Abstract
Introduction:
The Research Alliance for Veterinary Science and Biodefense BSL-3 Network (RAV3N) convened an “Applied Biorisk and Biosafety Gap Assessment Workshop” held February 9–10, 2023, in Baltimore, Maryland. As the global prevalence, complexity, and severity of infectious and transboundary veterinary diseases and emerging zoonotic diseases are increasing, there is growing recognition and concern that biorisk management data required to understand and counter these threats are lacking. With sponsorship from the U.S. Department of Agriculture and the U.S. Department of State, RAV3N partnered with Gryphon Scientific and ABSA International to organize, plan, and deliver this biosafety gap analysis workshop.
Methods:
The workshop brought together U.S. and international subject matter experts on veterinary and agricultural biorisk management from seven different countries to methodically identify, categorize, and assess the most pressing biosafety or biocontainment evidence gaps related to research and diagnostic activities of agricultural and veterinary importance.
Results:
The workshop findings aligned into the following categories: applied biorisk gap identified and required experiment proposed; research performed, but data not published or shared, mechanisms to share data required; risk assessment tools and process enhancements required; literature review and published summaries required; and additional survey, workshops, and “lessons learned” activities required. RAV3N members further analyzed the original dataset and report 25 prioritized applied biosafety research recommendations of importance to biocontainment facility and veterinary biorisk managers. These recommendations warrant consideration by agencies and policy makers considering funding research to ensure that biorisk management practices and regulations for biocontainment laboratories are evidence-based and built upon the best available science.
Introduction
Severe infectious disease threats affecting both animal and public health are emerging across the globe with increasing frequency. High-containment research facilities capable of working safely and securely with emerging disease pathogens in a range of potential host animal species play a critical role in maintaining preparedness against these disease threats. Traditionally, these facilities have relied on routinized practices based on a dated evidence base or, in many cases, develop and validate their own biosafety, biocontainment, and biosecurity practices and procedures, which potentially lead to variability and redundant efforts—factors which can impede critical research activities that need to be performed rapidly and safely. While the CDC’s Biosafety in Microbiological and Biomedical Laboratories (BMBL) 1 serves as an invaluable tool in guiding biosafety practice and risk assessment, there would be great value in applying its principles toward generating data-driven best practices for specialized applications, such as loose-housed large-animal biocontainment settings. A 2017 biosafety risk assessment performed by a private firm identified large gaps in biosafety knowledge, which could be addressed with primary research efforts to improve biosafety practices and reduce overall biorisk. 2 Similarly, an international working group conducted a comprehensive literature search and identified gaps in areas of applied biosafety research critical in supporting the safety and sustainability of global research programs. 3 This same group identified critical knowledge gaps in biosafety and biosecurity related to a number of persistent agricultural threats such as Foot-and-Mouth Disease virus (FMDV), 4 along with zoonotic agents, including SARS-CoV-2 virus, 5 Crimean-Congo Hemorrhagic Fever virus, 6 and zoonotic Avian Influenza. 7 Nevertheless, the voices of biosafety professionals are often underrepresented in conversations to determine strategic and funding priorities for applied biorisk research. There are limited incentives for life science stakeholders and funding organizations to discuss, share, analyze, and learn from issues related to biosafety and biosecurity. 8 Domestically and globally, the biocontainment research community would greatly benefit from research recommendations and priorities identified by experts in “biorisk management” to improve the evidence base that underpins the operational application of biosafety and biosecurity best practices in biocontainment facilities and potentially other settings (e.g., field biosafety).
The Research Alliance for Veterinary Science and Biodefense BSL-3 Network (RAV3N) is a network of 19 university and federal U.S. ABSL-3Ag/BSL-3/BSL-4 large-animal and laboratory agricultural research facilities that fosters collaboration and knowledge sharing among its members.
