Abstract
Preventing HIV transmission is a crucial step in ending the HIV epidemic. Safe and effective pre-exposure prophylaxis (PrEP) has been available in the United States since 2012. We set out to determine if persons at greatest risk for HIV acquisition were receiving HIV PrEP. HIV-negative individuals from the Observational Pharmaco-Epidemiology Research & Analysis (OPERA) cohort who were prescribed daily PrEP were contrasted with newly diagnosed HIV persons without PrEP use between July 16, 2012 and October 31, 2020 to determine if the PrEP prescriptions reached the populations who were seroconverting. Poisson regression was used to estimate incidence rates of seroconversion to HIV among PrEP initiators, as well as new diagnoses of sexually transmitted infections among both the PrEP group and the newly HIV+ group. Out of the 14,598 PrEP users and 3558 persons newly diagnosed with HIV in OPERA, demographics varied widely. Older individuals, those of non-Black race, men, nonintravenous (IV) drug users, and those with commercial insurance were proportionally overrepresented among those prescribed PrEP compared to persons newly diagnosed with HIV during the same time period. Over 82% of new HIV+ individuals received care in the southern United States compared to only 45% of PrEP users. Seroconversion to HIV among PrEP users was generally uncommon, although more frequent among those who identified as Black individuals, especially in the 13–25 years old age range. In conclusion, providers need innovative programs to better identify, educate, and link those at greatest risk of HIV acquisition, especially young people, women, Black individuals, and IV drug users, to PrEP.
Introduction
According to the Centers for Disease Control and Prevention (CDC), there have been around 38,000 newly diagnosed people per year between 2014 and 2018; 1 as of 2018, about 1.2 million people were living with the human immunodeficiency infection (HIV). 2 While overall incidence went down slightly over this period, some groups experienced increases in new HIV diagnoses. 2 Treatment alone cannot stop the HIV epidemic. 3 –5 Prevention has been identified as a key piece of any strategy to end the epidemic. However, prevention is most efficient when targeted to those at greatest risk.
Pre-exposure oral chemoprophylaxis (PrEP) with tenofovir disoproxil fumarate (TDF)/emtricitabine (FTC) was first approved in the United States in 2012; 6 PrEP with tenofovir alafenamide (TAF)/FTC was approved in 2019. 7 Both PrEP regimens are safe and effective in a variety of populations, protecting against sexual and intravenous drug use (IVDU) transmission. 8 –18 However, risk compensation as a result of feeling protected by PrEP is a concern. 19 Increases in condomless sex and sexually transmitted infections (STI) have indeed been noted in observational studies. 13 PrEP could also be viewed as an opportunity to provide education and encourage STI testing. 20
The World Health Organization (WHO) recommends that PrEP be offered to individuals at substantial risk of HIV infection [i.e., incidence rate (IR) >3 per 100 person-years without PrEP]. 21 The CDC has estimated that 492,000 sexually active men who have sex with men (MSM), 115,000 people who inject drugs, and 624,000 heterosexually active adults would benefit from PrEP. 22 In the United States, racial, ethnic, and geographic disparities in PrEP access have been reported. 23 –26 A cross-sectional study of population-level data assessing whether PrEP prescriptions are proportional to the underlying epidemiological need in the United States found considerable variation in the number of PrEP users relative to the number of new HIV diagnoses for populations stratified by geographic location, policies, race, ethnicity, poverty level, sex, and age. 25,26
This study sought to determine if those prescribed PrEP represented those at greatest risk for HIV infection in the United States. We operationalized this by characterizing the HIV-negative population being prescribed PrEP in the Observational Pharmaco-Epidemiology Research & Analysis (OPERA) cohort compared to the HIV-negative individuals not prescribed PrEP who seroconverted to HIV-positive during the same calendar period, under the assumption that seroconverters would have benefited from PrEP. Further, we quantified the incidence of select STIs between the groups, as well as the incidence of HIV seroconversions on PrEP.
