Abstract
Introduction
O
Nevertheless, it remains still uncertain whether oxidative stress increases secondary to the derangement of a specific subcellular compartment or to the imbalance of myofiber antioxidant systems. Increased iron levels and byproducts of lipid peroxidation significantly accumulated in the microsomal compartment of rat soleus muscles after 12-day unloading (21, 32). Sarcoplasmic reticulum (SR)/sarcolemmal NADPH-oxidase and cytosolic xanthine oxidase appeared to contribute to ROS production in the diaphragm muscle exposed to prolonged mechanical ventilation (41, 72). Dysregulated nitric oxide (NO) production, due to untethering of neuronal nitric oxide synthase (nNOS) from sarcolemma, and increased basal hydrogen peroxide formation in inner internal membrane of mitochondria also characterized unloading and immobilization of hindlimb muscles (42, 60). In addition to NO (33), increased levels of cytosolic calcium trigger mitochondrial ROS production (11). Except for a contrasting report, available body of evidence suggests the occurrence of dysfunctional calcium homeostasis in disused muscles (15, 25, 71, 74).
Skeletal muscle atrophy occurring after immobilization or prolonged bed rest represents a major invalidating condition. Disuse-induced disruption of neuronal nitric oxide synthase (nNOS) subsarcolemmal localization is recognized as an upstream event leading to myofiber atrophy. In this study, using an experimental animal model of muscle disuse atrophy (the hindlimb-suspended rat) and in vivo cDNA transfer, the authors identified the molecular chaperone Grp94 as a novel nNOS interacting partner, which was responsible for maintaining nNOS localization at myofiber sarcolemma and, thence, countered myofiber atrophy and oxidative stress.
Besides the antioxidant defense systems, a relevant role in antioxidant cytoprotection is attributed to molecular chaperones/stress proteins (27), among which the endoplasmic reticulum (ER) chaperone Grp94 distinguished itself for preventing protein carbonylation and cell death through its participation to the control of calcium homeostasis (4, 38, 46, 67). Grp94 binds calcium (7, 30) and passive ion release from the stores appears to be negatively related to the cellular content of the protein (4, 46).
Another interesting feature of Grp94 expression is its exclusive requirement for folding and secretion of insulin-like growth factor I and II (IGF-I and -II), the major positive autocrine regulators of muscle growth and regeneration (63, 69). IGF-I protein levels decreased in unloaded muscles (20), whereas increased expression of a recombinant muscle IGF-I isoform countered wasting accompanying sarcopenia and muscular dystrophies (63).
We then wondered whether Grp94 overexpression would exert an anti-atrophic role in unloaded skeletal muscle fibers, either by increasing myofiber antioxidant cytoprotection or by improving IGF maturation and release. Therefore, we investigated the effects on myofiber cross-sectional area (CSA) and presence of carbonylation after in vivo manipulation of Grp94 protein levels in soleus muscles of the tail-suspended rat. Our results indicate that increased Grp94 expression countered both oxidative stress and atrophy progression of unloaded soleus muscles, acting through an intrinsic pathway that hampered nNOS untethering from sarcolemma.
Results
Grp94 expression in unloaded soleus muscles
Soleus muscle weight/body weight ratio was used to monitor the presence of atrophy. In tail-suspended rats, it significantly decreased by about 28% after 7-day unloading compared with ambulatory rats (0.32×10−3±0.01×10−3 and 0.44×10−3±0.03×10−3, mean and standard error (SE) values, respectively, n=6, p<0.004), consistent with literature data (10, 26).
Protein carbonylation was evaluated by Oxyblot analyses (Fig. 1A). Unloaded soleus muscles displayed increased intensity on several polypeptides, compared with muscles from ambulatory rats. Densitometric analyses showed that normalized levels of protein carbonylation increased about twofold after 7 days of unloading compared with those of ambulatory muscles (p=0.01).
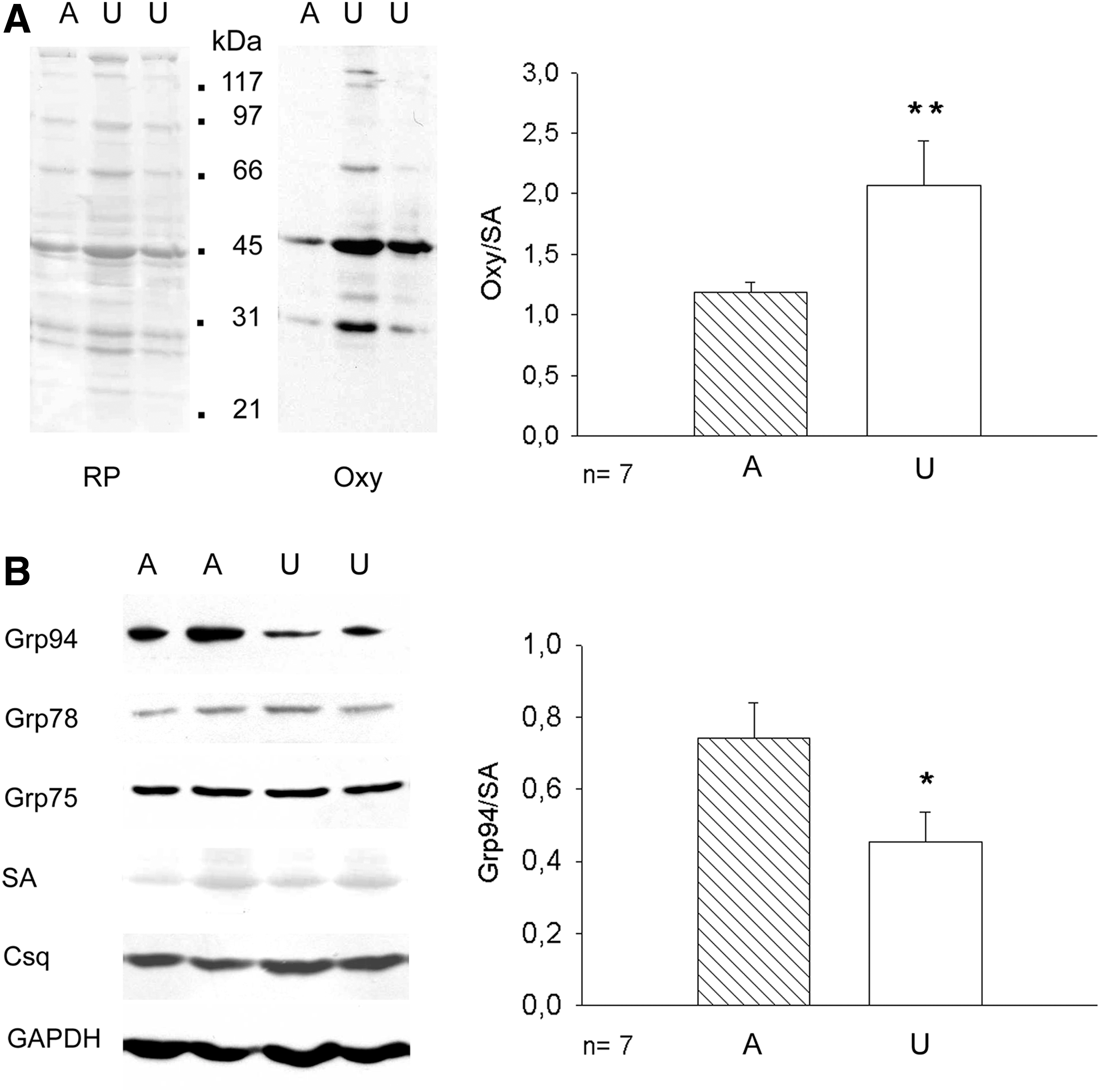
Western blot analyses showed that the relative amount of Grp94 protein significantly decreased in unloaded soleus muscles, either using as loading control serum albumin (SA; p=0.05, Fig. 1B) or glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (mean values±SE 0.44±0.05 and 0.16±0.02 for ambulatory and unloaded solei, respectively, n=7, p=0.001, Student's t-test). Consistent with previous reports (10, 23), protein levels of other SR/ER chaperones such as Grp78 and Grp75 did not appear to significantly change after 7 day-unloading (mean values±SE 0.53±0.11 and 0.44±0.09 for Grp78 levels of ambulatory and unloaded solei, respectively, p=0.53; mean values and SE 0.94±0.20 and 0.71±0.16 for Grp75 levels of ambulatory and unloaded solei, respectively, p=0.42), nor were affected those of calnexin and calreticulin (Supplementary Fig. S1A; Supplementary Data are available online at
In vivo transfer of Grp94 cDNA decreases myofiber atrophy
Grp94 protein was overexpressed in soleus myofibers by means of in vivo transfection of the pT94 plasmid, which contained grp94 cDNA (67). Control transfections were performed using the empty vector codifying for green fluorescent protein (pT vector). Transfected fibers were identified by the presence of the green fluorescent protein (GFP), whose cDNA was carried by the vector (Fig. 2A). The presence of recombinant Grp94 in GFP-positive fibers was visualized using a monoclonal antibody (mAb) (clone 3C4; 65) that does not recognize the rat protein (67; Fig. 2B). Myofiber CSA was measured 8 days after surgery, excluding myofibers that expressed embryonic myosin (ɛMy), because of regeneration induced by the transfection protocol (Fig. 2B).
