Abstract
Background:
Containment Level 3 (CL3) laboratories may require boundary integrity testing. Existing guidelines for CL3 room leakage are often subjective, lacking a definitive standard for what constitutes a “sealed” room.
Methods:
This study reviews global biocontainment guidelines and standards, and it compares multiple test results from global CL3 facilities by standardizing test data to an equivalent test pressure of 250 Pa.
Results:
Our analysis revealed that 55% of rooms constructed using typical CL3 methodologies met the proposed testing criteria. The United States Department of Agriculture (USDA) Agricultural Research Service (ARS) greenhouse leakage rate acceptance criterion of 0.139 L/s per square meter (0.027 cfm per square foot) at a room differential pressure of 250 Pa was found to be a challenging, yet achievable standard.
Conclusions:
A two-step process is recommended for boundary integrity testing: (1) Initial leaks are identified using smoke pencil or soap bubble tests, followed by necessary repairs; (2) The room is then subjected to quantifiable leakage rate testing to verify it meets minimum requirements. In the absence of definitive local guidelines, we recommend the published ARS greenhouse leakage rate at a room differential pressure of 300 Pa of 0.152 L/s per square meter of surface area as an acceptable criterion for testing construction boundaries of CL3 laboratories built using current CL3 construction practices. For primary containment CL3 rooms, a more stringent criterion following the German Verein Deutscher Ingenieure guidelines at a room pressure differential of 250 Pa is noted as 0.03620 L/s per square meter of room surface area is more appropriate.
Introduction
Biological research, diagnostic, and bio-response laboratories and facilities that handle infectious agents can mitigate the risk of a potential agent release by performing boundary integrity testing, also referred to as leak/air tightness testing.
Boundary integrity testing of the laboratory space for biological containment facilities achieves three primary objectives:
To minimize the potential for release of aerosolized infectious agents to the general public and staff, as well as minimize cross-contamination of multi-agent use facilities or within larger facilities with varied program requirements within the same biological containment facility (e.g., during a simultaneous occurrence of a spill and a ventilation system malfunction causing room positive pressurization). To minimize the migration of fumigants (decontamination gases) into adjacent spaces within the building. To verify construction quality of the space and whether it is sufficient for withstanding the maximum design pressure limit without compromising the structural integrity of the Containment Level 3 (CL3) containment boundary.
For level 3 agriculture (CL3 Agriculture [CL3-Ag], Biosafety Level 3 Agriculture [BSL-3Ag], Specified Animal Pathogens Order [SAPO]-4) and containment level 4 (CL4, BSL-4, Advisory Committee on Dangerous Pathogens [ACDP]-4, Physical Containment [PC]4) spaces, industry-accepted standards are well defined, and the purpose of this report is to focus on boundary integrity testing methods for CL3 (BSL-3, SAPO-3, ACDP-3) laboratories. For the purposes of this report, the acronym for CL3 laboratories will be used and is directly comparable to other international acronyms (BSL-3/Animal Biosafety Level 3 [ABSL-3], ACDP-3, SAPO-3, PC3, etc.).
Most industry testing strategies and acceptance criteria for CL3 room leakage are often subjective rather than definitive. This paper is not necessarily intended to be adopted by guidelines or standards but is a review of current guidelines and standards in the industry, an analysis of case studies, and proposes a testing methodology for new construction of CL3 laboratories. The aim is to ensure that an acceptable level of quality is achieved in newly constructed CL3 facilities using common CL3 construction techniques such as gypsum board and concrete masonry unit (CMU).
Guidelines and Standards
The only definitive testing criteria for CL3 spaces from the guidelines and standards reviewed are from the Australia/New Zealand standards and German Verein Deutscher Ingenieure (VDI) standards. Requirements from other standards and guidelines are included for reference. Where applicable, we have taken criteria defined within the guidelines and converted it to an equivalent range of acceptance at a room differential pressure of 250 Pa (1″ of wc) to allow for a cross-guideline comparison.1,2
Australian Standards/New Zealand Standards 2243.3–2022—Safety in Laboratories—Part 3: Microbiological Safety and Containment
Appendix F of Australian Standards/New Zealand Standards (AS/NZS) 2243.3 “Recommendations for achieving acceptable room airtightness” states a prescriptive recommendation for maximum air leakage: “The recommended maximum leakage rate, β, for PC3 and PC4 laboratories is 10−5, at a test pressure of 200 Pa (Figure F.1 and Clause F7). This corresponds to a leakage rate of 120 L per minute, or 2.0 L per second at this test pressure.”
AS/NZS 2243.3 also makes an exception, “It is not recommended that a laboratory be designed for gaseous decontamination if the leakage rate exceeds 10−4 at 200 Pa differential pressure, without specialist advice. A leakage rate of β = 10−4 corresponds to a leakage rate of 1200 l per minute, or 20.0 l per second at this test pressure.”
Given the exception, the 10−5 criteria is the AS/NZS standard; however, a higher leakage rate may be acceptable for rooms that are not exposed to frequent fumigation. Rooms may be permitted to have a leakage rate as high as 10−4 (20 L/s) at 200 Pa differential pressure (42 cfm at 0.8″ of wc) if precautions are taken during decontamination to ensure that risks of exposure to fumigants are minimized in adjacent spaces.