Methods
Study population
The study population was selected from the OPERA cohort, a collaboration of 14 health care organizations with over 80 clinics in 18 US states and territories from which electronic health records are uploaded daily or weekly (Supplementary Figs. S1 and S2). At the time of the analysis, over 748,000 individuals were prospectively followed through their electronic health records during their regular care with their primary care providers (Supplementary Table S1). The study population included HIV-negative individuals who were at least 13 years of age at the time of a prescription of daily PrEP (TDF/FTC or TAF/FTC) or a new diagnosis of HIV between July 16, 2012 and October 31, 2020. Individuals with hepatitis B virus (HBV) were excluded as these individuals may have been on TDF/FTC or TAF/FTC for treatment rather than PrEP. Among the PrEP users, those who were prescribed any other PrEP modality [e.g., a variety of antiretroviral therapy (ART) as PrEP ranging from monotherapy to full regimens off-label use] were also excluded, as it was not the focus of this work.
The comparison group consisted of people newly diagnosed with HIV (non-PrEP seroconverters). It can be surmised that seroconverters would have received some protection from PrEP use had they been prescribed it and that their behaviors represent those of individuals at high risk of acquiring HIV. Thus, comparing seroconverters to PrEP users can identify vulnerable subgroups of individuals who should be targeted for PrEP use. Among those newly diagnosed with HIV, individuals exposed to ART before seroconversion via PrEP or post exposure prophylaxis, or had a viral load <1000 copies/mL at the time of HIV diagnosis (an indicator of likely prior ART use), and those who did not have a prior documented HIV-negative test were excluded as they may not reflect individuals who were true seroconverters at that specific timepoint (Fig. 1). All individuals were followed from the date of PrEP initiation or HIV seroconversion to study end (October 31, 2020).
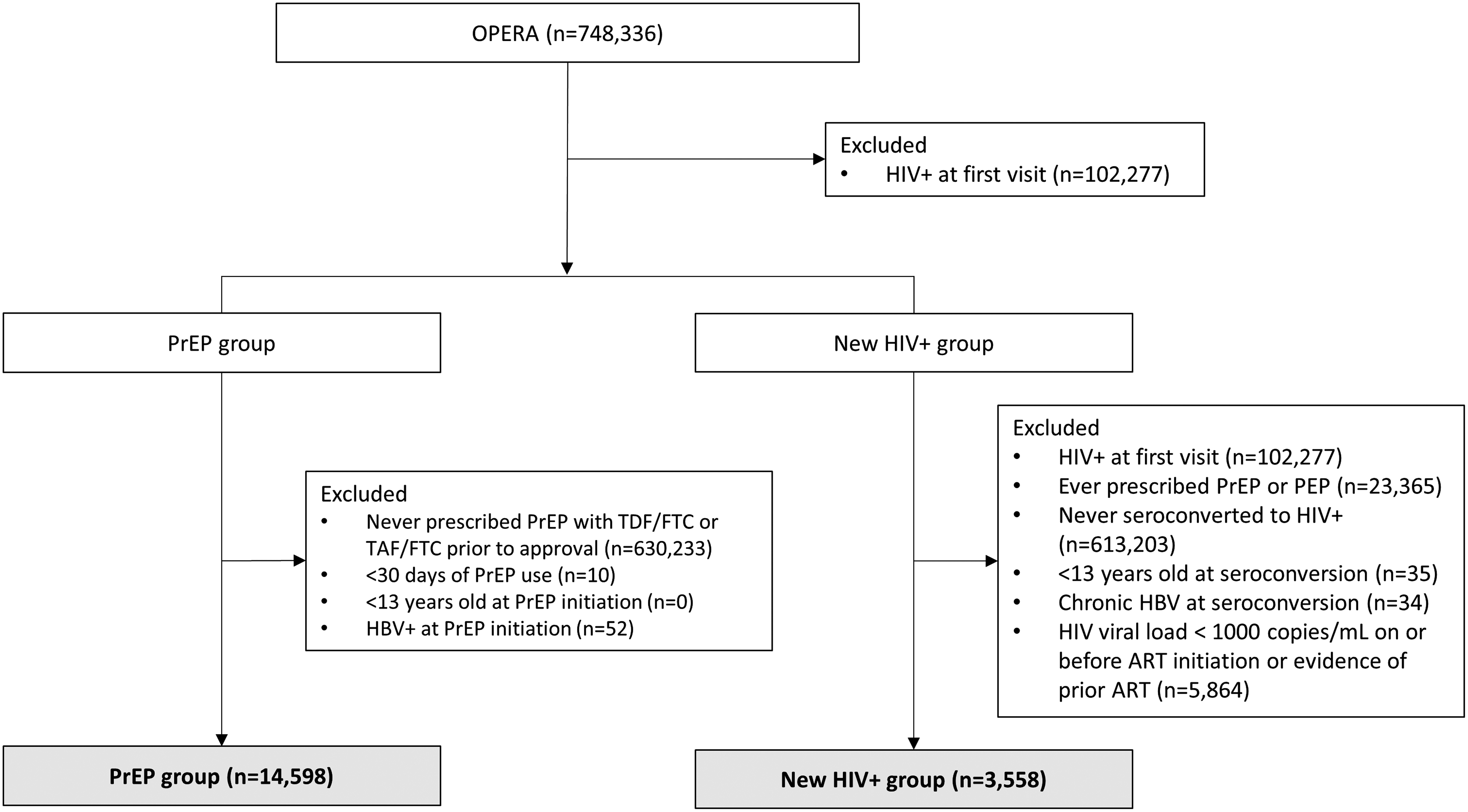
Flow diagram of study inclusion. ART, antiretroviral therapy; HBV, hepatitis B virus; OPERA, Observational Pharmaco-Epidemiology Research & Analysis; PrEP, pre-exposure prophylaxis; TAF, tenofovir alafenamide; TDF/FTC, tenofovir disoproxil fumarate/emtricitabine.
OPERA® complies with all HIPAA and HITECH requirements and received annual Institutional Review Board (IRB) approval from Advarra IRB, including a waiver of informed consent and authorization for the use of protected health information.
Measurements
