Abstract
The Gender, Race And Clinical Experience (GRACE) study was conducted between October 2006 and December 2008 to evaluate sex- and race-based differences in outcomes after treatment with a darunavir/ritonavir-based antiretroviral regimen. Between June 2010 and June 2011, former participants of the GRACE trial at participating sites were asked to complete a 40-item questionnaire as part of the GRACE Participant Survey study, with a primary objective of assessing patients' characteristics, experiences, and opinions about participation in GRACE. Of 243 potential survey respondents, 151 (62%) completed the survey. Respondents were representative of the overall GRACE population and were predominantly female (64%); fewer were black, and more reported recreational drug use compared with nonrespondents (55% vs. 62% and 17% vs. 10%, respectively). Access to treatment (41%) and too many blood draws (26%) were reported as the best and worst part of GRACE, respectively. Support from study site staff was reported as the most important factor in completing the study (47%). Factors associated with nonadherence, study discontinuation, and poor virologic response in univariate analyses were being the primary caregiver for children, unemployment, and transportation difficulties, respectively. Patients with these characteristics may be at risk of poor study outcomes and may benefit from additional adherence and retention strategies in future studies and routine clinical care.
Introduction
A
The Gender, Race, And Clinical Experience (GRACE) trial was designed to evaluate sex- and race-based differences in the outcomes associated with darunavir/ritonavir-based therapy in treatment-experienced HIV-infected adults. 9 This was a phase 3b, open-label, multicenter trial that enrolled 429 patients. The trial successfully enrolled a high percentage of women (67%) across North America, including women of color, through specific recruitment and retention strategies. 10 Patients received darunavir/ritonavir plus an optimized background regimen for 48 weeks. Sex-based safety and efficacy results have been previously reported. 9
We present here the findings from an exploratory patient survey conducted after the GRACE trial was completed, with the purpose of collecting additional data regarding issues inherent to clinical trials (such as retention). The impact of patients' characteristics, experiences, and opinions on treatment outcomes was assessed.
Methods
Study design of GRACE trial
Detailed study design and results of GRACE have been reported. 9 –15 Briefly, GRACE was a phase 3b, multicenter, open-label, 48-week study conducted in treatment-experienced HIV-infected adults at 65 sites throughout the United States, Puerto Rico, and Canada between October 2006 and December 2008. Patients enrolled in GRACE were treated with darunavir/ritonavir 600/100 mg twice daily in addition to an investigator-selected optimized background regimen.
GRACE participant survey design and objectives
The present study was a noninterventional, multicenter, cross-sectional survey completed by former participants of the GRACE trial. Of the 57 GRACE sites located in the United States and Puerto Rico that enrolled patients, 22 chose to participate in the survey study, which was conducted between June 2010 and June 2011. The primary objective of the survey was to examine GRACE participants' characteristics (beyond data obtained during the study), experiences, and opinions about study participation. The secondary objective was to explore statistically the associations between survey responses and adherence to study medications, study discontinuation, and virologic response. Patients completed a 40-question multiple-choice and open-ended survey in a single visit.
Statistical analyses
Descriptive statistics of survey responses were compiled. Univariate logistic regression was performed to determine the importance of individual survey responses on each of the following GRACE study outcomes: study discontinuation, adherence, and virologic response. Although the current study was not powered for statistical analyses, a p value<0.10 was used as a broad selection criteria for association of survey responses with respective end points.
Results
Patient population and baseline characteristics of survey respondents versus nonrespondents
In the total GRACE population (N=429), 67% were female, with a median age of 43 years (range 19–78; Table 1). Sixty-two percent were black, 22% Hispanic, and 15% Caucasian. Tobacco, alcohol, and recreational drug use were reported by 37%, 35%, and 14% of patients, respectively, and 39% had HIV stage C disease. Median baseline CD4 cell count was 200 cells/mm3 (range 1–1125). Median baseline viral load was 4.71 log10 copies/mL (range 1.7–6.8).
n, 120; b n, 60; c n, 180; d n, 133; e n, 313.
ITT-TLOVR, intent-to-treat time-to-loss of virologic response; VF, virologic failure.
