Abstract
We conducted a 24
Introduction
In sub-Saharan Africa, where breastfeeding is the norm, strategies for prevention of mother-to-child transmission of HIV-1 (PMTCT), which can occur intrauterine (IU), or intrapartum (IP), or postpartum (PP) mainly through breastfeeding, have dramatically evolved over the past 20 years, based on the World Health Organization (WHO) recommendations.
The WHO's recommendation of 2013, for PMTCT, is a program called Option B+ combination antiretroviral therapy (cART) where all HIV-infected pregnant women should be offered cART for life without any CD4 counts being quantitated or WHO staging of HIV disease assessed. In addition, all the infants from these HIV-infected mothers should be offered nevirapine prophylaxis in the first 6 weeks postnatally. Further, the infants born to HIV-infected mothers should be exclusively breastfed in the first 6 months postnatally. 1
By 2015, most countries in sub-Saharan Africa, including Zimbabwe, had implemented the WHO-recommended Option B+ in their national PMTCT programs. 2,3
We conducted a 24-month prospective follow-up study, at a primary health care clinic, to determine cumulative mother-to-child transmission of HIV-1 (MTCT) rate and the contributions of IU, IP, and PP to MTCT, as well as maternal and infant mortality rates in the era of Option B+ cART.
Materials and Methods
The study was conducted at Mabvuku Polyclinic (MPC), a primary health care clinic in Harare. The clinic has a laboratory on site where we installed a four-module GeneXpert machine for point-of-care (POC) early infant diagnosis of HIV-1 infection (EID) and viral load (VL) quantitation, desktop computer, uninterrupted power supply, and a biosafety cabinet for our study as we previously described. 4
HIV-infected pregnant women at various gestational periods, who had registered for antenatal clinic services at MPC, were enrolled into the study. The study was approved by the Medical Research Council of Zimbabwe (MRCZ/A/2113). All mothers signed the informed consent. Maternal VL quantitation was conducted at enrolment and every 6 months thereafter up to 24 months. Mothers with VL measurements >1000 copies/mL (cps/mL) had VL quantitation repeated within 2 weeks. Those with VL >1000 cps/mL on two consecutive tests were referred to MPC for consideration of switching to alternative cART regimens if suspected to have developed drug resistance, but with good adherence to cART, as we previously described. 4
Assessment of adherence was based on self-report. Infants were tested at birth, 4–6 weeks, and every 3 months thereafter up to 24 months. HIV-infected infants were referred to MPC for consideration of cART commencement. Those infants initiated on cART were then followed up every 6 months up to 24 months with VL quantified.
Viral load quantitation
Whole blood in ethylene diamine tetraacetic acid (EDTA) was collected and centrifuged within 6 h of phlebotomy to obtain plasma for VL quantitation. VL was quantified using the Cepheid GeneXpert HIV-1 Quantitative test (Cepheid, Sunnyvale, CA) following manufacturer's instructions as we previously described. 4
Diagnosis of infant HIV infection
From the infants enrolled into the study, aliquots of 50 μL of finger-prick blood were collected (five spots per card, using No. 903 Protein Saver cards; Whatman) at birth, 4–6 weeks, and every 3 months thereafter up to 24 months, air dried, and used for EID. The Cepheid GeneXpert HIV-1 Qualitative test (Cepheid) was employed for EID, as we previously described. 4
Timing of infant HIV infection
Timing of IU infection was defined as infants testing HIV positive at birth. Infants who tested HIV negative at birth, but subsequently first tested HIV positive at 4–6 weeks were considered to have evidence of IP infection. PP infection was defined as infants who tested HIV negative at birth and at 4–6 weeks, and subsequently tested first HIV positive at 3 months or later, as we previously described. 4,5
Statistical analysis
Rates of mode of transmission, cumulative mortality, and overall MTCT rates at 24 months were calculated. All mother-infant pairs who withdrew from the study were not included in the analysis. Continuous variables were summarized using parametric and nonparametric methods and also categorized where appropriate. Categorical variables were summarized using proportions and percentages. All statistical tests with a p-value of ≤0.05 were considered showing evidence of statistical significance. Statistical analysis was performed using STATA 15 (StataCorp, College Station, TX), as we previously described. 4
Results
Five hundred and two mothers were enrolled into the study between December 6, 2017, and December 31, 2018 (Fig. 1). 4 Six spontaneous abortions, and 10 stillbirths, including 1 twin set, were recorded. There were 492 live births, including 6 twin sets. Twelve babies died before they were enrolled into the study. The cause of death in 9/12 of these babies was respiratory distress; 2 babies died from birth asphyxia and 1 baby died from biliary obstruction. Twenty-seven infants were withdrawn from the study by their parents after delivery. 4 Thus, 453 infants, including 2 twin sets and 2 single twins, were enrolled into the study and together with 475 mothers (including mothers with fetus losses and those whose babies died before enrolment) were followed up to 24 months.
