Abstract
Glycerophospholipids represent a common class of lipids critically important for integrity of cellular membranes. Oxidation of esterified unsaturated fatty acids dramatically changes biological activities of phospholipids. Apart from impairment of their structural function, oxidation makes oxidized phospholipids (OxPLs) markers of “modified-self” type that are recognized by soluble and cell-associated receptors of innate immunity, including scavenger receptors, natural (germ line-encoded) antibodies, and C-reactive protein, thus directing removal of senescent and apoptotic cells or oxidized lipoproteins. In addition, OxPLs acquire novel biological activities not characteristic of their unoxidized precursors, including the ability to regulate innate and adaptive immune responses. Effects of OxPLs described in vitro and in vivo suggest their potential relevance in different pathologies, including atherosclerosis, acute inflammation, lung injury, and many other conditions. This review summarizes current knowledge on the mechanisms of formation, structures, and biological activities of OxPLs. Furthermore, potential applications of OxPLs as disease biomarkers, as well as experimental therapies targeting OxPLs, are described, providing a broad overview of an emerging class of lipid mediators. Antioxid. Redox Signal. 12, 1009–1059.
Accumulation and Potential Role of OxPLs in Pathology
OxPLs as Biomarkers of Disease and Targets for Therapy
I. Mechanisms of Phospholipid Oxidation
A. Oxidation of PLs: General mechanisms and biologically active products
Glycerophospholipids comprise an abundant class of lipids consisting of a glycerol backbone, phosphate-containing polar head group and two fatty acid residues. PL-bound polyunsaturated fatty acids (PUFAs) represent the major target for nonenzymatic or enzymatic oxidation that is not linked to the generation of metabolic energy. Although oxidative modifications of polar head groups have also been reported (316, 386), the biological importance of these reactions in insufficiently studied and will not be discussed here.
Oxidative fragmentation of a PL molecule generates several biologically active products, including small chemically reactive fragments of PUFAs, such as unesterified oxidized fatty acids (e.g., hydroperoxides and isoprostanes) and lyso-phospholipids. These products demonstrate multiple biological activities but will not be discussed here since the review specifically focuses on oxidation products having complete PL structure: glycerol backbone linked to polar head groups and two fatty acid residues, one or both of which are oxidized.
Available evidence suggests that nonenzymatic oxidation of PL-PUFAs proceeds according to the same basic mechanisms as oxidation of free (unesterified) PUFAs. This assumption is supported by identification of similar classes of molecular species generated by oxidation of free and PL-bound PUFAs that are described below. In contrast to the nonenzymatic oxidation, oxidation of PL-PUFAs by enzymes significantly differs from oxidation of unesterified PUFAs. While free PUFAs can be oxidized by multiple enzymes belonging to different protein families and introducing various oxidized groups, only one group of lipoxygenases (12/15 lipoxygenases) accepts PL-PUFAs as substrates producing PL-hydroperoxides (376). Further oxidation and rearrangements continue without participation of enzymes, and therefore oxidation initiated by enzymatic and nonenzymatic mechanisms produces many similar advanced PL oxidation products. Below follows a brief overview of pathways generating OxPLs. The description is focused on PUFA oxidation products that were shown to exist in PL-esterified form. Therefore, some original publications describing oxidation of unesterified PUFAs are not cited.
B. Initiation of oxidation
1. Nonenzymatic oxidation of PL-PUFAs
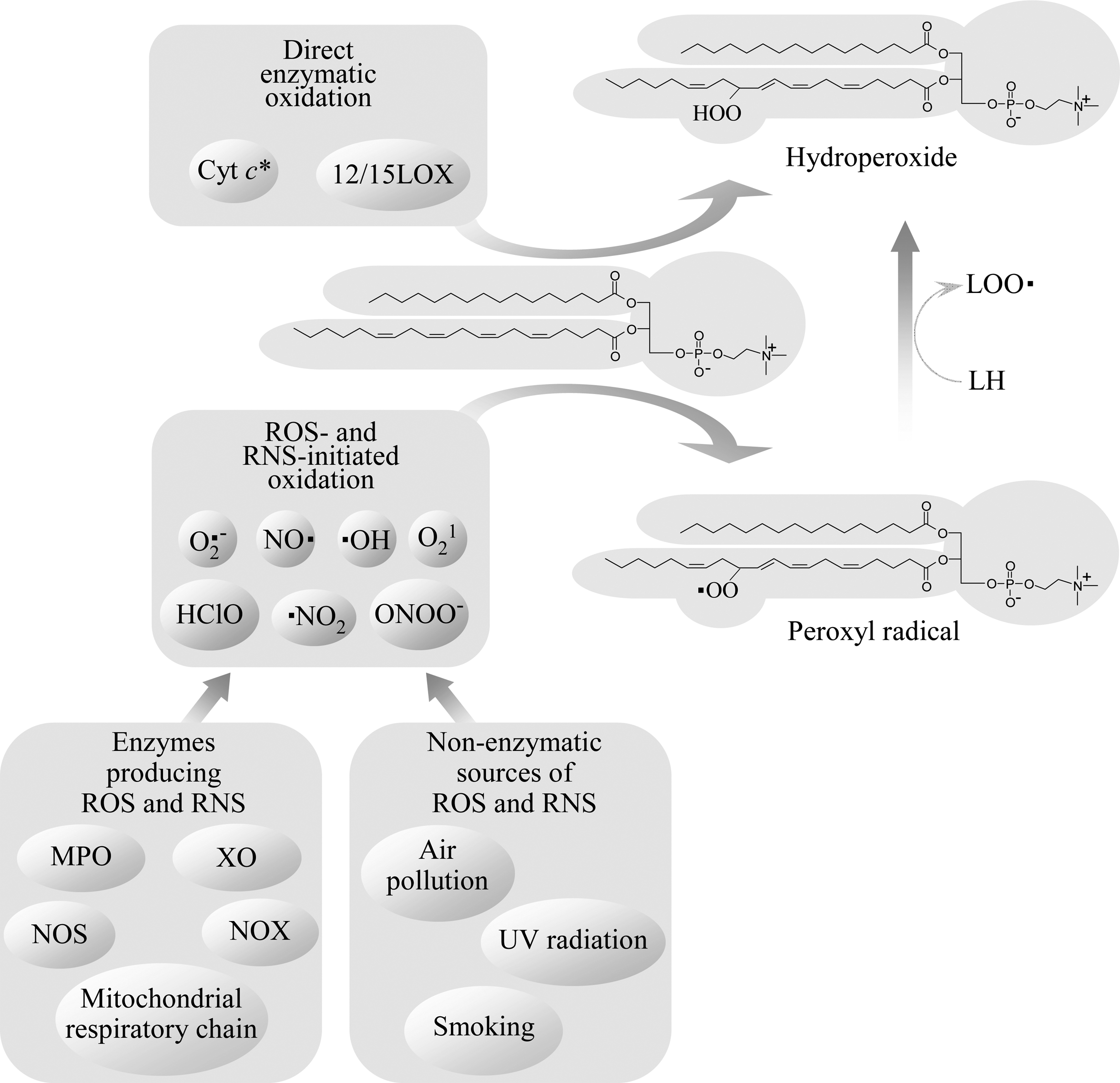
Nonenzymatic oxidation of PLs containing mono- and polyunsaturated fatty acids can be initiated by free radicals or nonradical reactive oxygen species (ROS) (Fig. 1). Free radical-mediated chain reaction initiated by formation of carbon-centered radicals and/or hydroperoxides of PUFAs (peroxidation of PUFAs) is one of the best understood and biologically relevant oxidative processes. The mechanisms described below were characterized for free PUFAs, but they are likely to be similar for PL-bound PUFAs.
PUFAs are more susceptible to oxidation as compared to saturated FAs due to the presence of methylene groups located between double bonds (bisallylic methylene groups) and as a result characterized by weakened hydrogen-carbon bonds. As a consequence, free radicals easily abstract hydrogen from bisallylic methylene leading to the formation of carbon-centered radicals within PUFAs. Carbon-centered radicals rapidly react with molecular oxygen, producing peroxyl radicals. This reaction represents the initiation step of lipid peroxidation. Peroxyl radicals react with bisallylic methylene groups in other PUFA molecules, leading to the transformation of peroxyl radicals to hydroperoxides and generation of new carbon-centered radicals. Thus, additional cycles of peroxidation are initiated. PUFA hydroperoxides in turn produce reactive alkoxyl and hydroxyl radicals via iron- or copper-catalyzed Fenton-like reactions, further propagating the chain reaction.
2. Sources of free radicals inducing oxidation of PLs
Oxidation of PUFAs can be induced by organic or inorganic free radicals. In addition to exogenous physical and chemical sources, such as air pollution, smoking, UV-light, or ionizing radiation, also multiple endogenous sources of free radicals exist. Several enzymes (i.e., NADPH oxidase, xanthine oxidase, uncoupled NO synthase) and electron transport system in mitochondria (Fig. 1) produce superoxide anion radical that interacts with other radical and nonradical oxidant species to produce several biologically important oxidant molecules, the most important of which are presented in Fig. 1. Myeloperoxidase produces highly reactive radicals (e.g., NO2 radical) from ROS characterized by lower reactivity, such as H2O2.
Enzymes inducing oxidation of PLs due to their ability to generate free radicals should be distinguished from enzymes directly oxidizing PUFAs (i.e., lipoxygenases and cytochrome c) (Fig. 1).
3. Oxidation of PUFAs by nonradical ROS
PL-hydroperoxides can be formed as a result of oxidation by unstable, energy-rich singlet molecular oxygen characterized by significantly higher reactivity as compared to ground state (triplet) oxygen (117). Singlet oxygen is generated by recombination of other ROS (224), or produced from ground-state triplet oxygen after irradiation with UV-light in the presence of endogenous photosensitizers such as all-trans retinal, NAD(P)H, FAD, etc. (12, 377). UV-light was shown to stimulate oxidation of PUFA-PLs in vitro and in vivo (125, 128, 331).
Ozone, which is produced in the atmosphere by photochemical reactions and also generated by industry, represents an important oxidizing air pollutant attacking epithelial cells and surfactant of lungs. Ozone readily reacts with double bonds in mono- and polyunsaturated fatty acids yielding fragmented species, such as 1-palmitoyl-2-(9-oxononanoyl)-PC that was found in lung surfactant extracts treated with ozone (349). Ozonolysis is used for generation of oxidatively fragmented saturated species of OxPLs (294).
4. Nitration and halogenation of PLs
In addition to peroxidation mechanism resulting in insertion of oxygen into PUFA residues, PLs can be oxidized by halogen- and nitrogen-containing compounds leading to the formation of PL-PUFAs containing Cl or Br atoms, or NO2 group.
NO2 is a highly reactive radical molecule produced by myeloperoxidase or by nonenzymatic reactions (e.g., breakdown of peroxinitrite) (274). NO2 can induce peroxidation of PUFAs (51); however, in addition to that, reaction of NO2 with mono- and polyunsaturated PL-esterified FAs can generate nitrated forms (246). The balance between oxidation and nitration of PUFA is determined by several factors including NO2 concentration and oxygen tension (103). Presence of esterified (including PL-esterified) nitrated FAs has been documented in normal human plasma and red blood cells (13). Biological activities of nitrated PLs were not studied.
MPO and eosinophil peroxidase produce HClO and HBrO, which are able to modify PLs via two types of reactions (i.e., PL peroxidation or halogenation). Recombination of HClO with oxygen superoxide anion, Fe2+, or hydrogen peroxide generates highly reactive hydroxyl radical or singlet oxygen, both of which are potent inducers of lipid peroxidation (205). Direct oxidation of PL-esterified FAs by HClO and HBrO results in insertion of halogenide and hydroxy groups into double bonds leading to formation of halohydrins (53, 158, 374). Alternatively, HClO reacts with amino groups of PLs to form N-chloramines of PE and PS (172). N-chloramines of PE readily break down with formation of N-centered radicals, which can further initiate PL peroxidation (172). Both peroxidation and chlorination of PLs were detected in HClO-treated lipoproteins and cells (158, 317). Chlorohydrins of PL-esterified oleic acid were shown to induce proinflammatory effects in vascular wall (79).
5. Enzymatic oxidation of PUFA-PLs
Lipoxygenases (LOXs) recognize 1,4-pentadiene motifs within unsaturated fatty acids and introduce molecular oxygen with high regio- and stereoselectivity producing hydroperoxides. LOXs are classified according to the position of oxygen insertion in the arachidonate substrate. The majority of LOXs (e.g., physiologically important 5-LOX enzymes) oxidize only unesterified PUFAs. Among known LOXs, only one group (12/15-LOX) is capable of oxidizing PL-esterified fatty acids. This class of enzymes is present in different biological species and includes mouse, rat, rabbit, bovine, and porcine leukocyte-type 12-LOX, rabbit and human reticulocyte-type 15-LOX, and soybean LOX (146, 376). These enzymes can oxidize PL-PUFA substrates in micellar form and in organized structures such as cellular membranes (183) or lipoprotein particles (16).
Available evidence suggests that 12/15-LOXs may play a role in pathological lipid oxidation. Oxidation of PLs by mammalian 15-LOX selectively generates (S)-isomers of esterified hydroperoxides. In contrast, nonenzymatic lipid oxidation produces a racemic mixture of (R)- and (S)-hydroperoxides. The analysis of 15-LOX products by chiral chromatography showed accumulation of (S)-isomers at early stages of atherosclerotic lesion formation, thus pointing to the important role of this enzyme as initiator of PL oxidation in vivo (184). However, in advanced lesions enantioselectivity of 15-LOX products was lost, suggesting that nonenzymatic mechanisms also play important role in oxidation of lipids in arterial wall. Which factors determine the balance between enzymatic and nonenzymatic oxidation of PLs at initiating and advanced stages of atherosclerosis and other pathologies, requires further investigation.
It has been suggested that cytochrome c (cyt c) may switch its activity from electron transport in mitochondria to peroxidase activity (166). The transformation begins from binding of cyt c to negatively charged cardiolipin (CL), leading to conformational changes with consequent release of PL-protein complex from mitochondria into cytosol. The complex of cyt c with CL upon activation by traces of PUFA-OOH or H2O2 acquires the ability to oxidize CL, PS, or PI, with formation of PL-OOH (164). CL-containing linoleic acid residues were preferentially oxidized by cyt c in vitro (149, 345) and in vivo (15, 347). Other PLs like PC or PE were not peroxidized by cyt c in spite of much higher unsaturation grade (164). The PL-peroxidation caused by cyt c may be essential for cell apoptosis (149, 346).
An alternative mechanism generating OxPLs is re-esterification of free oxidized PUFAs into lyso-PLs. Several types of OxPLs were shown to be formed via this mechanism in vitro and in vivo, including PLs containing monohydroxylated PUFAs (6, 27, 324), PUFA epoxides (86), esterified hepoxillins, and other oxidized residues (5). 5- and 12-LOXs and enzymes from 2C and 2J classes of cytochrome P-450 are examples of enzymes oxidizing free PUFAs whose products can be re-esterified into PLs (5, 42, 69, 169, 315). Further studies are required in order to clarify the impact of re-esterification as compared to other enzymatic and nonenzymatic pathways producing OxPLs under normal and pathological conditions.
C. Evolution of OxPLs
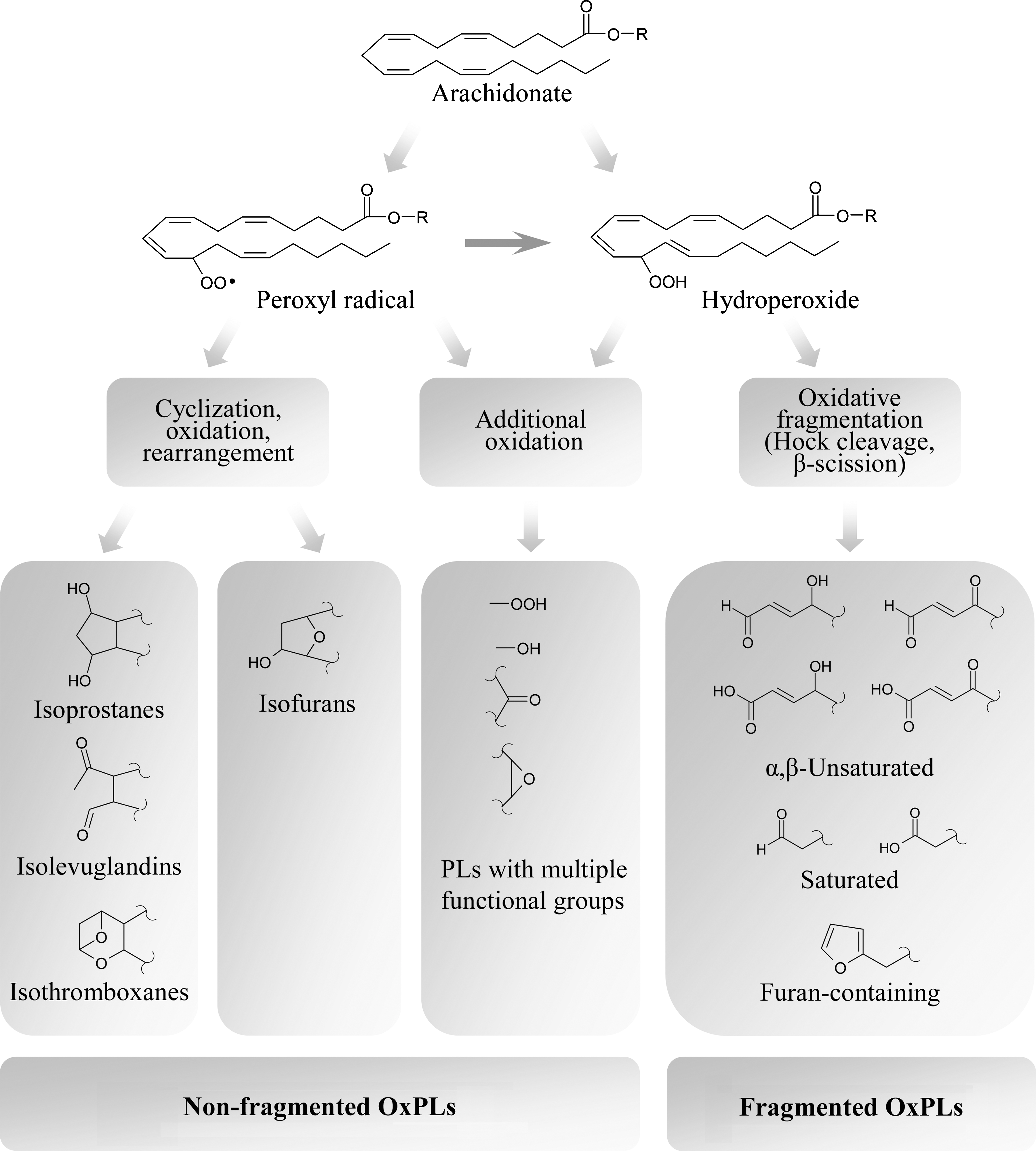
Oxidation of PUFAs can be initiated by a wide spectrum of reactions including enzymatic and nonenzymatic, free-radical, and radical-free processes. However, the vast majority of these reactions produce identical primary oxidation products (i.e., peroxyl radicals and hydroperoxides). Following the initiating step, subsequent oxidation of OxPLs is an enzyme-independent stochastic process producing a wide spectrum of OxPLs (Fig. 2).
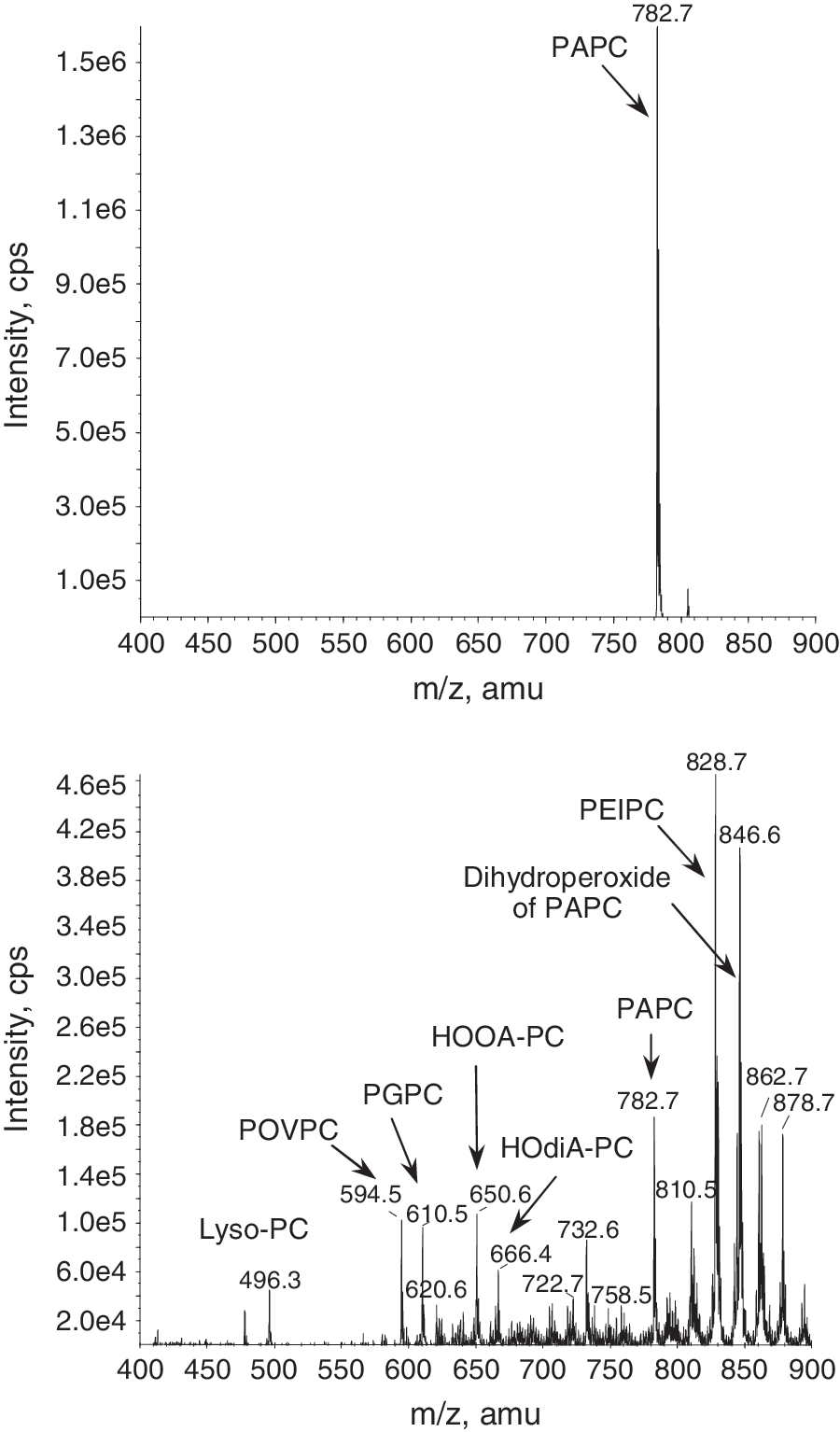
Generation of advanced peroxidation products proceeds according to several major mechanisms finally generating either full-length residues incorporating several oxygen atoms, or shortened fatty acid residues. These mechanisms include further oxidation of PUFA residue, cyclization of peroxyl radical, or oxidative fragmentation of esterified PUFAs (Figs. 3 and 4).
1. Formation of polyoxygenated PLs
Introduction of additional oxygen atoms into PUFAs is a common mechanism increasing complexity of OxPL mixtures. Since monohydroperoxides of PUFAs contain double bonds, further peroxidation is possible, yielding advanced oxidation products with variable combinations of hydroxy-, hydroperoxy-, keto-, and epoxy groups. Some of these complex OxPLs, including dihydroxy-PC and epoxy-monohydroxy-PC, were detected in vivo (81). Biological activities of polyoxygenated PLs are not characterized.
