Abstract
CASTLE was a randomized 96-week study that demonstrated that atazanavir/ritonavir (ATV/r) was noninferior to lopinavir/ritonavir (LPV/r) in treatment-naïve HIV-infected patients. Analyses were carried out among patients who received ATV/r in the CASTLE study to better understand the clinical significance of unconjugated hyperbilirubinemia associated with administration of boosted ATV. Hyperbilirubinemia was defined as total bilirubin (conjugated and unconjugated) elevation greater than 2.5 times the upper limit of normal (grade 3–4). Patients in the ATV/r arm were assessed based on the presence or absence of hyperbilirubinemia through week 96. Analyses included number of confirmed virologic responders (CVR; HIV RNA <50 copies per milliliter), impact of hyperbilirubinemia on symptoms, elevations in liver enzymes, patient quality of life, and medication adherence. Through 96 weeks in the CASTLE study, 44% of patients who received ATV/r had hyperbilirubinemia at any time point, and between 12.5% and 21.6% had hyperbilirubinemia at any single study visit. At 96 weeks, 74% of patients overall and 84% and 69% of patients with and without hyperbilirubinemia, respectively, achieved CVR. Symptoms of jaundice or scleral icterus occurred in 5% of patients overall and in 11% with hyperbilirubinemia and 0% without hyperbilirubinemia. Four percent of patients with and 3% of patients without hyperbilirubinemia had grade 3–4 elevations in liver transaminases. Less than 1% of patients discontinued treatment due to hyperbilirubinemia. There were no differences in quality of life or adherence between patients with or without hyperbilirubinemia. In the CASTLE study, hyperbilirubinemia observed in the ATV/r group did not negatively impact clinical outcomes in HIV-infected patients.
Introduction
A
In previously reported studies involving ATV, the prevalence of bilirubin elevations greater than 2.5 and 5 times the upper limit of normal (ULN) range (grades 3 and 4) was 33–41% among patients receiving the unboosted ATV dose of 400 mg daily 8,9 and 40% to 49% in those on once daily 300 mg of ATV boosted with 100 mg of ritonavir (ATV/r). 10 Unless clinical signs of jaundice and scleral icterus, which may impact a patient's appearance and quality of life, 11 are observed, hyperbilirubinemia does not require management and is both reversible and independent of hepatocellular toxicity. Data from the BMS study AI424-089 indicated that bilirubin levels correlated with ATV plasma exposure and thus may indirectly inform physicians about drug exposure and adherence. 12 However, the trends of ATV-related hyperbilirubinemia over time and its clinical significance have not been well characterized in clinical studies.
This analysis aims to assess the presence or absence of hyperbilirubinemia among patients who received ATV/r in the CASTLE study and includes the proportion of patients achieving a complete virologic response, defined as HIV RNA less than 50 copies per milliliter. CASTLE was a randomized study that demonstrated that ATV/r was noninferior to lopinavir/ritonavir (LPV/r) in treatment-naïve HIV-infected patients through 96 weeks. 13,14 The objectives of the current analysis are to better understand the patterns of hyperbilirubinemia, defined as laboratory abnormalities of grade 3–4 total bilirubin adverse events associated with administration of ATV/r, and to investigate the clinical impact of hyperbilirubinemia on treatment outcomes, patient quality of life, and medication adherence.
Methods
CASTLE was a randomized, open-label, multicenter 96-week study to assess the efficacy and safety of ATV/r (300/100 mg once daily) compared with lopinavir/r (LPV/r; 400/100 mg twice daily) administered with fixed-dose tenofovir/emtricitabine (300/200 mg once daily) in treatment-naïve patients. Full methodology and primary end points have been reported. 13,14
This study was performed in accordance with Good Clinical Practice and the ethical principles of the Declaration of Helsinki. The protocol was approved by the institutional review board at each study site, and patients provided written informed consent before participation in the study. The trial is registered with ClinicalTrials.gov, number NCT00272779.
