Abstract
Introduction:
The current global situation with increasing zoonotic transmissions of pathogens, rapidly changing ecosystems due to the climate change and with it the distribution of potential vectors, demands new ways of teaching and educating students in the field of infectious disease research.
Methods:
The international master program “Infectious Diseases and One Health—IDOH” started its second application period in 2019. Biosafety is an integral part of IDOH, exemplified by a biosafety level 3 hands-on training at the Animal Health Research Center IRTA-Centre de Recerca en Sanitat Animal (CReSA), Barcelona. At Hanover Medical School, biosafety is expanded to a comprehensive biorisk management approach with focus on risk assessment, bioethics, and training in a mobile containment laboratory. This article illustrates in depth the intention and the individual teaching units of the biorisk management module within the third semester of the IDOH master. Risk assessment is taught based on the new WHO Laboratory Biosafety Manual 4th edition, which represents a paradigm shift toward a risk-based approach instead of a prescriptive definition of biosafety levels. This methodology will enable the international IDOH students to cope with different national legislations and to provide guidance on biosafety in their home countries. In the final unit, the students operate a mobile containment laboratory and simulated a viral hemorrhagic fever outbreak in Africa.
Conclusion:
In sum, this module combines theoretical risk assessment and its practical implementation in the mobile laboratory as a future direction for training infectiologists. In addition, our report may serve as a blue print for others to amend their education with the herewith mentioned pillars of biosafety teaching.
Introduction
Biorisk Management
The past years have seen a significant increase in the emergence of zoonotic infections followed by epidemics or even pandemics. A few examples are the recent SARS-CoV-2 pandemic, the Ebola epidemic affecting Guinea, Sierra Leone, and Liberia in 2014–2016, and Chikungunya and Zika 2016 in the Americas.
In addition, climate change increases the occurrence of virus-transmitting vectors, for example, in Germany. The first mosquito-borne infections with West Nile Virus (WNV) were reported in Germany in late summer 2019. It is to be expected that WNV will move north as a consequence of its permanent mosquito reservoir now established in the middle of Europe with seasonal occurrence of WNV disease cases. 1
Basic research on highly pathogenic agents in containment laboratories is mandatory to develop vaccines and novel antivirals as well as diagnostic methods to control outbreaks. However, they also pose a risk to the public in case those organisms may escape to the environment due to a laboratory incident, leaky containment, poor laboratory practices, or intentional removal and subsequent release by a terrorist attack.
An initial international approach to compile a comprehensive set of requirements for biorisk management was the CEN Workshop Agreement 15793:2008. 2 This consensus article achieved broad international agreement in subsequent years and was upgraded to ISO Standard 35001 “Biorisk Management for laboratories and other organizations” in 2019. 3 This allows biorisk management to be incorporated into existing quality management systems within a company.
The realization that technically oriented safety measures alone are not sufficient to prevent incidents even in modern containment laboratories 4 led to a rethinking of biosafety. As early as the 1980s, biosafety experts pointed out that not only the pathogen is crucial for the risk assessment of the associated work, but also the intended activities and also the competence of the personnel involved. 5 The difference risk factors for a qualitative risk assessment have been elaborated, 6 but also the approach of quantifying biological risks has been formulated in the Biosafety Risk Assessment Model (BioRAM) approach (see also BioRAM chapter 7 ). The 5-P-concept (pathogens, procedures, personnel, personal protective equipment [PPE], and place) directed attention to the establishment of a risk management approach. 8
Nevertheless, until recently, the predominant emphasis was placed on technical safety measures and the description of biosafety levels based on the risk groups of the organisms (DIRECTIVE 2009/41/EC of the European Parliament, World Health Organization (WHO) laboratory manual (LBM) 3rd edition 2004). With the 4th edition of the WHO LBM 2020, 9 a paradigm shift in the evaluation of biosafety has occurred. The biorisk management approach herein establishes biosafety measures based on risk assessment and hazard evaluation of the activities involved.