Demographic characteristics were collected from intake information as well as social and behavioral history notes in the electronic health record data. Reporting and recording of these vary by provider-patient relationship and tend to grow more descriptive over time. Payer information was collected from the billing software and categorized as “other” when a patient received a local grant to pay for their care; missing payer data occurred when a patient was cared for pro bono or received other support not billed through the software. Clinical characteristics were obtained through the history and present care of the patient.
PrEP initiators were HIV-negative individuals prescribed TDF/FTC or TAF/FTC for the first time during the study period. Most individuals on PrEP for <30 days had difficulty getting PrEP insurance coverage and were thus excluded. PrEP discontinuation was defined as a gap of >45 days without a refill or a new PrEP prescription. With prescription data, we have observed that most gaps of <45 days represent delayed start and/or incomplete adherence as opposed to discontinuations.
Seroconverters were HIV-negative individuals with a new diagnosis or positive laboratory result for HIV during the study period. STI were defined as a new diagnosis or positive laboratory result for active syphilis, gonorrhea, chlamydia, trichomoniasis, chancroid, lymphogranuloma venereum, Mycoplasma genitalium, acute hepatitis C virus, or acute HBV during follow-up.
Statistical analyses
To assess whether PrEP prescriptions reached the subpopulations most at risk of HIV infections, baseline demographic and clinical characteristics were described using median and interquartile range (IQR) for continuous data and frequencies and proportions for categorical data. Groups were compared using Wilcoxon rank-sum test for continuous variables and Pearson's chi-square test for categorical variables. IRs of seroconversions with exposure to PrEP were estimated using Poisson regression among PrEP initiators. IRs of new diagnoses of STIs were estimated among both the PrEP group and the New HIV+ group and defined as new diagnoses among individuals with neither history of nor prevalence of STIs at baseline.
Results
The PrEP analysis population consisted of 14,598 persons initiating PrEP for the first time between July 16, 2012 and October 31, 2020 and met all eligibility criteria (Fig. 1) were followed for a median of 15 months (IQR: 12– 26). The majority were prescribed TDF/FTC (n = 8283), some started with TDF/FTC and switched to TAF/FTC (n = 3968), and fewer took TAF/FTC exclusively (n = 2347). The newly HIV-diagnosed population included 3558 HIV-negative individuals who seroconverted to HIV-positive between July 16, 2012 and October 31, 2020 and met all other eligibility criteria (Fig. 1) were followed for a median of 33 months (IQR: 18–56). PrEP uptake showed important increases over the observation period, from 115 PrEP initiations in 2012 to 3821 initiations in 2019 (2391 initiations in 2020 through October). Meanwhile, new HIV diagnoses were relatively stable until 2020.
