Abstract
Patients' adherence to antiretroviral medications is a primary determinant of both the effectiveness of treatment and the clinical course of HIV/AIDS. This empirical review is intended to compare the relative importance of patient and treatment characteristics on nonadherence behavior and the impact of nonadherence on treatment failure. Articles cited in PubMed and published between 2006 and June 2008 (n = 200) were reviewed to select those that address patient or treatment characteristics associated with nonadherence. Twenty-two articles were selected that provided odds ratio or hazard ratio statistics that quantified predictors of patients' level of nonadherence (e.g., <80%, 80%–95% and >95%). Results were summarized using random effects meta-analytic models. Predictors of nonadherence were divided into four predictive clusters (clinical predictors, comorbid predictors, treatment competence predictors, and dosing predictors). The summary odds ratios (ORs) of nonadherence for each cluster (in order of strength) were treatment competence 2.0 (95% confidence interval [CI]: 1.6–2.6), clinical predictors 1.6 (95% CI: 1.4–1.8), comorbid predictors 1.6 (95% CI: 1.4–1.8), and dosing predictors 1.5 (95% CI: 1.3–1.7). The effect of nonadherence on treatment failure supported the findings of two prior empirical reviews (OR 2.0, 95% CI: 1.6–2.5). Within dosing predictors, a pill burden of more versus less than 10 pills per day was associated with a much higher odds of nonadherence than twice versus once daily dosing or small differences in the number of types of antiretroviral treatments in a regimen. These results provide insight into the relative importance of various determinants of patient nonadherence that may inform the design of patient educational initiatives and initiatives to simplify treatment regimens.
Introduction
A
There have been two major reviews of medication nonadherence and HIV clinical outcomes over the last 4 years. One performed by Simoni et al. 2 reports on 77 articles published between 1996 and 2004. A second meta-analysis by Nieuwkerk and Oort 3 reports on 65 articles published between 1996 and 2003. Both articles suggest that the risk of HIV regimen failure is doubled among patients who are nonadherent to treatment. The meta-analysis by Nieuwkerk and Oort examined the adherence predictors of plasma HIV RNA concentration outcomes among adult patients prescribed ART using self-reported adherence measures. Across all studies, the pooled OR of detectable plasma viral load in nonadherent patients was 2.3 (95% CI: 2.0–2.7) compared to adherent patients.
Simoni and colleagues 2 also report an overall doubling of the risk for viral breakthrough among nonadherent patient samples, although they suggest that a number of complexities in the field hinder clear specification of a precise relationship between nonadherence and proximal clinical outcomes. These include: (1) the absence of standard approaches to define and operationalize adherence; (2) great variability in the time periods over which adherence data are observed (days to years); (3) the wide variety of methods used to assess medication adherence; and (4) the great diversity of study samples. In addition to specific measurement and methodological problems, biologically based interactions between medication adherence, viral replicative capacity, and medication pharmacokinetics in vivo preclude clear specification of adequate versus poor adherence behavior. Since publication of these reviews, some progress has been made to address these concerns.
Control of HIV replication and viral resistance
Maintaining a therapeutic plasma level of multiple antiretroviral agents is the primary determinant of effective HIV suppression and prevention of viral breakthrough. Ctrough levels describe the lowest plasma concentration between dosing cycles, and are affected by inadequate adherence to HIV medication regimens as well as drug metabolism. Because poor adherence to HIV medication often leads to incomplete suppression of viral replication, plasma viral load (described by log of HIV-1 RNA or viral copies per microliter) is the most easily observed and immediate response to treatment failure. As viral breakthrough intensifies, a patient's CD4+ lymphocyte count drops. This decline has a more direct relationship to clinical symptoms and AIDS-related morbidities than viral load. 4 For this reason, the clinically based Centers for Disease Control and Prevention (CDC) criteria for HIV disease progression is based primarily on CD4+ count. 5
These antiretroviral agents belong to one of five main HIV treatment classes: Binding or entry inhibitors (CCR5), PIs; nucleoside analogue reverse transcriptase inhibitors (NARTIs or NTRTIs), NNRTIs, and the integrase inhibitors (IIs). The class membership is defined by the treatment mechanism of action and by which step in the HIV virus adsorption, integration, or replication pathway is interrupted by treatment. 5
In the 1990s combinations of HIV therapeutic agents, appropriately named highly active antiretroviral therapy (HAART), were shown to be more effective at preventing viral replication and HIV/AIDS-related morbidity than treatment regimens containing a single medication class. The use of multiple classes creates a series of simultaneous barriers to viral replication at multiple points in the replication pathway. In order for the HIV virus to overcome and “breakthrough” multiple treatment barriers, a greater number of resistance mutations would have to occur in close temporal proximity.
Resistant viral strains can be either acquired at the time of infection or spontaneously occur through a process of selection in the presence of only partially effective blockades to viral synthesis. Medication levels below minimally therapeutic plasma concentrations (Cmin) allow for slow viral replication of resistant strains. The capacity of HIV to replicate in the presence of suboptimal levels of medication (i.e., replicative capacity) results in the emergence of viral resistance. 6 Viral resistance has serious treatment implications. With few exceptions, previous failure of a particular type of HIV medication renders all others in its class ineffective as a future treatment options. 7 Moreover, such resistance can be passed onto other individuals, with serious implications for available treatment options.
