Abstract
Prolyl hydroxylases are members of the iron- and 2-oxoglutarate–dependent dioxygenase enzyme family. Collagen prolyl hydroxylase is well known for its involvement in scurvy, in which ascorbate deficiency inhibits the enzyme and results in characteristic signs of the disease. Several distinct prolyl hydroxylases that hydroxylate (and thereby regulate) the hypoxia-inducible factor (HIF) transcription factors were discovered in 2001. These HIF prolyl hydroxylases, termed prolyl hydroxylase domain enzymes (PHDs), are the subject of this forum. HIF coordinates the cellular response to hypoxia, and the PHDs have attracted widespread interest as potential therapeutic targets in a wide range of diseases including anemia, ischemic heart disease, stroke, cancer, and pulmonary hypertension. Novel PHD-based pharmaceutical agents are now undergoing clinical trials. As well as original data, this forum includes reviews discussing recent advances in the biochemistry and therapeutic manipulation of PHDs, the potential role of PHD inhibitors in neuroprotection, and the involvement of PHDs in the complex interaction between oxygen homeostasis and iron homeostasis. Antioxid. Redox Signal. 12, 431–433.
HIF, Prolyl Hydroxylases, and Drug Discovery
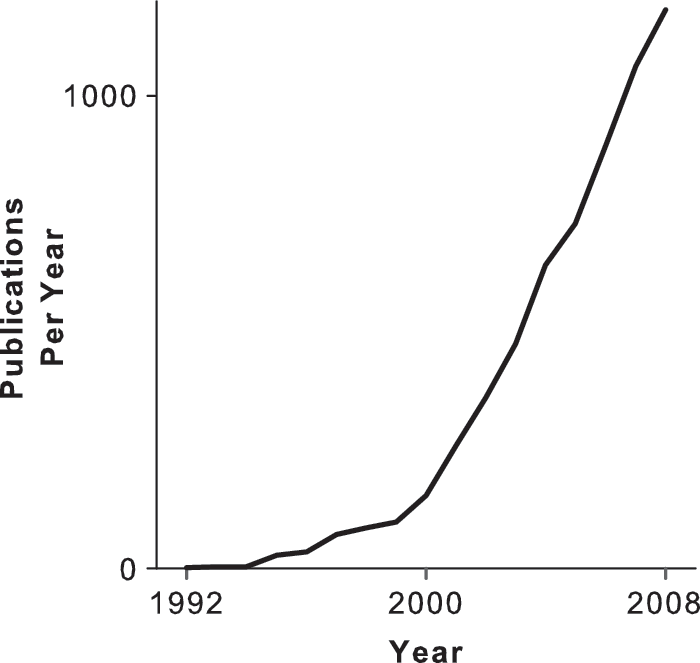
The importance of HIF in coordinating the cellular response to hypoxia is well established and is reflected by the accelerating growth in HIF-related scientific publications (Fig. 1). Oxygen-dependent PHDs negatively regulate HIF and, crucially, confer its oxygen sensitivity. Manipulation of responses to hypoxia is desirable in many disease states, and the PHDs are consequently an attractive therapeutic target. For example, inhibition of PHDs might upregulate beneficial HIF-dependent processes in anemia, ischemic heart disease, and stroke; conversely, controlled activation of these enzymes might usefully moderate HIF-dependent processes in the growth of solid organ tumors or in some forms of pulmonary hypertension.
Several pharmaceutical companies are believed to have an interest in PHD-based drug discovery (6). One of these companies, FibroGen, had been investigating collagen prolyl hydroxylase inhibitors as antifibrotic agents when the HIF-hydroxylating enzymes were discovered. FibroGen has since developed a number of PHD-inhibitors that are now undergoing clinical trials. Peer-reviewed data are not yet published, but the company reports promising preliminary phase I and II results in the areas of cytoprotection and renal anemia, respectively. We are not aware of any other clinical trials specifically targeting the PHDs to date, but preclinical work both in vitro and in vivo continues at pace. This forum includes a broad review of the field, which summarises the biochemistry of PHDs and the progress toward effective inhibitor and activator agents (8). Also included is a focused review of potential neuroprotective applications for PHD inhibitors (4), a particularly promising area, which is complemented by a comprehensive set of in vitro experiments that expand these potential neuroprotective applications to include conditions associated with mitochondrial dysfunction and metabolic stress, notably Huntington's disease (9).
Prolyl Hydroxylases as Iron Sensors
Although it has long been known that hypoxia and iron metabolism are somehow linked (hypoxia stimulates erythropoiesis, which requires iron), in recent years an ever more complex picture of interdependence between oxygen homeostasis and iron homeostasis has emerged, with the PHDs at its heart. Catalytic activity of PHDs is both oxygen dependent and iron dependent, and although they are better known as oxygen sensors, we increasingly think of the PHDs as cellular iron sensors. The multiple overlapping pathways now known to regulate cellular oxygen and iron metabolism, and the pivotal role played by PHDs, are the subject of a timely review in this forum (7). This growing understanding of integrated iron/oxygen homeostasis has recently converged with our own work in human hypoxia physiology. From studies in patients and volunteers, we now have evidence not only that HIF is involved in cardiopulmonary regulation (11, 12), but also that the interaction between iron and hypoxia extends beyond the cellular level to encompass systemic lung physiology (10). We and others (5) believe this interaction between iron, hypoxia, and pulmonary physiology is likely to be important in clinical practice, and this is supported by very recent work reporting that iron status modifies hypoxic pulmonary hypertension (13). Available evidence supports the hypothesis that PHDs are at least partially responsible for these pulmonary phenomena.
Future Directions
Early hopes of developing revolutionary PHD-based therapies have yet to translate into novel licensed pharmaceutical agents. One of the greatest challenges is the enormous complexity within the HIF signaling system, in which multiple PHD and HIF isoforms regulate the transcription of a large number of disparate genes while intersecting with multiple other signaling pathways. This presents abundant research opportunities but also complicates progress towards effective therapeutic agents, as illustrated in this forum by data demonstrating effects of PHD inhibitors that are apparently independent of HIF (9). This complexity also raises the issue of pharmacological and physiological selectivity, and whether an agent targeting the PHDs can be sufficiently selective to avoid nontherapeutic effects, while retaining the pleiotropy that distinguishes PHD inhibition or activation from therapeutic targeting of specific downstream HIF-regulated gene products.
Although the challenges are daunting, it is worth remembering how a disease as ancient and deadly as scurvy was overcome by the pursuit and application of scientific understanding. We are encouraged by the remarkable advances made in this field and remain enthusiastic about its future.
Footnotes
Author Disclosure Statement
No competing financial interests exist.
