Abstract
Introduction:
Cephalosporins can trigger hypersensitivity reactions in certain individuals. Consequently, strict regulations restrict the production of non-beta-lactam substances during or after cephalosporin manufacturing. Dry chlorine dioxide gas (dClO2), together with ultra-performance liquid chromatography Mass spectrometry/mass spectrometry (UPLC-MS/MS) detection methods, has emerged as a promising method for decontaminating cephalosporin compounds. This study aimed to assess whether a standardized dClO2 and testing protocol could provide successful decontamination of a broad spectrum of cephalosporins while also providing an indicative assessment of degradants and their biological activity.
Methods:
Chemical indicators (CIs) mimicking different surfaces (stainless steel, Perspex®, aluminum) were contaminated with 1 μg/cm2 of each cephalosporin and exposed to 9600 ppm-h of dClO2, followed by UPLC-MS/MS analysis (phase 1). Cephalosporins underwent degradation assessment after exposure to ClO2 in an aqueous solution (phase 2). In total, 100μg of each compound was subjected to 400 ppm of dClO2 for 24 h (9600 ppm-h), followed by UPLC-MS/MS analysis. Antimicrobial susceptibility disks (30 μg) of cefaclor underwent identical treatment cycles and UPLC-MS/MS analysis. Subsequently, these disks were placed in Escherichia coli cultures to evaluate the biological activities of the degradants.
Results:
The 9600 ppm-h of ClO2 exposure effectively degraded all cephalosporin compounds to levels <0.002 μg/cm2 on surfaces (phase 1), <2 ppb in solution, and <0.02 μg/disk (phase 2). The antimicrobial efficacy of cefaclor was nullified after the same exposure, confirming complete inactivation of the degradants.
Conclusion:
A decontamination protocol utilizing dClO2, combined with UPLC-MS/MS and biological activity testing, has significant potential to enable facility repurposing for the production of non-beta-lactam compounds.
Introduction
Beta-lactam antibiotics, including penicillins, carbapenems, monobactams, and cephalosporins, are a predominant group of antibiotics administered globally, accounting for ∼65% of all antibiotics sold. 1 All groups contain a four-membered nitrogen-containing beta-lactam ring (Figure 1), which represents the active bactericidal component.
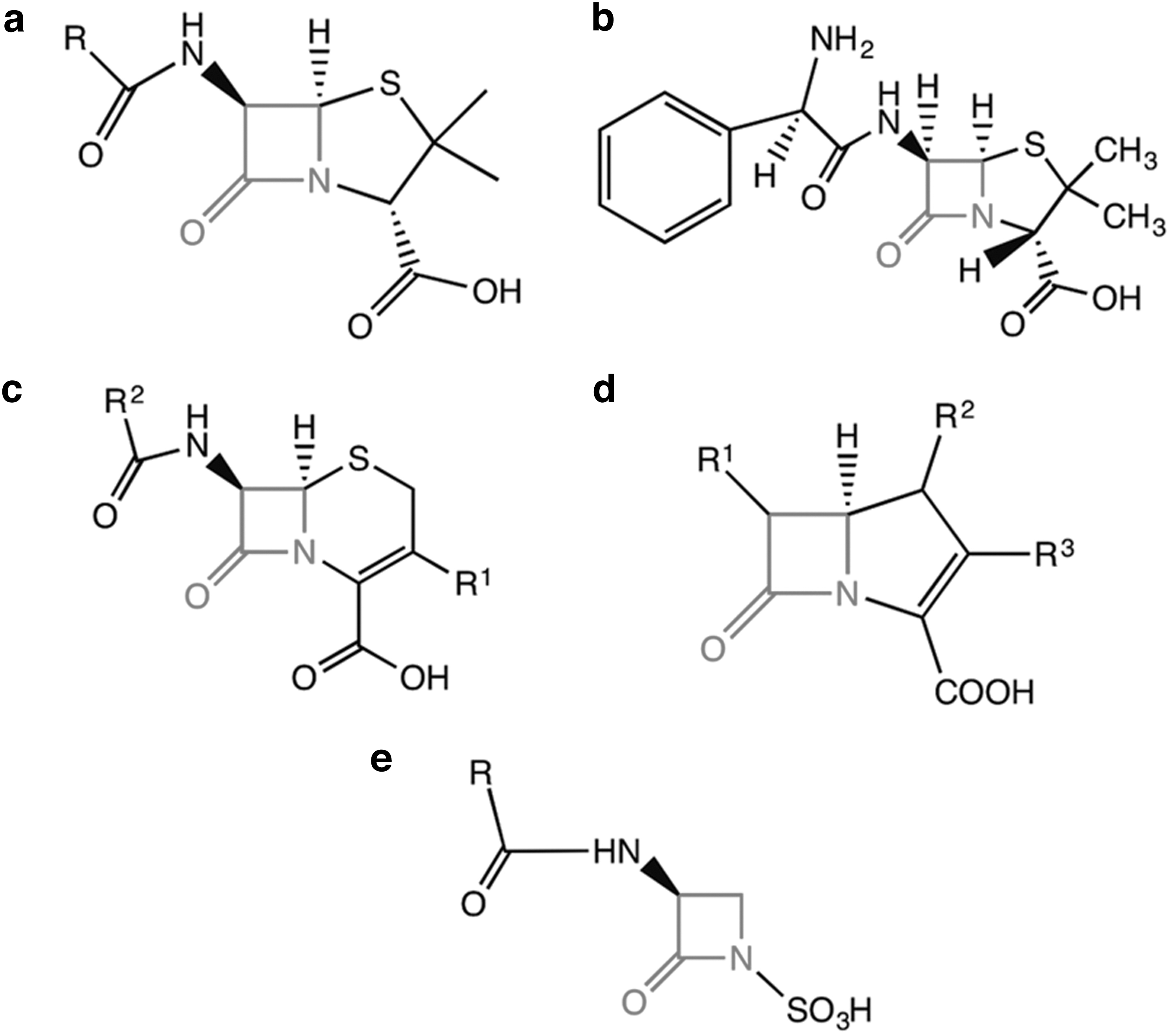
General molecular structures of the major beta-lactam antibiotic groups.
