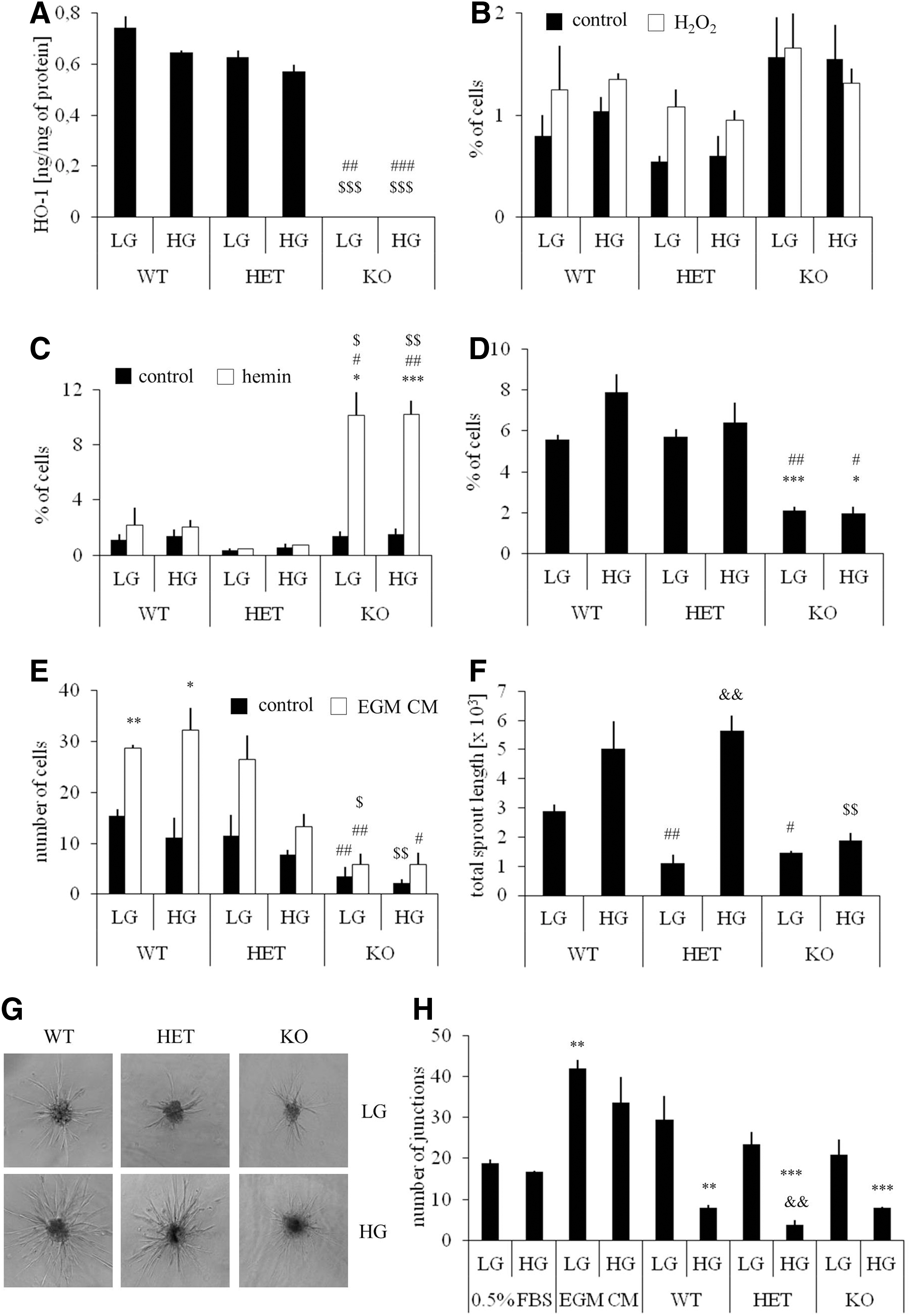
Cell cycle analysis revealed that the rate of spontaneous proliferation (percentage of cells at G2 and S phases) was lower in HO-1−/− cultures, and not modified by hyperglycemia (Fig. 1D). We did not observe differences in G0 to G1 transition. HO-1-deficient cells also exhibited significantly weaker migration capacity in control starvation medium, as well as in response to endothelial growth medium (EGM) complete medium, containing a mixture of growth factors (Fig. 1E). Thus, HO-1 deficiency, but not HO-1 haploinsufficiency, impairs viability, proliferation, and migration of BMDCs, features that are important for a proper cell function within the microenvironment of injured tissue.
HO-1 deficiency weakens angiogenic potential of BMDCs
To evaluate the angiogenic potential of HO-1+/+, HO-1+/−, and HO-1−/− BMDCs, we assessed the sprouting of capillary-like structures from the BMDC spheroids embedded in collagen gel (Fig. 1F, G). Under normoglycemic conditions, both HO-1+/− and HO-1−/− cells formed shorter capillaries than their HO-1+/+ counterparts. Surprisingly, the lengths of HO-1+/+ and HO-1+/− capillaries were increased in hyperglycemia. Such an increase was not observed in HO-1−/− BMDCs (Fig. 1F, G).
Since proangiogenic bone marrow cells were demonstrated to facilitate blood vessel formation mainly in a paracrine manner (45, 56), we plated the human aortic endothelial cells (HAECs) on Matrigel in conditioned media harvested from the BMDCs (Fig. 1H). Cells cultured in unconditioned starvation medium (0.5% fetal bovine serum [FBS]) and unconditioned EGM complete medium served as a negative and positive control, respectively. Quantification of the number of branch points showed that conditioned media (0.5% FBS) from normoglycemic HO-1+/+ BMDCs increased the formation of cord-like structures to a level which did not significantly differ from that of the positive control (p=0.15). Stimulations with media from normoglycemic HO-1+/− or HO-1−/− BMDCs were weaker (p=0.004 or p=0.015). Interestingly, media harvested from the BMDCs cultured in hyperglycemia, regardless of their HO-1 genotype, displayed strongly reduced paracrine proangiogenic potential (Fig. 1H).
HO-1 deficiency influences transcriptome of cultured BMDCs
To shed light on the possible pathways that are responsible for different angiogenic potential, we compared the transcriptome of HO-1+/+ and HO-1−/− BMDCs cultured for the last 24 h in normoxia or hypoxia (Supplementary Tables 1 and 2; all data will be provided on request). Results of Venn's analysis showed that HO-1 deficiency significantly changed the expression of 40 genes (Fig. 2A), namely 27 genes under normoxic and 18 genes under hypoxic conditions. Only five of them were regulated in the same way in both normoxia and hypoxia: kininogen-1 (Kng1) and V-set and immunoglobulin containing-4 (Vsig4) were down-regulated, while acetyltransferase like-1 (Aytl1), WNT1-inducible signaling pathway protein-1 (Wisp1), and solute carrier family-40 member-1 (Slc40a1) were up-regulated. Thus, in most cases, the effect of HO-1 deficiency was dependent on the oxygen level.
