Abstract
Introduction:
The transport of infectious substances is a highly regulated process. The European Agreement for International Carriage of Dangerous Goods by Road instructs that infectious specimens must be transported in a triple packaging system containing leakproof receptacles. Despite regulatory efforts, leakages occur. Current evidence for Parafilm usage is lacking. This study evaluates the use of Parafilm for preventing leakage from commonly used primary receptacles during ground transport.
Methods:
Two test methods, manual inversion and a rocker, were used to simulate standard ground transport. Testing was performed with two common receptacles and three differing volumes of test solution. Each test was performed in quintuplicate. Fluorescein solution was used to aid in visual identification of leakage in addition to the weight of the receptacles before and after testing.
Results:
Despite lids being correctly applied, 10% of full 30 mL universal tubes leaked as a result of the rocker testing (
Conclusion:
Clockwise application of Parafilm may decrease leakage in the event of cross-threading. Despite this, receptacles must be opened in the correct containment, as spillage during unwrapping and lid removal is common. Further investigation is required to assess the effects of pressure on modes of transport such as air travel.
Introduction
The transportation of infectious substances is required to support diagnostic services, scientific research, and disease surveillance. The most common method to transport infectious substances is road transport, although alternatives used include air, rail, and sea transport.
Infectious substances are classified as dangerous goods, 1 and as such, regulations for their transport are set out by national authorities, often guided by the international model regulations set by the United Nations Economic Commission for Europe. These regulations for road transport are enforced under “The European Agreement Concerning the International Carriage of Dangerous Goods by Road” (ADR), a treaty established by the United Nations (UN). 2 Infectious substances are classed under division 6.2 of the ADR and can be categorized further as Category A (an infectious substance that, when exposure occurs, can cause severe consequences to health) or Category B (an infectious substance that contains biological agents capable of causing infection in humans and animals but not meeting the criteria for Category A 2 ). These substances are assigned UN numbers, informing parties involved of the substance’s ability to affect human health.
The requirements for road transport of dangerous goods are specified in packaging instruction 650 of the ADR. 2 It advises a triple packaging system is used to contain the infectious material securely, protect the inner receptacles from breakage, and absorb any potential leakage during transit. There is no comprehensive list of suppliers or packaging that comply with the regulations, and as such, institutions that send and receive infectious substances often use a variety of packaging containers and materials. UN-approved triple packaging systems are available for purchase, yet inadequate practices and resource limitations in low-resource countries can lead to incidents and adverse outcomes. These outcomes include the increased risk of release to the environment, exposure to the courier, and exposure to the recipient during unpackaging, all of which pose a risk to public health.
Incidents within the transport of dangerous goods can occur for a variety of reasons but can be summarized into two groups: human factors and physical faults. Human factors are derived from human errors, poor practice, and nonadherence to the regulations provided. Alternatively, physical faults can occur within the packaging receptacles themselves. Some packaging systems available on the market have been shown to be unable to pass the +95 kPa overpressure test, which is a requirement within the ADR. 3 This testing by Sanchez et al. was designed to evaluate protection from environmental conditions that packages are often subject to.
One material that is often used in the packaging of tubular receptacles is paraffin film, commonly known by the brand name Parafilm. Parafilm is a semitransparent thermoplastic that is resistant to air, gases, alcohol, acids, and alkalis. Parafilm has water-resistant properties but is not completely waterproof. Despite the wide usage of Parafilm, no current literature evaluates the effectiveness in the sealing of sample tubes and whether it is a measure that can be implemented to reduce risk when handling and transporting infectious substances. A variety of protocols can be found online with contrasting instructions as to the use of Parafilm. Some state that the wrapping of Parafilm should be performed; however, others omit this detail or even specify Parafilm should not be used and instead suggest adhesive tape to secure the lid.4–6
While Parafilm itself is not considered a replacement for other layers of packaging, investigation of this topic provides valuable insight as to whether the use is beneficial in low-resource environments where the triple packaging system is not always adhered to. There is also relevance for the local transport of chemicals or hazard group one biological agents in which tubes containing samples are packaged and carried for internal transfer within facilities. The purpose of this study was to explore standard methodology for testing transport receptacles and examine differing applications of Parafilm and their effectiveness for sealing transport receptacles. This research will be used to provide guidance for the correct usage of Parafilm to reduce leakage from samples and subsequent release of infectious substances to the environment. Tubes were observed both correctly sealed and incorrectly sealed to represent the potential for “cross-threading” of screw-cap lids, a common error overlooked during packaging of materials. This study aimed to replicate common scenarios of transport and explore the application of Parafilm as a method of sealing tubes.