Hypersensitivity of some individuals to these antibiotics is well characterized, with beta-lactam antibiotics being the leading causative agents of drug-induced anaphylaxis, causing >1000 deaths a year.2,3 The potential hazards of accidental exposure of beta-lactams to sensitive individuals are well characterized, and steps to avoid cross-contamination in manufacturing facilities have been recommended in guidelines issued by organizations such as the FDA. 4 Such steps involve complete physical isolation of production and packaging operations from other processes and for purpose-built facilities restriction to production of beta-lactam product.4,5
Fundamentally, similar guidance exists for many regulated geographies around the world, such as European Medical Agency, China Food and Drug Administration, and Central Drugs Standard Control Organisation. 6 Nevertheless, guidelines specific to penicillin, including the 21 CFR 211.176 annex, state that a facility that previously produced penicillin dosages can be decontaminated to produce non-beta-lactam products. 7
Beta-lactam facilities have much of the instrumentation, expertise, and protocols in place to produce active pharmaceutical ingredients (APIs) of any nature; therefore, the repurposing of these facilities to produce nonbeta-lactam products provides an attractive proposition, providing flexibility, saving time, and costs in comparison with building a new facility elsewhere to produce the APIs of interest. 8 Decontamination restrictions limit the ability of pharmaceutical plants to switch from beta-lactam to non-beta-lactam production. 6
This has led organizations to analyze the risk to sensitive individuals of later-generation beta-lactams and build conceptual frameworks.9–13 These risk assessments require validated cleaning and decontamination of equipment and structures to allow safe servicing of equipment, repairs, renovations, and end-of-life cycle decontamination before repurposing or deconstruction, but these protocols are not definitive or standardized. 8
Current legislation states that the decontamination approach must allow for safe reuse of the facility, equipment, and provide evidence that any remaining beta-lactam residue has been reduced to an “acceptable level.” 7 Regulatory bodies do not set definitive quantitative acceptance criterion for any cleaning validation, that is, an “acceptable level” owing to the impractical nature of such a process. Rather, the onus is placed on individual firms to develop a rationale for an acceptance criterion for residual levels that needs to be logical, practical, achievable, and verifiable, based on the knowledge of materials used.
Acceptance criterion standards are present in other industries that specify maximum residual limits (MRLs) for penicillins, such as the veterinary medicine directorate of the United Kingdom and the European Communities Council (ECC), which define MRLs for penicillins in foodstuffs (5 ppb) and dairy products (50 ppb), respectively. 10
The acceptance criteria must include the sensitivity of the analytical methods used and the efficacy of decontamination agents must be scientifically proven. 7 Common acceptance criteria for decontamination of APIs within the industry are based on analytical, biological activity, and organoleptic levels of residues, with results of 10 ppm, 1/1000 of therapeutic dose, and no visible residues being used, respectively.
Owing to the risk of hypersensitivity of beta-lactam antibiotic production, analytical methods, such as liquid chromatography with tandem mass spectrometry (LC-MS/MS), provide the most sensitive methods to validate presence of beta-lactam antibiotics, with limit of detections (LODs) <0.01 μg/cm2 level for surfaces, and similar levels ∼0.1 μg/g (<100 ppb), for formulated materials being identified as achievable. 3 Although LC-MS/MS provides a suitable analytical testing methodology for beta-lactam antibiotics, a suitable and standardized biological activity testing method is currently lacking.
