Abstract
The epidemiology of human immunodeficiency virus (HIV) has shifted such that Black individuals disproportionately represent incident HIV diagnoses. While risk of end-stage kidney disease (ESKD) among people with HIV (PWH) has declined with effective antiretroviral therapies, a substantial racial disparity in ESKD burden exists with the greatest prevalence among Black PWH. Disparities in waitlisting for kidney transplantation, the optimal treatment for ESKD, exist for both PWH and Black individuals without HIV, but it is unknown whether these characteristics together exacerbate such disparities. Six hundred two thousand six ESKD patients were identified from the United States Renal Data System (January 1, 2007 to December 31, 2017), and HIV-status was determined through Medicare claims. Cox proportional hazards regression was used to determine waitlisting rates. Multiplicative interaction terms between HIV-status and race were examined. The 6250 PWH were significantly younger, more commonly Black, and less commonly female than those without HIV. HIV-status and race were independently associated with 50% and 12% lower likelihood of waitlisting, respectively [adjusted hazard ratio (aHR): 0.50, 95% confidence interval (CI): 0.36–0.69, p < 0.001; aHR: 0.88, 95% CI: 0.87–0.90, p < 0.001]. There was also a significant interaction present between HIV-status and Black race (aHR: 0.80, 95% CI: 0.66–0.98, p < 0.001) such that, while HIV-status and Black race were independently associated with decreased waitlisting, the interaction of Black race and HIV-status exacerbated those disparities. While limited by lack of HIV-specific data that may impact inferences with respect to race, additional studies are urgently needed to understand the interplay between HIV risk factors, HIV-stigma, and racism, and how intersectionality may exacerbate disparities in transplantation among PWH.
Introduction
The prognosis of a human immunodeficiency virus (HIV) diagnosis has evolved following the introduction of highly effective antiretroviral therapies (ART), yielding life expectancies among select people with HIV (PWH) who are comparable to those experienced by people without HIV. 1 Despite this progress, multiple studies have demonstrated that PWH remain at heightened risk of chronic comorbidities such as end-stage kidney disease (ESKD) compared to individuals without HIV, which is frequently attributed to HIV-specific characteristics such as nephrotoxic ART regimens, low CD4 counts, and HIV viremia and to health behaviors such as smoking and substance misuse. 2 –4 ESKD is also more prevalent among Black individuals independent of HIV-status, 5 and data suggest that the presence of an HIV diagnosis among Black individuals increases the risk of ESKD compared to Black individuals without HIV or White PWH. 6 –9
While the number of new HIV infections in the United States declined from 2015 to 2019, Black individuals still experience eightfold higher rates of new HIV diagnoses than White individuals. 10 Consequently, given the elevated risk of ESKD development among Black PWH, greater understanding of ESKD outcomes among PWH is needed.
ESKD is a substantial contributor to morbidity and mortality in the United States with over 800,000 individuals either on dialysis or the recipient of a kidney transplant in 2020. It also imposes a significant financial burden on the Medicare system generating 50 billion dollars in expenditures during 2020. 5 Kidney transplantation is the preferred treatment option for ESKD as it affords significantly higher quality of life and life expectancy compared to dialysis, 11,12 and transplantation is cost-effective compared to dialysis. 13 Despite this, given the high demand for transplantation and an inadequate supply of available donor organs, disparities in transplantation exist. 14,15 While racial disparities in transplantation have been thoroughly documented, 16 PWH are also subject to such disparities. PWH were historically denied transplantation due to concerns surrounding patient outcomes in the setting of intense immunosuppression. 17
Following numerous studies documenting excellent outcomes of kidney transplantation among PWH, 18 –20 this practice has increased 10-fold since 2003. However, to be eligible for kidney transplantation, PWH must meet both standard eligibility criteria and HIV-specific eligibility criteria, including undetectable HIV viral loads, stable ART regimens, lack of opportunistic infections, CD4 count >200, and, if diagnosed with hepatitis B virus (HBV) or hepatitis C virus (HCV), have no indication of present or likely fibrosis. 21,22 Moreover, despite evidence that transplantation affords a significant survival benefit for PWH over remaining on dialysis, 23 17% of kidney transplant centers still consider an HIV diagnosis an absolute contraindication to transplantation. 24 Thus, while the United Network for Organ Sharing (UNOS) does not discriminate based upon HIV-status, numerous studies have documented lower rates of waitlist addition and transplantation for PWH compared to ESKD patients without HIV. 25 –31
The HOPE Act was passed in 2013 with the goals of addressing the observed inequity in transplantation of PWH and reducing anti-HIV stigma in transplantation through utilization of organs from donors with HIV. 32 Despite projected high volumes of transplants, 33 the implementation of this practice has been limited to date. Given promising patient outcomes, 34 the Advisory Committee on Blood & Tissue Safety & Availability voted in November 2022 to recommend the removal of the requirement that these transplants be performed under research protocols, 35 which may further promote equity in transplantation for PWH.
