Abstract
Patients infected with HIV treated with highly active antiretroviral therapy (HAART) frequently develop body physical changes (BPC) that have an important psychosocial burden. The purpose of this study was to determine the prevalence of BPC observed by HIV-infected patients and their attending physicians and to assess the impact BPC had on daily life. In this epidemiologic multicenter study, patients with HIV infection and their treating physicians filled out parallel questionnaires about their perceptions of specific BPC and their impact on daily activities. A total of 965 patient–physician questionnaires were collected across 98 health centers. Patient's mean age was 43.7 ± 8.5 years and 72.6% were men. Adjusted prevalence of perceived BPC by patients and physicians was 55.1% (95% confidence interval [CI]: 52.0–58.1) and 55.2% (95% CI: 52.1–58.2), respectively (p = 1.000). Overall patient-physician agreement concerning perception of BPC was 83% (p < 0.0005). The most common BPC was lipoatrophy, described by 46.8% (95% CI: 43.7–49.8) of patients and 49.4% (95% CI: 46.3–52.5) of physicians (p = 0.033) followed by lipohypertrophy. No gender differences were observed in the global prevalence of BPC (p = 0.649). However, significantly more women reported lipoatrophy of the lower limbs (p = 0.009) and buttocks (p = 0.007), as well as lipohypertrophy (p = 0.007), than men; 58.2% (95% CI: 54.0–62.4) patients noted that BPC negatively affected their daily activities. This study reflects the high prevalence of patient and physician-perceived BPC in the HIV population, and the adverse impact on daily life. Physicians should be aware of the psychosocial consequences of BPC in HIV patients in order to improve patient well-being.
Introduction
B
The situation changed with the introduction of HAART, which markedly reduced opportunistic infections and HIV-related morbidity and mortality, as well as the outward signs associated with the development of AIDS. 8,9 On the other hand, the transformation of this fatal disease into a chronic illness led to the emergence of metabolic complications secondary to long-term HAART use. In effect, a clinical syndrome consisting of peripheral lipodystrophy, hyperlipidemia, diabetes, and insulin resistance was described in 1998 in HIV patients receiving at least one protease inhibitor (PI). 10 Since then, different risk factors, including HIV infection itself, have been identified as risk factors for the development of such syndrome. 11 –13 Noteworthy, the number of studies assessing HAART-associated metabolic disorders has increased steadily, leading to the identification of additional alterations affecting physical appearance. 14 –16 Frequent physical changes related with HAART include lipodystrophy, a wide range of dermatologic changes, and jaundice. 14
Lipodystrophy is a disorder characterized by an abnormal distribution of subcutaneous fat, and includes peripheral fat loss (lipoatrophy) in the arms, legs, face and buttocks, and lipohypertrophy (fat accumulation) in the trunk, chest, breasts, neck, or other areas of the body. 17,18 In addition, lipodystrophy has been associated with other metabolic features, including disturbances in lipid and glucose metabolism, mitochondrial toxicity, and elevated hepatic transaminases. 17,19 Although the prevalence of lipodystrophy varies widely, from around 7% to 84% 20 depending on the diagnostic criteria used, the overall prevalence of at least one physical change is approximately 50%. 21,22 The etiology of lipodystrophy is multifactorial, involving many overlapping risk factors related to the duration of HIV disease, degree of viral suppression, CD4 count at HAART initiation, gender, age, and duration of exposure to HAART. 17,23 –28 Visible changes in fat distribution appear early after the start of therapy, within 3 months to 1 year according to different studies, and can be progressive. 18,25,26 Some nucleoside reverse transcriptase inhibitors (NRTIs), particularly thymidine analogues, have been strongly associated with lipodystrophy, especially lipoatrophy, but currently, most of the available classes of antiretroviral drugs have been found to be involved with the abnormal fat distribution in HIV patients. 29 –33
Dermatologic disorders constitute another group of appearance-related side effects of HAART. Although their causes may be difficult to determine because HIV patients often take antiretrovirals in conjunction with other drugs, certain antiretroviral agents are associated with specific cutaneous manifestations. Skin rashes have been reported in association with abacavir, non-NRTIs, and some PIs. Skin and nail hyperpigmentation has been observed with emtricitabine and zidovudine use, and jaundice and conjunctival pigmentation secondary to hyperbilirubinemia are side effects observed with atazanavir and indinavir use. 14,34,35