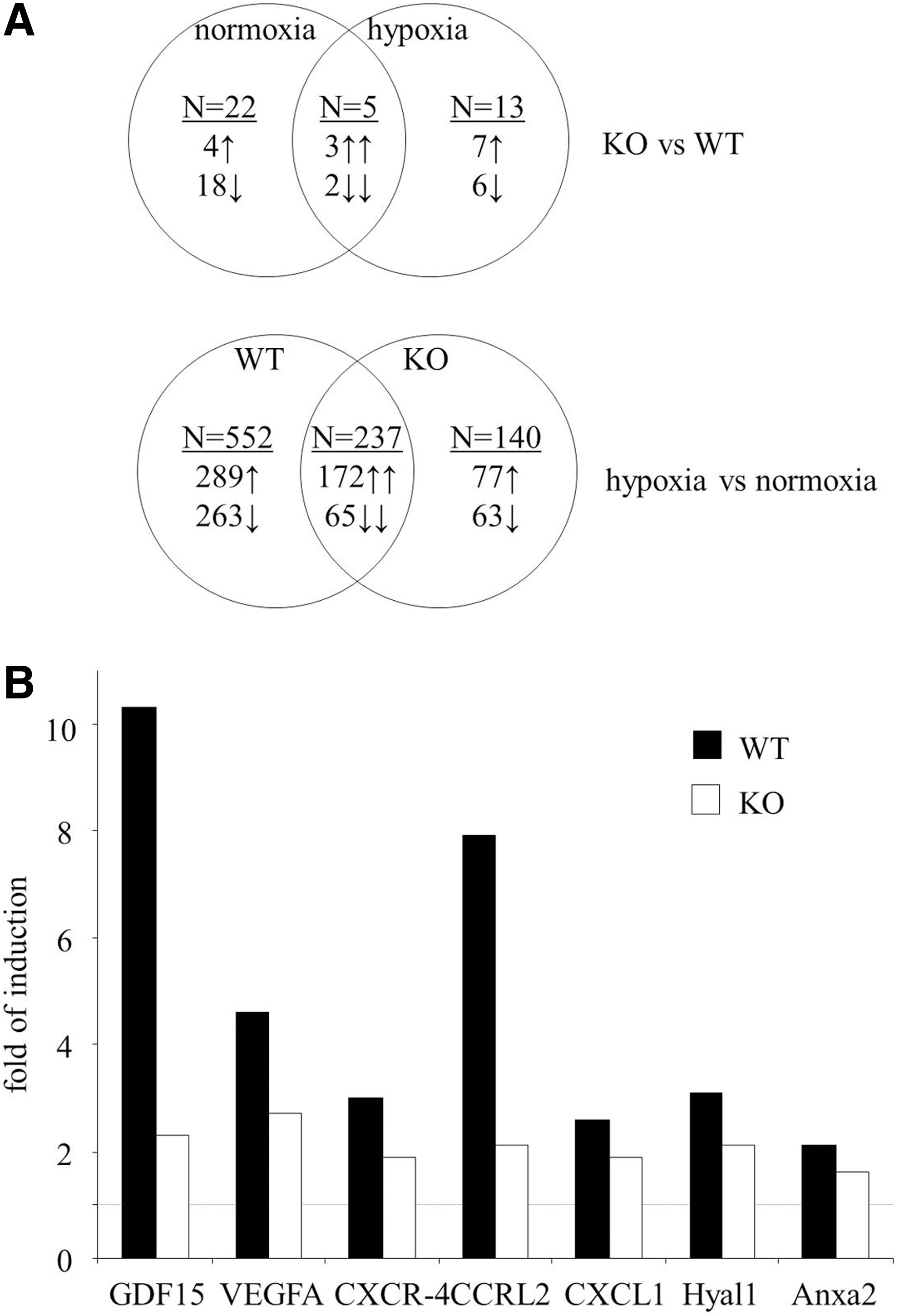
HO-1+/+ and HO-1−/− BMDCs differentially expressed some genes regulating angiogenesis, inflammatory reaction, proliferation, and apoptosis. Lack of HO-1 was associated with down-regulation of several proangiogenic genes, such as cyclin-dependent kinase-5 (Cdk5), activating protein-2 (AP-2), R-spondin-2 homolog (Rspo2), matrix metalloproteinase-11 (MMP-11), metallotionein-1, Sca-1 (lymphocyte antigen-6 complex, locus-A [Ly6a]), lymphocyte antigen-6 complex, locus-C1 (Ly6c), or ceruloplasmin, and up-regulation of Wisp1, which can exert antiangiogenic effects. In addition, among the 40 genes influenced by HO-1 deficiency, there were 9 genes of the WNT pathway, 5 genes associated with iron metabolism, and genes regulating adipogenesis (12) or osteogenesis (2) (Supplementary Table 1).
In the next step of transcriptome analysis, we checked the effect of hypoxia in HO-1+/+ and HO-1−/− BMDCs. According to Venn's diagram, 929 genes are differently expressed under normoxia and hypoxia in HO-1+/+, HO-1−/−, or in both cell types (Fig. 2A and Supplementary Table 2). The majority of them (552 genes) changed under hypoxic conditions only in HO-1+/+ BMDCs, while 140 changed only in HO-1−/− cells. Thus, HO-1 deficiency modifies the response to hypoxia, attenuating the hypoxic induction of several proangiogenic genes, including growth differentiation factor-15 (GDF-15), VEGF, chemokine receptor-like 2 (CCRL2), chemokine receptor-4 (CXCR-4), chemokine ligand-1 (CXCL-1), hialuronidase-1 (Hyal1), or annexin-A2 (Anxa2) (Fig. 2B and Supplementary Table 2).
BMDC transplantation improves tissue regeneration neither in cutaneous wound nor in hind limb ischemia models
To investigate the influence of HO-1 deficiency on the regenerative potency of BMDCs, we used two in vivo models. In the first one, either HO-1+/+ or HO-1−/− BMDCs were injected intradermally around the cutaneous wounds in HO-1−/− mice, in which the wound-healing process was previously shown to be impaired (22). We observed much more HO-1+/+ PKH67-positive BMDCs within the wounds in comparison to HO-1−/− cells at day 14 post-injury (Fig. 3A, B). However, there was no statistical difference in the time course of wound closure between all investigated groups, including the PBS-treated animals (Fig. 3C).
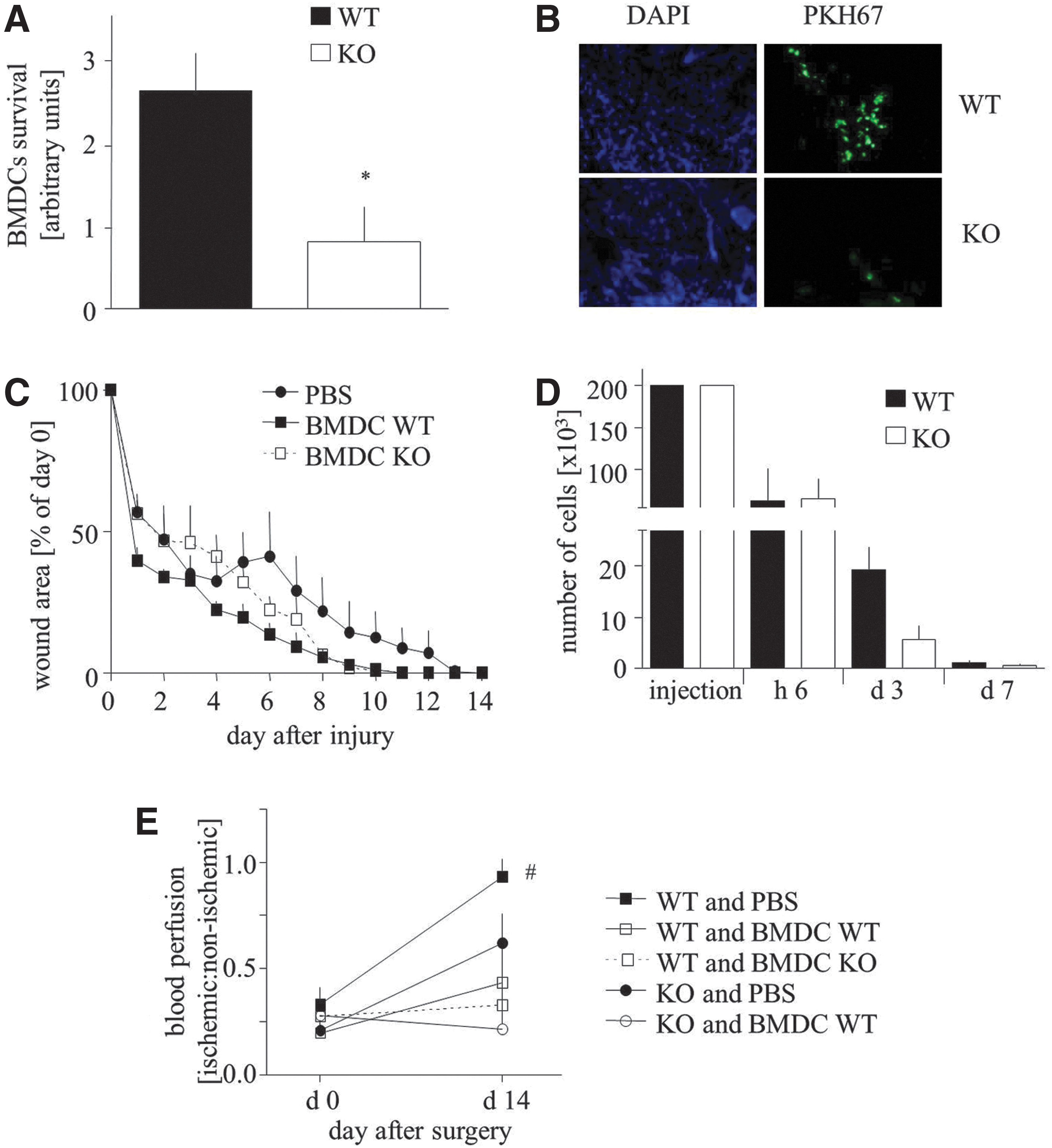
To determine the real proportion of transplanted cells surviving after an intradermal injection, we performed an additional analysis using sex-mismatched transplantation (male BMDCs transplanted to female skin), followed by quantitative polymerase chain reaction (PCR) to measure the level of male sry gene. Number of the surviving cells was calculated using a standard curve (Supplementary Fig. S2). The obtained results revealed that about 70% of BMDCs were eluted from the tissue within 6 h after an injection, regardless of their genotype (Fig. 3D). Next, 70% and 90% of HO-1+/+ and HO-1−/− cells, respectively, did not survive for subsequent 3 days. Importantly, BMDC endurance on day 3 tended to be weakened in case of HO-1−/− cells (p=0.068). Thus, of the 200,000 cells injected, only about 1100 HO-1+/+ BMDCs and 600 HO-1−/− BMDCs were still present in the skin 7 days after the injection (Fig. 3D), and 1 week later their number was less than 50, preventing catching the specific PCR signal.
