Abstract
Bacterial sexually transmitted infection (STI) incidences of gonorrhea, chlamydia, and syphilis are increasing in Washington, DC. Moreover, the availability of HIV pre-exposure prophylaxis for people at risk of HIV and condomless sex has increased, and bacterial STI rates have risen. This indicates the necessity of evidence-based strategies to ensure access to STI care and improve health outcomes for people with HIV in Washington, DC. Three clinics in Washington, DC, implemented three evidence-based interventions, including the use of a standardized audio computer-assisted self-interview to obtain an interval sexual history at each clinic visit, patient self-collection of chlamydia/gonorrhea nucleic acid amplification test specimens, and sexual minority welcoming clinical space indicators to normalize STI screening and testing. Three sites in Washington, DC, used a multi-level socioecological model to identify successes, challenges, and lessons learned from program implementation at the following three levels: (1) individual, (2) interpersonal, and (3) public policy. We conclude with a series of instructional strategies that may be useful for the implementation of similar interventions that may assist district-wide responses to decrease health disparities and increase STI prevention.
Introduction
HIV continues to be a major public health problem in Washington, DC. Despite a recent decrease in HIV incidence rates in DC, the incidence rates of sexually transmitted infections (STIs) continue to rise. 1 Many people living with HIV are reaching viral load suppression of fewer than 200 copies/mL, making them undetectable and therefore unable to transmit HIV sexually. Because the rates of viral suppression among people with HIV (undetectable = untransmissible) in the United States and the uptake of HIV pre-exposure prophylaxis among people at risk of HIV have increased, condom use has decreased and the possibility of STI transmission has risen. 2 STIs increase infectiousness of people living with HIV by increasing the viral concentration in the genital tract, and by increasing the potential for HIV acquisition in people at risk for HIV. 3
Further, many people at risk of having an STI are not routinely screened or tested for STIs by their medical providers, when necessary, despite well-established recommendations for STI/HIV testing. 4 Barriers to accessing STI screening and testing included lack of medical providers within rural communities, privacy and confidentiality concerns, and perceived stigma from providers. 5
As a result, significant issues persist, and progress in eliminating bacterial STIs is hindered, affecting structural challenges in the District. Structural challenges include health disparities linked to stigma based on racism, gender identity/expression, sexual orientation identity, and other social determinants of health such as poverty, lack of employment opportunities, housing instability, and behavioral health conditions. 6 Unique and substantial variations in sociodemographics and public policies increase vulnerability and cause discrepancies in STI screening, treatment, and prevention among this population. 7 As a result, the government has focused more attention and resources on increasing the physiological understanding of STIs, improving sensitivity and specificity of STI diagnostics, and developing evidence-based interventions to enhance STI screening, testing among individuals with or at risk of HIV. 6 Although different interventions suggest promising approaches, there are limited evaluative study findings demonstrating the effectiveness of these interventions when implemented in an outpatient ambulatory setting.
Background sociodemographics of the DC metropolitan area
Beyond the recommendations in the Sexually Transmitted Infections National Strategic Plan, Washington, DC, faces challenges in reaching populations affected by common bacterial STIs. 6 Years of systematic and structural influences have shaped the district's racial health disparities. 7 Racial health disparities translate into differences in health care need and demand, which are reflective of real-life barriers such as culture, economics, education, insurance status, and language. This has influenced difficulty in reaching disproportionately impacted ethnically and racially diverse groups, resulting in limited access to STI-related services.
Structural challenges such as policies, practices, norms, and conditions at the societal level jeopardize marginalized racial and ethnic group social standing and access to STI prevention education, STI testing, and inequitable STI-related outcomes. 8 We present results of a process evaluation, in which we used three evidence-based interventions to reach these persons burdened with racial health disparities and high infection rates. The jurisdictional goal of these interventions was to address sociodemographic risk factors associated with STI infections that impede routine screening, testing, and treatment; and improve clinical outcomes throughout the care continuum.
HIV/STI coinfection
Despite ongoing public health initiatives to minimize the rate of new STIs, people with HIV persist as a subpopulation with higher-than-average rates of STIs. These high rates are especially concerning for Black, Hispanic/Latino, American Indian, or Alaska Native populations in the United States, who have the highest rates of STIs and HIV among all races. 9 To address the disparate rates of STIs, we explored evidence-informed interventions to reduce health inequalities and mitigate the STI epidemic impacting people with HIV in Washington, DC.