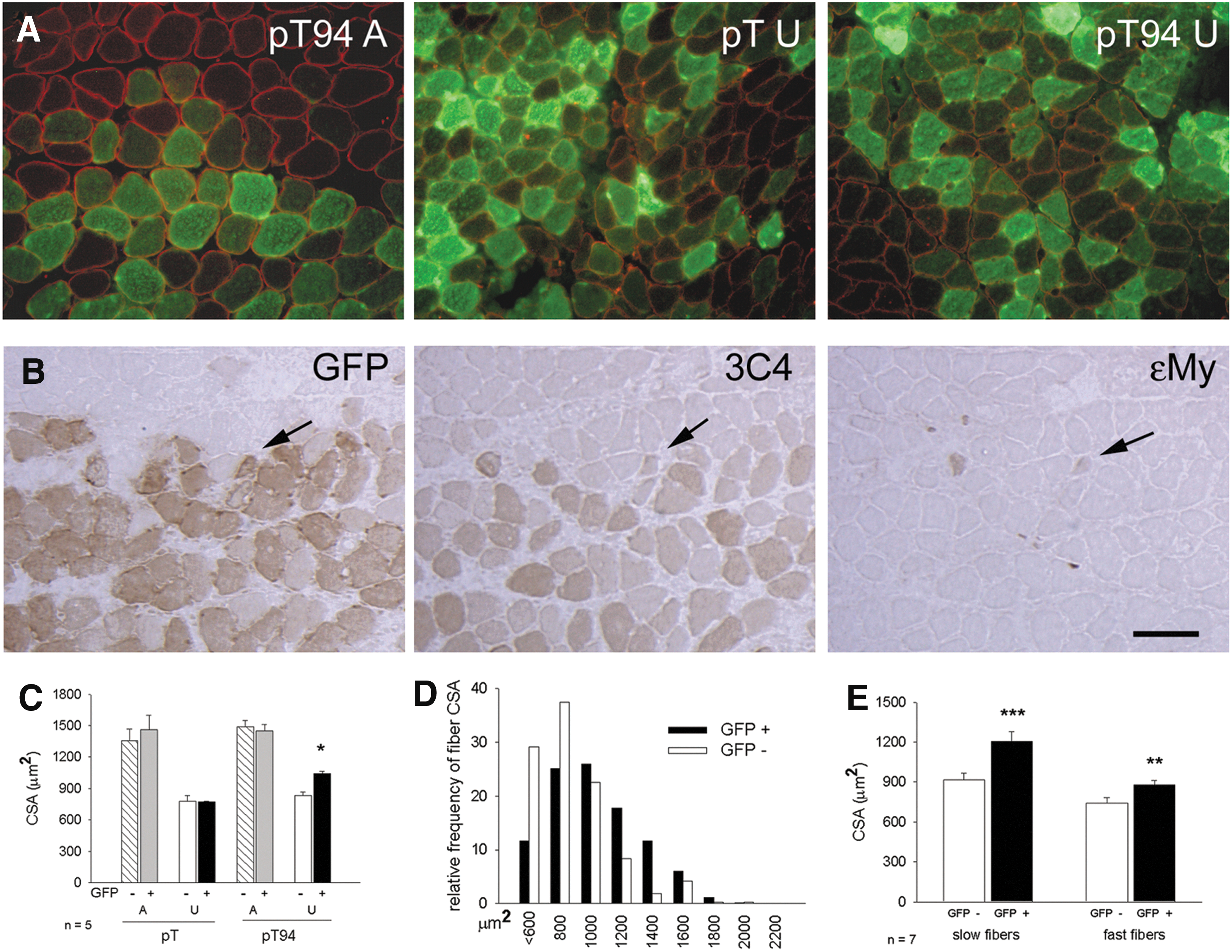
Fiber CSA of ambulatory soleus muscles was unaffected by the transfection protocol or by the presence of recombinant Grp94, because comparable values were measured in untransfected fibers and in those transfected with either pT or bicistronic vector codifying for recombinant Grp94 and GFP (pT94; Fig. 2C). When atrophy was induced in transfected soleus muscles by 7 days of hindlimb unloading, significantly higher CSA values were observed for recombinant Grp94-expressing fibers, compared with untransfected ones and with those transfected with the empty vector (p<0.01; Fig. 2C). Comparison of mean CSA values between transfected fibers of ambulatory and unloaded muscles showed that the degree of atrophy was about 28% after transfection with pT94, whereas it corresponded to 44% after transfection with the empty vector. The milder degree of atrophy displayed after pT94 transfection was confirmed by distribution analysis of fiber CSA between transfected and untransfected fibers of unloaded solei (Fig. 2D). We also separately determined the effect of pT94 transfection on CSA of unloaded slow and fast myofibers, which showed a significantly higher CSA, compared with untransfected ones (p=0.001 for slow fibers, and p=0.003 for fast fibers; Fig. 2E).
Although precursor levels of the Grp94 client proteins IGF-I and II were apparently unaffected in our unloaded muscles (Supplementary Fig. S1B), their distribution was investigated by immunohistochemistry in ambulatory and unloaded transfected muscles (Supplementary Fig. S2). No difference in staining with anti-IGF-I was observed. Conversely, IGF-II immunoreactivity appeared slightly more concentrated at the subsarcolemma of ambulatory myofibers compared with unloaded ones, and again stronger in unloaded transfected myofibers expressing exogenous Grp94.
In vivo transfer of Grp94 cDNA decreases myofiber carbonylation
We investigated whether recombinant Grp94 increased myofiber antioxidant protection, using carbonyls as marker of oxidative stress. To avoid artifactual oxidation, cryosections from only two samples (usually an ambulant muscle and an unloaded one or a pT94-transfected soleus and a pT-transfected one) were collected onto the same slide, reacted with 2,4-dinitrophenylhydrazine (DNPH), and the presence of carbonyl adducts demonstrated by indirect immunoperoxidase with anti-DNPH antibodies. Soleus myofibers showed usually weak and heterogeneous DNPH immunoreactivity (Fig. 3A, upper row).
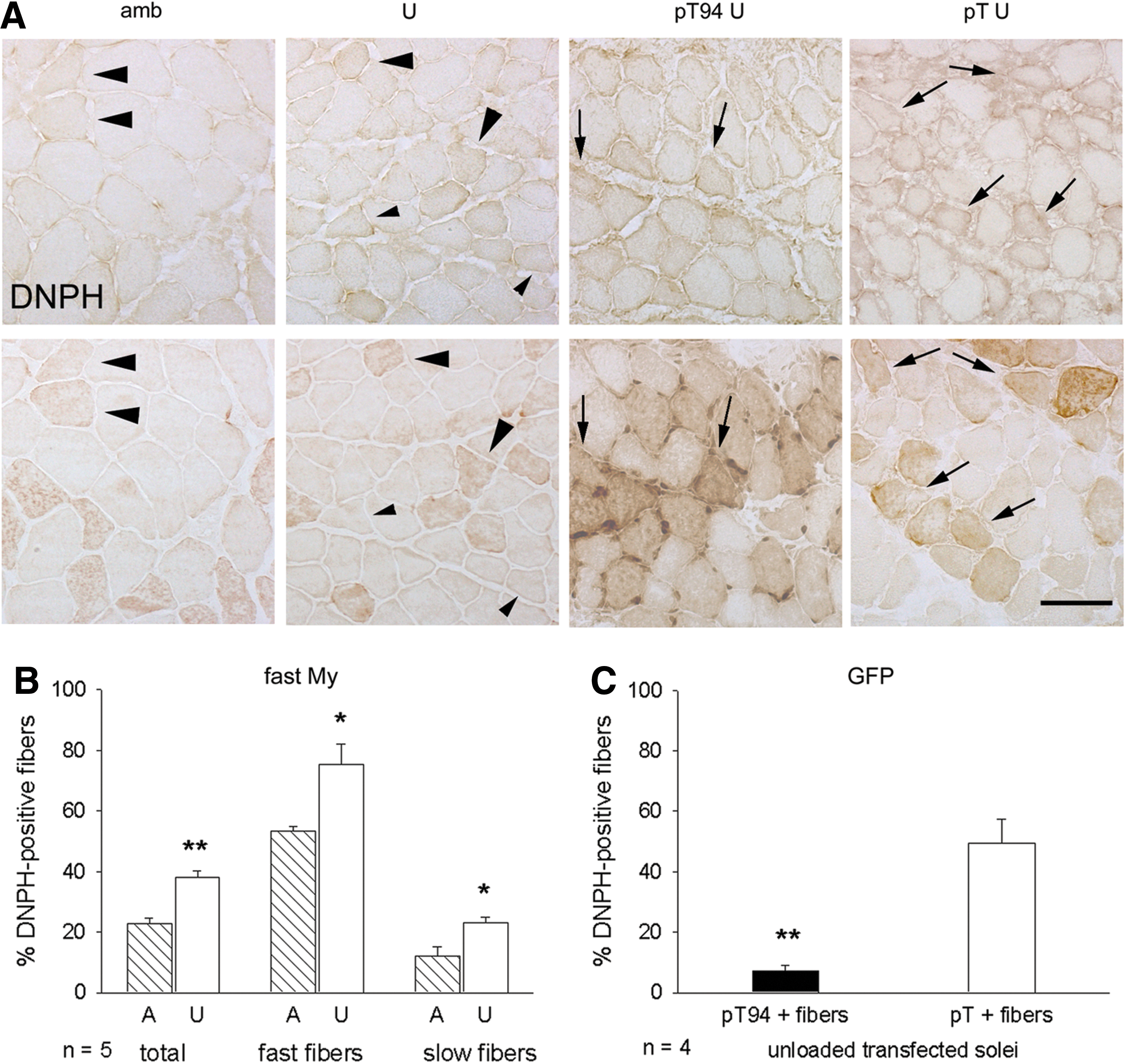
To validate visual inspection as an approach to discriminate between DNPH-positive and -negative myofibers, densitometry was performed using the Image J software on 10 positive and 10 negative fibers randomly collected from each soleus muscle of five ambulatory and five unloaded rats. Mean optical density (OD) of DNPH-positive fibers was significantly higher than that of negative ones in both unloaded and ambulatory muscles (mean and SE values of absorbance of DNPH-positive and -negative fibers of unloaded muscles were 118.20×10−4±8.78×10−4 and 106.15×10−4±5.07×10−4, respectively, p=0.013; mean and SE values of absorbance of DNPH-positive and -negative fibers of ambulatory muscles were 113.43×10−4±5.74×10−4 and 102.02×10−4±6.00×10−4, respectively, p=0.0019, paired Student's t-test).