9
RAV3N is headquartered and managed at the Texas A&M University Global Health Research Complex (GHRC) and supported by the U.S. Department of Agriculture (USDA) agencies,
One of RAV3N’s strategic initiatives is to exchange operational knowledge and collective best practices as related to operations and biorisk management in large-animal biocontainment research facilities. In alignment with this mission, RAV3N organized an “Applied Biorisk and Biosafety Gap Assessment Workshop” on February 9–10, 2023, in Baltimore, Maryland, with co-sponsorship from the U.S. Department of Agriculture and the U.S. Department of State. RAV3N partnered with Gryphon Scientific and the American Biological Safety Association - International (ABSA International) to organize, plan, and deliver the workshop. This workshop was scheduled to immediately follow the USDA–ARS 7th International Biosafety and Biocontainment Hybrid Symposium: “Biorisk Management in Modern Agriculture” to capitalize on the immense global expertise already convened, with domestic and international participants representing government, academia, and the private sector.
A steering committee was established with representation from all partnering organizations, including the USDA, U.S. Department of State, RAV3N secretariat (Texas A&M University), Gryphon Scientific, and ABSA International. The committee confirmed terms of reference and held several meetings to plan the workshop, with a focus on maximizing productivity, given the short duration of the workshop (one and a half days). The purpose of the workshop was to identify and prioritize data-driven opportunities for improving biorisk management, with a particular emphasis on high-consequence veterinary and agricultural pathogens. These expert recommendations could then be used to guide resource allocation to ensure that biorisk management practices, guidance, and regulations for containment laboratories are based on the best available science.
Workshop Format
The steering committee identified broad areas of importance, which were designated as topics for breakout sessions. Due to the breadth of topics to be covered and in recognition of the diversity of expertise and interest across the participants, the agenda was designed such that breakout sessions were held simultaneously and participants were given their choice of one of two breakout sessions to attend based on their personal interests and expertise. Each of the three workshop sessions was assigned an overarching theme, which was intended to guide discussion touchpoints across all breakout topics. These overarching themes were as follows:
Enabling evidence-based development of disinfection and inactivation methods. Enabling evidence-based biocontainment practices, operational procedures, and facility design options. Enabling evidence-based development of safe work practices and procedures.
Subject matter experts from within the steering committee led each breakout group, and scribes captured the transcripts of the small group discussions. A “Session Guide for Facilitators” was developed to assist group leaders with guiding discussions toward achieving targeted outcomes and to ensure continuity across the multiple sessions.
Participants were instructed to not only develop a list of applied biosafety gaps related to each particular session topic but also identify the types of data needed to address the gap and specific experiments to provide that data, if known. Within each breakout session, small group discussions were first held with participants seated around each table, and then these small groups each shared their findings with the other small groups within each topical breakout. This approach ensured the opportunity for all participants to share their perspectives. Once the small topical groups reached a consensus, the two simultaneous breakout sessions reconvened and a high-level breakout recap was held along with discussion with the entire group. As such, participants were able to select topics of interest to dig deeper into, as well as hear and discuss the findings of all other topical groups.
Workshop Outcomes—Development of RAV3N Task Group
The inclusion of worldwide subject matter experts convened for the preceding USDA–ARS biosafety symposium allowed broad discussions inclusive of domestic U.S. and international perspectives from 40 invited participants representing government, academia, and the private sector (Figure 1, group photo of participants; Figure 2, affiliations of workshop participants). The invited participants were typically biorisk management program directors or biosafety officers representing a national agricultural research, animal health, public health, or regulatory mission-oriented governmental agency; a veterinary health and animal production service provider; or an animal pharmaceutical company. Facilitators led workshop participants through 12 breakout discussion sessions encompassing the following themes: development of disinfection and inactivation methods; biocontainment practices, operational procedures, and facility design options; and development of safe work practices and procedures. Data were compiled into a singular summary database for each breakout session topic to enable review, sorting, and analysis. The data yielded findings that could be summarized into the following categories: (1) applied biorisk gap identified, missing data outlined, and detailed experiment proposed; (2) research has been performed, but data not published, not shared, incomplete, and mechanisms to share data required; (3) risk assessment tools and process enhancements required; (4) literature review and published summaries required; and (5) additional survey, workshops, and “lessons learned” activities required.

Group photo of participants in the RAV3N Applied Biorisk and Biosafety Gap Assessment Workshop, held on February 9–10, 2023 in Baltimore, Maryland. RAV3N, Research Alliance for Veterinary Science and Biodefense BSL-3 Network.