Hyperbilirubinemia was defined as total bilirubin (conjugated and unconjugated) elevation greater than 2.5×ULN (grade 3) and greater than 5×ULN (grade 4). The numbers of patients taking ATV/r who had hyperbilirubinemia were assessed at each study visit. Hyperbilirubinemia (grades 3 and 4) and total bilirubin through week 96 were reported in patients receiving ATV/r (as-treated patients).
For this analysis, patients in the ATV/r arm were assessed based on the presence or absence of hyperbilirubinemia through week 96. Analyses included proportion of patients achieving a confirmed virologic response (CVR, modified intent-to-treat [ITT], noncompleter=failure [NC=F]), defined as HIV RNA less than 50 copies per milliliter. The impact of hyperbilirubinemia on symptoms of jaundice or scleral icterus, elevations in liver enzymes (aspartate transaminase [AST] and alanine transaminase [ALT] greater than 5×ULN), patient quality of life, medication adherence, and reasons for nonadherence was also assessed. Quality of life was assessed at baseline and weeks 12, 24, 48, and 96 using the Medical Outcomes Study HIV Health Survey (MOS-HIV), a 35-item questionnaire to assess physical and mental well-being. 15 The summary scores of physical and mental health range from 0 to 100, with higher scores indicating better health. Adherence was measured and reasons for nonadherence were assessed at every visit from week 4 through week 96 using the Multicenter AIDS Cohort Study (MACS) adherence questionnaire. 16,17 Adherence was determined by comparing study medications received with those reported on the MACS questionnaire at the same visit. Adherent was defined as taking all doses and number of pills as prescribed for each medication.
Statistical analysis
Statistical methods for the 96-week analysis have been published 14 and are summarized briefly below. Efficacy results are presented by the as-randomized treatment regimen (ITT). Safety results are presented by the as-treated regimen (i.e., by the treatment regimen actually received). The hyperbilirubinemia analysis was not powered to detect statistical differences, and no statistical comparisons were carried out for this report. The proportions of patients with hyperbilirubinemia (grade 3–4), the median total bilirubin, and the proportions of patients who were responders (CVR NC=F, HIV RNA <50 copies per milliliter) through week 96 in patients receiving ATV/r (as-treated patients) were demonstrated with plots. Scatter plots of total bilirubin versus AST and ALT were produced for patients with hyperbilirubinemia at weeks 24, 48, and 96. The impact of hyperbilirubinemia on symptoms (jaundice or scleral icterus), grade 3–4 liver transaminase elevations, quality of life (MOS-HIV physical and mental summary scores), and adherence (MACS adherence questionnaire) were summarized. Adherence was assessed in the NC=missing (NC=M) analysis.
Results
A total of 441 treatment-naïve HIV-infected patients were randomized to and received at least one dose of ATV/r (as-treated patients). Full details of patient disposition and primary and secondary end points have been reported previously. 13,14
Patients with hyperbilirubinemia in the CASTLE study
Overall, 44% of patients receiving ATV/r had hyperbilirubinemia (grade 3–4) at any time point during the 96-week study, compared with 12.5–21.6% of patients with hyperbilirubinemia at any single study visit (Fig. 1). At baseline, median total bilirubin was 0.4 mg/dL (6.8 μmol/L) among patients receiving ATV/r. During weeks 4 through 96, median total bilirubin ranged between 1.3 mg/dL (22.2 μmol/L) and 1.8 mg/dL (30.8 μmol/L; Fig. 2).