About 20% of total myofibers of ambulatory soleus muscles displayed positive immunoreactivity for DNPH. These reactive fibers corresponded to about 50% of the fast fiber population (Fig. 3A, large arrowheads, B) and less than 10% of the slow fiber one. In unloaded muscles, staining for DNPH involved about 40% of total myofibers, corresponding to about 75% of the fast fiber population and significantly doubling their percentage within the slow fiber population (Fig. 3A, small arrowheads, B; p<0.02). In unloaded transfected muscles, the distribution of DNPH immunoreactivity was analyzed considering GFP-positive and GFP-negative fibers (Fig. 3A). The percentage of unloaded transfected fibers showing DNPH immunoreactivity was sevenfold less after transfection with pT94, the large majority appearing unstained, at variance with myofibers transfected with the empty vector (Fig. 3C, p=0.001).
Similarly, a lower percentage of DNPH-positive myofibers was detected among pT94 transfected myofibers of ambulatory muscles, compared to untransfected ones (Supplementary Fig. S3).
No effect on myofiber atrophy after expression of N-terminal deleted Grp94
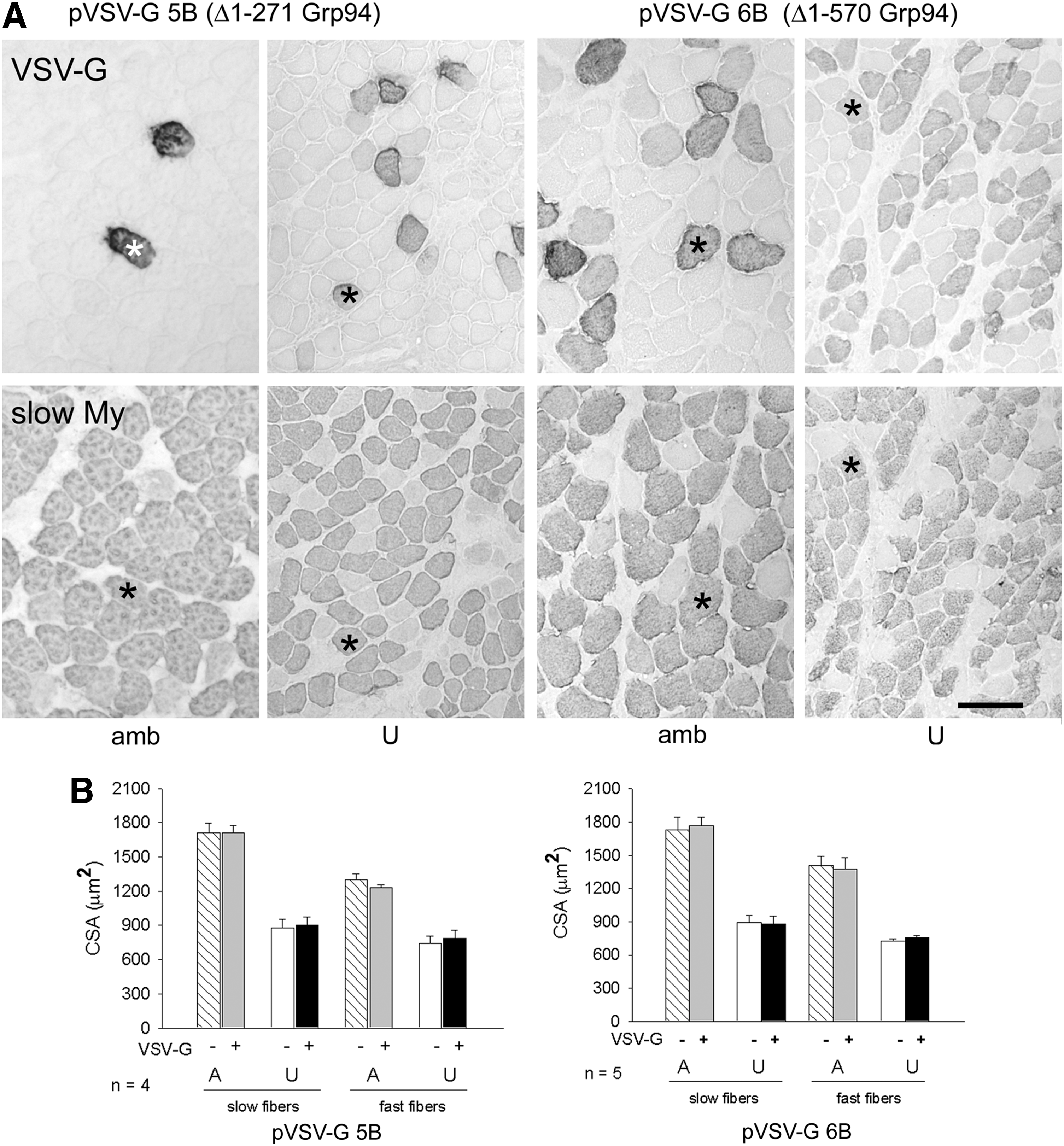
To validate the involvement of Grp94 in attenuation of muscle mass loss, transfection was performed using constructs (pVSV-G5B and vector codifying for VSV-G tagged N-deleted Grp94 [pVSV-G6B]) that codified for deleted N-terminal Grp94 forms. The former expressed a Δ1-271 Grp94 protein that lacked the nucleotide binding domain (Supplementary Fig. S4A); the latter expressed a Δ1-570 Grp94 form, which lacked three N-terminal putative protein-binding sites (3, 58), but retained one (AA 652–754) (73). The Δ1-570 Grp94 protein also participates to calcium homeostasis in a manner similar to the full-length Grp94 (46 and P. Pizzo, C. Scapin and L. Gorza, unpublished observations). The expression of the exogenous proteins was monitored by the presence of the VSV-G tag, which was added to the construct at the N-terminal region, immediately downstream the signal peptide required for the ER import. Figure 4A shows representative VSV-G immunohistochemistry of transfected ambulatory soleus muscles and 7-day unloaded ones. Fiber CSA of ambulatory soleus muscles was unaffected by the expression of the deleted Grp94 forms, either considering fast or slow fiber populations (Fig. 4B). A comparable result was observed for unloaded myofibers, whose CSA appeared reduced compared to myofiber values of ambulatory muscles, independently from the presence of deleted Grp94 (p<0.01; Fig. 4B).
nNOS interacts with Grp94
The effect of recombinant Grp94 expression upon atrophy and carbonylation appeared circumscribed to transfected fibers, therefore excluding a major mechanistical contribution through increased IGF secretion (53) and suggesting the chaperone involvement in other pathways. Immunoprecipitation experiments, performed using two different anti-Grp94 antibodies to explore physiological interactions with molecules promoting myofiber atrophy development, showed that Grp94 co-immunoprecipitated from lysates of both skeletal muscle and cerebellum with a 160 kDa polypeptide recognized by anti nNOS antibodies (Fig. 5A). Similarly, anti-nNOS polyclonal antibodies (pAbs) immunoprecipitated Grp94, but not Grp75, another ER chaperone/stress protein involved in antioxidant cytoprotection and known to interact with Grp94 (10, 29; Fig. 5B). Specificity of the antibodies used is illustrated in Supplementary Figure S4B.