Education on Biorisk Management
Training opportunities for biological high-containment laboratories are elaborate and usually designed for scientists. For students, such educational exercises are rare. The European Commission's chemical, biological, radiological, and nuclear (CBRN) action plan 2009 calls on member states to develop education and training concepts, including best practices for higher education for biosafety, raising awareness of possible misuse of data and biological agents and toxins, and bioethic issues for undergraduate, graduate, and doctoral students. 10
The University of Göttingen (Germany) first implemented an EU-funded biosafety level 3 (BSL-3) biosafety and biosecurity training for scientists and staff in 2009. 11 Hannover Medical School (MHH, Germany) joined in and served as co-coordinator for the second funding period. Overall, this initiative also created a continuous platform for the exchange of experiences on biosafety issues between scientists.
From 2019 on, the MHH has been participated in the EMJMD “International Master of Science in Infectious Diseases and One Health (IDOH+).” It is a 2-year master program (https://www.infectious-diseases-one-health.eu), taught in English and delivering 120 European Credit Transfer System. It is a joint master of the Université de Tours (UT, France), the Universitat Autònoma de Barcelona (UAB) in Spain, and the Medizinische Hochschule Hannover (MHH) in Germany (Table 1).
The Infectious Diseases and One Health program and the individual modules held in the participating institutions
This multidisciplinary master's degree aims to provide a broad program, which educates students from all over the world about the fundamentals of the biology and transmission of infectious diseases, ethical and environmental concerns, and new and innovative ways to cure infectious diseases in a “One Health” approach (summarized in Table 1). The One Health concept aims at “addressing health risks at the animal-human-ecosystems interface” (The FAO-OIE-WHO Collaboration 2010 12 ).
The EU, (or European Union) Erasmus mundus program requires a mobility scheme (Figure 1) and a joint degree after graduation. Hannover Medical School's role in the consortium is to represent human and translational medicine as well as biorisk management. The latter aspect was poured into a newly designed teaching module. A BSL-3 module in Barcelona, held at the CReSA together with the University of Barcelona within the second semester, accompanies the module described herein. At CReSA, the students experienced a unique opportunity of a hands-on training in a BSL-3 facility.
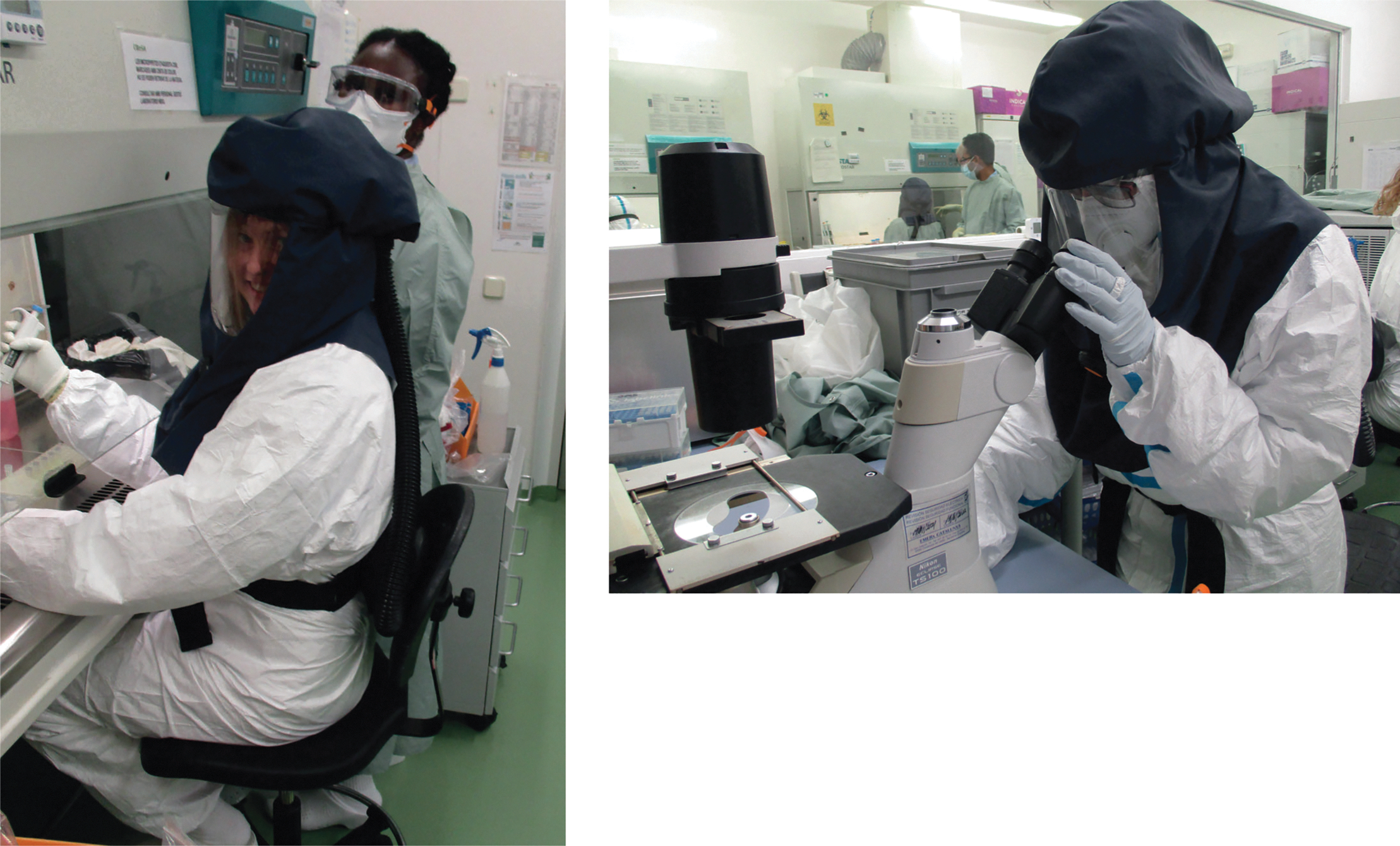
The pictures show the practical BSL-3 training at CReSA, Barcelona. Students wore full protective PPE (left side) and analyzed BSL-2 samples using a microscope (right side). BSL-3, biosafety level 3; CReSA, Centre de Recerca en Sanitat Animal; PPE, personal protective equipment.
The Module Biorisk Management
