Abstract
Introduction:
High-containment biological laboratories (HCBLs) are specialized facilities designed for handling high-risk biological agents. The lack of data regarding the quantity, condition, and distribution of these laboratories in Brazil poses challenges for governmental strategic decisions.
Objective:
This research sought to offer a comprehensive view of the present condition of Brazilian HCBLs, highlighting their quantity, geographical distribution, function, operational range, and commissioning and certification processes.
Method:
A questionnaire was designed to collect data on the construction and operation of Brazilian HCBLs and was disseminated to facilities nationwide, identifying themselves as HCBLs.
Results:
In this survey, 66 HCBLs were identified across Brazil, with 32 participating in the study. A majority of the laboratories were associated with public universities, predominantly in the Southeast region. The pathogens most frequently handled were SARS-CoV-2 and Mycobacterium tuberculosis. Among the responding laboratories, ∼85% were engaged in research activities, <50% reported being part of a national network, a mere 15.6% were connected to international laboratory networks, and ∼50% reported obtaining certification before operational launch.
Conclusion:
Establishing a national regulatory framework in Brazil for the design, construction, commissioning, and certification of HCBLs is crucial to standardize and harmonize procedures across the country. Furthermore, implementing a national policy for laboratory biosafety and biosecurity, alongside establishing a “National HCBLs Network”, is essential to foster multidisciplinary and collaborative efforts, thereby optimizing resource allocation. Such enhancements in biosafety and biosecurity protocols will significantly benefit both animal and public health domestically and contribute positively to global health outcomes.
Introduction
High-containment biological laboratories (HCBLs) serve critical functions in diagnosing, researching, and producing materials that involve hazardous biological agents.1–2 Although four biosafety levels (BSLs) have been traditionally recognized, with BSL-3 and BSL-4 providing high- and maximum-containment, the World Health Organization (WHO) and the World Organization for Animal Health (WOAH) have moved away from this classification system. 2 The variability in definitions and criteria across countries suggests that comparing laboratories within the same BSL may not always be valid. 3
The global count of HCBLs remains indeterminate, yet it is clear their numbers have surged significantly since 2011. 4 Estimates suggest that there are over 1,300 HCBLs in the United States 5 and between 16 and 44 in India. 6 China’s goal is to establish at least one HCBL in each of its 22 provinces by 2025. 7 In addition, United Kingdom, Canada, Russia, South Africa, Australia, and Sweden are acknowledged for their considerable numbers of HCBLs. 4
The process of gathering data on the quantity and types of HCBLs in Latin America is fraught with difficulties because of the absence of standardized information. This inconsistency and lack of coherence in data collection hinder the ability to accurately understand the infrastructure for researching dangerous pathogens in the region. It implies that there may be laboratories under construction or in existence, whose details are either unavailable or challenging to access. 8
Currently, official data on the operation of HCBLs in Brazil are scarce, and there are limited legal mechanisms to oversee the risks these laboratories pose. 9 This deficiency largely stems from limitations in Brazil’s legal framework regarding laboratory biosafety and biosecurity, notably the lack of oversight mechanisms beyond self-regulation. 10 Furthermore, the limited scientific research on this topic exacerbates the information gap regarding the status of Brazilian HCBLs, including details about their commissioning and certification protocols. 11
Although Brazilian regulations do not establish commissioning criteria, it is recommended according to various international guidelines. Commissioning an HCBL necessitates thorough installation, inspection, performance testing, and confirmation of meeting predefined criteria. This ensures that structural and system components are correctly functioning and various systems operate effectively even in complex scenarios. 12
Certification is another crucial concept. It entails a review by an authorized entity, often through an audit, assessing the laboratory’s technical construction and operation, biosafety management documentation, and commissioning process. 13 Currently, Brazil does not have a dedicated organization to certify HCBLs. 11
Understanding the quantity, condition, and placement of HCBLs is vital for nations aiming to structure their laboratory infrastructure strategically and plan new facilities. The absence of a detailed HCBL inventory in many countries may lead to various challenges, including resource distribution, identification of critical needs, prevention of overlapping initiatives, and promotion of collaboration among laboratories. 4
Owing to the current lack of data and the acknowledged value of this information, this study aims to offer a snapshot of Brazilian HCBLs, concentrating on their approximate count, distribution, function, range, and the procedures for their setup and certification.
Improving our grasp of laboratory biosafety and biosecurity in Brazil is a crucial component that will aid in supporting strategic decisions by the Brazilian government and other countries while also elucidating Brazil’s significant regional and global role. Brazil’s extensive land and large population make it a focal point of influence and an economic leader, fueled by its robust agricultural sector.
Methods
To identify Brazilian HCBLs, this study used various methods, including literature reviews, public announcements for institutional construction and remodeling projects, internet searches, and personal communications. Given the absence of an official certification process for verifying the biosafety and biosecurity status of laboratories in Brazil, all entities self-identifying as having an HCBL were included in the study.
A detailed questionnaire was crafted to collect data on the design, construction, and operations of laboratories. The questions were based on key international guidelines, as national regulations are outdated. These guidelines include the WHO Biosafety Manual, 1 the Biosafety in Microbiological and Biomedical Laboratories Manual, 2 and the ISO 35001:2019 standard. 14 The questionnaire comprised 382 questions, mostly multiple choice, with 38 specifically on scope, design, and construction. The questionnaire was distributed to 92 identified HCBLs nationwide. Responses were gathered between November 2021 and March 2023.
Participants who consented were given a Free Consent Agreement Form, underscoring the authors’ dedication to safeguarding the confidentiality of the information shared. The survey methodology and the Free Consent Agreement Form received approval from the Ethics Committee for Research with Human Beings at the Federal University of Viçosa.