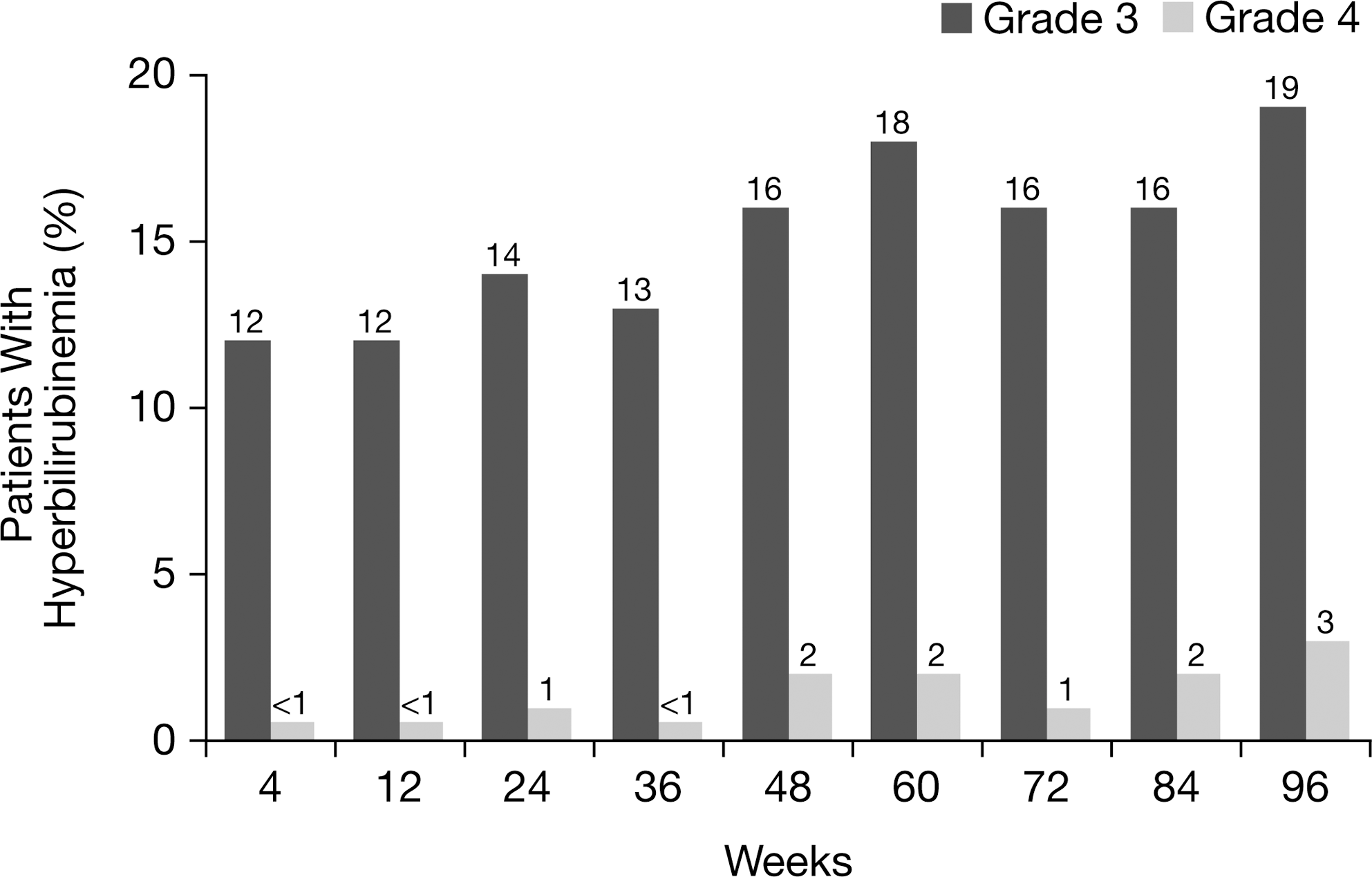
Hyperbilirubinemia through 96 weeks in patients receiving atazanavir/ritonavir (ATV/r; as-treated patients). Note: hyperbilirubinemia was defined as total bilirubin (conjugated and unconjugated) elevation >2.5×upper limit of normal (ULN) (grade 3) and >5×ULN (grade 4).
