Abstract
Background:
Inactivation of infectious liquid waste can be performed by different means, including autoclaving or chemical inactivation. Autoclaving is most widely used, but cannot always be implemented, so that chemical inactivation is a possible alternative. However, its efficacy has to be proven by in-house validation. Here, we provide an easy-to-implement validation process adapted from existing standards.
Methods:
The bactericidal activity of two commercially available disinfectants, containing glucoprotamine or peracetic acid, was tested on two gram-positive bacteria, methicillin-resistant Staphylococcus aureus (MRSA) and Streptococcus pneumoniae (SP). Quantitative suspension tests were adapted from the European standard EN 13727. Membrane filtration was used to remove any trace of the active antiseptic. The stability of working solutions and the effect of different organic loads were also assessed. Further, the aquatic toxicity of glucoprotamine was tested on the viability and behavior of zebrafish larvae.
Results:
Peracetic acid was highly efficient against both strains, even at a working concentration of 0.25% for 1 minute S. pneumoniae was also highly sensitive to glucoprotamine, whereas MRSA required either higher concentrations (1%) or longer exposure times (30 min) at lower concentrations. Further, the lethal dosis that kills 50% of the embryos (LD50) for glucoprotamine was 3–3.5 mg/L using the zebrafish Embryo Acute Toxicity Test.
Conclusion:
We could confirm the very high efficacy of glucoprotamine- and peracetic-based disinfectants to inactivate liquid waste containing gram-positive bacteria. Also considering aquatic toxicity, this methodology could help laboratories to establish validated procedures for chemical inactivation of infectious liquid wastes relevant to their institution, balancing efficacy, and environmental issues.
Introduction
Laboratories and facilities generating infectious waste must ensure their safe disposal. Many methods for decontamination (definition given in Supplementary Material) exist, each with their own advantages and peculiarities. The proper choice depends on different properties of the waste, such as chemical composition, state of the matter (solid vs. liquid), characteristics of the contaminating biological agent, and costs, all of which must be evaluated in a risk assessment. 1
Autoclaving is usually the method of choice for inactivation of most solid wastes, being safe, effective, reliable, and nontoxic, but may implicate a high initial investment and regular maintenance costs. It is not always practicable in case of material incompatibility, resulting in undesired effects such as corrosion or melting.1,2 Autoclaving of liquids is more challenging, requiring an autoclave suitable for the task. Further, the thermal conductivity of vessels containing the liquid waste needs to be considered and autoclaving large amounts of liquids may pose specific hazards such as over-boiling or the generation of toxic vapors or fumes.1,3,4 Chemical disinfection, when applicable, may therefore represent an alternative, provided that the efficacy of the chemicals is tested. 1
Microorganisms differ greatly in their innate sensitivity toward chemical disinfectants. Generally, spores are more resistant than vegetative bacteria or enveloped viruses.1,2 These differences are among the first things to be considered when selecting a disinfectant. 1 Disinfectants generally become more efficient with higher concentrations, though exceptions exist (i.e., iodophors). Densities of the microorganisms, temperature, pH and water hardness are other factors influencing disinfectant efficacy.
The presence of organic matter such as serum, blood, pus, fecal, or lubricant material in the liquid waste might also interfere with antimicrobial activities. 2 The properties of the disinfectant itself are also important factors to consider. This includes its stability, toxicity, corrosiveness, the presence of irritants, or any potential incompatibility with other chemicals that might be found in the solution to be inactivated. 1
Manufacturers must provide proof for the potency and efficacy of their disinfectants before market approval by carrying out standardized tests, defined, among others, by the Food and Drug Administration (FDA) in the United States and by methods outlined in several European Norms (EN) in Europe. These tests recommend the use of standard reference microorganisms. The end-users also need to conduct their own validation on microorganisms likely to be present in the waste they generate.
Peracetic acid (PAA) is a strong and highly reactive oxidant that denatures proteins by oxidizing oxidation-sensitive amino acids, sulfhydryl, and sulfur bonds. It also damages DNA and RNA by inducing strand breaks.2,5 PAA is, however, incompatible with the presence of heavy metals, reducing substances and alkali and is corrosive for different materials, though these effects may be reduced by additives and pH modifications.2,5 PAA has a broad spectrum of activity against viruses, gram-positive and gram-negative bacteria, fungi, and yeasts.2,5–7 It is not inactivated by organic matter and remains sporicidal even at low temperatures.
However, it is ineffective against helminths and certain protozoa. PAA poses no significant environmental hazard since it is halogen-free, leaves no residue, and its decomposition products (i.e., acetic acid, water, oxygen, hydrogen peroxide) are harmless or short-lived. However, PAA is instable, especially when diluted, and is readily inactivated by blood.2,5
Glucoprotamine is the conversion product of
It dissolves well in water, is non-volatile 8 and highly stable, retaining its efficacy in undiluted form up to 8 years of storage. 9 Glucoprotamine induces the disorganization and destruction of the cytoplasmic membrane of bacterial cells 10 or the lipid bilayer of enveloped viruses, while interacting with lipophilic groups of the protein capsid of naked lipophilic viruses. 9
The disinfectant is active against vegetative bacteria,8,9,11–13 including mycobacteria,9,14 fungi,8,9,12 enveloped and nonenveloped lipophilic viruses 8 but not against unenveloped hydrophilic viruses.9,15 There is evidence of sporicidal activity in its undiluted form.9,11,16 Microbiostatic and microbiocidal efficacy is greater in basic pH range.8,9
Another aspect to consider when choosing chemical inactivation is the disposal of the waste. Disposal is most often performed “down-the-drain” to sewage treatment systems. Therefore, the greatest potential threat is on the aquatic environment, and liquid waste should be treated as special waste if the biocide exhibits toxicity. Hazard assessment, in this case, requires to evaluate the potential for the substance to persist in the environment, to accumulate in biota, and to be toxic.
It is therefore usually assessed by the manufacturers and is described in their safety data sheets. For a lot of substances, high biodegradability or efficient removal during wastewater treatment mitigates their ecotoxicity by limiting the predicted environmental concentration to low levels. Glucoprotamine was originally described to have favorable ecotoxicological properties, being easily biodegradable in water treatment plants.8,17 However, one study classified the substance as potentially hazardous to the aquatic environment and recommended routine analysis of its concentration in surface waters. 18
The acute fish embryo toxicity test is a test that uses short-term exposure to a chemical substance to determine the concentration that is lethal to 50% of zebrafish larvae (50% lethal concentration [LC50]), as an indicator of acute fish toxicity. It is now considered as a valuable alternative for the acute fish toxicity test 19 and has been adopted in 2013 in a guideline of the Organisation for Economic Co-operation and Development (OECD).20,21
As complementary analyses, other phenotypic or behavioral tests can also be performed, especially at sub-lethal concentrations. 22 For example, phenotypic screening is performed by assessing free swimming behavior of the larvae under sequenced illumination conditions.23,24
The aim of this study was to develop an in-house methodology to validate chemical disinfection with glucoprotamine or PAA on liquid waste containing bacterial strains commonly encountered in our institution. Further, although not mandatory for in-house validation, aquatic toxicity of glucoprotamine was tested on zebrafish embryos. These tests are meant to help in the selection of the most appropriate method for chemical inactivation.