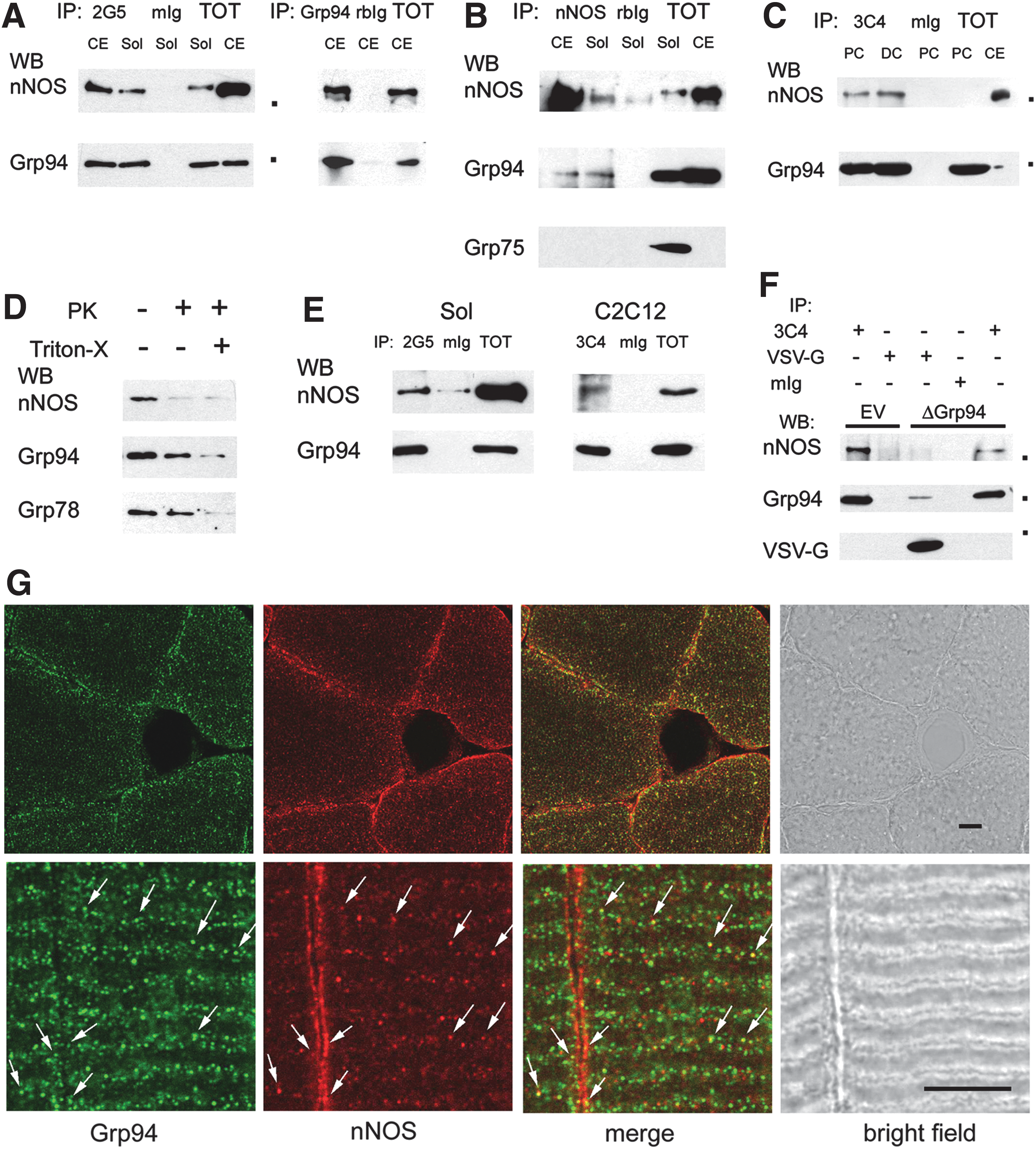
In order to exclude the aspecific nature of such interaction, cultures of the murine myogenic cell line C2C12 were exposed to a cell-permeant protein cross-linker before lysis and Grp94 was immunoprecipitated using a third different anti-Grp94 mAb (19, 66). Western blot analysis confirmed the presence of interaction of the chaperone with nNOS, in both proliferating and differentiating C2C12 myoblasts (Fig. 5C).
Since these results suggested an interaction between a cytosolic protein (nNOS) and an essentially lumenal one (Grp94), topology of this interaction was explored using crude membrane preparations obtained from soleus muscle of ambulatory rats (Fig. 5D). Microsomes were exposed to proteinase K (PK) digestion in the absence or presence of Triton-X (14). Addition of 200 ng/ml PK was sufficient to abolish nNOS immunoreactivity, whereas the presence of 0.5% Triton-X was required to blunt Grp94 immunostaining, which however appeared already reduced after exposure to PK alone. Immunostaining of the same blots for another lumenal protein, such as Grp78, revealed a 25.00±5.85% less decrease in labeling intensity (mean and SE of three different experiments), suggesting that a proportion of Grp94 molecules assumed a transmembrane localization (see also Supplementary Fig. S4C). Cross-linked microsomes, either directly prepared on crude membrane fraction obtained from soleus muscle or fractionated from cross-linked C2C12 cultures, demonstrated further co-immunoprecipitation of nNOS and Grp94 (Fig. 5E).
To ascertain whether the interaction with nNOS occurred also with Δ1-570 Grp94 form, immunoprecipitation assays were performed on lysates obtained from C2C12 cells after transient transfection with pVSV-G 6B and chemical cross-linking. Δ1-570 Grp94, immunoprecipitated by anti-VSV-G antibodies, showed an apparent Mr of 34,000 Da and did not co-immunoprecipitate nNOS, at variance with immunoprecipitated endogenous Grp94 from parallel aliquots of the same lysates and from C2C12 cells transfected with the empty vector (Fig. 5F).
Analysis of the subcellular distribution of nNOS and Grp94 was performed by confocal microscopy on both transverse and longitudinal cryosections of soleus muscles (Fig. 5G). Grp94 labeling, obtained using two different anti-Grp94 antibodies, concentrated at the subsarcolemmal region and extended amid myofibrils (Fig. 5G, upper panel, green fluorescence, and not shown), diffusely staining the I band, but the Z line (Fig. 5G, lower panel, and not shown). Anti-nNOS antibodies strongly labeled the subsarcolemmal region and weakly the sarcomeric I band (Fig. 5G, red fluorescence). Co-localization of Grp94 and nNOS signals involved a minor population of globular structures, adjacently to the A-I boundary and sarcolemma (Fig. 5G, arrows).
Grp94 upregulation recovers subsarcolemmal NADPH-diaphorase activity in unloaded muscles
nNOS untethering from sarcolemma represents a major initiating event leading to disuse muscle atrophy (60). Antibodies to nNOS stained sarcolemmal portions in cross sections of ambulant rat muscles (Figs. 5G, upper row, and 6A, arrows; 50), whereas immunolabeling at these sites appeared grossly reduced in unloaded solei (Fig. 6A), although western blot analyses (Fig. 6B) did not reveal any change in the relative amount of the protein (mean values±SE 0.77±0.13 and 0.73±0.13 for ambulatory and unloaded solei, respectively, n=5, p=0.83, Student's t-test).