Currently, the pharmaceutical industry has no defined acceptance criteria for decontaminating a broad spectrum of cephalosporin antibiotics, and there is no analytical approach for identifying surface contamination by ultra-performance liquid chromatography Mass spectrometry/mass spectrometry (UPLC-MS/MS), with past work limited to penicillin G.3,8,14 The development of an analytical detection method for cephalosporin surface contamination, such as UPLC-MS/MS, and specification of standardized detection limits are required to provide a logical, practical, achievable, and verifiable acceptance criterion for the industry to validate the success of cephalosporin decontamination procedures.
Gaseous Decontamination Methods
In addition to the development of an acceptance criterion, the industry lacks a standardized decontamination protocol for the repurposing of cephalosporin-contaminated equipment and facilities. A successful and consistent decontamination protocol must ensure that all surfaces are exposed to the decontamination agent at an effective concentration for a proven period of time. 6 Manual liquid deep cleaning is insufficient for complete decontamination due to the inability of the liquid cleaning process to reach all surfaces, providing a process limitation regarding facility repurposing, such limitations are common in biological contamination issues. 15
In the pharmaceutical and healthcare industries, containment facilities include gaseous decontamination methodologies within their decontamination protocols associated with microbial contamination, in addition to liquid cleaning, to ensure that all surfaces are treated. 16 However, such gaseous protocols are rarely applied and are not standardized for the contamination control of APIs and other sensitizing agents. 6
The application of dry chlorine dioxide gas (dClO2) has emerged as a potential gaseous solution for beta-lactam decontamination protocols.17,18 Previous studies have shown that dClO2 can successfully decontaminate beta-lactam residues following conventional cleaning methods, providing a practical and achievable approach to both equipment and facility decontamination.17–19
In 2009, Lorcheim, Lorcheim, and Czarneski reported successful beta-lactam decontamination with dClO2 gas. 17 Subsequently, extensive studies by Lorcheim reported successful results, with >99.9% destruction of eight beta-lactams, including amoxicillin, ampicillin, cefadroxil, cephalexin, cefazolin, imipenem, penicillin V, and penicillin G. 17 This study cited a range of decontamination conditions and established an optimum cycle for achieving acceptable decontamination. Exposures of 7240 ppm-h or higher were found to be effective at beta-lactam destruction (see Figure 2).
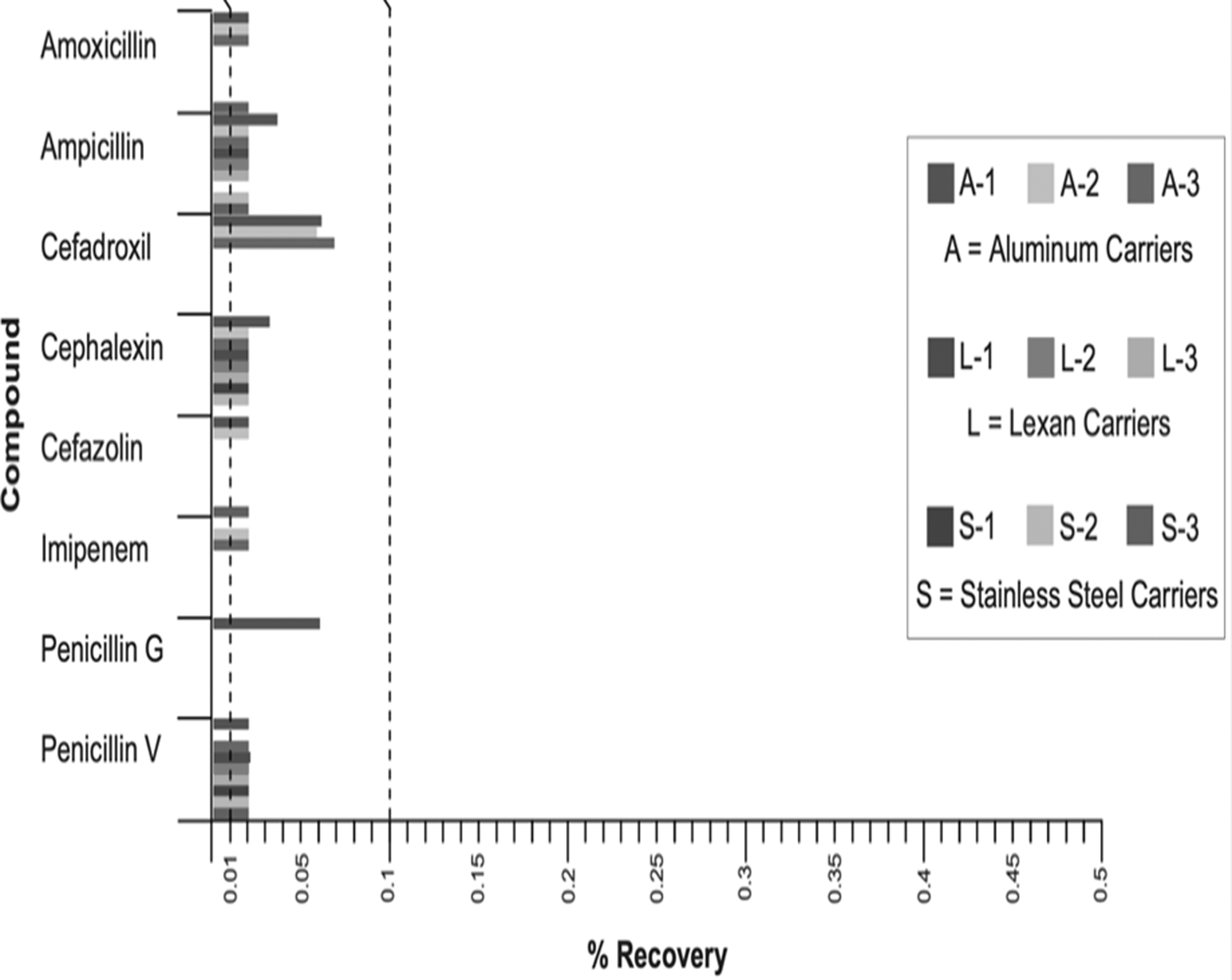
Lorcheim 17 >7240 ppm-h exposure results.