Materials and Methods
Bacterial Strains and Growth Conditions
Chemical inactivation was tested on a penicillin-sensitive Streptococcus pneumonia (SP) serogroup 3 strain isolated from the cerebrospinal fluid of a patient with bacterial meningitis 25 and on a Methicillin-resistant Staphylococcus aureus (MRSA) strain that was isolated from a patient with bacteraemia and has been recently sequenced. 26
Bacterial stocks were stored at −80°C using the Protect Microorganism Preservation System (Technical Service Consultants, United Kingdom). A bacterial aliquot bead was transferred from the stock into 10 mL Brain Heart Infusion Broth (BHIB; Becton Dickinson) for SP and incubated overnight (ON) at 37°C and 5% CO2. One milliliter of the ON culture was diluted into 9 mL of prewarmed fresh BHIB, further incubated at 37°C and 5% CO2 for 6 h, and referred to as test suspension N.
Similarly, MRSA was grown ON in 10 mL Trypticase Soy Broth (TSB; Beckton Dickinson) at 37°C and 75 rpm on an orbital shaker (Heidolph Instruments GmbH & Co. KG, Germany). The ON culture was used directly as test suspension N, since no significant difference in bacterial density was found between ON and 6-h logarithmic cultures.
The bacterial concentration of the test suspension N was determined in duplicates from 200 μL of 10−6 and 10−7 dilutions plated on Columbia sheep blood agar (CSBA) plates.
Bactericidal Solutions
The disinfectant solutions were prepared from commercially available concentrated formulations, recommended for both the cleaning and disinfection of instruments (Ecolab GmbH, Germany) (Table 1).
Test products with bactericidal activity
Liquid concentrate.
Powder.
EN, European Norms.
Product A, containing 25 g glucoprotamine/100 g, is recommended at working concentrations of 1%, 2.5%, and 4% with contact times of 60 min respectively 30 and 15 min. Product B is available in powder form, should be dissolved in water, and mixed for 15 min. Indicated working concentrations and exposure times are 1% at 60 min and 2% at 5 min. A 2% solution generates >1000 ppm peracetic acid.
In the present study, the final concentrations of the disinfectants are stated as the dilutions of the manufactured concentrate or powder. The following dilutions were evaluated: 0.1%, 0.25%, 0.5%, 1% and further 1.5% for SP. These are equivalent to 250 mg/L (0.025%), 625 mg/L (0.065%), 1.250 mg/L (0.125%), 2500 mg/L (025%), and 3750 mg/L (0.375%) of pure glucoprotamine for product A, respectively >50 mg/L (0.005%), 125 mg/L (0.0125%), 250 mg/L (0.025%), and 500 mg/L (0.05%) of peracetic acid for product B. For the tests, the disinfectant solutions were prepared at 1.25 times of the final working concentrations since they will be diluted to 80% during the tests and controls. NaCl 0.85% was used as diluent.
Organic Load Solutions
In the test conditions, we used bacterial culture medium (BHIB or TSB) as organic load, since this is the most relevant situation in the research laboratories. We also additionally included fetal bovine serum (FBS) 10% (final concentration during test) and NaCl 0.85% in some tests.
Quantitative Suspension Test
The method has been adapted from Chemical disinfectants and antiseptics—Quantitative suspension test for the evaluation of bactericidal activity in the medical area—Test method and requirements (phase 2, step 1); German version EN 13727:2012+A2:2015 27 for a customized use in our laboratory. Filtration was chosen as the mean of neutralizing the biocidal activity of the disinfectant, which represents a method equivalent to chemical neutralization, if validated. 28 All tests were carried out in duplicate and repeated at least three times at room temperature under a Class II Biosafety Cabinet (BDK Luft- und Reinraumtechnik GmbH, Germany).
One milliliter of a test suspension N or N−1 (dilution 1:10) was mixed by vortex with 1 mL organic load solution (interfering substance). After 2 min, the disinfectant solution (8 mL) was added and the tube shortly vortexed. The mixture was kept at room temperature for specified exposure times. At the end of this exposure time, the mixture was vortexed again, and 100 μL was transferred into a tube containing 1.9 mL of NaCl 0.85%.
This suspension was immediately filtered through 47 mm × 0.45 μm filters (S-Pak, HAWG047S6; Merck Millipore) mounted on a glass filter holder system (XX1014700; Merck Millipore). The filters were washed with 50 mL NaCl 0.85% to remove any trace of the bactericidal substance, transferred onto CSBA agar plates, and incubated for 24 h. The number of colony forming units (cfu) on the filters was counted.
The plates were further incubated for 20 to 24 h. The filters that no longer showed well-separated colonies were not re-counted. For the remaining filters, if the number of cfu had increased after the additional 24 h incubation, only the higher number of cfu was used for further evaluation (Figure 1).