Therefore, per the AS/NZS, the recommended range of leakage from a CL3 laboratory is 2 L/s (4.2 cfm) to 20 L/s (42 cfm), with a room differential pressure of 200 Pa (0.8″ wc). For comparison purposes in this report, the equivalent range of acceptance at a room differential pressure of 250 Pa is calculated at 2.2 L/s (4.7 cfm) to 22.4 L/s (47.5 cfm). 1
VDI Guidelines Tightness of Containments VDI-2083 Part 19–2018—Germany
This guideline provides criteria for room tightness classes for cleanrooms, isolator areas, and containment spaces. For CL3, it recommends that a class 4 criteria be used at a rate of 0.03620 L/s per square meter (0.007 cfm per square foot) of room surface area at a pressure differential of 250 Pa (1″ wc). Also, for CL4 spaces, the guideline recommends that a class 5 criteria be used at a rate of 0.01205 L/s per square meter (0.002 cfm per square foot) at a pressure differential of 250 Pa (1″ wc). Refer to the publication for the equivalency table for acceptable air permeability rates of different classes at multiple differential test pressures. 2
Canadian Biosafety Standard (3rd Edition) 2022
Paragraph 5.2.8 in the Canadian Biosafety Standard (CBS) identifies minimum testing requirements of containment boundaries for CL2-Ag zones and all CL3–CL4 zones as follows: “Where inward airflow is required, integrity of the seals of penetrations through the containment barrier of the containment zone, animal cubicle, and PM room to be tested with a smoke pencil or other aid that does not influence the direction of airflow.”
Paragraph 5.2.13 in the CBS identifies the testing requirements for CL3-Ag and CL4 zones as follows: “Integrity of the containment barrier, including ductwork and filter housings, to be verified by room pressure decay testing. Acceptance criteria include two consecutive tests with a maximum loss of 50% of maximum operational pressure over a 20-minute period.” The CBS references the Canadian Biosafety Handbook 2nd edition (2016), in which chapter 10 specifies a starting pressure of negative 500 Pa for pressure decay testing.
There is no guidance on an acceptable leakage rate for CL3 spaces. 3
US Department of Health and Human Services—Biosafety in Microbiological and Biomedical Laboratories—6th Edition—2020, USA
Published by the United States Department of Health and Human Services, the Centers for Disease Control and Prevention, and the National Institutes of Health, the Biosafety in Microbiological and Biomedical Laboratories is considered by many to be an international standard for biological containment facilities. However, for CL3 boundary integrity testing, it simply states, “Seams, floors, walls, and ceiling surfaces should be sealed. Spaces around doors and ventilation openings should be capable of being sealed to facilitate space decontamination.”
There is no guidance on an acceptable leakage rate for CL3 spaces. 4
Agricultural Research Service 242.1–2012—Agricultural Research Service Facilities Design Standards, USA
The Agricultural Research Service (ARS) Facilities Design Standard's criteria for testing of leakage are only provided for BSL-3Ag zones and are identical to the test identified in the CBS. Acceptance criteria are outlined in paragraph 9B-4.E as, “Two consecutive pressure decay tests demonstrating a minimum of 1″ wc (250 Pa) negative differential pressure remaining after 20 minutes, from an initial negative pressure differential of 2″ wc (500 Pa).”
In addition, CL3-Ag (BSL-3Ag) containment for greenhouses is separately identified in paragraph 9B-6 as “The test pressure difference will be 6.24 pounds per square foot positive static pressure (300 Pa); the allowable leakage rate is 0.03 cfm per square foot” (0.152 L/s per square meter). For comparison purposes within this report, the equivalent allowable leakage rate at a room differential pressure of 250 Pa is 0.139 L/s per square meter (0.027 cfm per square foot) of room surface area.
There is no guidance on an acceptable leakage rate for CL3 spaces.
Note that this guideline is currently under review for a future update. 5
World Health Organization—Laboratory Biosafety Manual Fourth Edition—2020
The World Health Organization manual, often used in developing countries lacking their own standards, states for boundary testing of CL3 facilities: “Where gaseous disinfection is selected as a heightened control measure for decontamination and waste management, laboratory room/space airtightness will need to be enhanced. This is achieved by sealing all surfaces and/or laboratory penetrations to prevent the escape of hazardous gases.”
There is no guidance on an acceptable leakage rate for CL3 spaces. 6
American National Standards Institute/American Society of Safety Professionals Z9.14–2020, USA
The American National Standard Testing has published a recent standard, “Testing and Performance-Verification Methodologies for Ventilation Systems for Biosafety Level 3 (BSL-3) and Animal Biosafety Level 3 (ABSL-3) Facilities.”
Section 8.4.8.2 states, “When required by the facility risk assessment, room tightness (room air-leakage test) shall be performed before initial operation, periodically thereafter (as determined by the facility risk assessment and SOP) …” While no definitive testing criteria is stated, it does identify that the boundary is to be confirmed using smoke or soap bubbles with room pressures ranging from normal operating pressures to higher values (e.g., twice the normal value) as appropriate. It also indicates that additional test methods may be appropriate for initial commissioning of new construction and/or renovations, such as:
Operating the room at higher differential pressure values for the smoke/bubble tests (e.g., up to 2″ wc [500 Pa]): Testing at this pressure may be useful in conjunction with ventilation controls testing.
“Room porosity” tests using airflow/pressure testing equipment similar to equipment used for duct-leak testing.
There is no guidance on an acceptable leakage rate for CL3 spaces. 7
Health and Safety Executive, United Kingdom
The Health and Safety Executive (HSE) published a document titled, “Sealability of Microbiological Containment Level 3 and 4 Facilities,” which requires that, for CL3 and CL4 facilities, the workplace is to be sealable to permit disinfection. The sealability test methods identified include:
Smoke testing is the main method, with no test criteria identified. They also go on to state, “Recent research has indicated that the use of smoke plume-generating devices is an inherently sensitive method. However, the limits of its sensitivity are heavily dependent upon the skill of the person performing the test.”