In another model, mice underwent femoral artery ligation and were subsequently (next day) intravenously injected with PBS, HO-1+/+, or HO-1−/− BMDCs. Unexpectedly, in wild-type mice, the adoptive transfer of BMDCs impaired tissue reperfusion, regardless of cell genotype (Fig. 3E). Measurements performed at day 14 also revealed the impaired blood flow recovery in HO-1−/− animals in comparison to wild-type counterparts. Again, the adoptive transfer of HO-1+/+ BMDCs did not improve the reperfusion but, instead, appeared to result in a disadvantageous effect (Fig. 3E). Quantitative PCR analysis of Sry level after a sex-mismatched intravenous injection of 500,000 cells was not sensitive enough to detect the injected cells in the injured muscle, either 6 h or at the later time points, which indicates that the homing of injected BMDCs to the ischemic tissue was weak (data not shown).
Hyperglycemia affects expression of proangiogenic genes in HO-1+/− mice
In the next set of experiments, we investigated whether a decrease in HO-1 expression may influence revascularization of ischemic tissue in diabetes. Since HO-1 promoter polymorphism in human populations leads to a strong variability in HO-1 expression and insufficient induction of HO-1 can be associated with cardiovascular complications in diabetic patients (11), we decided to perform the study using the more clinically relevant model of HO-1+/− instead of HO-1−/− mice.
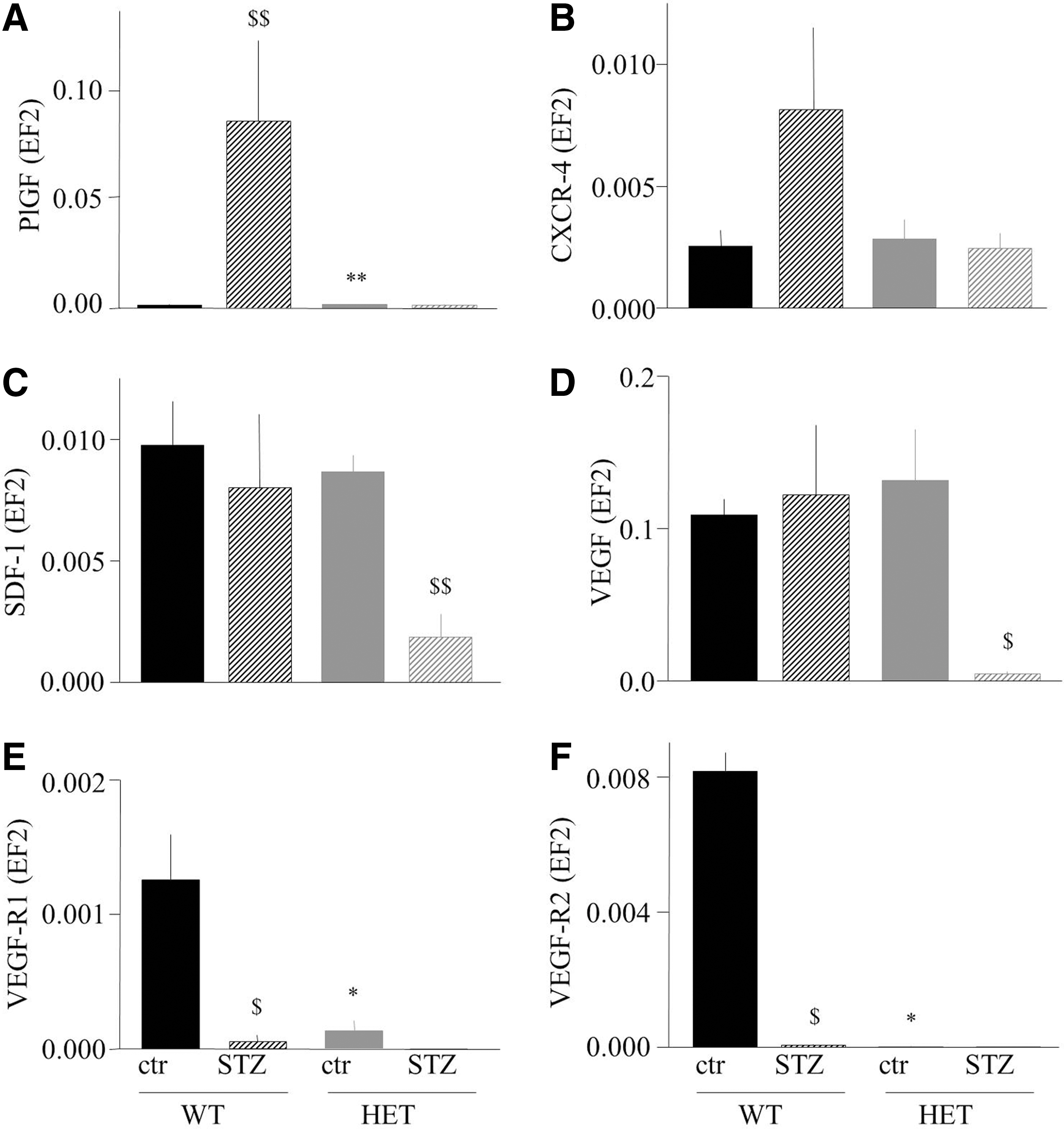
Hyperglycemia in mice was induced by injections of streptozotocin (STZ), with 100% efficiency of the procedure in both genotypes. Nevertheless, we observed a significantly higher mortality of HO-1+/− mice in comparison to HO-1+/+ counterparts (42.7% vs. 16.6%, respectively; p<0.05) 4 weeks after STZ dosing. Interestingly, there were obvious differences in the expression of proangiogenic genes in the caput gastrocnemius muscle between the wild-type and heterozygous animals (Fig. 4A–F). Namely, placental growth factor (PlGF) (Fig. 4A) and CXCR-4, the SDF-1 receptor (Fig. 4B), were up-regulated by STZ-induced hyperglycemia (in case of PlGF, significantly; in case of CXCR-4, it was a tendency, p=0.15) in the HO-1+/+ but not in the HO-1+/− individuals. On the other hand, the expression of SDF-1 (Fig. 4C) and VEGF (Fig. 4D) was lower in hyperglycemic HO-1+/− individuals in comparison to control HO-1+/− mice or to both HO-1+/+ groups, whereas the PlGF and VEGF receptors (VEGF-R1 and VEGF-R2) were strongly down-regulated in hyperglycemic groups and in normoglycemic HO-1+/− mice (Fig. 4E, F). Thus, gene expression analysis may suggest that tissue revascularization might be impaired in hyperglycemic HO-1 haplodeficient individuals.