Chemical indicators (CIs) were developed and used with three material bases: stainless steel (SS), aluminum (Al), and polymer (Lexan), and assessed by LC-MS/MS. Such indicators provide a basis for in situ verification of the dClO2 gaseous decontamination process. However, although this study highlights dClO2's ability to degrade beta-lactams, acceptance was based on a log reduction and does not state the sensitivity of the analytical test used, conflicting with current guidelines.
Cole 18 confirmed the results of the dClO2 method in the repurposing of a facility that had been used for amoxicillin and penicillin production, supporting the findings of Lorcheim. 17 Augmenting on previous studies, the Cole study (2014) 18 implemented the use of CIs and an accepted level of cleanliness that was based on absolute amount rather than log reductions.10,18 Postdecontamination results were reported to be <50 ppb found on the tested surfaces (SS, Al, and Perspex®).
A level of cleanliness derived from the ECC guidelines 1990 (<50 ppb final product) was used as the acceptance criterion.10,18 The conditions used in this confirmatory study again showed that a minimum 7240 ppm-h cycle was effective for the decontamination of the respective compounds. Results for amoxicillin demonstrated a reduction from 130 to 0.05 ppb, and penicillin V from 317 to 1.5 ppb. These results were in general agreement with those of previous studies and demonstrated minor differences between individual beta-lactam reactivity and variations in recoveries on various surfaces.17,18 Both studies demonstrated that superior results were obtained using a gaseous decontamination method, as contact with surfaces was more effective and complete than conventional cleaning alone.17–19
Study Aims and Objectives
Although previous studies have shown promise for the decontamination of cephalosporins, they have not adequately addressed the critical issues of standardization of a decontamination protocol for industry-wide applications. Our primary objectives are as follows:
Create a UPLC-MS/MS and CI technique that covers a wide range of cephalosporins and has detection limits comparable with those observed in prior research.
3
Establish a broad range cephalosporin decontamination protocol using gaseous chlorine dioxide.
In addition, we aimed to evaluate the degradation products post-decontamination and their biological activity, which has previously been of concern, in addition to the goals stated above. 6 Although LC-MS/MS can show degradation, it cannot guarantee that sensitizing compounds have been inactivated. To close this gap, we plan to research additional testing techniques outside the traditional LC-MS/MS, with an emphasis on presenting proof of biological activity for inactivation of the investigated cephalosporins.
According to our hypothesis, the disk diffusion test, which is frequently used in microbial diagnostic laboratories to evaluate bacterial resistance to antibiotics, may be utilized to ascertain whether cephalosporins exposed to dClO2 are inactivated. Growth of typically susceptible bacteria within 1 mm of an antimicrobial susceptibility disks (ASDs), impregnated with a known amount of antibiotic, after dClO2 exposure may signal compound deactivation. This test will be used to supplement or contrast the results of UPLC-MS/MS.
To Achieve These Objectives, Our Research Comprises Two Distinct Phases
Phase 1: Gaseous decontamination
Gaseous treatment of common surface materials loaded with 1st, 2nd, 3rd, and 4th generation cephalosporins, applying a standardized exposure of 9600 ppm-h to dClO2, will be conducted to evaluate the completeness and efficacy of the decontamination process. This phase will also involve the development of a UPLC-MS/MS detection method for these surfaces. An exposure of 9600 ppm-h was selected due to ∼10,000 ppm-h accumulative exposure showing consistent results in previous studies. 17
Phase 2: Degradation study
Known quantities of each cephalosporin compound will be treated in an aqueous chlorine dioxide solution for 24 h, equivalent to 9600 ppm-h. These experiments aim to identify degradation products and establish detection limits using UPLC-MS/MS for comparison with previous studies. 3 In addition, in this phase, we will perform biological activity testing to confirm or deny antibiotic inactivation. Precoated ASDs were treated with gaseous chlorine dioxide and then tested on agar plates inoculated with Escherichia coli to assess the effectiveness of the remaining antibiotics. ASDs will also be subjected to UPLC-MS/MS testing to provide comparative results.