However, understanding access to the waitlist for PWH, a requisite first step for deceased donor transplantation, is critical. Moreover, given the disproportionate representation of Black PWH among the ESKD population, identifying whether there is a significant exacerbation of known disparities for Black PWH is essential for both individual health outcomes and health equity. While limited in the ability to examine HIV-specific characteristics, we leveraged the US Renal Data System (USRDS) to examine waitlist addition rates among PWH compared to counterparts without HIV and, specifically, determined whether known racial disparities in access to the kidney transplant waitlist were exacerbated in the presence of an HIV diagnosis. We hypothesized that Black PWH would have lower likelihood of waitlist addition than White PWH or Black individuals without HIV.
Methods
Study population
This study utilized data from the United States Renal Data System, 36 a national database capturing data on all ESKD patients in the United States and their accompanying Medicare claims. The Institutional Review Board at the University of Alabama at Birmingham (IRB-161212003) approved this study. ESKD patients in this study were ≥18 years of age at time of dialysis initiation, first initiated dialysis between January 1, 2007 and December 31, 2017, were not listed for transplant nor did they receive a living kidney donor transplant before dialysis initiation, did not die nor were they removed from dialysis within 90 days of dialysis initiation, and had Medicare as their insurance payer.
Exposure of interest
HIV-status was defined using the Chronic Conditions Data Warehouse definition, requiring one HIV-specific inpatient claim or two outpatient claims within a 2-year period. 37 Race, sex, ethnicity, and geographic location were captured on the CMS 2728 form, which must be completed when a new ESKD patient initiates dialysis. While the practice of transplanting PWH has rapidly expanded, HIV-specific criteria for waitlist addition and transplantation exist. 38 Specifically, PWH who have active AIDS or HIV viremia will not be transplanted given the necessary immunosuppressive regimens needed to maintain a kidney transplant. Moreover, PWH must maintain an undetectable viral load, CD count >200, adherence to a stable ART regimen provided that they are not an elite controller, have no active opportunistic infections, and, if HBV or HCV-positive, have no indication of present or likely fibrosis. 22
The limitations of Medicare claims to distinguish between HIV and AIDS are well documented, but the Charlson comorbidity index recommends that PWH with evidence of HIV infection and a claim for an opportunistic infection may be classified as having AIDS. 39 While not fully encapsulating all HIV-specific waitlist criteria, any PWH with a claim for opportunistic infection within 90 days of dialysis initiation was excluded to create a more select cohort of PWH. 39
Demographics
Age, sex, race, ethnicity, clinical characteristics, and geographic location were assessed at time of ESKD diagnosis. Characteristics were compared by HIV-status using Wilcoxon ranked sum tests and chi-square tests of independence for continuous and categorical variables, respectively. Given the size of the cohort, all comparisons were statistically significant. To permit discernment of meaningful differences, standardized differences were calculated using a threshold of <0.1 to delineate between meaningful and less consequential differences in any given characteristic. 40
Method of analysis
The analyses conducted were time-to-event analyses using Cox proportional hazards regressions. ESKD patients entered the study and began contributing time-at-risk on the date of dialysis initiation. They were then followed until the earliest of waitlist addition, death, living donor transplant before waitlist addition, or administrative end of study (December 31, 2018). Those ESKD patients experiencing any outcome other than waitlist addition were censored. All models contained fixed effects for ESKD network and calendar year. 41 –43 The fixed effects for ESKD network were incorporated to account for variations in waitlist addition practices, 44,45 and the fixed effects for calendar year incorporated to account for increasing effectiveness of care for PWH and increasing comfort with transplantation of PWH. 46 Robust standard errors were used at the dialysis center-level.