Blood flow recovery is impaired in hyperglycemic HO-1+/− mice in spite of progenitor cell mobilization
We investigated the mobilization of progenitor cells (Sca-1+CXCR-4+) into peripheral blood in response to femoral artery ligation at day 1, 2, and 3 after surgery. Flow cytometry analysis showed that in wild-type mice, either normoglycemic or hyperglycemic induction of hind limb ischemia did not mobilize Sca-1+CXCR-4+ cells. In contrast, in HO-1+/− counterparts, the same procedure resulted in a significant increase in circulating progenitor cells, especially in hyperglycemic individuals, with a peak of mobilization at 24 h after surgery (Fig. 5A). Accordingly, SDF-1 gradient between ischemic muscle and bone marrow, measured at mRNA level, was the highest in hyperglycemic HO-1+/− mice (Fig. 5B).
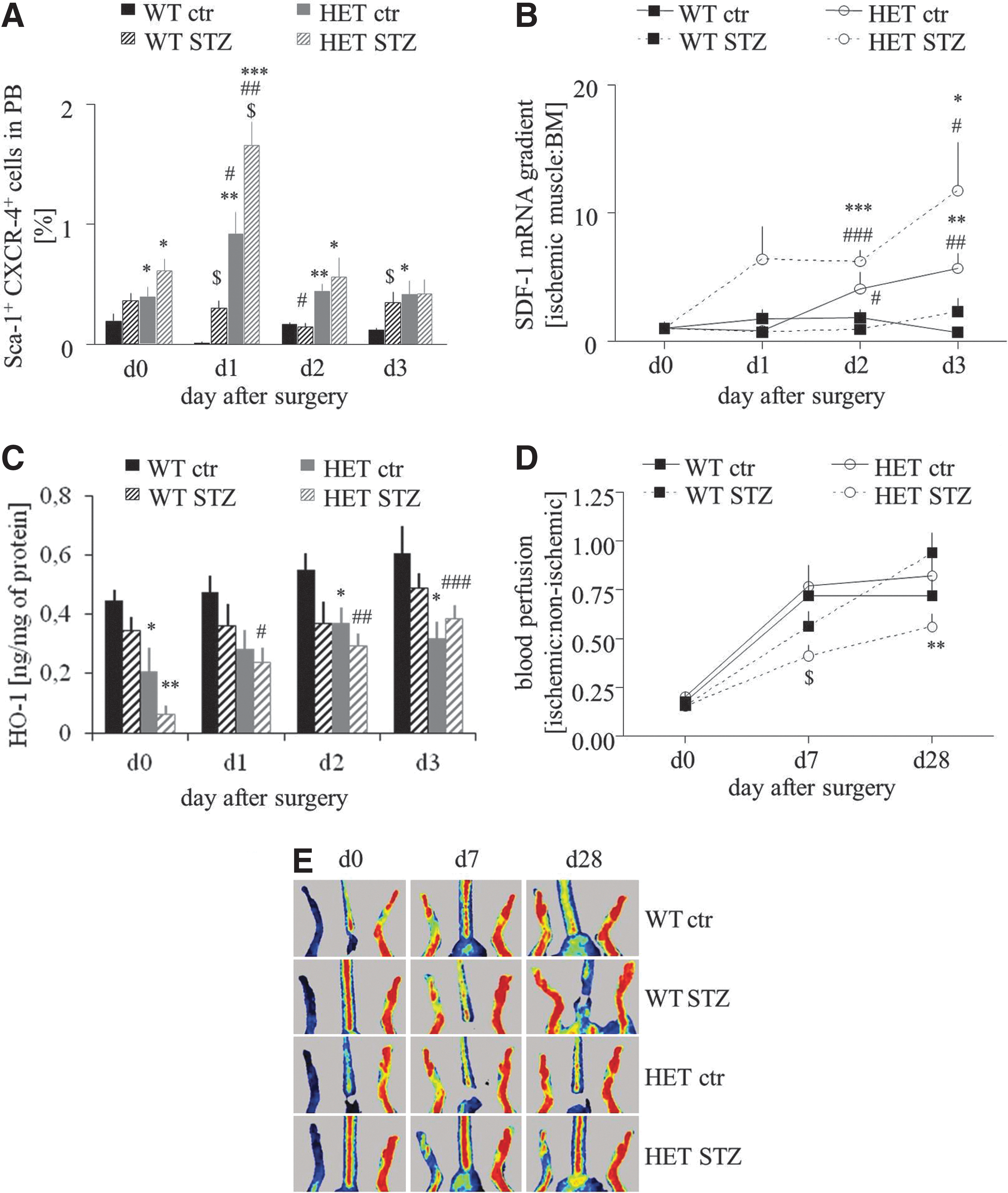
SDF-1 gradient did not correlate with the expression of HO-1 in the caput gatrocnemius muscle. Concentration of HO-1 protein in the healthy muscle of HO-1+/− mice was lower roughly by 50% in comparison to that of HO-1+/+ counterpart, and in both groups it was decreased after STZ treatment (Fig. 5C). Induction of ischemia led to a gradual increase in HO-1 protein levels, but this effect was significant only in the hyperglycemic HO-1+/− mice. Despite this up-regulation, concentration of HO-1 protein in the muscle remained lower in the HO-1+/− mice in comparison to the HO-1+/+ mice (Fig. 5C).
Thus, HO-1 haploinsufficiency in hyperglycemic mice diminishes the expression of proangiogenic genes (Fig. 4); on the other hand, it leads to the strongest mobilization of progenitor cells into peripheral blood in response to ischemia (Fig. 5A). To check which of these two processes is more significant in tissue regeneration, we investigated blood flow recovery in HO-1+/− and HO-1+/+ mice. Laser Doppler scanning did not reveal any difference between perfusion in normoglycemic HO-1 heterozygous and normoglycemic HO-1 wild-type animals at any time point. However, reperfusion was significantly impaired at day 28 post-surgery in hyperglycemic HO-1+/− animals when compared with hyperglycemic HO-1+/+ (Fig. 5D, E). Collectively, these results show that blood flow recovery is impaired in hyperglycemic HO-1+/− despite progenitor cell mobilization.