When comparing demographic characteristics (Table 1), the PrEP group tended to be older than the New HIV+ group, with the greatest difference being in the youngest age group, 13–25 years of age (PrEP: 24%; New HIV+ 32%). A greater proportion of women, Black individuals, and IVDUs were diagnosed with HIV than were initiated PrEP during this period. Over 80% of new HIV diagnoses occurred in the south of the United States, while only 45% of PrEP prescriptions originated there. While most PrEP initiators and New HIV+ patients were covered by commercial insurance, no information was available on PrEP coverage specifically (Table 1).
Demographic Characteristics of Pre-Exposure Prophylaxis (PrEP) Users and Individuals Newly Diagnosed with HIV, n (%)
Statistical significance may be an artifact of the large sample size and may not represent clinically meaningful differences between groups.
ADAP, AIDS Drug Assistance Program; IVDU, intravenous drug use; MSM, men who have sex with men; PrEP, pre-exposure prophylaxis.
IRs of HIV seroconversion after PrEP initiation were low across most groups with the exception of Black PrEP initiators between 13 and 25 years of age [2.19 cases per 100 person-years, confidence interval (CI): 1.58–3.03], which was double the rate among Black PrEP initiators between 26 and 49 years of age (1.12 cases per 100 person-years, CI: 0.78–1.60; Fig. 2). Compared to PrEP nonseroconverters, PrEP seroconverters tended to be younger and were more likely to be male and Black (Table 2). Seroconversion was observed after a median of 13.6 months (IQR: 7.1–25.6) following PrEP initiation. Most (70%) had at least one visit every 90 days and 75% still had a prescription for PrEP when they seroconverted. In comparison, PrEP users who did not seroconvert were less likely to have at least one visit every 90 days (62%; Table 3).