Peer support
Several barriers to getting tested among persons with STIs have been identified in previous studies, including expected stigma from health care providers, lack of knowledge of STIs, fear of potential consequences of a positive test result, lack of time to attend care, privacy concerns, and low-risk perception. 10,11 In an effort to combat STI barriers, this multi-site evaluation drew on conceptualizations of patient self-collection shaped by factors at multiple levels of interpersonal influence, including peer support. Patient self- collected STI testing for GC and CT can be identified as vaginal swabs, rectal swabs, pharyngeal swabs, and urine samples done by the patient in clinical and nonclinical settings. 12 Peer support refers to emotional, social, and practical assistance provided by nonprofessionals to help people sustain health behaviors. 13
As a result, it is a nonhierarchical interpersonal process that promotes mutual healing in the framework of community, defined by equitable interactions among individuals who have shared experiences and a commitment to growing beyond the constraints of one's current situation. Peer support for people living with HIV has gained increasing traction and is considered a way to take an active role in self-management. 14 This explains the concept that support is a form of self-help, autonomy, positive risk-taking, and self-awareness as a practice. To address STI barriers, we examine the role of peer support in shaping patient self-collection related to STI prevention by evaluating patient's accessibility, comfort, and preference for self-administered specimen collection.
In this article, the authors employ qualitative and quantitative data and contextual factors to better understand implementation successes and challenges identified across three clinical sites, an academic medical center clinic, a community-based clinic, and a Health Resources and Services Administration (HRSA) Health Center Program-funded clinic providing HIV care in Washington, DC. The authors compare sociodemographic, geographic, and social determinant factors with quantitative findings from three evidence-based interventions. The authors evaluated the facilitators and barriers to implementation of the bundled interventions: self-collected chlamydia/gonorrhea nucleic acid amplification test (CT/GC NAAT) specimens, audio computer-assisted self-interview (ACASI) sexual history, and LGBTQ+ welcoming clinical space indicators (WCSIs) at the following three program implementation levels: (1) individual, (2) interpersonal, and (3) public policy. Qualitative methods aid in comprehension of contextual factors and antecedents that affect implementation and are best suited to access diverse perspectives.
Methods
Data collection
We used a mixed-method process assessment to examine the sufficiency, accuracy, and experience of innovative methods for enhancing HIV screening, testing, and treatment among people with HIV. This process evaluation consisted of qualitative and quantitative data from three jurisdictional sites and was collected over a 1-year period. Two sites targeted HIV patients, while one site enrolled both patients with HIV and those at high risk into the multi-site evaluation. The quantitative data were collected through a Patient Satisfaction Survey to measure the efficacy and need for STI interventions within the three clinical demonstration sites across the DC jurisdiction. The Research Assistant and Peer Support Specialist administered the survey to the patients, along with managed enrollment and patient consent. Both personnel bridged together research-supported health care engagement realms and self-management interventions, offering an alternate perspective to challenges faced by patients.
Overall, qualitative data included systematic information about the obstacles and facilitators that influenced implementation in the jurisdiction. Other key staff included in the process of observation included a designated Change Champion (a clinical prescriber serving as the lead for the study at each clinical demonstration site), a nonclinical prescriber (medical assistant), a data coordinator to collect and enter patient- and intervention-specific data into the data portal for this project, and a jurisdictional convener.
Quantitative implementation data consisted of data from the ACASI-based sexual history and electronic medical record (EMR) data. The ACASI privately screened for GC/CT every 3–4 months and measured the patients' sexual behaviors, STI-related symptoms, and intervention satisfaction. If for some reason a patient was unable to use the ACASI-based sexual history, the RA or PSS would ask the sexual history questions, while the patient was in the examination room and document the answers using the ACASI-based tablet.
The ACASI-based surveys were uniquely customized and programmed in the primary languages of the study participants: English, Spanish, and Amharic. The eligibility criteria for enrollment into the multi-site evaluation required that patients were 18 years of age or older, had HIV or were at high risk and receiving care at the designated Ryan White HIV/AIDS Program (RWHAP)-funded clinic or Bureau of Primary Health Care (BPHC)-funded health center, and could read or understood audible English, Spanish, or Amharic.
The ACASI sexual history asked patients a series of questions about their sexual behaviors since last seen in the clinic or Health Center, and based on those results, provided advice to the clinical and nonclinical prescribers on which specimens (extragenital site-specific GC/CT NAAT specimens) to collect. Patients were also asked using the ACASI-based sexual history their preference regarding self-collected or provider-collected NAAT specimens for each of urogenital, throat, and rectal sites. Patients who agreed to provide self-collected GC/CT NAAT specimens were offered self-management support by both clinical and nonclinical prescribers, in which they were given a self-collection kit with personalized labels and step-by-step instructions to perform testing themselves. GC/CT NAAT specimen test results were identified in the patients EMR and trends in GC/CT STI testing, and positivity of the multi-site results were examined.
Each session (participant visit) is saved on its own line in the text file, and additional sessions are appended to the participant's data file. After completion of the ACASI sexual history survey, a summary of recommended STI tests will be auto-generated based on client responses. ACASI data files are automatically saved on the tablet and EMR data are entered in real time. Both were imported into a statistical analysis software program, SPSS, for analyses to determine the number/percent of bacterial STI at-risk participants screened, tested, and diagnosed with GC/CT (genital and/or extragenital sites) and how intervention(s) affected STI screening, testing, and treatment, by subpopulation [race, heterosexual, and men having sex with men (MSM)].