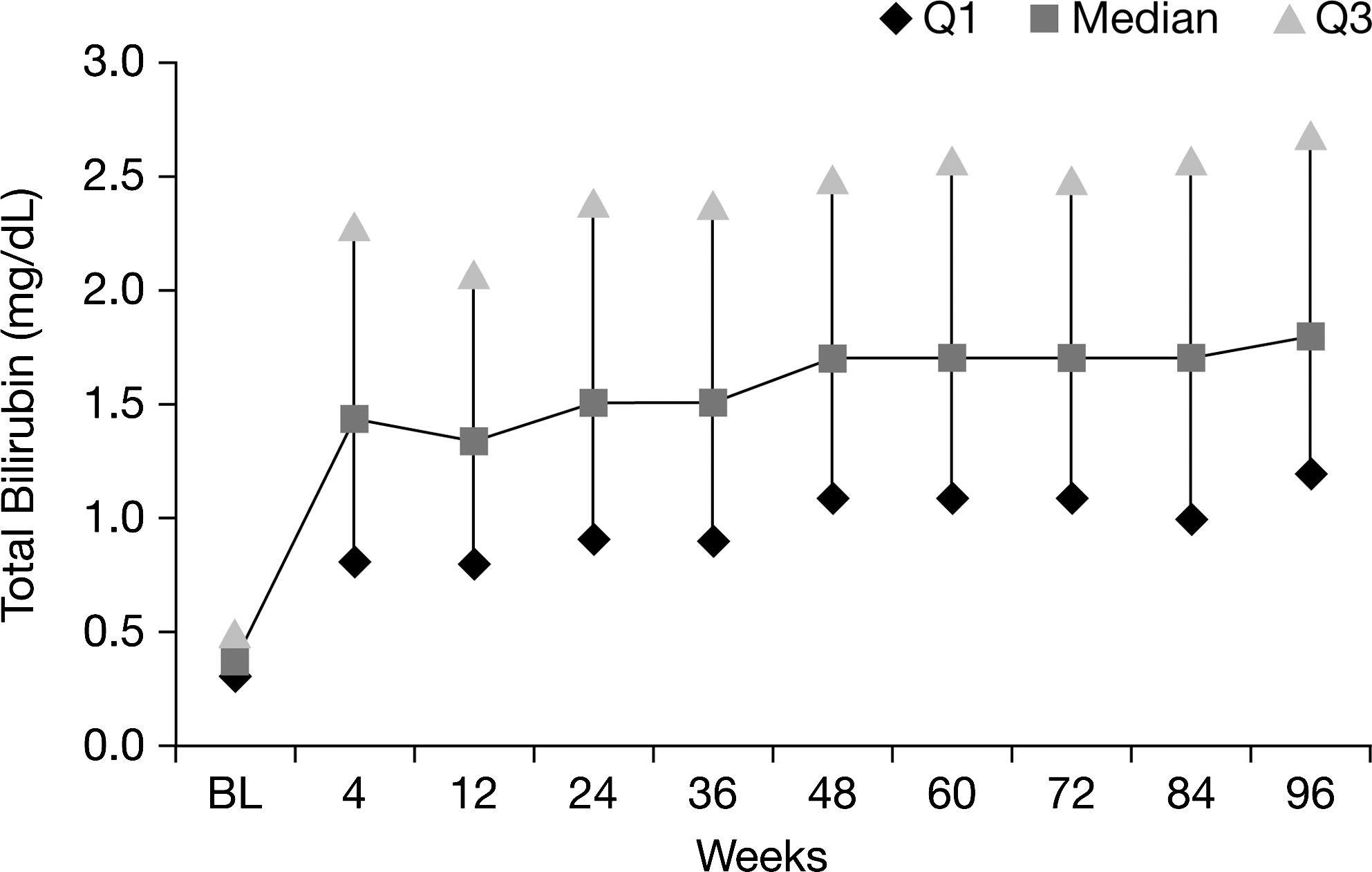
Median total bilirubin through 96 weeks in patients receiving atazanavir/ritonavir (ATV/r; as-treated patients).