Room pressure decay testing, which requires the room to be held at a specified negative pressure relative to atmosphere for a prescribed period and measurement of any loss of the pressure differential at regular intervals during that time. The leak rate must then be compared with a predetermined acceptance value. A reference to Canada's pressure decay testing methodology is also made.
In addition, there is a pointed note that applies to all responsible owners that deal with room disinfection that states, “Therefore, regardless of whether the fumigant of choice is formaldehyde, hydrogen peroxide or some other chemical, the legal requirement for room sealability remains and it is the responsibility of the duty holder to ensure the sealable status of the facility.”
There is no guidance on an acceptable leakage rate for CL3 spaces. 8
ACDP—2018, United Kingdom
The HSE “Management and Operation of Microbiological Containment Laboratories” identifies the following requirements:
[28] “At CL3, the laboratory must be capable of being sealed to allow it to be effectively disinfected. An ongoing programme of formal assessment, e.g., an annual test is recommended to make sure sealability is maintained. However, more frequent visual inspections should be undertaken, e.g., for cracks, or dust trails, which may provide early indication of breaches in sealability of the facility. In this event, remedial work should be carried out and verified as effective.”
[31] “Sealability is usually carried out with the laboratory operating at normal working pressure and leaks are detected by observing any deviation of the smoke plume (Figure 13) and should be performed by a competent person.”
There is no guidance on an acceptable leakage rate for CL3 spaces. 9
Merrick Industry Best Practice
In the absence of local standards or guidelines for CL3 leakage, Merrick has coordinated with internal biosafety professionals and has often specified (based on experience and lessons learned) an acceptable leakage rate of 2% of room air volume per minute at a room differential pressure of 500 Pa (2″ of wc). For comparison purposes within this report, the equivalent allowable leakage rate at a room differential pressure of 250 Pa (1″ of wc) is 1.41% of room volume per minute.
Guideline Comparisons
The graphic given next illustrates the wide variance in test criteria. Note that the only defined test criteria for CL3 laboratories are the AS/NZS guidelines and German VDI. ASHRAE 90.1 (whole building envelope leak testing) and the National Sanitation Foundation/American National Standards Institute 49 (biosafety cabinet leak testing) standards are not relevant to CL3 construction but are shown to illustrate extreme limits (Figure 1).10,11
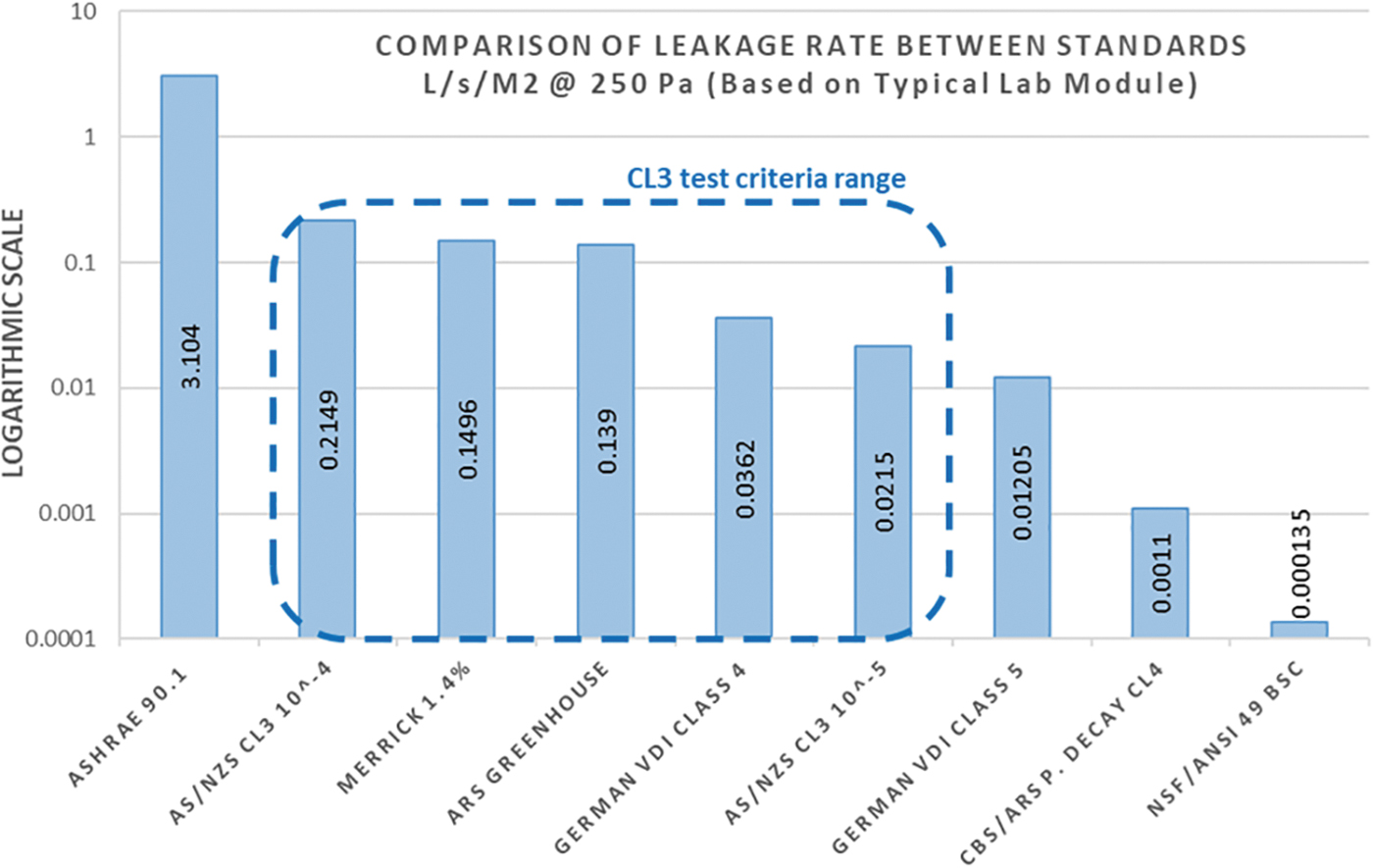
Logarithmic comparison of acceptable leakage rate limits between standards normalized to a dP of 250 Pa; Based on a typical lab module of 6.7 m(L) × 3.3 m(W) × 3.0 m(H). ARS, Agricultural Research Service; ASHRAE, American Society of Heating, Refrigerating, and Air-Conditioning Engineers; AS/NZS, Australian Standards/New Zealand Standards; CBS, Canadian Biosafety Standards; CL3, Containment Laboratory 3; NSF/ANSI, National Sanitation Foundation/American National Standards Institute; VDI, Verein Deutscher Ingenieure (Association of German Engineers).