Materials and Methods
Phase 1: Gaseous Decontamination
CI preparation and testing
Testing was performed using CIs prepared by inoculating 100 cm2 of three carrier materials (SS, Perspex®, and Al) with five types of cephalosporin antibiotics (see Table 1). The carrier materials evaluated in this study were polycarbonate polymers (Perspex®), SS (304L, passivated), and Al (nonanodized). Each CI was loaded with 0.1 mL of a 10 μg/mL mixed solution of five cephalosporin compounds (representative of 0.01 μg of contaminant per 1 cm2 (Supplementary Data S1 for further detail). Linear ranges and LODs, quantification, and reporting were determined during the method development.
Details of cephalosporin compounds used
After completion of decontamination cycles, CIs and positive controls were extracted and sent to AMAL Analytical for UPLC-MS/MS testing to assess the presence of cephalosporin. CIs were tested in replicates of five for each compound and carrier material. A “pass” criterion for successful decontamination was applied based on the remaining amount of cephalosporin being undetectable at or below the limit of reporting (LOR) of the UPLC-MS/MS method developed, as previously defined for each compound and each surface.
Gaseous chlorine dioxide
A ∼1.1 m3 test chamber was prehumidified to appropriate levels by humidity fogging to ∼80% relative humidity (RH). The humidity levels were assessed using a calibrated Testo RH probe (Model 440). The chamber was loaded with CIs (coupons), impregnated with five cephalosporin compounds, decontaminated, and sealed for gassing in a 9600 ppm-h chlorine dioxide cycle (see Supplementary Data S3). The chamber was then safely vented through activated carbon, and the coupons were analyzed for the remaining cephalosporins by LC-MS/MS (Waters Acquity TQs micro UPLC-MS/MS system).
dClO2 was directed into the sealed chamber, by means of a Clordysis method gas generation system (Clordysis Corp., NJ, USA). The flow rate and pressure of the gas were carefully monitored to be 20/L (+/−1) per second of gas. The concentration was monitored using a calibrated Clordysis EMS single-beam spectrophotometer/integrating instrument (Clordysis Corp.). The concentration time data for decontamination were recorded. At the conclusion of the decontamination run, the gas was safely withdrawn through a dedicated scrubber unit filled with ∼25 L of active carbon (AAF activated carbon in the valid date range). Staff monitored the chlorine dioxide levels until readings were at or <0.1 ppm (see Supplementary Data S3: Graph S1) when the chamber was deemed safe to open.
Phase 2: Degradation Study
To assess ClO2's ability to degrade cephalosporins, 0.25 mL of a 2500 μg/mL cephalosporin solution of five different cephalosporins (Table 1) was inoculated into separate 15 mL high-density polyethylene plastic (HDPE) vials, and each vial was then inoculated with 5 mL of 400 ppm chlorine dioxide solution and left at room temperature for 24 h. Upon completion, 1 mL of each chlorine dioxide solution was transferred into a separate HDPE vial containing sodium thiosulfate to neutralize the reaction. Each sample was then transferred to an autosampler vial for LC-MS/MS analysis.
Control solutions were prepared by inoculating each cephalosporin into 5 mL solution without the addition of ClO2, and after 24 h, each control was transferred to an autosampler for LC-MS/MS analysis. All samples were run on a Waters Acquity TQs micro UPLC-MS/MS system using a C18 BEH column; the eluents were 0.1% formic acid in ultrapure water (A) and 0.1% formic acid in acetonitrile (B). Degradation studies were performed in MS scan mode with any peaks identified background corrected, whereas quantitative analysis was performed in mass reaction monitoring mode for maximum sensitivity. The study was performed in triplicate.
Biological activity testing
Preloaded 30 μg cefaclor ASDs (Oxoid® Thermo-fisher, USA) were treated with 9600 ppm-h dClO2 exposure in a gassing chamber. After gassing, the disks were subjected to a disk diffusion test, being contacted with agar plates (Nutrient Agar plates Med 3.30; Southern Biological, Australia) loaded with E. coli (K-12 Strain; Southern Biological). The ASDs were arranged five times per plate with two replicates. A separate set of plates was arranged with five control ASDs (no dClO2 exposure) and two replicates. The plates were allowed to develop in an incubator under optimal growth conditions. The plates were visually assessed over a 4-day period, with growth within 1 mm of the disk demonstrating inactivation.