Descriptive statistical methods, including percentages and frequencies, were used to evaluate categorical data. For categories with low frequencies (n ≤ 5), the Fisher’s exact test was applied to assess relationships and correlations between variables in the statistical analysis. 15
Results
Respondent Profiles
Approximately half of the respondents to the questionnaire (15/32, 46.9%) identified as professors or researchers. Regarding their roles within HCBLs, an equal proportion serves as laboratory heads (16/32, 50.0%), with biosafety officers or their equivalents (7/32, 21.9%) and managers (6/32, 18.8%) following.
Identification of Operational HCBLs in Brazil
In this study, 92 laboratories 1 across 81 institutions were identified. Upon further examination and direct contact when possible, it was found that 17 facilities were no longer operating as HCBLs but had been downgraded to BSL-2 laboratories. Furthermore, nine laboratories under the Brazilian Hospital Services Company (EBSERH) were excluded because of unconfirmed status. 2 Therefore, the total number of HCBLs considered in this study was 66, belonging to 54 institutions.
The classification of these laboratories largely relied on self-reporting because of the lack of a national certification system, except for two that received international evaluation from a Food and Agriculture Organization’s reference center. Of the 66 self-declared HCBLs, 32 laboratories (48.5%) from 31 institutions (57.4%) participated in the questionnaire. This study did not aim to verify their BSLs.
Affiliation
The majority of HCBLs were linked to universities, followed by public health laboratories linked to the Ministry of Health or the State Health Secretariat (Table 1). In addition, more than 86% of the laboratories are supported with public funding (Table 1). The Oswaldo Cruz Foundation (Fundação Oswaldo Cruz) is the leading institution in terms of the number of HCBLs, hosting nine facilities.
Affiliation of self-reported HCBLs in Brazil
Geographical Distribution
The geographical analysis of HCBLs across Brazil indicated a significant clustering in the Southeast region, with 45 out of 66 facilities (68.2%) located there. Specific areas with a high density of laboratories included São Paulo, Rio de Janeiro, and Minas Gerais (Figure 1). These states host 34.9%, 18.2%, and 13.6% of the country’s HCBLs, respectively.
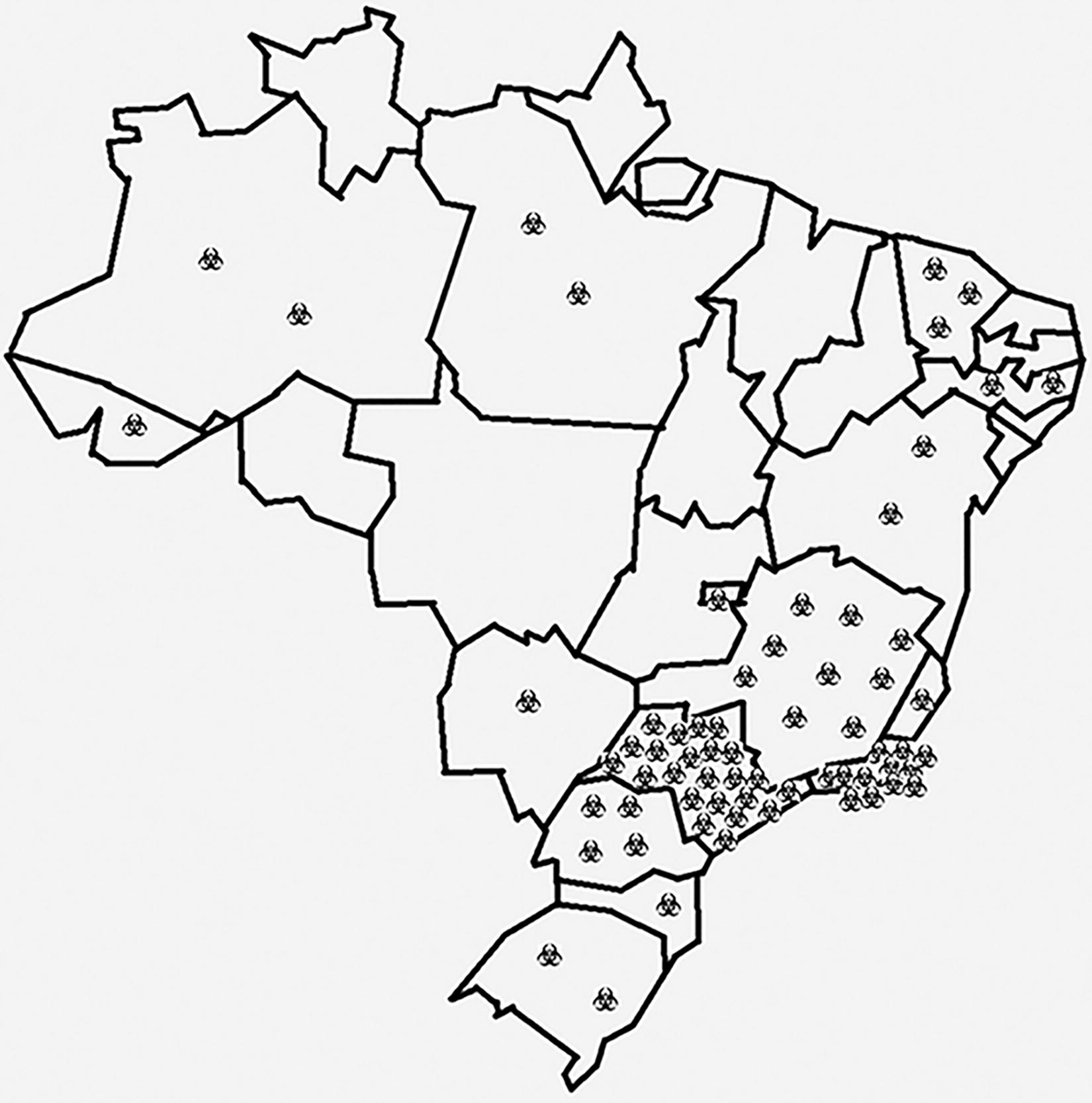
Geographical distribution of self-reported HCBLs in Brazil. Each symbol denotes an HCBL. HCBL, high-containment biological laboratory.