ATV/r efficacy among patients with and without hyperbilirubinemia
The percentages of patients in the ATV/r treatment group with and without hyperbilirubinemia achieving CVR (HIV RNA <50 copies per milliliter) at any point during the study are illustrated in Fig. 3 and Table 1. At 96 weeks, 74% (327/440) of patients overall and 84% (162/192) and 69% (167/243) of patients with and without hyperbilirubinemia had achieved CVR. Virologic failures occurred in 9% of the patients with and in 9% without hyperbilirubinemia (Table 1).
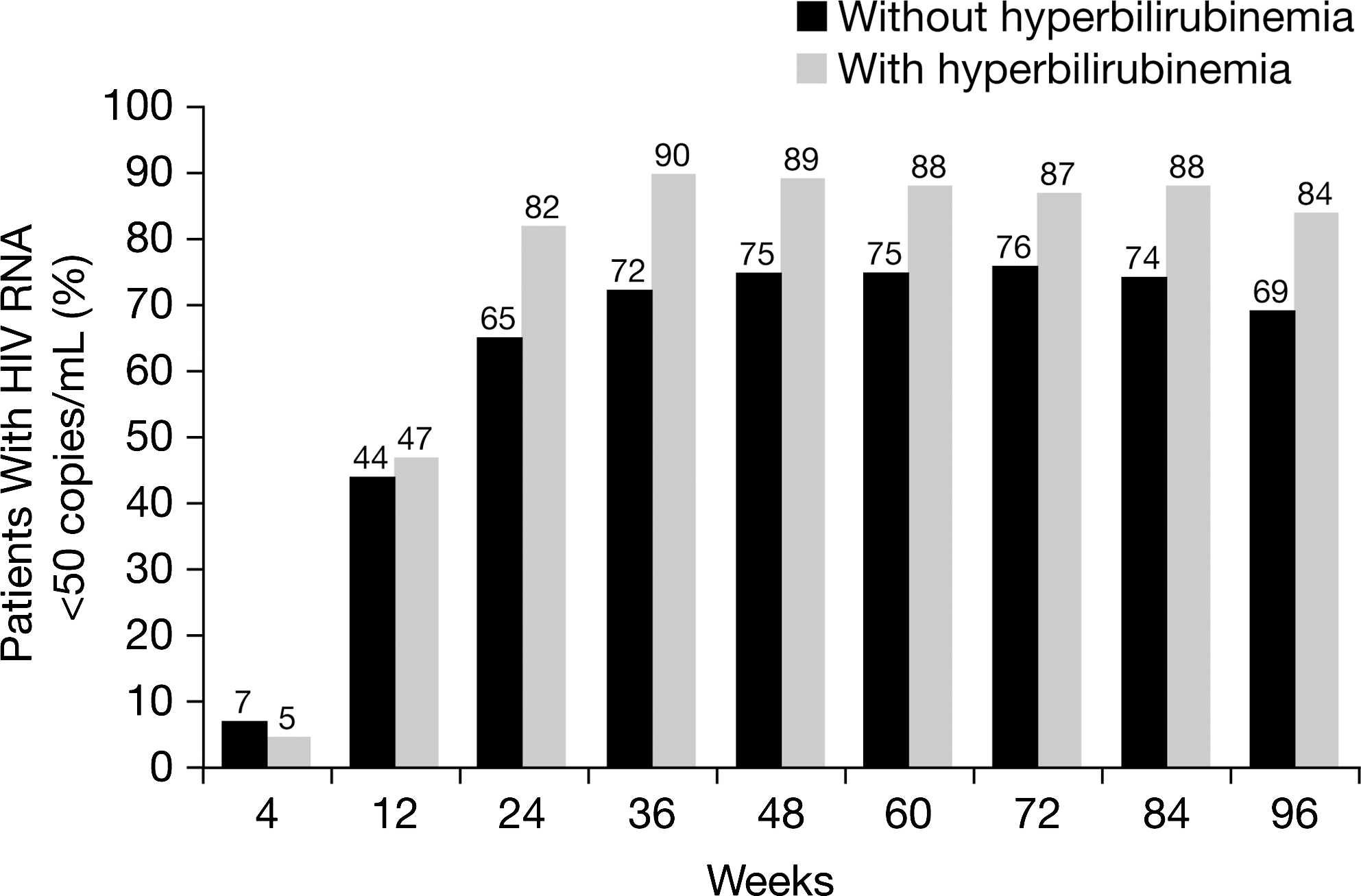
Percentage of patients with (n=192) and without (n=243) hyperbilirubinemia and HIV RNA <50 copies per milliliter receiving atazanavir/ritonavir (ATV/r) through 96 weeks (CVR, NC=F) (as-treated patients). Note: hyperbilirubinemia was defined as total bilirubin (conjugated and unconjugated) elevation >2.5×upper limit of normal (grade 3–4).