2. Cyclization of peroxyl radical/generation of nonfragmented OxPLs
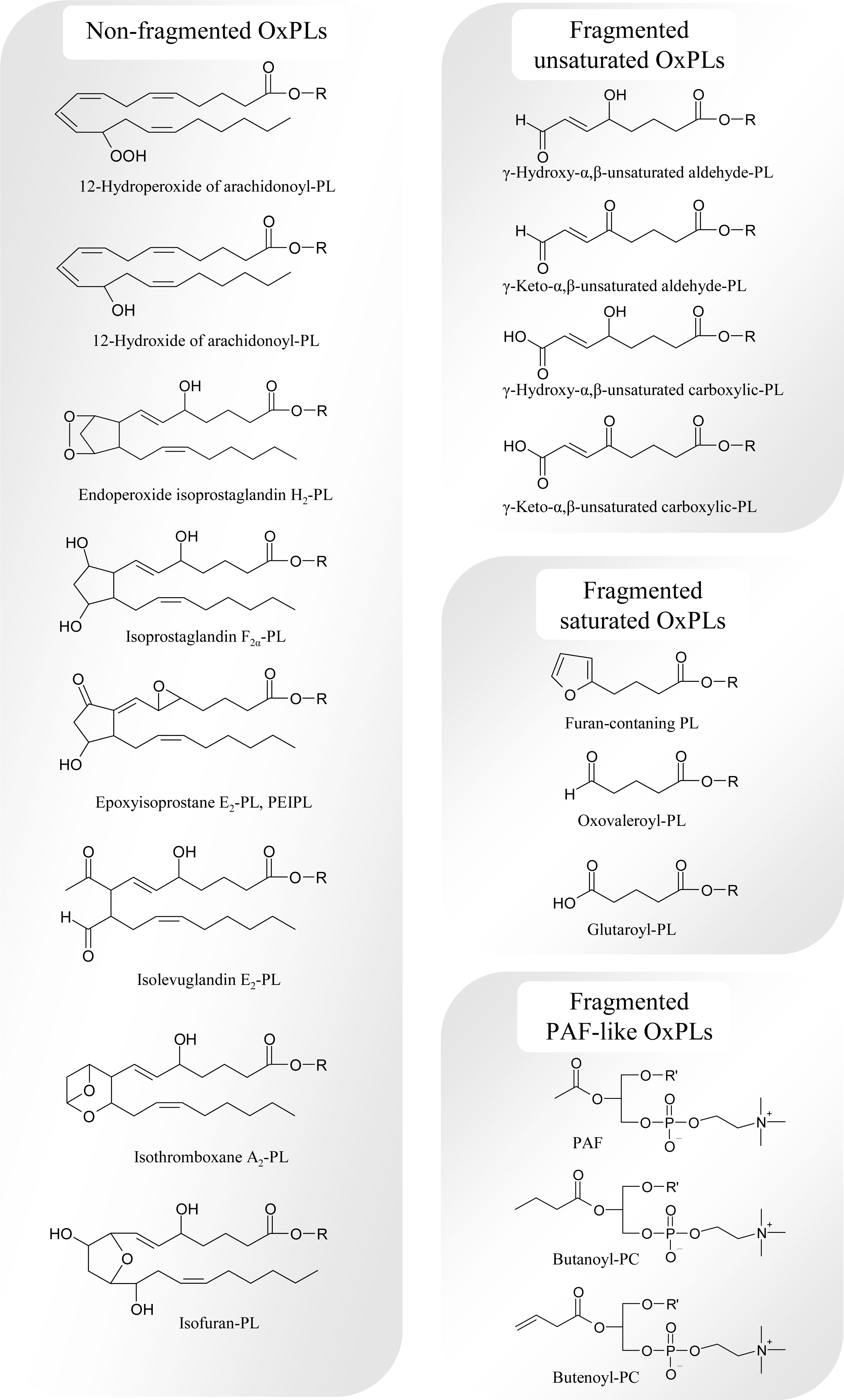
Cyclization of peroxyl radical produces cyclic peroxide, which undergoes either rearrangements yielding bicyclic endoperoxide, or oxidation introducing additional noncyclic or cyclic peroxide group. Cyclization of peroxyl radical is only possible for FAs having three or more double bonds (291). Bicyclic endoperoxide is a precursor of isoprostanes, isothromboxanes, and isolevuglandins, whereas rearrangement and further oxidation of molecules containing one cyclic and one noncyclic peroxide groups produces isofurans (282).
a. Esterified isoprostanes
The name “isoprostanes” originates from prostanoic acid, a 20-carbon carboxylic acid containing cyclopentane ring (also called prostane ring) within the aliphatic chain. Isoprostanes (isoPs) are prostaglandin-like compounds that differ from prostaglandins by stereo isomeric position of side chains relatively to cyclopentane ring, as well as stereo position of substituents within the prostanoic acid residue. The side chains in isoPs are located predominantly in cis- orientation in contrast to trans-position in prostaglandins (282). Moreover, naturally produced isoPs consist of several regio isomers, while prostaglandins are regio-specific. Prostaglandins are produced from unesterified arachidonic acid via enzymatic conversion by cyclooxygenases followed by PG-synthases (313). In contrast, isoPs are produced in vivo mainly from PL-esterified arachidonic acid via a free radical-induced oxidation (227). Bicyclic endoperoxide isoPG is formed as a random mixture of regio- and stereoisomers. As a result, rearrangement of bicyclic endoperoxides generates all possible stereoisomers of four regio isomers of isoPs, including stereospecific prostaglandins (104). Several classes of isoPs differing by type of substitutions and their location within the cyclopentane ring are formed, including F, E, and D series of isoPs (222). IsoPs of F-series are stable and therefore are widely used as markers of oxidative stress, both as PL-esterified and free forms (225), whereas isoPs E and D undergo spontaneous dehydration within the cyclopentane ring, yielding cyclopentenone-isoPs A and J, respectively (62). Formation of cyclopentenone-isoPs at least partially occurs in PL-esterified form since cyclopentenone-isoP-PLs were found in vivo (e.g., in liver and brain) (62, 231). Cyclopentenone-isoPs are highly reactive and readily form covalent complexes with reduced glutathione and cysteine residues of proteins (223).
An alternative way of transformation of bicyclic endoperoxide-containing PLs is formation of epoxyisoprostane-PLs such as PEIPC (Fig. 4) (370). Similarly to other isoPs, different regio- and stereo isomers of epoxyisoprostane-PC are formed (329). Dehydration of the cyclic isoprostane ring of PEIPC produces PECPC containing highly reactive cyclopentenone group (329). Elevated levels of PEIPC and PECPC were detected in ECs upon stimulation with inflammatory cytokines (329). Different positional isomers of PEIPC demonstrate similar ability to induce IL-8 expression and stimulate monocyte binding by ECs (329). In addition, epoxyisoprostane-PC demonstrates anti-inflammatory (360), angiogenic (40), and endothelial barrier-protective properties (28), and also induces antioxidant genes (125, 161).
Isoprostane-like compounds analogous to isoPs F, A, and J can be formed from PUFAs having three or more double bonds, including linolenic acid, eicosapentaenoic acid, adrenic acid, and docosahexaenoic acids (50, 154, 356, 383). PL-esterified forms of isoP-like compounds were detected in vivo in normal human samples (50, 280, 356, 383).
b. Esterified isothromboxanes
Apart from formation of (epoxy)isoprostanes, bicyclic endoperoxide spontaneously transforms into isothromboxane (isoTx) A2 (Fig. 4), which undergoes further transformation into isoTxB2 (283). PL-esterified isoTxs were found in rat liver at concentrations comparable with isoPs, and were strongly elevated during oxidative stress induced by CCl4 (228). Biological activities of PL-esterified isoTx A2 and B2 were not studied.
c. Esterified isolevuglandins
Isolevuglandins (also called isoketals, Fig. 4) represent an additional product of bicyclic endoperoxide rearrangement (292). Similarly to isoPs, isolevuglandins (isoLGs), are produced mainly in PL-esterfied form (290). Due to high reactivity of aldehyde groups, isoLG-PLs rapidly form covalent complexes with proteins. Following phospholipase cleavage, isoLG-protein adducts are formed that were found in oxidized LDL and inflamed tissues, and due to their long half-life in circulation were suggested as integral markers of oxidative stress (267, 293).
d. Esterified isofurans
Isofurans (Fig. 4) are PUFA oxidation products with a substituted tetrahydrofuran ring within the aliphatic chain (94). Isofurans can be formed either from cyclic peroxide or from monohydroperoxide according to different mechanisms (281). Formation of isofurans from cyclic peroxides is preferred over isoPs at enhanced oxygen tension (281). Isofuran-PLs are stable oxidation products and therefore can be used as conventional biomarkers of lipid oxidation. Isofurans formed from docosahexaenoic acid are called neurofurans (314). Elevated levels of PL-isofurans were found in hyperoxia-injured lungs (94), brain tissue from patients with Parkinson disease (93), and in brain samples of mouse model of Alzheimer disease (314).
3. Oxidative cleavage/formation of fragmented OxPL species
Fragmentation of hydroperoxides represents third important pathway whereby peroxides/peroxyls are transformed into advanced oxidation products.
γ-Hydroxy (or oxo) α,β-unsaturated PLs with terminal aldehyde groups (Fig. 4) are produced from hydroperoxides via oxidation/fragmentation or polymerization/cleavage. Oxidative fragmentation of hydroperoxides occurs via several mechanisms including β-scission, Hock rearrangement, or cyclization of alkoxy radical produced from hydroperoxide (128). On the other hand, peroxyl radical can cross-react with double bonds present in hydroperoxides yielding peroxydimers; these are unstable products and spontaneously break down forming either new radicals or α,β-unsaturated aldehydes (297).
a. Oxidatively truncated unsaturated OxPLs
γ-Hydroxy (or oxo)-α,β-unsaturated OxPLs containing terminal aldehyde (also called core aldehyde) or carboxyl groups (Fig. 4) demonstrate several biological activities, including interaction with scavenger receptor CD36 (264), induction of proinflammatory effects in ECs (327), and inhibition of toll-like receptor 4 (TLR4) (327). These OxPLs were found in vivo, and their levels are known to be elevated in plasma of hyperlipidemic mice, atherosclerotic plaques of mice and rabbits, and human damaged retina (263, 264, 331).
γ-Hydroxy (or oxo)-α,β-unsaturated aldehyde PLs are highly reactive compounds, able to covalently link to amino groups of proteins, as well as thiol groups of biomolecules (137). Alternatively, they can be further oxidized either to carboxylic α,β-unsaturated fragmented PLs or to saturated fragmented species.
OxPLs with terminal furan groups are produced from γ-hydroxy-α,β-unsaturated aldehyde PLs via spontaneous intramolecular reaction between aldehyde and hydroxyl groups (105) (Fig. 4). These are stable oxidation products generated in physiological oxidant systems in vitro and in vivo (105). Due to the loss of aldehyde groups, these compounds cannot react with proteins and are not toxic. In contrast to the parent γ-hydroxy-α,β-unsaturated aldehyde PLs, terminal-furan-PLs are not recognized by CD36 (105).
b. Oxidatively truncated saturated OxPLs
In addition to the products described above, oxidative fragmentation of PUFA-PLs also directly produces saturated fragmented species containing terminal carbonyl groups. Most common species are oxononanoate and azelaoate formed from linoleic acid, oxovaleroate, and glutaroate generated from arachidonic acid (Fig. 4), or oxobutyrate and succinate produced from docosahexaenoic acid (126, 265). In addition to direct formation from hydroperoxides, saturated fragmented OxPLs can be formed by further oxidation of γ-hydroxy (or oxo)-α, β-unsaturated PLs (265). Due to the lack of double bonds, saturated fragmented OxPLs are resistant to further oxidation. The absence of substituents and double bonds within fragmented chains results in diminished reactivity of aldehyde-containing saturated OxPLs as compared to α,β-unsaturated fragmented OxPLs. Thus, saturated fragmented OxPLs are chemically stable end products found inter alia in plasma and vessels of hypercholesterolemic patients and apoe −/− mice (263, 264, 368). These compounds demonstrate a variety of activities in vitro, including proinflammatory (368), angiogenic (40), anti-LPS effects (360), induction of antioxidant genes (161), and modulation of adaptive immune reactions (302).
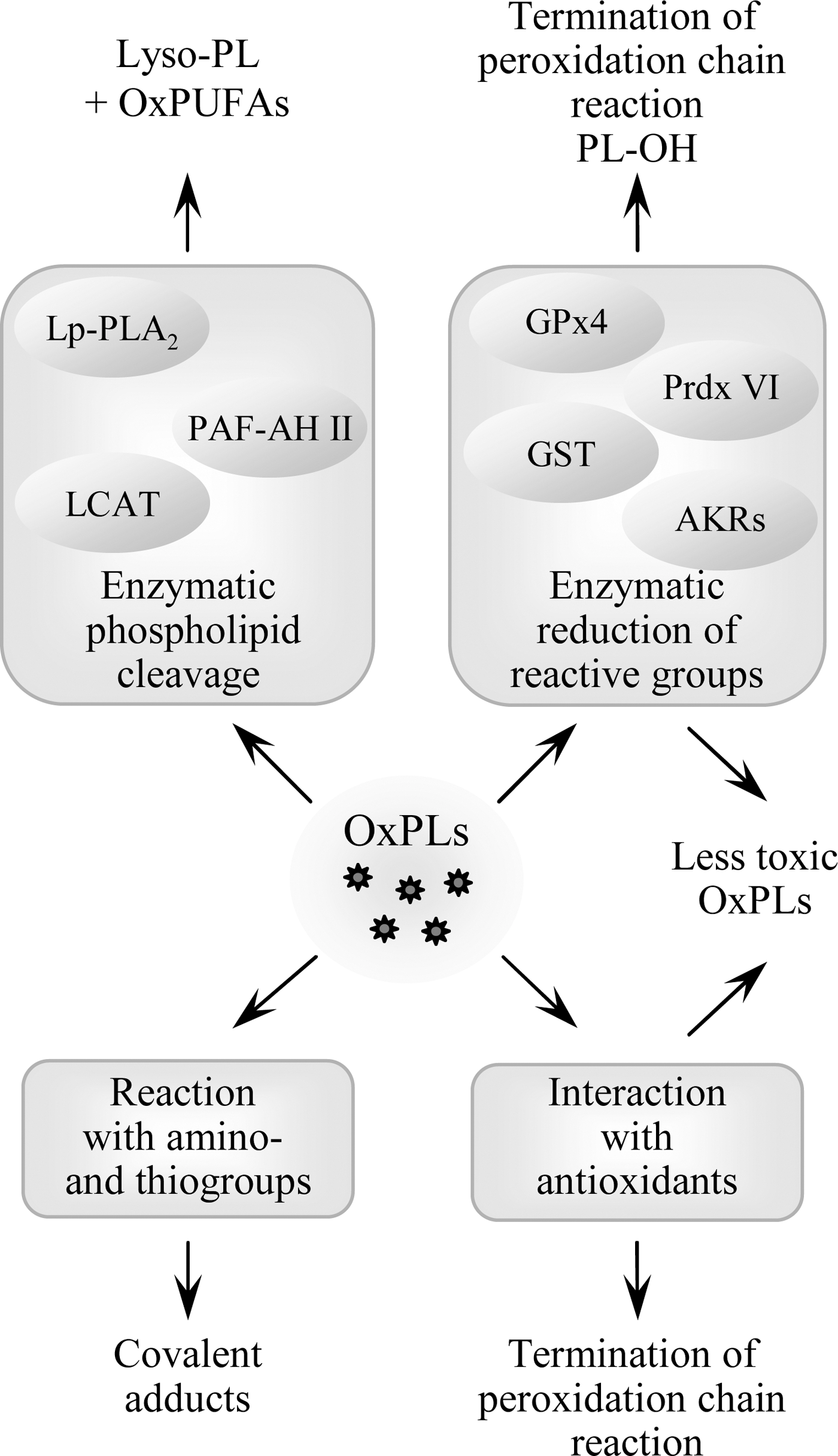
D. Termination of PL-oxidation and detoxification of reactive OxPLs
Several processes contribute to the elimination and detoxification of OxPLs, including the mechanisms terminating peroxidation chain reaction and inactivating toxic chemically reactive groups produced by oxidation (Fig. 5). While the reactions of PL-esterified carbon-centered and peroxyl radicals with low molecular weight antioxidants are likely to be similar to those of free fatty acids, enzymatic pathways are different since only a limited number of enzymes accept OxPLs as substrates. In addition, enzymatic cleavage of OxPLs generating lyso-PLs and free oxidized fatty acids represents another level of regulation of activity and biodistribution of OxPLs.
1. Enzymatic reduction of hydroperoxides
As compared to hydroperoxides, hydroxides are characterized by significantly lower chemical reactivity and therefore are considered to be stable and relatively non-toxic oxidation products (319). The most important type of enzyme catalyzing reduction of hydroperoxides to hydroxides is glutathione peroxidase (GPx). Lipid hydroperoxides are reduced in a reaction involving selenocysteine residue of GPx and glutathione. As a result, lipid hydroxide and oxidized glutathione are generated. Several species of GPx are known that are characterized by different tissue, cell and organelle distribution as well as variable substrate specificity (68). Among GPx enzymes, PL glutathione peroxidase (GPx4) has the highest activity in respect to membrane-bound hydroperoxides of PL-esterified PUFAs (295). GPx4 is expressed in many cell types where it is located in cytosol, mitochondria, and nucleus (295). This enzyme has been shown to suppress peroxidation of PLs induced by lipoxygenases (298) or UVA light (372).
Other enzymes such as peroxiredoxin VI (208) and glutathione transferase (379) can also reduce PL-hydroperoxides to corresponding hydroxides. In analogy to GPx, peroxiredoxin VI and glutathione transferase also use glutathione to recover their catalytic cysteines.
2. Reduction of carbonyls in OxPLs by aldo-keto reductases
Upon oxidation of OxPLs, a variety of products containing aldehyde and keto groups are formed. A class of enzymes called aldo-keto reductases transforms these functional groups to respective hydroxyl groups. Apart from their physiological role in metabolism of sugar aldehydes, aldo-keto reductases play a role in detoxification of toxic (phospho)lipid aldehydes (160). Aldo-keto reductases of the families AKR1A and B recognize as substrate aldehyde-containing OxPLs including diacyl-phosphatidylcholines, -ethanolamines, -glycerols, phosphatidic acids, as well as alkyl-acyl PCs (318, 320).
3. OxPL cleavage
Platelet activating factor acetylhydrolase (PAF-AH), also known as lipoprotein-associated phospholipase A2 (Lp-PLA2), is well recognized for its ability to cleave and thus inactivate PAF (217). This enzyme is very selective in respect to short sn-2 residues and does not cleave unmodified long-chain residues. However, upon oxidation of residues, this selectivity is lost and OxPLs become substrates for Lp-PLA2 even when they have nonfragmented oxidized sn-2 residues. The enzyme was shown to hydrolyze fragmented saturated OxPLs (326), as well as long-chain OxPLs, including esterified F2-isoprostanes, PC-hydroperoxides and PEIPC (73, 181, 321). In addition to circulating Lp-PLA2, also intracellular PAF-AH type II is known to hydrolyze fragmented OxPL species (133), and, with lower efficiency, isoP-PCs (321). Mice genetically deficient in PAF-AH type II demonstrated enhanced accumulation of esterified isoPs in response to CCl4-induced oxidative stress, and were more prone to oxidative injury (180). These data show that PAF-AH type II enzyme is important for degradation of PL-esterified isoPs and protection from oxidative stress in vivo. In addition to Lp-PLA2 and PAF-AH type II, also other isoforms of PLA2 can cleave OxPLs. Biolological importance of these reactions requires further investigation (241). Furthermore, it was hypothesized that paraoxonase-1 (PON-1), an HDL-associated organophosphonate triesterase, can cleave oxidized residues in OxPLs; however, more detailed study suggested that PON-1 has no PLA2 activity and that the phospholipase activity observed in previous experiments was explained by contamination of PON-1 preparations with Lp-PLA2 (212).
Lecithin:cholesterol acyltransferase (LCAT) catalyzes formation of cholesterol esters from free cholesterol present in lipoproteins using PCs and PEs as donors of acyl chains. As a result of the reaction catalyzed by LCAT, lyso-PLs and cholesterol esters are formed. The enzyme uses as substrates not only native but also oxidized PLs (233). In addition, LCAT was shown to hydrolyze OxPCs generating free oxidized residues rather than cholesterol esters (121). In contrast to PAF-AH, LCAT has preference for long-chain oxidation products as compared to OxPLs containing truncated residues (330). LCAT was shown to prevent accumulation of OxPLs and inhibit oxidation of LDL and PL micelles (358).
E. Formation of adducts
Formation of covalent adducts of OxPLs with proteins represents an additional pathway of their inactivation but at the same time can damage sensitive proteins. OxPLs containing aldehyde or electrophilic α,β-unsaturated carbonyl groups react with nucleophilic groups such as thiol and amino groups (for example, cysteinyl and lysyl residues of proteins) forming covalent Michael adducts or Schiff bases (126, 334) (Fig. 6). Spontaneous secondary reactions of Schiff bases produce pyrroles, lactams, and hydroxylactams that are stable protein–lipid adducts, which can be detected in vivo (392).
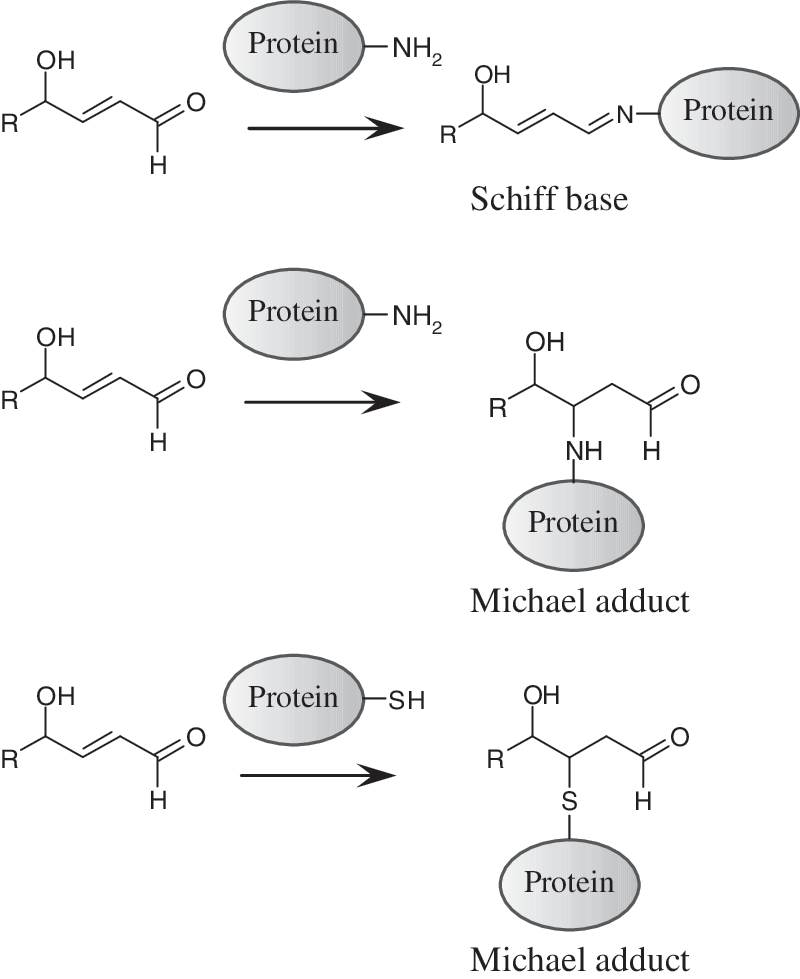
Formation of covalent adducts with OxPLs can inactivate important amino acid residues or induce polymerization of proteins resulting in protein dysfunction. Formation of PL–isolevuglandin–protein adducts was shown to impair function of cardiac K+ channel (47). Furthermore, γ-hydroxy-α,β-unsaturated aldehyde-PCs react with cysteinyl residues of cathepsin B, forming Michael adducts and as a consequence reducing its proteolytic activity (137).
