Abstract
Introduction:
The effective sterilization of isolators prior to placement of germ-free birds/eggs is crucial to ensuring that the environment is free of potential contaminants. With the use of formaldehyde for sterilization becoming less popular owing to its carcinogenicity, the need for an alternative agent with the same efficacy is essential in the preparation of isolators. Chlorine dioxide dry gas has previously been shown to be a highly effective sterilization method, providing a promising alternative for germ-free avian egg facilities.
Methods:
Polyvinyl chloride (PVC; n = 32) and stainless-steel production isolators (n = 7) were sterilized using approximately 4 h of exposure to chlorine dioxide (PVC isolator) and after approximately 6 h of exposure to chlorine dioxide (stainless-steel production isolator).
Results:
Each isolator type was sterilized effectively using chlorine dioxide. Samples collected for microbiological analysis from the isolators after sterilization confirmed that the isolators were sterilized and remained sterile for at least 3 weeks after sterilization.
Discussion:
The results of this study highlight the first use of chlorine dioxide dry gas for germ-free avian sterilization practices, augmenting on its use as a fogging agent seen in germ-free mice practices. As described in previous animal laboratory studies, values >1440 ppm-h cycle achieved in this study provided consistently adequate antimicrobial efficacy for the sterilization of germ-free egg facilities.
Conclusion:
Chlorine dioxide dry gas is a highly effective sterilization solution for germ-free avian egg facilities, providing long-lasting sterility to isolators without the safety concerns associated with other fumigants such as formaldehyde.
Introduction
Germ-free animals play a major role in researching the roles microorganisms (single or multiple) play in the functioning of the microbiome and immune system in humans and animals. 1 However, the establishment of colonies of germ-free animals is not without its technical challenges, with sterility being the primary objective that has to be met at all times. 2
Traditionally, the method of gaseous fumigation is used to achieve sterility within a contained environment such as a pharmaceutical isolator or other containment facility.3,4 Gaseous fumigation methods are used to sterilize or disinfect areas exposed to harmful organisms and constitute a risk to workers and the public if left uncontrolled.3,4 Gas laws support the assumption that the ideal fumigant should be gaseous at standard temperature and pressure to penetrate all surfaces and disperse uniformly across the fumigated area, which highlights formaldehyde consistency and efficacy as a fumigant.3,4 However, the utilization of formaldehyde is restricted owing to the carcinogenic nature of the agent.
Fumigation techniques have used formaldehyde as the preferred fumigant since the late 1880s.3,5 Simple formaldehyde vaporization involves heating a formalin solution with water in a temperature-controlled device.3,6 Formaldehyde is a colorless gas at room temperature. 3 However, safety concerns over its application and subsequent residues have led authorities to consider safer options.3,5–7
Gaseous chlorine dioxide (ClO2), vapor hydrogen peroxide (VHP), and peracetic acid (PAA) have been proposed and utilized as noncarcinogenic alternatives to formaldehyde for a variety of different applications. Historically, in the case of animal facilities, isolators have been sterilized prior to animal placement, using a multitude of approaches, many of which focused on the use of PAA 8 and formaldehyde. 9 Previously, germ-free animal facilities have utilized ClO2 methods 10 to achieve sterility as an alternative to formaldehyde. However, such work has been focused on germ-free mice (GFM), whereas no ClO2 method has been evaluated as a sterility method for avian germ-free egg facilities. The purpose of this study was to evaluate the use of gaseous ClO2 as a noncarcinogenic alternative to formaldehyde for germ-free egg facilities to assess whether the agent is suitable for avian germ-free egg applications. The utilization of other noncarcinogenic alternatives to formaldehyde such as VHP or PAA may also provide a satisfactory alternative, although the evaluation of such agents is outside the scope of this study.