Leakage Criterion Discussion
Containing Aerosols
CL3 laboratories typically rely on directional airflow to contain potentially infectious agents, by supplying less air into the space than what is being exhausted (extracted) from the space. CL3 spaces are not usually primary containment areas; work involving aerosols or animals is typically conducted within self-contained apparatuses, minimizing the risk of aerosol presence in the general CL3 environment.
Containing Fumigant (Decontamination Gases)
The applications of expensive welded metal enclosures or monolithic concrete is not required to achieve appropriate sealability for CL3 applications. Many construction techniques can effectively contain fumigants during a room decontamination process, often utilizing controlled pressure differentials to adjacent spaces.
Standard stud and drywall construction—with appropriate coatings, sealed joints at floors, ceilings, service openings, and door frames—has been successful. Fumigation standard operating procedures (SOPs) need to be considered to maintain fumigant concentration and minimize exposure risk to adjacent spaces. The room(s), being decontaminated, are commonly isolated from the rest of the facility, and thus consideration should be given to control the building ventilation system by extracting a very small amount of air from the space during decontamination to maintain the room at a negative pressure compared with adjacent spaces.
Construction Quality
The term “sealed” is often undefined in guidelines, leading to disputes during construction. Other guidelines require smoke pencil testing, but even that is difficult to quantify since interpretation is highly subjective of the person doing the testing.
Some guidelines have more prescriptive requirements, such as those for Australia/New Zealand, which specify a fixed rate; Canada and the United States Department of Agriculture (USDA), which specify a volumetric leakage rate for CL3-Ag; the USDA greenhouse requirements, which specify a limit on leakage per surface area; and the German VDI guidelines, which specify a limit on leakage per surface area.1–3,5
To avoid conflicts during construction and with testing officials, best practice involves a combination of tests, including smoke pencil or soap bubble tests, followed by a quantifiable leakage test. The leakage test should be specific, easily quantifiable, allow the contractor to be held liable for achieving the set parameters/standard, and easily repeatable in future testing.
Given that aerosol and fumigant (decontamination gases) containment can be effectively managed with well-designed SOPs, the acceptable leakage criterion for CL3 laboratories does not need to be extreme. They should, however, be achievable and repeatable, aligning with current construction practices for CL3 laboratories, which often consist of gypsum board or CMU.
Pressure Decay Versus Airflow Measurement
Facility leakage tests and pressure decay tests are similar tests, as they both measure the amount of air that escapes or enters a space under pressure. Airflow movement measures the amount of air leaking into or out of a room to maintain a pressure, while the pressure decay testing records pressure changes as air volume in the room changes due to pressurization and flow through the leak points.
For practicality, airflows less than 2 L/s (4 cfm) are difficult to accurately measure and control, while for most CL3 rooms air leakage greater than 2 L/s (4 cfm) result in a pressure decay rate that is relatively short and only lasts a few seconds. Instrument sensitivity should be considered to ensure accurate measurement of expected leakage rates.
Fixed Rate Versus Volume Versus Area
A fixed leakage rate may not be the optimal solution for determining acceptable leakage compared with criteria that incorporate room geometry. To illustrate, consider the following lab module examples that highlight the differences between three geometries:
A typical lab module of 6.7 m(L) × 3.3 m(W) × 3.0 m(H) has a volume of 66.33 m3, and a surface area of 104.22 m2.
A double long lab module of 13.4 m(L) × 3.3 m(W) × 3.0 m(H) is twice as long as a typical lab module, and it has a volume of 132.66 m3, and a surface area of 188.64 m2.
A double wide lab module of 6.7 m(L) × 6.7 m(W) × 3.0 m(H) is twice as wide as a typical lab module, and it has a volume of 134.67 m3, and a surface area of 170.18 m2
Figure 2 given next indicates the minimum fixed leakage of 2.24 L/s (4.7 cfm) when subjected to a room differential pressure of 250 Pa (1″ of wc). For lab modules of different geometry identified, this fixed leakage rate is compared with the equivalent leakage per unit area, leakage of %volume per minute, and an equivalent pressure decay rate in seconds (from initial test pressure to 1/2 its pressure).