Most of these antiretroviral-induced physical changes usually occur soon after the start of therapy and, with the exception of lipodystrophy, are usually reversible or treatable. Fat changes are not easily treatable, and might even cause chronic disfigurement and pain. 36 Current strategies for the treatment of lipodystrophy involve regimen switching, for example NRTI sparing, and reconstructive-cosmetic techniques, such as facial lipofilling, which has shown promising results in the long-term treatment of this condition. 14,37
Although much of current research has focused on those alterations that increase the risk of mortality, the development of appearance-related changes can be stigmatizing and socially distressful, producing a significant impact on quality of life (QOL), 38,39 which can lead to decreased adherence to therapy, suboptimal drug levels, and development of drug resistance. 40,41 Moreover, a study demonstrated that HIV-infected patients were willing to trade length of life for the avoidance of appearance-related adverse drug effects. 42
Given the importance of the timely recognition of body physical changes (BPC) caused by HAART, we sought to determine the prevalence of BPC as perceived by patients with HIV infection, and compared it to the prevalence of BPC as perceived by their treating physicians. Other objectives of this study were to compare the perception of the types of BPC perceived between patients and physicians, and the impact that BPC had on daily life according to gender.
Patients and Methods
Study design
This epidemiologic study consisted of a stratified multistage probabilistic survey to evaluate the prevalence of perceived BPC by HIV patients and their treating physicians. Patients were eligible to be included if they had documented HIV infection and were willing to participate in the study. The study protocol was approved by the Ethics Committee of Hospital Clínico San Carlos (Madrid), and an informed consent was obtained from all patients before they were included in the study.
Study procedures
Data were gathered throughout several health centers in Spain. The survey consisted of two parallel questionnaires designed specifically for this study, which were to be completed simultaneously, but independently, by the physician for a specific patient and by that particular patient during a single visit. Both questionnaires included an initial section where baseline patient characteristics were recorded, including age, gender, route of HIV infection, duration of HAART, route of infection and occupational status. Subsequently, patients and physicians were asked if they had noted the presence of BPC since the start of HAART; if the answer was “no,” the questionnaire ended. If, on the other hand, the physician and/or the patient answered “yes,” they proceeded to provide details about these changes, which included the perception of changes in skin and conjunctival pigmentation, and whether or not they had perceived fat loss in the face, upper and lower limbs and buttocks, or fat accumulation in the neck, lower back, chest, and trunk. In addition, patients and physicians were asked to provide a subjective rating of the severity of the fat loss or accumulation (mild, moderate or severe). Finally, physicians were asked for how long had they noticed the presence of BPC in their patients (less than 6 months ago, 6 to 12 months ago, or more than 12 months ago), and patients were asked if the presence of BPC had influenced in any way the following aspects of their daily life: mood, daily activities, personal relationships, and self-esteem.
Physical changes were defined as the perception of an increase (lipohypertrophy) or reduction (lipoatrophy) in bodily fat of whatever severity in at least one of the abovementioned body parts.
Statistical analysis
The primary objective of this study was to estimate the prevalence of BPC perceived by HIV-positive patients receiving HAART and their attending physicians, which had been previously estimated as greater than or equal to 25%, with a relative error less than or equal to 10%. Considering a finite population of 60,000 HIV-seropositive patients receiving HAART, and a design effect of 15%, a sample size of 813 evaluable patients was calculated. In addition, taking into account a phenomenon of nonresponse, and lack of evaluability due to any cause, the sample size was increased in 20% to 1016 patients and rounded to 1020. Secondary objectives were to compare the perception of BPC between patients and physicians, to compare the prevalence of BPC in men and women, and to determine whether subjective perceptions of its importance were gender-specific.