Until very recently, the need for multiple medication classes to maintain an effective blockade, along with different dosing schedules due to varying pK characteristics have meant that patients are typically required to take a large number of different types of pills, several times a day. Further complicating the situation is the need to follow strict dietary requirements, since therapeutic dose levels are affected by gastrointestinal absorption rates. As a result, patients are asked to adhere to strict administration requirements in terms of timing, dosing, and dietary conditions, all of which over time, are associated with various types of nonadherence events. 8
Challenges facing definition of optimal HIV treatment nonadherence
Strict adherence to a medication regimen is necessary in order for medications to be clinically effective and result in a sustained suppression of HIV viral replication. 9 –11 Therefore, drugs that are associated with better adherence are clinically favored, and factors affecting adherence are important when deciding on an appropriate anti-HIV treatment regimen. 12 As an example, PI-sparing medication regimens are clinically chosen over those that contain a PI, because PI-based regimens are typically associated with more severe side effect profiles, dietary administration requirements, and lower levels of medication adherence. 13
The level of treatment nonadherence affects not only whether viral breakthrough and a decline in CD4+ count occurs within a given time period, it also affects the length of time that ART continues to work after initial viral suppression. 14,15 Moreover, a number of studies have observed a dose-response phenomenon between nonadherence and decrements in the durability of treatment response assessed by viral load and CD4+ count. 16 For example, the time to virologic failure has been shown to be associated with the frequency of dosing in a lopinavir/ritonavir PI-based regimen, likely because of worse diarrhea in twice-daily than once-daily dosing schedules. 15 While a primary goal of “medication simplification” is reduced dosing requirements, this not always easily achieved. 17
Based on the pharmacokinetics of medications and the associated plasma clearance rates, some medication regimens maintain an effective barrier to viral replication (plasma C min levels) even when patients are less temporally consistent with medication use. For example, efavirenz has a therapeutic dose half-life of 40 hours, allowing for longer effective viral suppression than other antiretrovirals. 18 As a result, researchers will occasionally use different nonadherence criteria based on the pharmacologic characteristics of a particular medication regimen and choose nonadherence criteria at the point where nonadherence becomes problematic.
Also frustrating attempts to settle on a standard adherence threshold is the observation that the HIV virus begins to replicate at different drug plasma concentrations based on the medication and drug mechanism. 6 For example, boosted PIs may allow for HIV replicative capacity just below effective therapeutic dosing, thereby requiring more strict adherence, while HIV replicative capacity in patients on an NNRTI regimen may begin at much lower subtherapeutic plasma concentrations and be more forgiving of minor nonadherence.
Determinants of medication nonadherence
Nonadherence is the extent to which a person's use of medication fails to coincide with medical advice. There have been a number of recent empirical reviews of the predictors of medication nonadherence and the impact of various levels of nonadherence on viral breakthrough. These reviews identify certain patient characteristics are associated with medication nonadherence, such as drug addiction, psychiatric comorbities (e.g., anxiety, depression) and poor social support 19 –21 ; while clinical variables of high baseline viral load, low CD4 counts and coinfections are associated with poorer treatment outcomes. 19 Moreover, levels of medication nonadherence and rates of treatment failure can be reduced by providing comprehensive and structured clinical management services, particularly those that address patients' problems with drug addiction and limited psychosocial resources. 20 Such findings often result in the elimination of more problematic subpopulations such as drug users and homeless from trials and research summaries. 22
When patients are asked to give various reasons for missing doses and taking less ART medication than prescribed, these reasons generally fall into four categories: conflicts with daily routines; poor medication tolerability; regimen complexity or burden; and financial and access barriers to treatment. 23 –25 More specifically, treatment characteristics that contribute to nonadherence are the frequency and severity of side effects, as well as interference of administration requirements with daily routines (e.g., dietary requirements, dosing frequency, and the number and of medications in the regimen). 8,15,26 Drug development strategies designed to simplify regimens therefore seek to reduce the common reasons for nonadherence and ultimately improve clinical outcomes. 13
Because of the complexity of treatment regimens, inexperienced patients may be less well informed and be inadvertently nonadherent or ineffective with HIV medication use. 27,28 Adherence to complex medication regimens may also be compromised by mental health comorbidities associated with poor comprehension, forgetfulness, and functional disorganization. Finally, the therapeutic alliance between patients and their caregivers can affect adherence by influencing patients' expectations and knowledge about effective medication use. To date, however, there has not been a systematic comparative review of the effects of either medication characteristics or patients' level of administration competence, both of which appear to affect medication adherence.