The third semester at Hannover Medical School broadens the biosafety and biosecurity perspective to a comprehensive biorisk management approach. The MHH biorisk management module consist of two main pillars: (1) the new biosafety concept of the WHO Laboratory Biosafety Manual 4th edition 13 and (2) operating a mobile containment laboratory.
All teaching units within this module are designed to equip the students with the ability to use an overarching biorisk assessment in their future careers, wherever they may be employed. ISO 35001:2019 is taught to students as an international standard for establishing biorisk management. Regardless of which country the students come from and regardless of which country they will be working in the future, ISO 35001 helps them with a structured approach and integration into existing quality management systems.
Students recognize that such a risk-based approach to biosafety-related activities will enable safe handling of highly pathogenic agents even in laboratories with limited resources. There are mainly low- and middle-income countries that face particular challenges from pathogenic and highly pathogenic agents due to their geographical location.
Infectious disease research and biosecurity aspects in particular are obviously not restricted to biological or biomedical fact knowledge but touch on pervasive ethical, societal, and political questions that become even more intricate when a One Health perspective (considering human, animal, and environmental health) is chosen. With respect to biosecurity the important aim of minimizing potential threats to society must be weighed against innovation in the prevention and treatment of infectious diseases.
Different forms of handling dual-use of research of concern (DURC) have been developed internationally, many of the them putting a strong emphasis on researchers' own responsibility and sensitivity toward potentially dangerous aspects of their research. 14 Against this background, adequate ways of addressing biorisk management in undergraduate and postgraduate education seems of utmost importance.
The construction of a mobile containment laboratory infrastructure is the response to outbreaks of infectious diseases caused by highly pathogenic agents, mostly viruses. In 2013, the European Commission funded the project “Establishment of mobile laboratories up to risk group 4 in combination with CBRN capacity building in sub-Saharan Africa” (project reference: IFS 2011/272-372). The aim was to establish three field deployable mobile laboratory units for the diagnosis of pathogens up to risk group 4. In the following years, this initiative evolved into the European Mobile Lab project and consortium.
The European Mobile Labs operated their field deployable laboratories during the largest Ebola Virus epidemic to date in West Africa. 15 The effectiveness of these laboratories in the Ebola epidemic together with the idea of collaborative capacity building for highly virulent pathogen diagnostics kick started further projects to train future generations of scientists. In this approach, biosafety and biosecurity are crucial focal points within the overarching idea of a One Health concept.