In all, 243 patients who took part in the GRACE trial (57% of the total GRACE population), across 22 participating sites (39% of total GRACE sites), were asked to complete the survey. Of these 243 potential survey participants, 151 (62%) completed the survey; this represents 35% of the overall GRACE population. Survey respondents were predominantly female (64%), with a median age of 44 years (range 19–74), which was similar to nonrespondents (Table 1). However, fewer respondents were black (55% vs. 62% for nonrespondents) and more respondents were Hispanic (29% vs. 23% for nonrespondents). More survey respondents reported tobacco, alcohol, and recreational drug use during their participation in GRACE, compared with nonrespondents (38%, 38%, and 17% vs. 34%, 33%, and 10%, respectively). Fewer respondents had HIV stage C disease (34% vs. 40% for nonrespondents) and more had a higher median baseline CD4 cell count (221 vs. 187 cells/mm3 for nonrespondents). There was no difference in median baseline viral load (4.75 log10 copies/mL for respondents vs. 4.76 log10 copies/mL for nonrespondents). No significant differences in baseline characteristics were observed between sites that participated in the survey versus those that did not participate, ruling out potential bias in participating site selection (Table 1).
Adherence, discontinuation, and virologic response rates of survey respondents versus nonrespondents during participation in GRACE
Adherence rates of ≥95% were reported in 65.6% of survey respondents compared with 58.7% of nonrespondents. Similar rates were observed between survey respondents and participants of the GRACE trial as a whole (65.8% reported ≥95% adherence). 9 Fewer survey respondents had discontinued participation in the GRACE trial compared with nonrespondents (27% vs. 40%), and similar rates of discontinuation were observed between survey respondents and participants in the GRACE trial as a whole (27% vs. 30%, respectively). 9 Survey respondents achieved higher rates of HIV-1 RNA suppression below 50 copies/mL, with week 48 intent-to-treat time-to-loss of virologic response (ITT-TLOVR) values of 61%, compared with 46% for nonrespondents, and TLOVR nonvirologic failure (VF) censored values of 77%, compared with 70% for nonrespondents. However, among the overall GRACE population, 53.4% and 73.2% achieved HIV-1 RNA below 50 copies/mL in the ITT-TLOVR and TLOVR non-VF censored populations. 9
Characteristics of survey participants
While participating in the GRACE trial, 18% of survey respondents did not have health insurance and 27% were on Medicaid (Table 2). When not in the study, 25% of respondents relied on the AIDS Drug Assistance Program, other prescription assistance programs, or paid on their own for their HIV medications. In all, 73% of respondents were high-school graduates. At the time of entry into GRACE, 58% were not working and only 7% were in school. Less than 5% of the survey respondents were homeless or lived in a shelter, and only 2% spent a night in jail while participating in the GRACE trial. The majority (71%) of respondents had children, but only 38% reported that they were the primary caregiver for children/grandchildren. Before enrolling in GRACE, 39% of respondents reported having difficulty taking HIV medications as directed, mainly because they did not like how the drugs made them feel; 15% had trouble keeping medical appointments, which was primarily due to a busy lifestyle. Most respondents (73%) had disclosed their HIV status to others and over half (56%) reported receiving strong support from friends/family.
Not all survey respondents answered every question, resulting in an n that does not always add up to 151. Percentages were calculated based on actual number of responses. ADAP, AIDS Drug Assistance Program; PAPs, Prescription Assistance Programs.
Survey participants' experiences during the GRACE trial
The majority (71%) reported that GRACE was their first HIV study and that they learned about the study from the staff at their clinic (82%; Table 3). The most common reasons to participate in GRACE were the need to access medications (37%) and the desire to contribute to something bigger/help others (36%). Most (79%) felt that receiving other medications in addition to the main study drug, darunavir, was very important in their decision to participate in the GRACE study. The majority (76%) participated in GRACE at their primary HIV care location, and 77% did not have any difficulty arranging transportation to the study site. Nearly all (99%) participants reported being comfortable or very comfortable with the study site, indicating that the site was very flexible with scheduling visits (93%). In most cases, the key contact person at the study site was the study nurse (70%) and most respondents reported that it was not important to them that the key contact person was of the same race/ethnicity (73%) or sex (76%) as themselves.
Not all survey respondents answered every question, resulting in an n that does not always add up to 151. Percentages were calculated based on actual number of responses. ASO, AIDS Service Organization.