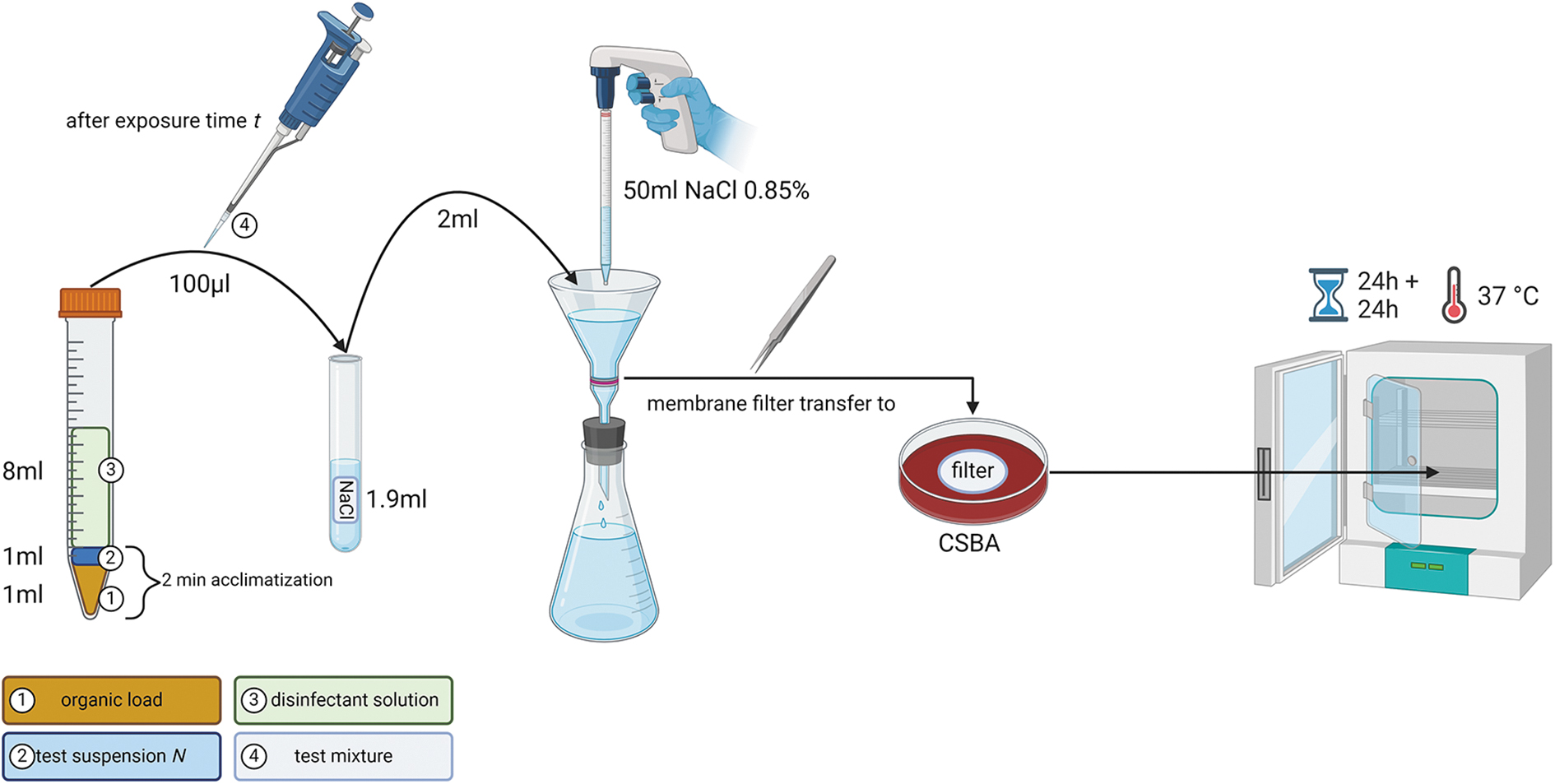
Graphical representation of the quantitative suspension test procedure for the determination of the bactericidal concentration. Created with BioRender.com. CSBA, Columbia sheep blood agar.
Calculation methods for germ reduction are based on those described in EN 13727, paragraph 5.6. Along these principles, the following values are determined: VC (colonies counted on the plates or the filters), N (initial cfu/mL in culture), N0 (cfu/mL in test suspension right at the onset of the exposure to the disinfectant at t0, corresponding to N/10), Na (surviving cfu/mL after t had elapsed), and NV (cfu/mL in the validation suspension).
The germ reduction R (R = N0/Na) is given in decimal-logarithmic values and calculated separately for each product concentration and exposure time with the following formula: log10 R = log10 N0–log10 Na. Different calculation rules related to the lower (<14 cfu) and higher (>165 on filters, >300 on plates) admissible limits for counting bacteria were also applied, according to EN13727.
Process Validations
It is crucial to conduct different process validations as described in EN 1373727 (description also provided in Supplementary Materials). For our own validation, we used only the highest disinfectant concentration and the more resilient strain (MRSA). Validation of experimental conditions (control A in EN 13737) was not performed, since nutrient broths or FCS, used as organic load, do not contain microbicidal constituents. Filtration control to check bacterial recovery and enumeration on filters (control B) and the validation of the method for disinfectant neutralisation by filtration (control C) were performed as described in the following two paragraphs.
Filtration Control B
An ON culture of MRSA was prepared, according to the paragraph “Bacterial strains and growth conditions” of the Materials and Methods section. From this culture, dilution series were performed in quadruplicates and 100 μL of the 10−6 dilutions was either plated on CSBA or filtered, using the same protocol as for the quantitative tests. A mean bacterial titer was calculated for both methods. The test was independently repeated three times.
Validation of Disinfectant Neutralization by Filtration (Control C)
We verified that our washing procedure efficiently removed any traces of the disinfectant on the filters, which may inhibit bacterial growth (Supplementary Figure S1). For the test, we chose MRSA, the most resilient strain, and the highest bactericidal concentration (1%) tested on this strain.
The bacterial validation suspension (NV) was prepared by diluting 500 μL of a 10−5 dilution of the test suspension N into 1.5 mL of NaCl 0.85%. Enumeration was made by plating 200 μL of the validation suspension NV on CSBA, performed in duplicate. Optimally, it should contain between 3.0 × 102 and 1.6 × 103 cfu/mL 3 .
One milliliter of organic load (FBS) was mixed with 1 mL of NaCl 0.85% and 8 mL of the disinfectant solution (final concentration of 1%). The mixture was kept at room temperature for the same exposure time as the quantitative suspension test. At the end of this exposure time, a 100 μL aliquot was taken, transferred into a tube containing 1.9 mL of NaCl 0.85%, and processed by membrane filtration, followed by washing with 50 mL NaCl 0.85%.
One hundred microliters of the validation suspension NV diluted in 1.9 mL NaCl 0.85%, was added onto the membrane filter, and processed for filtration and washing as described earlier. The number of bacteria on the filters was used to determine the bacterial concentration (Nc) in each sample and compared with the concentration of the validation suspension determined before filtration (NV).
Disinfectant Stability Determination
Solutions of 0.5% and 1% of glucoprotamine-containing product A were prepared and stored for 4 weeks before testing. Similarly, 0.25% and 0.5% solutions of peracetic acid-generating product B were prepared and stored for 3 and 5 days before testing. All solutions were stored in the dark at room temperature in a climate-controlled room (21°C ± 2°C). Stability was determined using MRSA for the quantitative suspension test.
Acute Fish Toxicity Test
Assessment of LC50
All experimental procedures involving zebrafish larvae were according to local animal welfare regulations for the care and use of animals. Adult wild-type zebrafishes (strain AB) were maintained at 28.5°C in 3.5-L tanks with 10/14 h dark/light cycle. AB zebrafish embryos were collected within the first hours post fertilization (hpf) and kept at 28.5°C in E3 medium (5 mM NaCl, 0.17 mM KCl, 0.33 mM CaCl·H2O, 0.33 mM MgCl2·7H2O) for zebrafish embryo supplemented with 0.3 mg/L methylene blue in an incubator equipped with a dark/light cycle similar to the husbandry conditions (Memmert, ipp110plus; Memmert GmbH+Co. KG, Schwabach, Germany).