In order to perform a stratified, multistage study, 17 regions were grouped into 12 strata, and the number of patients and centers involved in each stratum was calculated according to the overall HIV population in the area, with an overall sampling fraction of 0.017 (0.014–0.026). In the first stage, 102 health centers or hospitals were randomly selected with a probability proportional to the number of HIV patients followed up during the last year. In the second stage, attending physicians were selected by simple random sampling from each participating center. Finally, in the third stage, 10 patients from those scheduled by the physician during the study period (January 13 until March 18, 2008) were selected by systematic sampling to fill out the questionnaire. There was no reposition of patients who refused to participate in the study.
Qualitative variables are shown as absolute and relative frequencies, and adjusted percentages with 95% confidence interval (CI) considering the entire study population overall or per gender. Impact on daily life was only assessed in patients (overall and per gender) who reported having perceived BPC. Continuous variables were described using mean and standard deviation. Degree of patient–physician agreement was measured using Cohen's κ. Comparison of percentages between independent groups was performed with the χ2 test, and within the same group with the McNemar test or generalized marginal homogeneity test. To study the association between BPC and duration of HAART treatment, quartiles of time under HAART were calculated and classified as follows: less than three years, from three to seven years, from seven to ten years, and more than ten years. Statistical analyses were carried out using SPSS v15.0 software (SPSS Inc., Chicago, IL). Statistical significance was set at p < 0.05.
Results
Description of participants
Between January and February 2008, a total of 965 patient–physician questionnaires were collected across 98 health centers. Mean age of the patients was 43.7 ± 8.5 years. Seven hundred one patients surveyed (72.6%) were men and 255 (26.4%) women; gender was not reported in nine subjects. Nine hundred eight patients (94.1%) acquired HIV infection through a single route, either through intravenous drug use (39.0%), homosexual (26.9%) or heterosexual contact (25.9%). Mean time of HAART was 88.3 ± 55.5 months. Of the patients included in the study, 74.3% had received HAART for a period longer than three years, and 26.3% had received HAART for more than 10 years. Regarding occupational status, 627 (65.0%) were currently employed, 85 pensioners or retired (8.8%), 57 unemployed (5.9%), and 31 students (3.2%). Details of baseline patient characteristics are shown in Table 1.
HAART, highly active antiretroviral therapy; IDU, intravenous drug use; SD, standard deviation.
Perception and severity of BPC by patients and physicians
There was a concordance of 88.3% in the perception of BPC between physicians and their corresponding patients (κ = 0.682; p < 0.0005); in other words, most physicians and patients agreed that they had developed BPC. On the contrary, 7.7% of the patients considered they had not developed BPC, while their treating physicians thought they had, and 7.8% of the patients perceived they had developed BPC, while their treating physicians thought they had not. More than half of the patients (55.1%; 95% CI: 52.0–58.1) perceived they had suffered BPC since they started receiving HAART; similarly, more than half of the physicians (55.2%; 95% CI: 52.1–58.2) perceived their patients had developed BPC. No significant differences were detected between these percentages (p = 1.000). The most common BPC identified by patients and physicians was lipoatrophy, although it was mentioned by significantly more physicians (49.4%; 95% CI: 46.3–52.5%) than patients (46.8%; 95% CI: 43.7–49.8%; p = 0.033). The face was the body part most affected by lipoatrophy, followed closely by the lower limbs, buttocks and upper limbs. No significant differences were observed between patient and physician perceptions regarding the body part affected by lipoatrophy (Table 2). Lipohypertrophy was the next BPC most frequently noted by patients and also by physicians. The percentage of patients mentioning lipohypertrophy differed significantly from the percentage reported by physicians (28.7% [95% CI: 25.9–31.4] versus 25.7% [95% CI: 23.0–28.4]; p = 0.032). The trunk was the body part most commonly affected by lipohypertrophy, perceived by 24.9% (95% CI: 21.3–28.5) of the patients and 22.8% (95% CI: 19.3–26.2) of the physicians, followed by the chest, neck and lower back. There were no significant differences between frequencies observed by patients and physicians (Table 2). Skin pigmentation changes were perceived significantly less often by physicians than by patients (2.4% [95% CI: 1.4–3.3] versus 5.9% [95% CI: 4.5–7.4]; p < 0.0005). No significant differences were observed regarding conjunctival pigmentation, which was noted by 3.9% (95% CI: 2.7–5.1) of patients and 3.1% (95% CI: 2.1–4.2) of physicians (Table 2).