Scope
All laboratories, except one, handle or intend to handle pathogens that have an airborne route of transmission. Figure 2 lists the most frequently manipulated pathogens in the laboratories that participated in the survey. SARS-CoV-2 and Mycobacterium tuberculosis were most often mentioned. The following pathogens appeared only once: adenovirus, African swine fever virus, Bacillus anthracis, Bluetongue virus, Brucella spp., Burkholderia pseudomallei, classical swine fever virus, Clostridium botulinum, Coccidioides immitis, Cryptococcus sp., hantavirus, hepatitis virus, infectious laryngotracheitis virus, Leishmania sp., lentivirus, M. avium, M. leprae, Mayaro virus, Newcastle disease virus, Plasmodium sp., retrovirus, Rickettsia sp., Trypanosoma cruzi, vesicular stomatitis virus, and Yersinia pestis.
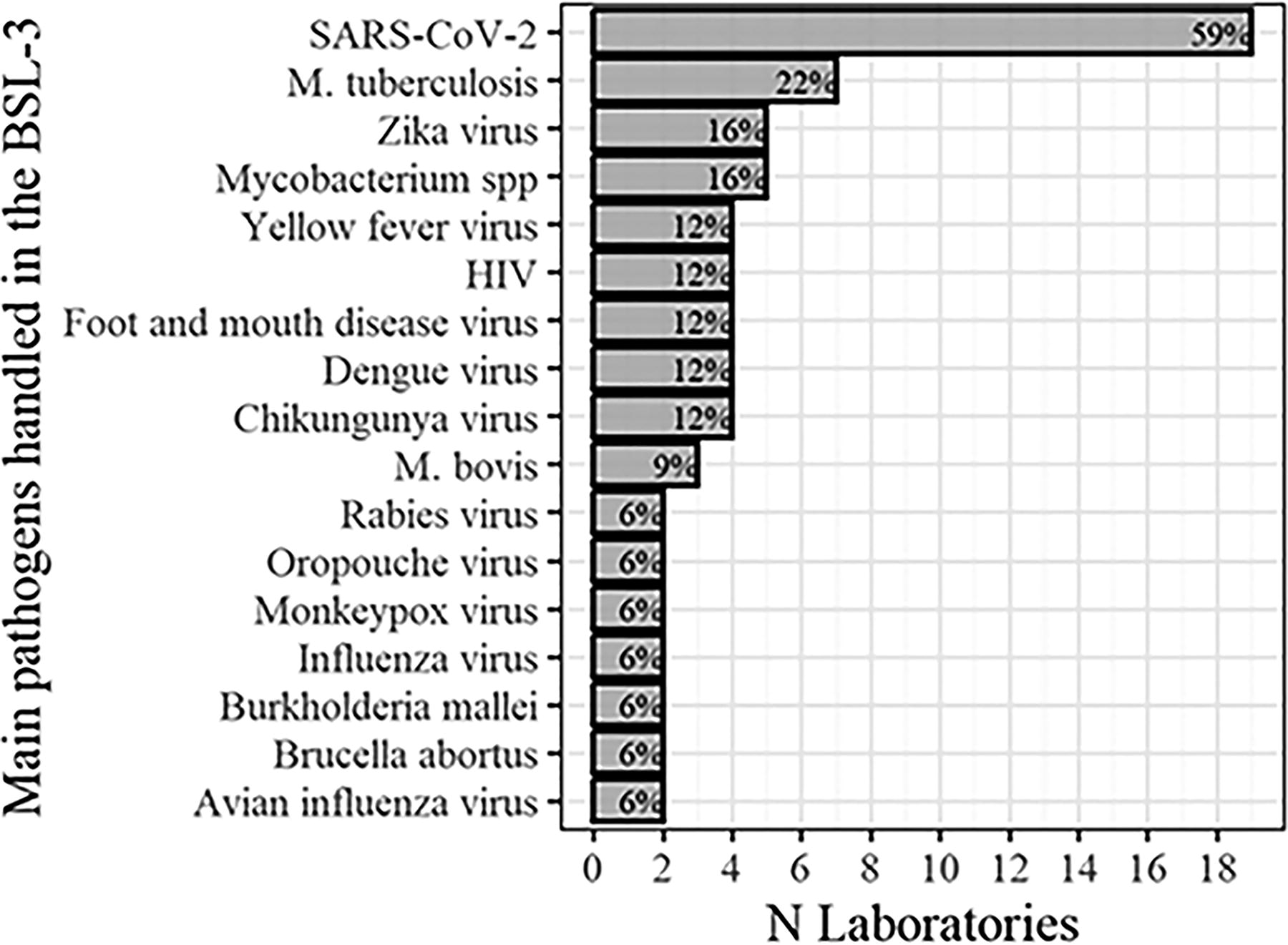
List of most frequent pathogens handled in the 32 laboratories participating in the survey.
The majority of laboratories surveyed (56.3%) serve multiple purposes, with a significant portion (84.4%) conducting research as a primary function. Over half (56.3%) are service providers, about a third (31.3%) participate in educational activities, and a smaller group (21.9%) is involved in producing materials such as vaccines and reference materials. Notably, a quarter of these HCBLs are primarily focused on research, whereas nearly 60% are engaged in at least two different types of activities, as shown in Figure 3.