Interaction analysis
To assess for potential effect modification, an interaction term between HIV-status and individual race was examined. First, the interaction term was assessed in a univariable model. When significant at p < 0.10, a multivariable model was built adjusting for potential confounders. The interaction analysis consisted of three relevant measures of association, the two main effects and the multiplicative interaction term between the two characteristics, represented as follows: β1 for HIV-status, β2 for a dummy indicator for Black race, and β3 for the interaction between the two characteristics. The listed β terms were then summed and exponentiated to allow for meaningful comparisons between groups. 47 A three-way interaction term between HIV-status, race, and sex was also examined.
Sensitivity analyses
As comfort in transplantation of PWH increased throughout the study period, support for waitlist addition among PWH may have correspondingly increased throughout the study period, leading to an overestimation of the disparity in waitlist addition. While calendar year was included as a covariate in our analysis to account for these secular trends, full follow-up time was used to confirm our findings of lower waitlist addition rates. In addition, the initial cause for Medicare enrollment may contribute to some of the observed association. For example, an ESKD patient who first enrolled in Medicare due to aging may perhaps be less likely to be added to the waitlist than an ESKD patient who first enrolled in Medicare due to their ESKD diagnosis as older patients are less likely to be added the transplant waitlist. 15 Consequently, waitlist addition rates may differ by initial reason for enrollment in Medicare.
Thus, analyses restricted to ESKD patients who first enrolled in Medicare due to their ESKD diagnosis were performed. Given lower waitlist addition rates and lower retention in HIV care, 48,49 interaction terms between HIV-status and individual sex, ethnicity, and rurality were examined for potential significance and for confounding of our inferences. These interactions, however, did not meet statistical significance after adjustment for other critical individual and center characteristics. Finally, to further interrogate whether increased comfort with transplantation of PWH might confound our inferences, we replicated our analyses in a cohort of prevalent ESKD patients as of January 1, 2012. These data indicated that, while the disparities observed in our primary analyses were reduced, they were not completely ameliorated and remained both statistically significant and clinically meaningful. All analyses were replicated in a cohort of PWH inclusive of those with evidence of opportunistic infection.
All analyses were conducted in SAS 9.4 (Cary, NC) and Stata 17.0 (College Station, TX). Statistical significance was defined as a two-sided p < 0.05.
Results
Cohort characteristics
There were 595,756 ESKD patients without HIV and 6250 PWH with ESKD. There were substantial differences between the two groups. PWH were significantly younger with a median age of 51 compared to 65 among those without HIV [PWH: 51, interquartile range (IQR): 43–60 vs. HIV−: 65, IQR: 55–75, p < 0.001, standard deviation (SD): −0.99] (Table 1). 70.0% of PWH were Black in contrast to 26.7% of ESKD patients without HIV (p < 0.001, SD: 0.96). PWH were less commonly female (31.7% vs. 43.4%, p < 0.001, SD: −0.24). Diabetes and body mass index of 30–34.9 kg/m2 were less prevalent among PWH (25.4% vs. 40.4%, p < 0.001, SD: −0.32; 13.9% vs. 19.1%, p < 0.001, SD: −0.14). Reported alcohol dependence and drug dependence were higher among PWH than those without HIV (3.0% vs. 1.4%, p < 0.001, SD: 0.11; 7.8% vs. 1.0%, p < 0.001, SD: 0.34).
Patient-Level Characteristics by Human Immunodeficiency Virus Status at Time of End-Stage Kidney Disease Diagnosis
BMI, body mass index; ESKD, end-stage kidney disease; HIV, human immunodeficiency virus; Std. diff., standardized difference.
