Abstract
Introduction:
Snap-cap microcentrifuge tubes are ubiquitous in biological laboratories. However, limited data are available on how frequently splashes occur when opening them. These data would be valuable for biorisk management in the laboratory.
Methods:
The frequency of splashes from opening snap-cap tubes using four different methods was tested. The splash frequency for each method was measured on the benchtop surface and on the experimenter's gloves and smock, using a Glo Germ solution as a tracer.
Results:
Splashes occurred very frequently when opening microcentrifuge snap-cap tubes, no matter which method was used to open the tube. The highest rate of splashes on all surfaces was observed with the one-handed (OH) opening method compared with two-handed methods. Across all methods, the highest rate of splashes was observed on the opener's gloves (70–97%) compared with the benchtop (2–40%) or the body of the researcher (0–7%).
Conclusions:
All tube opening methods we studied frequently caused splashes, with the OH method being the most error-prone but no two-handed method being clearly superior to any other. In addition to posing an exposure risk to laboratory personnel, experimental repeatability may be affected due to loss of volume when using snap-cap tubes. The rate of splashes underscores the importance of secondary containment, personal protective equipment, and good protocols for decontamination. When working with especially hazardous materials, alternatives to snap-cap tubes (such as screw cap tubes) should be strongly considered. Future studies can examine other methods of opening snap-cap tubes to determine whether a truly safe method exists.
Introduction
Standard microbiology laboratory apparatus and procedures have previously been shown to routinely generate splashes and aerosols that can pose biosafety risks to personnel, including common devices, such as centrifuges and mixers, as well as procedures such as pipette mixing, serial dilution, plating, and opening sample tubes.1–3 Snap-cap microcentrifuge tubes (also known as snap-top or plug-top tubes) are frequently chosen for convenience, but surprisingly little research has been conducted into how frequently contaminated splashes or aerosols are generated with their handling, and whether any specific opening methods reduce the risk of contamination, and are thus inherently safer.
Biosafety recommendations and guidance documents generally state that snap-cap tubes are less safe than other types of tubes. For sample transfer within the laboratory, the World Health Organization (WHO) Laboratory Biosafety Manual recommends using screw-capped tubes over ones with snap-on lids. 4 When working with potentially hazardous or infectious material, the Canadian Biosafety Handbook and other guidance documents specifically advise using externally threaded tubes with screw caps to minimize lid surface contamination.5–7
The strongest language is found in the Guidelines for Safe Work Practices in Human and Animal Medical Diagnostic Laboratories, recommendations from a CDC-convened, Biosafety Blue Ribbon Panel. 8 It states that microcentrifuge and other snap-cap tubes can produce aerosols and splatter when opened, and screw-cap tubes can reduce risk. Yet, no guidance documents, including the HHS Biosafety in Microbiological and Biomedical Laboratories (BMBL) and the WHO's Laboratory Biosafety Manual, prohibit or even strongly discourage the use of snap-cap tubes.4,9
Moreover, many of us remember being trained when first entering our graduate or undergraduate laboratories, to open snap-cap tubes with two hands to reduce the frequency of splashes and spills. However, we were unable to identify any research that forms the evidence basis of that recommendation.
Limited research has been conducted into the potential risks of snap-cap tubes. Pottage et al 3 simulated a series of common microbiological procedures and accidents with high concentration spore suspensions (107–109 CFU/mL), including flicking open vortexed Eppendorf tubes with the thumb, while measuring aerosols generated with air samplers. Of all procedures and accidents tested, flick opening of these snap-cap tubes generated the highest levels of aerosols with both suspension titers (up to 104 CFU/m3). Further, this event produced the second-highest number of 105 discrete splashes, behind only an accidental tube drop, on the surface under where the procedure was completed in a five-minute testing period, although the total number of individual tube openings during this period was not reported.