It is known that formation of covalent adducts with oxidized lipids plays a key role in activation of redox-sensitive transcription factor NRF2. Covalent reaction of cyclopentenone isoPs to specific cysteines within NRF2 partner protein KEAP-1 allows NRF2 to escape degradation and translocate to the nucleus, and thus promotes transcription of NRF2 target genes (195). For PL-esterified isoPs such covalent reaction has not yet been shown. However, Gugiu et al. showed that OxPLs can penetrate into the cell and covalently modify intracellular proteins (129). These data raise a possibility that OxPLs can directly regulate activity of redox-sensitive transcription factors inside the cell.
Many protein–OxPL adducts are stable and can serve as biomarkers of oxidative stress. The majority of circulating OxPCs, which can be recognized by specific E06 antibodies, is present in plasma in association with Lp(a). More than 85% of E06 immunoreactivity in human plasma co-immunoprecipitated with apo(a) (17). The linkage between OxPLs and Lp(a) is ∼50% covalent and ∼50% noncovalent (extractable lipids) (17). Two specific lysines of the kringle V domain of apo(a) were identified to be covalently modified by OxPCs (82). Moreover, Lp(a) but not LDL is modified by isolevuglandins (291). Thus, Lp(a) may serve as a major carrier of OxPLs in circulation (343). It has been hypothesized that Lp(a)-bound OxPLs retain their proinflammatory properties (82) and may represent one of factors underlying the well-known association of Lp(a) levels with atherosclerosis (343).
II. (Patho)physiological Effects of OxPLs
A. OxPLs as markers of “modified self”
Peroxidation of PLs generates a wide spectrum of reactive molecules that can modify endogenous structures, including proteins and other lipids. This leads to the generation of altered lipids as well as oxidized lipid–protein adducts, yielding “modified-self molecules” or so-called “neo-self determinants” that are recognized by specific innate and adaptive immune responses (Fig. 7).
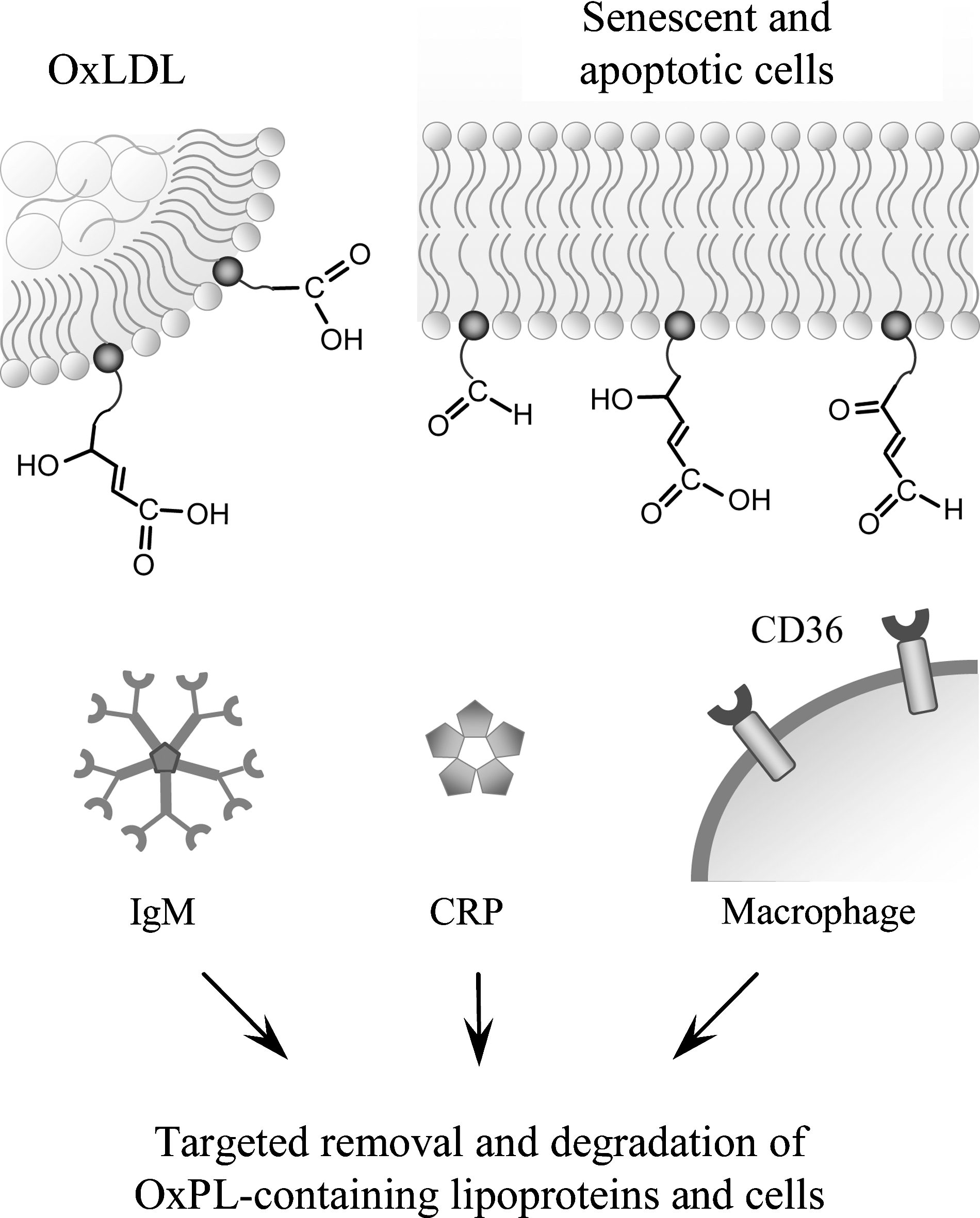
1. OxPLs as antigens
It is well known that subtle modifications of autologous proteins can render these immunogenic (322). Similarly, modifications with products of PL oxidation can lead to the generation of immunogenic molecules. In general, these antigenic adducts are recognized in a hapten-specific manner, in which neo-epitopes are formed on different endogenous proteins and lipids (e.g., PLs like PE containing a free amino group). For example, when the abundant PL phosphatidylcholine (PC) present in LDL and cell membranes is oxidized, decomposition of the unsaturated sn-2 fatty acid generates a wide spectrum of reactive molecules, such as malondialdehyde (MDA) and 4-hydroxynonenal (84), as well as the “core aldehyde” of the residual OxPL backbone such as POVPC (20). All these products are highly reactive and can form neo-epitopes. Importantly, apoptotic cells have been found to display an enriched content of OxPLs in their membranes, which renders them pro-inflammatory and immunogenic (56).
Both IgG and IgM antibodies to OxLDL have been documented in animal models of atherosclerosis and in humans (25, 140), and some of these antibodies recognize specifically OxPL epitopes that are present in OxLDL. While many studies show direct relationship of anti-OxLDL antibody titers with surrogate markers of disease or clinical endpoints, others fail to do so (152, 339). In recent years, evidence has accumulated demonstrating a different role for IgG and IgM antibodies against OxLDL. These studies suggest that titers of IgM but not IgG anti-OxLDL antibodies show an inverse relationship with atherosclerosis (170, 339). Of particular interest is the finding that some antiphospholipid antibodies (aPLs) of patients with antiphospholipid syndrome (APS) recognize oxidation-specific epitopes in OxLDL (143, 350). The APS is characterized by the presence of circulating aPLs and clinical features such as venous or arterial thrombosis, fetal loss, and autoimmune thrombocytopenia. Horkko et al. showed that many aPLs bound to CL only after it had been oxidized (OxCL), but not to a reduced CL analogue that could not undergo oxidation (142). Importantly, cardiolipin is also present in LDL particles (74). Horkko et al. further identified the neoepitopes of some aPLs as adducts formed between OxPLs and associated proteins, such as β2 glycoprotein 1 (β2GP1) or apoB (142). Thus, many aPLs seem to be directed at neo-epitopes of OxPLs. Antibodies against OxCL are clearly associated with atherosclerosis. In a subsequent study, it was shown that the levels of autoantibodies to OxCL correlated with the levels of aortic isoP F2α-VI and with the severity of atherosclerosis in apoe −/−mice, indicating that antibodies to OxCL reflect lipid peroxidation in vivo (270). The importance of these antibodies is further demonstrated by the fact that aPLs in patients with systemic lupus erythematosus were found to be primarily directed against OxCL and that the same patients exhibited increased levels of apoB-associated OxPLs as determined by the mAb E06 (99). Thus, antibodies to OxPLs are found in autoimmune diseases that predispose to cardiovascular disease and are associated with increased oxidative stress. This is further underscored by the characterization of a natural IgM Ab derived from the spleen of atherosclerotic ldlr −/−mice. This IgM was selected for binding to OxCL and binds to OxLDL, apoptotic cells, and immunostains atherosclerotic lesions (344).
Clearly, multiple OxPL epitopes exist, but the best described one is the phosphocholine group of oxidized phosphatidylcholine (OxPC). Indeed, phosphocholine-containing OxPLs have been identified as important antigens, in which the phosphocholine headgroup of oxidized but not of native PC is the epitope recognized by specific antibodies. A detailed understanding about the importance of phosphocholine evolved out of the detailed characterization of the prototypic monoclonal anti-OxLDL IgM Ab E06 that was derived from a panel of B-cell hybridomas from the spleens of atherosclerotic apoe −/−mice (251). E06, which was originally selected for the binding to copper-oxidized LDL, was subsequently found to specifically bind to OxPLs containing the phosphocholine headgroup but not to native unoxidized PCs (141). Moreover, in studies using synthetic OxPLs it was found that the specific OxPC epitope for E06 was the phosphocholine headgroup (98). Although the phosphocholine headgroup is present in native unoxidized PCs, only upon oxidation it becomes available for immune recognition by E06. Thus, it is hypothesized that structural changes as a result of PL oxidation lead to the generation of this neo-epitope. By the sequencing of the variable region of E06, Shaw et al. discovered that E06 is in fact a germline-encoded natural antibody identical to the previously characterized antibody T15 (303), which is known to bind the phosphocholine present in the capsular polysaccharide of S. pneumonia and many other microbes, and to protect mice from pneumococcal infections (48). Thus, molecular mimicry exists between the phosphocholine headgroup of OxPCs and the phosphocholine found in pneumococci and other microbes. E06 binds both the lipid and the protein moiety of OxLDL (containing free and protein-conjugated OxPCs) and inhibits its uptake by macrophages (141) as well as the uptake of apoptotic cells (55), suggesting that the same OxPL moiety is important for the recognition by macrophage scavenger receptors. Thus, phosphocholine is an important target of innate immunity recognized by a whole array of innate immune receptors (22).
2. OxPLs as ligands for scavenger receptors
Apart from serving as antigens for natural antibodies, OxPLs may also function as ligands for scavenger receptors. Scavenger receptors expressed on macrophages recognize both free OxPLs present in OxLDL, as well as OxPLs covalently linked to apoB-100 (26). Interactions of OxPLs with CD36, a member of class B scavenger receptor family (89), are by far the best characterized. Macrophage CD36 is involved in recognition and engulfment of apoptotic cells, and it also mediates the uptake of OxLDL and foam cell formation (89). The necessity of CD36 for the macrophage uptake of OxLDL and atherogenesis has also been demonstrated in vivo (90, 275), although a conflicting report exists (226). Podrez et al. have studied extensively the molecular characteristics of OxPLs interacting with CD36, and have demonstrated that a structurally conserved family of OxPLs with a truncated sn-2 acyl group incorporating a terminal γ-hydroxy(or oxo)-α,β-unsaturated carbonyl serves as high affinity ligand for CD36, and that these species are enriched in atherosclerotic lesions (264, 265). These oxidatively modified side chains protrude from lipid bilayers and lipoprotein particles to the aqueous compartment, thereby enabling their recognition by CD36 in neighboring cells (122). Recently, the structural basis of OxPC binding to CD36 has been studied in detail and amino acids 160–168 were identified as the core of the OxPC binding site, with two positively charged lysine residues 164 and 166 interacting with sn-2 acyl chains of OxPLs, thereby being indispensable for the binding (168). In addition to PC, also oxidized PS and phosphatidic acid were characterized as ligands for CD36 (122, 123) additionally suggesting that oxidized sn-2 residue but not polar head group is the major structural motif recognized by CD36. However, Boullier et al. presented conflicting data (46). They showed that binding of OxLDL to CD36 was inhibited by POVPC covalently bound via sn-2 group to a short peptide. This adduct does not contain γ-hydroxy(or oxo)-α,βunsaturated carbonyl motif and therefore is likely to interact with CD36 via different mechanisms possibly involving recognition of phosphocholine group (46).
In addition to CD36, OxPLs are recognized by another member of the class B scavenger receptors, SR-BI, and were hypothesized to serve as ligands for SR-BI-mediated internalization of OxLDL (116). Furthermore, OxPC species that are ligands for CD36 were shown to inhibit SR-BI-dependent HDL binding and reverse cholesterol transport (10). Thus, OxPLs may promote atherosclerosis not only by enhancing the uptake of modified lipoproteins by macrophages, but also by interfering with the reverse cholesterol transport by preventing SR-BI-mediated cholesterol uptake in hepatocytes.
3. OxPLs as ligands for CRP
The notion that OxPLs constitute a class of pathogen-associated molecular patterns (PAMPs) that are recognized by innate immune receptors is further supported by the finding that the ancient innate defense molecule C-reactive protein (CRP) binds OxPLs (57). CRP is an acute-phase protein that specifically binds to phosphocholine of the capsular polysaccharide of many microbes (359). Elevated levels of CRP are an independent risk factor for cardiovascular disease (199). In analogy to E06/T15 IgM, CRP binds to OxLDL and oxidized PC in a Ca2+-dependent manner, but does not bind to native LDL nor to unoxidized PC (57). CRP binding is mediated through the recognition of the phosphocholine moiety, as the binding to OxLDL was competed by KLH conjugated with phosphocholine and vice versa. Moreover, binding of CRP to phosphocholine-KLH could be competed by OxPC but not native unoxidized PC. Consistent with the presence of OxPLs in the membranes of apoptotic cells, CRP was also found to bind apoptotic cells via the phosphocholine moiety of OxPC. These data suggest that recognition of OxPCs in apoptotic and damaged cells may play a role in their clearance via CRP-dependent mechanisms (112).
B. Modulation of intracellular signaling by OxPLs
The data discussed in the previous chapter show that OxPLs are markers of disease of the ‘modified-self’ type that are recognized by natural antibodies and pattern-recognition receptors, and direct elimination of oxidized lipoproteins as well as senescent and apoptotic cells. However, increasing evidence shows that OxPLs are not only markers of disease but also ‘makers’ that are likely to play an active role in variable pathological states. This section describes intracellular signaling mechanisms activated by OxPLs.
1. Signal-transducing receptors initiating effects of OxPLs
OxPLs were shown or hypothesized to stimulate several types of signal-transducing receptors located on the cell surface or in the nucleus, including G protein-coupled receptors, receptor tyrosine kinases, Toll-like receptors, receptors coupled to endocytosis, and nuclear ligand-activated transcription factors such as PPARs.
a. PAF receptors
Probably the best understood molecular mechanism whereby OxPLs initiate biological effects is activation of receptor specific for PAF, which is an important lipid mediator of inflammation and platelet aggregation. This receptor specifically recognizes alkyl-acyl-phosphatidylcholines containing ether bond at the sn-1 position in combination with unusually short sn-2 acetyl residue. A proportion of PCs present in LDL particles contains sn-1 alkyl residues. Oxidative fragmentation of sn-2 PUFAs in alkyl-PCs generates products such as 1-alkyl-2-butenoyl and 1-alkyl-2-butanoyl (Fig. 4) that are recognized by PAF receptor (4, 210). These and similar ligands, usually referred to as PAF-like lipids, were found in atherosclerotic lesions and are known to form in vitro during oxidation of LDL (211). Although the affinity of PAF receptor for these ligands is 10-fold lower than for authentic PAF, they are likely to reach concentrations sufficient to activate the receptor (210). PAF-like lipids were shown to activate all major types of cells expressing PAF receptor (213). Whether similarly to alkyl-acyl PCs also fragmented diacyl-PCs can activate PAF receptor is still an open question. Similarly to PAF, POVPC was shown to stimulate adhesion of neutrophils to a gelatin matrix; the effect was inhibited by three different PAF receptor antagonists (312). In addition, POVPC competed with PAF for binding to macrophages and mimicked some (but not all) effects of PAF in this cell type (259). The role of the PAF receptor in the overall biological activity of OxPCs is not characterized. Many effects of OxPCs on ECs, for example, their proinflammatory and angiogenic action are neither mimicked by PAF, nor inhibited by PAF receptor antagonists (40, 191).
b. Prostaglandin receptors
OxPCs containing esterified isoPs (PEIPC, Fig. 4) activate receptors recognizing prostaglandins E2 and D (197) (EP2 and DP receptors, respectively). The EP2 receptor is expressed in all cell types relevant to atherosclerosis including endothelial cells (ECs), monocytes, macrophages, and vascular smooth muscle cells (VSMCs) (197). Activation of EP2 receptor on ECs results in activation of integrins and increased binding of monocytes similar to that induced by OxPAPC, while EP2 receptor antagonists inhibit action of OxPAPC. It has to be established whether prostaglandin receptors are activated by esterified isoPs directly or after release of isoPs from the glycerol backbone.
c. Scavenger receptors
Scavenger receptors were described in Section II.A.2 in the context of removal of oxidized lipoproteins and apoptotic cells containing OxPLs. In addition to that, scavenger receptor CD36, which is known to recognize fragmented unsaturated OxPLs (265), is likely to initiate some signaling effects of OxPLs. CD36 was shown to mediate activation of platelets by OxLDL acting by recruiting SRC family kinases FYN and LYN followed by activation of MKK4 and JNK2 protein kinases (60). It was hypothesized that CD36-dependent signaling stimulated by unsaturated fragmented OxPLs may explain hyperreactivity of platelets in patients with dyslipidemia (263).
d. VEGF receptors
Some effects of OxPLs may be mediated by VEGF receptors. Zimman et al. demonstrated enhanced phosphorylation (activation) of VEGFR2 within the first minutes of incubation with OxPAPC (396). Blocking antibodies to VEGF-A inhibited activation of VEGFR2 by exogenously added VEGF-A but did not influence the effects of OxPAPC, suggesting that the activation of VEGFR2 was ligand independent. It was hypothesized that trans-activation of VEGFR2 in OxPAPC-treated cells was mediated by c-SRC (396). Inactivation of VEGFR2 by siRNA and pharmacological inhibitors showed that this receptor plays a role in various effects of OxPAPC on ECs, including activation of signaling pathways (SREBP, ERK1/2) and expression of IL-8, tissue factor, and LDL receptor (396). In addition, OxPAPC, OxPAPG, OxPAPA, and OxPAPS were shown to induce VEGF-A production by ECs, peripheral blood mononuclear cells, monocyte-derived macrophages, fibroblasts, keratinocytes, lung epithelial cells, and epithelial tumor cell lines of different tissue origin (40). Therefore, in addition to transactivation of VEGFR2, VEGF-A can stimulate OxPL-treated cells acting as an autocrine and paracrine mediator.
e. Sphingosine-1-phosphate (S1P) receptor 1
Another example of receptor transactivation induced by OxPLs is S1P1 receptor (310). It was shown that OxPAPC stimulates recruitment of S1P1 to caveolin-enriched membrane microdomains, and induces its phosphorylation (activation) by AKT. These processes were important for OxPAPC-induced activation of RAC-1, leading to cytoskeleton reorganization necessary for enhancement of endothelial barrier. These results suggest that transactivation of S1P1 by OxPAPC plays a role in barrier-protective function of OxPLs (Section III.A.8).
f. Toll-like receptor 4
Several publications suggest that OxPLs may activate toll-like receptor 4 (TLR4). In particular, a role for TLR4 in OxPAPC-mediated induction of IL-8 in HeLa cells was suggested (366). More recently, OxPAPC was shown to induce lung injury and IL-6 production by mouse lung macrophages via the TLR4–TRIF–TRAF6 pathway (153). The conclusion about the involvement of TLR4 was based on the results of TLR4 knockout or knockdown experiments demonstrating attenuated effects of OxPLs. Whether OxPLs act as direct ligands of TLR4 is questionable, and it is more likely that a whole complex of receptors is involved in the recognition of OxPLs. Accordingly, several groups have shown that OxPLs do not induce many genes that are upregulated by LPS via TLR4. It has been shown that unlike LPS, various classes of OxPLs do not influence basal levels of E-selectin, ICAM-1, VCAM-1, TNFα, IL-6, IL-1α, IL-1β, and COX-2 in whole blood or individual cell types, including human umbilical vein ECs, blood monocytes, macrophage cell line, or fibroblasts (38, 83, 360). In contrast to LPS, OxPLs did not activate NF-κB-driven luciferase reporter in HEK cells stably transfected with TLR4 and MD-2 (360). These data show that OxPLs are not canonical TLR4 ligands inducing the same inflammatory effects as LPS or agonistic lipid A species. However, this conclusion does not exclude a role for TLR4 in OxPL-induced inflammation. TLR4 was shown to play an important role in aseptic inflammation induced by ischemia-reperfusion or mechanical ventilation of lungs (8, 355). It was hypothesized that in such pathologies TLR4 is stimulated by endogenous ligands (8). Similar mechanism might be activated by OxPLs. Furthermore, it is possible that OxPLs stimulate TLR4 only in combination with specific co-receptors and/or intracellular signaling adaptor proteins. Thus, agonistic effects of OxPLs on TLR4 may be selective for certain cell types and cell differentiation/activation state. Interestingly, in several in vitro and in vivo models OxPLs were shown to counteract acute inflammation induced by LPS. The hypothesized mechanisms of the anti-endotoxin activity of OxPLs are described in Section III.A.7.
g. PPARα and PPARγ
Peroxisome proliferator-activated receptors (PPARs) are intracellular ligand-activated transcription factors. Diacyl-OxPLs stimulated a PPAR response element-driven reporter construct in transfected HAECs (189). The effect of OxPAPC, POVPC, and PGPC was mediated by PPARα as indicated by activation of the ligand-binding domain of PPARα, but not PPARγ or PPARδ (189). Long-chain diacyl-OxPCs, such as PEIPC and PECPC, also activated PPARα in transfected HeLa cells (329). In addition to diacyl-OxPLs, also alkyl-PC hexadecyl-azelaoyl-PC was identified as a ligand and agonist of PPARγ in monocytes, where it stimulated expression of CD36 and COX-2 (72, 268), as well as in epidermal cells where it stimulated expression of COX-2 (391). Since oxidized FAs such as 9- and 13-HODE were shown to activate PPARs (234), it has to be established to which extent activation of PPARs by OxPLs depends on their cleavage catalyzed by PLA2 producing unesterified oxidized fatty acids, or on activation of LOX enzyme producing oxidized fatty acids intracellularly (77, 130).
h. Nonreceptor mechanisms
It is likely that certain cellular effects of OxPLs are initiated through nonreceptor mechanisms. The experimental support for this hypothesis is provided by the data showing that OxPLs induce depletion of cellular cholesterol leading to disruption of caveolae, which in turn results in activation of lipid-sensitive transcription factor SREBP and enhanced expression of IL-8 (380). Furthermore, OxPLs were shown to activate signaling events characteristic of electrophilic and unfolded protein stress responses. These cellular reactions, which are initiated independently of classical signal-transducing receptors, are described in detail in Section II.B.5.