Analysis
Next, we used a comparative approach to make comparisons of themes, perspectives, experiences, and processes to develop an explanatory framework for understanding program implementation across the sites. We used a multi-level socioecological model (SEM) to identify successes, challenges, and lessons learned (Fig. 1). Our analysis identified facilitators and barriers to program implementation at three levels: (1) individual, (2) interpersonal, and (3) public policy. 7
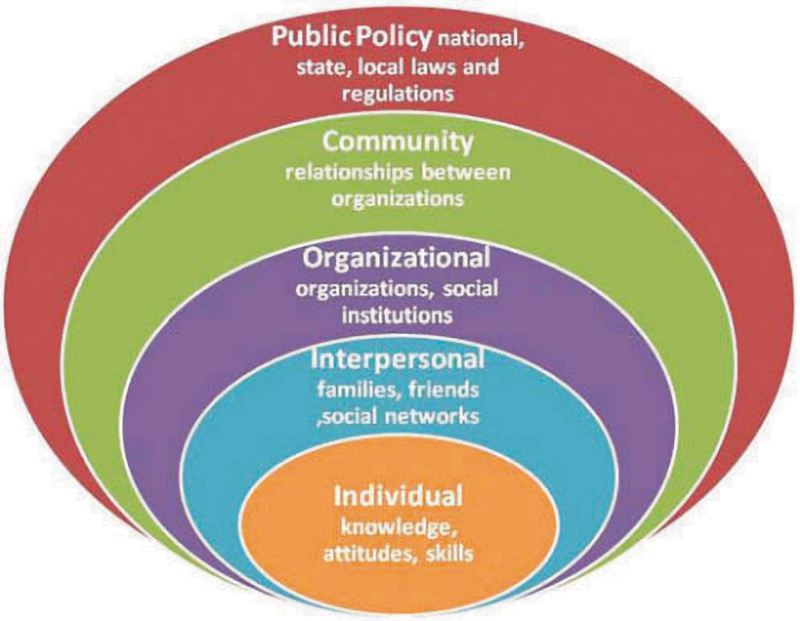
Socioecological model: framework for prevention. 15
Socioecological model
The fundamental principles of the SEM are as follows: (1) multiple factors influence an individual's behavior, including intrapersonal and interpersonal factors, with increasing effect at organizational, community, and public policy levels, (2) there are different degrees of influence, (3) this model should be used to analyze specific behaviors, and (4) multi-level interventions can be the most successful for modifying behaviors (Fig. 2). 15
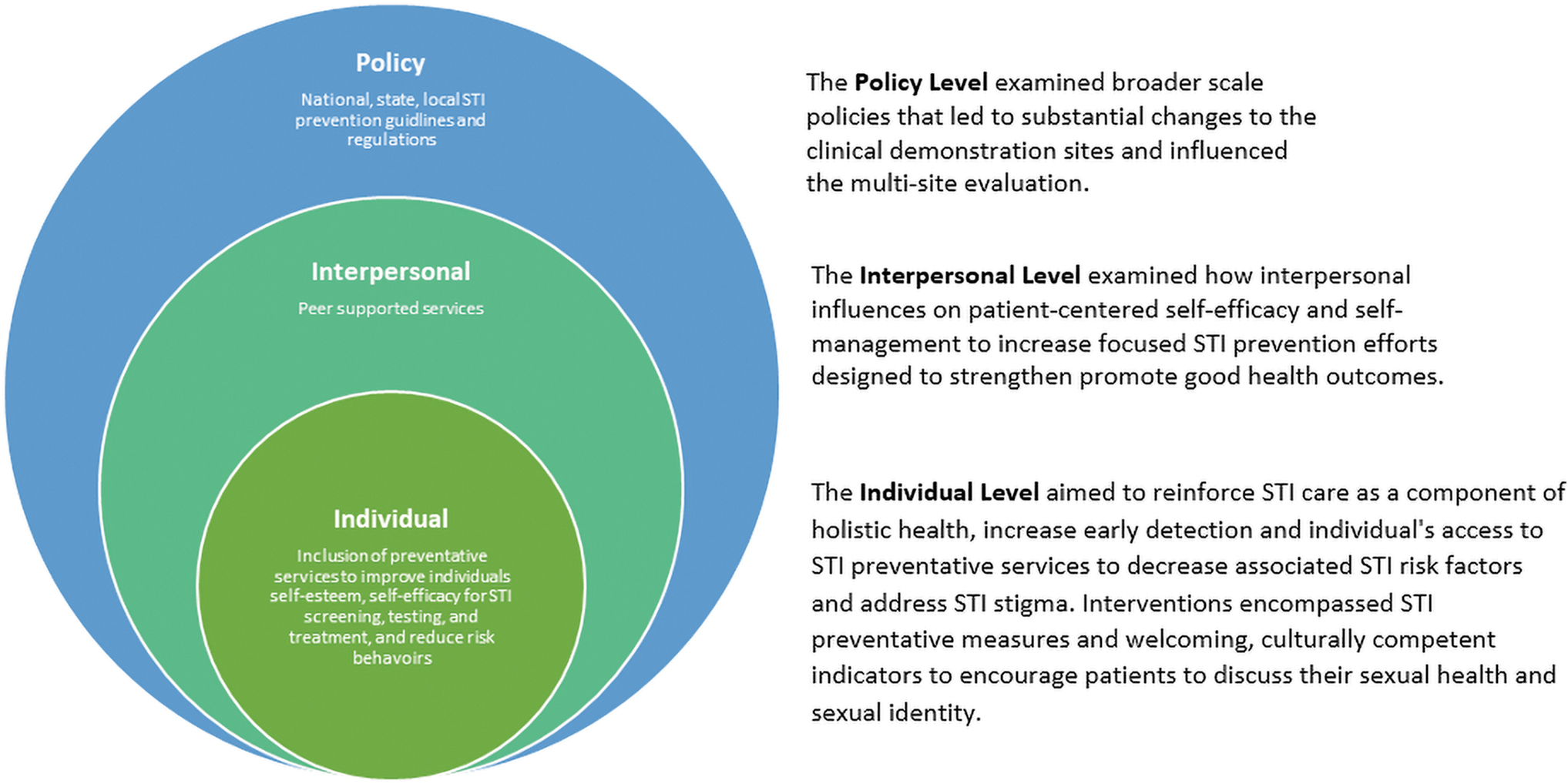
Modified socioecological model to address STI screening and testing interventions. STI, sexually transmitted infection.
At the model's core is the individual under the influence of interpersonal and policy interventions. Individual-based interventions are aimed to minimize the health and socioeconomic burdens of STIs and associated risk factors. The interpersonal sphere includes factors related to self-management support that may influence the individual's comfort with STI testing and health outcomes. Finally, at the outer sphere of the figure is the public policy level that includes federal/state/local/organizational regulations (with significant persuasive powers) that will extend to everyone in the society and demonstrate a destigmatized social norm.
Results
During the 12-month study period, 294 patients 19–81 years of age participated in the DC multi-site evaluation. The mean age of all participants was 50.13 years, and the majority identified as Black or African American (87.41%) (Table 1).
Participant Demographics (n = 294)
SD, standard deviation.
Individual level
LGBTQ+ WCSIs
There were 12 LGBTQ+ WCSIs (Table 2). Before implementation of the interventions, two out of three of the DC clinics already had more than half of the indicators in place. Pre-study, three clinical demonstration sites had a combined total of 63% LGBTQ+ WCSIs in place. Post-study, 90% of the LGBTQ+ WCSIs were implemented at each clinical demonstration site. For those not in place at the end of the study, the reason was due to systematic bureaucratic hurdles (see Limitations).