Cell-free strategy for therapeutic angiogenesis is an effective way to improve blood flow recovery
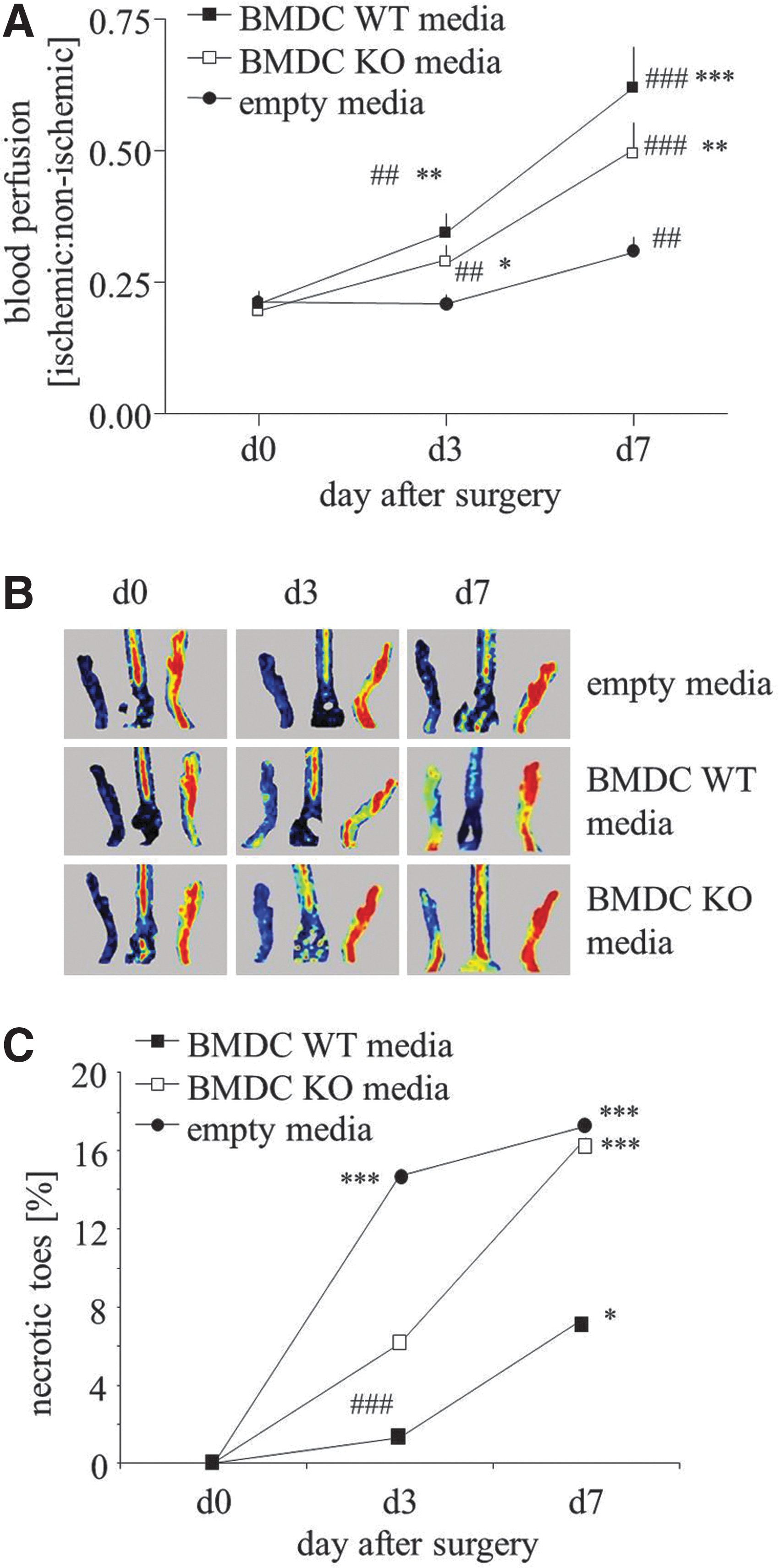
Since survival rate of locally injected cells was low (Fig. 3D) and an intravenous injection of BMDCs did not improve blood flow recovery and even resulted in deleterious effects (Fig. 3E), we used a cell-free strategy for therapeutic angiogenesis in a model of hind limb ischemia. For this purpose, hyperglycemic HO-1+/− mice, in which tissue revascularization is impaired (Fig. 5D, E), were subjected to angiogenic therapy utilizing conditioned EBM-2 media (with 0.5% FBS) either from HO-1+/+ BMDCs or from HO-1−/− BMDCs. Unconditioned EBM-2 medium with 0.5% FBS was used as a control.
Media injections were administered for 3 consecutive days after femoral artery ligation. Blood flow increased in ischemic limbs on day 3 post-surgery in animals treated with conditioned media in comparison to control group. This effect was prominent on day 7 (Fig. 6A, B). Although the improvement reached higher values in animals injected with media harvested from HO-1+/+ than HO-1−/− BMDCs, these differences were not statistically significant. Interestingly, the number of necrotic incidences (appearance of necrotic toes) was not fully parallel to the measured blood flow. Namely, 3 days after surgery, the number of necrotic toes significantly increased in control group (p<0.001), showed a tendency to increase in animals injected with media from HO-1−/− BMDCs (p=0.051), and did not significantly increase in counterparts injected with HO-1+/+ BMDC-derived media (p=0.485). On day 7, the increase in necrotic events became significant in all groups; however, the animals injected with media from HO-1−/− BMDCs were very similar to those injected with control media, whereas there was a tendency for weaker increase in animals treated with media from HO-1+/+ BMDCs (p=0.086).
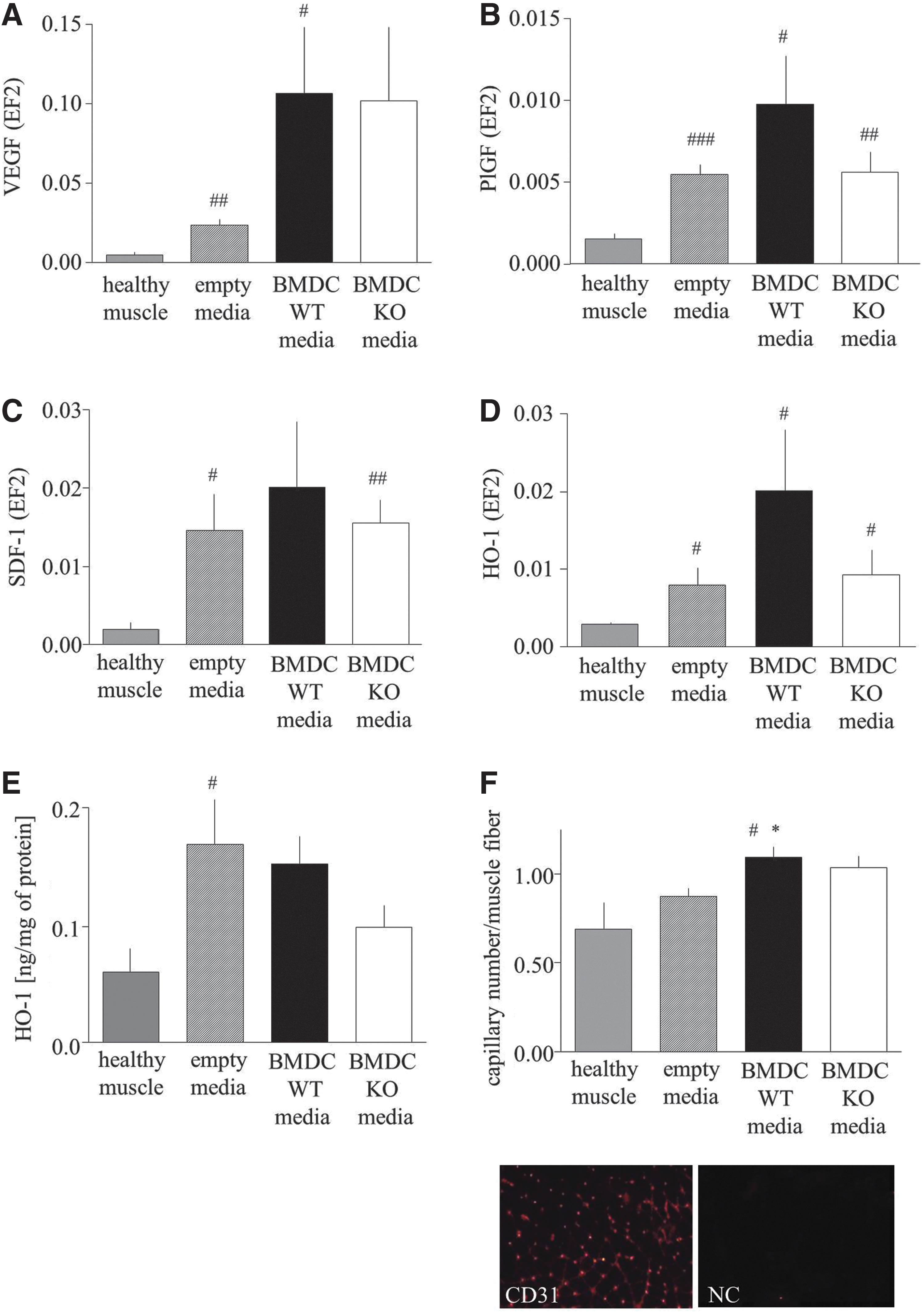
Analysis of gene expression revealed the tendency or significant up-regulation of VEGF, PlGF, and SDF-1 mRNAs in particular groups at day 7 after surgery (Fig. 7A–C). Interestingly, the HO-1 expression also was elevated in all investigated groups, which was shown at both mRNA (Fig. 7D) and protein (Fig. 7E) levels. Immunohistochemical staining for CD31 showed that the number of capillaries was significantly augmented in ischemic tissue in animals injected with HO-1+/+ BMDC-conditioned media (Fig. 7F).