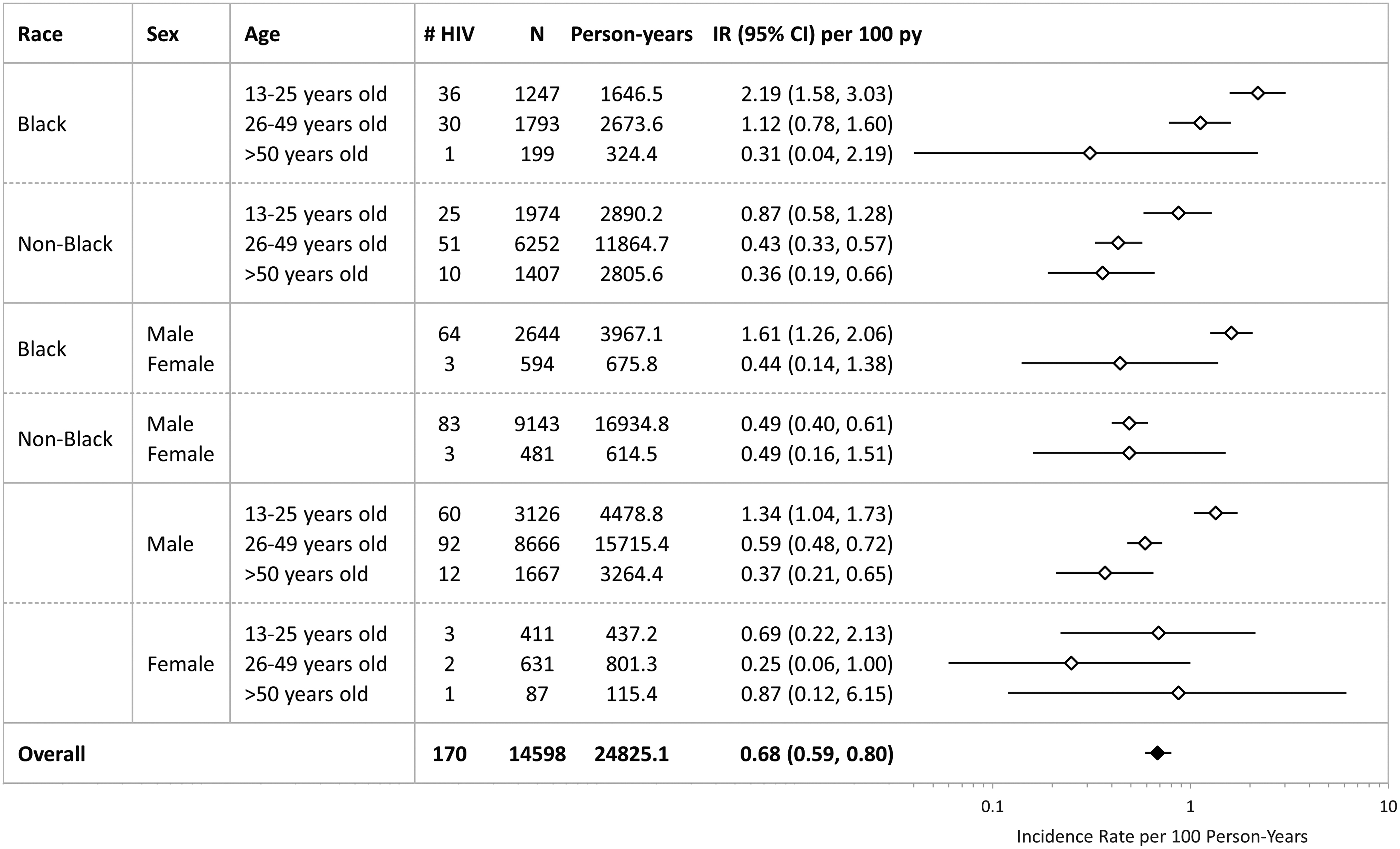
HIV seroconversion IRs per 100 person-years for PrEP recipients (N = 14,598) by demographic groups. Individuals with missing race or sex were excluded from stratified estimates as appropriate. IR, incidence rate; PrEP, pre-exposure prophylaxis.
Demographic Characteristics of Pre-Exposure Prophylaxis (PrEP) Nonseroconverters and Pre-Exposure Prophylaxis (PrEP) Seroconverters, n (%)
Statistical significance may be an artifact of the large sample size and may not represent clinically meaningful differences between groups.
ADAP, AIDS Drug Assistance Program; IVDU, intravenous drug use; MSM, men who have sex with men; PrEP, pre-exposure prophylaxis.
Pre-Exposure Prophylaxis (PrEP) Persistence
Statistical significance may be an artifact of the large sample size and may not represent clinically meaningful differences between groups.
IQR, interquartile range; PrEP, pre-exposure prophylaxis.
Past (prevalence) and new diagnoses of STIs (incidence proportions) were compared between the PrEP and New HIV+ groups; the large sample size may have resulted in statistically significant differences despite the absence of clinically relevant differences between groups (Table 4). When accounting for differential follow-up time between groups, the overall IRs per 100 person-years for new STIs were higher among PrEP users (IR: 14.76; 95% CI: 14.29–15.25) than among non-PrEP seroconverters (IR: 10.02; 95% CI: 9.45–10.61; Fig. 3). When assessing the IR for each of the most common STIs, both gonorrhea and chlamydia rates were higher among PrEP users than the non-PrEP seroconverters. However, rates per 100 person-years of syphilis were lower among PrEP users (IR: 4.21; 95% CI: 3.97–4.48) than the non-PrEP seroconverters (IR: 5.45; 95% CI: 5.04–5.90; Fig. 3).