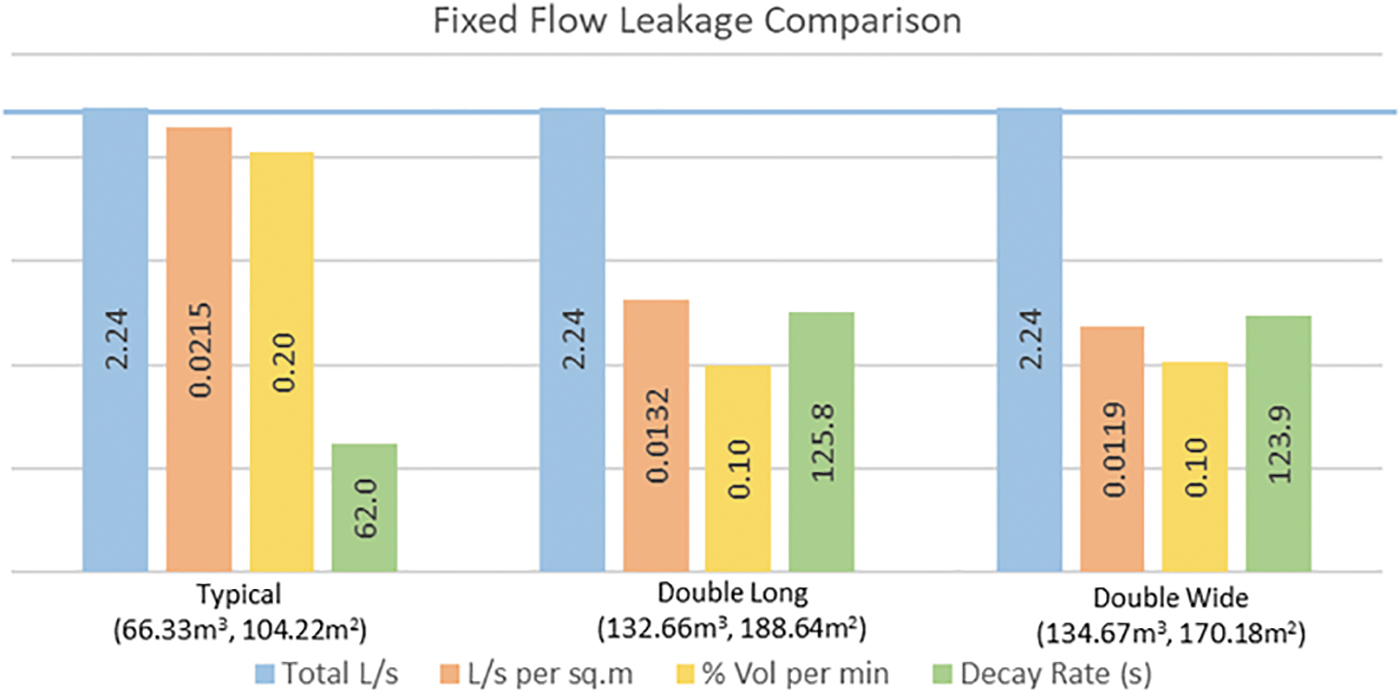
Fixed flow rate: AS/NZS minimum leakage comparison.
From the figure, we see that comparable leakage per unit area and leakage of percent volume per minute are essentially double for a typical lab module that is half the size of the other two lab modules. If all the lab modules have the same construction materials and techniques, then it is difficult to justify why a room that is twice the volume must be twice as tight in construction to achieve an acceptable result.
Quantifiable Testing Results
Background
The following examples are of real CL3 laboratories around the world where Merrick has compiled quantifiable leak testing results. These results are benchmarked against different guideline testing requirements of the AS/NZS total leakage of 22.4 L/s, the ARS Greenhouse standard of 0.139 L/s per square meter of room surface area, the Merrick standard of 1.41% of room volume per minute, and the German VDI standard of 0.036 L/s per square meter of room surface area all at a differential pressure of 250 Pa. Since testing at all facilities was at slightly different pressures, all data are modified using the orifice equations and ideal gas laws to equate comparable leakage rates at 250 Pa, and all units of measure are presented in metric.1,2,5
Australian Study
In 2009, Gordon B. McGurk published “A Study of Airtightness in Australian High-level Bio-containment Facilities” in the ABSA journal, Applied Biosafety. This study included leakage testing comparisons to the Australian standards for 18 different PC3 (CL3) facilities in Australia. Two facilities could not be successfully tested due to structural integrity. 12
We analyzed the 16 Australian facilities, compared accepted leakage rates based on the AS/NZS 2243.3 standard, and calculated flow per square meter of room surface area, percent of room volume of leakage per minute at equivalent room differential pressures of 250 Pa, and pressure decay rate from 500 to 250 Pa. Of the 16 facilities tested, only 10 met the minimum Australian criteria of 22.4 L/s when measured at a differential pressure of 250 Pa, and only two facilities achieved the leakage rate of 2.2 L/s.
North American Facilities
Merrick tested and analyzed 3 different facilities in the United States and Canada. These facilities were constructed using various materials: CMU, single layer drywall on metal studs, and double layer gypsum drywall on metal studs. Leakage rates were calculated on flow per square meter of room surface area, percent of room volume of leakage per minute at equivalent room differential pressures of 250 Pa, and pressure decay rate from 500 to 250 Pa.