Discussion
Exploitation of autologous patients' cells, subjected to ex vivo genetic modification to improve their regenerative potency, could be a promising strategy for the treatment of PAD. Hmox1 gene, encoding cytoprotective and proangiogenic HO-1 enzyme (17), is one of candidates considered for such therapy.
We found that the lack of HO-1 decreases the number of EPCs in murine bone marrow, and strongly inhibits the proangiogenic potential of cultured BMDCs, affecting their survival under oxidative stress, proliferation, migration, formation of capillary-like structures, and release of paracrine growth factors, although the influence on paracrine functions seems to be weaker than that on direct vasculogenic activities. These effects are accompanied by changes in BMDC transcriptome profile, including the reduced up-regulation of several proangiogenic genes in response to hypoxia. In addition, STZ-treated diabetic HO-1+/− mice exhibit lower expression of proagiogenic genes and impaired revascularization of ischemic muscles subjected to hind limb ischemia, in spite of strong mobilization of bone marrow-derived proangiogenic progenitors into peripheral blood.
We also observed that the administration of STZ causes a much higher mortality of HO-1+/− animals in comparison to their HO-1+/+ counterparts, with a concomitant down-regulation of HO-1. Since STZ is nephro- and hepatotoxic (36), and HO-1 was shown to be protective in these organs (48, 73), a low level of hyperglycemic HO-1 protein may explain the increased mortality of HO-1+/− individuals. Such decreased expression of HO-1 was also found in db/db mice (22) and in the Goto-Kakizaki rats (46), as well as in type 2 diabetes patients (1, 8, 12, 50).
Lack of HO-1 attenuates revascularization in HO-1−/− mice (26). Here, we demonstrated that haploinsufficiency of HO-1 does not influence the recovery of blood flow after hind limb ischemia in normoglycemic animals, but impairs it in hyperglycemic individuals. This corresponds to a clinical situation, as there is no association of PAD occurrence with Hmox1 promoter polymorphism (58), but less active Hmox1 alleles can increase the risk of coronary adverse events in diabetic patients (16). Noteworthy, the expression of HO-1 protein was lower by ∼40% in humans with less active variants of promoter (≥29 GT repeats) in comparison to those with more active (≤23 GTs) alleles (16). This indicates that HO-1 haploinsufficiency in mice is relevant to the diversity of HO-1 expression in the human population.
Blood flow recovery in our hyperglycemic wild-type mice was very effective. This might be astonishing, as some reports show a hyperglycemia-induced impairment in revascularization (9). However, the restoration of perfusion in STZ-treated mice may depend on the interval between diabetes induction and artery ligation (52). When surgery is done 4 weeks after STZ treatment, diabetic mice regenerate as effectively as normoglycemic animals (52), which has also been confirmed by the present study. Differently, reperfusion in hyperglycemic HO-1+/− mice was both delayed and diminished at the endpoint by 40% in comparison to HO-1+/+ animals. To our knowledge, this is the first report on STZ-treated HO-1 heterozygous mice. Earlier, Liu et al. showed that in the wild-type hyperglycemic animals the myocardial expression of HO-1 was strongly up-regulated in response to ischemia-reperfusion injury, whereas the lack of HO-1 exacerbates tissue damage (39). An insufficient level of HO-1 might be one of the possible reasons of impaired revascularization in hyperglycemic HO-1+/− animals.
It has been shown that the transfer of HO-1 transgene into ischemic muscle diminishes cell apoptosis, induces angiogenesis, enhances muscle perfusion, and limits autoamputations in mice and rats (6, 26, 60). In diabetic db/db mice, adenoviral delivery of HO-1 cDNA facilitates neovascularization and improves wound healing (22). Likewise, intracoronary transfer of HO-1 overexpressing bone marrow stromal cells increases expression of VEGF as well as capillary and arteriole density in a porcine model of myocardial ischemia reperfusion (27). Moreover, simultaneous transplantation of VEGF and HO-1 overexpressing cells facilitated blood flow recovery and decreased the number of necrotic incidents (41). These data indicate that in vivo revascularization is dependent on HO-1.
Impaired blood flow recovery in HO-1+/− hyperglycemic mice corresponds to down-regulation of proangiogenic genes in caput gastrocnemius muscle. Only in these animals, the expressions of all genes tested, namely PlGF, VEGF, SDF-1, VEGFR-1, VEGFR-2, and CXCR-4, are strongly decreased. In hyperglycemic HO-1+/+ and normoglycemic HO-1+/− animals, which regenerate efficiently, there is a low expression of VEGFR-2, but VEGF can also bind to neuropilin-1 and influence endothelial cells independently of VEGFR-2 signaling (3).
Many studies have described the beneficial effects of proangiogenic progenitors in tissue vascularization and regeneration [reviewed in Asahara et al. (2)]. We investigated the progenitor cell mobilization into peripheral blood in response to femoral artery ligation. Taking into account the monocytic origin of EPCs and endothelial-monocyte mimicry, we focused on Sca-1+CXCR-4+ cells, especially because such widely defined populations (e.g., MNCs, CD34+ cells, CD34+CD133+ cells), enriched in EPCs, were used in all clinical trials for the treatment of PAD (30, 33, 62). Mobilization of Sca-1+CXCR-4+ cells occurs only in HO-1+/− mice, and hyperglycemia enhances this process. However, blood flow recovery is impaired in hyperglycemic individuals. Therefore, we suppose that the mobilized cells are dysfunctional and do not contribute to tissue regeneration. They can be unable to home to injured muscle or can have reduced regenerative potency, and their release into circulation can only be a marker of stress reaction. Accordingly, Sca-1+CXCR-4+ cells were not mobilized in the effectively regenerating wild-type animals, although we cannot exclude that here the mobilization occurred earlier than 24 h after surgery and, therefore, was not captured in the study. Dispensability of Sca-1+CXCR-4+ progenitors in blood flow restoration is in line with the results of the REGENT clinical trial, where intracoronary infusion of CD34+CXCR-4+ cells (analogs of Sca-1+CXCR-4+ in mice) gave similar results as unselected bone marrow cells (63).
Interestingly, BMDCs seeded on Matrigel form capillary-like structures spontaneously, such as endothelial colony-forming cells (4) or late EPC (47), despite their phenotype resembling early EPC (47). This indicates that the cell phenotype is of secondary importance, and the regenerative potency of progenitors should be assessed in functional tests.