Description and Learning Objectives of the Module
The goal of the biorisk management module within IDOH+ is to deliver a comprehensive overarching approach to the subject. Bioethical aspects are integrated in addition. Risk assessment based on the WHO 4th laboratory manual, the BSL-3 training module in the second semester and the virology module also taught at the MHH all cumulate in the mobile laboratory part, where both theoretical knowledge and practical skills are needed to deal with the simulated outbreak situation.
The Module “Biosafety and Biosecurity” (Second Semester, Barcelona)
The module “Biosafety and Biosecurity” teaches the basics of biocontainment, biosafety, and biosecurity. The lectures cover all relevant topics on high-containment laboratories and research therein.
Risk assessment is the cornerstone of any biosafety and biosecurity-learning concept. The objective here is that students understand that risk assessment is a systematic process. Collecting information and evaluating the likelihood and consequences of exposure to or release of workplace hazards are key to set up the appropriate precautions and should result in an acceptable risk (WHO monograph “Risk assessment,” 2020; 9 see also chapter on BioRAM). Through guided discussion and interactive exercises, students learn to understand these key terms and to acknowledge that effective risk communication is an interactive process with both experts and staff.
Students learn about the different risk control measures and can identify the advantages and disadvantages. They learn that different measures represent a hierarchy of controls, through elimination, following engineering controls, administrative controls, practice and procedures, and lastly PPE. At the end of the module, the students demonstrate their newly acquired competence by preparing a risk assessment of their own practical activities in the BSL-3 laboratory using a simplified form. 16
Special attention is paid in the second semester to the construction and equipment requirements of biocontainment laboratories. In this study, learning objectives are to introduce students to the concept of primary and secondary barriers. 17 They get to know which facility features are used to mitigate different types of biorisks. Key engineering controls and equipment typically found in a biomedical research laboratory are discussed. Students should understand the laboratory zone concept and containment borders. 18
After the introductory lectures on the 1st day, the students can choose from different tasks dealing with design and planning of high-containment facilities from different angles as a homework in working groups. The student groups work in parallel to the lectures and their practical training. On the last day, one oral presentation per group is given, including time for comments from the students, suggestions, and remarks from the lecturers.
This feedback should allow the three teams to refine their approach and present a final written homework to be evaluated. The practical exercises include entrance and exit of a BSL-3 containment laboratory, the use of appropriate PPE, dexterity training, and inactivation procedures for viruses and bacteria (Figure 1).
The Module “Biorisk Management and Bioethics” (Third Semester, Hannover)
An international master's program on higher education in Biosafety, Awareness of Potential Misuse of Data and Biological Agents and Toxins, and Bioethics Issues must not be limited to the laws and the guidelines of individual countries only. Rather, international students need to understand the concepts and principles of biosafety and biosecurity and must be able to transfer and apply them to the individual situation of their home country. The WHO Laboratory Biosafety Manual 4th edition is excellently suited as a basis for such education and training.
Until the third edition of the WHO Laboratory Biosafety Manual, the classification of biological agents and laboratories was described in terms of risk/hazard groups and biosafety/containment levels. Although intuitive, it has led to the misconception that the risk group of a biological agent directly corresponds to the biosafety level of a laboratory. However, the pathogenicity of an agent is by far not the only factor determining the actual risk, but also by the procedure being performed and the competence of the laboratory personnel performing the experiments.
The 4th edition now shifts to a new paradigm. The handbook takes a risk-based, technology-neutral, and cost-effective approach to biosafety. The guiding principle describes the feasibility of laboratory activities, clinical diagnosis, and the promotion of biomedical research even in resource-limited environments, without compromising safety. This is increasingly important for combating infectious diseases. 13
In the 4th edition, risk control measures are sorted into three categories: core requirements, heightened control measures, and maximum containment measures based on increasing level of risk. Core requirements set the minimum standards for work in a low-risk biological laboratory. Heightened control and maximum containment measures are then added based on increased risk. Besides that, the importance of a safety culture in the laboratory is emphasized. This includes risk assessment, good microbiological practice and procedures and SOPs, and appropriate training of personnel.