Facility Summary
The rooms tested within the facilities were tallied with pass rates identified for each of the three comparative leakage rates at a room differential of 250 Pa (1″ wc). The results are shown in Figure 3 given next.
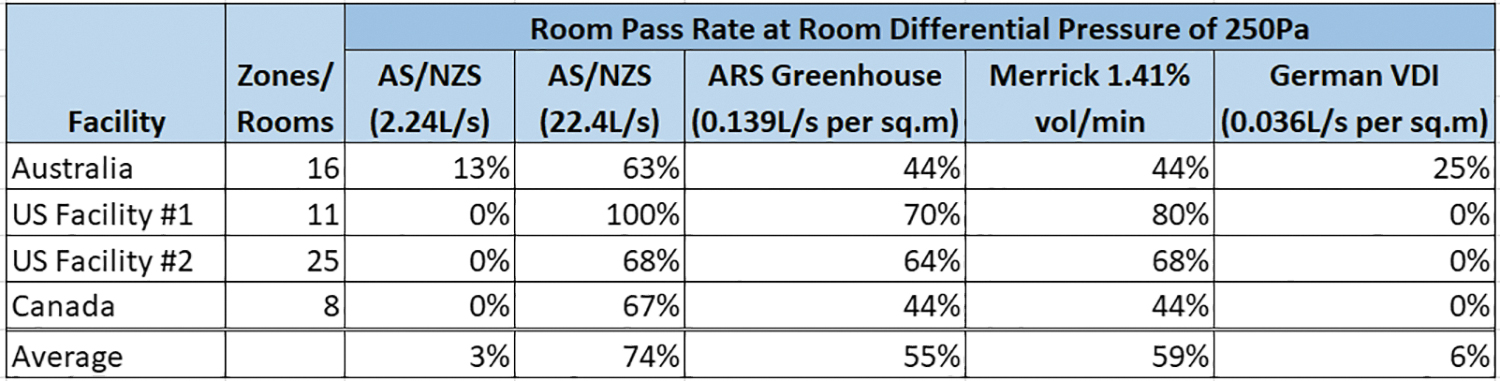
Leakage pass rate comparisons across projects.
Note that none of the earlier mentioned facilities in the United States or Canada passed the German VDI standard, and yet all the facilities reported are acceptable to national biosafety regulations and meet national requirements to operate at CL3. Additional tests confirm that fumigation of these spaces does not adversely affect adjacent areas. This suggests that the German VDI standard may be more stringent than is required for typical CL3 construction.
The ARS greenhouse leakage rate acceptance criteria of 0.139 L/s per square meter (0.027 cfm per square foot) or room surface area at a room differential pressure of 250 Pa (1″ of wc) seems to provide a room integrity test standard that is challenging but achievable. Fifty-five percent of the rooms tested pass the criteria with typical CL3 construction methodologies, including single and double wall gypsum and CMU.
The Merrick best practice rate of 1.41% of room volume at a room differential pressure of 250 Pa appears to offer similar challenging but achievable test results with 59% of rooms tested passing the criteria with the same CL3 construction methodologies.
Although not all rooms tested pass the ealier suggested criteria, it is important to note that such requirements were not originally specified and thus there was no reason for the contractor to apply better quality control techniques to achieve improved performance.
We also note that for rooms that are primary containment but still rated as CL3 (not CL3Ag or CL4), a risk assessment should be undertaken to validate acceptable leakage rates. Such spaces may have an increased requirement to contain potentially aerosolized agents and will likely see more frequent room fumigations occurring several times per year. Under these circumstances, consideration should be given to higher quality construction, and increased testing criteria such as that identified by the German VDI Guidelines.2,5
Recommendations
Industry standard construction materials and methods for CL3 Laboratories around the world typically use gypsum board or CMU with impermeable coatings. Achieving room pressure differentials above 250 Pa can be challenging with such construction, making it necessary to test at lower pressures. At the same time, pressures less than 125 Pa can yield inaccurate leakage rates and are not recommended. If the structure can handle the pressures, it is recommended to test the spaces at a minimum of 250 Pa (1″ of wc) or greater, and at positive or negative pressure differentials, or both.
To ensure that an acceptable level of quality is achieved in newly constructed CL3 facilities, the following two-step testing procedure is recommended:
Qualitative boundary testing: Identify initial leaks using smoke pencil or soap bubble tests and seal all visible leaks.
Quantitative boundary testing: Verify the room meets minimum leakage requirements through quantifiable leakage rate testing.
This procedure avoids several problems common to CL3 construction and testing practices and ensures a better end-product for the user:
Sole reliance on visual methods may lead to contractors overlooking small leaks while fixing easily identifiable, larger leaks.
The effectiveness of smoke pencil and soap bubble tests is subjective to the operator's skill level.
Quantitative testing alone might leave small holes that would be found with visual indicators, such as smoke pencil or soap bubbles.
Testing is repeatable at construction acceptance and to verify construction integrity is maintained on an annual basis or after major renovations or repairs.
In the absence of definitive local guidelines, we recommend the published (or equivalent) ARS greenhouse leakage rate at a room differential pressure of 300 Pa (1.2″ of wc) of 0.152 L/s per square meter (0.03 cfm per square foot) of surface area as an acceptable criterion for testing construction boundaries of CL3 laboratories built using current CL3 construction practices.