Our in vitro experiments reveal that HO-1 is necessary for proper activity of BMDCs. As expected (7, 51, 61), HO-1-deficient BMDCs are less viable when exposed to oxidants or after transplantation into the wound bed. They also display a weaker proliferation and migration either in control conditions or in response to growth factors. Similar effects were observed in murine mature or progenitor endothelial cells (13, 28) and in human endothelium with less active variants of Hmox1 promoter (61). We also demonstrated a weaker angiogenic potency of HO-1−/− BMDCs in capillary sprouting assay, confirming the results obtained earlier in human endothelial cells treated with HO-1 inhibitors (28) or in murine endothelial cells isolated from HO-1+/+ or HO-1−/− mice (13).
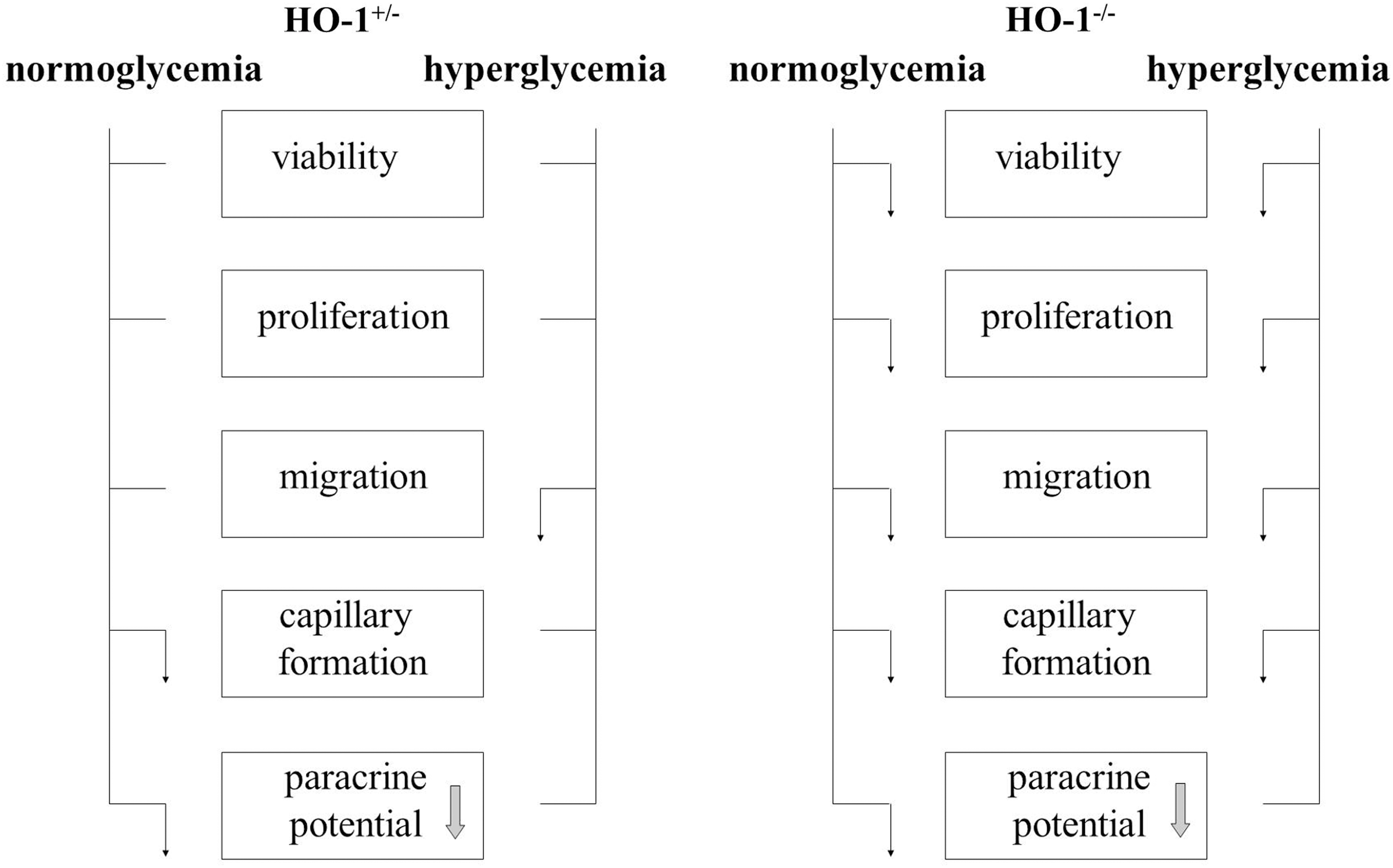
Reduced levels of HO-1 in HO-1+/− BMDCs cultured under normoglycemic conditions did not significantly affect cell viability, proliferation, and migration, but impaired angiogenic potential (summarized in Fig. 8). We observed such a limited effect of HO-1 haploinsufficiency in earlier experiments, where wound healing was strongly delayed in HO-1−/− animals. In 3-month-old HO-1+/− mice, this process was almost as efficient as in their wild-type counterparts. In older HO-1+/− individuals, however, the wound healing was significantly delayed (22). In current experiments, we found that hyperglycemia, in addition, reduced migratory capacities of the HO-1+/− BMDCs. Our in vitro experiments also indicate that hyperglycemia does not impair the formation of capillaries by BMDCs but significantly attenuates paracrine stimulation of endothelial cells.
In our hands, BMDC transplantation did not influence tissue regeneration (in a wound-healing model) or caused even deleterious effects (in hind limb ischemia model). Why an injection of BMDCs impaired blood flow recovery is not clear. We cannot exclude the fact that cells formed aggregates which occluded small blood vessels. We injected, however, only 0.5×106 cells, whereas 2×106 cells were used in some earlier experiments (10). Moreover, transplantation of 105–106 of the cells in murine models investigating therapeutic vascularization seems to be justified as a safe number of the cells that are able to promote re-endothelialization/neovascularization (25, 29, 32, 69, 72). The second possibility is that an injection of BMDCs, which contain monocyte-like cells, might lead to an increased inflammatory reaction. In case of the wound-healing model, lack of effect of BMDC injection on tissue regeneration might be related to the very fast clearance of these cells from the skin (∼70% after 6 h, ∼95% after 3 days, and ∼99.5% after 7 days). A small number of HO-1+/+ BMDCs can be recognized in the sections from the wounds 2 weeks after the injection, whereas HO-1−/− cells are almost lacking. Mobilization of the endogenous BMDCs might have also overridden the activity of exogenously administered cells. However, a similar situation may occur in patients in whom such an approach might be tested; hence, we believe that the observations provided here are of relevance for clinical conditions.
Since postnatal neovascularization is mostly regulated by paracrine signals (5), we employed a cell-free strategy for the stimulation of blood flow recovery in ischemic limbs. Such an approach may have two advantages: It is safer than an injection of cells (what applies all the more so to cells transduced ex vivo with viral vectors), and it avoids the problem of a large number of cells that are needed to be injected into a patient. Applications of proangiogenic growth factor cocktails produced ex vivo by the cells have been already carried out with positive effects (5, 15, 35, 37, 56, 64). Clinical trials with regard to monotherapy with proangiogenic proteins were not satisfying (21), and possibly a synergistic action of growth factors is needed for the formation of stable blood vessels (15). Our experiments show that an injection of conditioned media harvested from BMDCs accelerates the blood flow recovery in ischemic muscles of diabetic HO-1+/− mice. The therapeutic effect is clear for media harvested from both HO-1+/+ and HO-1−/− BMDCs; however, those from HO-1−/− cells tended to be less efficient. A limitation of this study is a lack of a precise phenotypic characterization of cell population(s) in heterogeneous BMDC cultures, which are responsible for this beneficial effect. Nevertheless, the protocol used enables obtaining heterogeneous but reproducible cell cultures. We suppose that augmented angiogenesis not only results from a direct stimulation of blood vessel formation by growth factors present in conditioned media, but is also accompanied by induction of proangiogenic genes at the site of the injection. Improved blood flow recovery might be also related to the vasodilatory effect of HO-1-derived CO; however, we did not observe a significant difference in control limb perfusion between HO-1+/+ and HO-1−/− hyperglycemic mice, which suggests that this mechanism does not play a crucial role here.