Chlorine Dioxide Dry Gas
Chlorine dioxide dry gas (dClO2) has shown promise as a safe, noncarcinogenic alternative to formaldehyde while preserving the efficacy. 6 As ClO2 is neither mutagenic nor carcinogenic in humans, it offers significant safety advantages when compared with formaldehyde. 11 When evaluating alternate fumigation procedures in 2011, the UK Health and Safety Laboratory Executive (HSE) evaluated dClO2 in addition to other common fumigants such as formaldehyde, VHP, and Ozone. 3 The most efficient and effective fumigation agent, including formaldehyde, was dClO2. 3 Since the study, 3 dClO2 has been proposed as part of the UK HSE’s recommendation for fumigation procedures for high-containment facilities. 6 In addition to replacement of formaldehyde, recent studies have shown that dClO2 provides a novel gaseous alternative to ethylene oxide, another carcinogenic fumigant, for pinworm decontamination and medical device sterilization.12,13 In addition, owing to its antimicrobial efficacy and noncarcinogenic and gaseous nature, dClO2 is beneficial to fumigation processes owing to its speed and quality control. 14 The increased speed of dClO2 cycles is owing to its oxidation capacity being higher than that of other decontamination agents such as PAA, which only harbor two free electrons in comparison with the five free electrons harbored by dClO2. 14 In addition, owing to its green color, dClO2 cycles can be monitored in real time, meaning that concentrations are known before, during, and after a cycle, which provides additional safety aspects especially for isolator reentry. 14
However, ClO2 gas at over 10% can be explosive; therefore, the generation of a controlled concentration of ClO2 is essential to process safety. The utilization of the ClorDiSys process injects a consistent ClO2 concentration of 4% (as per the manufacturer’s recommendations); however, for large environments, other ClO2 generation methods may not be favorable, although such methods were not evaluated in this research and therefore are outside scope. A potential drawback of the method used was the requirement of pressurized 2% chlorine cylinders on site for generation of the ClO2 gas, providing safety risk associated with pressurized cylinders that requires consideration. ClO2 methods must accommodate for photodegradation of ClO2 in sunlight, which may provide complications for practices whereby sunlight exposure can occur.
Material Compatibility
Contrasting reports have been documented relating to the compatibility of dClO2 with various materials, especially whereby excessive relative humidity (RH) may apply as RH of 65%, essential to the dClO2 process, may exceed limits and provide risk to certain electrical equipment. 15
After repeated use of gaseous ClO2 over 5 years, neither did a Class III cabinet show any traces of corrosion on the stainless-steel interior nor did the stainless-steel inhalation exposure system kept inside the cabinet experience any ill effects. 16 These data and the ability of ClO2 to effectively and safely sterilize electrical/battery-operated equipment (e.g., incubators used for egg incubation and hatching) warrant its use in the sterilization of isolators that will be used to house germ-free poultry.