Unadjusted analyses
The reference group to which all groups were compared was White individuals without HIV. Before any adjustment, the main effect for Black race compared to White race was significantly associated with 23% higher likelihood of waitlist addition [hazard ratio (HR): 1.23, 95% confidence interval (CI): 1.21–1.24, p < 0.001] (Table 2). In contrast, the main effect for positive HIV-status had no significant association with waitlist addition compared to negative HIV-status (HR: 1.07, 95% CI: 0.93–1.24, p = 0.76). The interaction between HIV and Black race was negative and significantly associated with reduced likelihood of waitlist addition (HR: 0.71, 95% CI: 0.60–0.85, p < 0.001), suggesting the combination of Black race and positive HIV-status was associated with lower likelihood of waitlist addition beyond what would be expected if HIV and Black race acted independently.
Unadjusted Likelihood of Waitlist Addition Interactions Between Human Immunodeficiency Virus and Patient-Level Characteristics
Bold indicates significance at p < 0.05.
CI, confidence interval; HIV, human immunodeficiency virus; HR, hazard ratio; PWH, people with HIV; Ref., reference.
Summing the three betas for the main effect of Black race, HIV-status, and the interaction between Black race and HIV-status permitted comparison of Black PWH to White individuals without HIV and suggested no significant difference in likelihood of waitlist addition (HR: 0.94, 95% CI: 0.86–1.03, p = 0.70). To estimate the effect of HIV among Black individuals, the betas from the main effects for HIV-status and race and the interaction term were further combined, demonstrating positive HIV-status among Black ESKD patients was significantly associated with 23% lower likelihood of waitlist addition (HR: 0.77, 95% CI: 0.70–0.84, p < 0.001), further demonstrating the association of HIV-status with waitlist addition was greater among Black individuals than among White individuals.
Adjusted analyses
After adjustment for demographics, clinical characteristics, and geography, the main effect for Black race was significantly associated with 12% lower likelihood of waitlist addition [adjusted hazard ratio (aHR): 0.88, 95% CI: 0.87–0.90, p < 0.001] (Table 3). In contrast to unadjusted analyses, the main effect for HIV infection was associated with 50% lower likelihood of waitlist addition (aHR: 0.50, 95% CI: 0.36–0.69, p < 0.001). The multiplicative interaction between Black racial identity and HIV-status maintained its negative and statistically significant association with lower likelihood of waitlist addition, such that the combination of Black race and HIV was associated with 20% lower likelihood of waitlist addition beyond what would be expected if they acted independently (aHR: 0.80, 95% CI: 0.66–0.98, p = 0.01).
Adjusted Likelihood of Waitlist Addition Interactions Between Human Immunodeficiency Virus and Patient-Level Characteristics
Bold indicates significance at p < 0.05.
Adjusted for age, race, sex, ethnicity, World Health Organization BMI class, diabetes, hypertension, atherosclerotic heart disease, cerebrovascular disease, chronic obstructive pulmonary disease, peripheral vascular disease, institutionalized status, ambulatory status, functional status, alcohol dependence, drug dependence, tobacco use, year of dialysis initiation, and dialysis center.
aHR, adjusted hazard ratio; BMI, body mass index; CI, confidence interval; HIV, human immunodeficiency virus; PWH, people with HIV; Ref., reference.
These three betas were again summed to permit comparison of Black PWH to White ESKD patients without HIV, such that Black race and HIV were associated with 65% lower likelihood of waitlist addition compared to White individuals without HIV (aHR: 0.35, 95% CI: 0.25–0.48, p < 0.001). To further examine the effect of HIV among Black individuals, the betas from the main effects for HIV-status and race and the interaction term were further combined, demonstrating that positive HIV-status among Black ESKD patients was significantly associated with 60% lower likelihood of waitlist addition (aHR: 0.40, 95% CI: 0.30–0.54, p < 0.001), providing further evidence that the negative association of positive HIV-status with waitlist addition was exacerbated among Black individuals compared to White individuals. Predicted HRs were generated, revealing Black PWH had lower predicted likelihood of waitlist addition than White ESKD patients, White PWH, and Black ESKD patients (Fig. 1).