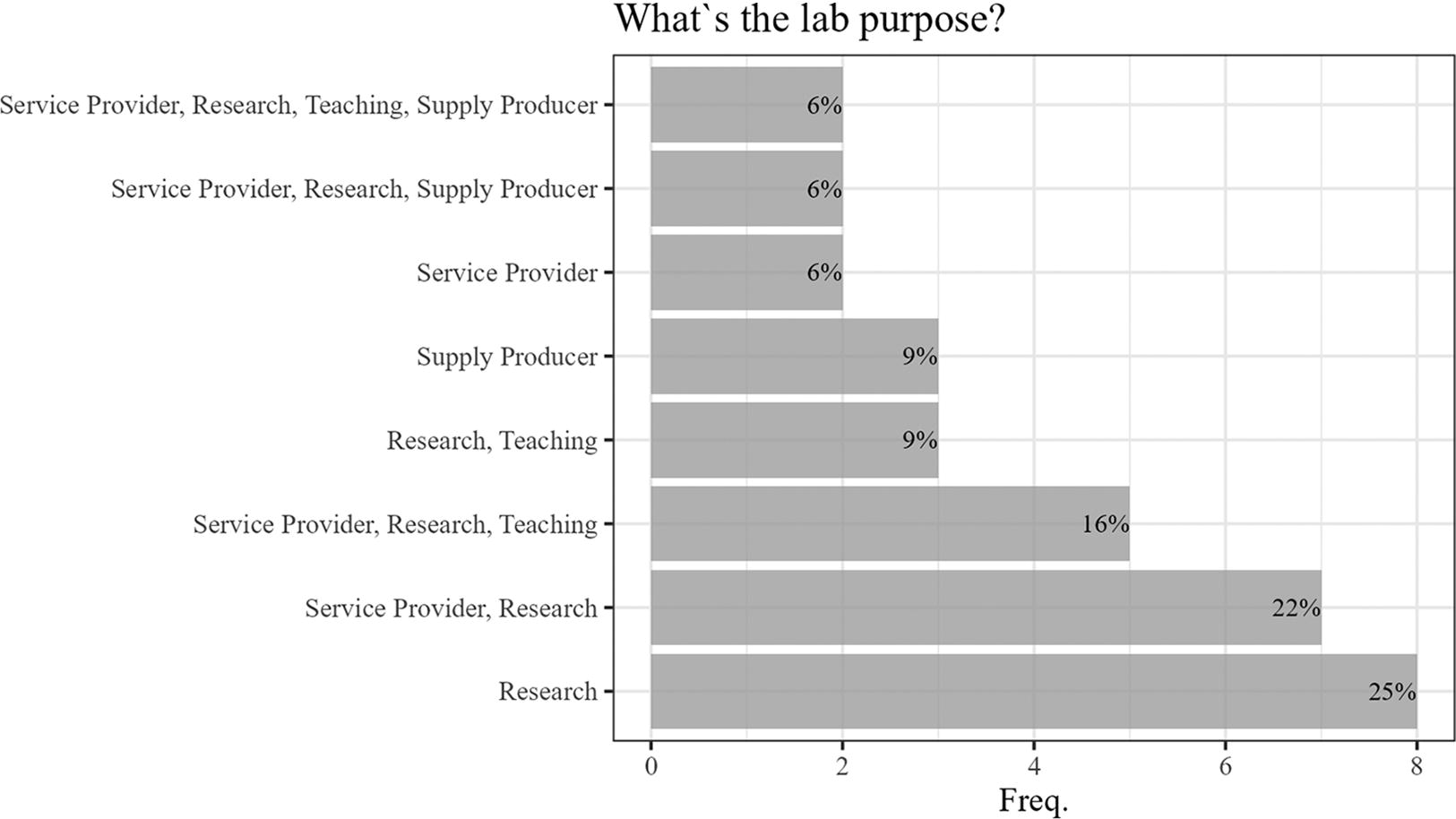
Purpose of the 32 HCBLs participating in the survey.
Networking
Less than half of the laboratories (40.6%) are part of some national network, with the Public Health National Laboratories Network being the most frequently mentioned. Notably, all HCBLs affiliated with the Ministry of Agriculture and Livestock (MAPA—Ministério da Agricultura e Pecuária) and the majority linked to the Ministry of Health (MS—Ministério da Saúde) and Health State Secretariats are part of national networks, in contrast to university HCBLs, which show no participation. This disparity indicates a significant difference in network participation based on HCBL affiliation (Fisher’s exact test <0.001).
A smaller fraction (15.6%) of HCBLs are involved with international networks, including WOAH, Pan American Health Organization, Pasteur Institute International Network, the Arbovirus Diagnosis Laboratory Network of the Americas, Entomological Virology Laboratories Network of the Americas, South American Laboratories Network for Avian Influenza and Newcastle Disease, and GABRIEL Network from the Mérieux Foundation.
Commissioning and Certification
The commissioning rate of HCBLs (60%) marginally surpasses the certification rate before operation (51.7%). No HCBL received certification without prior commissioning.
In the absence of national regulations regarding certification, significant questions arise concerning the validity and authority of these certifications. The MS and MAPA have committees empowered, according to their internal regulations, to assess HCBLs under their authority and endorse their operations. Despite the absence of specific guidance with defined requirements, this process can still be regarded as a form of certification.
Among the 15 laboratories that claimed certification, 3 did not disclose the responsible entity for the certification. Certification was predominantly conducted by official entities, notably the National Committee of MAPA and the Brazilian National Biosafety Technical Commission (CTNBio—Comissão Técnica Nacional de Biossegurança). Some laboratories indicated certification by local Institutional Biosafety Committees, constructors, or third-party companies. Nearly half of the laboratories (46.7%) renew their certification, with annual renewal being the most common (57.1%).