The objective of this study was to quantify the frequency of splashes that are produced from opening snap-cap tubes, and if any method of opening the tubes was superior at preventing splashes and aerosols to any other. Four opening methods were tested: a one-handed (OH) thumb extension technique and three different two-handed techniques. Using diluted suspensions of the fluorescent tracer GloGerm gel, the frequency of splashes was measured on the experimenters' gloves and smock covering the front of their body, and on the surface below the experimenter's hands.
In addition to determining the safest way to open snap-cap tubes, the data obtained from this study can be used to inform biological risk management for procedures using such tubes. Therefore, although Pottage et al focused primarily on quantifying the aerosols generated from opening snap-cap tubes, our study focused on determining how frequently splashes occur on various surfaces when opening snap-cap tubes. In addition, although Pottage et al only investigated one opening method (flicking the top open with the thumb), this study compared four methods.
Materials and Methods
Experimental Procedures
A one to four dilution of Glo Germ gel mixed with water was prepared. Glo Germ, 10 which contains plastic particles visible under blacklight (ultra-violet-A light) that simulate the presence of microorganisms, was used as a tracer solution for the identification of splashes on the experimenter and benchtop surface. The benchtop was covered in a sheet of black paper before each experiment to facilitate visualization of the splashes. The Glo Germ solution was stored at room temperature and re-suspended with a stir plate before each use. Before the experiment, calibration testing was preformed to determine how much Glo Germ could be visualized on the benchtop surfaces under the ambient light of the laboratory. It was determined that splashes as small as 0.1 μL could be visualized.
Eppendorf brand 1.5 mL plastic snap-cap tubes filled to 2/3 full (1 mL) with the Glo Germ dilution were used. Four snap-cap tube opening methods were tested, pictured in Figure 1: (1) OH, with the thumb extending to open the cap; (2) two-handed, with one hand stabilizing the tube and the thumb on the other hand extending to open the cap; (3) two-handed, with one hand stabilizing the tube and the thumb on the other hand pulling from above to open the cap; and (4) two-handed, with one hand stabilizing the tube and the index finger and thumb on the other hand working together to leverage open the cap.