Two additional preloaded 30 μg cefaclor ASDs (Oxoid® Thermo-Fisher, USA) were treated with identical conditions (9600 ppm-h) and sent for UPLC-MS/MS analysis after dClO2 exposure, duplicate untreated controls were subjected to UPLC-MS/MS analysis. The UPLC-MS/MS method development for disks was identical to that carried out for the CI, although extraction was carried out directly from the disks. Only the LOR was provided as LODs and limit of quantifications (LOQs) were ignored after initial CI results.
Results
Phase 1: Gaseous Decontamination
The UPLC-MS/MS CI method achieved a LOD, LOQ, and LOR of <0.00046, <0.0014, and 0.002 μg/cm2, respectively, for all compounds across all surfaces (see Table 2). Average recoveries between 95% and 107% and residual standard deviations between 8% and 12% were achieved for all compounds across all surfaces. Only the LOR of 0.002 μg/cm2 was consistent across all compounds and surfaces (see Supplementary Data S2 for more detail). dClO2 exposure for 9600 ppm-h provided complete degradation of the parent compounds of each cephalosporin compound across each surface to or below the LOR, meeting the “pass” criterion (Table 3).
Detection limits defined by the UPLC-MS/MS analytical method for each compound for all surfaces
AR, average recovery (%); LOD, limit of detection; LOQ, limit of quantification; LOR, limit of reporting; RSD, residual standard deviation (%); UPLC-MS/MS, ultra-performance liquid chromatography Mass spectrometry/mass spectrometry.
Gaseous dClO2 decontamination of cephalosporin inoculated chemical indicators
Pass criterion refers to the remaining amounts of each compound on each surface (columns 5, 6, and 7), being equal to or below that of the LOR postexposure.
dClO2, dry chlorine dioxide gas; LOR, limit of reporting.
Phase 2: Degradation Study
After exposure to 400 ppm of dClO2 over a 24-h period (9600 ppm-h), the parent compound for each cephalosporin showed complete degradation to the LOD of the UPLC-MS/MS method used, with the retention times and mass spectra for each compound being different postexposure (Table 4 and Figure 3). The instrument detection limits for the developed UPLC-MS/MS method were between 0.002 and 0.0005 μg/mL (2 and 0.5 ppb) for each compound in solution (see Table 4). Degradation products were detected for each compound post-exposure; however, degradation was not uniform across compounds, with different numbers and different sizes seen for each (see Supplementary Data S4 for MS images).