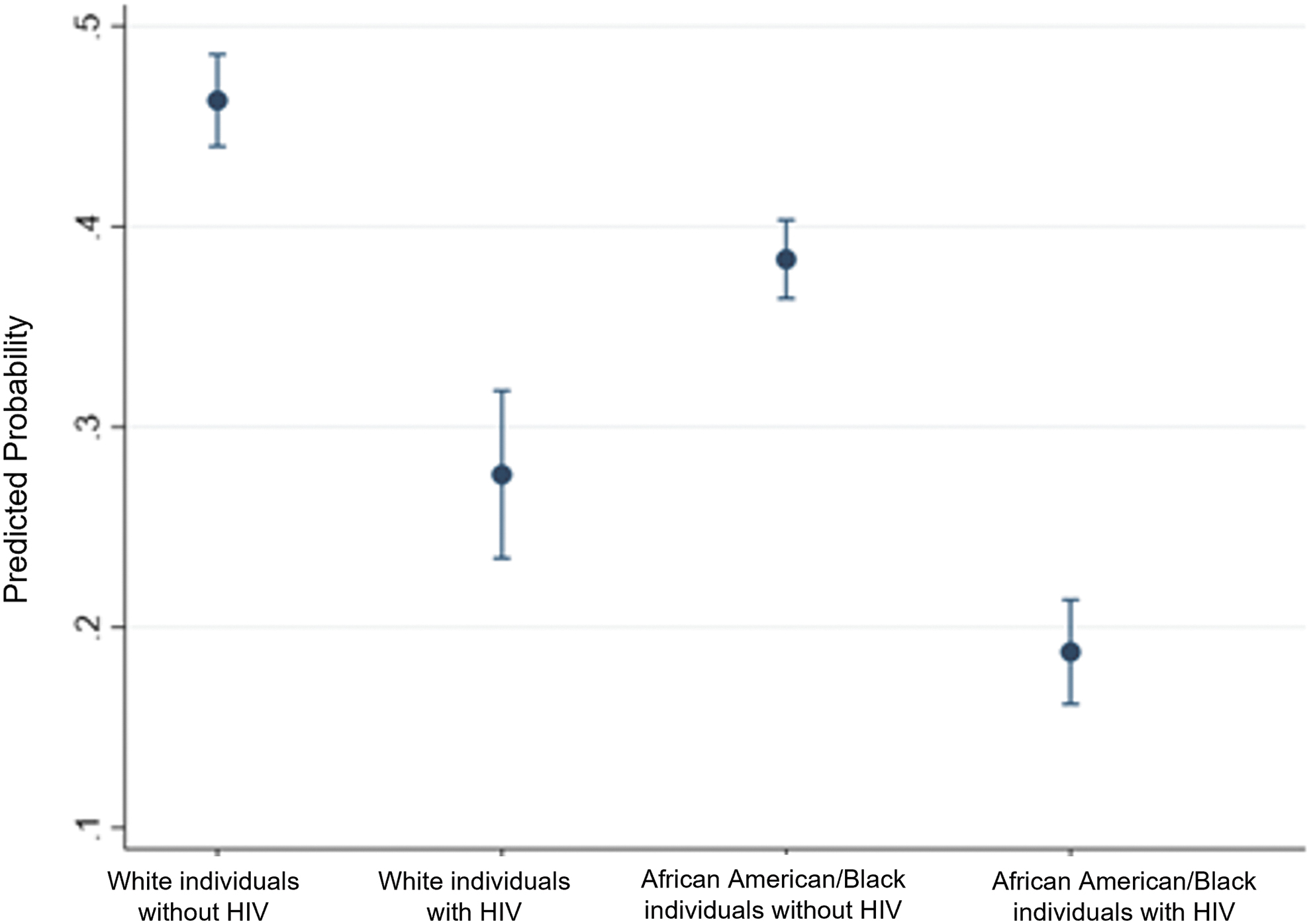
Predicted probability of waitlist addition within 5 years. Predicted probability for combinations of White and Black race and HIV-status were generated from the final adjusted Cox proportional hazards model. For White people without HIV, the predicted probability of waitlist addition within 5 years of dialysis initiation was 48%. For White people with HIV the predicted probability of waitlist addition within that same period was 27%. For Black people without HIV, the predicted probability of waitlist addition within that same period was 38%, and for Black people with HIV that predicted probability was 18%, significantly lower than any other group. HIV, human immunodeficiency virus.