Two days post-fertilization, manually dechorionated larvae were exposed to different dilutions of product A in E3 medium, corresponding to final concentrations of glucoprotamine ranging between 0.001 and 100 mg/L or to E3 medium only (control) for 3 days (n = 15–40 larvae per concentration). Lethality was determined for each concentration by observing the larvae under a stereomicroscope (Leica S9i) for the absence of heartbeat as criterion. The LC50 was calculated using simple logistic regression analysis.
Behavioral analysis
Behavioral analyses were performed using the observation chamber DanioVision powered by the video tracking software Ethovision XT version 15.0 (Noldus, Wageningen, Netherlands). In addition, constant water temperature at 28.5°C surrounding the experimental well plate was provided by the DanioVision temperature control unit. For the behavioral testing, larvae were exposed to LC50 and two sublethal concentrations of glucoprotamine. After 72 h, control (n = 72) and treated larvae (n = 30–36 larvae per concentration) were placed in a 96-well plate in 200 μL of the corresponding concentrations or E3 per well and transferred into the behavioral testing chamber.
For the locomotion testing, larvae were acclimatized in the dark for 1 h before recording. The total duration of the locomotion test was 50 min, starting with a 20 min dark period, followed by direct transition into a 10 min light period and ending with a second 20 min dark period, as described elsewhere. 24 The mean distance moved during the different illumination periods and the distance moved during the first minute after dark-light and light-dark transitions were analyzed. All behavioral experiments occurred in the morning between 9:00 and 12:00 a.m. All experiments were analyzed at 28.5°C with the same detection and acquisition settings.
Statistical Analysis
When two data sets were compared directly, a paired two-tailed t test with assumed Gaussian distribution and Tukey's test with significant difference (p = <0.05) was conducted. Where more than two data sets were analyzed, an ordinary one-way ANOVA with assumed Gaussian distribution and Tukey's test with significant difference (p = 0.05), was used instead. The determination of the lethal dosis that kills 50% of the embryos (LD50) of glucoprotamine on zebrafish embryos was performed using simple logistic regression analysis. GraphPad Prism 9.4.0 (GraphPad Software, LLC) was used for statistical analysis and graphical representations.
Results
Process Validations
Filtration control B
While a small decrease in the bacterial titer determined using the filter method could be observed (Figure 2A), this was not significant (log10 8.5 ± 0.11 vs. log10 8.62 ± 0.08, p = 0.07, paired t test). The mean recovery by filtration was 76.5% ± 9.8%, which is above the 50% tolerable recovery described in EN 13727.
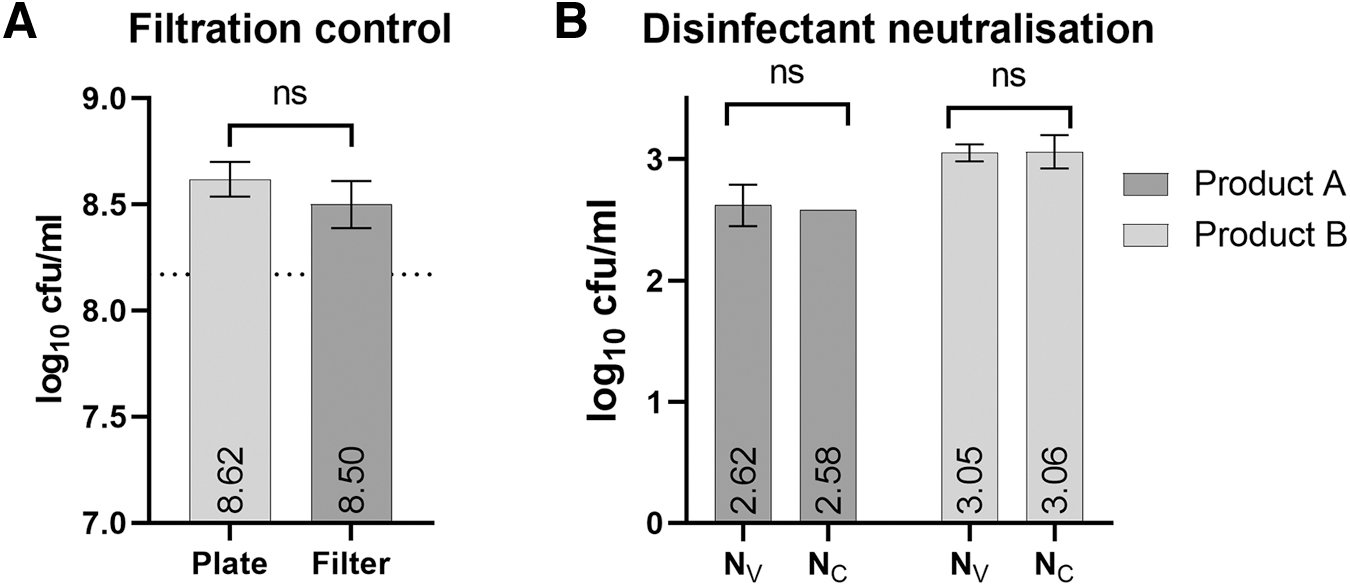
Validation of disinfectant neutralization by filtration (control C)
Both glucoprotamine and peracetic acid (at a product concentration of 1%) were efficiently removed from filters on washing (Figure 2B). There was no significant difference between bacterial concentration determined by plating of the validation suspension on agar plate (Nv) and after filtration on filters that have been previously exposed to the disinfectant and washed (Nc).
Quantitative Suspension Tests
Bactericidal activities of glucoprotamine and peracetic acid were assessed by exposing SP and MRSA to 0.1%, 0.25%, 0.5%, and 1% final working concentrations of the concentrate, respectively powder.
Glucoprotamine
SP was highly susceptible to glucoprotamine, with only a few colonies surviving after 1 min of exposure, even at the lowest concentration (0.1%). MRSA was more resistant to glucoprotamine, and the highest tested concentration (1%) was necessary to reach a ≥ 5 log10 reduction after 1 min exposition (Figure 3A). Exposure time of at least 30 min with a concentration of 0.1% or 0.25% also produced similar germ reduction of R = ≥5 log10 for MRSA (Figure 3B).