The WHO LBM 4th edition is also excellent for educational training, because the manual is complemented by seven monographs holding detailed information on how to implement systems and strategies on specialized topics. For the IDOH+ program, we harness the monographs “Risk assessment” 9 and “Outbreak Preparedness and Resilience.” 19 Didactically well structured, the monograph “Risk assessment” offers templates for the preparation of risk assessments.
There are short forms for standard procedures and long forms for handling, for example, highly pathogenic microbes requiring intensive consideration using the detailed information on the procedure. After completing the first module of virology, medical microbiology, and toxicology, the students are now given the task to prepare a comprehensive risk assessment of their own activities during this practical course in this first module of the third semester with additional challenges, for example, to include a scenario where a student is pregnant.
BioRAM as a Mean of Quantifying Biosafety Measures
To make biorisk management more experienceable we used the software tool BioRAM to value different aspects of biosafety and inherent features of the pathogens. The Sandia National Laboratories developed BioRAM.7,20 The program uses a classical risk graph composed of likelihood and consequences to illustrate the risk associated with various research questions (Figure 2). We invented different scenarios using three pathogens handled in laboratories of variable safety levels.
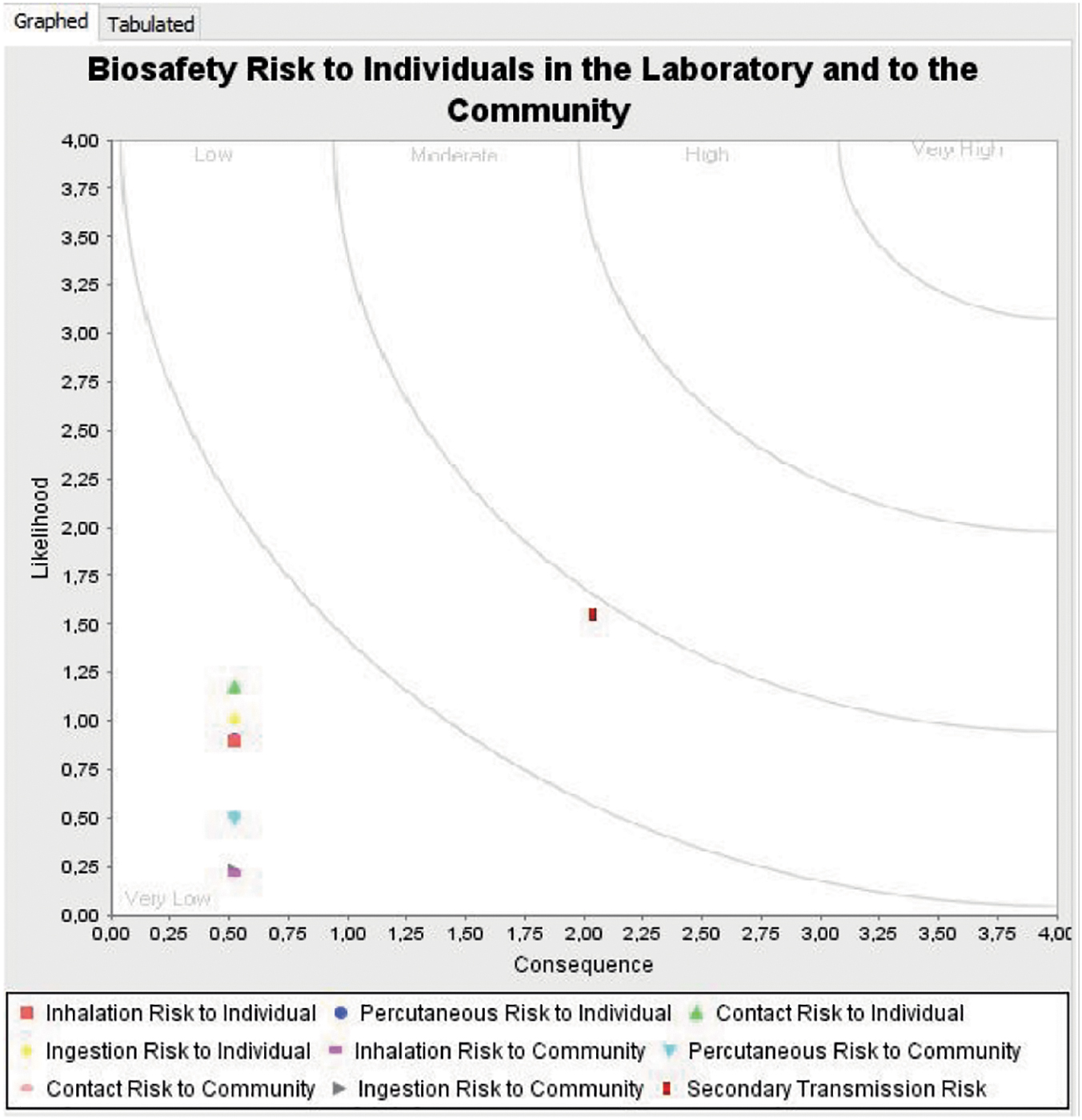
Risk graph produced by the BioRAM program. The examples given shows the risk associated with Monkey Pox Virus diagnostics. BioRAM, biosafety risk assessment model.