Methods
Packaging
Two different receptacles were used, 30 mL universal tubes (VWR) and 2 mL screw-cap micro tubes (Sarstedt Ltd.). Sarstedt tubes contained rubber O-rings, whereas universal tubes were only plastic threaded, as shown in Figure 1.

Examples of Sarstedt 2 mL (left) and universal 30 mL (right) tubes used for testing.
Fluorescein Solution
A fluorescein solution was used as the mock sample within this study. This solution was chosen as it provides a good comparison to low-viscosity biological samples, which are most likely to leak. Additionally, fluorescein’s ability to fluoresce under ultraviolet light was used to aid in the visual identification of leakage surrounding the test area and the operator’s hands. This provided a qualitative measure of the generation of spills. Previous testing (unpublished) observed the ability of the operator to identify fluorescein spills of small volumes. It was confirmed that the operator could identify droplets as small as 1 µL on benchkote, which was considered the limit of detection of spills.
A 0.05 g of sodium fluorescein salt powder (Sigma) was added to 500 mL of water to make a 0.01% (w/v) solution. Thirty-milliliter universal tubes were filled with either 30 or 15 mL, whereas 2 mL screw-cap micro tubes were tested with 2 mL of solution only. Further discussion of the receptacles within this article refers to the volume of solution within, not tube capacity. Three volumes of samples were tested (30, 15, and 2 mL) across two different methods of travel simulation.
Inversion Testing—Test 1
All tests carried out were performed at room temperature to represent common conditions for land transport. Each parameter set was tested in quintuplicate to ensure the validity and reproducibility of the results.
Sample receptacles were filled to their test volume, and lids were closed either correctly or cross-threaded. This is the state in which the plastic thread that screws the lid on is misaligned and can result in difficult-to-identify gaps despite the lid seeming secure. The mass of each receptacle was measured before a 15 × 5 cm rectangle of Parafilm was applied, regardless of receptacle type. In the three testing conditions where Parafilm was applied (over–under, clockwise, anticlockwise), the receptacles were weighed a second time, post application. The receptacles were inverted 10 times over benchkote (Scientific Laboratory Supplies) and left inverted for 60 min. After inversion, the receptacles were weighed to establish mass lost during the test in addition to visual identification of spillages. Subsequently, Parafilm was unwrapped (where applicable) and the receptacle opened. Both the internal screw thread and the outside of the receptacles were examined. The receptacles were weighed again to allow for calculation of mass lost post-test, accounting for any loss occurring due to the opening and unwrapping procedure. An ultraviolet torch was used to identify further spillages. This method was repeated with three differing applications of Parafilm. The third type of application involved wrapping the film from the bottom of the tube and over the top of the lid and is described as the “over–under” application.
Rocker Testing—Test 2
Sample receptacles were filled to their test volume, and lids were closed either correctly or cross-threaded. The mass of each receptacle was measured before a 15 × 5 cm rectangle of Parafilm was applied regardless of receptacle type. In the three testing conditions where Parafilm was applied (over–under, clockwise, anticlockwise), the receptacle was weighed a second time, post application. The receptacles were placed on top of benchkote on a rocker (Heidolph Instruments), which was set at 50 rpm. Receptacles were placed in either an east-facing orientation, where the rocking motion moved the liquid from base to lid, or a south-facing orientation, where the rocking motion moved the liquid from side to side. Receptacles remained on the rocker for 15 min before removal. After rocking, the receptacles were weighed to establish mass lost during the test. This was in addition to the visual identification of spillages. Subsequently, Parafilm was unwrapped (where applicable) and the receptacle opened. Both the internal screw thread and the outside of the receptacles were examined. The receptacles were weighed again to allow for calculation of mass lost post-test, accounting for any loss occurring due to the opening and unwrapping procedure. An ultraviolet torch was used to identify further spillages.