Historically, isolators have been sterilized prior to animal placement, using a multitude of approaches, many of which focused on the use of PAA 8 and formaldehyde. 9 In this article, we focus on the novel methodologies that we used to sterilize isolators used to house germ-free poultry for the production of germ-free eggs.
Methods
Isolator Types
Two types of isolators are used in the production of germ-free poultry, i.e., polyvinyl chloride (PVC) and stainless-steel isolators. The PVC isolator design is used for incubation and hatching of germ-free eggs and also for the purposes of transferring sterilized items into and out of isolators. The approximate dimensions of the PVC isolator are as follows: 1.6 m (l) × 0.84 m (w) × 1.0 m (h), giving a chamber volume of approximately 1.34 m3 (refer to Figure 1).
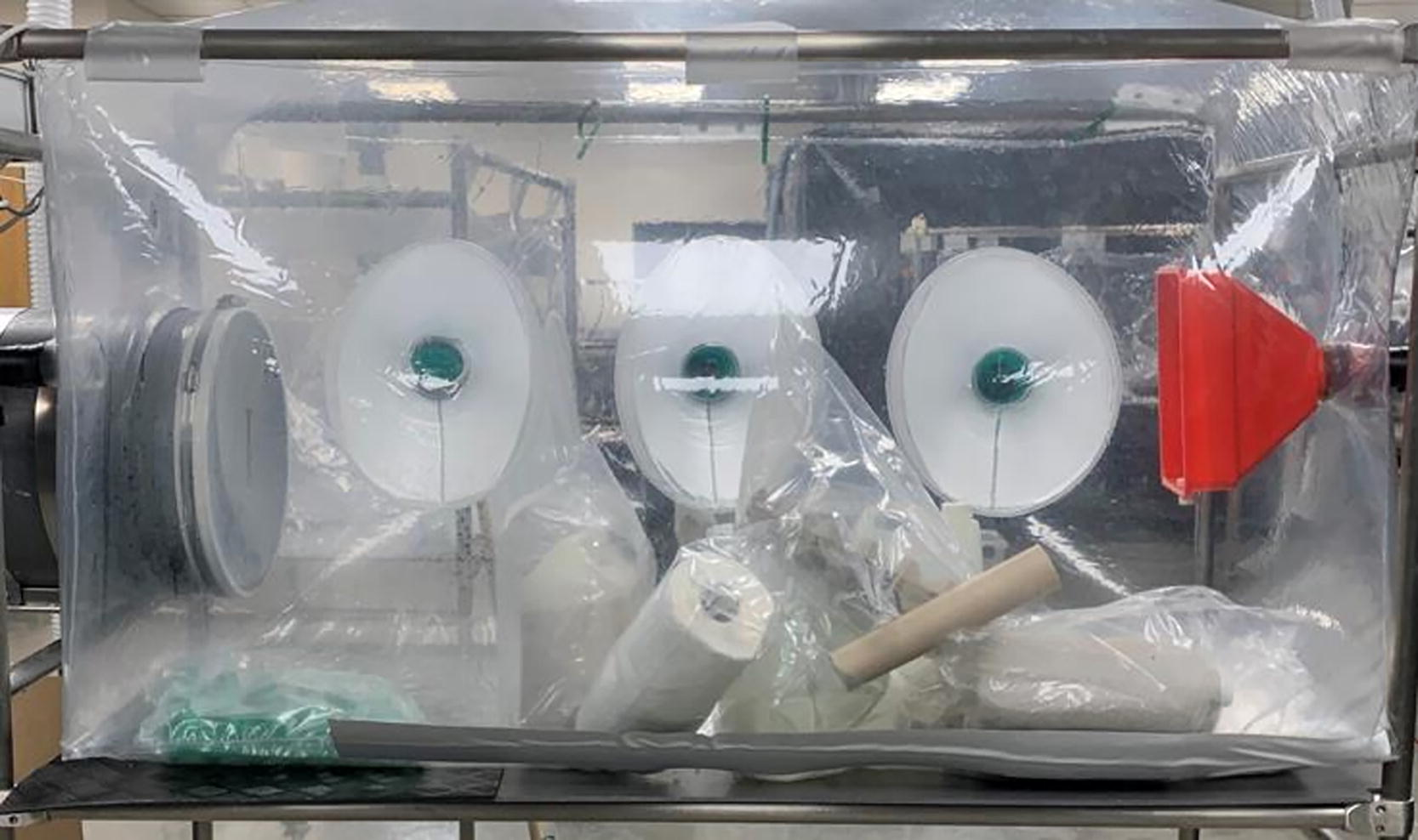
Side view of a PVC isolator with some contents visible on the floor of the interior. PVC, polyvinyl chloride.
The stainless-steel production isolator design is used for the purpose of maintaining adult laying hens and cockerels for the production of naturally laid germ-free eggs. The approximate dimensions of the stainless-steel production isolator are as follows: 9 m (l) × 1.2 m (w) × 1.7 m (h), giving a chamber volume of approximately 18.36 m3.
Prior to isolator processing, environmental monitoring was implemented on six flexible isolators and one production isolator (refer to Table 1). At least four samples were taken from each isolator, including various locations, and sent for analysis at Charles River Laboratories. Samples were positive across each isolator for either or both aerobic and anaerobic bacteria. Positive samples were sent for speciation, with Micrococcus luteus, Paenibacillus humicus, and Staphylococci species being detected. Together with the nonsterile nature of processing, it was concluded that the isolators were not sterile prior to fumigation.
Results of screening swab samples collected for microbiological analyses from PVC and stainless-steel isolators before and after sterilization with chlorine dioxide gas
Note: Swab samples were incubated under aerobic, anaerobic, and microaerophilic conditions. Plates were assessed for the presence or absence of bacterial contamination only. The number of colony-forming units was not determined when growth was detected.
ClO2, chlorine dioxide; FI, polyvinyl chloride (PVC) isolator; G, microbial growth; NG, no microbial growth; PI, production isolator.
Prior to sterilization of each isolator type with ClO2, all internal surfaces of the isolator were cleaned with 70% isopropyl alcohol. Each isolator type was then loaded with the various contents. To facilitate the entry of these items into the isolator, the isolator port cap was removed, thereby exposing the interior of the isolator to room air that was not sterile.
Chlorine Dioxide Dry Gas Generation, Monitoring, and Cycle Validation
A PVC isolator of 1.34 m3 volume was conditioned to ∼65%–75% RH and 17°C–19°C temperature for 10 min, with RH values logged via the Testo RH probe (Testo SE & Co, PE, USA). Chlorine dioxide dry gas (dClO2) was generated via the ClorDiSys dry gas chlorine dioxide generating system (Model 10-001: ClorDiSys Corp., NJ, USA). RH measurements highlighted ambient RH values of above 65%; therefore, humidity generation was not required. The requirement for additional humidity may have resulted in humidity generation that may have, in turn, caused condensation, which should be considered in future studies.