In commissioning, the most cited systems were biosafety cabinets (BSCs), heating, ventilation, and air conditioning (HVAC) system, autoclaves, and automation, as illustrated in Figure 4. Similarly, BSCs, autoclaves, and HVAC were the most commonly mentioned systems in periodic certifications, as depicted in Figure 5.
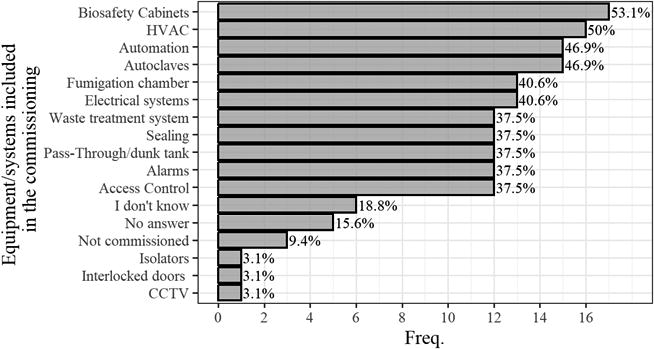
Proportion of equipment/systems contemplated in the commissioning process of the 32 HCBLs participating in the survey.
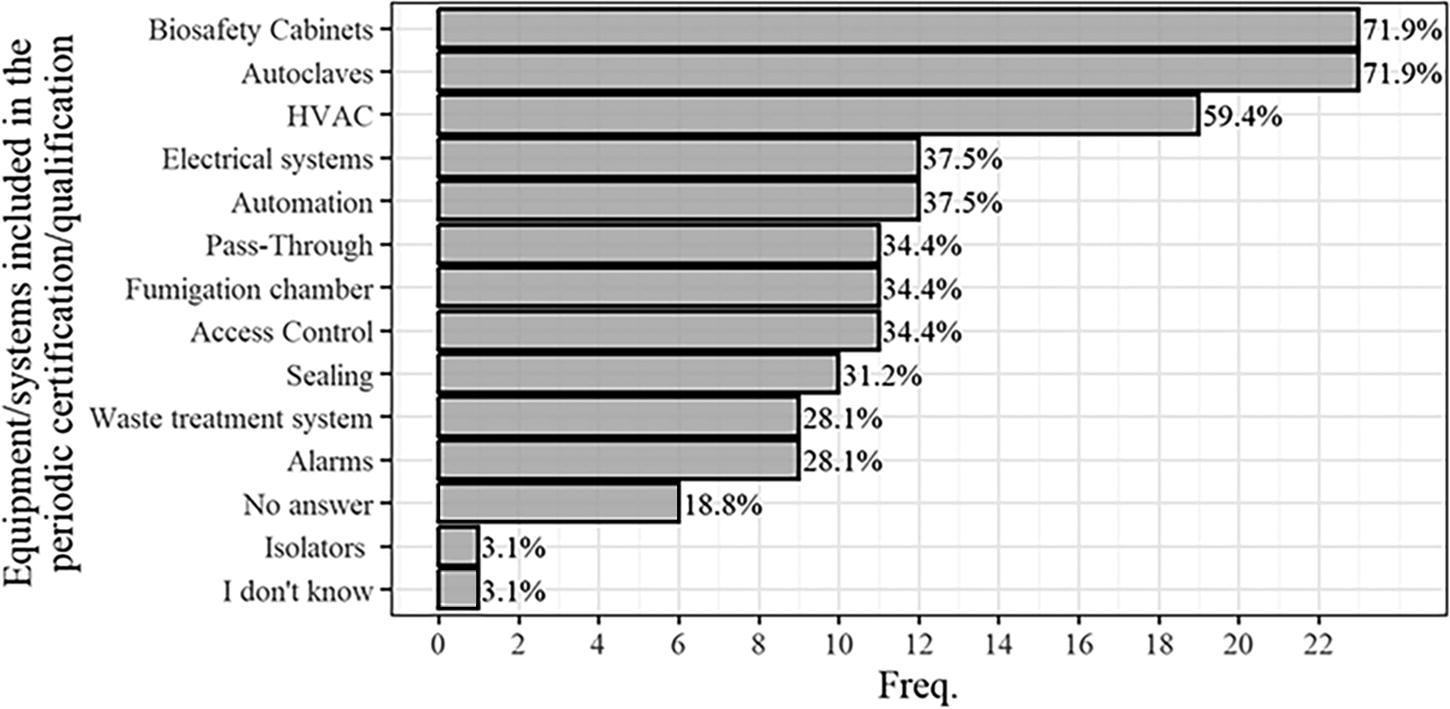
Proportion of equipment/systems contemplated in the periodic certification process of the 32 HCBLs participating in the survey.
Discussion
Respondent Profiles
The management of biological risks in Brazilian HCBLs presents significant concerns. A majority of these laboratories lack a dedicated biosafety officer or equivalent role to oversee biorisk management, a requirement according to ISO 35001 standards. The limited instances where such positions exist are a result of individual laboratory initiatives rather than a mandated regulatory requirement, which currently extends only to laboratories involved with GMOs.
Identification of Operational HBCLs in Brazil
Identifying the exact count of operational HCBLs in Brazil was challenging because of a lack of official data, self-declared BSLs, nonstandardized definitions of HCBLs, and difficulties in quantifying HCBLs within the same institution, especially when multiple laboratories share the same building.