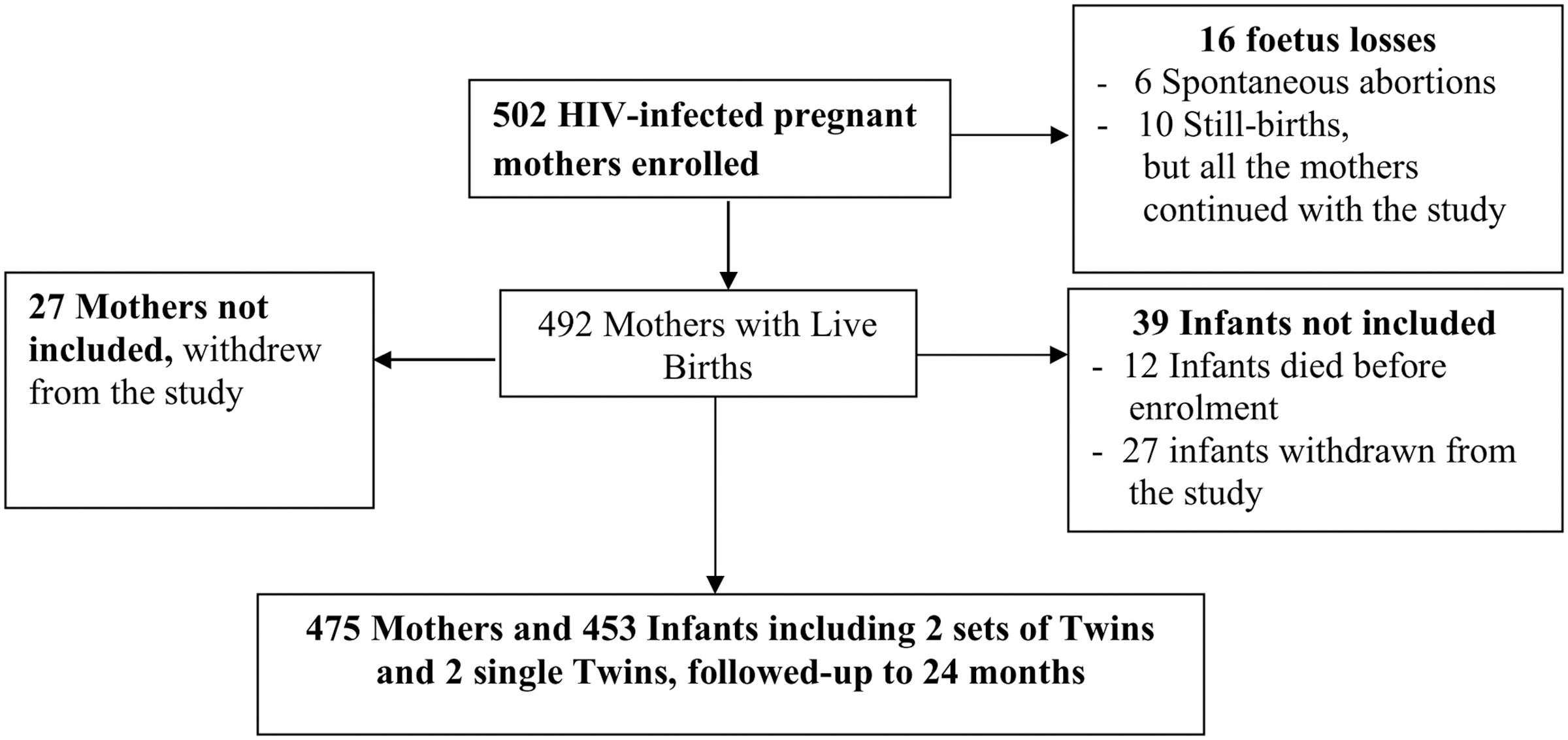
The figure shows enrolment of the mothers and their infants into the study.