Measurement of medication adherence
Accurate assessment of adherence behavior is an essential, yet enigmatic component of any attempt to study adherence to antiretroviral medications. 2 There is no gold standard for adherence assessment. Various methods have been developed to measure antiretroviral adherence: lab assessments of biologic levels of drugs or drug metabolites; the results of pill counts, typically taken at the time of clinic visit; the electronic recording of when medication bottles are opened; patients' self-report of their level of adherence to medication schedules 29 ; pharmacy refill records; and provider reports of directly observed treatment (DOT). 30 Each of these methods has its strengths and limitation in terms of the degree of random error and types of systematic biases that influence estimates of medication nonadherence. 31 –33
The precision of any nonadherence point estimate will be influenced by measurement error and systematic response bias associated with the assessment method. An example is the systematic bias observed using self-reported medication adherence measures, where the effects of response bias appears to result in greater self-reported nonadherence than clinical assessments, 34 which may explain why the concordance between electronic drug monitoring and self-reports are higher among less adherent groups than more adherent individuals. 35 Such biases likely account for low sensitivity estimates associated with many self-report measures, although the specificity (i.e., accurate identification of those that report nonadherent behavior against a viral load criterion) appears to be very good. 36 To balance such drawbacks, which are inherent in any method, some researchers recommend using multiple approaches. These include a “Composite Adherence Score” based on a primary measure (e.g., the Medication Event Monitoring System [MEMS] cap data) and additional measures (e.g., pill counts or interview data) to supplement missing or defective MEMS cap data. 37
Operational definitions and considerations
The most commonly reported ways that medication nonadherence statistics are: taking adherence (refill adherence); dose adherence (correct adherence to dose); schedule/timing adherence; dietary adherence; differential adherence (reported for each medication in a regimen); and time updated adherence (rolling average of adherence over time). Assessments that allow more fine-grained analysis permit computation of a number of different types of nonadherence estimates, and raise some interesting questions. For example, patients who are fully dose compliant may still be nonadherent with respect to dietary or scheduling requirements; which forms of nonadherence are most important to the research questions being asked? Also, which types of nonadherence statistics are most appropriate to use with the identified patient samples and treatments they are taking? As the number of compliance criteria increase, summary estimates of the pooled nonadherence in the sample should be considered carefully. 38
Nonadherence cut-points are often defined and reported as ranges, typically more than 95%, 95%–80%, and less than 80% adherence. In some studies these cut-points are chosen based on the degree of nonadherence that medication regimens can withstand before failing. In other studies, a standard scoring algorithm is available and applied for a particular assessment method. Still other researchers avoid the use of cut-point criteria altogether and report the effect sizes associated with incremental changes in nonadherence (e.g., 100% 99%–90%, 89%–80%, etc.) on clinical outcomes. This particular approach is very informative but provides quite small effect size estimates in comparison to the cut-point method. Variation across studies in which cut-point criteria are applied makes comparisons of adherence statistics difficult, and can attenuate a pooled effect size estimate across studies.
Adherence statistics can also be reported by regimen or by each individual medication within the regimen. Although this distinction is becoming less important as medications move towards combination dosage forms, “differential” nonadherence rates for each medication in a regimen can provide important information on drug performance in vivo. 39
Timing of the observational windows
The length of time nonadherence is assessed varies greatly across studies; from self-reported or observed behaviors occurring over the course of several days, to less detailed nonadherence assessments using pharmacy script refills over the course of many months or years. Each comes with a unique set of adherence metrics and computational methods to delineate levels of nonadherence.
Longer cohort pharmacy script studies of patients in large HIV treatment registries use mixed populations that differ in terms of the length of time patients have been on ART and what types of treatments they have taken. Often such studies cannot feasibly resolve the large number of interactions between diverse treatment regimens and treatment histories. In contrast, prospective adherence studies that use repeated, short observational windows are typically more detailed and can account for both medication and patient characteristics when modeling the impact of nonadherence on clinical outcomes. As discussed, such detail allows for operational definitions of adherence to include such things as missed doses, lax dosing schedules, 15 and failure to observe dietary restrictions. 38,40,41
Longitudinal models of clinically important outcomes such as virologic suppression and virologic failure over time reveal the temporal effects of a dose-response relationship with level of nonadherence, 14 such results are often displayed as a survival curve. The implication is that the points in time that assessments are made after patients' start therapy can influence strength of the observed relationships.
Objectives of the Study
This empirical review covers recent HIV treatment literature that has examined the relationship between the reasons for patients' nonadherence to HIV medication and treatment failure, and was intended to compare the relative influence of psychosocial and treatment factors on patient nonadherence. It was anticipated that a comparative evaluation of the various causes of nonadherence behaviors would help inform the educational and clinical programs aimed at promoting patient adherence to HIV treatment regimens. The key questions addressed in the study were: What patient characteristics and medication attributes affect HIV medication nonadherence? What types of clinical conditions and comorbidities affect nonadherence and short-term indicators of HIV treatment failure? What observations can be made with respect to the relative effect sizes between various predictors of nonadherence and medication failure?
Methods
A literature search of PubMed/MEDLINE was used to identify articles published between 2006 and 2008 that addressed various adherence predictive clusters. Articles were chosen that provided odds ratios (OR) and hazard ratios (HR) as effect size estimates of the relationships between treatment nonadherence and viral breakthrough or treatment-related predictors of nonadherence and nonadherence.