Statistically significant (p < 0.05).
BPC, body physical changes; CI, confidence interval.
To the question “For how long have you noticed the presence of BPC on your patient,” 79.2% (95% CI: 76.0–82.4) of the physicians answered they had noticed these changes for more than 1 year.
Regarding HAART duration, there was a significant association between the perception of BPC and the duration of HAART exposure (p < 0.0005). Overall, 19.8% (95% CI: 16.4–23.2) of patients on HAART for more than 10 years perceived BPC compared to 4.9% (95% CI: 3.6–6.3) of patients who had been on HAART for less than 3 years. McNemar's test did not evidence significant differences between patients' and physicians' perception in any of the time of HAART exposure groups (<3 years, p = 0.180; 3–7 years, p = 0.072; 7–10 years, p = 0.118; and >10 years, p = 0.417; Fig. 1).
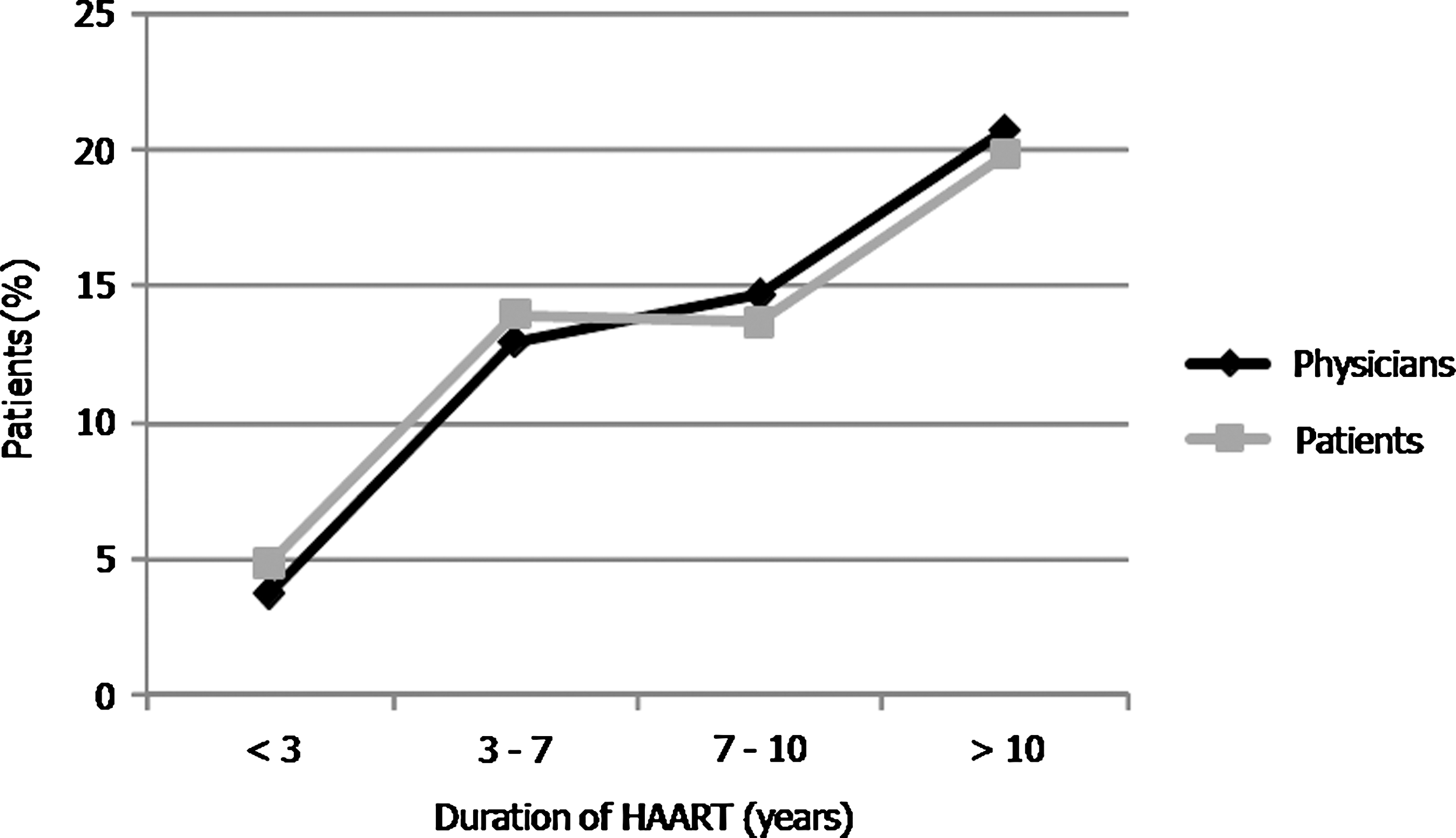
Percentage of patients referring body physical changes (BPC) in relation to years of highly active antiretroviral therapy (HAART) duration (n = 965).