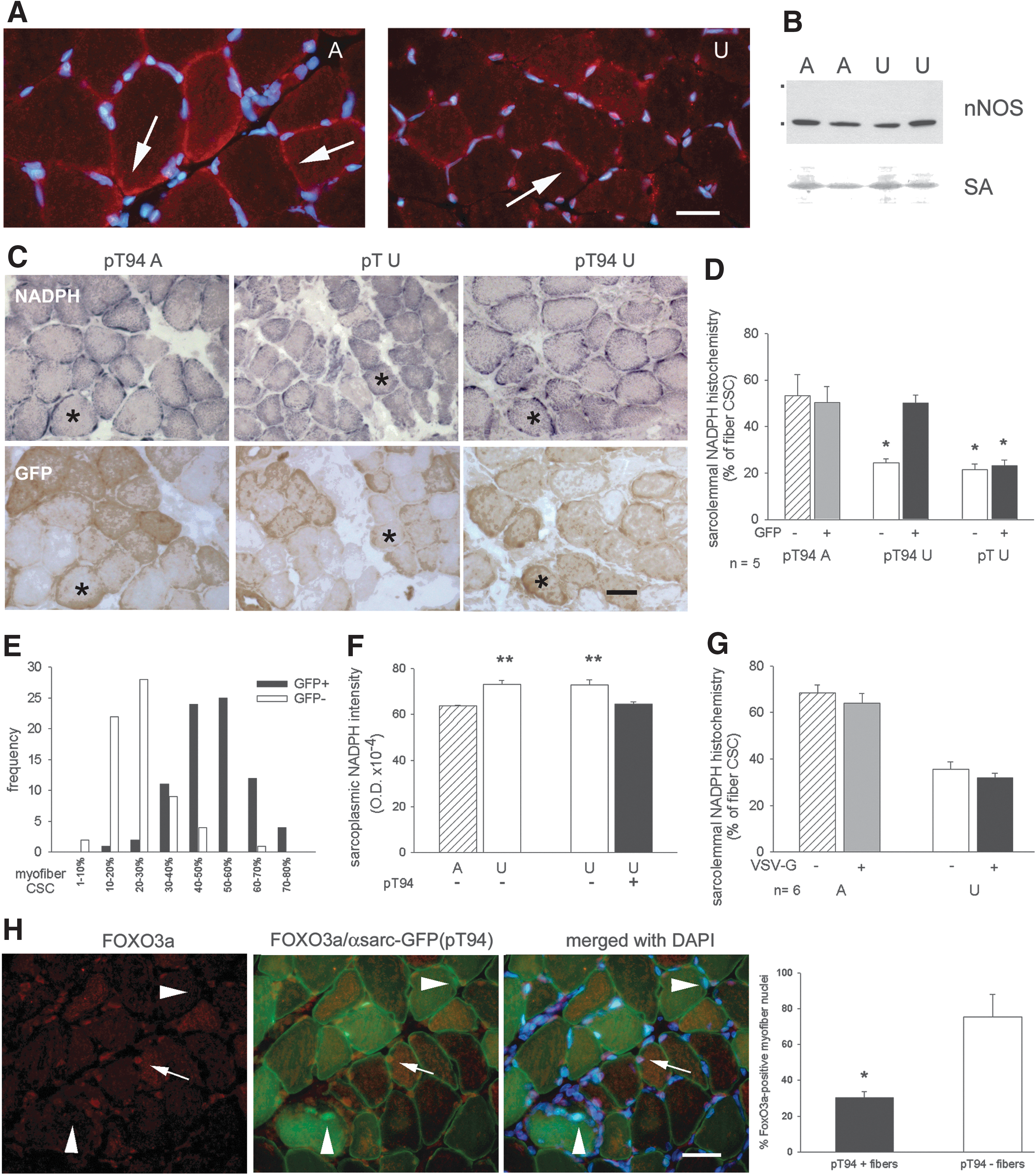
A consistent distribution was observed using the histochemical reaction for NAPDH-diaphorase (NADPH-d) (50), which had the advantage to reveal the presence of the active enzyme compared to immunolocalization (Fig. 6C). In ambulatory soleus muscle, staining appeared concentrated in discrete sarcolemmal regions corresponding to about 50–60% of fiber cross-sectional circumference (CSC). Enzyme histochemistry for NADPH-d was then used to evaluate the percentage of sarcolemma that bound active nNOS in transfected myofibers, identified in serial consecutive sections using anti-GFP antibodies and indirect immunoperoxidase. Sarcolemmal distribution of NADPH-d activity appeared significantly reduced in unloaded myofibers, either transfected with the empty vector or untransfected ones, compared to ambulatory muscles (p<0.01), whereas it appeared unaffected in myofibers transfected with pT94 (Fig. 6C–E).
On the other hand, the decreased sarcolemmal distribution observed in unloaded myofibers was accompanied by a significant, albeit slight, increased intensity of sarcoplasmic NADPH-d enzyme histochemistry, which was indicative of accumulated active nNOS (Fig. 6F). Transfection with exogenous Grp94 significantly decreased intensity of sarcoplasmic NADPH-d activity in unloaded muscles (Fig. 6F).
The same analysis on sarcolemmal distribution of NADPH-d activity was repeated in muscles transfected with the construct codifying for Δ1-570 Grp94. Unloaded untransfected myofibers showed a significantly reduced percentage of CSC positive for sarcolemmal NADPH-d activity, which, however, did not increase after transfection with pVSV-G 6B (p<0.01, Fig. 6G).
Since our transfection protocol allowed analyses at the fiber level only, no biochemical assay could be performed to investigate the effect of Grp94 overexpression on nNOS-released NO endpoints in unloaded muscles. Nevertheless, immunofluorescence studies revealed a negative effect on pro-catabolic signals, such as reduced nuclear localization of FoxO3a and ubiquitin labeling, in pT94-transfected myofibers (Fig. 6H; Supplementary Fig. S5).
nNOS mediates the trophic effects of Grp94 upregulation in unloaded muscles
To demonstrate that Grp94 acted upstream nNOS dysregulation in unloaded muscles, the specific nNOS inhibitor 7-nitroindazole (7-NI) was administered to ambulatory and hindlimb-suspended rats (60). Control rats received peanut oil only. Seven-day treatment almost abolished subsarcolemmal NADPH-d enzyme histochemistry in ambulatory muscles (Fig. 7A), without affecting both subsarcolemmal nNOS localization (not shown) and the co-immunoprecipitation with Grp94 (Fig. 7B).
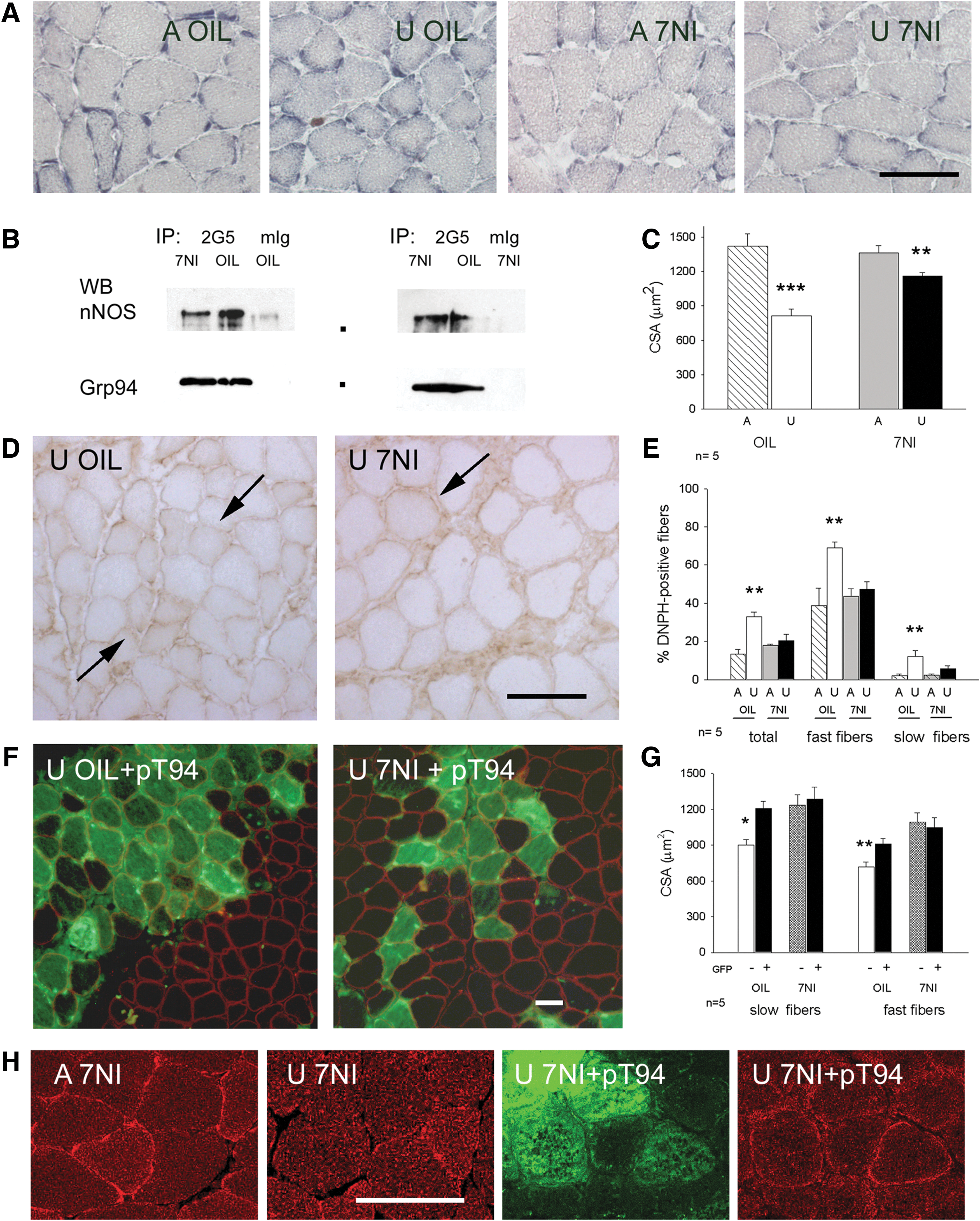
Consistent with Suzuki et al. (60), 7-NI treatment during hindlimb unloading significantly attenuated myofiber atrophy of soleus muscle, compared with oil-treated unloaded rats, (Fig. 7A, C; analysis of variance [ANOVA] p=0.003). Further, it affected myofiber carbonylation. The percentage of DNPH-positive myofibers remained at ambulatory level in 7-NI-treated unloaded solei, at variance with oil-treated unloaded muscles, where they significantly increased (p<0.01; Fig. 7D, E). Similarly, no significant increase in protein carbonylation levels was observed by Oxyblot analysis between ambulatory and unloaded solei of 7-NI-treated rats (Supplementary Fig. S6).