Concentrations were monitored via spectrophotometry, using the Environmental Monitoring System (ClorDiSys, Corp., NJ, USA). ClO2 was injected into the isolator until a concentration of 5 mg/L was attained. ClO2 generation and injection were halted once a target concentration of 5 mg/L (1810 ppm) was achieved.
Concentrations were monitored by using the Environmental Monitoring System. The system is based on a spectrophotometer and is calibrated and validated by its ability to detect concentrations of ClO2 via the manufacturer. The 5 mg/L (1810 ppm) concentration was then held in the isolator (defined as exposure) for a duration equating to 1440 ppm-h. Upon achieving 1440 ppm-h of exposure, the isolator was aerated through ventilation to atmosphere until a concentration of 0.00 mg/L was achieved. Geobacillus stearothermophilus biological indicators (n = 5), swabs (n = 6) collected from multiple locations in the isolator, and settle plates (n = 3) placed in three distinct locations in the isolator were used to verify the antimicrobial efficacy of 1440 ppm-h.
The process was replicated for the production isolator (18.36 m3), with the same ppm-h achieved, although the target concentration was ∼1.5 mg/L (543 ppm). Cycles were run in triplicate for each isolator type. Following achievement of 1440 ppm-h exposure, isolators were then aerated until a concentration of 0.00 mg/L was achieved.
Prior to introducing the ClO2 gas, leak testing, using ammonia solution, was performed to confirm the absence of leaks. The ammonia leak detection test was performed using Ammonia solution 0.88 (Romil Pure Chemistry) and ammonia leak detection cloths (Precision Europe, Northampton, UK). Unopened bottles of Ammonia solution 0.88 were placed in the isolator, and after the port cap was sealed and the internal pressure in the isolator increased to >250 Pa, the bottles of Ammonia solution 0.88 were opened and each aspect of the isolator, i.e., gloves, windows, and port caps, was checked for leaks using the ammonia detection cloths. To aid detection, additional leak testing using a Model D16 PortaSens III meter (Analytical Technology Inc. Collegeville, PA, USA) was performed after the ClO2 gas was deployed to confirm the absence of any leaks during the sterilization process.
Active Fumigation Procedure
Based on the cycle validation results, the following procedure was used for sterilizing PVC isolators: the gas was injected into the interior of the isolator for 80 s and then halted. The gas was left in the PVC isolator for at least 4 h exposure prior to aeration for at least 1 h.
The following procedure was used for sterilizing stainless-steel production isolators: the gas was deployed into the interior of the isolator for at least 5 min and then halted. The gas was left in the stainless-steel isolator for at least 6 h prior to aeration for at least 1 h. Exposure times were extended compared with the minimum cycle times defined by the cycle validation results to provide tolerance to the process. In the case of production isolators, an additional sterilization was performed approximately 24 h after the first sterilization. The use of two cycles of ClO2 for the production isolators was justified based on the volume of the production isolator and owing to the volume of contents placed in the isolator and also to mitigate the potential risk that one cycle was not sufficient. As our practice only took results after two fumigations were completed, we consider this to be a limitation of the study.
In the case of both PVC (n = 32) and stainless-steel production (n = 7) isolators, all loose items located on the floor of the isolator were rotated after approximately halfway through each cycle for both the PVC isolator and the stainless-steel production isolator to ensure all surfaces were equally exposed to the gas.
The isolators used to house germ-free eggs/poultry were always maintained under positive pressure with a working internal pressure of at least 100 Pa.
Microbiology Sampling of Isolators After Sterilization
A detailed microbiology sampling procedure was followed after PVC or stainless-steel isolators were sterilized. This sampling involved collecting swabs samples from multiple locations throughout each isolator type. For example, for the PVC isolators, samples were collected from the locations listed in Table 2. Note: In the case of production isolators, samples for microbiological analysis were not collected until after the second sterilization was completed. The sampling procedure involved three components:
Areas of the PVC isolator from which swab samples were collected
Note: At least three swabs were collected from each PVC isolator. The abbreviations M, O, and I were used to indicate the areas of the isolator/contents from which swab samples were collected.
Millipore Tryptone Soy Agar (TSA) settle plates containing neutralizers (lecithin, Tween®, histidine, and sodium thiosulfate) placed at defined locations throughout the isolator (four settle plates were used in PVC isolators initially, whereas nine settle plates were used in production isolators) and exposed for 4 h. The use of settle plates with neutralizers was purely precautionary in case there was any carryover of ClO2 after aeration. However, after aeration of each isolator type, the levels of dCLO2 were zero. Note: The double-wrapped settle plates were placed into each isolator type prior to sterilization. The settle plates were opened approximately 24 h after the sterilization was completed and the isolator was aerated. Following the completion of the validation of the sterilizing methodology, it was agreed that settle plates would not be used for subsequent routine sterilizations of PVC isolators.