For rooms that serve as primary containment but are still rated as CL3 (not CL3Ag or CL4), or for CL3 spaces that may have an increased frequency of room fumigations, a risk assessment is advisable to validate acceptable leakage rates such as the more stringent requirements of the German VDI guidelines. The German VDI guidelines' acceptable leakage rate at a room pressure differential of 250 Pa (1″ of wc) is noted as 0.03620 L/s per square meter (0.007 cfm per square foot) of room surface area. Due to the low acceptable airflows under the VDI standard, precise instruments such as a rotameter are recommended for accurate measurements.
In light of these guideline considerations, it is pertinent to note that CL3 facilities are unique and coordination with the end users and/or completion of a risk assessment is recommended to develop an appropriate criterion for each individual facility.
The tables in Figure 4 given next identify both the ARS greenhouse flow per unit area recommended leakage equivalence for different room differential pressures, with the yellow highlight as the “actual published value,” and the VDI flow per unit area recommended leakage equivalence for different room differential pressures.2,5
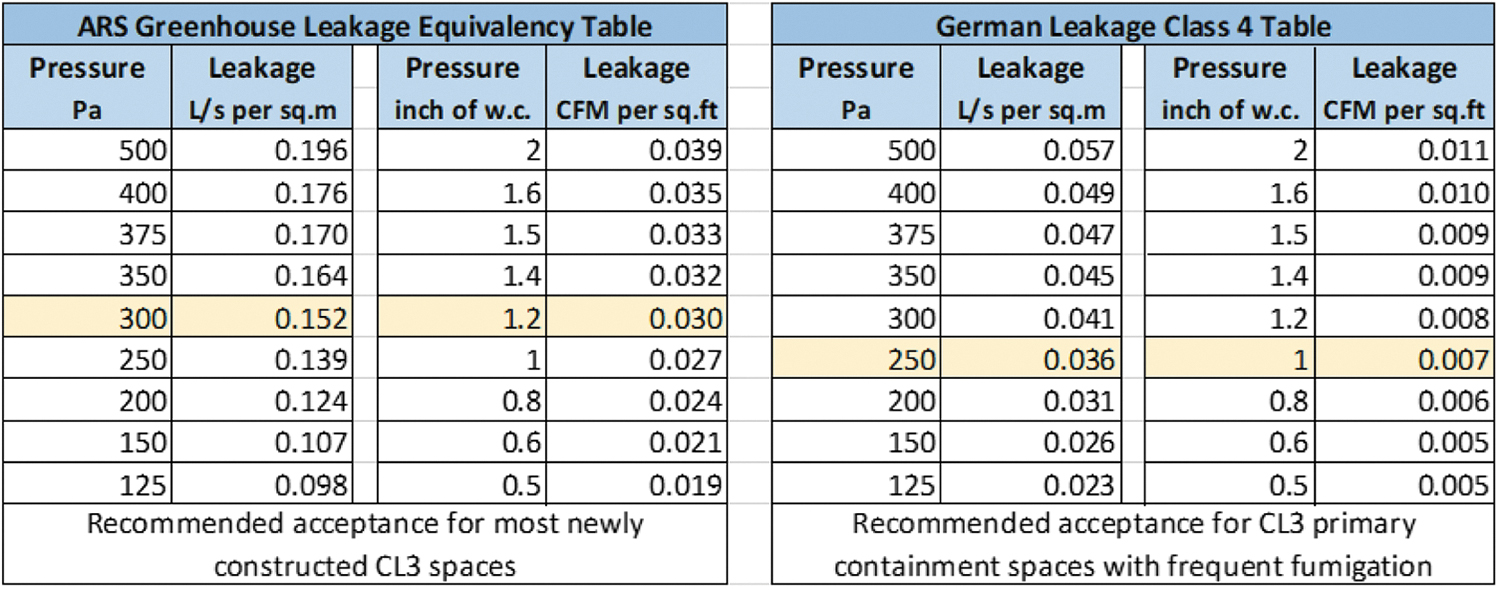
ARS Greenhouse and German VDI leakage criteria tables.
Integrity Testing Recommended Procedure
Room Preparation
• Fill all P-Traps with water.
• Seal ventilation openings with plastic and tape. If provided, use bubble-tight dampers in ductwork.
• Close and seal all doors with plastic and tape.
• Door Plate Installation (If using)
○ Must be installed on containment perimeter such that applied test pressure pulls/pushes door plate against door stop and allows for door frame to be part of leak test.
○ Door closers and any bump-stops located on door stop surface must be removed before fitting the door plate.
• Provide a calibrated digital manometer installed across the containment perimeter in a manner to minimize interference with wind or ventilation turbulence and to accurately represent the interior and exterior differential air pressure.
• Provide a portable fan to be used to achieve required test pressures in room.
○ Provide all adapters, fittings, bubble tight isolation valves, and flow monitoring stations required to perform testing as defined herein
Step 1 (Qualitative Boundary Testing)
Use a portable pressurization fan to pressurize room to a negative pressure (500 Pa [2″ of wc]) or maximum structural tolerance pressure. Use fan controls to maintain room at a steady negative pressure through the duration of smoke pencil/soap bubble testing. Be careful not to over pressurize the room such that structural damage may occur.
Use a smoke pencil and/or a liquid detergent for soap bubble testing at locations that could be point sources of air leakage.
Note that a room positive pressure is desirable for smoke pencil testing to easily see the smoke escaping through pin holes, while a room negative pressure is desirable for soap bubble testing to easily see bubbles forming on air being drawn in through pin holes.
Identify all relevant leak points for repair.
Repair any leaks found through a smoke pencil and/or soap bubble testing.
Repeat tests identified in step #1 earlier.