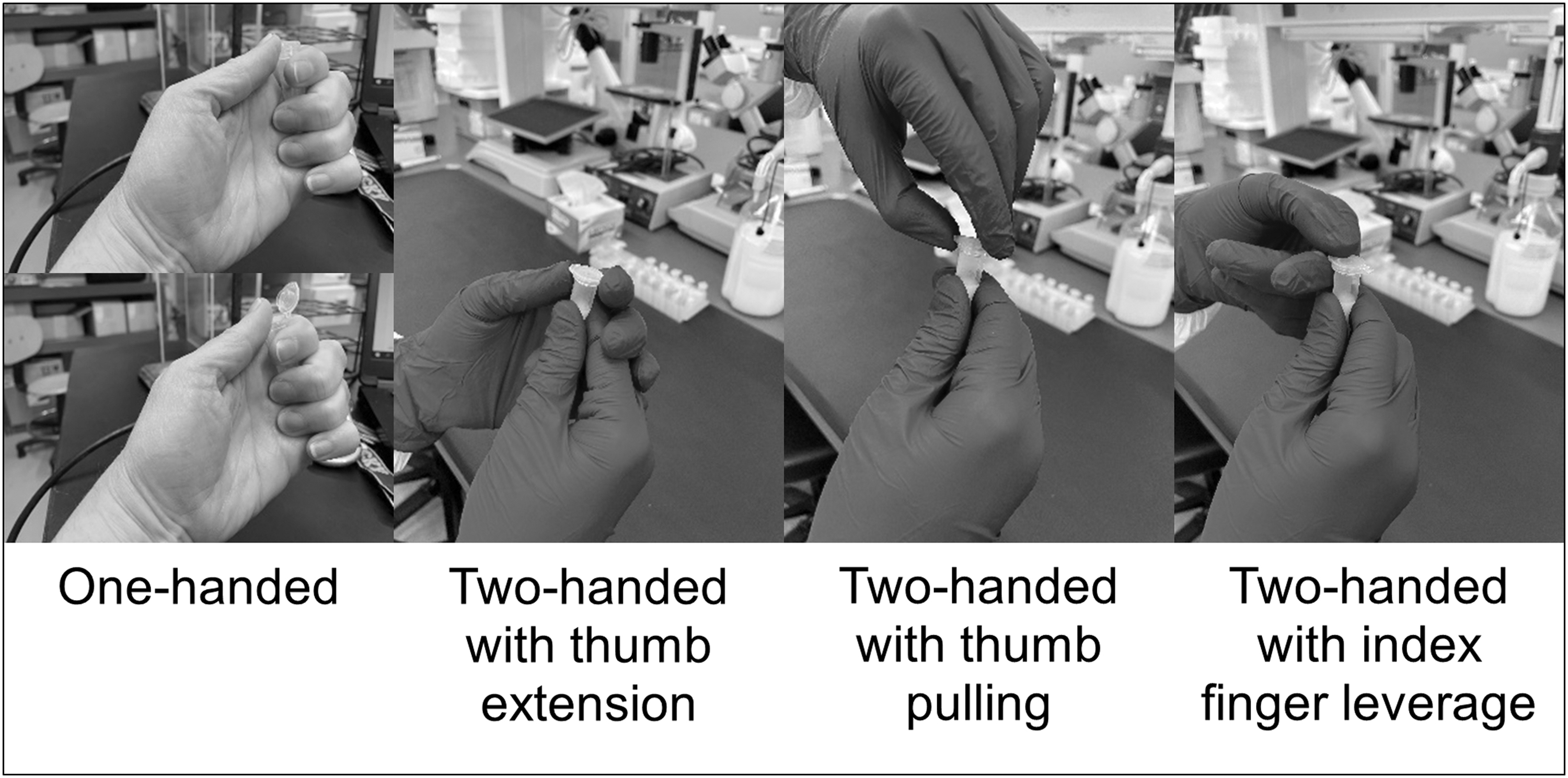
Images of the four snap-cap tube opening methods tested. The OH method is demonstrated with ungloved hands, and the two-handed methods are shown with gloved hands. OH, one-handed.
For each of the opening methods, five experimental sets were conducted, each consisting of filling and opening 20 snap-cap tubes. At the beginning of each run, the date and time were recorded, along with the names of the people filling, opening, and recording. All experimenters were undergraduate student researchers, with at least some previous laboratory experience and a basic foundation in research techniques and methodologies. The filler opened all 20 snap-cap tubes, aligned them in a clean rack, and used a bottle top dispenser to fill each with 1 mL of the Glo Germ solution.
The snap-cap tube exteriors were then checked with a black light for Glo Germ contamination and cleaned with isopropyl alcohol until contamination could no longer be detected with the black light. A total of 400 samples were tested, 100 per opening method, which were divided into 20 tube-sets.
The opener donned a clean white smock and nitrile gloves, then inverted the first tube once, and opened it using the designated method for that set. The number of Glo Germ splash droplets on the benchtop paper and experimenter's gloves and smock were visually counted with a blacklight. The results were coded as “Yes” if any drops were observed, “No” otherwise. Nitrile gloves contaminated with Glo Germ were removed and discarded before opening the next tube, whereas droplets on the benchtop surface and opener's smock were circled with a colored pen, enabling the paper and smock to be reused several times.
Statistical Interpretation
We started this project by writing down the analysis plan at a high level in a preregistration document. The mean number of splashes on the benchtop surface and experimenters' gloves and smock were calculated for each of the four opening methods. Human and environmental factors can affect the rate of errors, and the data are unbalanced across covariates. Therefore, the raw averages were also converted to estimated marginal means (EMM), using the emmeans R package.11,12 The EMMs are an “average of averages,” which control for variance between experimenters and thus better represent the true rate of splashes for each opening method investigated.