Biological indicators (Crosstex Biological Indicator Spore Strips for monitoring chlorine dioxide Geobacillus stearothermophilus—Cell Line 7953, each strip containing 2.9 × 106 per 6 mm × 30 mm strip) placed at defined locations throughout the isolator (four biological indicators were used in PVC isolators, whereas nine biological indicators were used in production isolators). Each biological indicator strip was placed inside the interior of a closed sterile 15-mL Falcon tube prior to sterilization. Conditioning involved ensuring that the biological indicators were exposed to RH of approximately >65% for at least 10 min before gassing commenced. Prior to the commencement of each sterilization, the cap of the Falcon tube was removed and replaced after the completion of the sterilization process. Each sealed Falcon tube containing the biological indicator strip was aseptically removed from the relevant isolator type after the sterilization was completed and sent to the microbiology laboratory for analysis. Following the completion of the validation of the sterilizing methodology, it was agreed that biological indicators would not be used for subsequent routine sterilizations of PVC isolators.
Swabbing, using COPAN Transystem 132C Cary Blair Agar gel swabs of defined areas (including difficult-to-reach locations) and surfaces in the interior of the isolators (in the case of PVC isolators, three swabs in total were collected from the interior of the isolator, whereas eight to nine swabs were collected from the interior of a production isolator). The tip of each swab was premoistened thoroughly, using sterile water for injection, prior to collection of each swab sample.
Placement of a mold trap(s), for the detection of any visual fungal growth, on the floor of the isolators. A mold trap is prepared by placing approximately 100 g of irradiated poultry diet (diet type is immaterial) into an irradiated 250-mL container and adding irradiated/ultrafiltered water to the diet to create a viscous consistency. Additional irradiated water was added as required to maintain the viscous consistency.
Positive and negative controls were used in all tests (refer to section Microbiological Analysis of Samples Collected from Isolators After Sterilization with dCLO2).
Note: Sterility was defined as achieving a 106 log reduction in Geobacillus stearothermophilus spores, which is in line with international standards for sterility assurance levels. 17
Isolator Sterility Maintenance
In order to confirm that the isolator was maintaining sterility, samples were collected once weekly, for at least 3 weeks, for microbiological analysis.
In one production isolator (PI #2) that was not scheduled for immediate use for the housing of germ-free birds, in addition to the settle plates and biological indicator samples referred to above that were collected after sterilization, swab samples were collected for up to 100 days after the isolator was sterilized.
Microbiological Analysis of Samples Collected from Isolators After Sterilization with dCLO2
Samples collected for microbiological analysis were analyzed in compliance with our standard operating procedures.
The settle plates were incubated for 5 days at 20°C–25°C followed by 3 days at 30°C–35°C. Nonexposed plates from the same batch used on the test were used as negative controls. As positive controls, plates from the same batch were opened inside a laminar flow cabinet for 4 h, and after that, the plates were inoculated with Staphylococcus aureus (NCTC 12981) and Candida albicans (NCPF 3179). Negative and positive controls were incubated together with the settle plates for the same period and under the same conditions. We did not expose plates to dCLO2 and then inoculate them—this could be regarded as a potential limitation of the study.
The biological indicators were aseptically transferred to 10 mL of Tryptone Soy Broth (TSB) (Fannin Ltd, Dublin, Ireland) and incubated at 55°C–60°C for 7 days. Noninoculated tubes were used as negative controls. A biological indicator strip from the same lot, nonexposed to dCLO2, was transferred to a TSB tube from the same batch as a positive control. Negative and positive controls were incubated together with the test strips for the same period and under the same conditions.
Each swab was rubbed on the surface of four culture media plates: TSA (Fannin Ltd, Dublin, Ireland) and incubated at 30°C–35°C for 7 days in aerobic condition, TSA + 5% sheep blood (Fannin Ltd, Dublin, Ireland) and incubated at 30°C–35°C for 7 days in anaerobic condition, TSA + 5% sheep blood (Fannin Ltd, Dublin, Ireland) and incubated at 30°C–35°C for 7 days in microaerophilic conditions, and Sabouraud dextrose agar (SDA) (Fannin Ltd, Dublin, Ireland) and incubated at 20°C–25°C for 14 days in aerobic conditions. A sterile swab was processed in the same way and applied to the same culture media from the same batch as the negative control. One plate of each media was inoculated with a reference strain microorganism as positive control (blood agar: Clostridium sporogenes NCTC 532 and Campylobacter jejuni NCTC 11351; TSA: Bacillus subtilis NCTC 10400; and SDA: C. albicans NCPF 3179). Negative and positive controls were incubated together with the test plates for the same period and under the same conditions. We did not expose plates to dCLO2 and then inoculate them—this could be regarded as a potential limitation of the study.