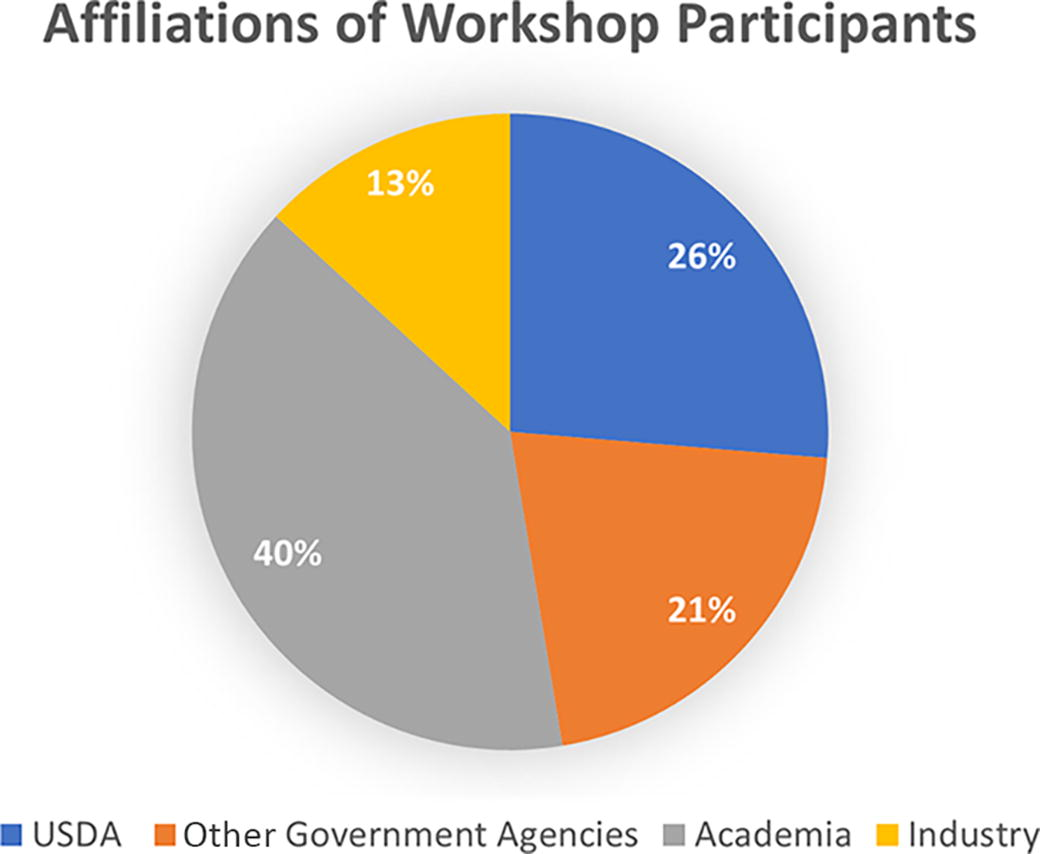
Professional affiliations of participants in RAV3N’s “Applied Biorisk and Biosafety Gap Assessment Workshop” held February 9–10, 2023 in Baltimore, MD. Participants represented 25 distinct institutions from the United States, Brazil, Nigeria, Philippines, Tunisia, India, and Kenya. RAV3N, Research Alliance for Veterinary Science and Biodefense BSL-3 Network. There were 32 participants from the U.S. and 1 from each other participating country.
Workshop partners and facilitators reviewed meeting notes captured by scribes and feedback from participants to complete the summary data file so that each separate partner could plan and pursue their respective next follow-up activities. Since RAV3N’s membership and scope of interest are primarily focused on activities occurring in the United States and Canada, their initial follow-up activity was to brief the entire RAV3N membership on the workshop discussions and present its summary findings. The
Following their workshop briefing, RAV3N members expressed strong interest in expanding upon the initial workshop findings to more specifically analyze the unique biosafety gaps and challenges of importance to agricultural and veterinary research and diagnostic activities. RAV3N members established an “Applied Biorisk/Biosafety Gap Assessment Task Group” (the “task group”), specifically charged with analyzing and compiling a more explicit list of applied biosafety gap areas, data requirements, and proposed experiments, which could potentially be performed across the network to address gaps associated with research involving agricultural, livestock, and wildlife animal models, zoonotic veterinary pathogens, and emerging diseases in BSL-3/ABSL-3Ag high-containment laboratory and animal holding facility environments. A secondary charge was to identify potential opportunities for compiling and sharing existing “applied biorisk management” research data among network members. The task group consisted of 11 members, including biosafety professionals, research leaders, and veterinarians representing U.S. academic and federal institutions.