Two pathogens were highly pathogenic to humans and the third one only harmful to animals. All chosen pathogens have in common that intended or unintended release into the environment would have drastic consequences for humans or livestock. The unit is performed in a media teaching room equipped with one personal computer per student. The students chose different scenarios and then answer the questions BioRAM puts forward.
The final result is a risk graph showing different outcomes such as impact on the public or on the experimentators. The students are now asked to alter the biosafety measures or increase the pathogenicity of the agents. The learning objective here is to assess the impact of the imputed variations on the risk graph. The quantitative output of BioRAM makes biosafety visible and helps the students in the future to assess their own biosafety precautions.
Scene Play to Justify DURC and Its Communication to the Public
As biorisk management should also include the biosecurity aspect, we implemented a teaching unit for DURC. In addition, the ethical impacts of dual use are also discussed in the bioethics part. Various approaches have been explored to raise awareness for dual-use research. Germany follows a bottom-up approach in which the responsible scientist along with local ethics committees clarifies dual-use issues. 21 Along these lines, DURC should also become a part of teaching in life science.
In a role-play, the students shall experience how to justify risky experiments to the public. In other words, they have to transform a complex research question into simple language and answer questions from journalists, which are represented by the other half of the study group. The teaching unit starts with a short lecture on DURC. Afterward the student group was split into scientists and journalists.
To the scientists a dual-use experiment was suggested. Their assignment was to clarify the associated risks, how to mitigate them, and how to communicate their research to the public. The journalist group should envision questions for a press conference to challenge the scientist. We specifically did not exclude the yellow press at this point. After a given time both groups reconvened and a press conference moderated by one of the students as a spokesperson was held.
Bioethics: Goals and Tasks
A significant part of the biorisk management module is dedicated to ethical issues occurring in the intersection of infection research, Public Health, and security. These bioethics sessions are taught by specialists (with backgrounds in philosophy and medicine) from the Institute of Ethics, History, and Philosophy of Medicine at MHH. The main aim of the seminars is to create a joint understanding of research ethics and Public Health ethics frameworks and to apply them to concrete cases in an interactive form.
To meet these learning objectives successfully, the diverse backgrounds of students need to be considered. In particular, the teaching group is highly heterogeneous regarding the individual level of bioethics training (or contact to the humanities at all) but also with regard to the students' national and cultural backgrounds, which might play a role for ethical evaluations as well.
To address this heterogeneity adequately the bioethics classes start by presenting key aspects of general research ethics, but also of Good Scientific Practice. For example, the question of “What makes a scientist good or bad?” is debated interactively and then discussed in relation to infection research. Individual aspects (scientists as persons) are considered as well as the academic and scientific systems that dominate research.
The students also get an introduction in the ethics of translational research and in Public Health ethics frameworks. The last part of the introductory sessions sheds light on the theory and ethics of One Health, addressing challenges on the conceptual and normative-ethical level in the intersection of human, animal, and environmental health.
This more theoretical part is amended by case studies in Public Health ethics (e.g., duty to vaccination) and by examples for DURC that have come into public attention in recent years. The students can successfully link these competencies to the more factual knowledge they gain from the other biosafety and biosecurity parts of the master's program.
The Mobile Laboratory
To apply all the theoretical knowledge the students gained during the course of the third semester, we designed the “Mobile Containment Laboratory” practical course. This unit simulates an infectious disease outbreak in which the students take on various tasks.