In summary, we demonstrated that HO-1 is necessary for proper proangiogenic function of BMDCs. Interestingly, angiogenic potential is also impaired in HO-1 haploinsufficient BMDCs. A low level of HO-1 in hyperglycemic mice leads to worse blood flow recovery in ischemic muscle, which can be rescued by an injection of conditioned media from cultured BMDCs, especially from cells with a higher expression of HO-1.
Materials and Methods
For a more detailed description of methods, please see the “Supplementary Materials and Methods” section.
Animals
HO-1−/− mice, HO-1+/− mice, and HO-1+/+ littermates (C57BL/6×FVB background, 12-week-old males) were used for all the experiments, except an analysis of EPC in bone marrow, where 1-year-old animals were employed.
Flow cytometric detection of EPC in bone marrow
Bone marrow cells were collected from tibias and femurs of adult mice, stained in PBS with 2% FBS for 20 min on ice with proper antibodies, and analyzed using an LRS II flow cytometer (BD).
BMDCs isolation and culture
Total bone marrow cells were flashed from tibias and femurs of HO-1+/+, HO-1+/−, and HO-1−/− mice and centrifuged on Ficoll gradient (400 g, 45 min, room temperature). Buffy coat cells were washed thrice with PBS and seeded on fibronectin (20 μg/ml) and gelatin (0.25%)-coated dishes in EGM-2 MV media (EBM-2 with supplement) containing 10% FBS and 5 mM glucose (LG) or 33 mM glucose (HG). All experiments were performed in cultures at a confluence of 90%, 7–10 days after isolation, except the measurement of proliferation, where cell confluence was 50%. Some cells were incubated for the last 24 h under hypoxic conditions (2% O2, 5% CO2, LG). To analyze the acLDL uptake and BS1 binding, the cells were incubated for 2 h in the presence of acLDL-DiI (20 μg/ml) or BS1-FITC (10 μg/ml) and then inspected under a fluorescence microscope (Nikon Eclipse TX-100).
HAECs culture
Cells were cultured in EGM-2 MV complete medium.
RNA isolation
RNA was isolated from tissues or cultured cells using a modified acid guanidinium thiocyanate-phenol-chloroform extraction method, precipitated in isopropanol, washed in ethanol, dissolved in water, and stored at −80°C.
Transcriptome analysis
Transcriptome of cultured BMDCs was analyzed using Agilent Whole Mouse Genome Oligo microarrays with binding sites for 44,000 sequences.
Detection of apoptosis
Cells were washed with PBS, stained with Hoechst 33342 and 7AAD according to the protocol by (59), and analyzed using an LRS II flow cytometer (BD).
Detection of proliferation
Proliferating cells were analyzed using flow cytometry after a Hoechst/pyronin Y staining.
Migration assay
Migration was measured using QCM Cell Migration Assay with inserts of a diameter of 8 μm (Chemicon).
Tube formation on Matrigel
Formation of tubes by HAECs was assayed using growth factor-reduced Matrigel in 96-well plates.
Spheroid culture assay
Spheroid culture assay was performed as previously described (68).
Cell therapy of wounds and ischemic limbs with BMDCs
For the wound-healing experiment, BMDCs isolated from HO-1+/+ or HO-1−/− mice were stained with PKH67. Two full-thickness excisional wounds were generated on either side of the dorsal midline of each mouse. Immediately after injury, 900,000 viable HO-1+/+ or HO-1−/− cells or PBS were injected intradermally into four places around the wound.
For the hind limb ischemia experiment, HO-1+/+ or HO-1−/− mice were subjected to femoral artery ligation (49). PBS or 500,000 BMDCs isolated from HO-1+/+ or HO-1−/− individuals were injected intravenously next day after the surgery.
Cell survival in vivo
Cell survival in vivo was determined after the transplantation of viable cultured BMDCs that were isolated from HO-1+/+ or HO-1−/− male mice into the tail vein (500,000 cells/animal) or into healthy skin (200,000 cells/animal) of HO-1+/+ female mice. The number of cells was quantified by detecting the male sry gene.
Induction of hyperglycemia
Hyperglycemia was induced by an intraperitoneal injection of STZ (50 mg/kg of body weight) for 5 consecutive days in HO-1+/− and HO-1+/+ mice. Individuals were regarded as hyperglycemic when fasting blood glucose level exceeded 240 mg/dl (31).
Gene expression analysis in caput gastrocnemius muscle
Total cellular RNA was isolated from caput gastrocnemius muscle and reversely transcribed. Gene expression at the mRNA levels was checked by real-time PCR. Total protein was isolated from caput gastrocnemius muscle. HO-1 expression at the protein level was checked by ELISA.
Hind limb ischemia in normoglycemic and hyperglycemic mice
Four weeks after STZ injections, normoglycemic and hyperglycemic mice were anesthetized and subjected to left femoral artery ligation to induce limb ischemia. The superficial blood flow of the ischemic and contralateral limbs was measured at day 0, 7, and 28 using a Laser Doppler Perfusion Imager System (PIM II; Perimed).
Progenitor cell mobilization
Peripheral blood was harvested from vena cava superior. Cells were stained with proper antibodies and analyzed using an LSRII cytometer (BD).
Cell-free therapy of ischemic limbs
BMDCs from HO-1+/+ or HO-1−/− mice were incubated in EBM-2 containing 0.5% FBS for 24 h. After this period, conditioned media were collected. HO-1+/− STZ-treated mice (n=12) were subjected to femoral artery ligation. Conditioned or control (EBM-2 with 0.5% FBS) media in a total volume of 30 μl were injected intramuscularly into caput gastrocnemius muscle at days 0, 1, and 2. Blood flow measurements and necrotic toe assessments were done at days 0, 3, and 7.
CD31 immunohistochemical staining
Gastrocnemius muscles were excised, embedded in OCT compound (Tissue-Tek), and snap frozen in liquid nitrogen. Cryostat sections were stained for CD31 to assess capillary density.
Statistical analysis
Results are expressed as mean±SEM. In vitro experiments were repeated at least thrice, while in vivo studies were performed in six mice per group, unless otherwise indicated. In the transcriptome study, the results were analyzed using one-tailed Bayesian ANOVA with post hoc Tukey HSD test, followed by p correction with Benjamini–Hochberg FDR test, at FDR <20%. In the other experiments, the two-tailed Student's t-test was used for a comparison of the groups and the two-tailed Fisher's exact test was used for a comparison of proportions.
Footnotes
Acknowledgments
The authors thank Ewa Zuba-Surma for technical help with some flow cytometry analyses. They truly appreciate the technical assistance of Agnieszka Andrychowicz-Rog, Aleksandra Bartelik, and Janusz Drebot. They are grateful to Anupam Agarwal, University of Alabama of Birmingham, for providing the HO-1+/− breeding pairs. This work was supported by European Union Framework Programs POIG 01.01.02-00-109/09 and 01.02.00-069/09 and by National Science Center grant N N301 460938. They also used the equipment obtained from EU Framework Programs POIG 02.01.00-12-064/08 and 02.02.00-00-014/08 (to Faculty of Biochemistry, Biophysics and Biotechnology). A.G.-P. is a recipient of L'Oreal Poland for Women in Science Scholarship and START Scholarship of Foundation for Polish Science (FNP). A.L. is the recipient of the POMOST grant from FNP, and Agnieszka Jazwa is the recipient of the HOMING Plus grant. Alicja Jozkowicz was an ISRF fellow supported by The Wellcome Trust. J.D. and Alicja Jozkowicz participated in the COST Action HypoxiaNet TD0901.
Author Disclosure Statement
The authors have no conflicting financial interests.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