The task group reviewed and analyzed the original workshop data and then added their own contributions to the combined dataset. The task group ultimately organized the combined dataset into the following five applied biosafety research domains for developing their subsequent gap analyses and applied research recommendations: (1) Mechanical and Facilities; (2) Workflow Analysis; (3) Animal Housing, Handling, and Contamination; (4) Human Factors; and (5) Validation of Inactivation, Disinfection, and Surrogate Organisms (Figure 3 and Table 1). The task group recognized that many of these domains likely overlap or interrelate with others, as depicted in Figure 3. For example, the influences related to efficacy of training or potential impacts of fatigue that would align under “Human Factors” would also likely impact each of the other four domains in some manner. The interrelatedness and concurrent interplay between elements of these five research domains also reflect the operational reality of maintaining safe and secure laboratories and animal facilities involved in performing BSL-3 and ABSL-3Ag level zoonotic and transboundary veterinary disease diagnostic testing and research.
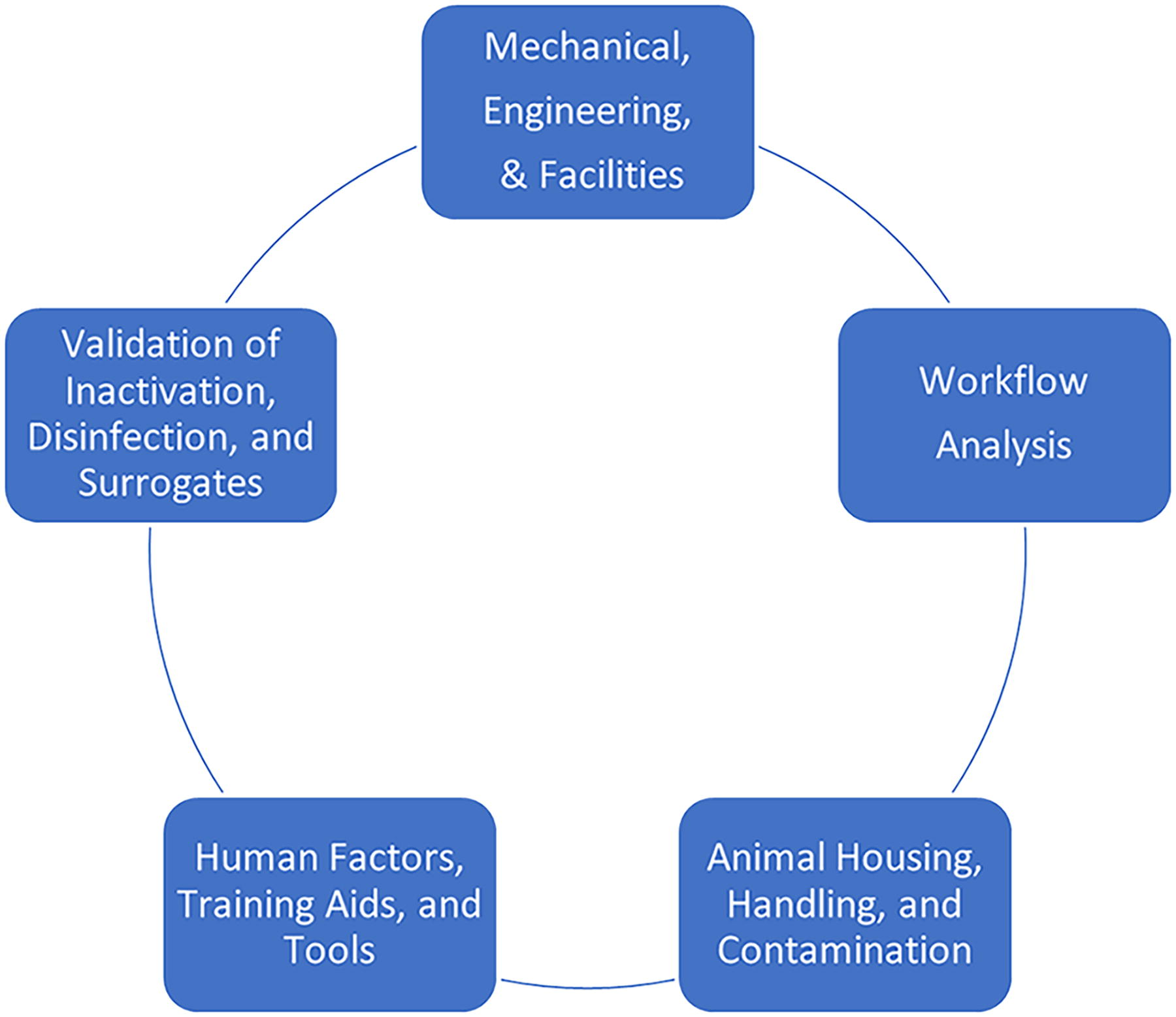
Interrelatedness of applied biosafety research domains identified for detailed gap analysis and specifying priority research recommendations.
RAV3N Task group summary of knowledge gaps and top five recommended research priorities for the applied biosafety research domains identified during the applied biorisk and biosafety gap assessment workshop
BSC, biological safety cabinet; EDS, effluent decontamination system; BSL-3, Biosafety Level 3; ABSL-3Ag, Animal Biosafety Level 3-Agriculture; ABSL-3, Animal Biosafety Level 3; BSL-4, Biosafety Level 4.
The task group identified and aligned two to three high-level “knowledge gaps” related to each of the five applied biosafety research domains and then identified their top five priority recommendations for applied biosafety research or the specific type of data required to address each of the respective knowledge gaps (Table 1). Group members were instructed to consider and develop their priority applied biosafety research recommendations upon the needs of the BSL-3/ABSL-3Ag biocontainment facility and research community overall, rather than at any specific institution. Each task group member independently ranked their own list of priority applied research recommendations and then the group reviewed the combined rankings to select the top five priorities for each research domain category. It is noteworthy that when the rankings from across the task group were combined, there was quick and unanimous consensus on the five “highest priority” gaps for each research domain (Table 1).
A persistent gap area and common theme that arose throughout the initial workshop and again during the task group discussions was the notion that a lot of applied biosafety research, especially regarding agent inactivation and facility disinfection, has been already performed, but it has either not been published in the public domain or shared widely within the professional biosafety community. The reasons cited for this varied. In many instances, agency or institutional confidentiality concerns, or internal security and nondisclosure policies were cited as a reason for not sharing or disseminating their