Discussion
In this national study of PWH in need of a kidney transplant, PWH were significantly younger, more commonly Black, more commonly male, and reported higher prevalence of smoking, drug use, and alcohol abuse than people without HIV. After adjustment for these critical confounding characteristics, positive HIV-status among White individuals was associated with 50% lower likelihood of waitlist addition compared to White individuals without HIV. Moreover, among Black individuals, positive HIV-status was associated with 60% lower likelihood of waitlist addition. In sum, among Black individuals with HIV, there was a 65% lower likelihood of waitlist addition compared to White individuals without HIV observed, a significant exacerbation of the inequity.
These data support the hypothesis that Black race and positive HIV status together are associated with lower likelihood of waitlist addition rates than Black race or positive HIV status independently is and, given the disproportionate burden of HIV and ESKD among Black individuals, warrants further study.
The barriers to waitlist addition are best illustrated by the Socioecological Model for Transplant, 50 with each component warranting individual examination to measure how it exacerbates or ameliorates this observed inequity. At the individual level, PWH must meet both standard eligibility criteria and HIV-specific criteria. Social support and the relationship between an ESKD patient and their social network may impact their likelihood of waitlist addition given its relative importance as an evaluation criteria 51 ; similarly, higher levels of functional social support among PWH may contribute to increased likelihood of viral suppression and ART adherence, particularly among Black PWH. 52 Black PWH, however, less frequently achieve and maintain viral suppression, which may be attributable to lower levels of experienced intrapersonal, health system, and structural support than White PWH. 53
Thus, while undetectable HIV viral loads and stable ART regimens present a barrier for all PWH, these known racial disparities in viral suppression and ART adherence may contribute to this increased inequity among Black PWH and perhaps explain some of our findings. 54 –56 PWH who are also HCV positive, however, must also demonstrate lack of fibrosis progression, but disparities in treatment for HCV also exist, which may potentially increase the likelihood of observed fibrosis. 57 Consequently, individual characteristics and the relationships between individuals, their support networks, and health care systems may meaningfully contribute to both the observed disparity among White PWH and also the greater disparity among Black PWH.
Seventeen percent of transplant centers still consider an HIV diagnosis to be an absolute contraindication to kidney transplantation. 24 Moreover, many centers have policies that restrict access to the waitlist for individuals engaging in active substance use. 58 While substance use was included in our analyses, given evidence that the convergence of Black race and substance use disproportionately decrease access to kidney transplantation, 58 it is perhaps similarly likely that the convergence of an HIV diagnosis and substance use exacerbate existing disparities in access. The disparity in waitlist addition may also manifest earlier in the transplant process, such as at time of referral or evaluation, and therefore may reflect patient preference or decision-making by the individual's nephrologist or dialysis provider. These data are not captured nationally, limiting the ability to ascertain their role in creating or exacerbating the observed inequities.
A single study, however, demonstrated lower rates of referral and of evaluation among PWH compared to ESKD patients without HIV. 28 Critically, though this study also lacked access to HIV-specific data, and thus, it is unknown whether PWH were not referred or evaluated due to failure to meet the HIV-specific criteria. Moreover, there are no data measuring nephrologist or dialysis provider support for transplantation of PWH, hindering our ability to extrapolate to what extent those health care providers may be contributing to our observations. Recent analysis of electronic health records documented racial bias on behalf of health care workers with Black patients having 2.5-fold higher odds of being described as “agitated” or “non-compliant.” 59 Vignette studies have demonstrated both race-based and sex-based bias in allocation of advanced heart failure therapies. 60,61
It is plausible that these same concerns observed in vignette studies about social support, finances, and adherence are present in the care of patients with ESKD. Given the need for PWH with ESKD to be adherent to both ART and to immunosuppressive regimens post-transplant, the relationship between health care providers and PWH, particularly Black PWH, warrants study to both measure support for and likelihood of advancing eligible PWH through the transplant process.