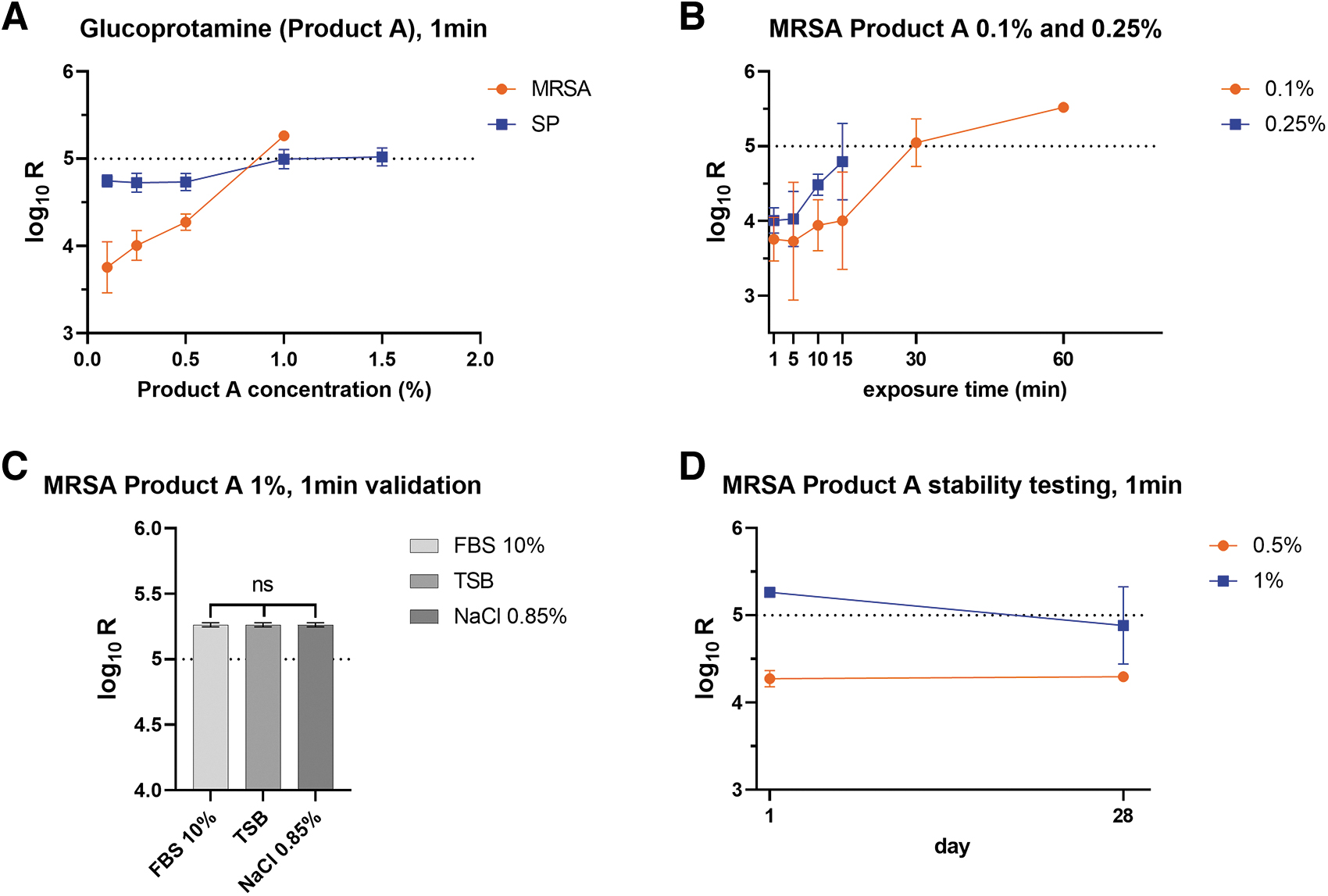
Efficacy of glucoprotamine in relation to
When used at a concentration of 1% for 1 min, the efficacy of glucoprotamine on MRSA was not influenced by the different organic loads we tested (Figure 3C). When stored for 4 weeks, the germ reduction for a 0.5% solution was similar (R = <4.3 log10) to the freshly prepared solution (R = 4.27 log10). However, for a 1% disinfectant solution, prolonged storage conditions resulted in a slight attenuation of the efficacy (R: from 5.26 log10 to 4.88 log10) (Figure 3D).
Peracetic acid
PAA proved highly effective against both strains at an exposure time of 1 min, with only the lowest concentration (0.1%) showing any bacterial growth on filters in most cases (Figure 4A). Most of the surviving colonies only became visible after >24 h of incubation on filters (not shown). Given the high efficacy demonstrated with 1-min exposition, longer contact times were not tested. Similar to glucoprotamine, we could not detect any interfering effect of the different organic loads we tested (Figure 4B). Finally, the working solutions of peracetic acid were found to still be efficient 1 day after preparation, but