internal applied biosafety research data. In other instances, participants indicated that available applied biosafety data were either at a preliminary pilot scale or otherwise not complete enough, or resources were not available for individuals to prepare and publish their results in peer-reviewed journals or external publications. Finally, it was often cited that any applied biosafety experiment and data were preliminary and only meant to enable performance of the “main experiments” that were planned once any potential biosafety issues were addressed; this was especially relevant regarding validation of inactivation protocols. The workshop participants suggested that this is an important issue that the broader professional biosafety and research communities need to consider and develop strategies for addressing. In response to the COVID-19 pandemic, a number of articles detailing various inactivation methods for the SARS-CoV-2 virus have been published, thus demonstrating a place and a utility for this type of publication.10–15 The task group also prioritized the need to establish a platform or other means of compiling and disseminating applied biosafety results and datasets to a broader user community (Table 1). This recommendation reinforces the 2022 Evidence-Based Laboratory Biorisk Management Science & Technology Roadmap report of the NSTC Health Security Threats Subcommittee on Laboratory Biosafety and Biosecurity recommendation #2 on Biorisk Management Data-sharing. 16
A noteworthy knowledge gap that spanned across several of the applied biosafety research domains and aligned with several of the recommended applied research priorities depicted in Table 1 dealt with the need to better understand and mitigate potential biorisks that arise in animal or laboratory spaces that concurrently support activities with multiple pathogens or animal species. This finding reflects workflows in many large high-containment facilities where it is common practice to concurrently perform multiple large-animal disease studies involving different pathogens and a diverse range of animal species across separate individual ABSL-3Ag large-animal holding rooms that are serviced by a common dirty corridor and supported by a multiuser BSL-3 enhanced laboratory. ABSL-3Ag rooms and BSL-3 laboratories in each facility are configured and operated slightly differently. Currently, it is challenging to perform detailed risk assessments and establish customized risk mitigation practices without having more specific and detailed pathogen dispersion data that correlate with actual workflow patterns and air flows within these containment spaces. For example, detailed data depicting zones within a BSL-3 laboratory or ABSL-3Ag large-animal room, where higher or lower levels of a pathogen may be found under typical operating parameters or workflows, could inform more detailed risk assessments and lead to refinement of workflow practices or operating conditions within the space. In the absence of more detailed data, some facilities restrict their BSL-3 laboratories to using a single pathogen at a time or impose potentially excessive risk mitigation requirements (e.g., PPE, quarantine periods) to assure that the greatest potential theoretical risks will be accounted for. These practices likely lead to increased operational costs and inefficient scheduling and use of