Eventually, transfection with pT94 of soleus muscles of unloaded rats treated with 7-NI did not significantly increase further CSA of either slow or fast myofibers, compared to oil-treated unloaded rats, proving that nNOS is the mediator of the trophic effects of exogenous Grp94 expression in unloaded muscles (Fig. 7F, G). Expression of recombinant Grp94 still counteracted the loss of nNOS immunofluorescence labeling at the sarcolemma of unloaded solei, since inhibition of nNOS activity by means of 7-NI did not hamper the untethering of the enzyme, as demonstrated by confocal microscopy (Fig. 7H).
Conversely, recombinant Grp94 further decreased the percentage of DNPH-positive myofibers compared with untransfected ones (mean and SE 8.87±2.53% and 20.28±3.49%, respectively, evaluated on 954 GFP-positive myofibers from four unloaded solei of 7-NI treated rats; p=0.04, Student's t-test).
Discussion
This study demonstrates that the SR/ER chaperone Grp94 plays a relevant role against myofiber carbonylation and atrophy of unloaded soleus muscle, and mechanistically explains its involvement identifying nNOS as a new interacting partner. The expression of recombinant Grp94 in unloaded muscle fibers affected nNOS subcellular distribution, favoring its localization at the sarcolemma and countering the occurrence of atrophy and oxidative stress.
Grp94 counteracts atrophy of unloaded soleus muscle
Grp94 is highly expressed by skeletal myogenic cells, where it is specifically involved in differentiation and maturation (14, 19, 44). The absolute requirement of this chaperone for IGF folding and secretion hampered muscle cell differentiation in Grp94 knock-out (KO) embryoid bodies (69) and decreased muscle size after conditional muscle-specific Grp94 KO (5). Further, Grp94 possesses calcium-binding ability and participates in the control of calcium homeostasis, displaying cytoprotective properties, which appear to be positively related to the intracellular chaperone protein levels (4, 46, 67). Different from developing muscles and other adult tissues, Grp94 is expressed at low protein levels in adult mammalian skeletal myofibers (24, 63, 65, 66), and here we show that unweighting decreases them further.
Stoichiometry represents a relevant feature of chaperone-client protein interaction (39). Increased expression of cytosolic chaperones/stress-proteins, such as Hsp70 and Hsp27, attenuated muscle atrophy secondary to limb immobilization (12, 55). Here, we demonstrate that muscle atrophy could be mitigated antagonizing Grp94 decrease by means of the expression of a recombinant one, whereas no effect on myofiber size was observed in standard caged rats. Due to the low number of transfected fibers we could not demonstrate changes in Grp94 protein levels; nevertheless, an indirect proof of the relevance to maintain adequate Grp94 protein amount during unloading was provided by the lack of effects after transfection with two differently deleted Grp94 forms.
A first mechanistic consideration has to take into account the possible involvement of IGFs. Although our findings would not exclude increased IGF maturation by recombinant Grp94, the lack of atrophy attenuation of adjacent untransfected myofibers questions IGFs as the actual mediators of the chaperone effects. Resistance to IGF signaling characterizes exposure to microgravity and hindlimb unloading (1, 20, 34, 56). Further, autocrine and paracrine effects of IGFs against muscle atrophy apparently occur concomitantly, since atrophy attenuation interested both transfected and untransfected myofibers after electroporation with IGF-I cDNA (53). Therefore, our results suggested the requirement of Grp94 for different intrinsic regulatory pathways, whose disruption contributed to muscle disuse atrophy.
Grp94 as a new partner of nNOS
Skeletal muscle fibers express multiple nNOS isoforms, which differentially localize at the subsarcolemma and Golgi (45). Although the Golgi nNOS isoform was recently identified as a major negative regulator of muscle contractility, decreased nNOS expression at sarcolemma (51, 60) was proposed as an upstream event that would foster disuse atrophy, as demonstrated by the NO-mediated increase of activity of the transcription factor FoxO3a and by the mild degree of muscle atrophy induced by hindlimb unloading in nNOS KO mice (60) and in 7-NI treated rats (60 and this article). At the subsarcolemma, nNOS participates in a multiprotein transmembrane apparatus composed by the dystrophin–glycoprotein complex (8), a linker between the extracellular matrix and the intracellular cytoskeleton. Sarcolemma directly connects to T-tubules and nNOS localization at the T-tubule and junctional SR level would be consistent with evidence showing transverse cross-striations after labeling with anti-nNOS antibodies or enzyme histochemistry for NADPH-d (50), decorated subsarcolemmal vesicles by immunoelectronmicroscopy (9, 68), localization of nNOS- and dystroglycan complex-interacting proteins like caveolin 3 in the SR (35), and co-immunoprecipitation with the SR calcium channel Ryanodine Receptor (RyR) 1 (52).
Our immunoprecipitation assays revealed Grp94 as a new putative partner of 160 kDa nNOS, which appeared recruited also in non-muscle tissues, like cerebellum. Such a finding suggests that Grp94–nNOS interaction might not be exclusive of the muscle-specific nNOS-μ isoform (57). The use of a cell-permeant chemical cross-linker to stabilize interactions among proteins further excluded the artifactual nature of this association and confirmed its occurrence in the microsomal fraction. Further, confocal microscopy revealed that both proteins co-localized, albeit partially, mainly at the A-I band boundaries, namely in regions containing T-tubules and junctional SR, and at the sarcolemma. Subcellular localization of nNOS is apparently driven by the binding to multiprotein complexes of different composition (75). Also the subcellular distribution of Grp94 varies: the protein translocates from ER to Golgi and plasmalemma in differentiating myogenic cells (14, 19), and in mammalian adult muscle it accumulates in the junctional SR, since anti-Grp94 antibodies decorated the Z-line in cardiac myocytes (64), and the I band in skeletal myofibers (this article). Proteolysis of intact microsomes suggested that the topology of the interaction between nNOS (a cytosolic protein) and Grp94 (a SR/ER lumenal one) might be explained by the presence of a minor proportion of Grp94 molecules showing a transmembrane configuration (14, 48 and this article). Alternatively, Grp94 and nNOS might participate to the same multimolecular complex without interacting directly between themselves. Although further studies will be required to identify additional partners, our results show that the expression of recombinant Grp94 was sufficient to hamper the loss of histochemically demonstrated nNOS activity from sarcolemma of unloaded muscle, leading to decreased FoxO-3a nuclear localization and myofiber ubiquitination. Conversely, no effect on subsarcolemmal nNOS localization was observed after the expression of an N-terminal deleted Grp94 protein containing a binding domain for integrins and Toll-like receptors (73). Although the Δ1-570 Grp94 could not be assayed for chaperone activity, since Grp94 is not required for folding of skeletal muscle integrins (5), it did not apparently bind nNOS, even after chemical cross-linking and despite the presence of some heterodimerization with endogenous Grp94.
Antioxidant effect of recombinant Grp94 expression
Dysregulation of nNOS activity is a recognized consequence of both decreased enzyme levels and sarcolemmal localization in several types of neuromuscular disorders (13, 17, 36, 60, 74). No apparent decrease in protein amount characterized our 7-day unloaded solei, whereas the increased NO availability to the sarcoplasm, indicated by increased cytosolic NADPH-d activity (70 and this article), would not only promote transcription of atrogenes (60), but also hypernitrosylate RyR (6, 17, 52), an event that would displace calstabin and lead to calcium leakage from the stores (6). Increased number and frequency of calcium sparks were described in the unloaded rat soleus (74), representing a putative trigger of mitochondrial ROS production (11), in addition to NO (33). Oxidative stress of unloaded myofibers was affected by the expression of recombinant Grp94, which reduced sevenfold the percentage of carbonylated myofibers and significantly decreased the presence of active sarcoplasmic nNOS.
In addition to stabilize nNOS at sarcolemma, recombinant Grp94 might exert antioxidant protection by independently contributing to the maintenance of calcium homeostasis. Overexpression of Grp94 in C2C12 cells reduced the amount of releasable calcium from the stores and prevented protein carbonylation after exposure to hydrogen peroxide (46).