AE, adverse event.
Impact of hyperbilirubinemia on bilirubin-associated adverse events and laboratory values
Through 96 weeks in the CASTLE study, 5% (21/441) of patients overall and 11% (21/192) of patients with hyperbilirubinemia (compared with 0% patients without hyperbilirubinemia) had grade 2–4 treatment-related adverse events of jaundice or scleral icterus at any time during the study (Table 2). Four percent (8/192) of patients with and 3% (8/243) of patients without hyperbilirubinemia had grade 3–4 elevations in liver transaminases. Only four patients had concurrent (within 1 week) hyperbilirubinemia and grade 3-4 ALT or AST abnormalities when receiving ATV/r during the study. Figure 4, a scatter plot of ALT, AST, and bilirubin levels, suggests that there is no relationship between hyperbilirubinemia and elevated liver transaminases at any time point during the study. Less than 1% (3/441) of patients discontinued treatment as a result of hyperbilirubinemia, jaundice, or scleral icterus before week 48, and no patients discontinued after week 48. Discontinuations due to adverse events occurred in 3% of the patients with and without hyperbilirubinemia (Table 1).
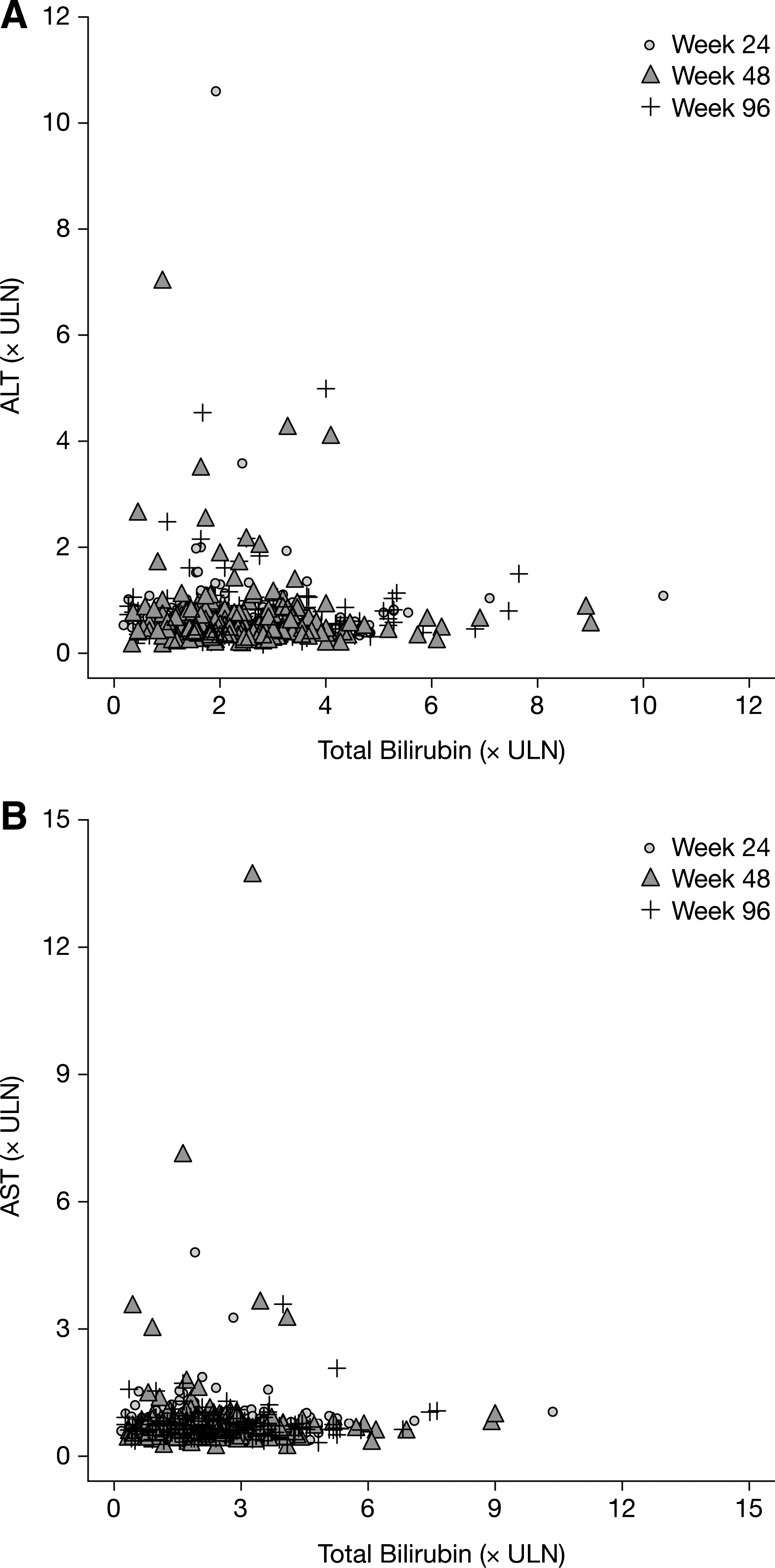
Total bilirubin versus (