LGBTQ+ WCSIs by District of Columbia Clinical Demonstration Site Pre- and Post-Study
LGBTQ+ WCSI, Welcoming Clinical Space Indicators.
A total of 240 Patient Satisfaction Surveys were collected for the DC jurisdictions, and of all Patient Satisfaction Survey respondents. Among the 240 surveys, 167 total surveys were completed by “Heterosexual” participants, 51 total surveys were completed by “LGBTQ+” participants, and 22 total surveys were completed by “Other” participants. Although being treated with respect had a positive influence on most of the participants, sexual gender minorities had a higher overall level of agreement with LGBTQ+ WCSIs (Table 3).
Patient Satisfaction with LGBTQ+ WSCIs
Fisher exact test used to determine if there are nonrandom associations between patient satisfaction (noticing, not noticing, liking, and disliking), the LGBTQ+ WCSI, and sexual orientation (Heterosexual, LGBTQ+, and Other).
Values in bold demonstrate there was no association between patient satisfaction, sexual orientation, and the following LGBTQ+ WCSI: [visible LGBTQ+ inclusiveness materials in the waiting room such as magazines, posters, and fliers; LGBTQ inclusiveness educational materials (gender diverse people and same-gender couple); fliers or cards in the waiting room with information about LGBTQ supportive organizations; and acknowledgment of LGBTQ awareness and recognition days or events, such as Transgender Day of Remembrance or LGBTQ Pride].
LGBTQ+ WCSI, Welcoming Clinical Space Indicators.
Of the 10 LGBTQ+ WCSIs, the following 6 measures were noticed and liked the most by participants whose sexual orientation was “Other”: acknowledgment of LGBTQ+ awareness and recognition days/events (54.55%), clinic registration/intake form for patient name and pronoun (77.27%), flyers or cards in waiting room about LGBTQ-supportive organizations (50%), gender-neutral bathrooms (54.55%), visible gender and sexual minority inclusiveness in waiting room materials (magazines, posters, and flyers) (63.64%), and respect from the clinical staff and providers (100%). For the LGBTQ+ participants, the highest rate of positive impact was associated with the clinic registration/intake form (68.63%), gender-inclusive educational materials (52.94%), and the nondiscrimination policy (72.55%).