Field deployed mobile containment and diagnostic laboratories are usually staffed with teams of four to six laboratory scientists and technicians, which poses particular challenges in team resource management and communication and coordination between the laboratory workstations. Unforeseen events have to be managed, for example, emergencies such as spills or injuries as well as non-laboratory-related events such as security threats or visits of the laboratory by officials and press representatives.
The students in the course are challenged with many of these aspects simulating real outbreak-like conditions. The scenario was created originally for the training course that is based on the wider experience and knowledge gained during many field deployments and training sessions of the European Mobile Labs. The students learn to perform the laboratory workflow in the field deployable laboratory for the molecular diagnosis (by PCR) of the scenario-based outbreak pathogen. Supervised and trained by experts from the Robert Koch Institute, Berlin and the Bernhard Nocht Institute for Tropical Medicine, Hamburg, they carry out work processes at four stations that are relevant for actual outbreak operations:
Patient sample reception, registration, and documentation. Transfer and inactivation of patient material in a glovebox. PCR analysis of the samples with real-time PCR. Theory/evaluation and communication with local partners.
After the introductory lecture, the students get the task of assessing the course rooms provided at Hannover Medical School and implement the necessary infrastructure and organization guided by the requirements of the laboratory workflow on biocontainment, zone concept, and biosecurity. 19 We would like to stress at this point that our scenario is as real as possible. Thus, the students start from unpacking the 18 boxes with equipment (Figure 3A). Nothing is preset. They even have to move furniture to create the sample reception.

Scenes from the mobile containment laboratory training.
Sample reception
The students learn how the sample shipments are received at the laboratory. This includes safe unpacking and documentation of sample and patient identity. It is here where the quality assurance process begins, which is continued downstream throughout the laboratory. The challenges of the sample reception are the arrival of potentially contaminated shipments.
They have to assess the risks concerning this interface of the relatively controlled laboratory environment to the more uncontrolled outside world (e.g., not well-trained transport staff handling shipments in an unsafe manner) and adapt accordingly (e.g., by using the appropriate level of PPE). They are trained in using the correct disinfection procedures (e.g., dunking) as well as disinfection solutions (e.g., correct dilution of bleach). Moreover, donning and doffing the appropriate personal protective equipment challenge the students right from the beginning (Figure 3B).
Good communication in the laboratory starts here, when the students inform their own colleagues at the other workstations about sample numbers, for example, so that further processes be well prepared.
Glovebox
Setting up the glovebox together and handling the samples inside the glovebox required effective coordination and communication. The students teamed up as pairs (the buddy and operator system) and followed the given SOPs. The patient samples are brought into the glovebox (high efficiency particulate air filter [HEPA] filtered negative pressure biosafety containment glovebox), inactivated for the PCR analysis and brought out after an approved decontamination process. The restricted size of the glovebox and the necessity to work with triple gloved hand posed additional challenges here. For the following sample, the students switched roles (Figure 3C).
PCR station
The complete PCR station was also packaged into the transport boxes and the students had to set up a pre-PCR “room,” which is a tent in the case of this mobile laboratory. Furthermore, they had to operate the quantitative PCR, machine according to the SOPs provided. Building up on our virology module the students had to perform and understand the quality control measures necessary for a valid diagnostic PCR result, which in our outbreak situation will have drastic consequences for a wider geographical region even. In addition, a plausibility check has to be done before the results can be communicated to the sending entity (e.g., hospital or Public Health officials). The students have to assure that the correct result, for the correct patient, reaches the correct sender in a timely manner.
Theory station
In parallel, the students attended a theory station. In mostly self-governed group work they were given theoretical tasks that serve as a debriefing wrap up of the training experience in the mobile laboratory deployment scenario. Mimicking a real-world deployment the students shall produce a mission report in the form of a presentation that summarizes their deployment, laboratory set-up planning, diagnostic activity with statistics as well as reporting on contingencies, incidents, and accidents.
The groups are also presented with a list of open questions, based on the topics and structure of the current WHO LBM “Outbreak and resilience” Monograph, 9 which guide the students to reflect about further laboratory safety and security issues, about human factors that come into play (training, psychological issues, etc.) and occupational health topics. The students' “Mission Report” had to include a risk assessment on the different stations they had passed through. Finally, the students get the opportunity to present their reports to their respective mission headquarters played by trainers, as a final mission debriefing in the deployment scenario (Figure 3D).