Existing policies for organ allocation and the methods by which transplant center performance is assessed prioritize the efficient and optimal use of deceased donor organs whereby both patient and graft survival are maximized to derive the greatest utility from a scarce resource. Given this, PWH may be disadvantaged given their observed higher rates of acute rejection, 34 the high prevalence of substance use among PWH, 29 and concerns over whether viral suppression and adherence can be maintained. PWH, however, have access to an exclusive pool of organs from donors with HIV. In addition, the practice of kidney transplantation of PWH with donor organs from PWH may soon be considered standard clinical practice 62 ; yet, it remains unclear whether the more permissive regulatory environment will lead to broader adoption of this practice.
With only 35 of 250 transplant centers approved to participate in the study of HIV-to-HIV transplants, 63 expansion of this practice may consequently necessitate increased access to the transplant waitlist for PWH to ensure that there are adequate numbers of candidates willing to receive organs from donors with HIV. Access to the transplant waitlist for deceased donor organs may be even more critical when considered within the context of living organ donation for PWH. While living kidney donation from PWH is permitted under research protocols, the practice of such remains limited despite high reported willingness from PWH to serve as a living organ donor. 64,65 Given previous reports of lower rates of living donor kidney transplantation among PWH, 30 there may be greater reliance upon the pool of organs from deceased donors to treat ESKD.
One reported successful strategy for encouraging adoption of HIV-to-HIV transplantation is collaboration with HIV advocacy organizations, which has been attributed to higher levels of adoption and greater support for utilization of organs from donors with HIV. 35 Thus, forging connections between dialysis centers, transplant centers, organ procurement organizations, and HIV advocacy groups may present a tangible and cost-effective way of increasing access.
There are limitations to this analysis, the first of which is the lack of HIV-specific data. We were unable to assess CD4 counts, HIV viral loads, adherence to ART regimens, or history of opportunistic infection, all of which are critical measures for ascertaining eligibility for kidney transplantation. Moreover, it is perhaps likely that these characteristics also differed by race, thus contributing to some of our observed finding. While this is a critical limitation, it is also one to which all national analyses of ESKD patients are subject due to the failure of national databases to capture HIV-status before transplantation, let alone needed HIV-specific measures. As such, our understanding of individual and provider behavior in the context of granular HIV data is limited to single-center data.
We also were unable to ascertain whether lower waitlist addition rates were attributable to lower rates of referral for transplant evaluation or lower rates of evaluation completion. As with HIV-specific data, these data are not captured nationally, impeding the ability to study disparities on a national basis. Consequently, we cannot attribute our findings to any specific step of the process or to provider or patient behavior. Given single center data illustrating lower rates of evaluation completion among PWH and regional data revealing lower rates of referral and evaluation, 27,28 there is an urgent need for data collection. However, while these data are limited, they contribute to our understanding of waitlist addition among PWH nationally and identify a group both at elevated risk of disease and at a disproportionate lower likelihood of waitlisting, motivating greater study with more comprehensive data.
To our knowledge, this represents the first study of disparities in waitlist addition among PWH and in comparison, to ESKD patients without HIV. As observed among individuals without HIV, Black individuals were less likely to be added to the waitlist, but Black individuals with HIV were less likely to be added than White individuals with HIV or Black individuals without HIV, suggesting an exacerbation of both the disparities attributable to HIV and race, respectively. Given the disproportionate burden of both HIV and ESKD diagnoses among the Black population, this disparity warrants further study to ascertain the factors and relationships limiting access to transplantation.
Data Availability and Access
These data are publicly available through the United States Renal Data System through an IRB-approved research plan.
Ethics Statement
The Institutional Review Board at the University of Alabama at Birmingham (IRB-161212003) approved this study.
Footnotes
Authors' Contributions
B.A.S.: Conceptualization, Methodology, Formal analysis, and Writing—Original draft. D.J.B.: Methodology and Writing. P.A.M.: Methodology and Writing. B.S.: Methodology and Writing. H.B.: Conceptualization, Methodology, Writing, and Supervision. J.E.L.: Resources, Conceptualization, Writing, and Supervision.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This work was supported by the National Institutes of Health/National Institute of Diabetes and Digestive and Kidney Diseases (R01 DK117675, PI: Jayme E. Locke) and National Institute of Mental Health (K01 MH116737, PI: Henna Budhwani).