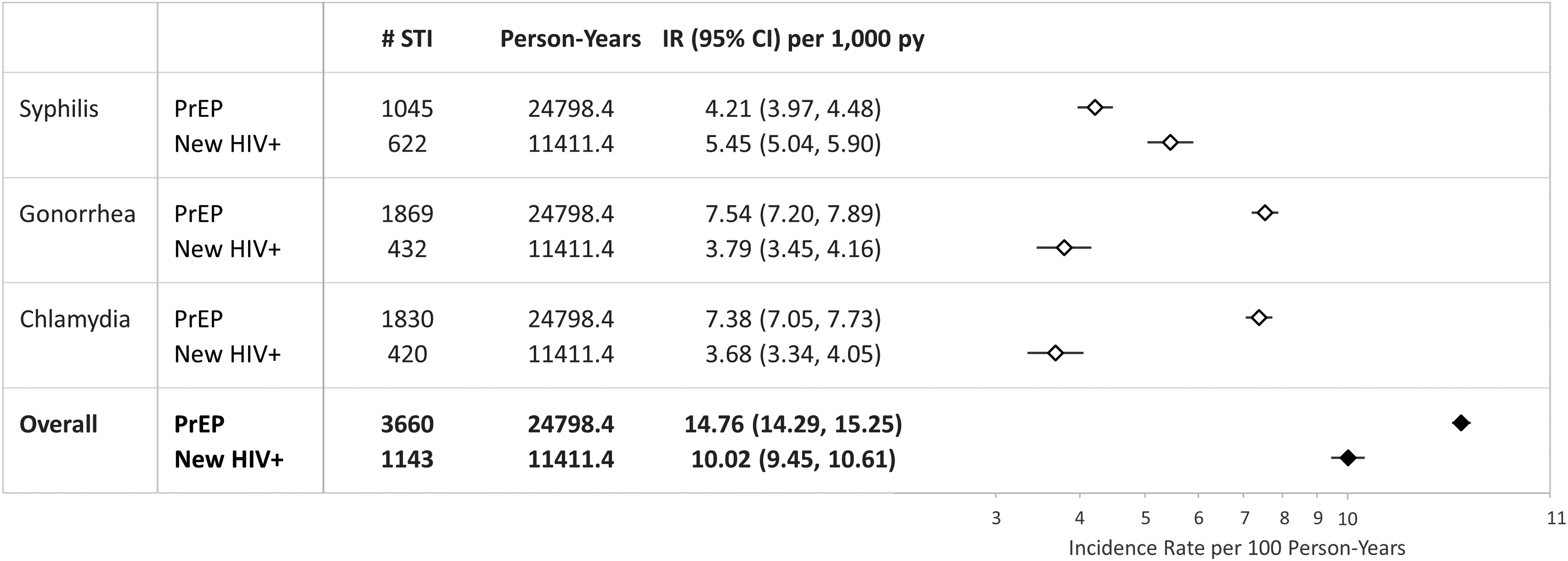
STIa IRs per 100 person-years for PrEP recipients (N = 14,598) versus new HIV+ individuals (N = 3558). aOverall STI: new diagnosis or positive laboratory result for active syphilis, gonorrhea, chlamydia, trichomoniasis, chancroid, lymphogranuloma venereum, Mycoplasma genitalium, acute hepatitis C virus, or acute HBV during follow-up. CI, confidence interval; HBV, hepatitis B virus; IR, incidence rate; PrEP, pre-exposure prophylaxis; PY, person-years; STI, sexually transmitted infection.
Pre-Existing and Newly Diagnosed Sexually Transmitted Infections, n (%)
History defined as a diagnosis of a given infection on or up to 3 months before start of PrEP or diagnosis of HIV.
Statistical significance may be an artifact of the large sample size and may not represent clinically meaningful differences between groups.
NA, not applicable; PrEP, pre-exposure prophylaxis; STI, sexually transmitted infection.