2. Second messengers upregulated by OxPLs
a. Elevation of Ca2+ i
Many hormones, growth factors, and lipid mediators induce biological effects by increasing cytosolic levels of calcium ions (Ca2+ i) and stimulating Ca2+-sensitive intracellular effector pathways. MM-LDL was shown to induce elevation of Ca2+ i in endothelial cells (139). In addition, OxPAPC was shown to induce rapid and reversible Ca2+-responses in ECs (39). One of the downstream effectors of OxPAPC in ECs was identified as Ca2+-sensitive phosphatase calcineurin that regulates nuclear translocation of transcription factor NFAT, which in turn mediates effects of OxPAPC such as induction of tissue factor (39).
b. Elevation of cAMP
MM-LDL causes a saturable dose-dependent increase in cAMP levels in aortic ECs (255, 261) that may result from stimulation of Gs and inhibition of Gi heterotrimeric G-protein complexes (255). Likewise, OxPAPC and its components POVPC and PEIPC increase intracellular cAMP levels (67). The amount of PEIPC required to cause cAMP-dependent cellular events was ∼10-fold lower than that of POVPC, suggesting that the PEIPC component of OxPAPC is a principal cAMP-elevating PL (67). Subsequent studies identified Gs-coupled prostaglandin E2 and D receptors (EP2 and DP) as mediators of cAMP elevation induced by OxPAPC and PEIPC, but not POVPC (197). Another study presented evidence for the existence of an adenylate cyclase-coupled membrane receptor that is activated by POVPC but not PGPC (192).
Elevation of cAMP induced by OxPAPC causes cellular effects relevant to inflammation. Intracellular cAMP elevation in ECs caused by OxPAPC, POVPC, or PEIPC activates small GTPase R-RAS, which stimulates apical expression of connecting segment-1-containing fibronectin and thus promotes the entry of monocytes into sites of chronic inflammation (67) (see Section III.A.1). On the other hand, cAMP-dependent PKA plays a role in OxPAPC-induced phosphorylation of transcription factor CREB leading to enhanced expression of heme oxygenase-1, an enzyme with prominent antioxidant and anti-inflammatory properties (182). In addition, cAMP was shown to inhibit activation of the major pro-inflammatory transcription factor, NF-κB (257). Furthermore, cAMP and PKA play mechanistic roles in barrier-protective effects of OxPLs in pulmonary vascular endothelium (30) (see Section III.A.8).
3. Intracellular signaling pathways activated by OxPLs
a. Protein kinases and phosphatases activated by OxPLs
OxPLs were shown to activate a number of protein kinases and phosphatases, which characterizes OxPLs as pleiotropic lipid mediators (Table 1). More detailed description of these enzymes as mediators of bilological effects of OxPLs is given in corresponding chapters.
b. Small GTPases regulated by OxPLs
Small GTPases control various cellular functions including cell proliferation, gene expression, cell motility, regulation of monolayer integrity, and barrier properties. The most extensively characterized members are RHO, RAC, and CDC42, which have distinct effects on actin cytoskeleton, cell adhesions, and cell motility (354), whereas RAS family GTPases mainly control MAP kinase signaling and gene expression (35). Different components of OxPLs activate small GTPases RAC, CDC42 (28), and R-RAS (67) in endothelial cells.
R-RAS
OxPAPC was shown to induce selective activation of R-RAS, without affecting H-RAS activity (67). OxPAPC components POVPC and PEIPC also activated R-RAS, leading to enhanced α5β1-integrin-dependent monocyte binding to aortic ECs. Notably, activation of R-RAS by OxPAPC, POVPC, and PEIPC occurred via a cAMP-dependent mechanism (67).
RAC and CDC42
RAC and CDC42 play a crucial role in enhancement of peripheral endothelial cytoskeleton and EC barrier properties via CDC42-mediated formation of filopodia (85, 335) and RAC-dependent lamellopodia extension, formation of new cell adhesions, and enhancement of peripheral endothelial cytoskeleton (220, 332, 335). Stimulation of pulmonary ECs with barrier-protective concentrations of OxPAPC or OxPAPS strongly activates RAC and CDC42 activities (28, 30, 31). HPLC-MS analysis revealed that PECPC co-eluted with barrier-protective activity (28). Another OxPAPC component PEIPC also induced barrier-protective effects in pulmonary and aortic endothelial monolayers (K. Birukov, J. Berliner, unpublished). Consistent with their barrier-protective effects, OxPLs containing full-length oxidized sn-2-residues induced activation of RAC and CDC42 (but not RHO) small GTPases (28). In contrast, barrier-disrupting fragmented products of PAPC oxidation (POVPC, PGPC, see Section III.8) did not activate RAC or CDC42. These findings demonstrate a key role of RAC- and CDC42-dependent mechanisms in EC barrier protection induced by OxPAPC.
Mechanisms of RAC and CDC42 activation by OxPAPC
Guanine nucleotide exchange factors (GEFs) facilitate exchange of GDP for GTP in the nucleotide-binding site of small GTPases (35, 41, 393). Recent study by Birukova et al. (32) showed involvement of RAC/CDC42-specific GEFs TIAM1 and βPIX in the OxPAPC-induced pulmonary EC barrier regulation. PAK-interacting exchange factor (βPIX) is localized within focal adhesions (209), whereas activated TIAM1 becomes recruited to caveolin-enriched cell membrane microdomains (311) or to adherens junctions in EC monolayers (207). Such localization of RAC-specific GEFs induced by OxPAPC appears to be important for local RAC/CDC42 activation, leading to active cytoskeleton remodeling and enhancement of cell–cell junctions critical for increased endothelial barrier function (7, 375). Protein depletion of TIAM1 and βPIX using siRNA approach abolished OxPAPC-induced RAC activation, cytoskeletal remodeling, and EC barrier protective response (32). Thus, OxPAPC via yet to be identified mechanisms, which may include PKC, PKA and tyrosine kinases (30, 32, 54, 301), activates TIAM1 and βPIX, which in turn stimulate RAC/CDC42 GTPases and induce cortical actin polymerization and EC barrier enhancement.
RAC–RHO crosstalk
Low concentrations of OxPAPC or PEIPC do not activate RHO (28, 32). OxPAPS and OxPAPC induce robust stimulation of RAC, and at the same time significantly attenuate RHO signaling activated by thrombin (31). Both OxPAPS and OxPAPC markedly attenuated RHO activation and endothelial barrier disruption in EC monolayers exposed to pathologic mechanical stress (244). Exact mechanisms of OxPAPC-induced downregulation of RHO activity remain to be clarified and may involve direct PKA-mediated RHO phosphorylation (63), or indirect suppression of RHO activation via PKA-catalyzed phosphorylation of RHO GDP dissociation inhibitor (271). Other mechanisms may involve modulation of RHO-specific GEFs by PKA, PKC, and SRC known to be activated by OxPAPC (29, 393). Activated RAC1 may downregulate RHO via p190–RHOGAP (RHO-specific GTPase activating protein)-dependent mechanism (135).
4. Transcription factors mediating effects of OxPLs
OxPLs have profound influence on gene expression. In human aortic ECs, OxPAPC modulates expression of ∼1,000 genes, including up- and downregulated mRNAs (107, 108). OxPLs regulate genes related to inflammation, lipid metabolism, cellular stress, proliferation, and differentiation. Transcription factors characterized as mediators of the effects of OxPLs are described in Table 2. The mechanisms of gene regulation by OxPLs often differ from those activated by well-characterized inducers of corresponding genes. For example, OxPLs induce VEGF-A in endothelial cells independently of its major transcriptional regulator HIF-1 but through activation of ATF4-dependent transcription (248). Another example is IL-8 that is regulated by inflammatory agonists predominantly through the NF-κB pathway. In contrast, the induction of IL-8 by OxPLs does not require NF-κB (382), but may involve SREBP, STAT3- and PPARα-dependent mechanisms (380, 381). Furthermore, expression of tissue factor is typically induced via NF-κB-dependent transcription, while OxPLs were shown to induce expression of this blood clotting initiator acting via transcription factors EGR-1 and NFAT (39). These examples illustrate that in some cases induction of gene expression by OxPLs involves specific combinations of transcription factors. As a result, kinetics of gene induction may be different such as significantly more prolonged elevation of IL-8 induced in ECs by OxPAPC as compared to the action of TNFα (382).
5. Cellular stress pathways activated by OxPLs
a. Electrophilic stress response
Exposure to electrophiles, including electrophilic α,β-unsaturatred aldehydes derived from oxidation of unsaturated FAs evokes a concerted response aiming at limiting their toxic effect in cells. This occurs through transcriptional induction of genes having a cis-acting element called antioxidant response element (ARE), or electrophile response element (EpRE), in their promoters (240). This sequence binds the transcription factor nuclear factor-E2-related factor 2 (NRF2), which forms heterodimers with other basic leucine zipper (bZip) transcription factors, particularly small MAF proteins, recruiting transcriptional coactivators and facilitating the expression of target genes (173). Under basal conditions, NRF2-dependent transcription is repressed by its negative regulator KEAP1, which functions as an adaptor for CUL3-based E3 ligase leading to proteasomal degradation of NRF2. When cells are exposed to electrophiles, NRF2 escapes KEAP1-mediated repression, translocates to the nucleus, and binds to the ARE (Fig. 8) (173). KEAP1 is a thiol-rich protein, the mouse Keap1 having a total of 25 and the human protein 27 cysteine residues. Many of these are prone to alkylation by electrophiles (80). Electrophilic lipid oxidation products such as 4-hydroxynonenal and 15-deoxy-A12,14-prostaglandin J2 are known to react to KEAP1 protein (144; 195). Although a number of cysteine residues in recombinant KEAP1 have been shown to be reactive towards various electrophiles (138), the functionality of Cys151, Cys273, and Cys288 has been demonstrated by testing the abilities of specific KEAP1 cysteine mutants to inhibit NRF2 activity in transient transfection assays (144, 195, 363, 389) as well as in vivo (378). The model proposed by Yamamoto et al. (378) suggests that Cys151 is essential for NRF2 activation in response to electrophiles while Cys273 and Cys288 are important for KEAP1 repression of NRF2 activity under unstressed conditions (378). In addition to direct sensing of electrophiles by KEAP1, a number of protein kinase pathways have been suggested to play a role in regulating NRF2 activity (173).
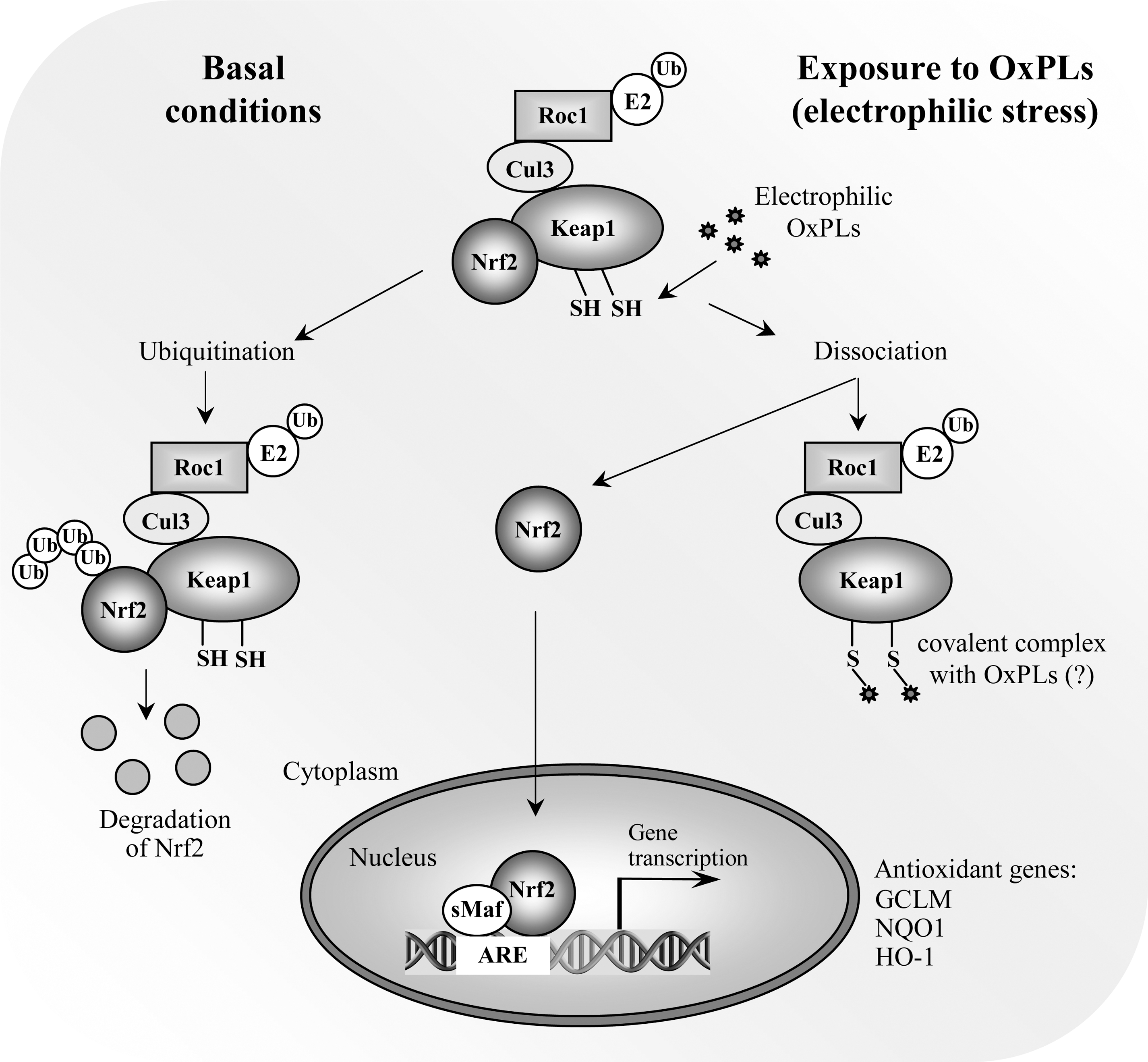
One of the earliest reports regarding the regulation of antioxidant genes by OxPLs was the observation that heme oxygenase-1 (HO-1) was induced by OxPAPC (157). The HO-1 gene is a classical ARE-regulated gene, and both human and mouse HO-1 promoters have two enhancer areas located approximately −4 and −10 kb relative to the transcriptional start site. Both enhancer areas contain multiple ARE-elements (287). Later on, a microarray analysis of human aortic ECs revealed that both modifier and catalytic subunits of glutamate-cysteine ligase (GCLM and GCLC, respectively), the rate-limiting enzyme of glutathione synthesis, were upregulated by OxPAPC (108). GCLM and GCLC also have well-characterized ARE sequences in their 5′-flanking regions. Later studies confirmed that OxPLs evoke a general NRF2-mediated response and induce antioxidant genes in ECs in vitro and mouse carotid arteries in vivo. Indeed, NRF2 was activated in OxPAPC-treated cells as assessed by its nuclear accumulation, while silencing of NRF2 expression by siRNA reduced HO-1 expression as well as the expression of GCLM and NAD(P)H:quinone oxidoreductase-1 (NQO1), one of the classical ARE-regulated genes (161). Moreover, chromatin immunoprecipitation (ChIP) experiments revealed a direct interaction of NRF2 with NQO1 ARE-containing promoter region as well as the distal enhancer region of HO-1 in OxPAPC-exposed cells. However, unlike with GCLM and NQO1, silencing of NRF2 with NRF2-specific siRNA had only a partial effect on HO-1 expression, suggesting that other redundant pathways such as that mediated by cAMP-responsive element-binding protein (CREB) contribute to its induction by OxPLs (182). It was also demonstrated that OxPAPC-inducible expression of HO-1, GCLM, and NQO1 was lower in NRF2-null than wild-type mouse carotid arteries in vivo (161). In addition to HO-1, GCLM and NQO1, also the expression of human tumor suppressor gene OKL38 by OxPLs has been shown to be NRF2-dependent (196).
The nature of NRF2-inducing OxPLs was also examined (161). Thiol reactivity of OxPAPC is important in the induction of NRF2-dependent genes, as small molecular weight thiol antioxidants glutathione and N-acetylcysteine were able to inhibit induction. On the other hand, reduction of electrophilic groups in OxPAPC by sodium borohydride significantly inhibited induction of ARE genes. Of the different species present in OxPAPC, isoP-PC was by far the most potent in inducing NRF2-dependent genes (161). However, the isoP ring structure was not absolutely necessary for the activity, as oxidized palmitoyl-linoleoyl-phosphatidylcholine (OxPLPC), unable to form the prostanoid structure (242), was almost equally effective in its ability to induce NRF2-dependent genes. Also other species such as PC-hydroperoxides were able to increase the expression of NRF2 target genes.
Selective reduction of PC-hydroperoxides to the oxidatively inert hydroxides by triphenylphosphine resulted in attenuated induction of ARE genes (161). These data implicate that the NRF2 activating capacity was shared with structurally rather diverse family of OxPLs demonstrating electrophilic properties.
What is the mechanism of NRF2 activation by OxPLs? One possible mechanism is that electrophilic OxPLs could be directly sensed by reactive cysteine residues in KEAP1, as intact PLs can be taken up by the cell by transbilayer movement (250, 337), or by receptor-mediated mechanisms (72). Moreover, using fluorescent or biotin labeling, OxPLs have recently been shown to be internalized (129, 229), and to bind to intracellular proteins such as H-RAS (129, 229). Intriguingly, structural analogs of 15-deoxy-A12,14-prostaglandin J2, a well-defined NRF2 activator known to bind KEAP1 protein thiols, have been found esterified in the sn-2 position in some of the OxPAPC species (144, 179, 195, 370). It is therefore alluring to speculate that KEAP1 would be a direct sensor of electrophilic OxPLs.
An alternative mechanism by which OxPLs activate NRF2 has been proposed (196). OxPAPC has previously been shown to increase endothelial superoxide anion production via NADPH oxidase (286). Li et al. (196) inhibited NADPH oxidase by diphenyleneiodonium (DPI) and found that the induction of OKL38 by OxPAPC was NADPH oxidase-dependent. As OKL38 induction was also inhibited by siRNA specific to NRF2, the assumption was made that NRF2 activation by OxPAPC is NADPH oxidase-dependent. However, activation of NADPH oxidase appears not to be necessary for the induction of all NRF2 target genes, as neither DPI, NADPH oxidase inhibitor apocynin, nor siRNA knocking down NOX4, the major endothelial NADPH oxidase isoform responsive to OxPAPC (1, 286), were able to affect the expression of ARE target genes in HUVECs (161). Regarding the role of NADPH oxidase in NRF2 signaling, there are only a few reports in the literature showing that NRF2 activation by any stimuli requires NADPH oxidase-derived radical production (254, 300, 367). It is unfortunate that these reports used DPI to inhibit NADPH oxidase. This compound is not a specific NADPH oxidase inhibitor, but inhibits flavoenzymes in general, and thus firm conclusions cannot be drawn from these studies. Moreover, the question still remains how NADPH oxidase-derived superoxide radical would impact on NRF2 signaling. With respect to KEAP1, protein and mixed disulfide formation with glutathione has been detected using recombinant KEAP1 protein exposed to different GSH/GSSG redox ratios (138). However, whether physiologic ROS messengers such as H2O2 are able to modify KEAP1 in this manner, is currently unknown. In contrast, there is strong evidence that KEAP1 thiol residues are directly alkylated by different inducers (138). Should modification of KEAP1 be the trigger for the ARE response by OxPLs, direct modification of thiol residues by them or their fragmented products should be the most likely mechanism.
b. Unfolded protein response
Endoplasmic reticulum (ER) is the site of folding and maturation of most secreted and transmembrane proteins within eukaryotic cells. Its complex protein folding machinery is highly sensitive to external pertubations, which may cause accumulation of unfolded or misfolded proteins within ER, a condition referred to as ER stress. The unfolded protein response (UPR) is an adaptive signaling pathway triggered by ER stress. It is a combined transcriptional and translational response aiming at restoring ER protein folding and homeostasis. The UPR signaling is sensed by three transmembrane signaling proteins residing in ER, double-stranded RNA-dependent protein kinase (PKR)-like ER kinase (PERK), inositol requiring 1 (IRE1), and activating transcription factor-6 (ATF6), each triggering downstream signaling events ultimately leading to increased expression of UPR target genes (Fig. 9) (299).
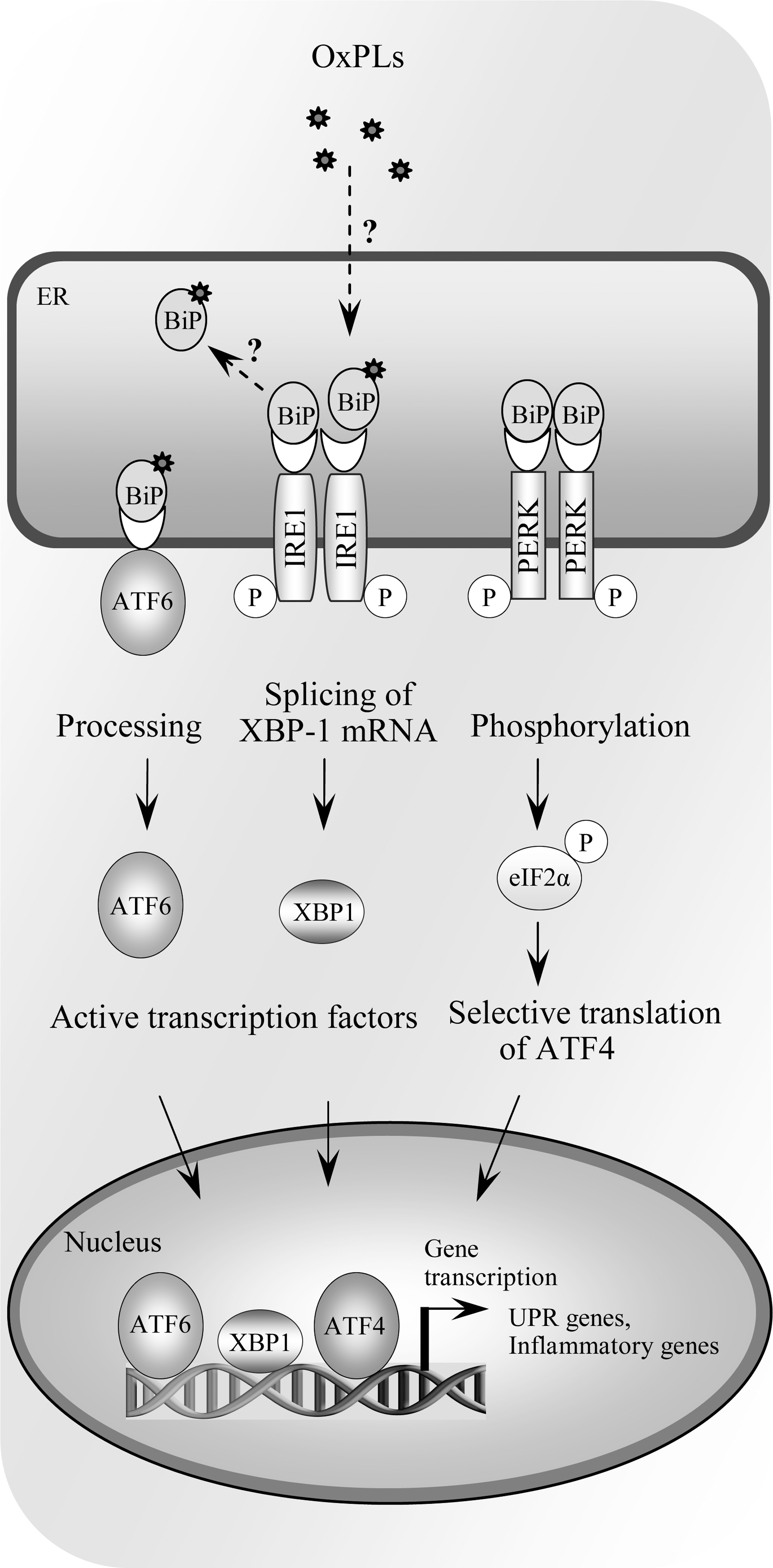
A microarray analysis of human aortic ECs exposed to OxPAPC revealed that a number of genes induced by OxPL treatment were known UPR target genes (108). Gargalovic et al. demonstrated activation by OxPAPC of all branches of UPR, including cleavage and nuclear translocation of ATF6, splicing of XBP1 mediated by IRE1, and phosphorylation of eIF2α catalyzed by PERK (107). Using the siRNA approach, the authors demonstrated that both ATF4 and XBP1 are important for transcriptional induction of proinflammatory genes IL-8, CXCL3, IL-6, and MCP-1 (107). In human atherosclerotic lesions, immunostaining for ATF3 and ATF4 revealed that ECs in shoulder areas of atheroma stained positively for both proteins (107). Moreover, lesion areas containing foam cells also stained positively. Staining with the E06 antibody recognizing oxidatively modified PCs showed positivity at the areas of lipid deposits and foam cells and in close proximity of endothelial cell layer (107). These data combined with earlier findings reporting increased expression of UPR marker genes at every stage of atherogenesis in apoe −/− mice (394) and the well-demonstrated role of UPR in macrophage apoptosis (92) and cytokine production (198) suggests that UPR may play an important role in the atherogenic process.