Search and selection strategies
PubMed/MEDLINE was searched for potential articles using the following three search strings:
Search string #1: Measurement of nonadherence (resulted in 125 articles)
(((“HIV”[Mesh]) OR (human immunodeficiency virus)) AND (adhere* OR complian*) AND (viral load OR breakthrough OR break through OR “Drug Resistance, Viral”[Mesh] OR “CD4 Lymphocyte Count”[Mesh])) AND ((composite adherence score) OR (ACTG adherence) OR (AIDS clinical trials group adherence) OR (pill count*) OR (prescription fill*) OR (“Drug Monitoring”[Mesh]) OR (“Self Assessment (Psychology)”[Mesh]) OR (mems caps) OR (adherence questionnaire) OR (self report* OR self-report* OR patient report* OR patient-report*) OR (drug level)) AND (“last 3 years”[PDat] AND (Humans[Mesh]) AND (English[lang]))
Search string #2: Determinants of nonadherence (resulted in 51 articles)
(((“HIV”[Mesh]) OR (human immunodeficiency virus)) AND (adhere* OR complian*) AND (side effects OR convenience OR effectiveness OR efficacy) AND (viral load OR breakthrough OR break through OR “Drug Resistance, Viral”[Mesh] OR “CD4 Lymphocyte Count”[Mesh])) AND ((composite adherence score) OR (ACTG adherence) OR (AIDS clinical trials group adherence) OR (pill count*) OR (prescription fill*) OR (“Drug Monitoring”[Mesh]) OR (“Self Assessment (Psychology)”[Mesh]) OR (mems caps) OR (adherence questionnaire) OR (self report* OR self-report* OR patient report* OR patient-report*) OR (drug level)) AND (“last 3 years”[PDat] AND (Humans[Mesh]) AND (English[lang]))
Search String #3: Dosing effects on nonadherence (resulted in 36 articles)
((“HIV”[Mesh]) OR (human immunodeficiency virus)) AND ((adhere* OR complian*) OR (composite adherence score) OR (ACTG adherence) OR (AIDS clinical trials group adherence) OR (adherence questionnaire)) AND ((pill burden) OR (pill number) OR (number of pills)) AND (“last 3 years”[PDat] AND (Humans[Mesh]) AND (English[lang]))
Article selection and data extraction
Candidate articles (n = 200) were identified that were published in English between January, 2006 and June, 2008. Only abstracts that included, or suggested the presence of ORs or HRs related to psychosocial or treatment characteristics were selected for review. This selection process resulted in 91 full-text articles being reviewed further, with 23 of these identified as containing OR or HR data that could be included in the empirical review (Table 1).
ART, antiretroviral therapy; NNRTI, non-nucleoside reverse transcriptase inhibitor; HAART, highly active antiretroviral therapy; VL, viral load; PI, protease inhibitor; NVP, nevirapine; BID, twice daily; MEMS, Medication Event Monitoring System; Tx, treatment; SSRI, selective serotonin reuptake inhibitor; 3TC, lamivudine; ddI, didanosine; HRQOL, health-related quality of life.
Because the dimensions assessed across studies varied widely, unadjusted bivariate OR and HR values were sought whenever available, although it was observed that the adjusted statistics rarely differed much from the unadjusted values. In cases where unadjusted values were not provided or extractable, multivariate values were used.
Statistical methods
Some ORs were inverted by computation using the original dataset so that ORs were expressed consistently across studies. Close attention was paid to making sure that the correct attributions were made with respect to interpretation based on the reference group. For example Hicks et al. 49 reported an OR of 0.45 for good versus inadequate adherence associated with persons who reported hazardous drinking compared to those who did not drink. This statistic was reversed so that hazardous drinking was associated with an OR for inadequate adherence.
A spreadsheet was created with each line conceptually classified based on the type of adherence assessment method used, the type of predictor and outcome groups used, and the predictive cluster the effect size estimate belonged within. Table 2 presents the codes that were evolved within each predictive cluster based on the data that were available within the identified studies.
VL, viral load; Tx, treatment.
A random effects model was used to estimate the summary OR/HR and 95% confidence intervals associated with each predictive cluster.
Results
OR/HR data gleaned from the 22 articles were grouped according to one of the four predictive clusters of nonadherence (Tables 3 –6) or nonadherence predictors of viral break-through (Table 2).
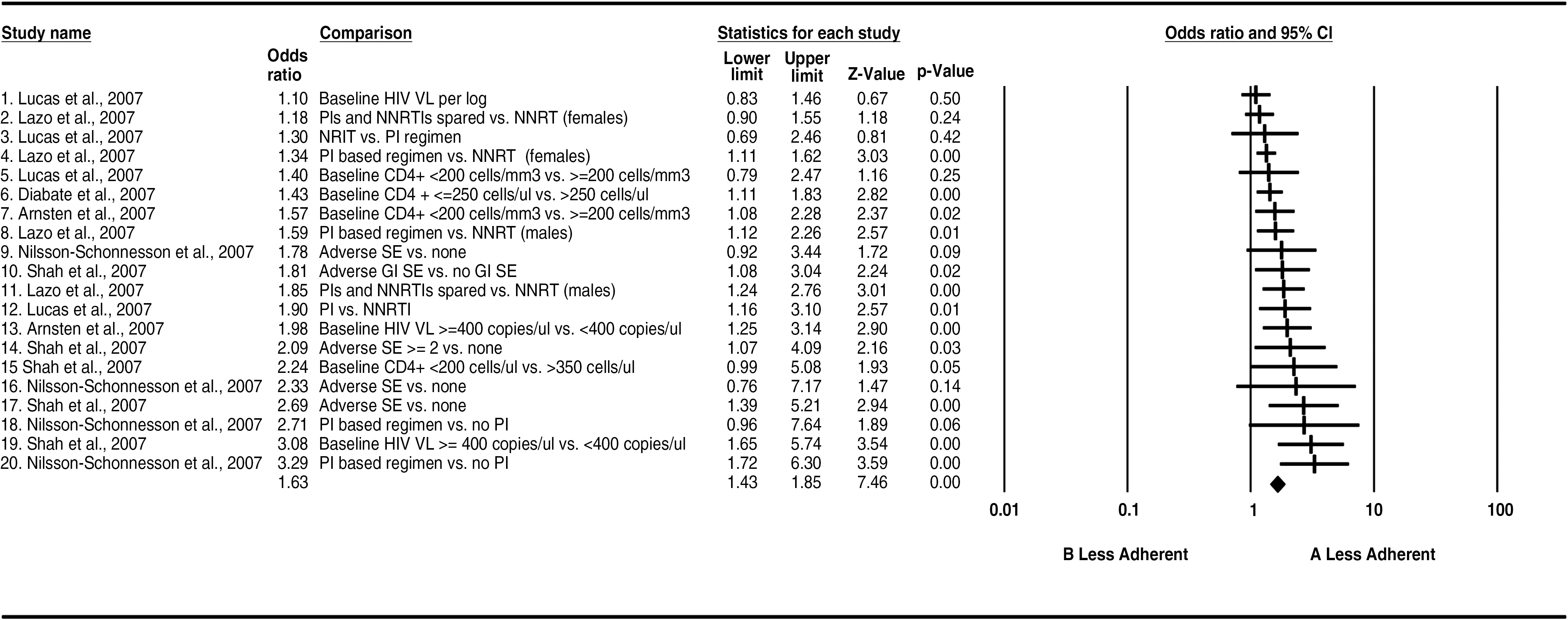
Note: The index group (A) and reference group (B) used for odds ratio (OR) computations for each statistic are listed in the Comparison column.