In relation to the severity of lipodystrophy-related BPC, most patients and physicians considered the changes to be mild or moderate, regardless of whether the BPC was lipoatrophy or lipohypertrophy, or of the body region affected (Table 3). The marginal homogeneity test only showed significant differences between the severity of lipohypertrophy affecting the trunk (p = 0.003) and the chest (p = 0.047), reported by patients and physicians, reflecting the similarity between patient's and physician's assessment of the perceived severity of BPC.
Percentage of severity grade declared by patients and/or physicians reporting each event.
Statistically significant (p < 0.05).
GHMT, Generalized marginal homogeneity test.
Perception of BPC between men and women
No significant differences were observed in the overall prevalence of BPC perceived by men (54.7%; 95% CI: 51.1–58.3) and women (56.9%; 95% CI: 50.8–63.0; p = 0.649). However, regarding specific BPC, significantly more women reported lipoatrophy of the lower limbs (42.7% versus 33.7%; p = 0.009) and buttocks (41.5% versus 32.3%; p = 0.007), as well as overall lipohypertrophy (34.7% versus 26.5%; p = 0.007), than men. No significant differences were observed between changes perceived by men and women regarding skin pigmentation changes and conjunctival pigmentation (Table 4).
Gender was not reported in 9 subjects.
Statistically significant (p < 0.05).
BPC, body physical changes; CI, confidence interval.
Impact of BPC on daily life
Three-hundred twenty-four patients (58.2% of those reporting BPC; 95% CI: 54.0–62.4) noted that the development of BPC negatively affected their daily life. When divided according to gender, 63.4% (95% CI: 56.3–70.5) of women and 56.1% (95% CI: 51.2–61.1) of men mentioned that BPC had an impact on their daily life overall, without significant differences between genders (p = 0.209). Additionally, as for the effect of BPC on specific aspects of daily life, significantly more women described mood changes (53.0%; 95% CI: 45.6–60.4) compared to men (31.5%; 95% CI: 26.8–45.6; p < 0.0005). Also, significantly more women (32.2%; 95% CI: 24.9–39.5) noted that BPC affected their self esteem than men (23.2%; 95% CI: 19.1–27.4; p = 0.030), whereas the effects of BPC on daily activities and personal relationships noticed by men and women were not significantly different (p = 0.2244 and p = 0.355, respectively; Fig. 2).