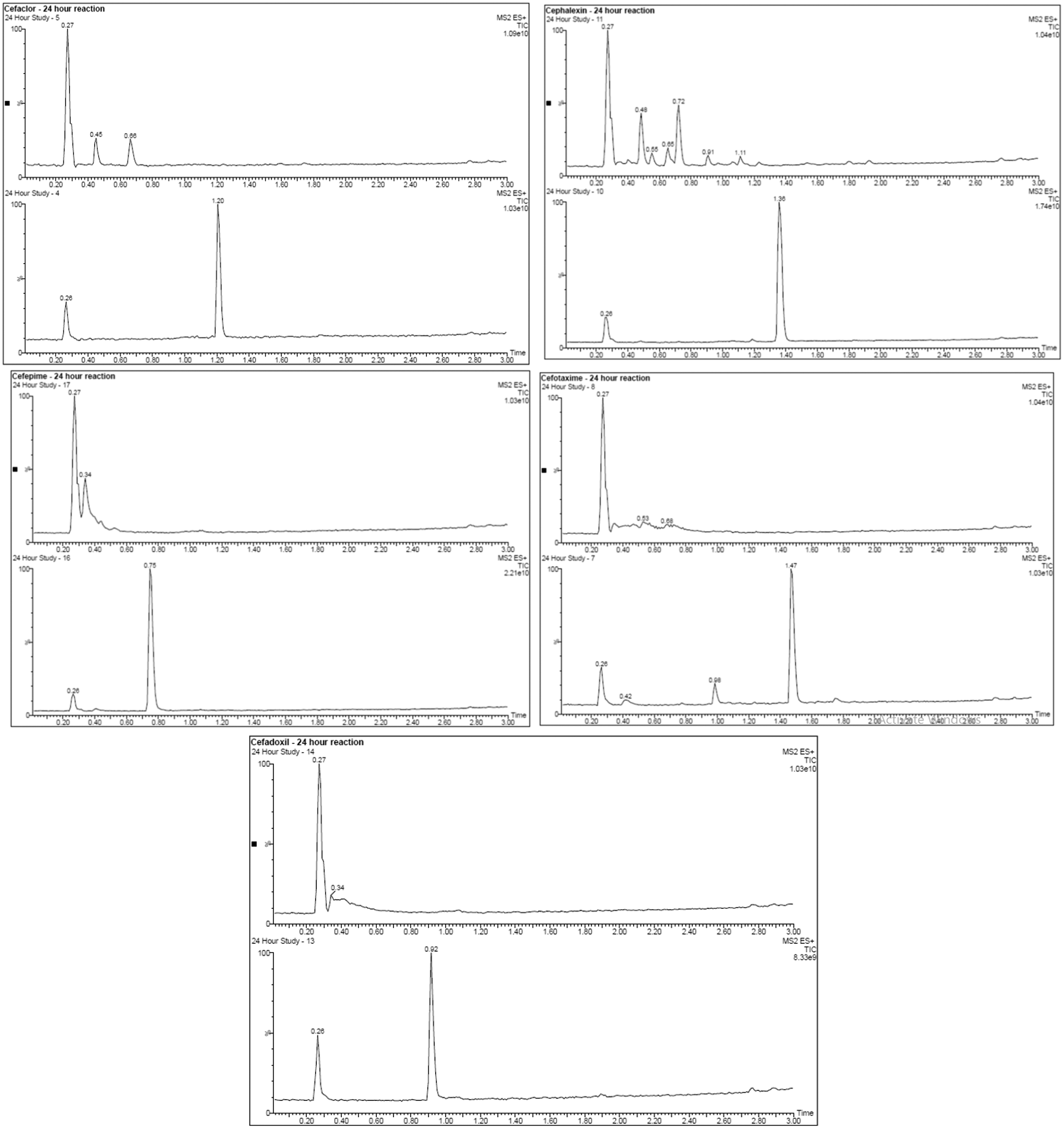
UPLC graphical data showing the breakdown of each cephalosporin compound after 24 h exposure (9600 ppm-h) to ClO2. For each compound, the bottom chromatograph shows the parent compound (untreated), upper chromatograph shows compound postexposure to dClO2 for each compound (treated). Row 1 Left = Cefaclor, Row 1 Right = Cephalexin, Row 2 Left = Cefepime, Row 2 Right = Cefotaxime, Row 3 = Cefadroxil. dClO2, dry chlorine dioxide gas; UPLC, ultra-performance liquid chromatography.
Showing details of the degradation results after exposure to 9600 ppm-h of dClO2, including the percentage of parent compound destroyed and the number and size of associated degradants
Biological Activity Testing
For ASDs exposed to dClO2, growth within 1 mm of the disk was recorded, highlighting any impregnated antibiotic or degradant as inactive (Table 5). Where ASDs were not exposed to dClO2, no growth within 3 mm of the disk was recorded, confirming that the impregnated antibiotic was active (see Supplementary Data S5 for organoleptic data). UPLC-MS/MS method development defined a LOR of 0.02 μg/disk for detection of cefaclor from the ASD (Table 6).
Antibacterial activity postgaseous decontamination of Cefaclor Disks
Growth, growth within 1 mm of disk; Nil, no growth within 3 mm of disk.
Gaseous dClO2 decontamination of preloaded cefaclor antimicrobial susceptibility disks
The starting quantity was 30 μg/disk. The remaining amount (μg/disk) is shown in column 3. Pass criterion was based on being below the LOR developed.
LC-MS/MS, liquid chromatography with tandem mass spectrometry.
Table 6 demonstrates that exposure to 9600 ppm-h of dClO2 successfully degrades the cefaclor present on the ASDs to acceptable levels, reaching below the LOR and achieving the “pass” criterion previously defined. The results demonstrated that dClO2 not only completely degraded the parent cephalosporin but also inactivated any degradant products (Tables 5 and 6).