Concerning the difficulty in counting laboratories, a U.S. Government Accountability Office report highlighted the issue of defining a “laboratory” in HCBL contexts. This is particularly challenging when containment facilities include independent rooms, leading to discrepancies. Some count each room as a separate laboratory, whereas others consider the entire complex as one unit. 16
The absence of official data on HCBL operations in Brazil presents a significant challenge. Data from facilities under governmental bodies like MS and MAPA are reliable but not public, whereas information on EBSERH, private, and university laboratories’ status is difficult to obtain.
Although the precise count of operational HCBLs in Brazil remains uncertain, this study identifies ∼66 self-reported facilities, significantly exceeding previous estimates by 2.1–4.4 times.4,17,18 A recent survey reported 47 HCBLs, 9 yet 12 of these were not functional as HCBLs at the time of this research, with no indication of imminent activation.
These data reveal fluctuations in the operational status of HCBLs across Brazil, attributed to the development of new sites and the closure of existing ones. Reports from staff members highlight that the primary cause for ceasing operations as HCBLs is the lack of proper maintenance, often because of financial constraints.
When compared with the United States and United Kingdom, Brazil’s count of HCBLs is notably lower. However, this number is similar to Canada, which has 66 licensed high- and maximum-containment laboratories across the country, 19 and much higher than Argentina, which has less than 10 BSL-3 laboratories 4 and other Latin American countries like Colombia (n = 1), Paraguay (n = 1), Mexico (n = 18), Venezuela (n = 2, one in container), and Cuba (n = 1, in container) (Cláudio Mafra, personal communication).
Countries with a high prevalence of disease often have limited resources and capabilities for diagnosing and monitoring diseases caused by these agents. However, simply increasing the number of laboratories may not be the best solution. 20 When constructing a new facility, it is essential to ensure the seamless integration of new capabilities into the existing public health infrastructure of a country. Avoiding duplication of existing public health laboratory capabilities and national program efforts is crucial for ensuring sustainability. 21
The requirement for HCBLs to conduct fundamental research on SARS-CoV-2 in Brazil presented a significant challenge. Laboratories previously successful in generating critical knowledge during Zika, dengue, and chikungunya epidemics have encountered lengthy waiting times to access BSL-3 facilities, either within their own institutions or through collaborations. The concentration of BSL-3 laboratories in the Southeast region and the urgent need to enhance the network by establishing additional federally funded BSL-3 and notably, ABSL-3 laboratories within the Federal universities system, are emphasized to increase the capacity to respond to health emergencies. 22 It is important to emphasize the support provided by academic laboratories during a public health crisis, as demonstrated during the COVID-19 pandemic.
Although the concerns about the need for more HCBLs in Brazil are valid, it is crucial to first gain a clearer understanding of the existing landscape. Expanding the number of HCBLs without careful consideration could lead to inefficiencies, such as underutilization or misalignment with national, public, and animal health objectives. Moreover, unnecessary expansion could introduce risks and burden regulatory bodies, particularly MS and MAPA, with increased oversight responsibilities.
Experts at the 2019 workshop Developing Norms for Biological Laboratories in Low-Resource Settings, organized by the U.S. Academies of Sciences, Engineering, and Medicine, emphasized the importance of understanding the existing number, condition, and distribution of HCBLs. This knowledge is essential for countries planning to enhance their national laboratory infrastructure and strategic development of new facilities. Currently, many nations do not possess a detailed inventory of HCBLs. Creating such a comprehensive list could potentially enable better resource allocation, identification of needs, prevention of redundant efforts, and promotion of collaboration among laboratories. 4
The overarching goals of a national laboratory biosafety and biosecurity framework should be clearly defined within a National Biosafety and Biosecurity Policy. The WHO states that the national laboratory strategic plan translates the prerequisites for an ideal laboratory system formulated in the national laboratory policy into strategic objectives that must be undertaken to turn the policy statements into reality. Consequently, developing such a strategic plan is a critical and logical progression following the establishment of a national laboratory policy. 23
The national strategic plan for laboratory biosafety should outline the optimal quantity, variety (BSL-3, ABSL-3, ABSL-3Ag, BSL-4, ABSL-4, etc.), distribution, connectivity, and function of these facilities throughout the nation, alongside the budgetary requirements for their implementation, particularly for those falling under the Federal government’s authority.
Although this study primarily addresses facilities dedicated to human and animal pathogens, it is crucial to recognize that similar concerns extend to high-risk plant pathogens. Therefore, these also warrant inclusion in national policies and strategic plans.
Geographical Distribution
The concentration of laboratories in the country’s Southeast region can be attributed to the region’s demographics and numerous industries, universities, and reference laboratories. When discussing the national strategic plan, it is essential to consider this issue, and the health authorities should assess the necessity of establishing additional laboratories in other regions. This evaluation must consider the logistical challenges associated with sample transportation, particularly from remote areas, weighed against the availability of qualified personnel and maintenance services.
Five HCBLs are strategically located in the Amazon region, an area of critical importance because of its vast biodiversity and potential for discovering new pathogens. 24
Scope
The significant number of laboratories processing SARS-CoV-2 during the COVID-19 pandemic, which was declared a global health emergency by the WHO from January 2020 to May 2023, led to an increased use of HCBLs. Despite this, not all activities related to the virus necessarily required HCBLs. Following WHO guidelines, the MS mandated that diagnostic assays, including hematological and serological tests, tissue fixing, molecular biology, clinical pathology, processing of fixed or inactivated samples, electron microscopy, and sample inactivation, be conducted in BSL-2 laboratories because of their low-risk and nonproliferative nature. 25 However, many institutions initially opted to use their HCBLs for diagnostic purposes, particularly polymerase chain reaction assays, as a precautionary measure amid uncertainties about sample handling risks. It is important to emphasize the possibility that this decision was not always grounded in a comprehensive risk assessment.