Baseline characteristics, at enrolment, of mothers who transmitted HIV infection to their infants (transmitters) and those who did not (nontransmitters), and the mothers with unknown HIV-1 transmission
Table 1 shows the baseline characteristics of the transmitters, nontransmitters, and mothers with unknown HIV transmission (women with fetus losses and those whose babies died before enrollment into the study). The small number (n = 7
Baseline Characteristics at Enrolment of All the Mothers
The table shows baseline characteristics at enrolment of mothers who transmitted (transmitters), mothers who did not transmit (nontransmitters) HIV infection to their infants, and mothers with unknown transmission.
Duration on cART (years) was calculated from the date cART was commenced up to the date the mothers enrolled into the study.
3TC, lamivudine; ABC, abacavir; ATV, atazanavir; ATV/R, atazanavir/ritonavir; AZT, zidovudine; cART, combination antiretroviral therapy; D4T, stavudine; DTG, dolutegravir; EFV, efavirenz; FTC: emtricitabine; IQR, interquartile range; NVP, nevirapine; SD, standard deviation; TAF, tenofovir alafenamide; TDF, tenofovir fumarate.
The majority of the women, 96.42%, were on first-line cART regimen: tenofovir/lamivudine/efavirenz. The median duration on cART of the transmitters was 0.02 years, interquartile range (IQR): 0–0.22, compared to 1.80 years, IQR: 0.10–3.55 for the nontransmitters, and 0.46 years, IQR: 0.08–2.83 for mothers with unknown transmission. While all the 7 transmitters had detectable VL (>1000 cps/mL), the majority of nontransmitters, 62.61%, and mothers with unknown transmission, 58.33%, had undetectable VL (<20 cps/mL).
Timing of HIV-1 infection from birth to 24 months
Figure 2 shows outcome of infant HIV testing from birth to 24 months. Seven infants were infected by 24 months. The estimated cumulative MTCT rate at 24 months was 1.55% (95% CI 0.68–3.22%). Four of the infected infants were HIV positive at birth, suggesting IU infection. The IU transmission rate was 0.88% (95% CI 0.26–2.33%). The fifth infant tested HIV-1 negative at birth and subsequently first tested HIV positive at the 4–6-week follow-up visit, implying IP transmission rate of 0.22% (95% CI 0–1.4%).