Audio computer-assisted self-interview
To improve the patient experience with STI screening and testing, each clinical demonstration site implemented computer assisted self-interview (ACASI)-based sexual histories into the clinic flow. Out of 188 participants who completed the Patient Satisfaction Survey, 97.34% reported finding the ACASI-based sexual history taking to be “very easy” or “easy” and 2.66% reported it being “very difficult” or “difficult” (Fig. 3).
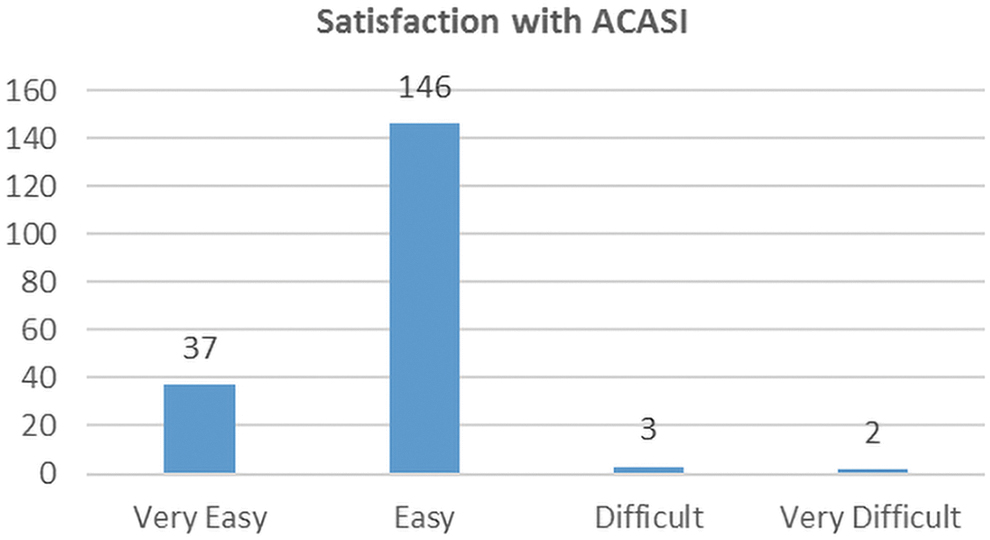
Participant satisfaction with ACASI-based sexual history. ACASI, audio computer-assisted self-interview.
Self-collected NAAT specimens
Two clinics executed this intervention into the clinic flow during pre-appointment laboratory services to address time constraints affecting STI screening and testing. One site did not have a laboratory on site, prompting specimens to be collected by the nonclinical prescribers during the follow-up examination. To determine the patient's preference of self-collected CT/GC NAAT specimens, participants completed the ACASI sexual history assessment, in which 70.7% of patients preferred for the provider to collect rectal GC/CT NAAT specimen, 55.5% preferred for the provider to perform throat GC/CT NAAT, and 64% patients preferred the self-collected urine tests (Fig. 4). Over the multi-site evaluation, there were 21 positive/reactive STI tests.
Participant comfort in GC/CT NAAT specimen self-collection. GC/CT, gonorrhea and chlamydia; NAAT, nucleic acid amplification test.
Fourteen (4.8%) of the 294 participants tested positive for GC and/or CT at least once during the study period. Of these 14 participants, 6 (42.86%) reported no STI-related symptoms at the time of being tested. There was a total of nine positive cases for GC/CT at urogenital sites, in which all six cases reported symptoms that make up 66.67%. Of the 14 that had a positive STI, 8 were only at urogenital sites and 2 had an extragenital and urogenital site infection at the same time.
Out of 6 participants who tested positive for GC/CT at only urogenital sites, 3 (50%) did not report symptoms. Seven out of 14 tested positive for GC/CT in extragenital (rectal and pharyngeal) sites, of which 2 (28.6%) did not report STI-related symptoms and would have otherwise been missed without screening and testing. Despite the low positivity rate (Table 4), increasing the frequency of screening and testing with the use of extragenital site self-collected swabs helped screen asymptomatic patients and detect repeat infection (Fig. 5).