Statistical Interpretation
Statistical analysis was performed on Graphpad Prism version 10.1 software. Nonparametric tests were used as the data recovered was not normally distributed. For comparisons between the Parafilm methods and tube types, the Kruskal–Wallis test was used with multiple comparisons to identify further significance between groups. A Mann–Whitney
Results
Inversion (Test 1)
The inversion testing and resulting spillages are shown in Table 1. The total mass loss was averaged from five replicates when leakage occurred. A classification system was used to differentiate between larger spills that are more obvious to an operator and minor spills that may be harder to identify and therefore increase the risk of further contamination as spillage procedures would not be followed.
Severity of spills generated from inversion testing
Testing was performed in quintuplicate with mean mass lost being used to categorize spill severity. Spills were classified as “major” (++) for >0.2 g and “minor” (+) for 0–0.2 g or “absence of an identified spill” (—). Parafilm applications are coded as such: No Parafilm (NP), over–under (OU), clockwise (C), and anticlockwise (AC).
No spills were observed when lids were applied correctly for both receptacles. Additionally, no spills were witnessed from screw-cap micro tubes with incorrectly secured lids. Incorrectly secured lids resulted in spills identified from receptacles containing 30 and 15 mL during the test. Four (20%) receptacles containing 30 mL and five (25%) receptacles containing 15 mL leaked during testing, which resulted in major spills for the “no Parafilm” test group and minor spills for the “over–under.”
Despite similar leakage identified during the test, post-test, Parafilm-wrapped 30 mL receptacles resulted in major spills (>0.2 g), while Parafilm-wrapped receptacles containing 15 mL experienced minor spills only. These post-test spills were identified in 80% and 90% of cases in receptacles containing 30 and 15 mL, respectively.
A Kruskal–Wallis test performed on incorrect lid data did not identify significance between the different applications of Parafilm (
Rocker (Test 2)
Within the rocker test, four of the universals containing 30 mL leaked post-test despite having correctly closed lids. This leakage was identified by the presence of fluorescein on the Parafilm or benchkote during and after removal of the lid. Three of the four tubes that leaked (despite a correctly applied lid) were wrapped with Parafilm in the anticlockwise direction. Mechanical loosening of the lid was observed during the wrapping of the lid prior to the test beginning. The 15 mL of fluorescein within the universals did not experience the same leakage rates when the lids were applied correctly.