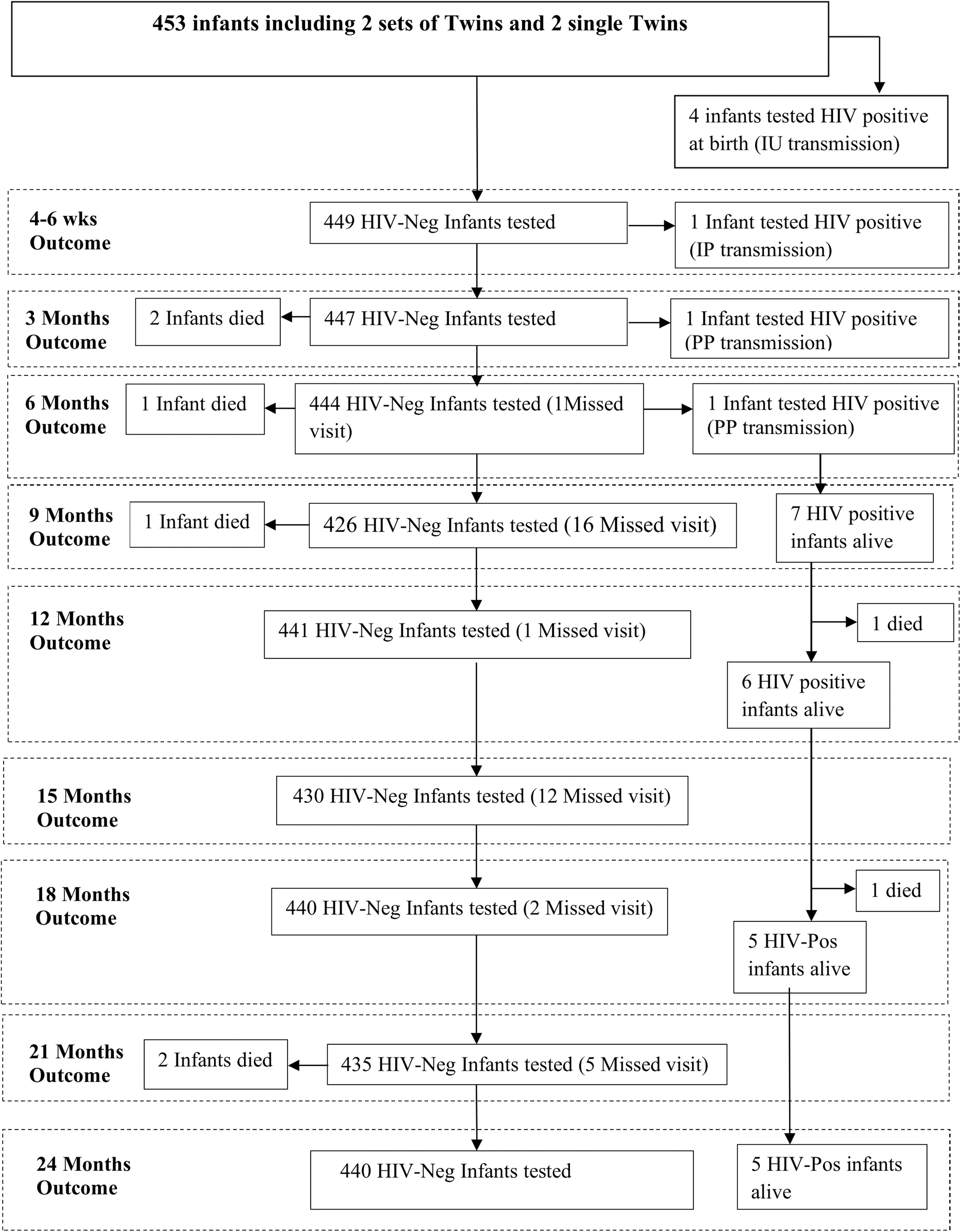
The figure shows HIV testing outcomes of the enrolled infants at birth, 4–6 weeks, 3 months, and every three months thereafter up to 24 months, as well as outcomes at each visit. IU, intrauterine; IP, intrapartum; PP, postpartum; HIV-Neg, HIV negative; HIV-Pos, HIV positive.
The sixth infant tested HIV negative at birth and at the 4–6-week follow-up visit, but subsequently first tested HIV positive at 3 months, and the seventh infant was HIV negative at birth, 4–6 weeks, and 3 months, but subsequently first tested HIV positive at 6 months. These two infants were classified as having PP transmission. The PP transmission rate was 0.44% (95% CI 0.01–1.7%). These results are similar to those that we reported after the 6-month follow-up of the cohort. 4
Characteristics of HIV-positive and HIV-negative infants
The majority (88.30%) of infants in both the HIV-positive and HIV-negative groups had normal birth weights, >2.5 kg. All the infants in the two groups, except one set of twins in the HIV-negative group (the mother was not able to produce sufficient breast milk), were exclusively breastfed in the first six months of life. The majority of infants in both groups (100% and 90.58% HIV positive and HIV negative, respectively) were from vaginal deliveries (Table 2). 4
Characteristics of HIV-Infected and HIV-Uninfected Infants
The table shows characteristics of HIV-infected and HIV-uninfected infants.