Survey respondents' opinions about the GRACE trial
Respondents reported that the best part of the GRACE experience was access to treatment (41%), being part of something bigger (18%), and feeling better (17%) (Table 4). Seventy-six percent felt that the GRACE trial made them feel differently about their health/HIV care, and most (82%) became more focused on their health, with 87% of respondents continuing treatment after GRACE. In all, 68% would be interested in sharing their GRACE experience, and 96% would recommend participation in a clinical trial to others. Some of the worst parts of the GRACE experience as reported by respondents were too many blood draws (26%), travel to the study site (13%), and the opinion that medicines were hard to take (12%). Although 88% reported having completed the GRACE trial, only 74% actually did so. Support from site study staff (47%) was noted as the most important factor in completing the study. Other common success factors were feeling better or healthier (16%), receiving support from family/friends (15%), and having access to medications (13%). Of the 12% who reported not completing the study, the most cited reason for discontinuation was intolerable side effects/not liking how they felt (50%).
Not all survey respondents answered every question, resulting in an n that does not add up to 151. Percentages were calculated based on actual number of responses. bMultiple answers were permitted. cFour of the 133 survey respondents who answered yes did not clarify what was most important in helping them complete the study. dFour of the 18 survey respondents who answered no did not give a reason.
Themes from the open-ended question
The survey respondents were asked to share additional opinions about their experience during GRACE through the open-ended question: “Is there anything else you would like to share about your experience in participating in the GRACE study?” The following themes emerged: (1) Improved adherence: respondents reported an increased awareness of the importance of continuing to take their medications. “I became more aware of the importance of the meds and I take my care more serious now.” (2) Increased self-confidence: respondents reported feeling more confident about themselves and their future. “More self-confident being with the staff that help me a lot.” (3) Better health: respondents felt healthier since participating in GRACE, with improved energy and quality of life. “Thank God for the GRACE study! I loved it. My health was a mess. After I took the medicine, my health was back on track. I am still taking the same medications.” (4) Access to effective HIV medications: respondents reported that the study provided them with medications that they would not otherwise have been able to afford. “I am very blessed to be in this study, because I can't afford the meds for a whole year.” (5) Race: respondents felt that the fear among people of color regarding participation in clinical trials needed to be dispelled and that people of color are now able to access effective treatment. “As a black woman the fear from clinical trials had to be dispelled.” (6) Positive relationship with study staff: respondents felt that the staff was professional, supportive, comforting, nonjudgmental, confidential, caring, and spent extra time to help the study participants. “The most important was the staff because I just felt like I was normal and however I could deal with the medicine and they were there. I appreciated the medicine because I didn't have any. When doing a survey or hiring staff the most important thing is that the staff doesn't judge the people. That's what I like, they were very confidential. They saved my life as well as the medicine. Thanks!”
Associations of responses with clinical outcomes
Univariate analyses were performed to assess any associations between survey responses and clinical outcomes, including adherence to treatment, study discontinuation, and virologic response to study medications.
Adherence was defined as ≥95% compliance with study medications over the course of the study. The following associations were found to be significant at a level of p<0.1 (Fig. 1): increased adherence was associated with not feeling differently about one's own health or HIV care since participating in GRACE, not having children and working. Less adherence was associated with having some difficulties with transportation to study site, not being very comfortable with study site and staff, having trouble taking HIV medications prior to the GRACE trial, or being a primary caregiver for children.