Mold traps were visually inspected at least once weekly for filamentous fungus growth.
Post-Sterilization Leak Testing
In addition to the leak testing performed before and during sterilization with ClO2, it is crucial to test the integrity of the sterilized isolator for the presence of leaks. For each of the isolators referenced above, leak testing of all surfaces of the isolator, including gloves and isolator sleeves, was performed on a weekly basis to confirm the absence of leaks. In order to test the integrity of the isolator, the internal pressure was increased to at least 400 Pa, the inlet and outlet valves were closed, and a soap solution was used to check for leaks. 18 We routinely also performed leak testing using soap solution, once weekly, once isolators were populated with germ-free birds.
Sampling Frequency After Isolator Sterilization
Samples were collected from isolators (PVC and stainless steel) at least three times after sterilization. When contaminations were detected, an investigation was performed to elucidate the possible cause of the contamination. In all cases of confirmed contamination, the relevant isolator was resterilized and swab samples were collected after sterilization to confirm the effectiveness of the resterilization process.
Results
Initial Cycle Validation Results
The results of the validation (refer to Figure 2) confirmed that 80 s of dClO2 injection achieved a target concentration and an exposure of 5 mg/L (1810 ppm) and 1440 ppm-h in the PVC isolator, respectively, equating to an absolute time of ∼50 min exposure (see Figure 2). An injection of 5 min of dClO2 achieved a target concentration and an exposure of 1.5 mg/L (543 ppm) and 1440 ppm-h in the stainless-steel production isolator, respectively, with an absolute cycle time of ∼2 h and 45 min). Aeration successfully reduced dClO2 levels to 0.00 mg/L following achievement of 1440 ppm-h across both cycle types, with complete aeration achieved in <10 min for both isolator types. The resulting minimum cycle times were confirmed as ∼1 h for the PVC isolators and ∼3 h for the production isolators. Biological indicators, swabs, and settle plates displayed no microbial growth at 1440 ppm-h.
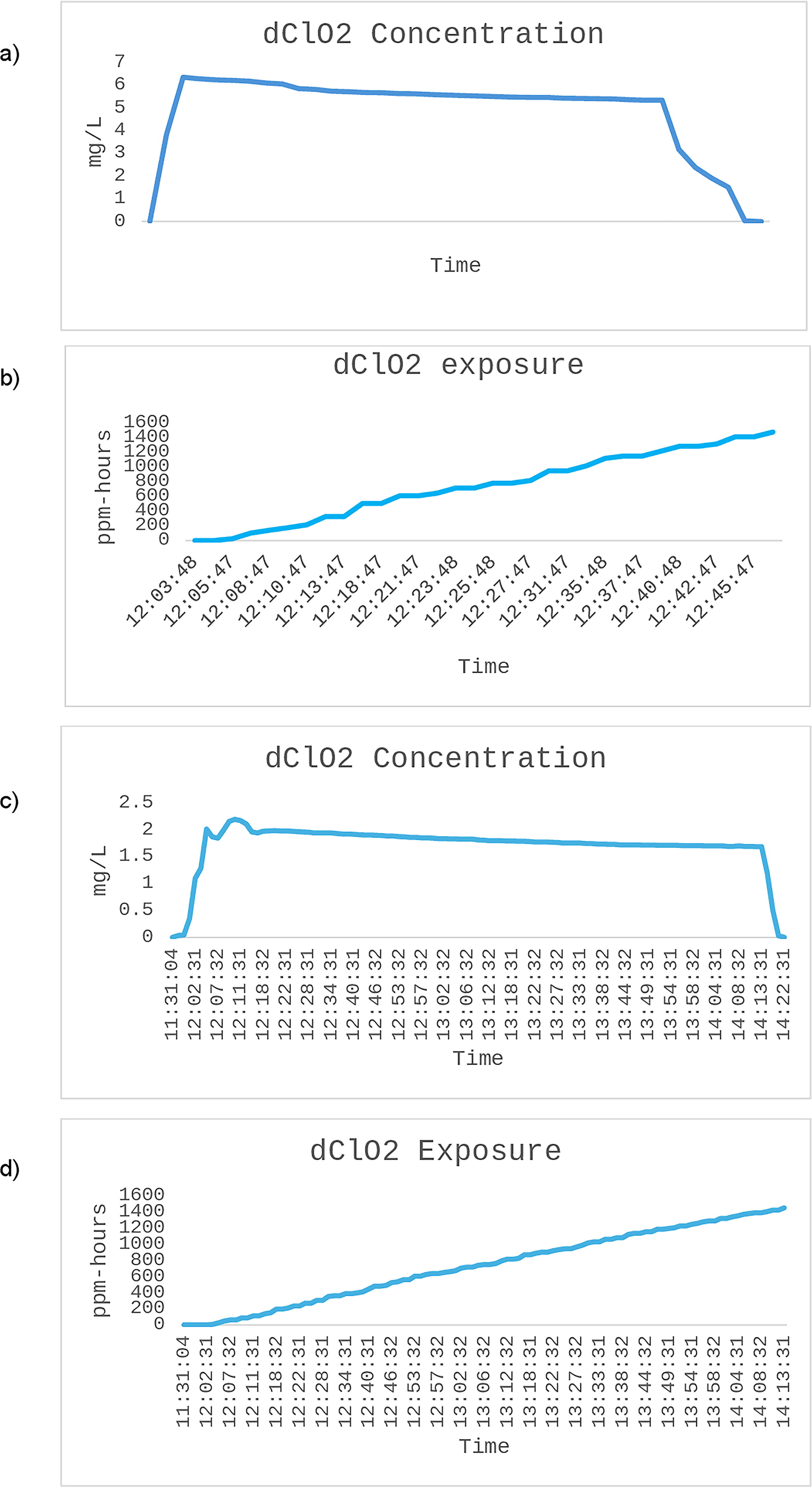
Active Fumigation Results
The results of the analysis of the various samples collected for microbiological analysis after isolator sterilization are presented in Table 3. Sterilization of each isolator type was achieved by using dry ClO2, following the previously described protocol (see Active fumigation procedure in methods), with reproducible results. Isolators maintained their sterility for up to at least 100 days (see data from PI #2 in Table 3) and after the transfer of germ-free birds/eggs into the isolator. Confirmation of maintenance of sterility after placement of germ-free birds/eggs was by analysis of fecal samples from birds in the isolator, which is a standard procedure for germ-free animals. 10