Baseline characteristics of mothers who transmitted HIV infection to their infants
Table 3 shows the characteristics of the seven transmitters. The small numbers of IU (4), IP (1), and PP (2) transmitters were considered not adequate to conduct any statistically meaningful comparison of the two groups, neither can we do any analysis to identify the risk factors associated with the three modes of transmission. All the transmitters were in their third trimester of pregnancy at enrolment. All, but one had been on cART for <6 months and all had detectable VL (>1000 cps/mL). The only mother who had been on cART for 5.92 years was on her third-line cART, while the remainder were on their first-line cART regimen. 4
Baseline Characteristics at Enrolment, of the Seven Mothers Who Transmitted HIV-1 to Their Infants, and Mode of HIV-1 Transmission
The table shows the baseline characteristics at enrolment, of the seven mothers who transmitted HIV-1 to their infants, and mode of HIV-1 transmission.
3TC, lamivudine; BMI, body mass index; cART, combination antiretroviral therapy; EFV, efavirenz; NVP, nevirapine; TDF tenofovir fumarate.
Maternal virologic responses to cART and retention in care
Table 4 shows the maternal virological responses and retention rates over the 24-month follow-up period. At enrolment, the majority of mothers, 61.47%, had VL <20 cps/mL. The proportion of mothers with VL <20 cps/mL increased to 89.81% at 6 months, and 89.79%, 90.00%, and 88.94% at 12, 18, and 24 months, respectively. Inversely, the proportion of mothers with VL >1000 cps/mL decreased from 26.11% at enrolment to 3.40% at 24 months. Retention in care at each of the four visits was greater than 98% with the rate at 24 months being 98.95% (Table 4).
Maternal Virologic Responses to Combination Antiretroviral Therapy and Retention in Care
This table shows maternal virological response to cART quantified as VL and retention in care. Maternal VL was quantified at enrolment into the study and every 6 months thereafter up to 24 months.
cART, combination antiretroviral therapy; VL, viral load.
Infant virologic responses
Table 5 shows VL of the seven infected infants before and after cART initiation. At 6 months, one infant had insufficient specimen, one missed the visit, and VL was <1000 cps/mL for three of five infants who had VL quantified. At the 12-month visit, one infant had died and two missed the visit, VL was quantified for four infants, three of four infants had undetectable VL, and one was viremic. One infant had died by the 18-month visit; of the five live infants, four had VL <1000 cps/mL, while one had VL of >1000 cps/mL. At 24 months, four of five live infants had undetectable VL, while the one who was viremic at 18 months remained viremic.
Infant Virological Responses to Combination Antiretroviral Therapy
Infant VL was quantified before commencement of combination antiretroviral therapy and every 6 months thereafter up to 24 months.
cART, combination antiretroviral therapy; N/A, not applicable; VL, viral load.
Infant mortality in the first 24 months of life
Eight infants (8/453) died by 24 months, giving an estimated infant mortality rate of 1.78% (95% CI 0.90–3.53). Six of these infants tested HIV negative at their last scheduled clinic visit before they died. The cause of death for the first infant, after testing HIV negative at birth and at 4–6 weeks, is unknown. The second infant who tested HIV negative at birth and at 4–6 weeks died of respiratory asphyxia. The third infant who tested HIV negative at birth, at 4–6 weeks, and at 3 months died of pneumonia.
The fourth infant died of unknown cause, having tested HIV negative at birth, at 4–6 weeks, and at 3 months. The fifth infant who was HIV infected and on cART died from severe dehydration, secondary to acute gastroenteritis. The sixth infant who was HIV uninfected died from bleeding from the mouth and nose. The seventh infant, HIV uninfected, drowned in an unprotected toilet, and the eighth infant on cART, but viremic, died of immunosuppression. 4
Maternal mortality in the first 24 months of life
Only one mother, with undetectable VL, died from severe anemia, dehydration, and severe weight loss by the 12-month scheduled clinic visit, giving an estimated mortality rate of 0.21% (95% CI 0–1.17).