costly BSL-3 laboratory and ABSL-3Ag animal spaces. Compiling more detailed pathogen dispersion, workflow pattern impact, and disinfection efficacy data from across multiple facilities representing a range of BSL-3 laboratory and ABSL-3Ag animal room configurations and operational parameters represents a crucial first step toward being able to improve risk assessment and mitigation, improve workflow planning, and enhance overall biosafety, biosecurity, and operational efficiencies in these complex biocontainment facilities.
It is not surprising that a high-priority research gap area identified by the task group was the need for more detailed data describing impacts of ventilation system design features (e.g., type and placement of diffusers) combined with operational considerations such as air changes per hour and positioning of animals within the room, and workflow pattern impacts on pathogen dispersion and risk localization within ABSL-3Ag large-animal holding rooms. The typical operational practice and recommendation to maintain between 10 and 15 air changes per hour (ACH) of single-pass conditioned air represent substantial facility design and construction costs that continue as a major ongoing operational cost (e.g., energy, equipment maintenance, building compliance monitoring and testing). More specific and detailed studies from a representative cross section of multiple ABSL-3Ag large-animal facilities working with a range of large-animal species could be immensely informative of how ventilation system design features, ACH, animal density and placement within the room, and workflow patterns potentially impact pathogen dispersion and bio-localization within these complex operational spaces. These data are needed to improve risk mitigation and employee safety, enhance animal comfort and welfare, and potentially decrease or modulate ACH based on actual risk data to optimize and potentially decrease operational costs.
In an ABSL-3Ag large-animal holding room, the room itself typically serves as the primary biocontainment barrier and needs to provide for animal welfare and comfort, support daily waste removal and transfer to an effluent decontamination system (EDS), and support safe and efficient workflow practices for animal caretakers and researchers involved in supporting animal experiments within a potentially infectious environment. The interaction between study animals, biological agent, and personnel in an ABSL-3Ag large-animal room during an infectious animal challenge study is a highly complex and specialized activity. Thus, it follows that one of the most cited, highly prioritized applied research gap areas identified during the initial workshop and by the RAV3N task group was the need for collecting data during routine ABSL-3Ag large-animal holding room operations across multiple facilities. Specific data gaps in this area include applied biorisk assessments and material performance data on performance and cleanability of animal room flooring surfaces, impacts of various types of bedding materials (e.g., sawdust, no bedding, straw, etc.) on animal comfort and pathogen dispersion within the room, and defining specific aspects of personnel workflows during room cleaning procedures that will assure proper and effective EDS waste treatment system performance (e.g., avoiding system breakdowns, clogs, potential releases).