Next, we investigated whether the apparent differences in splash rate between opening methods were statistically significant. Models were fit to the results for splashes on the gloves, benchtop surface, and smock. For each splash measurement, the choice of models considered the identities of the filler, opener, and filler/opener combination as random effects. These factors were used as potential confounders, as considering the identities of the experimenters allowed correction for differences in their skills. The Bayesian Information Criterion (BIC), which balances model fit and complexity, was used to measure model fit. 13 We then selected the model with the best BIC score, and we tried to account for the resulting selection variability in our inference.
We had the option of choosing between multiple reasonable inferential methods. These options included how to define the comparison summary statistic, how to deal with model selection uncertainty, whether to use one or two-sided intervals, and how to validate our approach. We tested multiple iterations of analysis, and deviated from our preregistration at certain points, which are described in the Supplementary Data. Our final choice was to use the relative risk as a summary statistic and to validate our confidence intervals with a simulation study. These intervals are designed to control the FWER (family-wise error rate) for a given splash target (gloves/bench/smock), which means that intervals produced by this procedure would all contain the true relative risk in 95% of similar experiments. We believe that the bounds produced by these intervals would be worth testing in a confirmatory experiment.14,15 For more information, please see our statistical supplement (Supplementary Data).
Results
Table 1 contains the EMMs and raw averages of the rate of splashes on the benchtop surface and experimenters' gloves and smock for each of the four snap-cap tube opening methods tested. Across all methods, the highest splash rate was observed on gloves, with 70–97% of tube openings resulting in the experimenter's gloves being contaminated with the tracer solution. The lowest rate of splashes across all methods was observed on the opener's smock, with the tracer solution being detected on the smock less than 10% of the time.
Each method was tested in 5 sets of 20 tubes each (100 tubes total per method).
IFL, two-handed with index finger leverage; OH, one-handed; TE, two-handed with thumb extension; TP, two-handed with thumb pulling.