In addition to the role of UPR in OxPL-induced inflammation, recent findings suggest that UPR is also important for proangiogenic effects of OxPLs (40, 248). Specifically, induction of the critical proangiogenic growth factor VEGF-A was shown to be dependent on transcription factor ATF4 (248). Moreover, the structural characteristics of UPR inducing OxPLs were explored in this study. The results show that several different OxPLs having varying oxidative modifications in their sn-2 position were able to induce both VEGF-A and ATF3 (248). However, on a molar basis, PEIPC is the most active species evoking the UPR pathway. Moreover, the presence of oxidized sn-2 residues is a prerequisite, as lyso-PC is dramatically less potent than OxPLs (248).
The mechanisms by which OxPLs evoke the UPR pathway remain unclear. The involvement of previously characterized OxPL receptors, including PAF-, prostaglandin E2 receptor, PPARs, and TLR4 is not likely (107, 248). One possible mechanism is the effect of PL oxidation on biophysical properties of ER membranes, as has been suggested to occur upon accumulation of free cholesterol in macrophages (92). PL oxidation products differ in structure and polarity from their parent molecules and therefore have profound effects on membrane physical properties. It has been shown that oxidation of unsaturated PLs within the membrane increases its rigidity (44), and that incorporation of OxPLs into PL monolayer causes monolayer expansion and discontinuity, with reorientation of oxidatively modified side chains into the aqueous phase (288). These modifications should impact on the structure and function of membrane-associated proteins, and it is therefore enticing to speculate that the transmembrane sensors of ER stress could be affected in this manner. Another, alternative explanation would be that OxPLs are recognized by a currently unknown receptor which transduces the UPR signal. One promising candidate for this is GRP78/BiP, which forms complexes with all three UPR signaling mediators, IRE1, PERK, and ATF6 (Fig. 9) (285). As GRP78/BiP has been shown to bind lipid peroxidation product 4-hydroxynonenal (357) as well as biotinylated oxPAPE (129), one may envision that covalent modification of GRP78/BiP disrupts the interaction with its binding partners eliciting the UPR response.
c. Membrane stress
In addition to activation of specific cell signaling cascades, oxidation of PLs can have profound effects on cellular functions via altering biophysical properties of cellular membranes. As referred to in the previous section, oxidation of unsaturated PLs increases membrane rigidity (44), and incorporation of OxPLs into DPPC monolayer causes monolayer expansion as well as reorientation of oxidatively modified side chains and subsequent partitioning of OxPLs into the aqueous phase (288). Oxidative modification of PL bilayers not only affects plasma membrane but mitochondrial membranes as well, drastically altering their biophysical properties (218, 219). This membrane disorganization influences the conformation and function of membrane-associated proteins (177), and also exposes functionally important oxidized fatty acyl chains for recognition by their receptors, such as CD36 (134). Interestingly, oxidized PS was hypothesized to act as a “nonenzymatic scramblase” promoting externalization of unoxidized PS characteristic of apoptotic cells (348). The PS externalization serves as a signal for clearance of apoptotic cells, as discussed below.
d. Apoptosis-related signaling
Apart from their ability to evoke stress signaling pathways aiming at restoring cellular homeostasis, OxPLs, when exceeding the cell's capacity for adaptation, can trigger apoptosis. POVPC and PGPC have been reported to activate apoptotic cell signaling cascades in vascular smooth muscle cells (201). Both species were able to activate acid sphingomyelinase, and inhibition of its activity suppressed caspase-3 activation (201). Moreover, azelaoyl-PC, abundant in oxidized LDL (336), can damage mitochondria and trigger apoptosis via the intrinsic apoptotic cascade (61). In addition to exogenous OxPLs, endogenous PL oxidation products, such as CL oxidized by cyt c in mitochondria, have been implicated to play a significant role in early execution of the intrinsic apoptotic program (166). Oxidized CL epitopes can also be recognized within apoptotic cells by a natural antibody (344). It has also been argued that externalization of PS, an early event in apoptosis involved in phagocyte recognition and engulfment of apoptotic cells by macrophages, requires PS oxidation (134, 165).
In addition to their role in triggering and execution of apoptosis, increased levels of biologically active OxPLs are found in apoptotic cells (56). OxPLs are highly enriched in membrane vesicles released from activated cells and blebs derived from cells undergoing apoptosis (151). Membrane vesicles, also called microparticles, are found in circulation, and the levels are increased in patients with increased atherothrombotic risk (45). Membrane vesicles, derived in part from apoptotic VSMCs, are also highly abundant in atherosclerotic lesions (193). OxPLs within membrane vesicles have been shown to have inflammatory effects via enhancing monocyte binding to ECs, an effect inhibited by OxPL-recognizing antibodies (56, 151).
III. Accumulation and Potential Role of OxPLs in Pathology
A. Major biological activities of OxPLs
1. Proinflammatory effects of OxPLs
Intensive study of OxPLs as biologically active compounds began after these lipids were identified as an active principle of MM-LDL capable of stimulating production of chemokines and adhesion of monocytes to ECs (21). Inflammatory effects of MM-LDL may be mediated by PAF-like (alkyl-acyl) OxPLs (309), as well as diacyl-OxPLs that were reproducibly shown to induce inflammatory reactions in cultured ECs and in animal vessels in vitro, in vivo, and ex vivo (79, 102, 368). Available data show that proinflammatory effects of diacyl-OxPLs differ from the action of classical inflammatory agonists. While LPS, TNFα, or IL-1 induce extravasation of both monocytes and granulocytes via ICAM-1, VCAM-1, and E-selectin-dependent mechanisms, OxPAPC and MM-LDL do not induce expression of these adhesion molecules, and stimulate ECs to bind monocytes but not neutrophils (102, 150, 176, 192). This difference is nicely illustrated in the model of air pouch. Injection of OxPAPC stimulated accumulation of monocytes/macrophages in pouch wall, but not in the lumen. In contrast, injection of LPS resulted in accumulation of monocytes and neutrophils both in the lumen and the wall of the pouch (162). Specific induction of mononuclear cell adhesion supports the notion that OxPLs represent initiating factors in atherogenesis (18), which is characterized by selective accumulation of mononuclear cells in arterial intima. The mechanisms of selective binding of monocytes by OxPL-treated ECs are not completely understood since diacyl-OxPLs are known to stimulate expression in ECs of granulocyte-specific chemokines and adhesion molecules capable of binding neutrophils such as P-selectin (see below). Following is a description of currently identified inflammatory mechanisms activated by OxPLs.
a. Cell adhesion molecules activated by OxPLs
Two mechanisms mediating adhesion of monocytes to ECs treated with MM-LDL or OxPLs were identified. First, MM-LDL was shown to activate β1-integrins on the ECs (306). Activated β1-integrins bind a splice variant of fibronectin (FN) containing a 25-amino acid sequence called connecting segment-1 (CS-1 FN). Thus, CS-1 FN is captured from plasma and deposited on the apical surface of the endothelium where it serves as a ligand for VLA-4, a major integrin mediating firm adhesion of monocytes (306). This mechanism is likely to operate in atherogenesis as illustrated by the presence of activated β1-integrins and accumulation of CS-1 FN in atherosclerotis lesions at the sites of mononuclear cell infiltration (306). Furthermore, chronic infusion of a short peptide mimicking the VLA-4-binding site of CS-1 FN inhibited formation of lesions in LDL receptor knockout mice (305). Similarly to MM-LDL, OxPAPC was shown to stimulate adhesion of monocytes to the ECs via CS-1 FN-dependent mechanism (192). The activation of β1-integrins by OxPLs is initiated by elevation of cAMP followed by the activation of GTPase R-RAS and PI-3-kinase pathway (67). IsoP-containing PC (PEIPC, Fig. 4) was identified as a potent component of OxPAPC activating this pathway via prostaglandin E2 receptor (197).
The second mechanism of leukocyte adhesion activated by OxPLs depends on P-selectin, known to bind both monocytes and neutrophils (216). MM-LDL and its component OxPLs upregulate P-selectin in HAECs (361). P-selectin was localized intracellularly in quiescent cells but was immediately translocated to the cell surface after treatment with histamine or LDL oxidation products. More recently, it was found that this effect can be reproduced by OxPAPC in isolated vessels. Blocking antibodies to P-selectin inhibited rolling of monocytic cell line in mouse carotid arteries preperfused for 4 h with OxPAPC but not its unoxidized precursor (102). OxPAPC did not significantly elevate the levels of P-selectin mRNA, suggesting that the effect most likely resulted from mobilization of Weibel–Palade bodies (102). Upregulation of P-selectin protein and P-selectin-dependent adhesion of leukocytes were also observed ex vivo in mouse aortic segments pretreated with PL-chlorohydrin generated by oxidation of esterified unsaturated fatty acid with hypochlorite known to be generated by myeloperoxidase (79). Podrez et al. described enhanced expression of P-selectin on the surface of blood platelets incubated with OxPLs, suggesting that in addition to monocyte-endothelial interaction, OxPLs can stimulate formation of platelet-monocyte aggregates thought to play a role in pathogenesis of vascular disease (263).
b. Chemokines
OxPLs stimulate expression in ECs and VSMCs of several chemokines (Table 3), some of which are specific for mononuclear leukocytes, while other attract both monocytes and granulocytes, or are selective for granulocytes. The induction of chemokines in ECs by OxPLs is regulated by genetic and/or epigenetic factors (108), as well as by shear stress (145).
VEGF-A produced in monocytes stimulated by OxPLs is another candidate cytokine that may direct migration of leukocytes. Enhanced secretion of VEGF-A in response to OxPAPC was shown in human blood monocytes and monocyte-derived macrophages (40). In addition to its angiogenic growth factor activity, VEGF-A is a potent chemoattractant for monocytes acting via FLT-1 receptor (14). Furthermore, overexpression of VEGF-A was shown to induce expression of VCAM-1 and PECAM-1 on endothelium and stimulate adhesion of monocytes leading to the generation of bigger and more inflamed atherosclerotic lesions in apoe −/− mice (202).
c. Direct effects of OxPLs on leukocytes
Experiments testing direct action of OxPLs on leukocytes produced controversial data describing both pro- and anti-inflammatory effects. Imai et al. showed that BSA-conjugated OxPAPC stimulated production of IL-6 in mouse lung tissue macrophages (153). The effects were inhibited by mAb E06 specific for OxPCs (see Section IV.A). Similar inhibitory effects of E06 were observed after stimulation of alveolar macrophages with bronchoalveolar lavage fluid derived from mice with acute lung injury, or in peritoneal macrophages treated with artificially oxidized lung surfactant (153). Because the lung injury was ameliorated in il-6 −/− animals, the authors concluded that IL-6 induced by OxPLs in macrophages plays important role in the pathogenesis of acute lung injury (153). Smiley et al. showed that similarly to PAF, POVPC stimulated adhesion of neutrophils to a gelatin matrix (312), while Pegorie et al. observed POVPC-stimulated expression of IL-8, TNFα, and IL-1β in human monocyte-derived macrophages (259). Furthermore, treatment of monocytic cell line THP-1 with PC-hydroperoxide and PC-hydroxide, but not with unoxidized or oxidatively truncated PCs, stimulated LFA-1-dependent adhesion of cells to immobilized ICAM-1 (9).
The limited data presented above describe several proinflammatory effects of OxPLs in monocytes/macrophages, which however seem to be different from the action of well-recognized inducers of inflammation. To make the picture more complicated, OxPLs induce in leukocytes certain effects that are usually regarded as anti-inflammatory. For example, OxPAPC and PEIPC acting via the EP2 receptor were shown to inhibit basal production of TNFα and enhance production of IL-10 by human macrophages and macrophage cell line (197). Another publication demonstrated OxPL-induced inhibition of IL-12 synthesis with concomitant elevation of anti-inflammatory IL-10 expression in primary human monocytes stimulated by TLR2/1 ligand (70). Treatment of murine bone marrow macrophages with OxPAPC-treated LDL did not upregulate inflammatory genes but induced genes of protective antioxidant response (124).
In addition to regulating production of cytokines or adhesion, OxPLs can influence other functions of leukocytes directly related to inflammation and antibacterial defense. OxPAPC and other classes of OxPLs interefere with the ability of leukocytes to kill bacteria by inhibiting oxidative burst in human neutrophils (37), suppressing phagocytosis of bacteria by mouse peritoneal macrophages and polymorphonuclear cells (178), and blocking production of antibacterial peptide cathelicidin by human monocytes stimulated by TLR2/1 agonist (70). It is likely that the anti-inflammatory effects may compromise antibacterial defense and aggravate the course of infectious disease (178).
In summary, OxPLs induce both pro- and anti-inflammatory effects in monocytes and macrophages. The net effect likely depends on the type of inflammation and differentiation/activation state of leukocytes.
2. Effects of OxPLs on generation of ROS
There are indications that OxPAPC, but not its unoxidized precursor, may increase intracellular and extracellular levels of superoxide radical in human and bovine aortic ECs (78, 196, 286). Elevated activity of NADPH oxidase, partially resulting from increased expression of NOX4 mRNA, was suggested as one potential mechanism mediating enhanced production of superoxide radical in OxPAPC-treated cells (286). Another mechanism of NADPH oxidase activation depends on recruitment of RAC1 to the membrane, leading to activation of superoxide anion radical production (190). On the other hand, activation and uncoupling of endothelial NO synthase was hypothesized to be an additional source of superoxide radical in human aortic ECs treated with OxPAPC and PEIPC (114).
3. Effects of OxPLs on blood coagulation and activation of platelets
a. Modulation of blood coagulation
Since membrane PLs are key components of blood coagulation cascade, it is not surprising that oxidation of esterified PUFAs influences clot formation. The inclusion of OxPLs in liposomes resulted in significant enhancement of prothrombinase activity (371). On the other hand, oxidation of liposomes containing PUFA–PLs stimulated activites of anticoagulant proteins C and S (289). However, oxidized PS and PE were also found to stimulate the opposite reaction, namely inactivation of protein C by protein C inhibitor (206). Thus, OxPLs were reported to exert functionally opposite effects on different steps of coagulation cascade.
Additional procoagulant mechanisms activated by OxPLs regulate expression or activity of pro- and anticoagulant proteins on the surface of endothelium. Three effects of OxPLs enhancing thrombogenic potential of endothelium were described (Fig. 10). First, MM-LDL (91), OxPAPC, and PGPC (39) were shown to upregulate on ECs expression and activity of tissue factor, which is a master-switch initiating clotting cascade. OxPAPC upregulated mRNA, protein, and activity of tissue factor acting via mechanisms involving protein kinases ERK1/2, phosphatase calcineurin, and transcription factors NFAT and EGR-1 (39). In addition, OxLDL and OxPCs downregulated expression of the major anticoagulant protein located on ECs, thrombomodulin. The effect resulted from decreased nuclear accumulation and promoter binding of transcription factors mediating basal expression of thrombomodulin, including retinoic acid receptor β, retinoid X receptor α, Sp1, and Sp3 (156). Furthermore, peroxidation products of PC and PE containing esterified linoleic acid were shown to influence the initial steps in blood coagulation by inactivating tissue factor pathway inhibitor. The effect was explained by direct binding of OxPLs to the C-terminus of tissue factor pathway inhibitor leading to its inhibition (247).
In summary, OxPLs induce multiple effects on blood coagulation, and the majority of these effects are likely to induce procoagulant shift in endothelium and promote blood clotting (Fig. 10).
b. Activation of platelets
Oxidized lipoproteins and lipids extracted from atherosclerotic lesions are known to induce proaggregant effects in platelets, which may be partially mediated by the action of OxPLs. PAF-like (alkyl-acyl) OxPLs and LysoPA accumulate in MM-LDL and atheroma and act as potent inducers of platelet activation working in concert with other agonists (132, 213).
In contrast to the well-characterized receptors mediating action of PAF-like (alkyl-acyl) OxPLs and LysoPA, the mechanisms whereby platelets are activated by diacyl-OxPLs are not completely understood. Diacyl-OxPCs were shown to activate platelets independently of PAF-, LysoPA-, LysoPC-, or thromboxane A2 receptors (119). Diacyl-OxPLs are weak agonists since they induce platelet shape change and surface expression of P-selectin but do not induce platelet aggregation when added alone (119, 131). Although relatively weak, the effects of OxPLs may be of biological relevance, as suggested by the ability of unsaturated fragmented diacyl-OxPCs to stimulate platelets synergistically with agonists such as ADP (263). Experiments using CD36 knockout mice showed that the proaggregant action of OxLDL and unsaturated fragmented diacyl-OxPLs on platelets may be initiated by CD36 (263, 265). The role for CD36 is further supported by the inhibition of platelet activation by peptide mimicking the CD36 binding site (168). Recognition of OxLDL by CD36 is followed by recruitment and activation of SRC-family kinases, MKK4 and JNK2. It is likely that activation of this cascade underlies the enhanced reactivity of platelets in hyperlipidemic patients known to have elevated plasma levels of OxPLs (168, 263).
In summary, in addition to inducing thrombogenic shift in the endothelium, OxPLs are likely to increase the risk of thrombosis through direct activation of platelets mediated by CD36, PAF, LPA and probably other unidentified receptors.
4. Modulation of vascular smooth muscle cell phenotype
A unique feature of vascular smooth muscle cells (VSMC) is their phenotypic plasticity. During early atherogenesis, SMCs undergo phenotypic switching from contractile to synthetic phenotype characterized by decreased expression of SMC differentiation marker proteins, a high rate of cellular proliferation, and increased synthesis of extracellular matrix proteins (249). Accumulating evidence suggests that OxPLs may play a role in phenotypic modulation of VSMCs. OxPAPC and its oxidatively fragmented component POVPC profoundly reduced protein expression of the markers of differentiated SMC such as smooth muscle (SM) α-actin and smooth muscle myosin heavy chain (SM MHC), while simultaneously increasing expression of the proinflammatory genes MCP-1, MCP-3, and cytolysin (260). OxPL-induced inhibition of SM α-actin and SM MHC expression were mediated at least in part by Krüppel-like transcription factor 4 (KLF4), the potent repressor of SMC differentiation marker genes. POVPC-induced KLF4 activation suppressed expression of VSMC differentiation transcription factor myocardin, thus pointing to a potential mechanism of POVPC effects on VSMC phenotypic transition (260).
In addition to phenotypic effects on vascular SMC, fragmented product of PAPC oxidation, POVPC, alters extracellular matrix production and stimulates VSMC migration (65). POVPC increased expression of type VIII collagen alpha1 chain (Col8a1) mRNA in vitro and in vivo, which was mediated by activation of KLF4 and Sp1 and increased KLF4 binding to the Col8a1 gene promoter. Such activation, as well as POVPC-induced VSMC migration were markedly reduced in KLF4- or type VIII collagen-knockout VSMCs (65). These findings suggest that changes in VSMC phenotype, extracellular matrix composition, and resulting stimulation of VSMC migration are induced by fragmented OxPL products and may contribute to the pathogenesis of atherosclerosis.
5. Angiogenic activity of OxPLs
High concentrations of OxPLs accumulate in atherosclerotic lesions; another characteristic feature of late atheroma is presence of neovessels that make the plaque prone to rupture. Since the density of neovessels does not always correlate with the thickness of plaque and corresponding level of hypoxia, it was hypothesized that additional intraplaque factors may play a role. Available data indicate that OxPLs are likely to be one of locally accumulated mediators promoting angiogenesis within the lesion. It was shown that diacyl-OxPLs stimulate migration of ECs, induce formation of EC sprouts, and promote angiogenesis in Matrigel plug model in mice (40). The authors presented evidence that angiogenic effects of OxPLs were mediated by autocrine mechanisms. According to the model, OxPLs stimulate production by ECs of VEGF-A, IL-8, and COX-2-derived prostaglandins that act in concert to induce angiogenic switch in ECs (40) (Fig. 11). Furthermore, OxPLs stimulated expression of metalloproteinase ADAMTS-1, further suggesting that OxPLs may play a role in plaque destabilization (40). Later studies demonstrated that OxPL-induced production of VEGF-A is mediated by ATF4-dependent transcription (248).
In contrast to diacyl-OxPLs, PAF-like (alkyl-acyl) lipids and PAF were shown in different models either to stimulate or inhibit angiogenic reactions. In a study describing negative effects on angiogenesis, PAF-like (alkyl-acyl) lipids present in OxLDL inhibited production of FGF2, a key growth factor crucially important for formation of neovessels (58). In addition, fragmented OxPLs such as POVPC inhibited in a CD36-dependent manner production of VEGF-A by macrophages and inhibited corneal neovascularization (232).
Taken together, available data show that depending on chemical structure and biological context, OxPLs can induce both pro- and antiangiogenic effects. Further studies are required in order to identify the factors determining the balance of these opposite activites.
6. Calcification of atherosclerotic lesions and bone
Advanced atherosclerotic lesions are characterized by calcification and express osteoblastspecific markers. Increasing evidence shows that OxPLs might represent one of the factors responsible for calcification of atheroma. Treatment of calcifying vascular cells derived from aortic smooth muscle cells with MM-LDL or OxPAPC but not unoxidized lipoprotein or lipids, stimulated expression of alkaline phosphatase, specific changes in cell morphology and formation of cellular aggregates containing calcium mineral–all characteristic features of osteoblastic differentiation (256).
Paradoxically, in many patients with atherosclerosis and calcification of arteria, parallel loss of bone mineral often leads to osteoporosis. Parhami et al. offered an explanation for this phenomenon by showing that OxLDL and OxPLs exert opposite effects on evolution of bone as compared to arterial wall. They demonstrated that MM-LDL and OxPAPC inhibited differentiation of bone-derived preosteoblasts (256). Furthermore, OxPAPC was shown to inhibit action of bone anabolic agents such as bone morphogenic protein-2 and parathyroid hormone (148). An in vivo study using LDL receptor knockout mice, characterized by increased levels of oxidized (phospho)lipids, suggested that hyperlipidemia may interfere with osteoanabolic effects of parathyroid hormone (147).
Taken together, the data suggest that OxPLs are likely to play opposite role in calcification of arterial wall and bone.