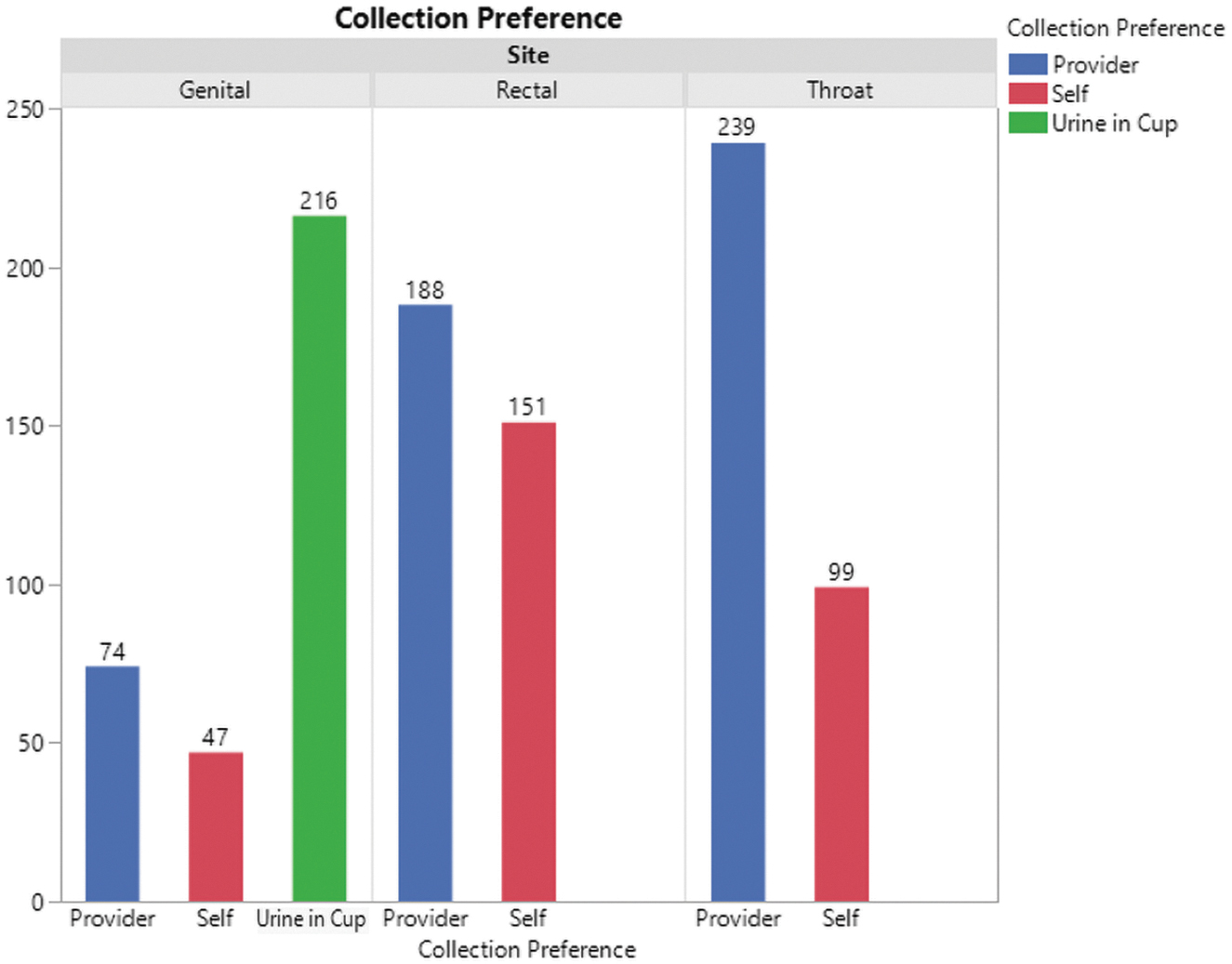
Participant specimen collection preference for GC/CT NAATs. GC/CT, gonorrhea and chlamydia; NAAT, nucleic acid amplification test.
GC/CT Nucleic Acid Amplification Test Testing Positivity Rates
CT, chlamydia; GC, gonorrhea; STI, sexually transmitted infection.
Interpersonal level
Peer support services
Peer support helped make STI testing more accessible for patients, encouraged regular screening, enhanced the convenience of testing through self-testing, and addressed enabling factors that affect the patients comfortability. The central approach of peer support was to improve self-management in patients diagnosed with an STI and contribute to the ease and access to STI screening, testing, and treatment services, improving health outcomes through patient education and promoting a common understanding of the participants' needs and preferences. Peer support bridged together meaningful engagement and self-management interventions, offering patients emotional support that improves their health condition and management, making them more comfortable to do a self-collected chlamydia/gonorrhea nucleic acid amplification test (CT/GC NAAT).