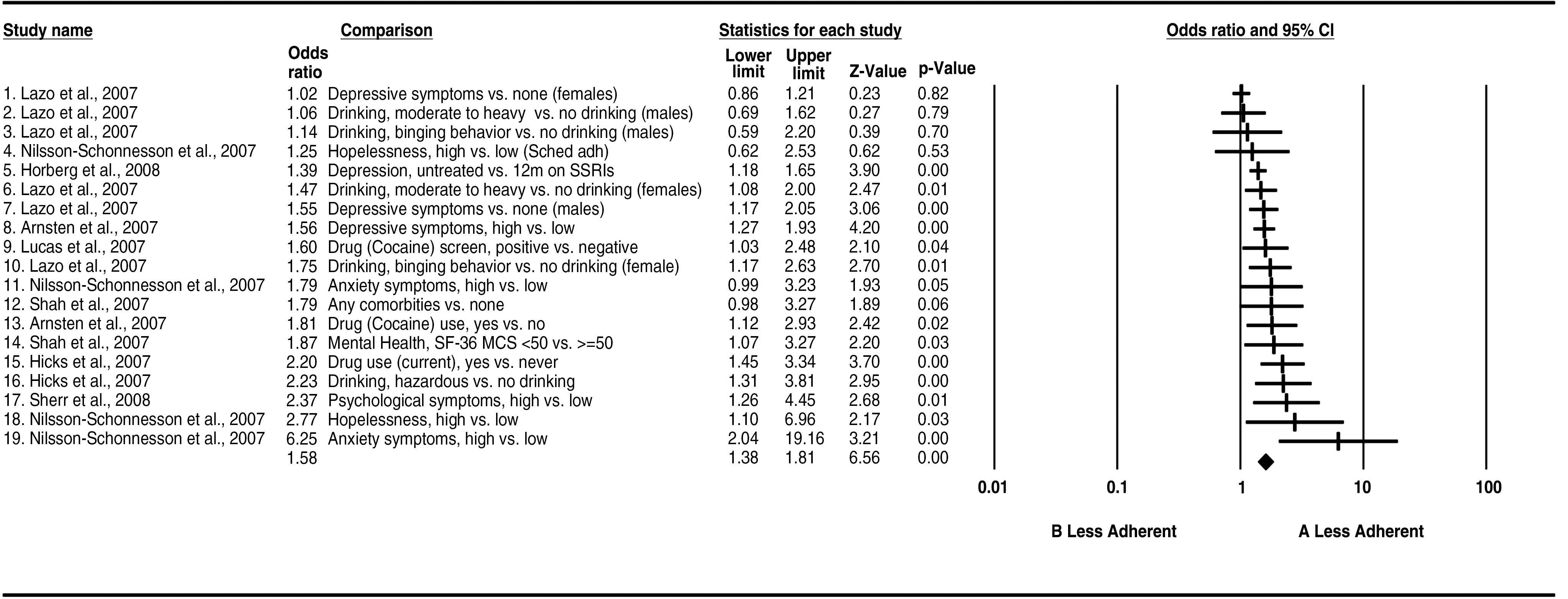
Note: The index group (A) and reference group (B) used for odds ratio (OR) computations for each statistic are listed in the Comparison column.