Discussion
In this large population of new PrEP initiators and individuals newly diagnosed with HIV, those prescribed PrEP were more likely to be older and were less likely to be women, Black individuals, IVDU, or MSM+IVDU than those newly diagnosed with HIV without PrEP during the same calendar period. Of note, the highest incidence for seroconversion among PrEP users was among Black individuals 13–25 years of age in alignment with the CDC report showing that 21% of new HIV diagnoses in 2018 occurred among youth 13–24 years of age. 27 While the overall trend of new HIV infection in youth in the United States has been declining from 2010 to 2017, 53% of youth between 13 and 24 years of age who were newly diagnosed with HIV in 2017 were Black individuals. 28 Prevention of HIV in this age group is crucial, as younger people living with HIV are more likely than older individuals to have suboptimal adherence to ART 29 and to fall out of care, 30 leading to an increased risk of virologic failure. 28,30
Thus, HIV prevention is of the utmost importance among younger people of color. Tailoring PrEP education, administration, and support to individuals between 13 and 25 years of age at highest risk for contracting HIV is of paramount importance. Unfortunately, as of the spring of 2018, only 55% of young MSM aged 15–17 years had ever heard of PrEP. 31 In OPERA, only 24% of PrEP initiators were between 13 and 25 years of age, while this age group represented 32% of the total pool of new HIV infections. These results reflect those from a study utilizing a national health data company aggregating clinic, patient, and provider data in the United States (Source Healthcare Analytics). 25 Indeed, in the fourth quarter of 2017, in the United States, 70,395 individuals received a prescription for PrEP, representing 26 PrEP users per 100,000 population. The prevalence of PrEP use was considerably lower among individuals <24 years of age (15.2 PrEP users per 100,000 population), compared to those aged 25–54 years (range, 28.1–61.5 PrEP users per 100,000 population). 25 Moreover, this study estimated that in this age group, for each new HIV diagnosis, there were only 0.9 PrEP users, suggesting that the level of PrEP provision did not meet the underlying epidemiological need for youths in the United States 25
The majority of PrEP initiators in OPERA returned for at least one follow-up appointment, and 53% remained on PrEP at study end over a median of 15 months (IQR: 12–27) of follow-up. This implies that 47% discontinued PrEP, although reasons for discontinuation are unknown. Similar or slightly higher levels of PrEP retention have been described in other studies. 32 –37 Of note, this study found that as many as 75% of PrEP seroconverters still had a PrEP prescription at the time of seroconversion, suggesting a suboptimal adherence to PrEP. Pharmacokinetic studies have suggested that at least four doses/week for rectal exposure and at least six doses/week for other exposures are necessary to protect against HIV. 38 –40 This highlights the importance of providing adherence support programs to ensure the success of PrEP in the United States.
Incidence of STIs have been increasing over time in the United States, with a 19% increase in chlamydia, a 63% increase in gonorrhea, and a 71% increase in primary and secondary syphilis from 2014 to 2018. 41 In 2018, the CDC reported high rates (cases per 100,000 population) of chlamydia (539.9), gonorrhea (179.1), and syphilis (10.8), but only three cases of chancroid. 41 Similarly, in OPERA, syphilis, gonorrhea, and chlamydia were the most common STIs in both the PrEP and New HIV+ groups. In the OPERA study, IRs per 100 person-years of STIs overall were statistically significantly higher among PrEP users (14.76; 95% CI: 14.29–15.25) compared to non-PrEP seroconverters (10.02; 95% CI: 9.45–10.61) driven by gonorrhea and chlamydia infection. However, IRs per 100 person-years of syphilis were statistically significantly lower among PrEP users (4.21; 95% CI: 3.97–4.48) than non-PrEP seroconverters (5.45; 95% CI: 5.04–5.90). The synergistic interplay between HIV and syphilis has been a well-known phenomenon, both behaviorally and biologically, notably with syphilitic ulcers promoting HIV transmission and acquisition. 42,43 Syphilis is therefore an indicator of high HIV risk, and any individual presenting with syphilis should receive sexual risk reduction counseling and STI screening and be offered PrEP immediately if HIV-negative.
Mitigation of the protective effects of PrEP through risk compensation has been a source of concern. Risk compensation may occur if PrEP users change other behaviors because they feel protected (e.g., increased condomless sex). 19 While several PrEP clinical trials have assessed risk compensation behavior changes and found no increases in sexual risk behavior or STI incidence, such trials are not representative of the real-world PrEP and STI counseling experience. 19 Several observational studies have reported increases in condomless sex and STI diagnosis with PrEP. 44 –48 Of note, if PrEP use is accompanied by a higher frequency of STI testing, increased STI rates are to be expected. 20 However, while higher STI screening in the PrEP group may have driven some of the differences observed in other studies comparing PrEP users to themselves before PrEP use, or to HIV-negative individuals not on PrEP, it is less likely in this OPERA study, as individuals newly diagnosed with HIV should also be offered regular STI screening and counseling.