then to quickly lose some antimicrobial activity, after 3 days (R = 4.83 log10 at 0.25% and R = 4.82 log10 at 0.5%) and further after 5 days (R = 4.5 log10 for both tested concentrations) (Figure 4C).
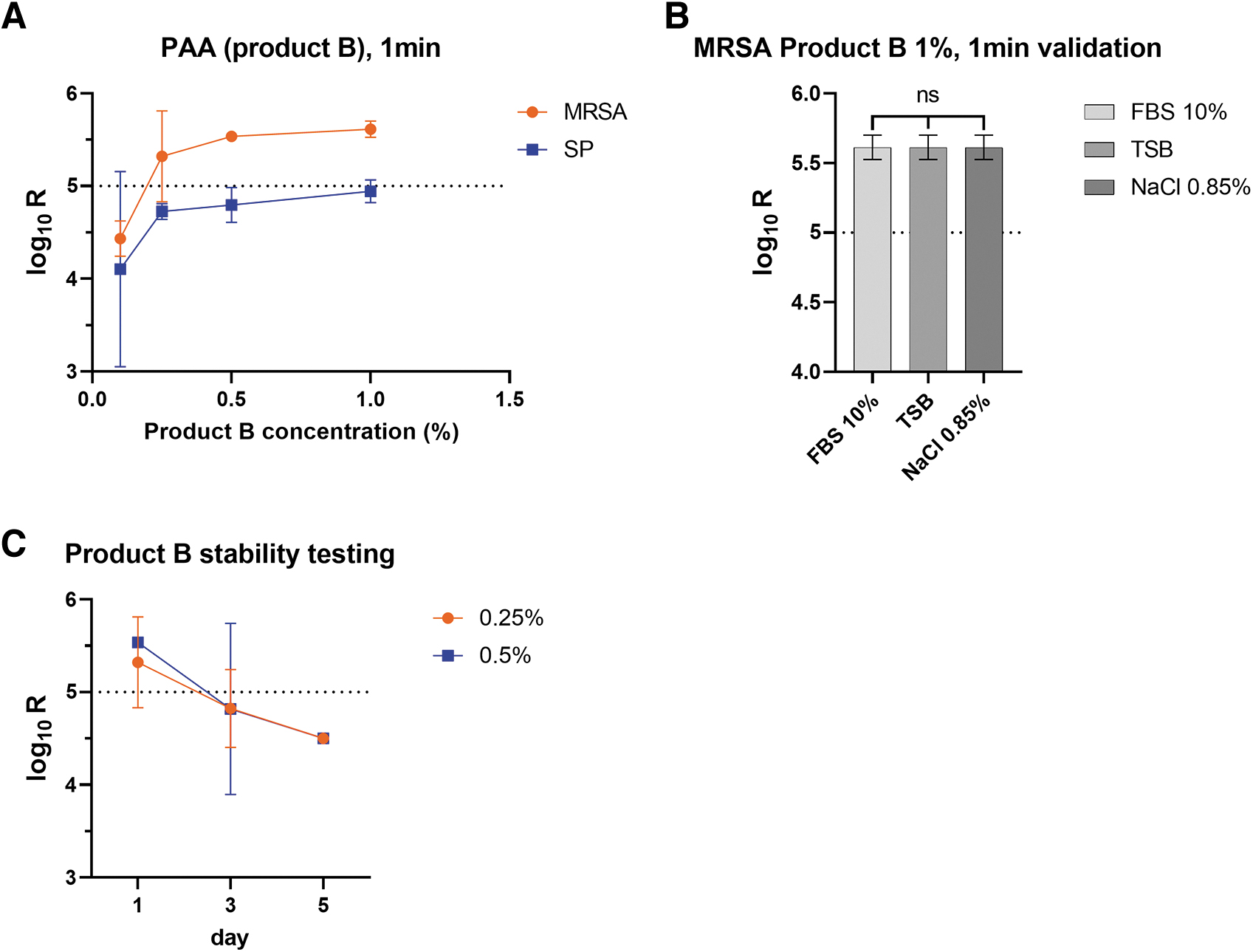
Efficacy of peracetic acid in relation to
Acute Aquatic Toxicity Test for Glucoprotamine
Determination of LC50 in zebrafish larvae
Larvae treated with glucoprotamine concentrations below 2 mg/L showed a survival rate comparable to control larvae (Figure 5A). In contrast, larvae treated with concentrations above 4.5 mg/L showed no survival after 3 days of treatment. Simple logistic regression analysis revealed that LC50 of glucoprotamine lies between 3 and 3.5 mg/L.
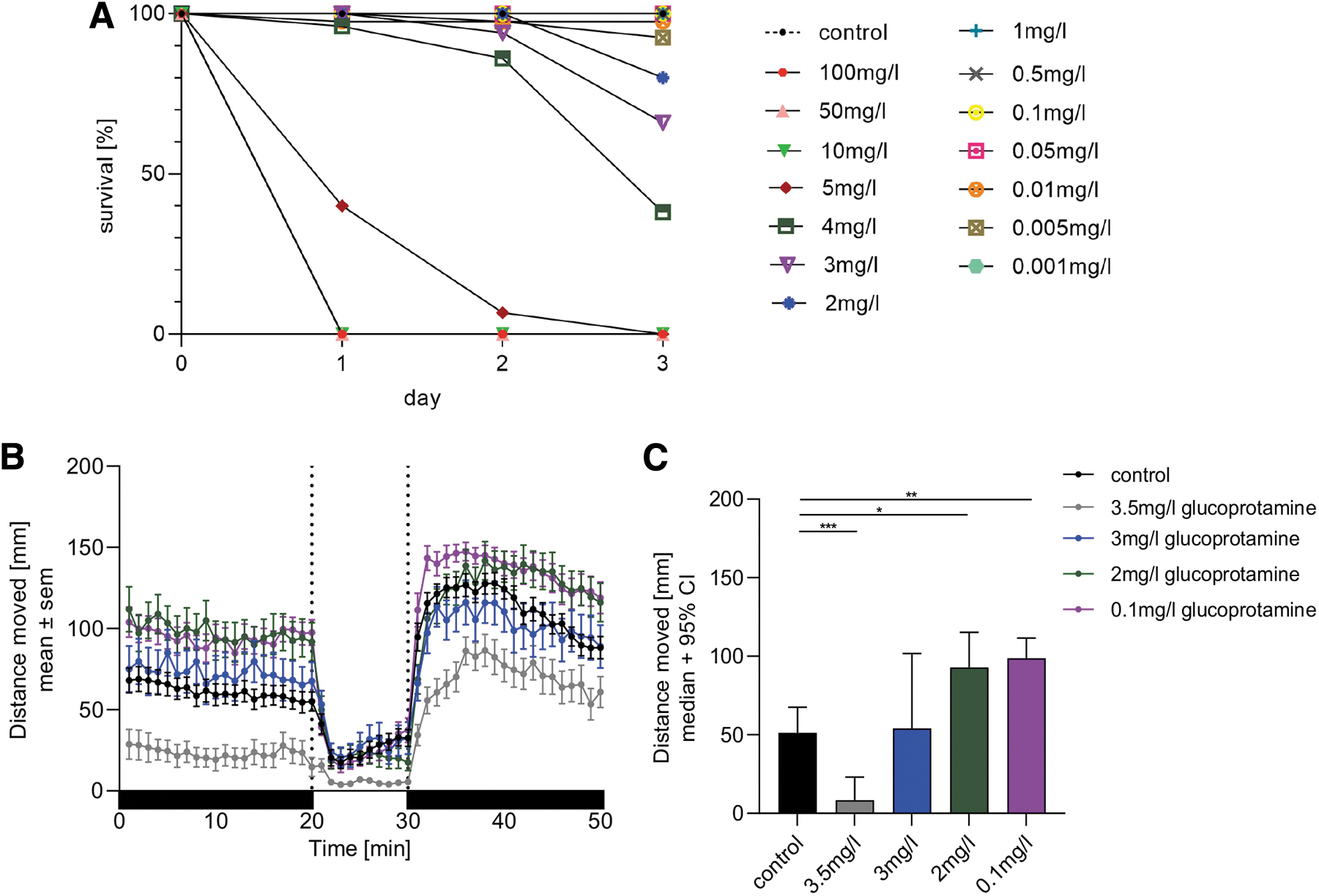
Zebrafish larvae show altered behavior on treatment with glucoprotamine
Swimming pattern of the larvae during alternating light and dark conditions was determined (Figure 5B). The basal locomotor activity (BLA), defined as the distance moved by the larvae in the first dark period of 20 min after acclimatization, was significantly reduced in larvae treated with 3.5 mg/L but not with 3 mg/L compared with controls (p < 0.001) (Figure 5C).