Hyperbilirubinemia reported as an adverse event (not a laboratory elevation), including the terms “blood bilirubin abnormal, blood bilirubin increased, blood bilirubin unconjugated, and blood bilirubin unconjugated increased.”
ALT, alanine transaminase; AST, aspartate transaminase.
Impact of hyperbilirubinemia on quality of life and adherence
There was no difference in change of quality of life among patients with and without hyperbilirubinemia throughout the study. Results at 96 weeks of the MOS-HIV survey are presented in Table 3. In the MACS adherence questionnaire, adherence to the regimen, 84% (147/176) versus 83% (154/186), and to ATV, 87% (153/176) versus 85% (159/186), was also similar among patients with and without hyperbilirubinemia (NC=M) at week 96. Reasons for nonadherence to the regimen reported at weeks 4 and 96 included: didn't want others to notice, fell asleep, felt depressed, felt sick or ill, were away from home or busy with other things, too many pills to take, ran out of pills, or simply forgot. There was no common pattern of reasons for nonadherence reported by patients with hyperbilirubinemia. None of the patients with hyperbilirubinemia and two patients without hyperbilirubinemia selected “feeling like the drug was too toxic” as a reason for nonadherence during the study.
ATV/r, atazanavir/ritonavir; MOS-HIV, Medical Outcomes Study HIV Health Survey.