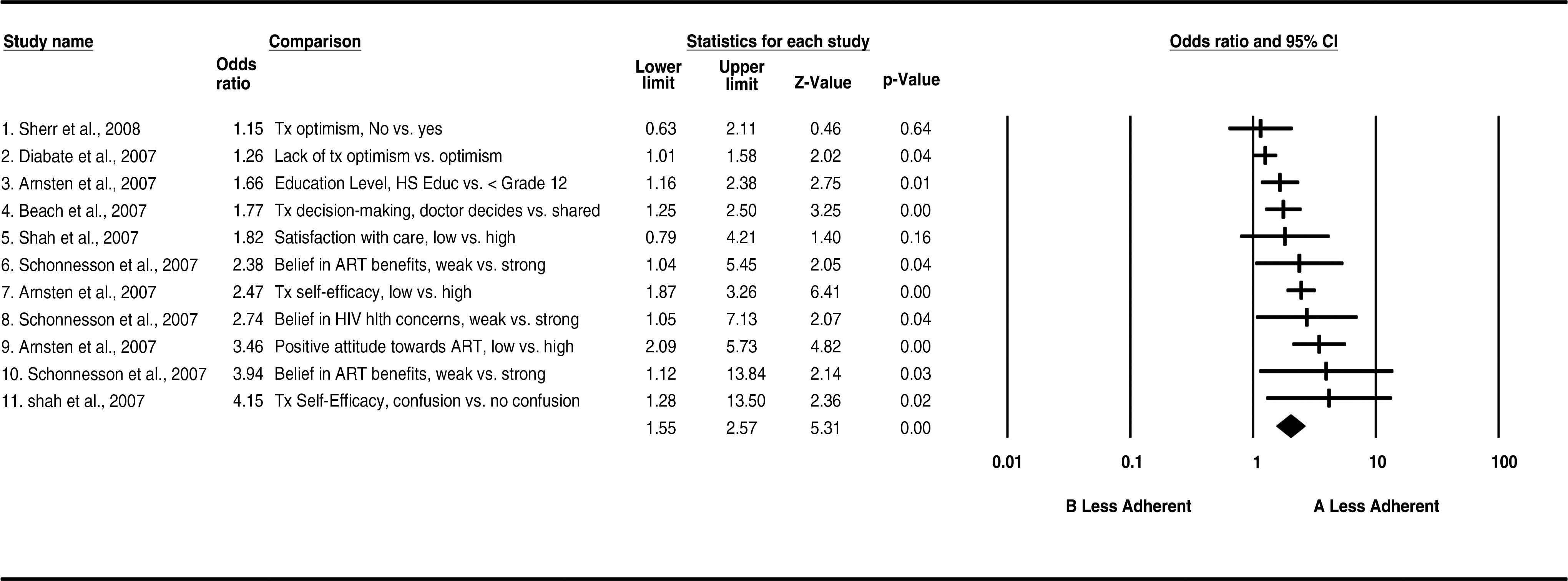
Note: The index group (A) and reference group (B) used for odds ratio (OR) computations for each statistic are listed in the Comparison column.
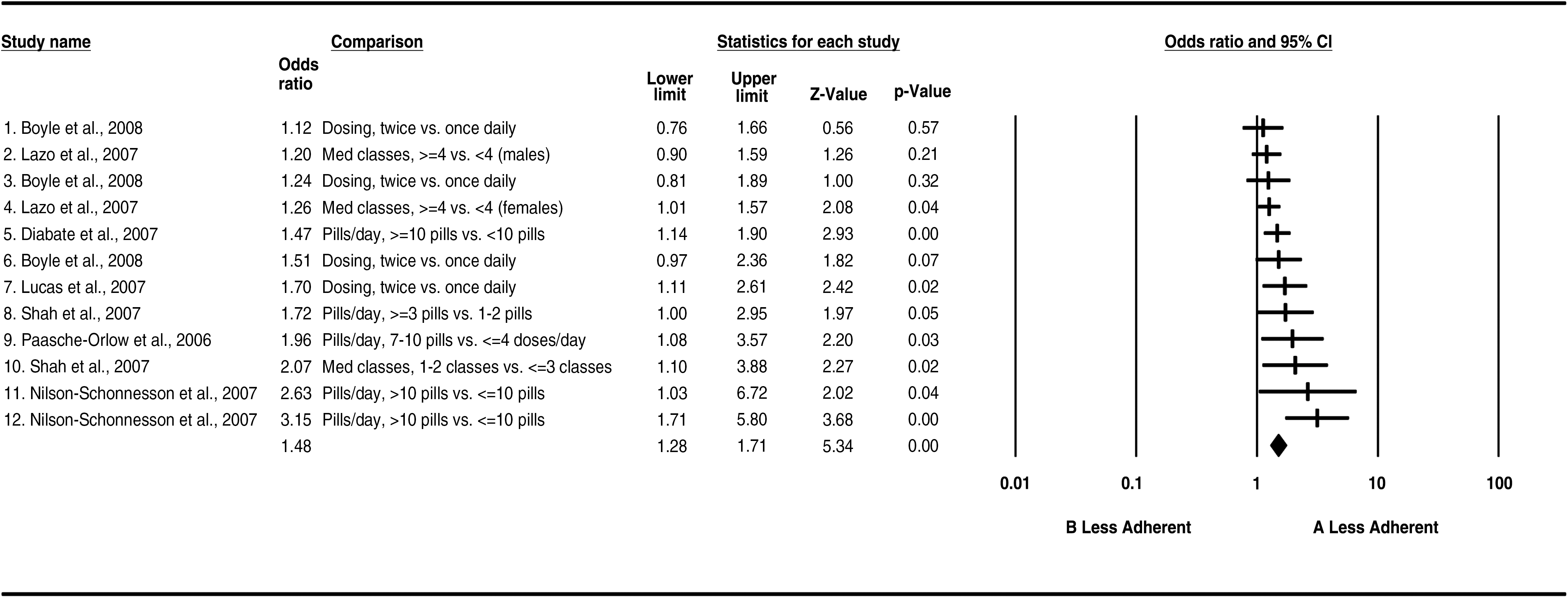
Note: The index group (A) and the reference group (B) used for odds ratio (OR) computations for each statistic are listed in the Comparison column.
The clinical predictors of nonadherence presented in Table 3 included the number and types of adverse events, baseline viral load, baseline CD4+ count, and the use of a PI regimen (Table 3). An overall OR of 1.6 (95% C.I.: 1.4–1.8) was reported for this cluster.