In summary, the whole module received very positive feedback from the students and ranked among the best teaching units in the whole master's program. There is a thorough follow-up of the IDOH alumni and some of them entered a career in national Public Health bodies or similar institutions. Our hope and goal is that they use and build up on the knowledge and skills acquired in our module.
Discussion
The guiding principle of the IDOH+ consortium is the One Health concept. The IDOH+ master program shed light on various aspects of human and animal health and their connection to the environment and ecology during all semesters. Building up the previously gained knowledge and practical experiences, we amended the third semester with an overarching biorisk management module, including the operation of a mobile laboratory with a simulation of hemorrhagic fever outbreak in Africa.
The Erasmus mundus funding gave us the opportunity to recruit instructors from Germany's leading Federal institutes for infectious disease diagnostic and prevention. Although initially designed to train scientists and technicians for deployment in outbreak situation, both institutions recognized and supported the benefit of educating the next generation of scientists. Many of IDOH alumni return to their home countries and serve in the respective governments or NGOs. Surely, they will implement the knowledge and skills acquired in this master program.
In the end, this will serve our goal to improve the situation for biomedical research in low resource situations as well as high-containment laboratories around the world. Still on the horizon, but not too farfetched, is a scenario where outbreaks can be handled immediately on-site and thereby preventing further spread. IDOH also benefits from its small group size (maximum of 25 students per intake), which enables everyone to participate in all, especially, practical parts of the program. The herein described module on biorisk management aimed to provide a comprehensive approach building up on previous modules.
In our view, the students should experience the benefit of an overarching and comprehensive biorisk management approach. The contents of the 4th WHO laboratory manual and the paradigm shift therein should broaden their thinking in terms of risk assessment and the opportunities and benefits of this approach. While working in the BSL-3 in Barcelona and in the mobile containment laboratory in Hannover, they should experience how important a widening the biosafety culture is for successful research.
A few examples of topics that require ongoing review include animal welfare, duty of vaccination, and dual use research. Researchers need a basic knowledge in all these areas to rethink their own research and decisions, especially if they move into Public Health and policy decision-making. Ethics are not static, but require constant adjustment as well as essential principles and moral perspectives.
This teaching unit has its limitations. Within the IDOH+ master program, it is only one of many modules and the way of approach biorisk management presented here has to be further trained and explored to become a guiding principle for the students in their future research.
In summary, we believe that equipping students with the ability to use biorisk management to improve working in the infections disease field and not to perceive it as a new administrative burden will help in pandemic preparedness and diagnostics in the long run. We truly support the bottom-up approach to raise awareness for dual use and ethical issues at this early stage of the student's careers.
Footnotes
Acknowledgments
We would like to thank the IDOH students of the intake 2020 for their permission to use the pictures in Figures 1 and ![]() . In addition, we thank Kathrin Baumann, Sabrina Weiß (both RKI), Beate Becker-Ziaja, Alexander Schlaphof, and Yemisi Ighodal (all BNITM) for their indispensable support and efforts to ensure a successful mobile laboratory training, and Anna Buch and Andrea Cuadra for administrative and technical support.
. In addition, we thank Kathrin Baumann, Sabrina Weiß (both RKI), Beate Becker-Ziaja, Alexander Schlaphof, and Yemisi Ighodal (all BNITM) for their indispensable support and efforts to ensure a successful mobile laboratory training, and Anna Buch and Andrea Cuadra for administrative and technical support.
Authors' Contributions
J.B. co-designed the module, held teaching units, and wrote the article. S.S. taught the bioethics part and wrote the article. M.G. was the lead instructor of the mobile laboratory training and wrote the article. X.A. designed the BSL-3 module in Barcelona and wrote the article. J.M. designed the module, taught several teaching units, organized the mobile laboratory training, and wrote the article.
Authors' Disclosure Statement
All authors declare no conflict of interest.
Funding Information
EU Erasmus Mundus Joint Master Degree; project number 6100556-EPP-1-2019-1-FR-EPPKA1-JMD-MOB.