In contrast, larvae exposed to 2 (p < 0.05) and 0.1 mg/L (p < 0.01) exhibited significantly increased BLA compared with controls. During the next 10-min light period, only larvae treated with 3.5 mg/L glucoprotamine showed significant reduced mean activity during the light period compared with control larvae (p < 0.01, Figure 5B). During the last 20 min dark period, larvae treated with the highest concentration showed reduced mean activity (p < 0.01), whereas larvae treated with the lowest concentration showed increased mean activity compared with the control (p < 0.05, Figure 5B).
Sudden increase or decrease in light intensity evokes a specific locomotor response in zebrafish larvae. 24 Only larvae treated with 3.5 mg/L glucoprotamine showed reduced activity in the first minute after the transition from dark to light (light-on visual motor response) compared with the control (p < 0.01) (Supplementary Figure S2A). Further, they also demonstrated a reduced light-off visual motor response to the sudden decrease in light intensity in the first minute of the second dark period compared with the control larvae (p < 0.0001) (Supplementary Figure S2B).
Discussion
The aim of this study was to develop a validation procedure for chemical inactivation of liquid laboratory waste containing bacterial pathogens as an alternative to autoclaving. The European standard EN 13727 specifies a test method and the minimum requirement for antibacterial activity of chemical disinfectant and antiseptic products (to be diluted or ready-to-use) in the medical area in the fields of hygienic handrub, hygienic handwash, surgical handrub, surgical handwash, instrument disinfection by immersion, and surface disinfection by wiping, spraying, flooding or other means. 27
The product must fulfill the requirement to be introduced on the market. It needs therefore to be tested on a representative panel of microorganisms under different conditions to cover most of its potential future applications. We adapted the methodology for a narrower application, that is, the inactivation of liquid laboratory waste containing some bacterial strains used in our research department. We directly used liquid bacterial culture, instead of bacteria streaked from agar plate and resuspended in a defined diluent.
Further, the protocol was adapted, using a test solution that reproduces the composition of laboratory waste that we may encounter in most of the situations, including bacteria in their culture media or in solution containing 10% FBS. We used filtration as the method for detergent neutralization. The advantage of this method is that it does not require the use of a validated neutralizer that may not always be available.
On the other side, filtration requires additional equipment and proper validation steps, and may be not applicable in the case when the test solution (with or without organic load) provokes the clogging of the filter. The filtration process was simplified, using lower volume (50 mL) of saline solution (0.8% NaCl) as rinsing liquid. Validation of these simplified processes (bacterial recovery from filtration, and detergent neutralization) was performed and is described in detail in the present study.
In contrast to EN 13727, process validations were not systematically performed in parallel to all quantitative suspension tests, but separately in three independent experiment concerning the filtration control, and only with the highest disinfectant concentration for validation of disinfectant neutralization. This allows to reduce the number of filtrations to be performed during a session, especially when only few filtration devices are available.
If properly performed and constant conditions are respected, we are confident that the methodology remains valid. The bactericidal efficacy of two commercially available disinfectants containing either glucoprotamine or peracetic acid was therefore tested on MRSA and SP. Filtration was chosen to suppress the bactericidal and/or bacteriostatic effect of the disinfectant before incubation of the surviving bacteria after their exposition.
The European standard indicates that the biocide passes the test when it demonstrates a 5 log10 reduction. When performing this standard for in-house validation on specific strains and culture media, this may reveal challenging. According to EN13727 (paragraph 5.4.1.4), the test suspension N should optimally contain 1.5–5 × 108 cfu/mL, which represents a concentration of 1.5–5 × 107 (log10 7.17–7.70) cfu/mL in the test mixture at the beginning of exposure (N0). 27
This is mathematically necessary to detect a ≥ 5 log10 reduction, given the lower limit for cfu quantification. Reaching this density with liquid cultures of Streptococcus pneumoniae as investigated here was challenging. Indeed, S. pneumoniae displays a particularly effective quorum sensing system that activates several potent autolysins. Therefore, instead of using standard culture conditions with BHI, it would have needed optimized culture conditions. 29 Alternative options would be to concentrate bacteria by centrifugation and resuspension or by a direct resuspension of bacteria grown on plates to reach bacterial density able to fulfill the EN 13727 conditions.
With 1-min contact time, only a few surviving colonies of S. pneumoniae could sporadically be documented at the lowest tested concentration of glucoprotamine-containing biocide (0.1%). With higher concentration or contact time, we did not observe bacterial growth. Although we could not document a 5 log10 reduction because of the insufficiently high initial bacterial density, we still consider S. pneumoniae to be highly susceptible to the disinfectant.
MRSA has proven to be more resistant to glucoprotamine, requiring 1 min exposure to a 1% solution to achieve the 5 log10 reduction in bacterial density. This criterion was also reached when the exposure time was increased to 30 min for the lower concentration of 0.25%. These results are in line with a previous study showing that a working concentration of 0.5% of product A with 1 min contact time was sufficient to result in over a 5-log reduction for all bacterial isolates tested (including S. aureus), regardless of their antibiotic resistance pattern. 12
High efficacy of PAA was observed in this study. Even at an exposure time of 1 min, only the lowest concentration (0.1%) exhibited visible postexposure bacterial growth on the filters for both strains. The calculated germ reduction R was below 5 log10 for S. pneumoniae cultures for the reason given above, but PAA can be considered as highly effective against both strains.
Concerning MRSA, our results are slightly superior to what has been found by others, where a treatment of 30 min with concentrations of PAA similar or higher than those used in the present study was not sufficient to reach the 5-log reduction. Higher concentrations of bacteria in the test suspension, different bacterial strains, and difference in the methodology (chemical neutralization of the biocide) may explain this difference. It should also be stated that PAA was highly efficient against other strains, such as Escherichia coli or S. aureus. 7
TSB, FBS 10%, or NaCl 0.85% as different organic loads influenced neither glucoprotamine nor PAA efficacy, which is consistent with what had been reported previously.2,5,8,9 For these tests, both biocidal products have been applied at a concentration of 1%. We cannot exclude an effect of organic matter when compounds are used at lower concentrations.
The choice of interfering substances for our test was driven by practical considerations, based on situations mostly encountered in research laboratories working with pathogens. However, tests would need to be adapted by using other interfering substances, according to specific fields of application. For example, since PAA is readily inactivated by blood,2,5 the test should be adapted accordingly, should the liquid waste contain significant amounts of blood. In that context, EN 13727 (paragraph 4) indicates the use of 3 mL/L sheep erythrocytes and 3.0 g/L bovine albumin as interfering substance for testing dirty conditions.
Using a contact time of 1 min on MRSA as test condition, we demonstrated that the two compounds varied in their stability profile, once diluted to their working concentrations. Solutions of either 0.5% and 1% glucoprotamine retained most of their activity after 4 weeks (28 days). At a concentration of 0.5%, the germ reduction (R = 4.3 log10) remained similar in comparison to the freshly prepared solution (R = 4.27 log10).