Acceptance Criteria: No visible leaks found after repairs completed.
Step 2 (Quantitative Boundary Testing)
Use a portable pressurization fan to pressurize room to a negative pressure (recommended 250 Pa [1″ wc]). Use fan controls to maintain room at a steady negative pressure through the duration of leakage testing. Be careful not to over pressurize the room such that structural damage may occur.
Record/trend room pressure, temperature, and airflow over a 10-min period at 5-s intervals.
Meters to have appropriate range and accuracy for variables being measured.
If temperatures are not constant due to equipment in the space, temperature trend data may be used to calculate equivalences.
Acceptance Criteria: Leakage rate is less then pre-determined quantifiable leakage rate (Figure 5).
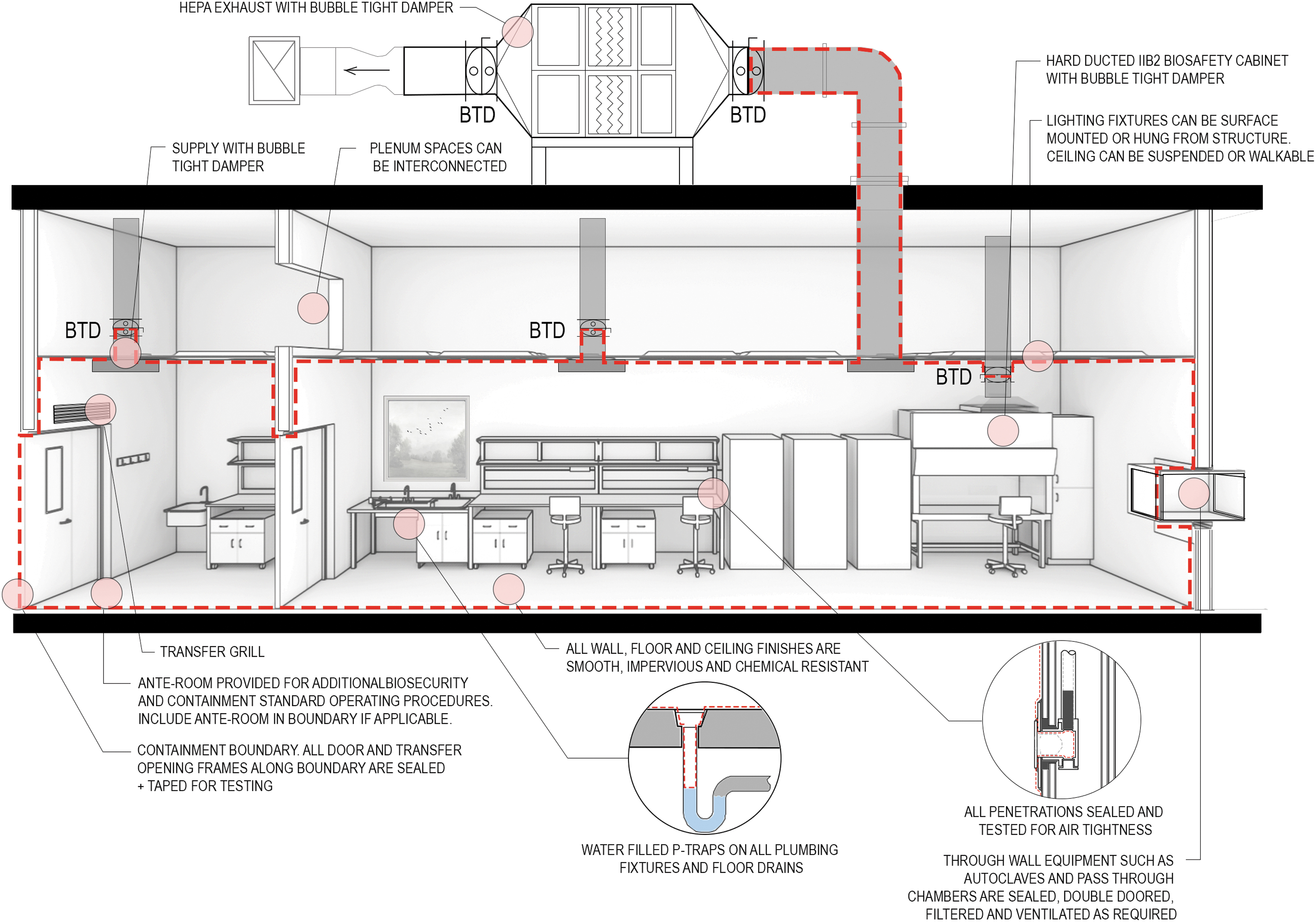
Example CL3 room testing boundary. BTD, Bubble Tight Damper; HEPA, High Efficiency Particulate Air.
Footnotes
Acknowledgments
The authors wish to thank Rachel Gamble, DrPH; Chris Kiley, PE; Draven Whitaker, PE; and Kate Worsham for their valuable contributions in reviewing and editing the article, and Ramon Renderos Soto for his assistance with visualization.
Authors' Contributions
C.Z. and G.T.: Conceptualization, data curation, methodology, validation, visualization, writing—original draft preparation, and writing—review and editing.
Ethical Approval Statement
The authors declared that no animals were used in this study.
Statement of Human and Animal Rights
The authors declare that no human subjects or animals were involved in generating data for this article.
Data Access Statement
The majority of the data used in this study is derived from pre-published information sources. Only the Merrick data/examples are new and can be made available upon request.
Authors' Disclosure Statement
The authors declares that there are no conflicts of interest.
Funding Information
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