7. Anti-inflammatory activities
Products of lipid peroxidation traditionally are regarded as toxic compounds inducing tissue damage and initiating inflammation. The data on the proinflammatory action of OxPLs in atherosclerotic vessels described above are consistent with this notion. Paradoxically, under certain conditions OxPLs also demonstrate tissue-protective and anti-inflammatory activites. The best documented anti-inflammatory effect of OxPLs is their ability to block acute inflammatory reactions induced by LPS (38, 192, 364). Below follows a description of TLR-specific and general anti-inflammatory mechanisms activated by OxPLs.
a. Modulation of TLR activity
Anti-endotoxin activity is characteristic of several OxPL species, including fragmented saturated OxPLs (192), fragmented unsaturated OxPLs (327), PL-hydroperoxides, PL-hydroxides, and isoP-containing PLs (360). In addition to TLR4, OxPAPC inhibited activation of TLR2 (83, 364), but did not influence the activity of other TLRs in transfected HEK 293 cells (83). Testing in several animal models confirmed the ability of OxPLs to inhibit acute inflammatory responses to exogenous agonists of TLRs 2 and 4 in vivo (38, 83, 204).
Available evidence suggests that OxPLs inhibit several steps in activation of TLRs, including extracellular components of the cascade (LBP, CD14, etc.) as well as cell-associated machinery (360) (Fig. 12). One hypothesis postulates that OxPLs bind to LBP and CD14, and that binding of OxPLs and LPS is mutually exclusive (360). Furthermore, OxPLs inhibit binding of LPS to MD-2 (83). Since LBP and especially CD14 and MD-2 are crucially important for presentation of LPS to TLR4, inactivation of these proteins is expected to inhibit action of LPS. In addition to TLR4, CD14 presents PAMPs of gram-positive bacteria to TLR2, explaining why OxPLs inhibit activation of TLR 2 and 4 but not other TLRs that are CD14-independent (83).
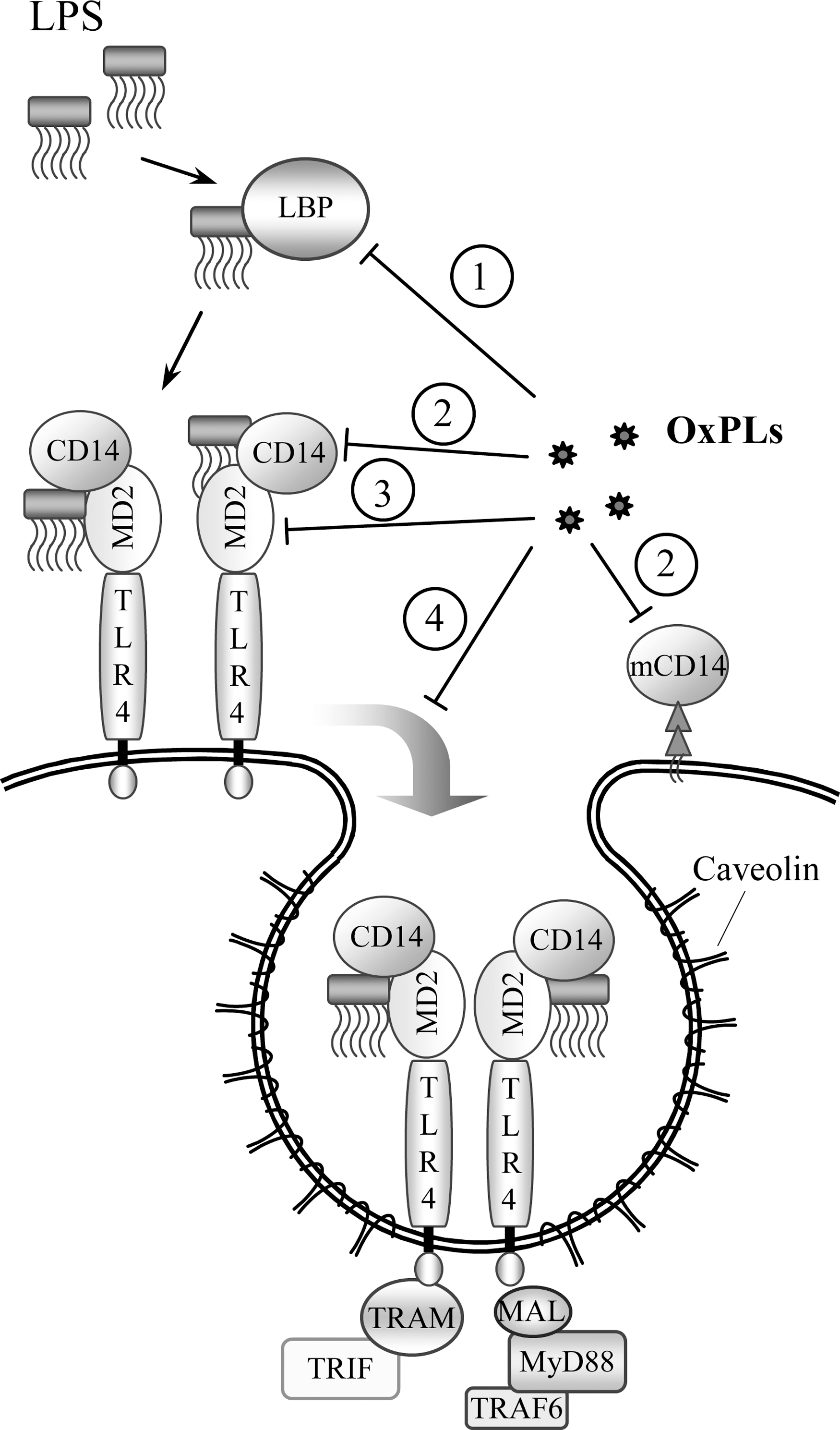
Another hypothesis suggests that OxPLs inhibit formation of TLR4 signaling complex with its downstream adaptors. This effect may be due to disruption of lipid rafts by OxPLs resulting from adsorption of cholesterol or action of sphingomyelinase products (364, 365), although another work showed that action of cholesterol-disrupting agents alone is not sufficient for inhibition of TLR2 and 4 (83), thus suggesting the involvement of more complicated inhibitory mechanisms.
In summary, it is likely that OxPLs inhibit action of LPS via a multi-hit mechanism targeting several steps in LPS recognition (360) (Fig. 12).
Some studies reported TLR4-dependent proinflammatory action of OxPLs (153, 366). These data suggest that under specific conditions or in certain cell types OxPLs may be agonistic for TLR4. One possibility is that alternative co-receptors, such as 37 kDa GPI-anchored protein (366) or scavenger receptors (136), rather than CD14, present OxPLs to TLR4. It is also possible that in those cases OxPLs upregulated endogenous TLR4 agonists or induced switch of TLR4 to other signaling adaptors. These speculative mechanisms require further investigation.
b. Induction of antioxidant and anti-inflammatory genes
An additional mechanism whereby OxPLs may exert anti-inflammatory and tissue-protective action is induction of drug metabolism phase II genes mediating protection from oxidant stress. Low concentrations of OxPLs do not damage cells but induce antioxidant enzymes such as glutamate-cysteine ligase and especially HO-1, well recognized for its prominent anti-inflammatory activity (2, 161, 182). These reactions are an important part of electrophilic stress response (ESR) and are discussed in more detail in Section II.B.5.
c. Inhibition of oxidative burst
In addition to modifying inflammatory reactions, different OxPLs including OxPAPC, OxPAPG, OxPAPS, OxPLPC were shown to inhibit oxidative burst induced by fMLP or PMA in neutrophil granulocytes without influencing viability or other functions of these cells (37). The effect resulted from the inhibition of assembly of phagocyte oxidase complex. Thus, oxidation of PLs may serve as a negative feedback preventing tissue damage by uncontrolled oxidative burst.
d. Lung barrier protection
Lung edema is a common life-threatening complication of acute systemic inflammation. OxPLs prevented lung edema formation in several in vivo models, including application of PAMPs or pathological lung ventilation. These protective effects of OxPLs are described in more detail in Section III.B.3.
8. Effects of OxPLs on cytoskeleton, noninflammatory cell adhesion, and permeability of endothelium
a. Cytoskeletal mechanisms of endothelial permeability
Paracellular pathway of endothelial permeability is regulated by a balance between competing contractile forces imposed by actomyosin cytoskeleton organized into stress fibers and opposing adhesive cell–cell and cell–matrix tethering forces produced by focal adhesions, adherens junctions, and peripheral actin cytoskeletal rim (220). Activation of myosin light chain kinase, MAP kinases, tyrosine kinases, and RHO-mediated signaling by edemagenic and pro-inflammatory agents causes phosphorylation of regulatory myosin light chains (MLC), cytoskeletal changes, activation of EC contraction, and destabilization of adherens junctions, leading to paracellular gap formation and increased EC permeability (220). RHO effectors MDIA and RHO-associated kinase (RHO-kinase) appear to be required for RHO-induced assembly of stress fibers, MLC phosphorylation, and actomyosin-driven cell contraction (110, 171, 353). In contrast, RAC-mediated enhancement of peripheral actin cytoskeleton and cell–cell junctions in OxPAPC-stimulated ECs is essential for restoration of EC barrier (28, 31, 33).
b. OxPAPC and EC permeability
Treatment of pulmonary ECs with OxPAPC in the range of 5–30 μg/ml caused dose-dependent enhancement of monolayer barrier (Fig. 13A), which lasted over 12 h (28). Of note, unoxidized PLs did not affect basal or agonist-induced permeability in ECs. At higher concentrations OxPAPC induced barrier disruption (Fig. 13A). Structure-function analysis showed that different molecular species present in OxPAPC have opposite influence on endothelial barrier. Nonfragmented sn-2-oxygenated OxPLs, especially cyclopenthenone-containing species (PEIPC, PECPC) are likely to be responsible for barrier-protective and barrier-enhancing effects of OxPAPC (28). In contrast, sn-2-fragmented OxPLs such as PGPC (Fig. 13B) and POVPC, increased endothelial permeability even at low concentrations (28, 272).
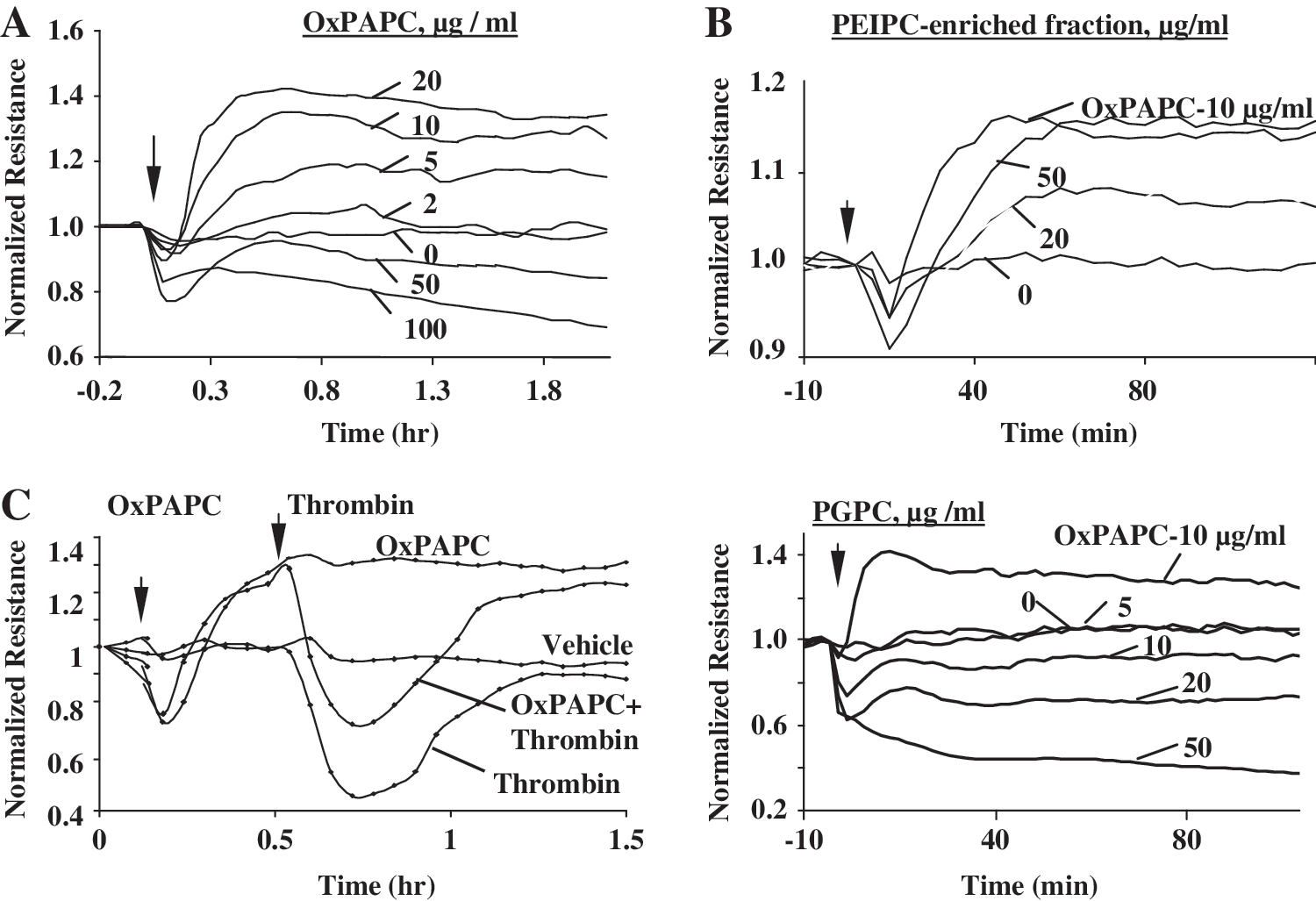
Oxidized phosphatidyl serine (OxPAPS) and phosphatidyl choline (OxPAPC) bearing negatively or positively charged polar head groups, respectively, both exhibited potent and sustained barrier-protective effects. However, OxPL lacking head group (oxidized phosphatidic acid, OxPAPA) only transiently enhanced EC barrier (31). All three classes of OxPLs attenuated agonist-induced EC permeability triggered by transient activation of RHO pathway, but only OxPAPC and OxPAPS abolished short- and long-term EC barrier disruption caused by LPS (31).
One potential clinically relevant feature of OxPAPC is its ability to suppress RHO-dependent elevation of EC permeability induced by inflammatory and edemagenic agents (Fig. 13C). OxPAPC attenuated elevation of endothelial permeability caused by thrombin, IL-6, LPS, or exposure of endothelial cells to high-magnitude cyclic stretch and thrombin (31, 244). Treatment with OxPAPC accelerates the recovery of the compromised EC barrier function (28, 31). Another important observation is the barrier-protective effect of OxPAPC in the in vivo model of ventilator-induced lung injury (VILI) (244). VILI-associated EC barrier dysfunction was also reproduced in the in vitro model in EC monolayers exposed to high magnitude cyclic stretch and thrombin (244). This study demonstrated suppression of RHO signaling as a critical mechanism of protective effect of OxPAPC in VILI.
c. OxPAPC and cytoskeletal remodeling
Barrier-protective OxPLs cause specific rearrangements of cytoskeleton. They reduce the number of central F-actin stress fibers and markedly enhance peripheral F-actin rim (Fig. 14A, shown by arrows) responsible for the maintenance of monolayer integrity and EC barrier function. These features are similar to effects of other barrier-protective agonists, such as sphingosine 1-phosphate and prostacyclin (34, 106). The unique feature of OxPAPC is formation of microspike-like actin structures at the cell–cell interface (28) mediated by simultaneous activation of RAC and CDC42 (28), which likely contributes to the sustained character of EC barrier protective responses to OxPAPC. RAC effectors, WAVE, WASP, and ARP-2,3 complex are required for actin polymerization and enhancement of peripheral actin rim (43, 59, 230). Consistent with RAC/CDC42-mediated mechanism of cytoskeletal remodeling, OxPAPC induced peripheral translocation of the regulators of actin polymerization, preferentially activated by RAC (cortactin, p21ARC), CDC42 (N-WASP), and RAC/CDC42 (ARP3, phospho-cofilin) (28).
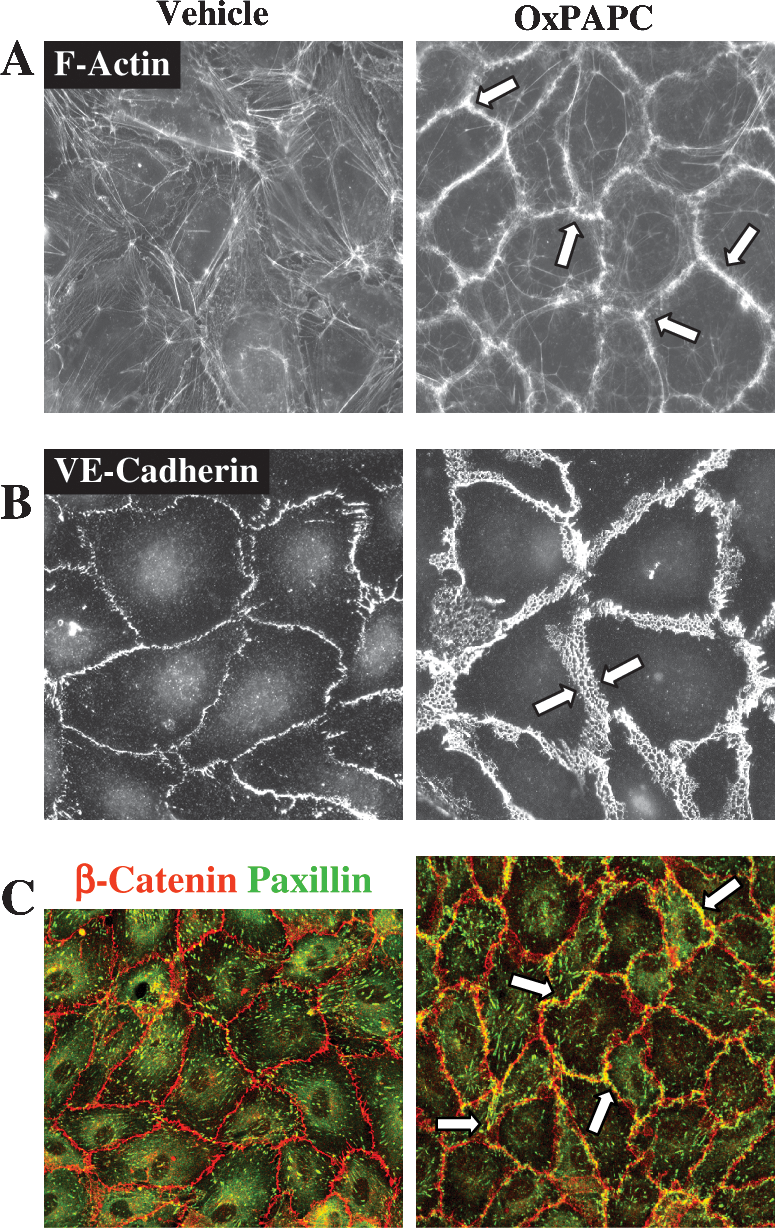
d. OxPAPC and assembly of adherens junctions
Assembly of adherens junctions (AJ) is critical for basal barrier enhancement and restoration of endothelial barrier disrupted by inflammatory agents or pathologic mechanical forces. Activated RAC stimulates association of VE-cadherin with intracellular catenin complex. This mechanism also provides physical integrity and cytoskeletal attachment of AJ complex, leading to increased EC barrier properties (76). OxPAPC at barrier-protective concentrations (5–30 pg/ml) causes enlargement of AJ areas in EC monolayers shown by arrows in Fig. 14B (33). Knockdown of AJ protein β-catenin attenuates OxPAPC-induced barrier enhancement in pulmonary EC (33).
e. OxPAPC induces FA-AJ interactions
OxPAPC induces peripheral accumulation of focal adhesion (FA) protein complexes containing paxillin (33) and leads to colocalization of FA complexes with enlarged adherens junction complexes (Fig. 14C, marked by arrows). Morphological and biochemical assays revealed novel OxPAPC-induced interactions between FA and AJ complexes via paxillin and β-catenin association mediated by RAC- and CDC42-dependent mechanisms (33). Disruption of paxillin-β-catenin interactions attenuated OxPAPC-induced barrier protective effects (33). This study suggests that novel paxillin-β-catenin interactions may form a “double rim of defense” in endothelial monolayers, which secures intercellular flux of solutes and macromolecules controlled by AJ and limits the fluid influx between EC and basal membrane by formation of peripheral FA rim. FA–AJ interactions thus may function as a double lock, preventing macromolecular transport between and under EC.
f. Gap junctions and tight junctions
As discussed above, high OxPAPC concentrations or EC treatment under serum-free conditions may cause EC barrier dysfunction. DeMaio et al. reported decreased expression of tight junction proteins occludin and ZO-1 and increased flux of 10-kDa dextran through EC monolayers indicative of EC barrier dysfunction in EC stimulated with 20–50 pg/ml OxPAPC in serum-free conditions. These effects were linked to oxidative stress induced by OxPAPC (78).
A study by Isakson et al. (155) showed OxPAPC-induced dysregulation of connexins CX37, CX40, and CX43 in the carotid artery EC and VSMC. These proteins form gap junctions that are assembled as dodecameric channels composed of a pair of connexin hexamers linking two adjacent cells. Gap junctions are involved in transduction of signal from cell to cell by intercellular transport of small signaling molecules. OxPAPC-induced changes in connexin expression markedly reduced biocytin dye transfer between ECs and VSMC, suggesting impaired communication between EC and VSMC. These results show that phospholipid oxidation products such as OxPAPC, which accumulate during the development of atherosclerotic lesions, may affect gap junction-mediated communication in the vascular wall.
In summary, OxPAPC and isoP-PLs demonstrate potent lung barrier-protective effects in cell and animal models of acute lung injury induced by inflammatory and edemagenic agonists and pathologic mechanical strain (Fig. 15). These effects are mainly mediated by activation of RAC and downregulation of RHO-mediated signaling, leading to enhancement of peripheral actin structures, cell–cell junctions, and novel interactions between cell–cell and cell–substrate adhesive protein complexes. Another important lung vascular protective mechanism is inhibition of inflammatory cascades triggered by bacterial components, which will be described below (Section III.B.3).