Discussion and Recommendations
Recent reports have reinforced the importance of establishing and maintaining a diverse and up-to-date science and evidence-based foundation to underpin and inform application of biorisk management principles in research, diagnostic testing, and industrial settings around the world.2,8,11 Establishing and implementing a robust applied biosafety research agenda will be a critical requirement for assuring that evidence-based biorisk management practices and technologies remain current and relevant. The initial workshop and subsequent analysis by the RAV3N task group were focused on identifying and prioritizing data-driven opportunities for improving biorisk management, with a particular emphasis on high-consequence veterinary and agricultural pathogens. Our particular focus on veterinary and agricultural pathogens is similar to recent applied biosafety gap analyses that employed a “literature review”-based approach to identify specific biosafety data and knowledge gaps specific to each pathogenic agent.3–7 In contrast, the workshop- and task group-based methodologies employed in this study yielded findings that were perhaps less agent specific and more operationally oriented toward BSL-3 and ABSL-3Ag facilities and research involving a wide range of wildlife and large-animal model systems.
The initial workshop resulted in a comprehensive dataset rich with specific “real-life applicable” examples provided by a diverse panel of international subject matter experts in biorisk management. This targeted workshop format proved to be a demonstrable process that evoked and channeled the passions and knowledge of workshop participants to identify and better conceptualize oftentimes nebulous applied biosafety gaps. Participants interacted and shared ideas from multiple perspectives, while recognizing various technical, international, institutional, and political considerations. Given the diversity of backgrounds and experiences of participants, the continuously repeated themes of shared priorities, challenges, and opportunities were striking. These findings highlight the inherent value and importance of convening this type of workshop comprising diverse and passionate subject matter experts with a focused intent on identifying and deeply understanding gaps so they can be subsequently addressed. It is strongly recommended that biosafety professionals continue to sponsor and conduct future workshops of this nature to assess, refine, and enhance our understanding of critical applied biosafety research gaps and develop informed strategies and a research agenda to address them.
The workshop resulted in the compilation of an expert-generated list of priority areas within the field of agricultural biorisk management to be addressed with future applied research. These five well-considered and deeply analyzed high-level themes warrant additional analyses, workshops, and other directed activities to mature them. The priority list from the original workshop was further refined and categorized by a focused task group of biosafety and research professionals from across the RAV3N network. The “five recommended priorities for applied biosafety research” listed under each of the respective “applied biosafety research domains” depicted in Table 1 were deemed to be the most important research targets overall. This “recommended priorities list” represents the field’s greatest needs, as determined by stakeholders within the U.S. veterinary and agricultural biosafety community and may be used to guide development of a national research agenda and allocation of funding for applied biosafety research. While the RAV3N network is focused on the United States, the ongoing related work of Gryphon Scientific demonstrates that these issues are indeed global in scope and align with the NSTC recommendation to establish a globally distributed applied biosafety research agenda. 16
A lack of consistent funding dedicated to applied biosafety research combined with a perception that some of this research has already been done, but not published, may be obstacles to establishing and implementing a comprehensive national applied biosafety research agenda. The fundamental gaps identified herein present an important opportunity for funding agencies, policy makers, and the biosafety profession itself to take stock and consider allocating the funding required to solicit research proposals to address these high-priority topics. Doing so would be an important next step toward achieving an overall goal of improving the safety, security, and efficiency of biological research through improving and standardizing practices and guidance related to biorisk management.
Footnotes
Acknowledgments
The authors gratefully acknowledge the members of the workshop steering committee, which includes representation from USDA-ARS, USDA-APHIS, U.S. Department of State, ABSA International, and Gryphon Scientific, as well as the volunteer workshop scribes from Georgetown University (Washington, DC). The authors appreciate the workshop participants and members of the RAV3N task group for contributing their knowledge and expertise to this project.
Authors’ Contributions
K.A.Z.: Conceptualization (lead), data curation (equal), and writing—original draft (equal). C.L.H.: Project administration (lead), data curation (equal), and writing—original draft (equal).
Authors’ Disclosure Statement
The authors declare that they have no competing interests.
Funding Information
This work was supported by USDA National Bio and Agro-defense Facility (NBAF) Cooperative Agreement No. 58-3022-2-011 and U.S. Department of State Cooperative Agreement No. SAQMIP23CA0004.