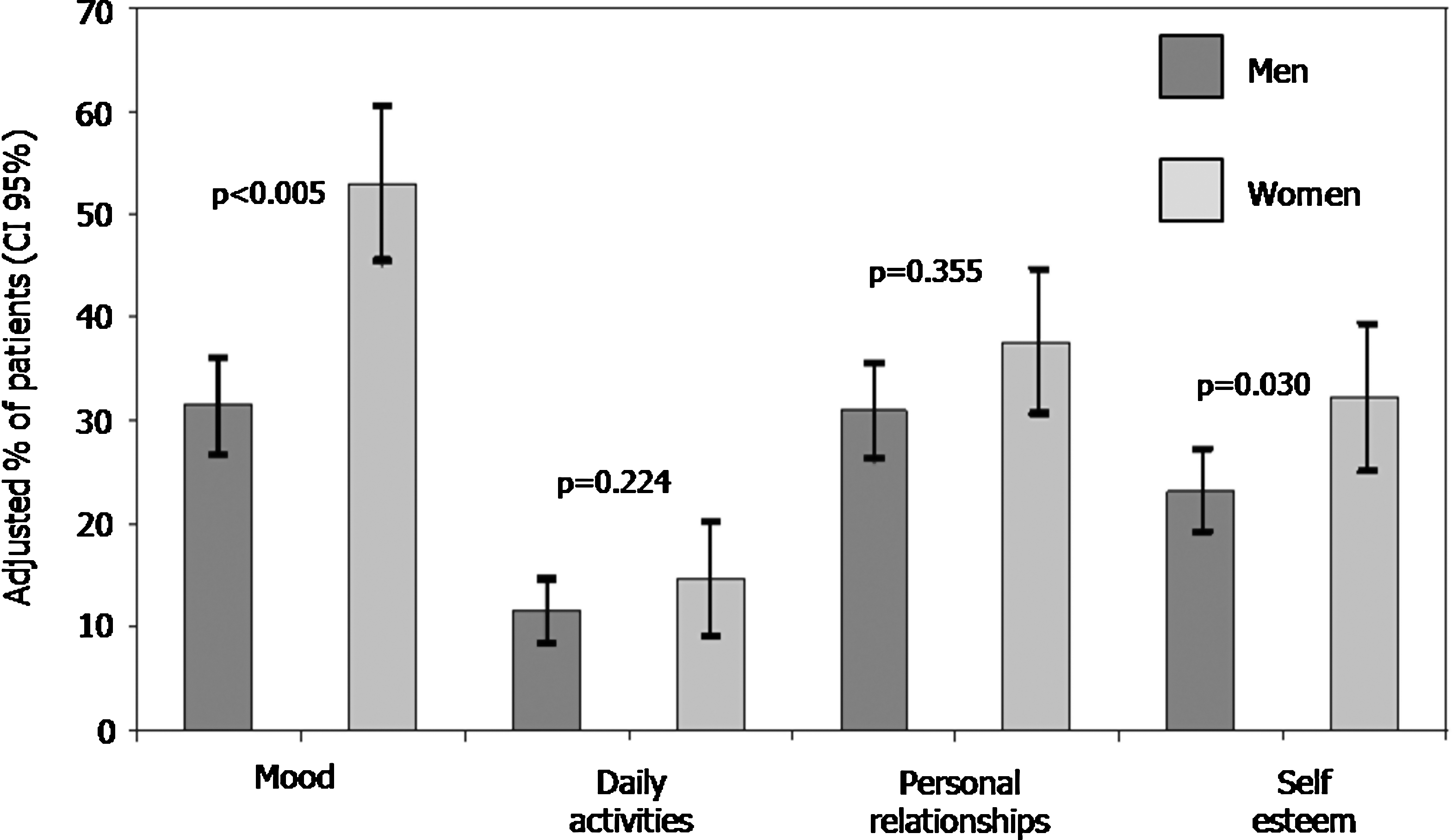
Impact of body physical changes on daily life among men and women reporting body physical changes (BPC).
Discussion
The results of our study demonstrate that BPC perceived by HIV-infected patients as well as by their treating physicians is highly prevalent. In fact, the estimated prevalence of perceived BPC doubled the initial statistical estimation of 25%, underscoring the significance of our findings. The most common BPC described by patients and physicians was lipoatrophy, with estimated prevalence rates of 46.8% and 49.4%, respectively. Lipohypertrophy was the next BPC most reported by patients and physicians, with prevalence rates of 28.7% and 25.7%, respectively. In relation to lipodystrophy as a whole, although studies have yielded variable results, 21,41,43 –47 those including a similar sample size as the one used in our study reported prevalence rates of lipodystrophy perceived by HIV-infected patients that range between 28% and 49%. 43,47 The reason behind these differences might be the lack of an objective definition and measurement of lipodystrophy, 21 and the presence of confounding factors such as age, gender, and type and duration of HAART. 22
When patients and physicians were asked to grade the severity of lipodystrophic changes, all of them were regarded, for the most part, as mild or moderate. Lipodystrophy is a complex syndrome, and its etiology is far from understood, 17 and so its treatment is equally complex. Antiretroviral switching has been rigorously studied and, although results on improvement of lipohypertrophy are confusing or even contradictory, 48 current data on improvement of lipoatrophy indicate that switching to less toxic NRTIs (e.g., avoiding thymidine analogue NRTIs) such as abacavir or tenofovir leads to a limb fat gain, but this fat improvement is slow and incomplete. 49 –51 Thus, given the difficulty of reverting lipodystrophic changes, and since there are less toxic therapeutic options available, it seems reasonable to prevent the development of this disorder at its earliest and less severe stages than to wait until it becomes clinically evident.