At a concentration of 1%, R decreased from 5.26 log10 to 4.88 log10. The manufacturer suggests working concentrations of 1.5%, 2.5%, and 4% with contact times of 60 min respectively 30 and 15 min. Based on the findings presented earlier, the concentrations and contact times indicated by the manufacturer may be higher than needed. For this product, the manufacturer also recommends that the diluted solution, when unsoiled, should be replaced after 14 days.
PAA, in contrast, is known to be unstable and decompose into acetic acid, water, and oxygen.2,5 Accordingly, we demonstrated a relatively rapid decrease in antimicrobial activity after the solution was prepared. Already on the third day, the germ reduction R had fallen below <5 log10 levels (R = 4.83 log10 at 0.25% and R = 4.82 log10 at 0.5%), with R falling to 4.5 log10 for both tested concentrations on the fifth day (Figure 4C). This is in line with what had been reported in earlier studies. 30
The manufacturer of product B recommends working concentrations and exposure times of 1% at 60 min and 2% at 5 min. These values are above what seems to be required in most cases for safe inactivation with a freshly prepared solution. Sufficient activity could therefore be reached over a longer period after preparation of the working solutions, even when the manufacturer recommends those to be freshly prepared.
It should also be considered that, similar to antibiotics, a reduction in biocide to sub-optimal concentration could possibly lead to selection pressure and contribute to the survival of less susceptible strains in the population, which could lead to biocide resistance or at least tolerance. However, due to the rather unspecific mechanisms of action of disinfectants and the usually high concentrations used under practical conditions, resistance has so far been of little concern, 31 but should not be neglected. Indeed, an enhancement of antibiotic resistance due to low-level exposure to biocides has been demonstrated for both gram-positive and gram-negative bacteria.32,33
We observed an increase in the numbers of cfu on filters after an additional 24 h incubation (total 48 h) after exposure to PAA (not shown). This possible post-exposure growth delay effect cannot be attributed to residual bactericidal or bacteriostatic activity on filters after washing.
During the validation of the disinfectant neutralization, there was no significant difference between the density of the test solution determined before filtration and from the bacteria retained on the filters, indicating that the disinfectant was efficiently removed by filtration. A similar observation was made for glucoprotamine.
PAA shows a very rapid biodegradation profile in all environmental compartments and is not considered as a persistent organic pollutant. 34 Glucoprotamine, in contrast, is classified as very toxic to aquatic life, but has been originally claimed to be easily biodegradable in water treatment plants, thus having favorable ecotoxicological properties.8,17 Limited data are, however, available on the aquatic toxicity of glucoprotamine. Acute toxicity tests revealed an EC50 of 0.5 mg/L on daphnia and LC50 of 0.6 mg/L on fishes. 8
Previous versions of the safety data sheets provided by the manufacturer also mentioned an acute LC50 of 1–10 mg/L for an exposition of 96 h in fishes, but in the most actual versions, this information has been removed. We therefore took the opportunity of having an in-house zebrafish facility and performed our own assessment, although this is not a mandatory requirement. Using the acute zebrafish embryo toxicity test, we found LC50 values (3–3.5 mg/L) that are in range with what has been published previously.
Using another behavioral test, we also noticed that surviving larvae at sublethal concentrations also displayed altered behavior that may be related to general health conditions or neurotoxicity.23,24 Although we did not determine an EC50 for this observation, it is of concern, especially considering the suggestion made by others that the predicted environmental concentration of glucoprotamine may reach a level superior to those of the predicted no-effect concentration. 18
Conclusion
Here, we present a validation protocol derived from the standard EN13727, used by manufacturers to prove the antibacterial efficiency of their chemical disinfectant and antiseptic products before marketing. Our protocol is meant to validate in-house chemical disinfection of liquid waste containing bacteria as an alternative to autoclave.
Using a simple and affordable filtration system, the test is especially suited for liquid waste of simple composition, such as bacteria in their culture medium, or liquids containing a relatively low amount of organic load. Some of the steps in EN13727 were simplified as described in more detail earlier. This should enable laboratories with limited resources and infrastructure to perform proper own in-house validation. A crucial point is to proceed to all necessary process validations.
The laboratory may choose to perform the necessary process validations in parallel or independently of the main quantitative suspension test. In the latter case, special care should be taken to respect constant test conditions throughout the process to validate the chosen inactivation procedure.
As a practical example, we tested PAA and glucoprotamine on two gram-positive bacteria often investigated in our laboratory. Using the test, glucoprotamine and PAA have proven to be very efficient antimicrobial agents even at lower concentrations than those recommended by the manufacturer. Using the concentrations and the strains described here, we would consider a 1 min contact time as sufficient.
However, in a practical setting, especially considering the presence of more resistant microorganisms, higher organic load, or the presence of biofilms, we consider that for both biocides, a concentration of 0.5% for at least 1 h, or better ON, before disposal can be recommended.
Finally, the definitive choice should also include organizational aspects. Glucoprotamine is more stable, and the working solutions can be used for longer periods, but an uncertainty still exists about its ecotoxicological properties and the need to treat the liquid waste as hazardous material. On the other hand, PAA quickly loses its effect and solutions should be replaced more often. However, the degradation products are non-toxic and largely harmless, so that they can be disposed of in the wastewater.
Footnotes
Acknowledgments
The authors thank Prof. Stephen Leib for providing the S. pneumoniae strain and Prof. Yok-Ai Que for providing the S. aureus strains.
Authors' Contribution Statement
J.R.: conceptualization; methodology; formal analysis; investigation; validation, visualization; and writing—original draft (equal). N.D.L.: conceptualization (supporting), methodology; formal analysis; investigation; visualization; writing—original draft (supporting); and reviewing and editing (supporting). F.L.L.: investigation (supporting). S.L.L.: resources; writing—review and editing (supporting). M.G.: conceptualization (equal); supervision (equal); methodology (supporting); and writing—review and editing (equal). J.F.: conceptualization (supporting); methodology (supporting); and writing—review and editing (supporting). K.S.: conceptualization (equal); supervision (equal); methodology (supporting); writing—review and editing (supporting); and resources. D.G.: conceptualization (equal); supervision (equal); methodology (supporting); formal analysis (supporting); visualization (supporting); writing—original draft (equal); and reviewing and editing (equal).
Availability of the Data
The datasets used and/or analyzed in this study are available from the corresponding author on reasonable request.
Ethical Statement
All experimental procedures involving zebrafish larvae were according to Swiss animal welfare regulations for the care and use of animals (Holding and Breeding License BE 3/2012).
Authors' Disclosure Statement
The authors declare that they have no commercial or associative interests that represent a conflict of interest in connection with the manuscript at the time of publication.
Funding Information
This project received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors, but benefited only from intramural funding.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