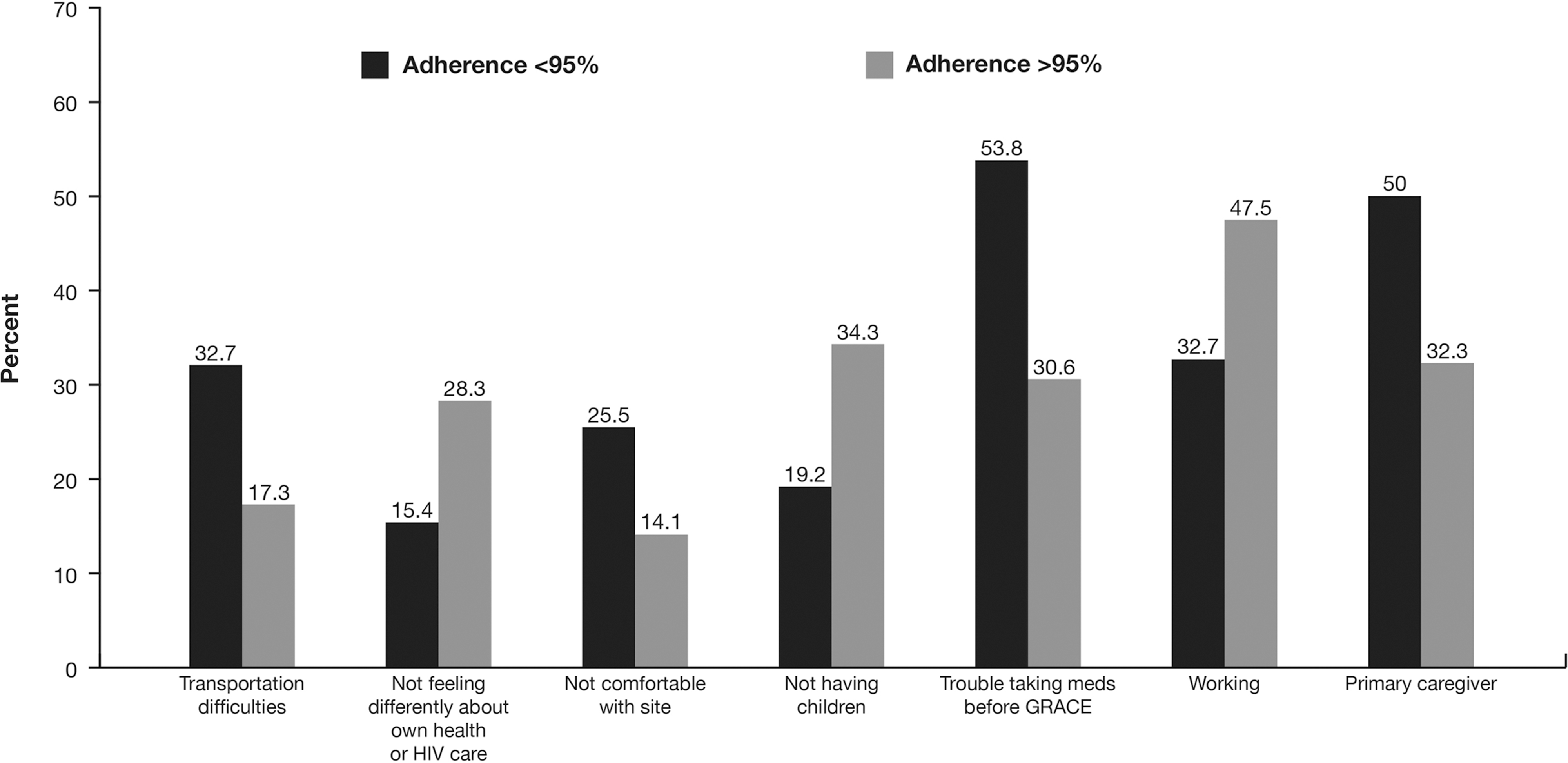
Associations with adherence. Univariate analysis was used to examine associations (p<0.1) between survey responses and adherence to study medication. The p value for univariate selection is based on all levels (survey answers) of the variable (survey question); only one level is shown here. Adherence was defined as compliance with study medications ≥95% over the course of the trial.
Study discontinuation was defined as discontinuing the study for reasons other than VF or adverse events. The following factors associated with study discontinuation were significant at a level of p<0.1 (Fig. 2): having spent at least one night in jail during the trial; not staying on treatment after GRACE; having Medicaid; having difficulty with transportation to study site; choosing not to disclose their HIV status to others; not having a high-school diploma; not feeling it was important that medications other than darunavir were provided during the study; or feeling that it was important that the study nurse was the same sex. Factors associated with study completion (significant at a level of p<0.1) were having private insurance, working, or not having children.