Eventually, the proof of concept demonstration that Grp94 attenuation of disuse-induced myofiber atrophy and carbonylation were mediated by nNOS was obtained by investigating the effects of Grp94 overexpression in unloaded muscles lacking active nNOS, that is, in the presence of significant attenuation of atrophy (60 and this article) and percentage of carbonylated myofibers at ambulatory levels. Expression of recombinant Grp94 did not increase further CSA of unloaded 7-NI-treated solei, whereas it significantly reduced the percentage of carbonylated myofibers to levels comparable to those observed after transfection of both untreated unloaded and ambulatory solei. These results indicate that Grp94 might exert antioxidant cytoprotection through two distinct mechanisms. The first one concerns pathological levels of oxidative stress, like those generated during unloading, and appears mediated by nNOS. The second one concerns oxidative stress levels compatible with physiological muscle activity and its actual effectors remain to be determined.
In conclusion, the findings presented here identify Grp94 as a new partner of nNOS and a major regulator of its localization at the subsarcolemma of unloaded skeletal muscle fibers. Our results also show that changes in protein levels of this ER chaperone/stress protein would occur upstream nNOS untethering from sarcolemma (60). Muscle Grp94 protein levels appeared significantly reduced after few days of mechanical silencing (49). Although such an early decrease remains to be mechanistically investigated, the results presented here reveal the obligatory participation of Grp94 to a multiprotein complex, whose integrity is required to preserve nNOS subcellular localization and ameliorate both trophism and degree of carbonylation of unloaded muscle fibers.
Materials and Methods
Antibodies
The following antibodies were used in this study: (i) anti-Grp94 antibodies: rabbit polyclonal anti-Grp94 antibody (pAb) (SPA851) and rat anti-Grp94 mAb 9G10, (Stressgen, Victoria, Canada); mouse anti-Grp94 mAbs 3C4 (65) and 2G5 (67); (ii) anti-ER chaperones and proteins: rabbit anti-Grp78 (SPA826) and anti-calreticulin (SPA600), and mouse anti-Grp75 mAb (SPS825; Stressgen); goat anti-calnexin pAb (C20; Santa Cruz Biotech., Heidelberg, Germany); mouse anti-Csq mAb (Affinity BioReagents, Golden, CO); (iii) loading control: mouse anti-GAPDH mAb, clone 6C5 (Millipore, Vimodrone, Italy); (iv) anti-tag antibodies: mouse anti-VSV-G mAb, clone P5D4, conjugated with peroxidase (Sigma, Milan, Italy); rabbit anti-GFP pAb (A11122; Invitrogen, Groningen, The Netherlands); (v) antibodies for muscle proteins: mouse anti-α sarcoglycan mAb (Monosan, Uden, The Netherlands); anti-myosin heavy chain (My) mAbs (18, 61), clone BA-D5 for type 1-β My, clone SC-71 for type 2A My, and clone BF-G6 for ɛMy; (vi) anti-nNOS antibodies: rabbit anti-nNOS pAb (H-299; Santa Cruz Biotech.); mouse anti-nNOS mAb clone 16 (BD Transduction Lab., Milan, Italy); (vii) non-immune rabbit and mouse immunogloblulins (rbIg and mIg; Sigma); agarose-conjugated mIg or rbIg (Santa Cruz Biotech.); (viii) goat anti-IGF-I and anti-IGF-II pAb (R&D System, Inc., Lille, France); rabbit anti-FoxO3 (Sigma) and anti-ubiquitin (Dakocytomation, Milano, Italy) pAbs; (xi) secondary antibodies: goat anti-mIg conjugated with horseradish peroxidase or Texas Red and rabbit anti-goat Ig conjugated with horseradish peroxidase (Santa Cruz Biotech.); swine anti-rbIg conjugated with horseradish peroxidase (Dakocytomation); goat anti-rbIg conjugated with Alexa 568 and goat anti-mIg conjugated with Alexa 488 (Invitrogen); anti-rbIg and anti-mIg conjugated with agarose (eBioscience, San Diego, CA).
Plasmids
The following constructs were used: (i) pT94 (67), a bicistronic vector that contained the cDNA sequences of rabbit Grp94 (clone 2.52) and GFP; (ii) pT, the corresponding empty vector that contained GFP cDNA only (Invitrogen); (iii) pVSV-G5B and pVSV-G6B, which contained the SacII–BamHI fragment and the StyI–BamHI fragment of rabbit Grp94 cDNA, respectively, codifying for N-terminal deleted proteins (AA 272–782 and 571–782, respectively), inserted downstream the sequences codifying for the 21AA of the signal peptide (67) and for a doubled VSV-G tag framed by a PPGP peptide (AYTDIEMQRLGKL-PPGP-AYTDIEMQRLGKL); (iv) pBK-RSV, the corresponding empty vector (Stratagene, Agilent Techn., Cernusco sul Naviglio, Italy).
Animals and experimental protocols
A total of 94 six-weeks-old female Wistar rats of about 140–160 g were used for hindlimb unloading associated or not to gene transfer experiments. Number of animal used in each protocol was specified in the text and/or in the following paragraphs.
Hindlimb unloading
The experimental protocol was performed following the recommendations provided by the European Convention for the protection of Vertebrate Animals used for Experimental and Scientific purposes (Council of Europe number 123, Strasbourg, 1985) and authorized by the Animal Ethics Committee of the University of Padova and the Italian Health Ministry (103/2007B).
Animals were caged individually and were randomly assigned to either of two experimental groups (n=7 in each group): freely ambulating controls and 7-day hindlimb-unloaded animals. Hindlimb muscles were unloaded using the tail-suspension model (10), by wrapping the tail root with tape under general anesthesia induced with intraperitoneal administration of 20 mg/kg of Zolazepam chlorhydrate and tiletamine chlorhydrate (Zoletil; Virbac SRL, Milano, Italy). Each animal was weighted before and after the suspension period. Animals were euthanized after inducing anesthesia with isofluorane. Soleus muscles were excised, weighed, and frozen in liquid nitrogen. Cerebellum was also excised from control rats.
In vivo gene transfer
After inducing general anesthesia with isofluorane, soleus muscle was exposed and bilaterally injected with 50 μg of purified plasmid. After closing the wound, a train of six 20 ms electrical impulses at 209 V/cm, with 200 ms intervals, was given with a BTX Electro Square Porator (Harvard Apparatus, Quebec, Canada) using two 19 mm-squared plate electrodes placed upon the anterior and posterior sides of the hindlimb skin, as described by Serrano et al. (54). The day after, animals were either tail-suspended or let free to ambulate to form different groups of at least seven rats each: (i) freely ambulating animals transfected with the empty vector; (ii) freely ambulating animals transfected with either pT94 or pVSV-G6B or pVSV-G5B; (iii) hindlimb-unloaded animals transfected with the empty vector; (iv) hindlimb-unloaded animals transfected with either pT94 or pVSV-G6B or pVSV-G5B. After 7 days, animals were sacrificed, and soleus muscles processed as described above. Muscles containing less than 20 transfected, non-regenerating, myofibers were excluded from the study.
7-Nitroindazole
The nNOS-specific inhibitor 7-NI was daily administered by intraperitoneal injection at a dosage of 50 mg/kg to 10 rats following previously described protocols (60). Additional 10 rats were injected with vehicle only. The next day, the left soleus of each animal was electroporated with pT94, whereas the right one was left intact. On the third day after drug or vehicle administration, animals were randomly tail-suspended or let free to ambulate, for 7 days, to form four different groups of five rats each: (i) 7-NI treated freely ambulating animals; (ii) oil-treated freely ambulating animals; (iii) 7-NI treated hindlimb-unloaded animals; and (iv) oil-treated hindlimb-unloaded animals.
Oxyblot and Western blot procedures
Oxyblot assay was performed as previously described (10), using about 12 μg of muscle proteins for derivatization with DNPH (Oxyblot Oxidized Protein Detection Kit; Millipore). The final mix was separated by denaturing gel electrophoresis and proteins transferred to nitrocellulose membranes, which were first stained by Red Ponceau (RP), and then saturated and incubated with anti-DNPH antibody, following manufacturer's instructions. Blots were developed using a chemiluminescence detection system (BioRad Lab., Rome, Italy).
Routine western blot analysis was performed as previously described (61). About 50 μg of muscle homogenate, lysed with electrophoresis loading buffer (ELB), containing 3% sodium dodecyl sulphate (SDS) and 5% β-mercaptoethanol, and boiled for 5 min, was loaded on 10% linear SDS-polyacrylamide gels and electrophoretically separated proteins were electrotransferred onto nitrocellulose. Blots were stained first with RP, saturated, incubated with primary and secondary antibodies conjugated with peroxidase and developed as described above. Densitometry of the reactive polypeptides detected in Oxyblots and western blots was performed on scanned autoradiographic films using the National Institute of Health Image J software, and values were normalized using the corresponding densitometric value of either SA visualized by RP staining (10) or GAPDH immunolabeling.