These observations highlight the critical role of HCBLs in responding to emergencies and the value of adaptable facilities. Nonetheless, it is important to recognize that using HCBLs for low-risk activities may lead to inefficient use of resources.
Similar considerations apply to work with Mycobacterium spp., classified as risk group 3 25 and the second most commonly handled organism in Brazilian HCBLs. According to MS guidelines, 26 in line with WHO recommendations, 27 bacilloscopy assays that adhere to good microbiological practices pose a low aerosol generation risk and can be conducted on an open bench with proper ventilation. However, for procedures with a higher aerosol generation risk, such as sample preparation for culture and other proliferative activities, the use of a biosafety cabinet or a biocontainment laboratory—not necessarily a BSL-3 facility—is advised for handling cultures for sensitivity or genetic probe tests.
Arboviruses, including Zika, dengue, chikungunya, and yellow fever viruses, are prevalent in Brazilian HCBLs, reflecting their significance in the country. Despite being classified as risk-group 2 pathogens by the MS, Brazilian regulations stipulate added safety measures when handling the wild yellow fever virus with techniques prone to producing significant infectious material and aerosols. Specifically, such activities, especially those involving high viral loads or volumes, necessitate a BSL-3 facility for the yellow fever virus. Likewise, procedures involving the chikungunya virus that generate substantial infectious aerosols are recommended to be conducted in BSL-3 laboratories. 25
Further studies are necessary to evaluate whether activities conducted in Brazilian HCBLs, which involve the manipulation of arbovirus, SARS-CoV-2, and Mycobacterium spp., require BSL-3 laboratories or whether they could potentially be performed in lower biosafety containment levels. The same concern applies to other risk-group 2 pathogens handled in HCBLs, as identified in this survey, including Adenovirus, Cryptococcus sp., hepatitis virus, Leishmania sp., Mayaro virus, Plasmodium sp., retrovirus, Trypanosoma cruzi, and Oropouche virus.
Among animal pathogens, Foot and Mouth Disease virus (FMDv), M. bovis, Brucella abortus, avian influenza virus, and Burkholderia mallei are most frequently encountered, all linked to notifiable animal diseases. 28 Laboratories handling FMDv, often involved in vaccine production, may close or undergo remodeling as Brazil moves toward FMDv-free status without vaccination by 2025 or 2026. 29 This study did not find laboratories handling other notable risk-group 3 animal pathogens, such as equine encephalitis virus and Aujeszky’s disease virus.
Brazil’s lack of ABSL-3Ag laboratories for handling animals such as cattle, sheep, horses, goats, or pigs in high-containment environments is a significant gap, limiting in vivo research, vaccine development, diagnostic testing, and the production of crucial supplies and reference materials for high-risk animal diseases and zoonoses, such as classical swine fever, FMD, avian influenza, anthrax, and brucellosis. In addition, the absence of BSL-4 laboratories to manage risk-group 4 pathogens—including Ebola virus, Marburg virus, Hendra and Nipah Henipavirus, Crimean–Congo hemorrhagic fever virus, and the Brazilian mammarenavirus (Sabia virus)—underscores a critical vulnerability in addressing potentially emerging or reemerging diseases.
This study found no HCBL in Brazil working with arthropods. This absence does not necessarily indicate that such facilities do not exist in the country, as approximately half of the laboratories did not participate in the survey. Nevertheless, it is crucial for researchers, particularly in academic settings, to be cognizant of the necessity to handle certain animals, such as mosquitoes and ticks, within high-containment environments for studies involving high-risk pathogens like Rickettsia spp., yellow fever, dengue, chikungunya, and Zika virus.
The role of university laboratories, as revealed by this study, is pivotal, with nearly half of Brazil’s HCBLs situated in public universities. This aligns with a U.S. survey of 245 BSL-3 laboratories, where 52.3% were academic. 30 However, within the Federal Select Agent Program, this figure drops to 35.2%. 31 Out of 600 BSL-3 facilities in United Kingdom in 2016, an estimated 150 are in universities (25.0%). 32 In Canada, the percentage of academic laboratories licensed to work with risk-group 2, 3, or 4 pathogens and toxins is even lower, at 20.4% (201/916). 19
The Brazilian Federal government is urged to assess the collaborative dynamics and impact of university-based laboratories on national public and animal health agendas. This includes determining if these laboratories can address needs unmet by government laboratories and evaluating the suitability of the higher prevalence of HCBLs in Brazilian universities compared with countries like Canada and United Kingdom, especially given existing regulatory oversight challenges in Brazil. 10 The performance of public university laboratories during the COVID-19 pandemic showcased the potential of these facilities in bolstering national public health services, especially during a public health emergency.
Networking
Networks significantly bolster laboratory capabilities, particularly in laboratory biosafety and biosecurity contexts. Effective networking is essential for rapid responses and minimizing the impact of incidents. 33
Multidisciplinary and collaborative efforts are necessary at both global and local levels because of the challenges posed by increased antimicrobial resistance, the extreme variability of RNA viruses, and the role of domestic and migratory birds and wildlife in transmitting diseases. This complexity underscores the need for regional and local networks to use highly sensitive and specific methods for early pathogen detection. 34
Despite occasional efforts, particularly by the MS and MAPA, Brazil lacks a unified strategy to establish a National HCBL Network. This gap is notably detrimental to university laboratories, which stand to gain significantly from such a collaborative framework.
Commissioning and Certification
This study highlights a concerning finding regarding the relatively low proportion of HCBLs that are commissioned before launch despite the absence of commissioning criteria in Brazilian regulations. Notably, the commissioning process for some HCBLs seems to be partial, lacking inclusion of essential systems and equipment like access control and autoclaves.