Discussion
Our study, conducted at a primary health clinic, is the first in Zimbabwe to report on the cumulative MTCT rate and timing of MTCT, as well as infant and maternal mortality in the era of Option B+.
The cumulative MTCT rate of 1.55% is within the reported rates of 1% to 2% for HIV-infected nonbreastfeeding women, who were on various cART regimens in the developed countries. 6 Before introduction of cART for PMTCT, the rate of MTCT in the early 2000 varied from 15% to 45% in breastfeeding sub-Sahara African mothers, 7 as we previously stated. 4
There were very few studies conducted in sub-Saharan Africa to investigate the contributions of IU, IP, and PP transmissions before the introduction of antiretroviral drugs (ARVs). A large study conducted in Zimbabwe reported the IU, IP/early PP, and late PP transmission rates of 9.7%, 16%, and 5.5%, respectively, in the first 6 months of life. 4,5 In sharp contrast, in this study, the IU, IP, and PP transmission rates were 0.88%, 0.22%, and 0.44%, respectively, among infants followed up prospectively from birth up to 24 months postnatally.
The IU rate in this study is comparable to the 0.80% and 0.91% for HIV-infected women in Botswana, who commenced dolutegravir/tenofovir/emtricitabine and efavirenz/tenofovir/emtricitabine cART regimens, respectively, during pregnancy. 8 The Botswana study reported IU transmission rate only among infants followed up to six months. Another study in South Africa also reported only the IU transmission rate of 0.90%, following the introduction of Option B+, 9 as we previously stated. 4
Our study showed that the majority of MTCT, during the first 24 months of life, occurs IU. This is a cause for concern as in Zimbabwe and indeed in many other countries in sub-Saharan Africa, as diagnosis of HIV-1 infection in infants born to HIV-infected women starts at 4–6 weeks postnatally. The 4–6 weeks was decided to coincide with the infants' first immunization clinic visit and additionally because of the financial constraints and logistics to conduct EID at birth. 4 However, the delayed EID will lead to delayed commencement of cART for the IU HIV-infected infants with a consequent mortality risk, which begins to increase at about 3–4 weeks of age, and may be as high as 10% by age 2 months for infants with IU or IP transmission. 10
Six of seven mothers who transmitted HIV infection to their infants (transmitters) were in their third trimester when they enrolled into the study, had been on cART for <6 months, and were viremic. The seventh transmitter who had been on cART for >5 years was also viremic and on third-line cART regimen. The switch from first cART regimen to alternate regimens was based on virological failure and not drug resistance testing (DRT) as this is not offered in public health care institutions in Zimbabwe and indeed in many countries in sub-Saharan Africa because of the high cost of DRT. Surprisingly, though, there was also a small proportion (24.77%) of women, in their third trimester, who had been on cART for <6 months and were viremic at enrolment, but did not transmit HIV-1 to their infants. 4
Recent studies in the era of cART have reported children being infected, despite the use of ARVs for PMTCT. 11 Previous studies conducted in the pre-cART for PMTCT era identified high VL as a risk factor for MTCT. 12 –16 All the transmitters in this study had detectable VL at enrolment into the study. It is plausible that the transmitters had already transmitted HIV-1 to their fetuses/infants before commencement of cART. Thus, early initiation of cART by HIV-infected women may be key to prevention of IU MTCT. Indeed, a study conducted in France reported zero MTCT in HIV-infected women who were on cART with undetectable VL before conception and continued cART throughout pregnancy and had undetectable VL at delivery. 17
Before the introduction of cART for PMTCT, one-third to one-half of PP transmission in sub-Saharan Africa was attributable to breastfeeding. 18 –21 Breastfeeding, mostly in the form of mixed feeding, is widely practiced in sub-Saharan Africa. Studies that were conducted in the early years of the HIV/AIDS epidemic reported lower PP transmission risk and mortality in exclusively breastfed infants compared to infants who received mixed feeding. 22 –24 Subsequently, several studies using short-course antiretroviral regimens given to either the mother or infant during breastfeeding also reduced PP transmission rate. 25 –31
All, but one mother in this study exclusively breastfed in the first 6 months of life. The PP transmission rate of 0.44% in our study in the first 6 months appears to indicate that the risk of PP transmission through breastfeeding can be reduced if the mothers are on cART and are virologically suppressed as the PP transmission rate remained the same after the introduction of mixed feeding after 6 months up to 24 months.