Results of microbiological analyses of samples (settle plates, biological indicators, and swabs) collected from PVC and stainless-steel isolators after sterilization with ClO2 gas
Eight plates had no growth; one plate fell on the floor and the plate cover fell off—the plate was sent for analysis and growth was detected.
Growth detected in one mold trap—contamination linked to preparation of the mold trap with water from an ultrafiltration system. PI #2 was emptied of all materials, all internal surfaces cleaned with 70% isopropyl alcohol, restocked with all relevant consumables, and resterilized twice with chlorine dioxide as per the Material and Methods section. Two new mold traps were prepared using irradiated poultry diet and irradiated water, and no growth was detected afterward.
Growth detected in one swab—contamination linked to procedural modification, which was then reversed.
FI, PVC isolator; G, microbial growth; NG, no microbial growth; PI, production isolator.
On occasion, swab samples indicated the presence of fungal/bacterial contamination, e.g., PI #2 and FI #3; however, investigation demonstrated no link with the dClO2 efficacy.
Discussion
The use of ClO2 fogging as a sterilant in GFM practices, 19 together with the superior safety, efficacy, and consistency of dClO2 over other fumigants described in previous studies, 3 provided a robust rationale for selecting dClO2 gas as the sterilant of choice for germ-free avian egg practices in this study. In support of previous findings, dClO2 exposure provided complete sterilization of the area being fumigated,3,14 with this study providing the first results of its kind, to the authors’ knowledge, for isolators used in germ-free egg facility practices. The results of this study highlight the first use of dClO2 gas for germ-free avian sterilization practices, augmenting on its use as a fogging agent seen in GFM practices. 19 As seen in prior studies, dClO2 provided rapid cycle times with no residual clean-up, with the production isolator cycle time being in line with previous dClO2 studies.14,16 However, quicker cycle times were achieved for the flexible isolators in this study. The minimum cycle time to achieve 1440 ppm-h, an exposure threshold previously used for sterilization practices in animal facilities, 13 was 1 h. This 1-h cycle time augments substantially on the turnaround time required for sterilization of isolators and biological safety cabinets seen in previous dClO2 studies.3,14,16 The use of two cycles of ClO2 for the production isolators was justified based on the volume of the production isolator and owing to the volume of contents placed in the isolator and also to mitigate the potential risk that one cycle was not sufficient. As our practice only took results after two fumigations were completed, we consider this to be a limitation of the study.