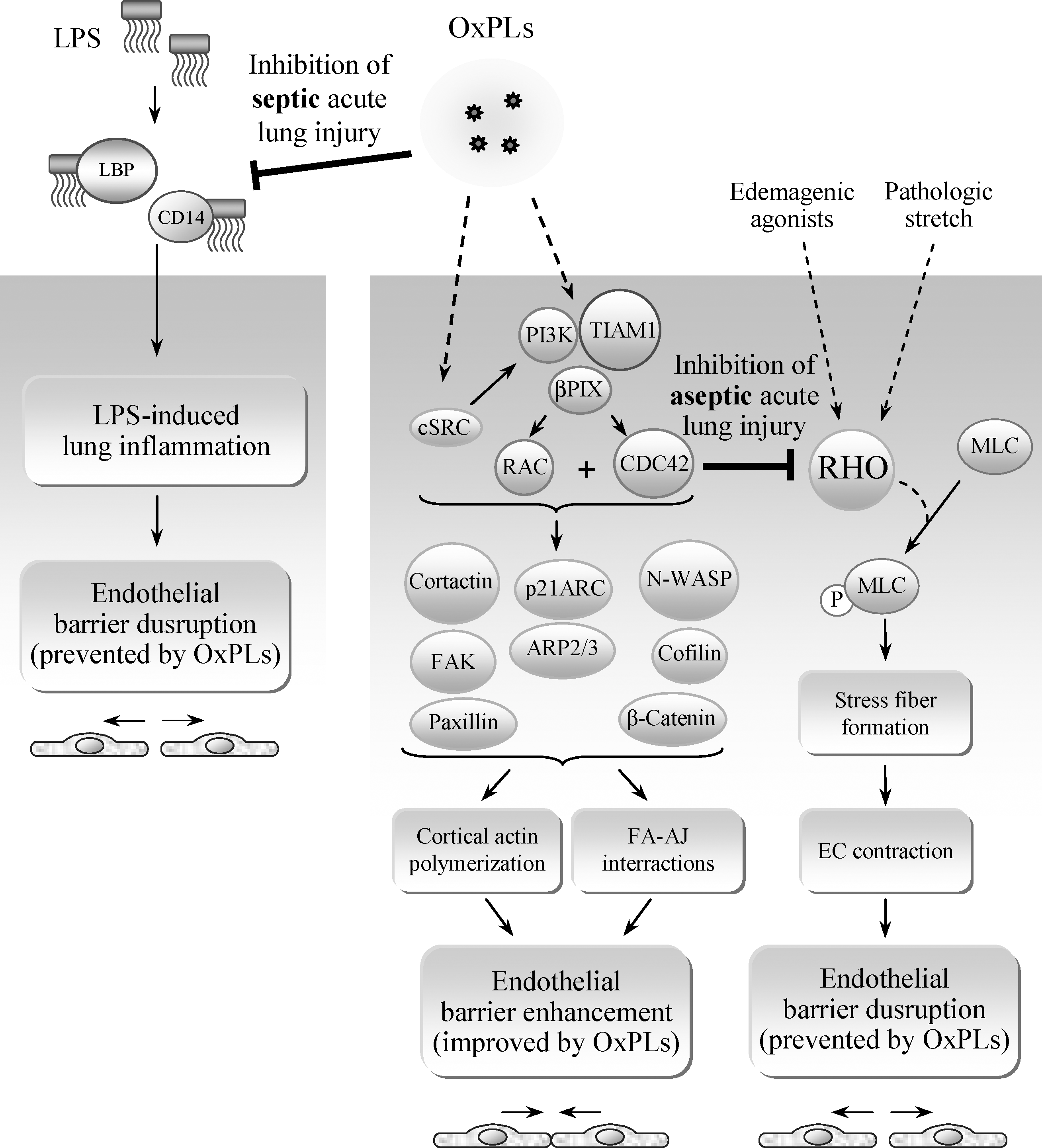
9. Regulation of adaptive immunity
Inflammatory signals play a key role in initiation of adaptive immune responses. Recent studies demonstrated that OxPLs can modulate adaptive immune system by acting on antigen-presenting dendritic cells as well as T cells.
a. Modulation of dendritic cell function by OxPLs
Dendritic cells (DCs) are key regulators of adaptive immunity. In the steady state, DCs reside in peripheral tissues as immature antigen-presenting cells and are considered to be tolerogenic. During infections, DCs are activated by stimulatory signals from invading pathogens. DC maturation is accompanied by translocation of MHC–peptide complexes to the cell surface and upregulation of co-stimulatory molecules such as CD40, CD80, and CD86. As a result, DCs become fully competent to activate T-cells. Recent studies demonstrated that OxPLs have no significant direct influence on phenotype of immature DCs except for modestly elevated surface expression of CD86 and MHC class II (36). Furthermore, OxPAPC alone did not have major impact on the functional behavior of DCs. However, OxPLs can strongly interfere with the activation of DCs induced by PAMPs recognized by TLRs 2, 3, and 4, as well as by CD40–CD40L interaction.
OxPAPC was found to inhibit in human monocyte-derived DCs upregulation of CD40, CD80, CD86 and CD83 induced by LPS (36) (Fig. 16). Moreover, LPS-induced surface expression of MHC classes I and II as well as production of IL-12 was significantly inhibited by OxPAPC. As a consequence, OxPAPC dramatically reduced T-cell stimulatory capacity of DCs treated with LPS (36). OxPAPC also strongly inhibited surface expression of chemokine receptor CCR7 in LPS-treated DCs. Downregulation of CCR7 is known to impair emigration of DCs from the inflamed tissue to the lymph node that is crucially important for the induction of an antigen-specific immune response (214).
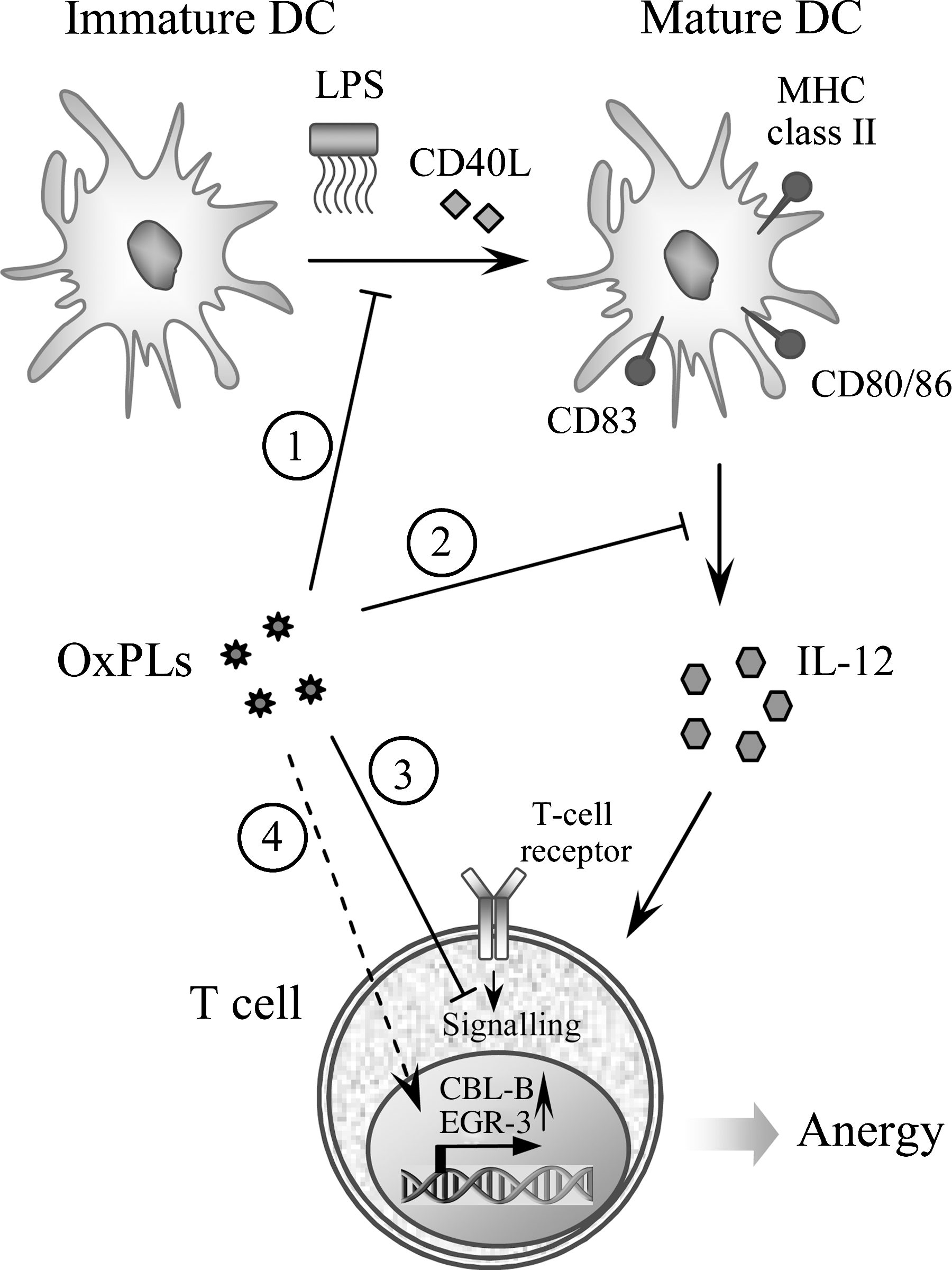
It is likely that many of the abovementioned effects result from the ability of OxPLs to inhibit LPS binding to, and activation of, TLR4 that is discussed in Section III.A.7. However, low concentrations of OxPAPC did not block LPS-induced signaling (i.e., NF-κB activation) or upregulation of the DC activation marker (CD83), but suppressed LPS-induced production of IL-12 and TNFα (36) (Fig. 16). Moreover, at low concentrations of OxPAPC, the ability of LPS-treated DCs to polarize T-cells towards IFN-γ-producing Th-1 cells was abrogated (36). These results suggest that in parallel to the inhibition of TLR4 activation, OxPLs modulate Th-1-driving capacity of DCs via currently unknown mechanisms.
OxPAPC also inhibited DC maturation induced by Poly I:C, which signals through TLR3. Similarly to the effects on LPS-induced DC maturation, addition of OxPAPC prevented induction of costimulatory molecules, production of cytokines and increase in T cell-stimulatory capacity in DCs activated by Poly I:C (36). Although to a lower extent, OxPAPC also inhibited maturation of DCs induced via TLR2 or CD40. OxPAPC did not influence upregulation of co-stimulatory molecules or activation of NF-κB induced by CD40L; however, CD40L-induced elevation of TNFα and IL-12 was potently suppressed by OxPAPC (36). Similarly, in case of TLR2-mediated maturation stimulated by Pam3CSK4 or proteoglycan, OxPAPC did not interfere with upregulation of co-stimulatory molecules, but potently inhibited TNFα and/or IL-12 production (36). These data identify OxPAPC as a strong negative regulator of IL-12 production in DCs treated with various maturation-inducing stimuli, pointing to a mechanism whereby OxPLs can impair adaptive immune responses.
b. Induction of T cell anergy by OxPLs
In addition to modulating DC function, OxPLs can directly regulate T cell activation. OxPLs having different polar head groups (OxPAPC, OxPAPS, OxPAPG, and OxPAPA), but not their unoxidized precursors, strongly inhibited the proliferation of purified human T cells stimulated with anti-CD3/CD28 or anti-CD3/CD63 mAbs (302). Inhibition of T cell proliferation by OxPLs was not due to their toxicity since T cell proliferation triggered by PMA/ionomycin was not diminished by OxPLs. T cells activated by anti-CD3/CD28 in the presence of OxPLs produced and released lower amounts of IFN-γ and IL-2, whereas IL-4 was only slightly diminished. OxPL-treated T cells produced only low amounts of IL-10, a well-established immunosuppressive cytokine. Thus, it is unlikely that the inhibitory effects of OxPLs on T cells were due to elevated production of IL-10. OxPLs prevented the expression of de novo- synthesized activation markers (CD25, MHC-II) but not expression of CD63 or CD69, which translocate from preformed intracellular pools to the cell surface of anti-CD3/CD28 mAb-stimulated T cells. Furthermore, T cells stimulated in the presence of OxPLs were characterized by low cytotoxicity of CD8+ T cell, which is pivotal mechanism in removing pathogens. Most importantly, T cells that were activated in the presence of OxPLs failed to proliferate in response to restimulation, a phenomenom called anergy. This hypoproliferative state was accompanied with an upregulation of the anergy-related genes early growth response gene 3 (EGR-3) and the E3 ligase Casitas-B-lineage lymphoma protein-B (CBL-B), which are both well-defined negative regulators of T cell activation (88). In summary, available data suggest that treatment of T cells with OxPLs rather modulates TCR/CD3 signaling than completely blocks it. In contrast to IL-10 and immunosuppressive drugs such as FK506, cyclosporine A, glucocorticoids, or rapamycin, OxPLs are known to repress the cytolytic function of cytotoxic T cells (307, 325, 388). These observations show that OxPLs demonstrate unique immunosuppressive characteristics and allow hypothesizing that direct inhibitory effects of OxPLs on T cells might contribute to the control of T cell function necessary to avoid overwhelming or even detrimental Th1-driven immune responses at sites of inflammation.
B. Accumulation and role of OxPLs in specific pathologies
1. Atherosclerosis
Quantification of OxPCs using liquid chromatography coupled to tandem mass spectrometry showed that atherosclerotic vessels contain high concentrations of OxPCs. Different species of OxPCs were detected in atherosclerotic vessels including PL-hydroperoxides and hydroxides (362), oxidatively fragmented PL species containing saturated and unsaturated truncated residues (137, 264, 328, 368), PL-esterified isoPs (118, 269, 328, 368, 370), and PL-isolevuglandins (267, 292). The analysis of OxPL spectra at different stages of human atherosclerosis (fatty streak, fibrous plaque, necrotic core, and calcified tissue) identified similar OxPL species at different stages of the disease, including early oxidation species (hydroperoxides) and advanced products (fragmented OxPLs) (276). These data suggest that during the evolution of atherosclerotic lesion, OxPLs are continuously generated and degraded.
In parallel with elevated levels of OxPLs, atherosclerotic vessels express high amounts of proteins known to be induced by OxPLs in vitro. These include for example MKP-1 (279), ATF3, ATF4 (107), SREBP-1 (380), HO-1 and IL-8 (64), MCP-1 and COX-2 (203), and GROα (253). Furthermore, quantification of OxPLs in atherosclerotic vessels showed that total concentration of active OxPLs reach locally in atherosclerotic vessels high levels comparable with those inducing biological effects in vitro (Table 4). These data support the view that OxPLs not only accumulate in atheroma but play mechanistic role in atherogenesis.
Several activities of OxPLs described above suggest that OxPLs are likely to be involved into all stages of atherogenesis. OxPLs can play a role in initiation of arterial inflammation (monocyte–endothelial interactions, production of chemokines), progression of disease (foam cell formation, smooth muscle cell phenotypic modulation and migration, extracellular matrix production, and calcification) and development of complications (angiogenesis, production of metalloproteinases, thrombogenic shift in endothelium, and activation of platelets). OxPLs act on all major cell types involved in atherogenesis including monocytes, endothelial and vascular smooth muscle cells, lymphocytes, and platelets. These findings raise a possibility that regulation of OxPL levels and inhibition of their deleterious effect may become a useful strategy for prevention and treatment of atherosclerosis. Current research in this direction is described in Section IV.B.
2. Acute inflammation
Acute inflammation is accompanied by massive production of ROS, which in parallel with other antibacterial mechanisms kill invading microorganisms. As a result of “friendly fire,” also host lipoproteins and membrane lipids become oxidized. The elevation of circulating levels of OxLDL in response to inflammatory stimuli has been shown in vivo in models of acute inflammation induced in hamsters by injecting LPS, zymozan, or turpentine (221). Elevated plasma levels of PL-hydroxides were detected in plasma of patients with dengue fever (188).
The production of OxPLs in response to inflammation may be induced in different cell types including leukocytes. Phorbol ester-stimulated neutrophils and monocytes incubated with PUFA-PCs produced mono- and bishydroperoxides of PC, as well as isoP–PC, thus suggesting that activated phagocytes can oxidize lipids in the surrounding medium (159). Furthermore, macrophages infected with mycobacteria accumulated isoP–PC (PEIPC) (70). Nonleukocyte cell types also generate OxPLs in response to inflammatory stimuli. It has been shown that accumulation of fragmented OxPLs (POVPC, PGPC), as well as OxPLs containing esterified epoxyisoprostanes (PEIPC, PECPC) was induced by IL-1 treatment of human aortic ECs (329). In addition, infection with influenza A virus stimulated production of POVPC, PGPC, and PEIPC by human alveolar type II cell line (352). Moreover, accumulation of OxPCs recognized by E06 mAb was observed in the lungs of humans and animals infected with H5N1 avian flu, anthrax, or SARS (153). These data demonstrate that accumulation of OxPLs is characteristic of different types of inflammatory reactions.
Functional importance of OxPLs accumulating during acute inflammation is insufficiently understood. OxPLs both positively and negatively regulate multiple inflammatory reactions and therefore may exert variable and context-dependent effects on the outcome of inflammation. Due to their ability to upregulate inflammatory cell adhesion molecules and chemokines (see Section III.A.1), OxPLs can promote inflammation. Proinflammatory action of IL-6 induced by OxPLs was suggested to play important role in the pathogenesis of acute lung injury induced by viruses, bacteria, and application of acid (see Section III.B.3). In addition, OxPLs inhibit activation of TLRs 2 and 4 by bacterial PAMPs (see Section III.A.7) and thus may interfere with innate immune responses to bacteria. Furthermore, OxPLs were shown to inhibit phagocytosis of bacteria by neutrophils and macrophages, leading to enhanced mortality of animals from abdominal sepsis induced by E. coli (178). Impaired clearance of bacteria can be aggravated by the ability of OxPLs to inhibit oxidative burst in neutrophils and to suppress adaptive immune reactions (see Sections III.A.7 and III.A.9). These deleterious effects characterize OxPLs as potential immunosuppressors impairing various facets of antibacterial defense. On the other hand, inhibition of TLRs and oxidative burst by OxPLs may serve as a negative feedback preventing excessive activation of antibacterial mechanisms in order to avoid damage to host tissue. This hypothesis is supported by several studies showing protective effects of OxPLs under the conditions of acute “sterile” inflammation induced by injection of purified PAMPs (see Section III.A.7). Further studies are required in order to determine factors influencing the balance of pro- and anti-inflammatory activities of OxPLs in vivo.
3. Lung injury
a. Oxidative stress and generation of OxPLs in lungs
The epithelial lining pulmonary surfactant is permanently exposed to high concentrations of oxygen and other oxidants present in the air. Ozone, a common pollutant in urban air, is a highly reactive gas readily attacking double bonds and producing oxidatively truncated PLs (349). PLs (mainly PCs) represent the major component of lung surfactant comprising 80% of its weight. Under physiological conditions, surfactant is protected from oxidation by low contents of PUFAs, antioxidant action of glutathione present in the lining fluid, and by surfactant proteins A and D (185, 187). However, in pathological states oxidation of surfactant PCs, membrane lipids and apoptosis of bronchial cells may result in accumulation of biologically active OxPL products.
Several types of lung injury are accompanied by oxidative stress. Infection of type II pneumocytes with influenza A virus dramatically increased secretion of PAPC oxidation products that induced MCP-1 expression (352). Imai et al. (153) used OxPC-specific E06 antibody to document elevated lung levels of OxPLs in murine models of lung injury caused by H5N1 avian influenza virus and acid instillation. Formation of OxPLs was also observed in inflammatory exudates and alveolar macrophages of H5N 1-infected human patients, anthrax-infected rabbits, and anthrax-, pox- and Yersinia pestis-infected monkeys. Oxidative stress is also characteristic of idiopathic interstitial pneumonia, as illustrated by accumulation of OxPC-positive cells in alveolar spaces of the lungs with desquamative or usual interstitial pneumonia (385). These cells were characterized as macrophages expressing high levels of CD36, suggesting that they play a role in scavenging of OxPLs generated in the inflamed lung (385).
b. Proinflammatory effects of OxPLs in the lungs: SARS, anthrax, H5N1 avian influenza virus, and acid-induced lung injury
Accumulation of OxPLs in the murine models of H5N1 avian influenza virus and acid-induced lung injury (153) was accompanied by increases in lung elastance reflecting compromised lung function. Mechanistic involvement of OxPLs in lung inflammation was demonstrated by inhibitory effect of OxPC-specific E06 antibody on production of cytokines by alveolar macrophages in response to bronchoalveolar lavage fluid (BAL) from injured lungs. Oxidized lung surfactant preparations increased lung elastance and IL-6 production. Similarly, OxPAPC increased lung elastance within an hour after intratracheal application. Based on amelioration of OxPL- and acid-induced increases in lung elastance in il-6, tlr4, and trif knockout mice, the authors proposed that IL-6 induced by OxPLs via the TLR4-TRIF pathway plays a crucial role in deleterious effects of OxPLs on lungs (153).
On the other hand, under certain conditions OxPLs can also exert anti-inflammatory effects and protect pulmonary vascular endothelial barrier. The lung-protective activity of OxPLs is discussed below.
c. Anti-inflammatory and barrier-protective effects of OxPAPC in the models of acute lung injury
Several studies showed that OxPLs protect lung function in animal models of acute lung injury (ALI) induced by application of pure PAMPs such as LPS and CpG DNA. Ma et al. showed that OxPAPC inhibits elevation of TNFα in mice upon intratracheal or systemic administration of LPS or CpG DNA (204). Another work demonstrated that lung inflammation and vascular leak induced by intratracheal LPS aerosol administration were markedly attenuated by OxPAPC (245). These results are in good correlation with and are likely to be explained by the ability of OxPAPC to reverse LPS-induced cytoskeleton remodeling and disruption of monolayer integrity (see Section III.A.8).
In addition to PAMP-induced models of acute lung injury, OxPAPS and OxPAPC markedly reduced lung vascular leak associated with ventilator-induced injury (244). Murine and rat models of ventilator-induced lung injury (VILI) demonstrated that intravenous injection of OxPAPC markedly attenuated protein and inflammatory cell accumulation in bronchoalveolar lavage fluid and lung tissue (244). An in vitro model of VILI showed that high magnitude stretch enhanced endothelial paracellular gap formation induced by thrombin via synergistic effects on RHOGTPase activation. Stretch-induced endothelial barrier disruption was markedly attenuated by OxPAPC and was accompanied by activation of RAC and suppression of RHO (244). Taken together, these findings suggest lung-protective effects of OxPAPC in “aseptic” type of ALI.
d. Summary: Dual effects of OxPLs in lung pathology
The data discussed above show that OxPLs may induce either beneficial or detrimental effects on lungs under different circumstances. Several factors may determine the balance of pro- and anti-inflammatory effects of OxPLs in the lung. First, due to the ability of OxPLs to inhibit inflammatory effects of PAMPs mediated by TLRs 2 and 4 (see Section III.A.7), OxPLs are likely to exhibit anti-inflammatory effects in lung injury induced by a variety of PAMPs from gram-negative and gram-positive bacteria. On the other hand, study by Imai et al. suggests that high OxPL levels may directly or indirectly engage TLR signaling, leading to lung injury progression (153). Second, due to direct effects on lung endothelial permeability, specific OxPLs can suppress lung vascular leak induced by aseptic edemagenic molecules (thrombin) or excessive lung distension during mechanical ventilation. Third, the action of OxPLs on the lungs may depend on their concentrations: whereas low levels of OxPLs protect endothelial barrier, high concentrations of the same OxPLs induce barrier-disruptive effects (28, 78). This biphasic action may be explained by the overwhelming action of barrier-disruptive fragmented and lyso-species at high concentrations of OxPLs (28, 272). Different mechanisms of oxidation produce different proportions of barrier-protective and -disruptive species. For example, oxidation of monounsaturated fatty acids in the lung surfactant by ozone selectively generates barrier-disruptive fragmented OxPLs such as POVPC (see Section III.A.8). In contrast, oxidation of PLs in membranes and lipoproteins by cell-generated free radicals produces a large proportion of esterified isoPs demonstrating prominent barrier-protective effects (28). Therefore, the net pro- vs anti-inflammatory effect of endogenous OxPLs likely depends on the intensity, duration, and mechanism of oxidative stress and efficiency of OxPL clearance since these factors determine total concentrations of OxPLs as well as contents of individual species. Quantitative analysis of systemic and tissue concentrations of molecular OxPL species is required in order to get more insight into the action of OxPLs in the lungs under different inflammatory states.
4. Ischemia
Ischemia and ischemia followed by reperfusion are characterized by massive production of ROS. Several studies demonstrated elevated tissue and systemic levels of OxPLs in response to ischemia/reperfusion. Plasma levels of esterified hydroxides and F2-isoPs were elevated in patients with acute ischemic stroke (188). Systemic and local levels of hydroperoxides of PE and PC were elevated upon ischemia-reperfusion of rat liver (101, 333). In addition, PAF-like (alkyl-acyl) OxPLs were detected within the first minutes after reperfusion of kidneys after warm ischemia (200). Furthermore, plasma concentrations of fragmented OxPCs were increased in patients during the reperfusion period after coronary surgery with cardiopulmonary bypass (97). Stroke induced by ligation of the middle cerebral artery in mice resulted in accumulation of enhanced levels of furan-PCs in infarcted brain tissue (105). Furan-PCs accumulated in ischemic hemisphere but not in contralateral control hemisphere (105). Myocardial ischemia-reperfusion models demonstrated elevated levels of PC-hydroperoxides and oxidatively fragmented PCs in the heart ventricles 6 hours after reperfusion that was preceded by 30 minutes of coronary artery occlusion (235). In addition, oxidation of mitochondrial CL was stimulated by ischemia-reperfusion in aged rat hearts (194).