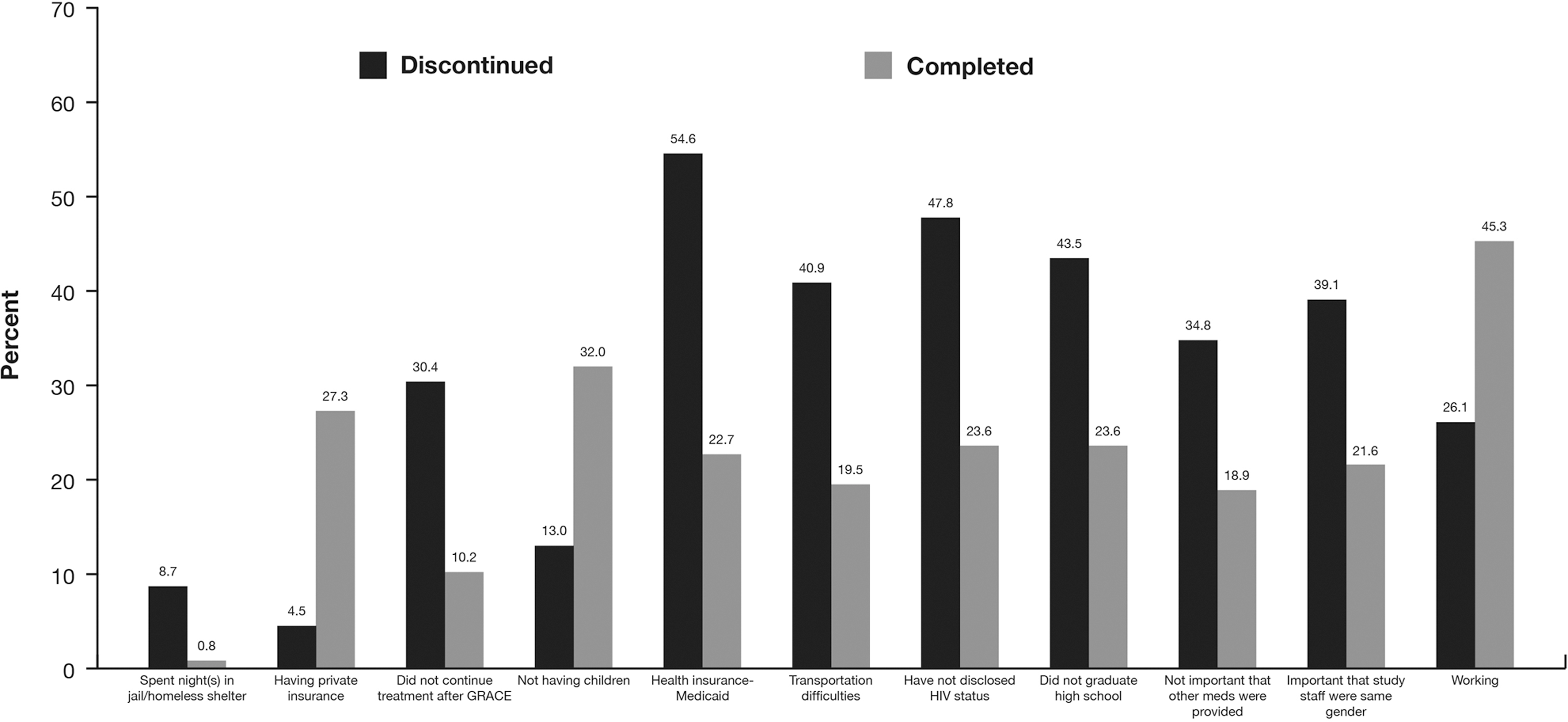
Associations with study discontinuation. Univariate analysis was used to examine associations (p<0.1) between survey responses and discontinuation from study for reasons other than virologic failure or adverse events. The p value for univariate selection is based on all levels (survey answers) of the variable (survey question); only one level is shown here.
Virologic response to study medications was defined as ITT-TLOVR <50 HIV-1 RNA copies/mL at week 48, and the following factors were significant at a level of p<0.1 (Fig. 3): paying for HIV medications with private insurance, being educated about HIV by research coordinator/HIV magazines/brochures/other, working, having private insurance and having the need to access medications as a main reason for participation in GRACE. Factors associated with not achieving a virologic response were difficulty keeping appointments prior to the GRACE trial, not feeling it was important that medications other than darunavir were provided during the study, having Medicaid, problems with transportation to study site, paying for HIV medications with Medicaid or Medicare, feeling it was important that the study nurse was the same sex, having trouble taking HIV medications prior to the GRACE trial, traveling to study site via transportation other than personal car, or being a primary caregiver.