VHP and PAA have been used for isolator fumigation in the past as a noncarcinogenic alternative to formaldehyde. 20 The evidence in this study shows that dClO2 provides an additional alternative to both VHP and PAA for isolator fumigation, with speeds comparable with using either VHP or PAA. 20 In contrast to VHP, dClO2 did not encounter any distribution issues, with concentrations being consistently above the desired amount for the cycles of both isolator types to achieve antimicrobial efficacy across all areas (see Figure 2), 20 supporting the advantages of gaseous protocols previously defined.3,6,20 dClO2 distributed evenly throughout the isolator based on the strategic placement of spore strips and settle plates at multiple locations and the collection of swabs samples from multiple locations in the isolator, including from the HEPA filters, achieving 106 log reduction across all challenges. Further studies may investigate distribution and penetration capabilities utilizing other methodologies, although such research is outside the scope of this study. As described in previous animal laboratory studies, >1440 ppm-h cycle achieved in this study provided consistently adequate antimicrobial efficacy for the sterilization of germ-free egg facilities. 13
As the egg incubation and hatching processes rely heavily on the use of electrical/battery-operated equipment (e.g., balances for egg weighing, incubators for incubating and hatching eggs, and heated mats used to provide supplemental heating for newly hatched chicks), it is crucial that any electrical/battery-operated equipment that is exposed to a sterilization agent is not adversely impacted by the use of that agent. In this context, dClO2 is a suitable agent that can sterilize the items listed above without leading to oxidization of these items or adversely impacting their functionality. 21 The data from the current study clearly show no adverse effects of dCLO2 on the functionality of electrical/battery-operated items used in isolators for germ-free eggs and poultry. Recent studies have highlighted material compatibility issues caused by fumigants including both dClO2 and VHP in other applications.11,22 Future research could consider additional material compatibility evaluations for ClO2 applications within avian germ-free egg facilities, although such material compatibility assessment was outside the scope of this study.
Two of the key requirements for any sterilization agent used in sterilizing isolators to be used in germ-free processes is the duration of efficacy and confirmation of the duration of efficacy by analysis of samples collected for microbiological analysis. 10 Companies involved in the production and supply of germ-free rodents such as Taconic Biosciences and Charles River Inc. recommend testing isolators for at least 3 weeks after sterilizations have been completed to confirm that the isolator has maintained its sterility after sterilization. 10 This approach helps to identify any slow-growing bacteria prior to placement of germ-free eggs or birds in an isolator. To gain data on the duration of sterility of the isolator, Midcalf and colleagues 18 recommend following a sampling plan when sampling isolators after sterilization. This plan should also include periodic random process sampling to ensure contaminations do not go undetected. We have used and continue to use this approach for isolator sampling after sterilization with dClO2, and the microbiological (bacteriological and fungal) data that we have generated support this approach.
Guitton and colleagues 9 noted that even though they sterilized rigid isolators with formaldehyde vapor on three occasions, using 60 mL of formalin (24% formaldehyde) added to 30 g of potassium permanganate per cubic meter, approximately 10% of the isolators were not germ free. Our representative results presented in Table 3 show 100% efficacy of ClO2 when administered at the rates described in this article. When compared with formaldehyde, these results highlight not only increased efficacy but also enhanced maintenance of sterility over time, including the stewardship of slow-growing organisms and augmenting germ-free status. However, when a contamination in an isolator was detected, it was, upon investigation, not attributed to a failure of sterilization with dClO2.
Conclusions
Chlorine dioxide dry gas (dClO2) provides a promising noncarcinogenic sterilization solution for germ-free egg facilities, providing sterility to associated isolators.
Footnotes
Authors’ Contributions
M.M.: Conceptualization, methodology, validation, and writing (original draft, review, and editing). R.J.: Conceptualization, methodology, validation, and writing (original draft, review, and editing). Ad.S.: Methodology and writing (original draft, review, and editing). M.A.: Methodology and writing (original draft, review, and editing). C.C.: Methodology and writing (original draft, review, and editing).
Disclaimer
The article contents, including any opinions and/or conclusions expressed, are those of the authors alone.
Ethics Approval
Ethics approval was not required for the work described in this publication, as it did not involve the use of animals.
Authors’ Disclosure Statement
No competing financial interests exist.
Funding Information
The results incorporated in this publication received funding from the European Union’s Horizon 2020 research and innovation program under grant agreement no. 858390.