Immunofluorescence and immunohistochemistry
Serial consecutive transverse cryosections from soleus muscle were assayed for indirect peroxidase immunohistochemistry and/or immunofluorescence, following previously described protocols (61). In brief, after a 10 min fixation at room temperature (RT) with 4% buffered paraformaldehyde (PFA), cryosections were incubated overnight at 4°C with the primary antibody, extensively rinsed with phosphate buffered saline (PBS), and incubated with the appropriate secondary antibody for 1 h at RT.
Incubation with anti-Grp94 antibody required a 10 min Triton-X100 permeabilization after fixation, as previously described (66).
Cold acetone was used to fix for 10 min at −20°C cryosections to be incubated with anti-My antibodies (61).
For indirect immunoperoxidase, fixation was followed by inhibition of endogenous peroxidase activity with methanol added with 0.3% H2O2 for 30 min at RT. Bound antibodies were then revealed by incubation with diaminobenzidine.
For control staining, sections were incubated with 1 μg/ml of non-immune mIg or rbIg as the primary antibody.
Consistency of the immunostaining was validated by independent analysis of adjacent cryosections. Analysis was performed using the Zeiss Microscope Axioplan equipped with fluorescence optics and 10×and 40×Plan-Neofluar objectives (Zeiss, Milan, Italy). Images were acquired using a Leica digital DFC 300FX camera and the IM50 software (Leica Microsystems SRL, Milan, Italy). Myofiber CSA was evaluated using Image J software. CSA value for each myofiber population (slow-fast fibers or transfected-untransfected ones) corresponded to the mean of the CSA values measured from 20 to 40 fibers of the same muscle (the experimental unit) and was expressed in μm2 (61).
DNPH immunohistochemistry
Cryosections were reacted with DNPH diluted 1:100 (Oxyblot Oxidized Protein Detection Kit) for 1 h at RT. After a 10 min rinse in 50 mM Tris pH7.6 added with 0.9% NaCl, sections were saturated with 3% bovine SA dissolved in the same buffer for 20 min, and incubated with the anti-DNPH antibody overnight at 4°C. After adequate rinses in PBS, sections were incubated for 2 h at RT with peroxidase-conjugated secondary antibodies and developed with diaminobenzidine.
Cell cultures and in vitro gene transfer
Cultures of proliferating and 6-h differentiating cells of the murine cell line C2C12 (ECACC) were performed in 10 cm Petri dishes, as previoulsy described (14). Chemical cross-linking was performed by exposing cells for 5 min at RT to 330 μg/ml dithiobis-succinimidylpropionate (DSP; Pierce; Euroclone, Pero, Italy) in PBS as previously described (14).
Transient transfection of C2C12 cells with pVSV-G 6B or the empty vector was performed following the procedure described in Pizzo et al. (46). About 10,000 cells/well were seeded in 18 wells of a 24-well plate and used 24 h after transfection. After extensive rinsing with PBS, cultures were cross-linked and processed for immunoprecipitation.
Immunoprecipitation
Samples from soleus muscle and cerebellum of ambulating control rats were lysed with 10 mM Tris-HCl pH 8.0 containing 150 mM NaCl, 1 mM ethylenediaminetetraaceticacid (EDTA), 50 mM NaF, and 1% NonIdet-P40, added before use with 1 mM phenylmethylsulfonyl fluoride (PMSF), 10 μM calpeptin, 1 μg/ml each of leupeptin, pepstatin, and aprotinin and supernatants collected after 30 min 10,000 g centrifugation at 4°C. Cross-linked cell cultures were lysed with 10 mM Na phosphate, 150 mM NaCl, 1% NP40, and 0.5% Na deoxycholate, pH 7.4 added with 1 mM EDTA (RIPA) and antiproteases and supernatants collected after 30 min 10,000 g centrifugation at 4°C. About 20 μg protein from cerebellum lysate, 200 μg protein from skeletal muscle, and 150 μg from cultures were precleared for 30 min at 4°C in orbital agitation with a mixture of agarose conjugated with either anti-mIg or anti-rbIg. Supernatants were then used for immunoprecipitation with 5 μg of primary antibody or non-immune rbIg or mIg conjugated with agarose. Samples were then incubated with 50 μl of the appropriate secondary antibody conjugated with agarose for 1 h at 4°C under agitation. After three rinses with 10 mM Na phosphate and 100 mM NaCl, pH 7.4 added with antiprotease cocktail, bound proteins were eluted with 35 μl ELB, boiled for 5 min, and further analyzed by 7.5% SDS-polyacrylamide gel electrophoresis and western blotting. A 6–12% gradient gel electrophoresis was used to separate samples of cultures transfected with the deleted Grp94 form.
Microsome fractionation, digestion, and cross-linking
Crude membranes were prepared from soleus muscles essentially as described by Margreth et al. (40). Briefly, dissected muscles were minced; homogenized in 10 mM HEPES pH 7.4, 20 mM KCl, 1 mM PMSF, and 10 μg/ml leupeptin; and centrifuged at 650 g×10 min at 4°C. Supernatant was then centrifuged at 10,000 g×10 min at 4°C to eliminate mitochondria and again at 120,000 g×90 min at 4°C to yield the total membrane fraction.
Pellet was resuspended with 10 mM Tris HCl pH 7.4, 0.25 M saccharose, and 1 mM EDTA for PK digestion assays, which were perfomed as previously described (14). For chemical cross-linking, microsomes were resuspended in 0.1 M Na phosphate pH 7.2, 3 mM MgCl2, and 0.1 mM EDTA at a concentration of 2.5 mg/ml. DSP was added to a final concentration of 0.5 mM and incubated for 30 min on ice. Reaction was stopped by adding Tris HCl pH 7.5 to a final concentration of 50 mM and lysing membranes with RIPA added with antiproteases.
Crude membranes were prepared from cross-linked C2C12 cell cultures, as described by Kang and Welch (28). After rinsing with PBS, cells were scraped and solubilized with Dounce in 10 mM Tris-HCl pH 7.4 added with anti-proteases. Lysates were equilibrated with sucrose to 0.25 M and centrifuged at 650 g×10 min at 4°C. Supernatant was centrifuged at 10,000 g×10 min at 4°C and then at 100,000 g×90 min at 4°C. Pellet was resuspended with RIPA added with antiproteases.
Confocal microscopy
Transverse and longitudinally oriented cryosections from soleus muscle were treated as described (64) and used either for single immunofluorescence with anti-Grp94 pAb or double immunofluorescence with anti-nNOS pAb and the anti-Grp94 mAb 2G5. After adequate rinses with PBS, sections were incubated for 2 h at RT with a mixture of appropriate goat secondary anti-Ig conjugated with Alexa 568 or 488. Slides were observed with a Leica SP5 confocal microscope equipped with Argon 488 nm and Helium 543 nm lasers (Leica Microsystem SRL). Acquisition was performed sequentially, collecting signals between 495 and 540 nm wavelenghts after excitation with Argon 488 laser, and between 584 and 630 nm for the Helium 543 one. Images were recorded at 1024×1024 pixel density using a Kalman four-collection average. A non-confocal bright field image was also collected from the same field. Images were analyzed using Photoshop software.
NADPH-d histochemistry
NADPH-d activity was demonstrated by histochemistry following previously described procedures (22, 50), with minor modifications. In brief, 12 μm-thick sections of frozen muscle were cut on a cryostat, collected on slides and fixed for 10–20 min with 2% PFA. After rinsing with PBS, sections were incubated in a medium containing 50 mM Tris-HCl pH 8.00, 0.2% Triton-X-100, 0.5 mM nitroblue tetrazolium chloride (Serva; Crescent Chemical Co., Islandia, NY), and 1 mM β-NADPH (Sigma) for 1 h at 37°C. Reaction was stopped by briefly rinsing with distilled water. Coverslips were mounted with glycerin jelly.
Statistical analyses
All data were expressed as mean±SE.
Statistical analysis between two groups was performed utilizing Student's t-test. Paired Student's t-test analysis was used when comparing average CSA or OD values between transfected and untransfected myofibers of the same muscles.
ANOVA or within-subject ANOVA, and Bonferroni test for post-hoc analysis were used for multiple comparison. In the case of within-subject ANOVA, average values of transfected and untransfected myofibers were compared with values obtained from transfected muscles obtained from animals of the same body weight (matched participants).
p value=0.05 was set as the limit for significance.
All these statistical analyses were performed using SigmaStat software, version 2.0 (Jandel Europe, Germany).
Footnotes
Acknowledgments
We thank R. Rostellato, M. Andreose, and B. Ravara for technical assistance. The financial support of Agenzia Spaziale Italiana (grant OSMA-WP1B51-2 to L.G.) is gratefully acknowledged.
Author Disclosure Statement
No competing financial interests exist.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