Incorrect application of the lids resulted in far higher volumes of leakage. In total, 50% (
Severity of spills generated from rocker testing
Testing was performed in quintuplicate with mean mass lost being used to categorize spill severity. Spills were classified as “major” (++) for >0.2 g and “minor” (+) for 0–0.2 g or “absence of an identified spill” (—). Parafilm applications are coded as such: No Parafilm (NP), over–under (OU), clockwise (C), and anticlockwise (AC).
As in the inversion test, no leakage was observed from the Sarstedt tubes, regardless of orientation or status of lid closure. The receptacles containing 30 and 15 mL were significantly more likely to leak than the 2 mL test group (
Results generated from both tests are plotted in Figure 2 as the mean of the total percentage lost. Each individual receptacle is also plotted. Figure 2 shows that during testing, the leakage from a receptacle was often very high or low with few values in between. A higher average spill was generated from receptacles in the rocker test, which can be noted by the difference in the
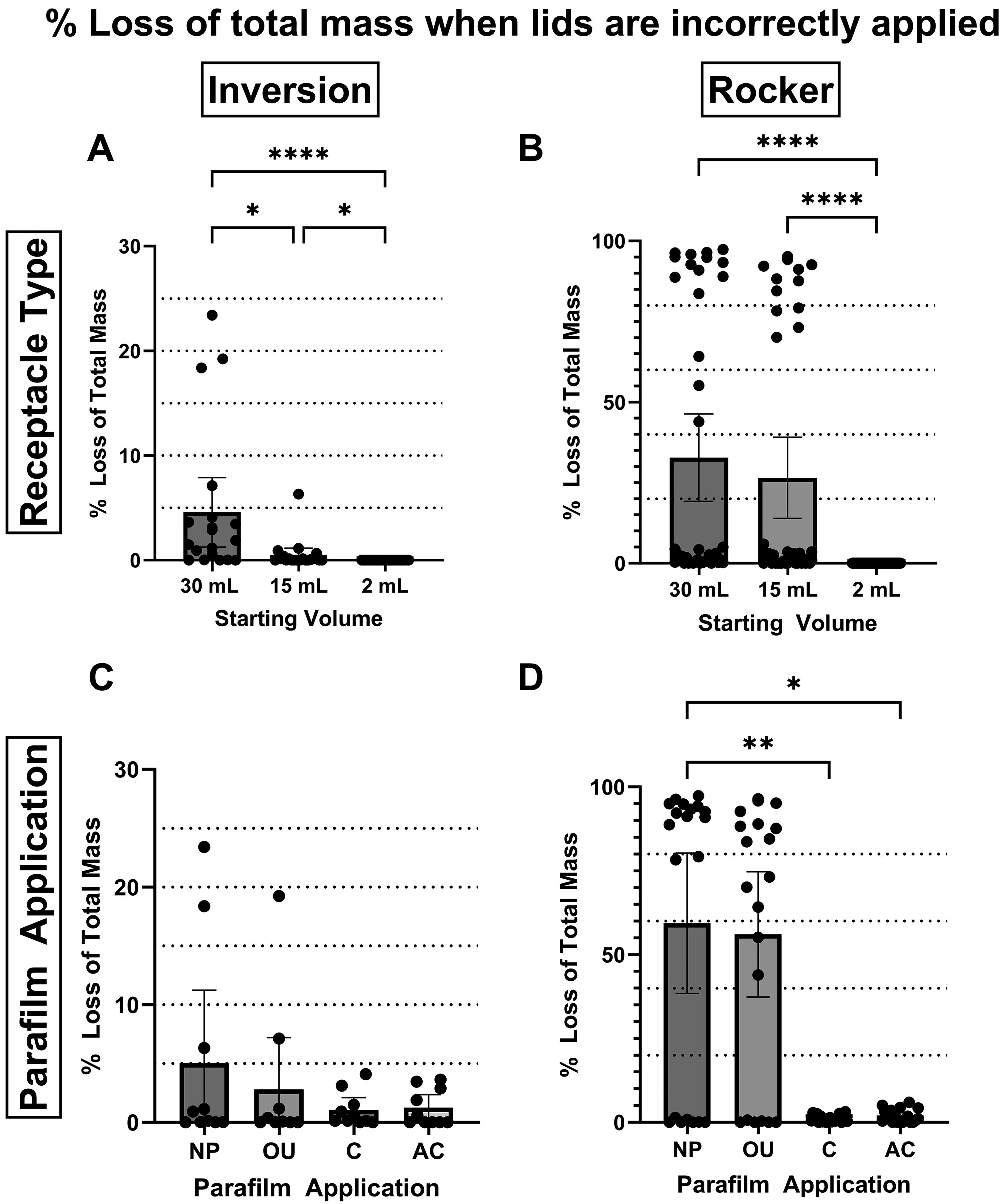
Percentage loss of mass from rocker and inversion testing (A and B). Comparisons between the Parafilm application and the tubes used are present. Parafilm applications coded as such: No Parafilm (NP), over–under (OU), clockwise (C), anticlockwise (AC). (C and D) consist of 30 and 15 mL starting volumes only.
With regards to the choice of receptacle, there was no significant difference observed in the percent mass loss between the receptacles containing 30 and 15 mL in rocker testing (
The differing Parafilm applications made no significant difference in the inversion test (
Orientation on Rocker
A Mann–Whitney
While environmental contamination was shown to often be contained by Parafilm, in some instances, droplets could form on the inner surface of the Parafilm. These droplets could then be transferred to the workers’ hands or external surfaces at varying levels of visibility. Droplets and examples of such hand contamination are shown in Figure 3.

Example of post-test spillage identified on the Parafilm, the side of the lid, and droplets on the operator’s gloves.