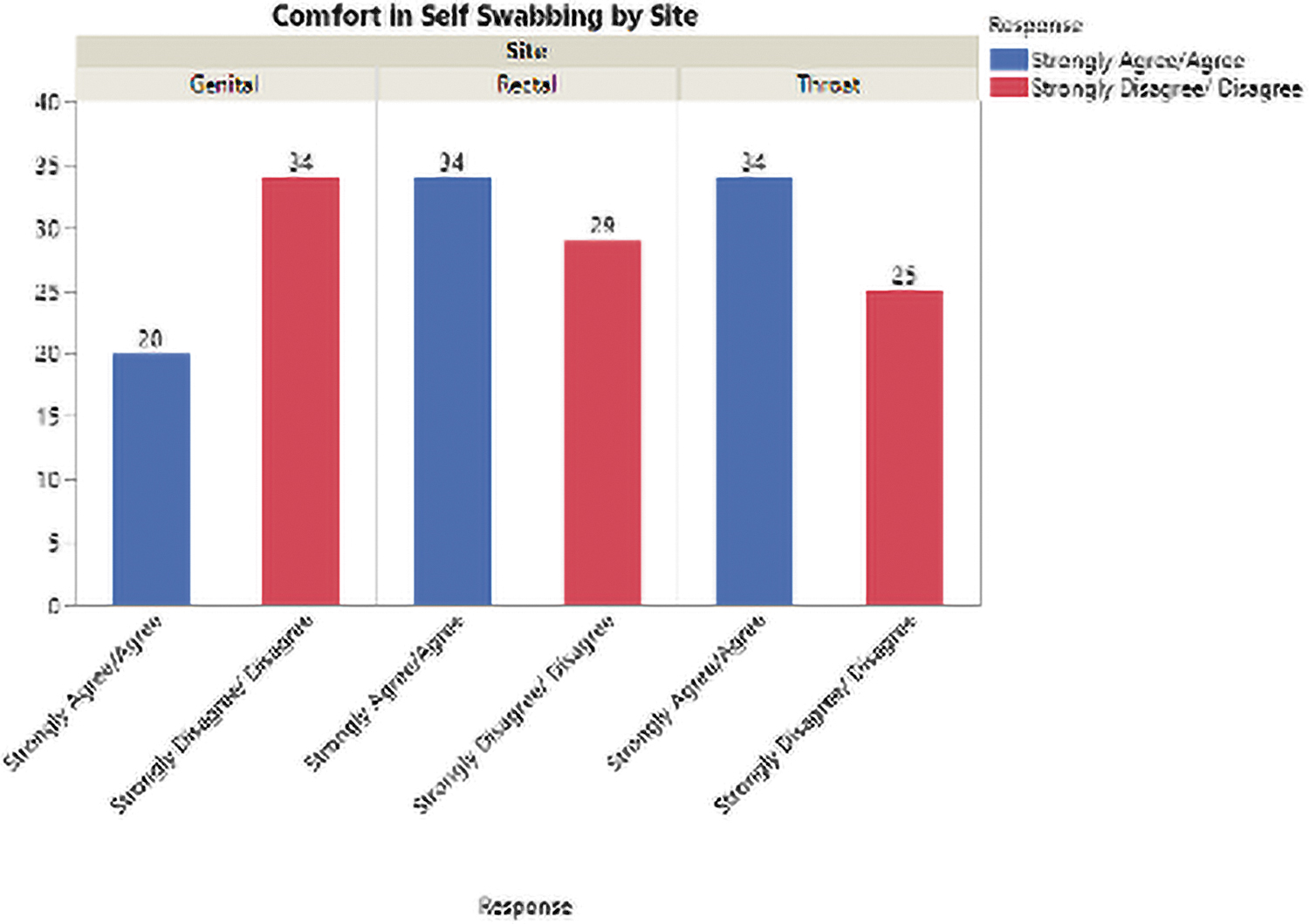
Of the 240 Patient Satisfaction Surveys completed for the DC jurisdiction, 56% strongly agreed or agreed being comfortable performing their own throat and rectal swab, because “it is easy to do,” and/or that its more private. Forty-four percent of participants reported not being comfortable performing their own throat and rectal swab and believed the providers swab “would be more accurate” (Fig. 5). Genital NAAT specimen self-collection had the lowest acceptance rate of 37% among participants completing the satisfaction survey; however, urine was primarily used instead of genital swabbing.
Policy level
At the outer level of the SEM are the larger-scale policies that influenced the individual- and interpersonal-level changes to the clinical demonstration sites and shaped the multi-site evaluation.
Discussion
Findings from this multi-site evaluation identified facilitators and barriers to the implementation of STI preventative interventions within the five levels of the SEM. Factors that facilitated implementation included subjective methodologies, access to an adequate pool of trained and certified personnel, and peer support approaches. Because of the minimization of face-to-face contact with patients due to COVID-19, recruitment efforts were impacted by the peer support approach. Welcoming measures were critical in improving intersectional stigma at the micro-level. The ACASI-based sexual history increased routine screening, and CT/GC NAAT extragenital site testing played an important role in increasing detection of asymptomatic bacterial STIs. Finally, an adequate understanding of the macro-level epidemiology at the local level was critical in directing program efforts. As a result, stigma and a lack of measures for evaluating participant engagement and facilitator performance were major roadblocks to implementation.
As a result of this review, the following recommendations may be relevant for the development of similar interventions aimed at increasing STI prevention. Our findings show that preliminary data gathering and piloting of the proposed intervention component before implementation are critical at the individual level.
Our findings also imply that regular training could impact the adoption of interventions at the individual level, easing implementation efforts. Clinicians who advocated for improving existing STI prevention measures in organizations were successful in implementing interventions. The latter opposed involvement that jeopardized the planned implementation. According to the findings, program components that are being implemented for the first time at an organization should provide development training to physicians and clinical personnel before implementation.