Limitations of this study include the influence of early prescribers of PrEP who may not have been targeting its use to “at risk” populations in the same way as later prescribers of PrEP, which could have skewed the PrEP population characteristics. However, the early years had the fewest PrEP initiators, with over 75% of all new PrEP use recorded between 2017 and 2020, so this effect should be modest at worst. In addition, this study does not account for the impact of the COVID-19 epidemic on HIV prevention and testing, which may have impacted 2020 estimates. However, the impact of the pandemic has varied greatly depending on the setting and geographic location. 49 –51 In addition, racial and socioeconomic disparities in both COVID-19 and HIV testing, infection, and outcomes have been documented. 52 –54
Demographic characteristics were self-reported and, in the case of race and ethnicity, could be left blank, limiting our ability to observe whether individuals of Hispanic decent were adequately provided PrEP. In addition, marital status, risk of infection, and gender identity were gleaned from the social and behavioral history notes of the providers. This information can vary by provider and time in care. The New HIV+ patients were followed for a median of 33 months (IQR: 18–56). In contrast, the PrEP users were followed for a median 15 months (IQR: 12–26). It is possible that screening rates varied between these populations, impacting the IRs. Moreover, not all STIs have the same risk or reflect recent risk behaviors. If focusing on STIs that indicate recent risk behavior while in care, different rates between PrEP users and new HIV+ patients may indicate a combination of differences in provider interventions, patient receptivity to risk modification, or other influences. Identification of these root causes could be used to inform interventions. This is an area for future research efforts.
In addition, this analysis was limited to first PrEP exposure. Large gaps in PrEP were not evaluated and may become a bigger issue as individuals experiment with on-demand PrEP. This too is an area for future research. A generous gap of >45 days between PrEP prescriptions was allowed before qualifying for a discontinuation. This definition was developed because OPERA includes provider prescription data, not pharmacy fill data. It was therefore important to allow for delayed start, suboptimal adherence, and possible use of samples. This approach may have been too conservative in the estimation of PrEP discontinuation. Finally, this analysis did not cover several other sources of disparities in PrEP access, such as socioeconomic status, state policies, or health care provider attitudes or knowledge. 25,26,55,56
In conclusion, although PrEP has been transformative, and its use has been on the rise, significant challenges in its implementation remain. This study revealed that PrEP was under-prescribed to individuals who were at the highest risk for HIV acquisition (13–25 years old, Black people, women, in the southern United States). Hence, raising awareness, reducing stigma, and enhancing PrEP access among the most vulnerable individuals are of paramount importance. The highest incidence of seroconversion among PrEP users was noted among young Black men, underscoring the need to monitor PrEP adherence closely and offer support in this vulnerable population.
Footnotes
Authors' Contributions
K.C.M., J.S.F., V.V., and K.R. share the responsibility for the design of this study. K.C.M., J.S.F., R.K.H., and G.P.F. contributed to the acquisition of data. J.S.F. and L.B. conducted all of the analyses. All authors have contributed to the interpretation of results, have critically reviewed and approved the article, and have participated sufficiently in the work to take public responsibility for its content.
Acknowledgments
In memory of Evelyn Byrd Quinlivan, MD, who brought this work to life. This research would not be possible without the patients and the healthcare providers who participate in the OPERA cohort. In addition, we are grateful for the following individuals: Robin Beckerman (SAS programming), Jeff Briney (QA), Bernie Stooks (Database Mgmt), Judy Johnson (Med Terminology Classification), and Rodney Mood (Site Support).
Author Disclosure Statement
K.C.M. has received research grants from Gilead Sciences, Merck, Janssen, and GSK/ViiV Healthcare and honoraria for Speakers Bureau and Advisory Boards from Gilead Sciences, Merck, Janssen and GSK/ViiV Healthcare; and advisory board participation with Epividian. J.S.F., L.B., and G.P.F. are employed by Epividian, Inc.; Epividian has had research funded by ViiV Healthcare, Merck & Co., Janssen and Gilead Sciences. R.K.H. has received a research grant from Gilead Sciences, speaker honoraria and advisory boards for ViiV Healthcare, BMS, Merck, Gilead Sciences and Janssen, and advisory board participation of Epividian. V.V., A.R., and K.R. are employees of ViiV Healthcare. M.S.S. was an employee of ViiV Healthcare at the time of the analysis. He is currently an employee of HengRui USA. K.R.F. has participated in Epividian advisory boards.
Funding Information
This work was supported by ViiV Healthcare.
Supplementary Material
Supplementary Table S1
Supplementary Figure S1
Supplementary Figure S2
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