The criteria used by respondents regarding commissioning and certification were not explored in this study, raising concerns about potential misunderstandings. Enhancing capacity-building training for managers, biosafety officers, and maintenance personnel is crucial for harmonizing understandings and spreading knowledge across Brazil. The Brazilian Biosafety and Biosecurity Society (Sociedade Brasileira de Biossegurança e Bioproteção) could significantly contribute here, particularly in offering training courses on biosafety and biosecurity risk assessment.
Establishing clear roles, responsibilities, and minimum qualifications for biosafety officers in Brazil is crucial. Presently, only one professional in the country has a valid certificate for biorisk assessment by the International Federation of Biosafety Associations, highlighting a cultural deficiency in recognizing the necessity of such qualifications across both public and private sectors.
Despite the absence of national regulations for certifying HCBLs in Brazil, over half of the surveyed laboratories claimed to have certification. This raises significant concerns regarding the legitimacy and credibility of these certifications in the absence of a regulatory framework. It is essential to identify the certifying entities and assess their legitimacy. For instance, some HCBLs mentioned being certified by national committees affiliated with the MS or MAPA. Although certification by such regulatory bodies might seem appropriate, there is currently no national standard defining the term “certification” and its criteria. In other instances, the certifying entity mentioned was wholly inappropriate, such as the constructor or self-assessment.
Furthermore, certifications from the National Technical Commission on Biosafety (CTNBio) and private entities should not be deemed sufficient. CTNBio primarily focuses on GMOs, and private certifications lack legal authority. Presently, Brazil lacks an authoritative body to certify HCBLs for their BSLs, underscoring the pressing need to strengthen the national regulatory framework.
Conclusions
Although the results presented offer a broad yet thorough insight into the present condition of Brazilian HCBLs, the lack of official data concerning the operation of HCBLs in Brazil persists as a significant concern.
The Brazilian government should consider carefully assessing the necessity and function of multifunctional BSL-4 and ABSL-3Ag laboratories in the country, taking into account the vulnerabilities associated with the absence of such facilities for public and animal health. However, before contemplating the expansion of HCBLs in Brazil, it is imperative to gain a better understanding of the current situation. Therefore, further studies are required to determine whether activities conducted in Brazilian HCBLs necessitate BSL-3 facilities or if they could be carried out at lower biosafety containment levels. For instance, conducting research and diagnostics for COVID-19 in BSL-3 laboratories solely for the purpose of justifying their existence or securing funding, rather than for genuine biosafety concerns, is unacceptable.
It is essential to promote the engagement of Brazilian HCBLs in both national and international networks. The formation of partnerships at both levels is crucial for the sustainability of HCBLs, an aspect that, unfortunately, appears to be neglected within the national context.
It is imperative to establish a national regulatory framework for the design, construction, commissioning, and certification of HCBLs. Drawing from the regulations implemented in other countries can offer valuable insights. This initiative would foster a basic level of standardization in construction, commissioning, and certification practices, thereby reducing the risk of misclassification of laboratory BSLs. Such a framework would facilitate the sharing of resources and promote a culture of safety and security at the national level. Ultimately, it would discourage the handling of high-risk pathogens in facilities that do not adhere to the requisite construction, commissioning, and certification standards.
There is an urgent need to establish a national policy for laboratory biosafety and biosecurity and to formulate a national strategic plan for HCBLs. This plan should encompass the optimal quantity, types, distribution, interconnections, research focus, and purpose of these facilities nationwide, ensuring their contribution to public and animal health while preventing redundancy and ensuring the efficient allocation of resources.
Strengthening biosafety and biosecurity in Brazilian laboratories through robust policies, a comprehensive regulatory framework, and standardizing biorisk management procedures, including defining roles and responsibilities of biosafety officers, promises significant benefits for both animal and public health within the country. In addition, these measures are poised to positively impact global health outcomes, given Brazil’s substantial contributions to feeding the world through its thriving agribusiness sector. Furthermore, Brazil’s strategic geographical location facilitates dynamic trade relationships with nations worldwide, further solidifying its significance on the international stage. As a key participant in forums such as BRICS (Brazil, Russia, India, and China) and Mercosur, Brazil asserts its leadership by shaping policies and fostering cooperation that transcends borders.
Footnotes
Acknowledgments
The authors would like to express their sincere appreciation to Prof. Elias Medeiros for his invaluable contribution to the compilation and analysis of the data in this research.
Authors’ Contributions
A.M.D.O. was primarily responsible for developing the survey, including gathering and assessing data as part of his doctoral program. He played a significant role in elaborating the article and ensuring the coherence and alignment of research findings with the study objectives. A.M.D.O. also contributed to the initial data analysis and participated actively in discussing the results, providing critical insights that shaped the final conclusions. C.M., as the advisor responsible for the doctoral program, oversaw all project steps with a keen eye on ensuring the research’s integrity and relevance. Beyond revising the article for intellectual content and clarity, C.M. was instrumental in structuring the questionnaire, conducting a thorough data analysis, and refining the article’s structure and presentation. His contributions extended to critical revisions of the draft, incorporating a deeper analytical perspective and offering strategic guidance that enhanced the article’s overall quality and impact. Both authors contributed significantly and substantively to the development and completion of this work. Their collaborative effort has been pivotal in advancing the research presented in this article, and both have approved the final version for submission.
Authors’ Disclosure Statement
No competing financial interests exist.
Funding Information
This research has been supported by the Project “Management and Governance in Biosafety,” an initiative of the Coordination of Superior Level Staff Improvement (CAPES) and the Ministry of Defense, Brazil (Grant number 88881.682224/2011-01).