In addition to the good maternal virologic responses to cART implying good adherence, our study also showed good rates of retention in care throughout the 24-month study period. In sharp contrast, several studies that have been conducted in sub-Saharan Africa in the era of Option B+ have reported high levels of poor adherence to cART, loss to follow-up, and low levels of retention in care during or after pregnancy. 32
The majority of HIV-infected infants also showed good responses to cART. There is dearth of data on virologic responses to cART among infants in sub-Saharan Africa in the era of Option B+.
The infant mortality rate of 1.78% at the end of the 24-month follow-up period is in sharp contrast with the 11.8% in the absence of any ARV. Before the introduction of ARVs, the majority of infants born to HIV-infected mothers, who died, were HIV infected. 5 Intriguingly, in this study, the majority (6/8) of infants who died tested HIV negative at the last scheduled clinic visit before they died. The only two HIV-infected infants on cART, who died, were viremic. Their mothers were also viremic from the period they enrolled into the study up to the 24-month follow-up period. The virological failure of the mother-infant pairs may have been due to HIV drug resistance.
The maternal mortality rate of 0.21% in the era of Option B+ in our study is lower than the reported rates of 2.6% and 2% at 6 and 12 months, respectively, post-cART initiation in Cameroon 33,34 and 0.5% in Malawi after 1 year on cART. 35
Our study has some limitations and strengths. We may have underestimated the IU transmission rate and the overall MTCT rate. We do not know the HIV infection status of the 12 born live babies, who died before enrolment. Further, we also do not know the HIV infection status of the 27 babies withdrawn from the study at birth before being HIV tested. The major reason for withdrawal of mothers and their infants from continued participation in the study was relocation from the study area to other parts of the country. 4
Because of financial constraints, we were not able to conduct DRT, which could have been useful in switching the few women who were viremic to more effective cART regimens rather the current practice of switching to alternative cART regimens based on virological failure and self-reported adherence to cART. 4
The major strength of our study is the prospective 24-month follow-up of the mothers and babies, where we showed that POC EID, starting at birth, and VL quantification can be conducted at a primary health care clinic with good clinical outcomes.
In conclusion, in the first 24 months of life, in the era of Option B+ cART, IU transmission is the major route of MTCT. The cumulative MTCT rate of 1.55% and low maternal and infant mortality rates of 0.21% and 1.78%, respectively, contribute to growing evidence that Option B+ cART not only drastically reduces MTCT but also maternal and infant mortality.
Footnotes
Authors' Contributions
L.S.Z. and D.A.K. made substantial contributions to the conception and design of the study. L.S.Z., M.M.C., W.B., T.B., and D.A.K. substantially contributed to acquisition of data. L.S.Z., T.B., M.M.C., and W.B. substantially contributed to data analysis. L.S.Z. prepared the article. L.S.Z., T.B., M.M.C., and W.B. revised it critically for important intellectual content. L.S.Z., T.B., M.M.C., and W.B. gave final approval of the submitted version of the article.
Acknowledgments
We thank the study participants and their families and the Mabvuku Polyclinic staff. We also thank Maureen C.F. Sahanga, Janice Martin, and Tafadzwa D. Nyakubaya for conducting the laboratory tests, and Brian Nyauzame and Mutsa R.J. Makunike for data capture.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This study is part of the EDCTP2 program supported by the European Union (Grant Nos: 1051 TESA II EDCTP-RegNet2015 and TESA III EDCTP CSA2020NoE-3104). The funder was not involved in the study design, collection, analysis and interpretation of data, writing of the report, and decision to submit the article for publication.