Discussion
Within this study, the use of Parafilm and its ability to contain infectious substances for transport was assessed. This is due to the importance of preventing exposure of infectious substances to laboratory staff receiving samples, workers handling the samples during transport, and the environment itself. Incidents of exposure may present a threat to public health depending on the agent being transported. The effectiveness of three differing applications of Parafilm was evaluated on two different commonly used receptacles. A new methodology was developed that could easily be replicated to standardize and produce a scientific method representative of land transport. Most importantly, transport by car, however the method used could apply to other modes of transport such as trains.
Previous testing of transport packaging systems has been undertaken in the literature. Singh et al. tested the effects of pressure on package integrity to simulate the effects of high-altitude shipping. 3 Within the Singh study, UN-approved packages were found to fail the +95 kPa pressure test. In addition to this, vibration of the packages could cause leakage, and the combination of altitude and vibration generated larger leakages than either alone. A more recent study conducted by Sanchez et al. 7 examined packages from the Spanish market (many of which are available internationally). They concluded that out of the UN-approved packaging systems tested, no secondary packaging receptacles were able to withstand the overpressure testing and that the use of suitable primary receptacles is essential in these systems. They state that information regarding resistance to high internal pressure is not clearly provided by manufacturers, which can lead to poor practice resulting in incidents and the use of unapproved packaging.
Further work by Singh et al. has shown multiple cases where UN-approved packaging does not pass low pressure and random vibration testing, but notably, receptacles that utilized stretch tape to secure the closure had a lower rate of failure.8,9
Comparison Between Tube Type
Within this study, the use of two types of primary receptacles allowed for comparisons between the receptacles and their effectiveness at containment of the test substance. Data was normalized by calculating the percentage of mass lost throughout testing which allowed for comparisons between the primary receptacles despite their differing volumes.
Throughout all testing, the 2 mL Sarstedt tubes did not leak on a single occasion (
The receptacles containing 30 mL were more likely to spill than the 15 mL test group with the inversion method used (
Comparison Between Parafilm Application
Differing applications of Parafilm were tested to identify potential methods of reducing the chance of leakage. The lack of literature relating to errors in transport meant these methods did not have a previous evidence base for their effectiveness. Instead, a variety of recommendations for and against Parafilm usage are available online.
During the inversion test, there was no significant difference identified between the Parafilm methods as shown in Figure 2C. While there was no statistical significance identified, the mean loss was lower in the receptacles wrapped clockwise and anticlockwise and prevented spillage during the test as shown in Table 1. Spillage was only identified during unwrapping. It is possible that the stationary nature of the tube formed a seal that reduced total leakage from the tubes rather than the Parafilm itself; however, the differences between the over–under method and the clockwise and anticlockwise methods suggest some sealant effect.
Alternatively, Figure 2D shows a significant reduction in the percentage of mass lost when Parafilm is applied in either the clockwise or anticlockwise direction when compared with no Parafilm. The stronger significance was identified when the clockwise wrapping technique was used in our testing. Therefore, we recommend that the Parafilm be applied in the same direction as the closure of the receptacle lid. For the receptacles used in this study, this was the clockwise direction. This prevents any possible unscrewing of the lid before transportation, which was observed when applying Parafilm in the anticlockwise direction.
This study has highlighted the importance of adherence to the triple packaging guidelines and the need for providing multiple layers of containment. This is in addition to the need for the correct containment facilities and precautions during the opening of Parafilm-wrapped tubes, especially with Category A pathogens. If incorrect application of the lid has occurred, 92.5% of 30 mL universals leaked. While Parafilm wrapping can reduce leaking during the transport, there is a high chance of droplet presence on the Parafilm. These droplets are difficult to observe visually, especially when working with colourless solutions. During unwrapping these droplets are likely to splatter onto the operator or the surrounding environment. This risk to the operator and receiver environment can be mitigated by following the relevant containment procedures for the sample.
These findings can also be applied to noninfectious samples where sterility is not required. In these cases, sealing the tubes with Parafilm in a clockwise direction will reduce the total chance of leakage and maintain the greatest possible volume within the tube in the event of an incorrect lid application.