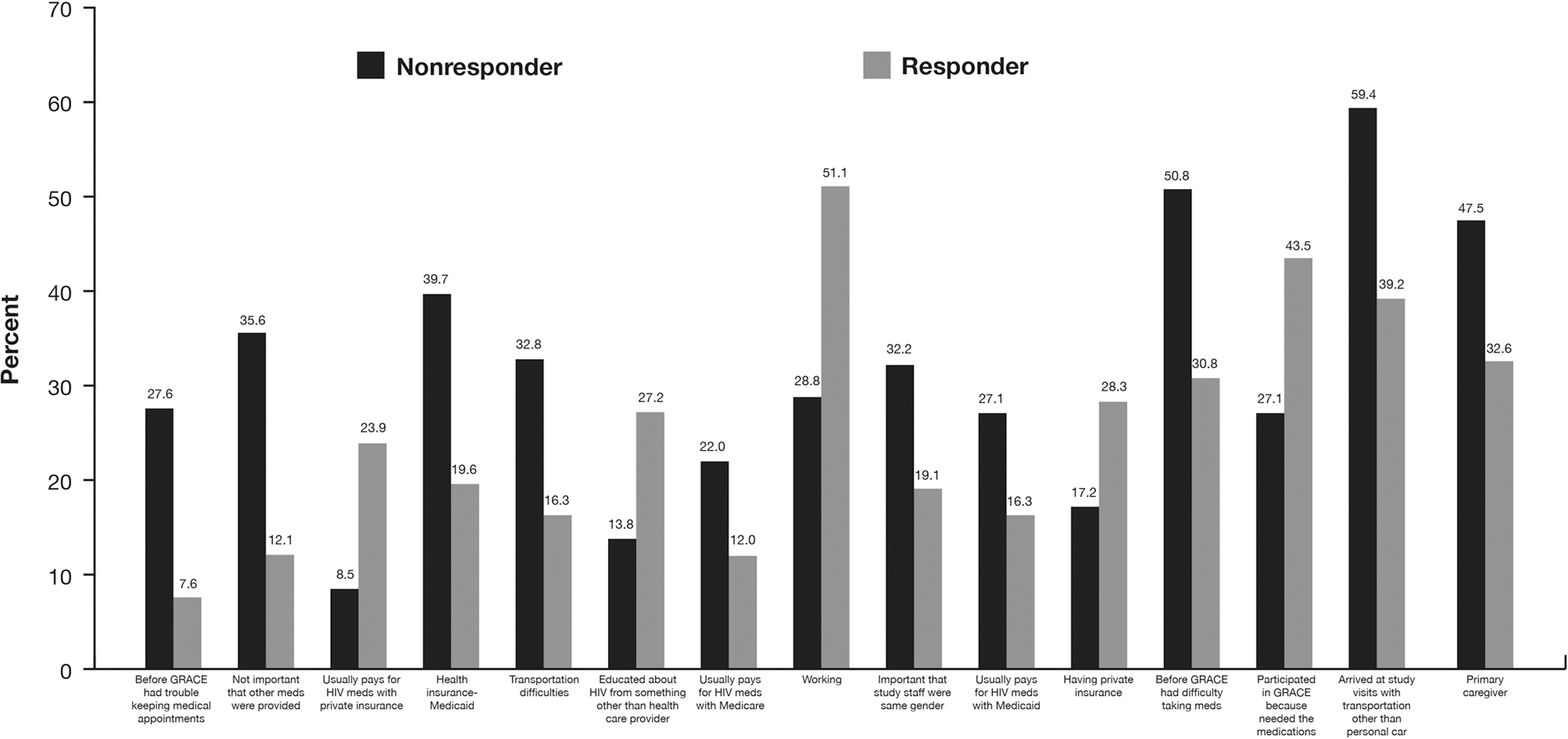
Associations with virologic response. Univariate analysis was used to examine associations (p<0.1) between survey responses and virologic response. Responders were defined as patients achieving intent-to-treat time-to-loss of virologic response <50 HIV-1 RNA copies/mL at week 48. The p value for univariate selection is based on all levels (survey answers) of the variable (survey question); only one level is shown here.