At the interpersonal level, peer support approaches that adopted the goals of patient self-management of specimen collection were key factors that supported implementation efforts. It is critical for sites to promote patient's sense of belonging and give them less stigmatizing and intrusive sources of support. This can ensure all patients feel safe and included, despite different behaviors, identities, and expressions.
At the macro-level, current epidemiology at the local level impacted the implementation of interventions at the clinical demonstration sites. Based on the CDC and DC Department of Health recommendations, system-wide responses to normalize patient-administered sexual history screenings, increase STI extragenital site testing, and expand the scope of sexual health education and treatments for bacterial STIs in people with HIV were implemented at the macro-level. Macro-level interventions consisted of both health and social policies at the local district level. Macro-level interventions aimed to improve population health and health equity and change or influence social, economic, or political environments (the root causes of health and health inequity) in ways that benefit large numbers of people at once 16,17 and promote population health or address health inequities. 18
Policies increase access to prevention, education, and information, as well as testing and treatment, to affect the distribution of STIs across and within the district's social and cultural norms. Societal factors influence health policy in STI prevalence and prevention, particularly among groups who are disproportionately affected by STIs, and target mechanisms to decrease the overall effect of STIs and STI inequities. Macro-level health and social policies served as a roadmap for all clinical demonstration sites to guide the development of LGBTQ+ WCSIs, initiatives, and actions for STI prevention and control. To support the achievement of these goals, we implemented them across all domains and levels of the multi-level socioecological framework.
Limitations
Although the multi-site evaluation facilitated several opportunities for the clinical demonstration sites, it posed a special set of barriers. Despite efforts to reduce stigma and improve STI preventative services, the clinics still faced many challenges to enhance patient adherence to STI interventions. This was primarily influenced by COVID-19, due to the need to minimize face-to-face contact with patients. During these unprecedented times, each clinical demonstration site faced many uncertainties and broad-ranging challenges encompassing a range of activities, including prioritizing patient safety amidst the pandemic, consenting, and recruiting new participants into the study, GC/CT NAAT specimen collection, and intervention delivery. Because the ACASI-based sexual histories produced timely information, it was difficult fitting time for ACASI sexual history taking into patient appointments. In many cases, recruitment was paused, while these mitigations were evaluated. Administrative hurdles, impacted by each clinic's unique culture, also affected implementation.
Hurdles were due to complicated rules and procedures within the clinic system, which caused prolonged delays in eventual approval. Although established with good intent, the clinic's rules and regulations stifled timely project implementation and hindered patients from receiving the full benefit of STI prevention interventions. This included inability to purchase LGBTQ+ WCSIs, long hiring process, despite staffing shortages, and systemic IT issues. Another challenge was political disturbances in the DC during the study period that exposed the racial undercurrents of national politics and required mitigations to maintain patient safety.
Lessons Learned
Despite challenges, we strive to share the most valuable information on lessons learned and promising practices specific to STI prevention. While recruiting ethnically and racially diverse demographics to prevent and treat STIs, we learned several lessons framed around stigma. Stigma and the lack of information and awareness regarding the commonality of STI diagnoses, combined with outdated beliefs, led many patients to fear contracting STIs. Fear clouded many patients' judgments, with regard to protecting their health, which turned healthy conversations about safer sex into awkward dialogs fraught with defensiveness.
Many patients believed the routine sexual history screening was an invasive way of underhandedly questioning their sexual responsibility (or lack thereof), while others understood that it was an essential part of their health, which suggests that stigma often tends to develop in area where knowledge is lacking. By introducing the component of surveys into the evaluation, we were able to measure the patient experience and determine an effective way to foster an environment that destigmatizes STI diagnosis.
Given the increased focus on identifying successful strategies to improve the routine screening, testing, and treatment of STIs, it is vital to learn what elements influence whether these interventions succeed or fail. The purpose of this study was to figure out what factors influenced the implementation of five STI prevention interventions. Findings identified barriers and facilitators to multi-site implementation. The findings have significant ramifications for STI interventions and highlighted several levels of influence to consider when planning and developing similar interventions. We also wish to emphasize the relevance of process evaluations and implementation research in future evaluations. In the realm of STI prevention, a detailed understanding of implementation is instrumental in the establishment and success of dissemination efforts.
Ethical Approval
The study protocol was approved by the Institutional Review Boards of Howard University and Rutgers University. Consent forms with Rutgers University contact information were distributed to all subjects following explanation of the surveys and before their participation in the study. Upon completion of the surveys, subjects received contact information to request an aggregate summary of study findings.
Consent
Informed consent was obtained from all participants
Footnotes
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This study was supported by the US Department of Health and Human Services (HHS) Health Resources and Services Administration's (HRSA) HIV/AIDS Bureau (HAB) and Bureau of Primary Health Care (BPHC) under grant number U90HA32147. The contents are those of the author(s) and do not necessarily represent the official views of, or an endorsement by, HHS, HRSA, HAB, BPHC, or the US Government.