Orientation of Tube on Rocker
During Test 2, we placed tubes horizontally on a rocker to simulate land transport. No significant difference was observed between placing the receptacles containing 30 and 15 mL either south or east on the rocker (
Limitations
Within this study, there were limitations. The standard conditions of 1 atmosphere pressure and 25°C are not representative of all land or even road travel conditions, particularly higher altitude regions through which packaging will be subjected to increased internal pressure. Pressure testing has been shown to influence the successful containment of transport receptacles in the literature,3,7 so further testing is required to identify if Parafilm is effective at mitigating leakage for high-altitude land transport.
Additionally, while the limit of detection was considered 1 µL for instances of spillage, pathogens can be transported in smaller volumes than this. The visual inspection of spills was therefore only able to provide qualitative evidence to support the other methods used.
Further Testing
Future research could adapt the methodology shown to investigate air transport testing and guidance. Singh and Sanchez both found that market-available receptacles were unable to withstand the +95 kPa overpressure requirements, and further examination of primary receptacles commonly used may be required. The ability of clockwise Parafilm wrapping to contain pressure-induced spillages has not yet been assessed. Parafilm usage under other environmental conditions such as decreased temperature could be assessed to represent high-altitude transport and reflect that biological samples will commonly be transported with a cooling component such as dry ice.
Additionally, further work on the method of receipt is required to identify methods to reduce contamination of the receiver when there is leakage onto the Parafilm. It is possible that methods such as centrifuging samples upon receipt could be effective at moving droplets within the lid and Parafilm to the main liquid reservoir and preventing the splattering observed on the Parafilm and operator in Figure 3. This need would be balanced with the risks of disrupting the sample and whether any existing safety procedures are in place. For example, class III biological safety cabinets are sufficient at reducing operator exposure, and therefore droplets within the cabinet are not of significant importance with regards to operator safety and only a concern with regards to sample contamination. Instead, issues arise for operators working at Biosafety Level 3 using open-fronted biological safety cabinets, as the protective air curtain at the front of the cabinet may prove insufficient to contain larger droplets generated from unwrapping. In such cases, there is a requirement for appropriate risk assessments, including suitable PPE to protect operators from spills and effective hand hygiene practices, all of which should be tailored to the relevant infectious agent.
Conclusions
The varied testing performed allowed for numerous comparisons and conclusions to be drawn. There were comparisons to be made between types of tubes, the use of Parafilm for preventing leakage, and the storage of tubes during transport.
Despite the transport of infectious substances occurring globally and the importance of safe and correct procedures, there is a lack of reporting and literature surrounding the issue. The regulations covered under the ADR and ICAO technical instructions provide the basis for legislation; however, noncompliance may still occur at a local level. This work identifies small measures that can assist in prevention and minimize the severity of incidents occurring due to the packaging instructions not being followed. All researchers have an obligation to work in a safe manner and prevent the exposure of infectious material to the environment.
In conclusion, there are several significant findings from this study that relate to the safe transport of infectious substances. It is important to remember that a multi-faceted approach is required in order to risk assess and apply the principles of safe sample transport to the user’s needs and context. Important factors include the selection of your transport receptacle. Screw-cap micro tubes with O-rings have been shown to be resistant to leakage. In addition to this, working with smaller volumes when possible reduces the consequences of spills. It is important that lids are correctly applied, and Parafilm may be appropriate to reduce the chance and severity of leakage if there are suitable decontamination and containment practices in place for the receipt of the samples. This includes the correct training and adherence to procedures regarding surface decontamination as some spills may not be visible.
In extremely resource-limited settings, usage of Parafilm or equivalent alternatives may be prioritized for use in scenarios where leakage is most likely or presents significant risks in line with the standard principles in risk assessment.
Footnotes
Acknowledgments
The authors would like to thank Kazunobu Kojima for his ongoing support and expertise relating to the Biosafety research field.
Authors’ Contributions
Conceptualization and methodology: A.S., S.P., T.P., and A.M.B. Investigation: A.S., S.P., T.P., and A.M.B. Formal analysis: J.R. Writing—original draft: J.R. and T.P. Writing—review and editing: All authors.
Disclaimer
The views expressed in this article are those of the authors, not those of the UK Health Security Agency or other funding source. Crown Copyright. UK Health Security Agency, 2024.
Authors’ Disclosure Statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and or publication of this article.
Funding Information
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the UKHSA Grant-in-Aid funding.