In our study, significantly more patients perceived skin pigmentation changes than physicians. Some of the reasons behind this might be that dermatological conditions are less evident than other BPC, and hence are perceived less frequently by the attending physician. On the other hand, conjunctival pigmentation was perceived by almost as many patients as physicians. Jaundice and conjunctival pigmentation, a consequence of hyperbilirubinemia, is a side effect developed by approximately 30% of patients taking the PI atazanavir. 52 Although these signs are reversible upon discontinuation, and elevations in unconjugated bilirubin do not seem to produce other significant side effects besides those related to appearance, 52 the presence of jaundice may lead to esthetic concerns and social distress.
One of the objectives of this study was to investigate the impact that the development of BPC has on daily life of HIV-infected patients. Given the high prevalence of BPC found in our study population, this matter is worth considering. Our results showed that more than half (58.2%) of the patients reporting BPC thought that these affected their daily life; this effect seemed to be greater within the female population, and significantly greater for mood and self-esteem implications with respect to men. Although few studies have assessed this issue, those that have done so agree that appearance-related changes do have a significant impact on daily life in HIV-infected patients. 39 The development of lipodystrophy in HIV-infected patients produces feelings of shame, influences choice of clothing, disrupts sex life, impairs psychosocial functioning and generates bodily discomfort, low self-esteem and depression. 38,53 In addition, patients with lipodystrophy often feel they can be recognized as HIV positive by their physical appearance, 46 a reminder of the early years of AIDS when BPC developed by HIV-infected patients were recognized as true landmarks of the disease. Our results confirm these observations, as lipodystrophy had a negative effect on body image perception and self-esteem, which could eventually lead to stigmatization of interpersonal and intimate relations, as has been demonstrated by other authors. 44,54,55 Above all, several studies have proven that the negative effects that BPC have on daily life are an important source of reduced adherence to therapy, 40,41,56 particularly in patient-perceived BPC in comparison to clinically diagnosed BPC. 57
We acknowledge that the results of this observational study should be interpreted in the light of potential limitations inherent in epidemiologic studies; also, the use of validated health-related quality of life questionnaires could have been helpful to determine with certainty whether HIV-infected patients with BPC have a worse quality of life and functionality than patients without BPC. Nevertheless, our study provides significant data on BPC prevalence in the HIV population on HAART in our country, which had not been previously estimated. Furthermore, we consider this study is of special relevance due to the lack of trials assessing daily life in HIV-infected patients, and the growing interest in the well-being of patients with this disease in the HAART era. Thus, based on these two main outcomes, it is of special importance that physicians treating HIV subjects actively search for BPC and ask their patients about the impact of BPC on their daily lives.
Finally, health professionals should be aware of the psychosocial consequences of BPC in HIV patients in order to reduce potential negative effects on therapy, such as reduced adherence to HAART. A reasonable strategy would be to start therapy early with a regimen not associated with lipodystrophy, or to switch to less toxic antiretrovirals as they become available.
Footnotes
Acknowledgments
The authors acknowledge Dr. Jesús Garrido for the statistical analysis of the study and Dr. Ximena Alvira from HealthCo SL (Madrid, Spain) for assistance in the preparation of this manuscript. Participation of the following investigators belonging to the HIV Body Physical Changes Study Group is gratefully acknowledged:
HIV Body Physical Changes Study Group Members:
Author Disclosure Statement
No competing financial interests exist.