Comorbid predictors of nonadherence included problematic alcohol and drug abuse, depression, and psychological symptoms (Table 4). On average individuals with these comorbid conditions had 1.6 (95% CI: 1.4–1.8) greater odds of treatment nonadherence than those without such concerns. Because multiple comorbidities are not uncommon, the combined disorganizational effects of comorbities on individuals' daily lives may be synergistic, and underestimate their impact in patients with multiple mental health concerns.
Factors associated with treatment competence were consistently strong predictors of patients' medication adherence (overall OR 2.0, 95% CI: 1.6–2.6). Within this cluster patients' optimism, understanding of HIV treatment benefits, treatment self-efficacy, and sharing treatment decision-making with their clinician were associated with increased odds that they would adhere to HIV treatment (Table 5).
The findings associated with dosing predictors show that a higher pill burden, greater versus less than 10 pills per day and to a lesser degree, a greater number of doses per day were all associated with higher nonadherence (OR: 1.5, 95% CI: 1.3–1.7; Table 6).
Table 7 provides a convincing demonstration that higher levels of nonadherence are associated with higher odds of viral breakthrough. This occurred despite the use of different adherence assessment methodologies being applied across the studies. The overall OR of treatment failure across study groups was 2.0 (95% CI: 1.6–2.5), results that are in line with the summary OR of 2.0 reported in the earlier meta-analysis of ART adherence by Simoni et al. 2 and 2.3 reported by Nieuwkerk and Oort. 3
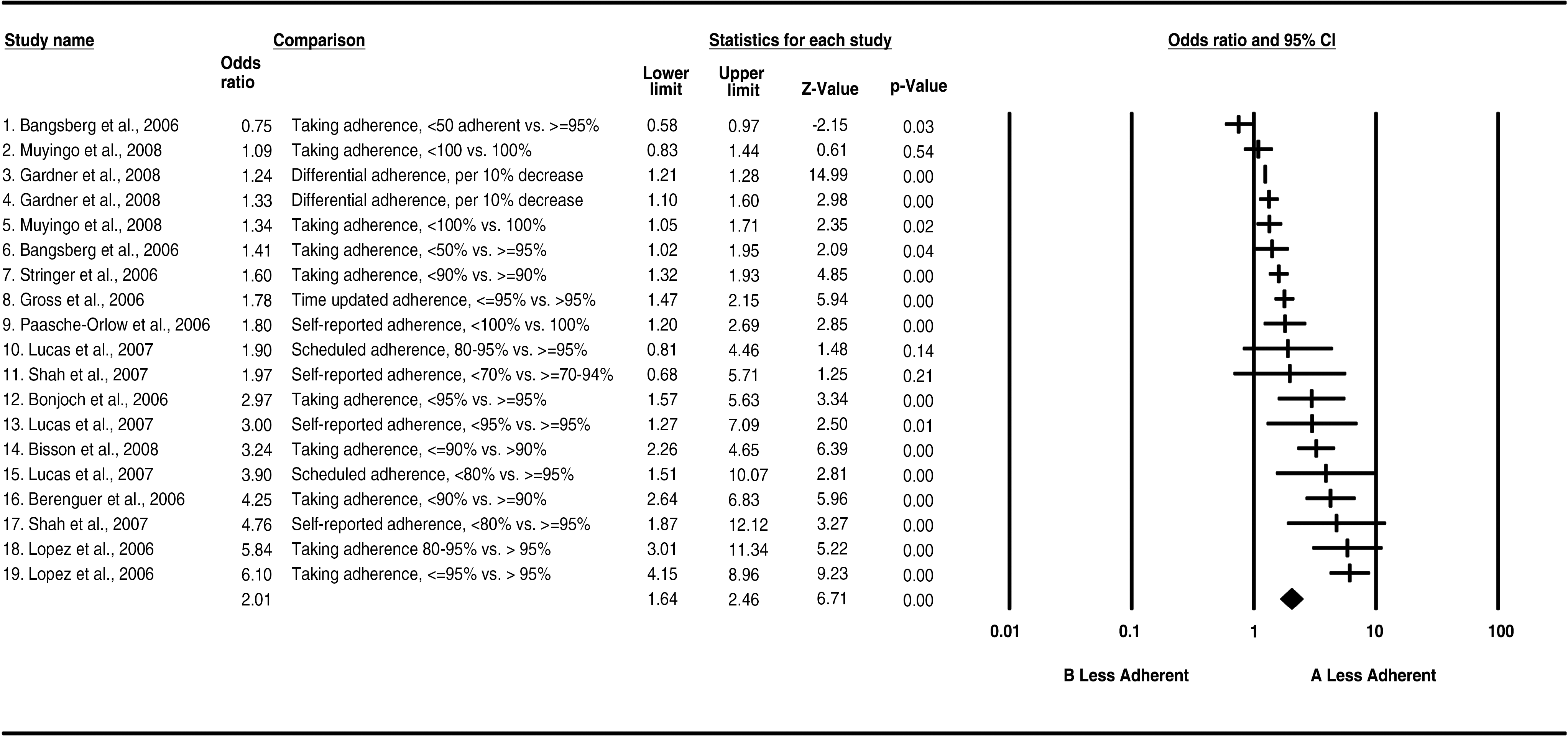
Note: The index group (A) and reference group (B) used for odds ratio (OR) computations for each statistic are listed in the Comparison column.
Table 8 provides insight into the relative importance of some specific determinants of medication nonadherence within each of the predictive clusters. Leading the list, poor treatment competence, heavy pill burden (>10 pills per day) and medication side effects are the strongest correlates of medication nonadherence. These are followed by various comorbid predictors of nonadherent behavior. Finally the number of times a day a medication is used and the number of medication classes are relatively weak correlates of medication nonadherence.