Discussion
GRACE was reported as a very positive experience for most survey participants. Although other reports have assessed patient experiences as they relate to HIV care in general, 16 to our knowledge, this is the first report of a post-trial exploratory survey to assess patient experiences as they relate to clinical outcomes following a large trial. Most respondents were recruited to participate in GRACE via their clinic, suggesting that media outreach programs may have less impact than originally thought or hoped. GRACE's enrollment success may be attributed to the access to treatment it provided for participants, as this was often cited as an important reason for participation. In addition, people who felt that availability of medications other than darunavir during GRACE was important were more likely to complete the study and had better virologic responses. The main factors linked to completion of the GRACE study were access to treatment and family and study site support. Although most survey respondents reported having no difficulty with arranging transportation to the GRACE site, those that did have difficulty and/or used a mode of transportation other than their own vehicle were more likely not to achieve a virologic response. Past experiences as they relate to difficulties adhering to HIV medications and keeping medical appointments were associated with lower adherence, study discontinuation, and lower virologic response rates while participating in GRACE. Similar respondent characteristics/responses were observed to be associated with adherence, study discontinuation, and virologic outcomes; this was not surprising given the fact that a lack of adherence to study medication, as well as study discontinuation are likely to affect virologic response. A prospective survey of antiretroviral-naïve and -experienced HIV-infected patients treated once daily with single-pill, fixed formulations of nucleoside/nucleotide reverse transcriptase inhibitors found that the reported adherence rate was more than 90%. Adjusting treatment regimens to more convenient formulations upon identification of patients with or likely to have poor adherence may ultimately improve patient outcomes. 17
There were some limitations of the survey. This was a post-hoc analysis, not prespecified in the protocol, and only 35% of the overall GRACE population (151 of 429) completed the survey. The survey was conducted 1.5 years after completion of the GRACE trial. This limitation may actually be beneficial because better insight may have been obtained after the excitement and emotion of the trial were over. A potential confounding factor may be HIV care providers' negative perceptions of female versus male patients, resulting in gender disparities in HIV care. 18 This may potentially lead to lower adherence rates in women compared with men. Although the survey respondents overall represented the participants in the GRACE trial, there were some small differences. Fewer respondents were black, more used recreational drugs, responders had less HIV stage C disease (which is indicative of progression to AIDS), and they had a higher median baseline CD4 cell count. Fewer respondents had discontinued GRACE and more had achieved higher response rates. As survey completion was not mandated for GRACE trial participants, those who had a lower discontinuation rate, higher response rate, and greater adherence were more likely to (and did) complete the survey.
Patients with certain characteristics or lifestyles may be at greater risk for experiencing poor study outcomes. Study respondents with or who are the primary caregiver of children and/or grandchildren, those who were not working, and those who experienced difficulties arranging transportation experienced less adherence, study completion, and virologic response.
Conclusions
Overall, baseline characteristics of survey respondents were representative of participants in the GRACE trial. Respondents cited access to treatment as the main reason for participation in the study, and completion of the study was attributable to support from the study site staff. Patients with certain characteristics may be at risk for poor study outcomes and may benefit from additional efforts with the aim of improving drug adherence, study retention, and, ultimately, clinical outcomes. Acknowledging these characteristics may be useful when designing future clinical trials for patients with HIV, especially women.
Footnotes
Acknowledgments
The authors thank the subjects and their families, the principal investigators, and the study sites for their participation in this survey. The authors acknowledge internal study support staff, as well as Jennifer Granit, PhD, and Francesca Balordi, PhD, Medicus International New York, for editorial assistance. Funding for the study analyses and for editorial support was provided by Janssen Therapeutics.
Author Disclosure Statement
RR, SS, YKD, and JM are employees and stockholders at Janssen. JC has received honorarium from Gilead for consulting and from Merck for a research grant for a clinical trial.
JF has received grant support from Janssen, Roche, BMS, GSK/ViiV, and Pfizer/ViiV, speaker support from Janssen, BMS, Merck, GSK/ViiV, and Pfizer/ViiV, and served as a consultant to Janssen, GSK/ViiV, and Merck. KS has received grant/research support from Biocryst, Gilead Sciences, GSK, Janssen, Merck, and Vertex, and has served on advisory boards (for scientific purposes) for Abbott, Gilead Sciences, Merck, Janssen, Tobira, and ViiV, and advisory boards (for marketing purposes) for Gilead Sciences and Janssen. KS has also served as a consultant to GSK, Merck, and Tobira and participated in data safety monitoring boards for Pfizer. DB has nothing to disclose.