Discussion
Although elevated levels of bilirubin are associated with the use of some PIs, including atazanavir, the effect of these elevations on clinical outcomes in HIV-infected patients is not well characterized in clinical studies. 11 In the CASTLE study, 14 hyperbilirubinemia occurred in 44% of patients receiving ATV/r at any time through 96 weeks, and this prevalence rate is similar to the rates reported in other studies. 8 –10,18 The mechanism by which some patients experience hyperbilirubinemia and others do not is not fully understood. Genetic polymorphisms of the UGT1A1 gene impact UGT activity and have been previously reported to contribute to hyperbilirubinemia. Haplotype variants of UGT1A1, UGT1A3, and UGT1A7 genes have previously been associated with occurrence of unconjugated hyperbilirubinemia among patients receiving ATV/r. 19 –23
Our study showed similar low rates of hepatotoxicity in patients with and without hyperbilirubinemia. Hepatotoxicity, chronic liver disease, and cirrhosis are common with HIV/HCV co-infected individuals, and HIV infection may accelerate liver damage caused by hepatitis C virus. 24 In addition to liver toxicity, people with HIV infection have been suggested to be at greater risk of cardiovascular disease (CVD) than the general population. 25,26 Several cohort studies have identified an inverse relationship between bilirubin and CVD risk. 7,27 –33
Overall, patients receiving ATV/r responded well to treatment, with 74% achieving a CVR 14 ; a greater percentage of patients with hyperbilirubinemia than without achieved a CVR. Previous studies have reported that bilirubin levels correlate with ATV plasma concentrations. 23,34 –36 It is possible that a higher number of patients with hyperbilirubinemia than without achieved a CVR due to higher ATV plasma concentrations, but supporting pharmacokinetic sampling and data were not available in this population.
Quality of life and adherence were similar in patients with and without hyperbilirubinemia, and were similar to values reported in previous studies. 37,38 Hyperbilirubinemia was not associated with any increase in abnormalities in liver transaminases compared with absence of hyperbilirubinemia, which supports the results of previous reports that ATV-associated hyperbilirubinemia does not impact hepatic function. 3,9 –11,39 Most antiretroviral regimens are safe in HIV/HCV-coinfected individuals, including those patients with cirrhosis. 35,40 The prevalence of symptoms of jaundice or scleral icterus among patients receiving ATV/r was low overall in the CASTLE study (5%) and in patients with hyperbilirubinemia (11%). Furthermore, few patients discontinued treatment as a result of hyperbilirubinemia, jaundice, or icterus, and none of the discontinuations were between weeks 48 and 96. 14 Similarly low rates of discontinuations because of hyperbilirubinemia were also reported in other ATV studies. 9,10
The limitations of this study are that it is a post hoc analysis of a subset of patients and is therefore not powered to make robust conclusions. Additionally, CVD was not assessed. However, the study provides a valuable opportunity to assess the general trends of hyperbilirubinemia and its clinical impact in patients treated with ATV/r in the CASTLE study.
Although hyperbilirubinemia was common among patients receiving ATV/r at any time through 96 weeks in the CASTLE study, it was less frequent at specific time points and was not associated with related symptoms in most patients. The majority of patients responded well to treatment overall, with numerically more patients suppressing HIV RNA (<50 copies per milliliter) with hyperbilirubinemia than without. The presence of hyperbilirubinemia did not affect AST/ALT elevations, quality of life, or adherence. These data suggest that hyperbilirubinemia observed with ATV/r does not negatively impact clinical outcomes in HIV-infected patients.
Footnotes
Acknowledgments
Editorial support was provided by Cheryl Jenkins and Jean Turner of PAREXEL and Emily Cullinan of Bristol-Myers Squibb and was funded by Bristol-Myers Squibb.
Author Disclosure Statement
C.M. has served on speakers' bureaus for Bristol-Myers Squibb, Gilead, and ViiV Healthcare, and has received research funding from Bristol-Myers Squibb, Gilead, GlaxoSmithKline/ViiV Healthcare, Boehringer-Ingelheim, Merck, and Pfizer. J.U., W.H., V.W., S.J., D.B., D.M., and A.F. are employees and stockholders of Bristol-Myers Squibb. G.M. has received research funding from Abbott, Ardea Biosciences, Bionor, Bristol-Myers Squibb, Gilead Sciences, GlaxoSmithKline, Merck, Pfizer, Theratechnologies, and Tibotec. He has from received honoraria as a speaker and/or advisor from Boehringer-Ingelheim, Bristol-Myers Squibb, Gilead Sciences, GlaxoSmithKline, Merck, Pfizer, Theratechnologies, Tibotec, and ViiV Healthcare.