Discussion
This study demonstrates that a gaseous chlorine dioxide cycle at 9600 ppm-h exposure provides effective decontamination for a broad spectrum of cephalosporins, providing the first definitive cycle of its kind and augmenting previous dClO2 studies regarding beta-lactam decontamination.17,18 This study also provides the first evidence that UPLC-MS/MS methods can be successfully applied for the detection of multiple generations of cephalosporins from common pharmaceutical surfaces and solutions, with detection limits similar to those reported in recent studies specific to penicillin G. 3
The use of a UPLC-MS/MS method provided a LOR of 0.002 μg/cm2 which could be consistently detected across all surfaces and cephalosporins tested, providing a promising detection limit for standardized acceptance criterion for cephalosporin decontamination. Such a limit would be in accordance with the current regulatory and cleaning validation methods used to ensure a safe environment for work.3,20
Phase 2 of this study provides the first investigation into the degradation products associated with ClO2 beta-lactam degradation, indicating that the ClO2 process not only degrades the parent compound, but also inactivates the biological activity of the remaining degradants. The analysis of the degradation in phase 2 was performed in a liquid solution. Consequently, the detection limits defined in phase 2 (<2 ppb for each compound) are comparable with and support previous studies that highlight the practicality of achieving detection limits of <2 ppb utilizing a standardized UPLC-MS/MS method for formulated materials. 3
This 2 ppb limit achieved during the degradation study is significantly below the 0.3 ppm threshold previously defined by the US FDA for penicillin G and amoxicillin, 18 while also being lower than the 5 and 50 ppb thresholds set for dairy products by the ECC and Veterinary Products by the UK VED, respectively. 18
Previous studies utilizing UPLC-MS/MS for detection of Cephalosporins in formulated material (at <2 ppb) have been restricted to Cefuroxime. 3 This study extends on this restriction, proving that UPLC-MS/MS could be equally applicable for cefaclor, cefalexin, cefotaxime, cefadroxil, and cefepime detection from formulated material, with detection limits similar to that seen for cefuroxime in a previous study. 3
As stated, previous studies have raised concerns about the potentially intact and active beta-lactam ring in the remaining degradants after decontamination. 21 Although UPLC-MS/MS is sensitive in detecting residual parent compounds, it does not indicate whether the degradants are still active. ASDs are widely used in diagnostic and drug discovery laboratories to assess the susceptibility of clinical bacterial isolates to antibiotics. 22 A potent antibiotic results in a significant zone of inhibition (no bacterial growth within 1 mm) around the ASD, whereas an ineffective antibiotic shows observable growth within 1 mm of the disk. 22
This study introduced a novel approach to determine the activity of residual degradants after dClO2 exposure by evaluating growth within the zone of inhibition. The dClO2-exposed disks showed no antibiotic activity, with E. coli growth occurring within 1 mm of the disks. Inactivation was supported by UPLC-MS/MS results demonstrating complete degradation of cefaclor on the disks to 0.02 μg/disk (LOR) after dClO2 exposure. These results provide the first evidence that the dClO2 decontamination method also inactivates any remaining degradant products, thus addressing the concerns raised in prior research. 21
Such an analysis has not previously been shown in other dClO2-related studies.17,18 We hypothesized that dClO2 could inhibit the activity of degradants by fracturing the beta-lactam ring, as reactive oxygen species have demonstrated this effect in previous studies.23,24 However, the results of phase 2 highlight that there was no uniformity in the degradant products across the cephalosporin compounds analyzed in this study. Therefore, even if beta-lactam cleavage is the cause of inactivation for cefaclor post-dClO2 exposure, this may not be the case for the other cephalosporin compounds studied.
Previous research has highlighted that degradation of various beta-lactam compounds can provide different degradant products, 23 which may or may not include an intact beta-lactam ring, after exposure to decontamination agents, including oxidizers. 23 Significantly more research is needed to pinpoint the inactivation mechanisms of beta-lactams post-dClO2 exposure if they are indeed inactivated.
In addition to the aims of this study, the results of phase 2 provide indicative evidence that ClO2 can also be used in liquid solutions to degrade cephalosporins, opening the door to a potential routine cleaning agent. Sodium hypochlorite has previously shown efficacy in cleaning cephalosporins, 14 however, it is harsh on materials and releases toxic by-products. 25 This study provides evidence that ClO2 could provide an alternative solution to sodium hypochlorite, which remains effective over a wider pH range and does not produce harmful by-products such as trihalomethanes and halo-acetic acids. 25 However, the evaluation of cleaning agents is beyond the scope of this study.
Conclusion
dClO2 and associated chromatographic and mass spectrometry validation provide a promising solution for cephalosporin compound decontamination, opening the door for facility repurposing for non-beta-lactam products. In addition to penicillin G, UPLC-MS/MS analytical testing provides a standardized solution for surface contaminant testing for cephalosporins, with a 0.002 μg/cm2 LOR providing a uniform acceptance threshold that holds promise as an industrial standard. The use of ASDs and associated diffusion tests to assess cephalosporin activity after dClO2 exposure could provide a useful tool to assess the biological activity of degradant products and the first CI that provides analytical, biological, and organoleptic information for beta-lactam contamination control.
Further research is required to assess whether the degradation of cephalosporins other than cefaclor yields similar biological activity results. Future studies including more beta-lactam compounds are required to assess dClO2's ability to provide a broad-spectrum method for beta-lactam facility repurposing.
Footnotes
Acknowledgments
We thank AMAL analytical for carrying out the analytical services required for the UPLC-MS/MS methods developed and used for this study.
Authors' Contributions
R.T.J., B.C., and A.H. contributed to the design and implementation of the research, analysis of results, and writing of the article. R.T.J. and S.R. conceived the original research and supervised the project. J.M., A.H., and C.W. contributed to the practical laboratory work.
Ethical Compliance
Ethical approval was not sought for this study because no human participants were involved in the study.
Authors' Disclosure Statement
R.T.-J. is employed by Serosep UK. B.C. and A.H. are employed by Biosafety International. All other authors have no conflict of interest with the research work carried out.
Funding Information
The authors thank Serosep UK and Biosafety International Pty for financial support for this research project.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