OR, odds ratio; CI, confidence interval; Tx, treatment.
Discussion
Patients' ability to adhere to rigorous and complex administration schedule is demanding. Patients should appreciate the risks of not taking their medication regularly, understand how and when to use each medication, and be organized enough to do this several times a day, with little room for error. Within each predictive cluster are findings that contribute to our understanding of why people occasionally, and sometime regularly, have difficulties with precise and consistent medication self-management.
Cluster 1: Clinical predictors of nonadherence
The clinical predictors of nonadherence suggest that persons with high baseline viral loads and compromised immune function at baseline were more likely to be nonadherent to their future HIV treatment regimen. It is possible that the effects of earlier nonadherence resulted in poorer outcomes and this behavior was simply carried forward with subsequent treatment. As expected, the number and type of side effects, and particularly those associated with PI-based treatments, exert a consistent negative effect on adherence behavior. Nonadherence estimates comparing naïve versus experienced ART patients appear to vary greatly; possibly because of the amount of education naïve patients receive when initiating HIV therapy for the first time. Supporting this idea, changes in adherence levels have been shown over time among ART-naïve patients but not among experienced patients. 27
Cluster 2: Comorbid predictors of nonadherence
The comorbidities of alcohol/drug abuse, depression and anxiety consistently predicted nonadherence behavior. Given the high degree of organization and discipline required to remain highly compliant, it is possible that the underlying causes of nonadherence among binge drinkers or drug users are impulsivity, avoidance and inadequate planning. This may not be the same for persons who are depressed and/or anxious, for whom feelings of anxiety about coping with life and hopeless attributions may affect motivation associated with effective medication management, ultimately with undesirable clinical outcomes.
Cluster 3: Treatment competence predictors of nonadherence
Patients' lack of ART competence appeared as the strongest nonadherence predictors; with poorer medication self-efficacy, incorrect beliefs about the treatment and the disease, lower optimism, and poorer understanding of treatment were associated with worse medication adherence. This cluster speaks to the central importance of patients being informed, actively involved in the treatment process, and competent to manage their own treatment. Moreover, such competencies are clearly essential to good clinical outcomes.
Cluster 4: Dosing predictors of nonadherence
This set of predictors was ranked as second in importance to treatment competence. The complex administration requirements associated with many medication regimens (e.g., a large number of pills, different types of medications with differing dietary requirements) often result in doses being delayed, missed, or not taken with the right food. The value of efforts to simplify dosing is demonstrated in studies showing that providing a pillbox organizer almost doubles the odds of patients remaining adherent while on complex regimens. 55 The relatively minor difference between once versus twice daily dosing seemed to be a weak predictor of medication nonadherence.
Cluster 5: Nonadherence as a predictor of treatment failure
This cluster underscores the general clinical importance of medication adherence in the effective treatment of HIV/AIDS. Interestingly, in contrast to predictors of nonadherence, the results presented in Table 7 came from a set of more technical and clinically focused research studies. These results provide evidence of the dose-response relationship between level of nonadherence and the odds of treatment failure. Notably, the summary OR for this cluster would have been still higher if the per unit nonadherence OR estimates, such as those reported by Gardner et al. 39 were omitted.
Summary
It was encouraging that some of the recent adherence research had acted on the earlier recommendations by Simoni et al., 2 providing results that discriminate between the various forms of nonadherence. Such discrimination allows for a deeper understanding of the causes of nonadherence and their relative importance among different patient subgroups. The current findings may help inform the design of educational interventions to address specific determinants of nonadherence that lead to undesirable clinical outcomes. Moreover, the use of differential nonadherence estimates (i.e., for each medication class) could be used to identify the relative importance of specific aspects of nonadherence as they impact medication failure, thereby informing directions for drug development. 56
The strength of relationships between predictors of nonadherence and observed nonadherence are often subject to great variation based on the chosen referent group used to compute the OR estimates. It is difficult to infer magnitude of effects associated with medication nonadherence from any particular study from the effects of researchers' choice of patient reference group, medication type/regimen, and method of adherence assessment. Across a larger number of studies, these methodological effects are averaged out.
This analysis did not apply meta-analytic regression or analysis of variance (ANOVA) methods to model the effects of estimate variation across studies. Such an approach might prove informative, although despite efforts to standardize, some problems might persist due to constraints inherent in the individual study design. For example, longer retrospective cohort studies often confound length of time on a particular therapy with nonadherence.
There is some room for improvement within the field with respect to consistent reporting of data and effect size estimates. In situations where a linear trend or dose-response is observed between the causes of nonadherence and nonadherence it would be helpful to describe the trend per unit change in the predictor, as is frequently reported regarding viral load and some dimensions of comorbidity. For example, some authors have taken steps to standardize the dose-response of nonadherence level on clinical outcomes by reporting ORs per 10% decline in observed adherence.
Limitations of the current review and future research
This review was not intended to be a comprehensive summary of all articles in the field. It was intended to clearly represent the effects of various clinical, medication-related, and patient factors on nonadherence that have been reported within recent HIV treatment literature. The relatively small number of studies on which some of the comparisons are based may not be sufficient to provide precise effect size estimates, and more study may be required.
Footnotes
Acknowledgments
Funding for this empirical review was funded through an independent grant from Merck & Co. Inc.
Author Disclosure Statement
No competing financial interests exist.