In summary, accumulated data show that ischemia/reperfusion is a pathological state characterized by elevated local and circulating levels of OxPLs. In some of these studies, amelioration of reperfusion-induced symptoms was achieved by application of antioxidants (200), suggesting that oxidized lipids and OxPLs in particular may play a role in the pathogenesis of this condition.
5. Light- and radiation-induced stress
Formation of OxPLs can be stimulated by visual and UV-light. Several oxidatively fragmented OxPEs including sn-2-succinyl-, sn-2-(9-oxo) nonanoyl-, and sn-2-(4-oxo)butaroyl-PE were found in rat retina (128). Furthermore, acute exposure of rats to green light induced accumulation in the retinas of saturated and unsaturated fragmented OxPCs that are ligands for scavenger receptor CD36 (331). OxPLs accumulating in retinas serve as ligands for CD36-dependent phagocytosis of shed photoreceptor outer segments by retinal pigment epithelium; this process is necessary for normal function of the retina (331).
Generation of OxPLs induced by light exposure was also demonstrated in skin cells. UVA-1 ultraviolet radiation (340–390 nm) stimulated accumulation of E06-reactive OxPCs in dermal fibroblasts via singlet oxygen-dependent mechanism (125). UVA-1-irradiated PAPC containing several OxPL species induced expression of antioxidant and anti-inflammatory enzyme heme oxygenase-1 in dermal fibroblasts, keratinocytes, and in a three-dimensional epidermal equivalent model (125). Thus, OxPLs are likely to play a protective role in UVA-irradiated skin by inducing HO-1. In addition, UVB radiation was shown to stimulate production in skin of PAF and PAF-like (alkyl-acyl) OxPLs that are likely to play a role in UVB-induced hyperalgesia (390). Furthermore, UVB irradiation of cultured epidermal cells stimulated production of fragmented alkyl-PCs such as hexadecyl-azelaoyl-PC that is a ligand for PPARγ (391). PAF-like (alkyl-acyl) lipids generated in response to irradiation promoted in a PPARγ-dependent manner expression of cyclooxygenase-2 and production of PGE2—events potentially relevant to UVB-induced immuno-suppression (391).
Tissue injury induced by gamma-irradiation was shown to induce oxidation of PLs. The analysis of OxPLs in small intestine upon whole body gamma-irradiation of mice demonstrated selective oxidation of CL in mitochondria and PS outside of mitochondria, leading to formation of several hydroperoxides of PS and CL. The authors hypothesized that elevated levels of these OxPLs may result from radiation-induced apoptosis, which is characterized by selective oxidation of CL and PS (347).
6. Leprosy
Oxidized PCs recognized by E06 mAb were detected in lepromatous (disseminated) leprosy lesions, but not in tuberculous leprosy characterized by stronger host immune response and self-contained infection (70). OxPLs mainly accumulated in macrophage-derived foam cells. Analysis by mass spectrometry confirmed elevated levels of isoP-containing PEIPC in mycobacteria-infected human macrophages (70).
In order to demonstrate functional importance of OxPLs accumulated in leprosy lesions, the effects of PEIPC on innate immune responses were analyzed. It was shown that PEIPC prevented upregulation on differentiating dendritic cells of CD1b, which is important for presentation of lipid antigens to T cells (70). The effect was accompanied by impaired activation of M. leprae-reactive CD1b-restricted T cells. Furthermore, OxPLs modulated the activity of TLR2/1 known to recognize mycobacterial triacylated lipoproteins. PEIPC inhibited secretion of proinflammatory IL-12, but stimulated production of anti-inflammatory IL-10 induced by TLR2/1 ligand. Furthermore, PEIPC inhibited TLR2/1-induced production of antimicrobial peptide cathelicidin, known to protect human monocytes and macrophages against myco-bacteria (70).
In summary, lepromatous leprosy lesions are characterized by accumulation of OxPLs, which can counteract innate and specific immune responses to mycobacteria and thus promote their survival.
7. Multiple sclerosis
Multiple sclerosis (MS) is an autoimmune disease of the brain, which targets the myelin sheath of the central nervous system and results in neurodegeneration. A number of potential culprit protein antigens have been identified, but there is also increasing evidence that lipid-specific autoimmune responses are equally involved. With more than 70%, lipids comprise a major part of myelin sheaths. Kanter et al. have tested this hypothesis by analyzing cerebrospinal fluids of MS patients for the presence of antibodies to specific lipids using a newly developed large-scale lipid microarray (167). Antibodies in MS serum samples showed strong reactivity to various lipids, such as sulfatide, sphingomyelin, and PLs, but also to three forms of OxPC (PAzPC, PONPC, and PGPC). While the authors show that sulfatide-specific antibodies can worsen disease in mice in the experimental allergic encephalitis (EAE) model, the pathogenic role of antibodies to OxPLs is not clear. Nevertheless, the importance of OxPLs in MS was further supported in a later report by Qin et al., who demonstrated the presence of OxPLs (alone and conjugated to a 15 kDa protein) in extracts of MS lesions directly by Western blot analyses using the E06 antibody (273). Interestingly, the authors also demonstrated the presence of natural E06 antibodies themselves using an anti-idiotypic Ab that specifically recognizes the idiotope of E06/T15. Thus, both OxPLs and OxPL-reactive antibodies are found in plaques of MS patients and mouse models. Likely, OxPLs promote the inflammatory process in MS lesions, whereas the role of specific anti-OxPL antibodies is more complex and requires further evaluation.
IV. OxPLs as Biomarkers of Disease and Targets for Therapy
A. Association of OxPL levels with disease
1. Methods of quantification of OxPLs
Quantification of OxPLs in vivo is a challenging analytical task since OxPLs are significantly more variable as compared to unoxidized precursors (see Fig. 2), and each of multiple oxidized species is present in vivo in significantly lower amounts than precursor PL (Table 4). OxPLs can be detected using specific antibodies (337a) or a combination of HPLC with reagents specific for oxidized groups (e.g., carbonyls or hydroperoxides) (97, 101, 109). However, the most universal and sensitive method for quantification of OxPLs is mass spectrometry. Since PLs are thermolabile and nonvolatile molecules, their analysis is performed using soft ionization techniques such as electrospray ionization (ESI) and matrix-assisted laser desorption ionization (MALDI). Complex biological samples such as animal tissues and blood plasma contain multiple molecules having mass-to-charge (m/z) ratios very close to OxPLs, so-called isobaric compounds. As a result, OxPLs cannot be detected in such samples by the routine methods of “shot-gun” lipidomics. Typically, reliable identification of OxPLs requires separation of isobaric lipids by high-performance liquid chromatography followed by tandem mass spectrometry. Each molecular species is characterized by a “signature” consisting of its m/z value, chromatography retention time, and m/z value(s) of its characteristic degradation product(s). Detailed description of mass spectrometry of OxPLs can be found in the review of Domingues et al. (81).
OxPLs were detected in a variety of normal human and animal tissues, and their elevated levels were found in different pathological states (Table 4). High levels of OxPLs reaching 1% of precursor PL were observed in plasma and arterial wall in patients and experimental animals with hypercholesterolemia and atherosclerosis. Normalization of OxPL amounts to the volume of plasma or wet weight of tissue showed that under physiological conditions individual species of OxPLs are either nondetectable, or present at low concentrations (Table 4). However, in pathology their concentrations can reach the levels of micromoles or even tens of micromoles per liter. Importantly, the majority of biological effects of OxPLs that were detected in vitro (e.g., monocyte binding to ECs, production of IL-8, induction of electrophilic genes, activation of UPR, the angiogenic switch of ECs, inhibition of LPS effects, enhancement of endothelial barrier, etc.) are also observed in the range of micromoles to tens of micromoles per liter, and on the other hand these effects are induced by different classes and molecular species of OxPLs. Therefore, combined concentration of active species, rather than their individual levels, should be taken into account. Furthermore, Table 4 mainly summarizes data on OxPLs generated from palmitoyl-arachidonoyl- and palmitoyl-linoleoyl-PC, while other molecular species and classes of OxPLs, which are likely to have similar biological activities, were not analyzed. Thus, concentrations of OxPLs detected in certain pathologies are close to those inducing biological effects in vitro, and are likely to be of biological importance.
Which of multiple species of OxPLs is the most convenient and informative marker of general oxidative stress, PL oxidation, and disease is currently under investigation. Two groups of OxPLs look promising due to their relatively long half-life as compared to other species that are rapidly reduced or cleaved by phospholipases and other enzymes. The measurement of isolevuglandin/protein adducts generated after cleavage of PL–isolevuglandin/protein complexes by PLA2 may be preferred as compared to quantification of isoPs due to slower clearance of protein adducts from the circulation (71). It was suggested that isolevuglandin/protein complexes may serve as integral markers of oxidative stress (266). Significantly elevated levels of isolevuglandin–protein adducts were found under different pathological conditions, including atherosclerosis (392), renal disease (292), Alzheimer disease (387), mouse sepsis model (266), and glaucoma (120). In addition to isolevuglandin–protein adducts, also isofuran–PLs are relatively stable and therefore can be used as conventional biomarkers of lipid oxidation. Elevated levels of PL-isofurans were found in a number of human and experimental pathologies, including hyperoxia-induced lung injury (94), Parkinson disease (93), and a mouse model of Alzheimer disease (314). Although these data characterize isolevuglandins and isofurans as potential biomarkers of disease, clinical relevance of their quantification is insufficiently studied and requires further analysis.
The most convincing data characterizing OxPLs as potential disease biomarkers were obtained in clinical studies using an ELISA test (343) based on detection of OxPCs (but not OxPLs containing other polar head groups) by mAb E06 that specifically recognizes phosphocholine group present in oxidized PCs. The mAb also binds to bacterial products such as capsular polysaccharide of Streptococcus pneumoniae but does not interact with the phosphocholine group present in unoxidized choline-containing PLs (303). E06 recognizes a variety of, but not all, oxidized species of PC, including polymerized species and protein adducts (98), and thus may serve as an integral marker of PC oxidation. Drawbacks of E06 represent the opposite side of its advantages, including complex and still not exhaustively characterized antigen specificity, cross-reactivity with bacterial wall antigens, as well as the lack of reactivity with other classes of OxPLs (98). In spite of these limitations, clinical studies reproducibly showed that E06 reactivity normalized to apoB-100 levels (OxPL/apoB-100 ratio) demonstrates good correlation with the presence, extent, and prognosis of cardiovascular disease. Clinical studies on ∼10,000 individuals showed that OxPL/apoB-100 levels are elevated in patients with coronary, carotid, and femoral atherosclerosis, during acute coronary syndrome and after percutaneous coronary intervention (338, 340, 341). Furthermore, increased levels of OxPL/apoB-100 predict myocardial infarction, stroke, and cardiovascular death (175). Importantly, the prediction of cardiovascular events by OxPL/apoB-100 levels was independent of Framingham atherosclerosis risk factors (175). In addition, OxPL/apoB-100 levels were increased in patients treated with low-fat diet or statins, raising the possibility that OxPL/apoB-100 ratio may serve as a biomarker of efficiency of hypolipidemic therapy and removal of atherogenic OxPLs from the vessel wall (95, 186, 284, 308).
It was found that values of OxPL/apoB-100 demonstrated high and reproducible correlation with the levels of Lp(a) and especially its forms containing the lowest number of kringle IV type 2 repeats (340). This correlation is likely due to the fact that the vast majority of E06-reactive lipids in circulation is bound to the lipid phase and apo(a) component of Lp(a) particles (17) (see Section I.E). Taking into account close correlation between OxPL/apoB-100 and Lp(a) levels, further clinical studies are required in order to establish which additional diagnostic information can be provided by quantification of OxPL/apoB-100 using E06 antibody as compared to existing Lp(a) tests.
B. Experimental therapies involving inactivation of OxPLs
Identification of important pathological activities of OxPLs raises a question whether inactivation or removal of such lipids could be used for therapy. Currently, there are no approaches selectively targeting OxPLs in vivo. However, several experimental therapies in parallel to other beneficial mechanisms also interfere with the generation or action of OxPLs.
1. HDL and apolipoprotein peptide mimetics
HDL and its mimetics are regarded as potential therapeutic agents against atherosclerosis (373). In addition to the reverse cholesterol transport, beneficial effects of HDL particles may be explained by multiple mechanisms including inactivation of OxPLs by HDL-associated PAF-AH and LCAT (121, 369). Apart from enhancing degradation of OxPLs, HDL can antagonize their effects by decreasing sensitivity of cells. Pretreatment of aortic ECs with HDL was shown to inhibit activation of several signaling pathways and reduce induction of chemotactic activity and monocyte binding in response to OxPLs (115). At the same time, HDL did not inhibit induction of antioxidant genes (115). The authors hypothesized that the protective effects of HDL may be partially explained by stabilization of caveoli and inhibition of OxPAPC-induced production of superoxide radicals (115).
Interestingly, the ability of HDL to inhibit formation and biological activity of OxPLs may be compromised as a result of disease. The loss of protective properties of HDL was observed in patients with coronary disease, diabetes, and lepromatous leprosy, inter alia (70, 215, 239). Quantification of the anti-OxPL activity of HDL was suggested as a clinical test for prediction of atherosclerosis (239).
Anti-inflammatory and anti-atherogenic effects of HDL can be mimicked by short peptide fragments of apoA-I or apoJ (238). An advantage of short peptides as compared to HDL or apoA-I to be used as therapeutic agents is that they can be applied per os. Such peptides that were chemically modified in order to increase stability in vivo, were shown to protect vessels in animal models of atherosclerosis, diabetes, and other diseases (236). Moreover, the reversal of atherosclerosis was achieved by combined treatment of apoE knockout mice with apoA-I peptide mimetics and statins (237). Anti-atherosclerotic activity of apoA-I peptide mimetics is based on multiple mechanisms including direct binding of OxPLs. Interaction of most active peptide mimetics with OxPLs was characterized by very high affinity that was several orders of magnitude higher as compared to the interaction with apoA-I (351). This binding is likely to inhibit biological effects of OxPLs as suggested by a correlation between the ability of different apoA-I mimetic peptides to bind OxPLs and inhibit development of atherosclerosis in mice (351).
In summary, inactivation of OxPLs is a recently identified mechanism likely to play a role in the anti-atherogenic and anti-inflammatory activity of HDL independently of reverse cholesterol transport. OxPL-binding peptide mimetics of apoA-I are promising pharmacological agents for prophylactics and treatment of atherosclerosis.
2. CS-1 fibronectin peptide mimetics
An alternative peptide-based approach used synthetic peptide fragments of CS-1 fibronectin (CS-1 FN). CS-1 FN mediates VLA-4-dependent binding of monocytes to OxPL-treated ECs (see Section III.A.1). It was shown that long-term infusion of CS-1 peptide mimetic applied using osmotic pump inhibited monocyte accumulation and fatty streak formation in aortic sinus of LDL receptor knockout mice (305). This study provided a proof-of-principle that peptide mimetics can be used for inhibition of OxPL-induced CS-1 FN-dependent monocytic inflammation.
3. Immunization against OxPLs: Vaccine against atherosclerosis?
Immunization studies of atherosclerosis-prone rabbits and mice with models of autologous OxLDL have demonstrated that OxLDL-specific immune responses are accompanied by atheroprotective effects, as all these interventions led to decreased lesion formation (3, 23, 96, 111, 243, 252, 395). Interestingly, vaccination with adjuvants alone, without antigen, also provides protection against atherosclerosis, which may result from normalization of lipoprotein profile, stimulation of Th2 responses or increased serum titers of antibodies to MDA-LDL (174). However, as discussed below, induction of specific response to OxLDL or its components is much more effective in preventing vessel disease. The exact mechanisms by which immunization with OxLDL is atheroprotective are still unknown, which is in part due to the heterogeneity of the model antigens used, which make the analyses of the induced responses more difficult to interpret. In this regard, we have focused on the role of phosphocholine-specific immune responses, as phosphocholine-specific antibodies constitute a part of anti-OxLDL antibodies.To test this, the molecular mimicry between phosphocholine groups present on the surface of pneumococci and phosphocholine present in OxPC was exploited. Cholesterol-fed ldlr −/− mice were immunized with phosphocholine-containing pneumococcal extracts. This immunization led to a near monoclonal expansion of E06/T15 idiotypic IgM antibodies, but not IgG antibodies to OxLDL or phosphocholine, and significantly decreased lesion formation (24). Thus, expansion of the phosphocholine-specific IgM E06, which specifically binds OxPL, provided atheroprotection. The atheroprotective effect of E06/T1 5 was further corroborated in a study by Faria–Neto et al., in which the authors showed that passive transfusion of purified T15 idiotypic IgM decreased lesion formation in a carotid artery vein graft model in apoe −/− mice (87). There are multiple potential mechanisms by which E06/T15 can mediate atheroprotection (25). E06 IgM has been shown to block the uptake and degradation of OxLDL by macrophages which in vivo would prevent foam cell formation (141). In addition, E06/T15 may neutralize the many proinflammatory effects of OxPLs, as has been shown for the activation of ECs in response to apoptotic cells (56) and blebs (151) carrying OxPLs, as well as the production of IL- 6 by macrophages stimulated with OxPLs (153). Many of these effects may be dependent on the isotype of the induced antibodies, as potential pro-inflammatory effects mediated by binding to Fcγ receptors are absent with IgM Abs. Nevertheless, Caligiuri et al. have followed another strategy to induce phosphocholine-specific antibodies by immunization of ldlr −/− mice with phosphocholine conjugated to KLH (52). In their study the immunization led to development of both IgG and IgM Abs specific for phosphocholine and OxLDL, and mice still developed less atherosclerosis. The contribution of the induced IgG antibodies in this study is unclear, as the IgG subclasses, which have different affinities for Fcγ receptors and different Fcγ-mediated effects, were not differentiated. Humans also have IgM and mostly IgG2 antibodies against phosphocholine, which exhibit cross-reactivity to OxLDL (24, 296). However, their role in cardiovascular disease is unknown. Identification of the protective subset of anti-phosphocholine antibodies and development of strategies to selectively induce them, may enable the future development of vaccines against atherosclerosis.
C. OxPLs as targets for imaging techniques
Advanced atherosclerotic lesions are rich in oxidized lipoproteins, thus suggesting that antibodies specific for oxidized epitopes can be used for detection of plaques in vivo. Indeed, several studies described selective accumulation of radioactively labeled antibodies specific for malondialdehyde-LDL in atherosclerotic lesions of rabbits and mice (304, 342). Recent studies showed that in addition to MDA-LDL-specific antibodies, also E06 mAb, which recognizes a variety of OxPCs, selectively interacted with arteria of hypercholesterolemic apoe −/− mice. Magnetic resonance imaging was performed after injection of antibodies coupled to micelles containing gadolinium (49). The study demonstrated significant and specific enhancement of signal in atherosclerotic vessels of apoe −/− but not wild-type mice, suggesting that OxPL-specific antibodies may be used for noninvasive detection and quantification of atherosclerosis.
V. Open Questions and Perspectives
It is increasingly recognized that OxPLs are markers of disease of the “modified-self” type that are recognized by several PRRs including membrane receptors (e.g., CD36) and soluble proteins such as CRP, CD14, and natural antibodies. OxPLs can be easily distinguished from normal membrane PLs since truncated oxidized residues protrude into the water phase where they have spatial access to PRRs. Since lipid oxidation in vivo is strictly controlled, and on the other hand oxidized molecules are rapidly eliminated, accumulation of OxPLs in cell membranes and lipoproteins (formation of “lipid whiskers” (122)) may be a universal marker of senescence and disease, processes that typically are accompanied by enhanced production of ROS and impaired antioxidant defense. Importantly, recognition of OxPLs by PRRs and other receptors not only targets OxPL-containing structures for elimination, but also modulates cellular functions.
A common theme in this review is that OxPLs often exert functionally different, and even opposite, effects on various steps within one functional pathway. Examples are pro- and anti-inflammatory, pro- and anti-angiogenic, and pro- and antioxidant effects of OxPLs. Apparently, the net outcome depends on the biological context. One explanation for variability of biological effects of OxPLs is activation of several receptor-mediated and nonreceptor signaling mechanisms. In this respect, OxPLs are analogous to the family of prostanoids known to act via multiple plasma membrane and nuclear receptors, and also capable of inducing electrophilic and unfolded protein stress responses.
The data on biological activities of OxPLs were mainly obtained in vitro after treatment of cells with artificially oxidized pure lipids that were added as emulsions. Therefore, physical state of OxPLs was different from that in lipoproteins and cell membranes. However, available evidence suggests that published effects of pure OxPLs adequately mimic the action of their more physiological forms. As mentioned in corresponding sections, the majority of biological effects of OxPLs were reproduced using OxLDL or its more physiological form MM-LDL. Furthermore, apoptotitc blebs and apoptotic cells were shown to induce proinflammatory effects similar with those of pure OxPLs (56, 151). In addition, a close parallel exists between induction of several genes by OxPLs in vitro, and enhanced expression of these genes in atherosclerotic lesions known to contain high levels of OxPLs (Section III.B.1). In summary, accumulating evidence supports the notion that biological activities of OxPLs are not artifacts of in vitro experimentation but correclty reflect their effects in vivo. The influence of the physical state of OxPLs on their biological activity deserves further investigation because it may give a clue to the mechanisms initiating cellular responses to OxPLs.
It has to be established to which extent pleiotropic effects induced by OxPLs depend on their structural variability. The data on the structure–function relationship show that while some activities are specific for molecular species containing certain chemical groups (e.g., activation of PAF receptor by PAF-like OxPCs, induction of electrophilic stress, enhancement versus disruption of endothelial barrier, and activation of prostaglandin receptors), other important effects such as monocyte adhesion and induction of IL-8 in ECs, inhibition of LPS effects or stimulation of unfolded protein response are induced by essentially all studied molecular species of OxPLs. Therefore, further analysis of structure–function relationships in combination with quantification of individual and combined species of OxPLs is required in order to understand the biological role of these lipid mediators.
Significant complexity of signaling mechanisms activated by OxPLs hampers evaluation of their role in vivo. Variable chemical structures of OxPLs, enzymatic and nonenzymatic mechanisms of their generation and inactivation, multiple receptors and nonreceptor mechanisms mediating action of OxPLs together dictate the necessity of using a broad panel of gene knockout and knockdown models, as well as enzyme inhibitors and receptor antagonists. The progress in understanding the role of OxPLs in vivo may be promoted by the success of novel approaches to modulation of effects of OxPL (see Section IV.B), which potentially may evolve into the methods of experimental therapy. In addition, potential therapeutic applications of OxPLs such as inhibition of TLR2 and 4 and protection of lung vessel endothelial barrier deserve further validation. Since effects of the same OxPL may be pathologic in one setting (e.g., induction of proinflammatory changes in ECs) and beneficial in another setting (protection from lung edema during acute aceptic inflammation), possible use of OxPLs must be considered in relation to particular pathology. Last but not least, OxPLs are increasingly recognized as promising diagnostic markers.
In summary, OxPLs represent an emerging family of lipid mediators. Analysis of gene expression in OxPL-treated cells identified very high number of regulated genes (107, 108), suggesting that our current knowledge on OxPLs is just the tip of the iceberg, and that future studies will discover new biological activities of OxPLs and improve our understanding of the role of OxPLs in health and disease.
Footnotes
Acknowledgments
This work was supported by grants from Fonds zur Förderung wissenschaftlicher Forschung (FWF, projects P20801-B11 and P18232-B11 to VNB) and Österreichischer Forschungsförderungsgesellschaft (project 815445 to VNB).
Abbreviations Used
Reviewing Editors: Judith Berliner, Denis Blache, Clett Erridge, Valerian Kagan, Vishwanathan Natarajan, Sampath Parthasarathy, Yulia Tyurina, and Fulvio Ursini