The highest rate of splashes on all surfaces (97%) was observed with the OH method (flicking the lid with the thumb) compared with two-handed methods, although all methods produced splashes on the gloves most of the time.
Then, the splash rate across opening methods on the gloves, benchtop, and smock were compared in a pairwise manner, beginning with the splash rate on the opener's gloves. The best-fitting model used the identity of the filler/opener pair (as a random effect). No statistically significant differences in error rates between methods were observed for splashes on gloves (Figure 2).
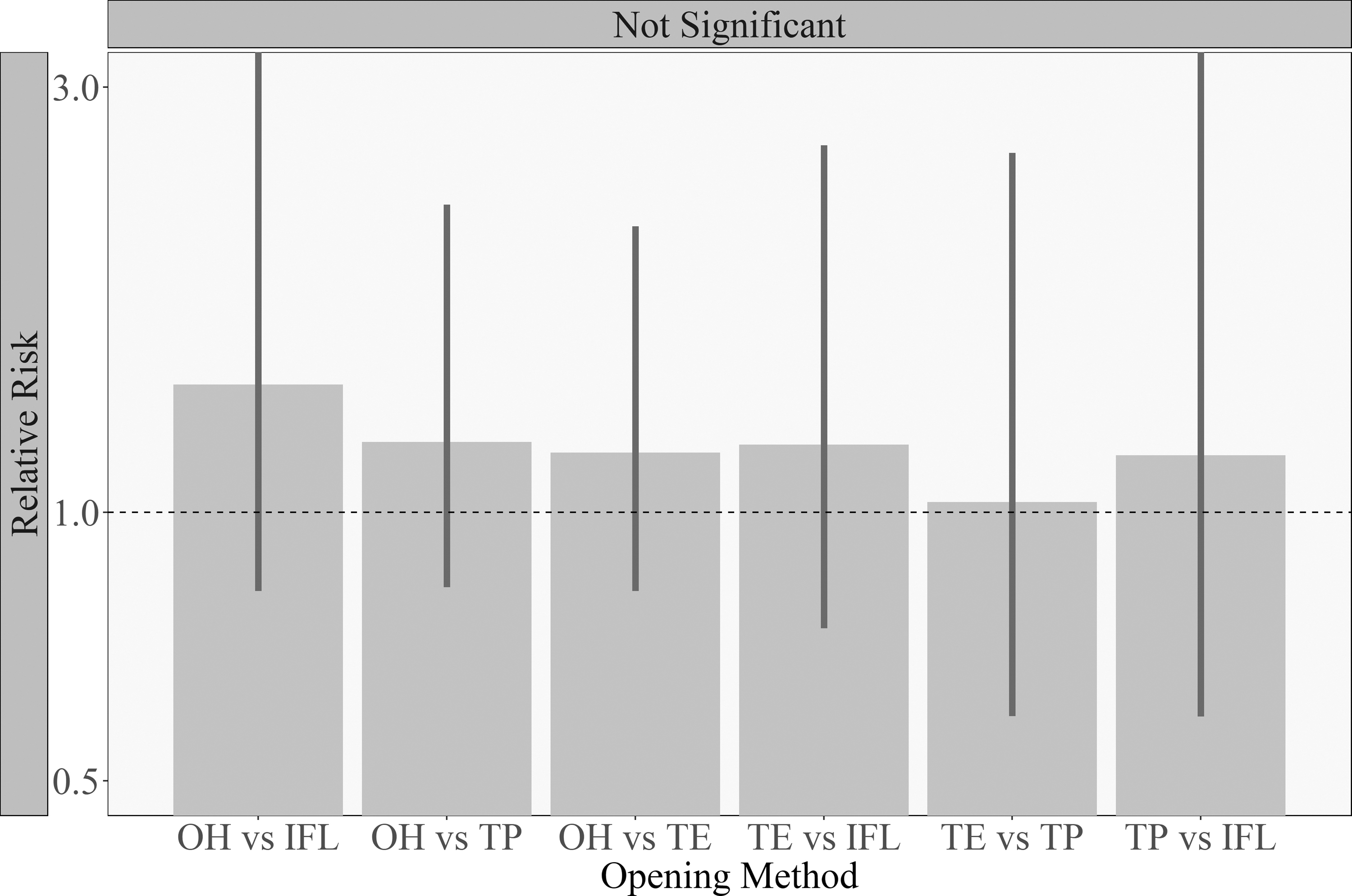
Testing for significant differences in splash rates between methods for drops on glove. Inferential results for the rate of splashes on the tube opener's gloves, showing the increase in risk as a ratio of the splash rates for pairwise method comparisons. The bars represent confidence intervals for the relative risk. Solid vertical bars are confidence intervals for the relative risk that keep the FWER below 5%. Bars that reach the top of the plot were cut off for readability. When the error bars overlap 1 (dotted line), a difference between the opening methods cannot be corroborated. For example, the relative risk of the OH versus index finger leverage methods is the ratio of their EMMs: 0.97/0.7, which is ∼1.4. The lower and upper bound of the confidence interval for this pairwise comparison are 0.8 and 3.3. For the interpretation and justification of these values, please refer to the Materials and Methods section and the statistical supplement. EMM, estimated marginal mean; FWER, family-wise error rate; IFL, two-handed with index finger leverage; OH, one-handed; TE, two-handed with thumb extension; TP, two-handed with thumb pulling.
Next, the model that best fit the splash rate on the benchtop considered the confounding effect of the filler's identity (as a random effect). In pairwise comparisons of the splash rate between opening methods (Figure 3), several significant differences were observed. These were between the OH versus thumb pulling, OH versus thumb-extension, and finally index finger leverage versus thumb pulling methods. The observed relative risk of the EMMs for the first comparison was especially large (∼20), and the lower limit of our confidence interval is still large (∼2.8). Our interpretation of this result is that it indicates that thumb pulling is at least 2.8 times less likely to generate a splash on the benchtop compared with the OH method; however, since we did not intend to test this specific hypothesis, we do not want to claim that it has been corroborated by the data.
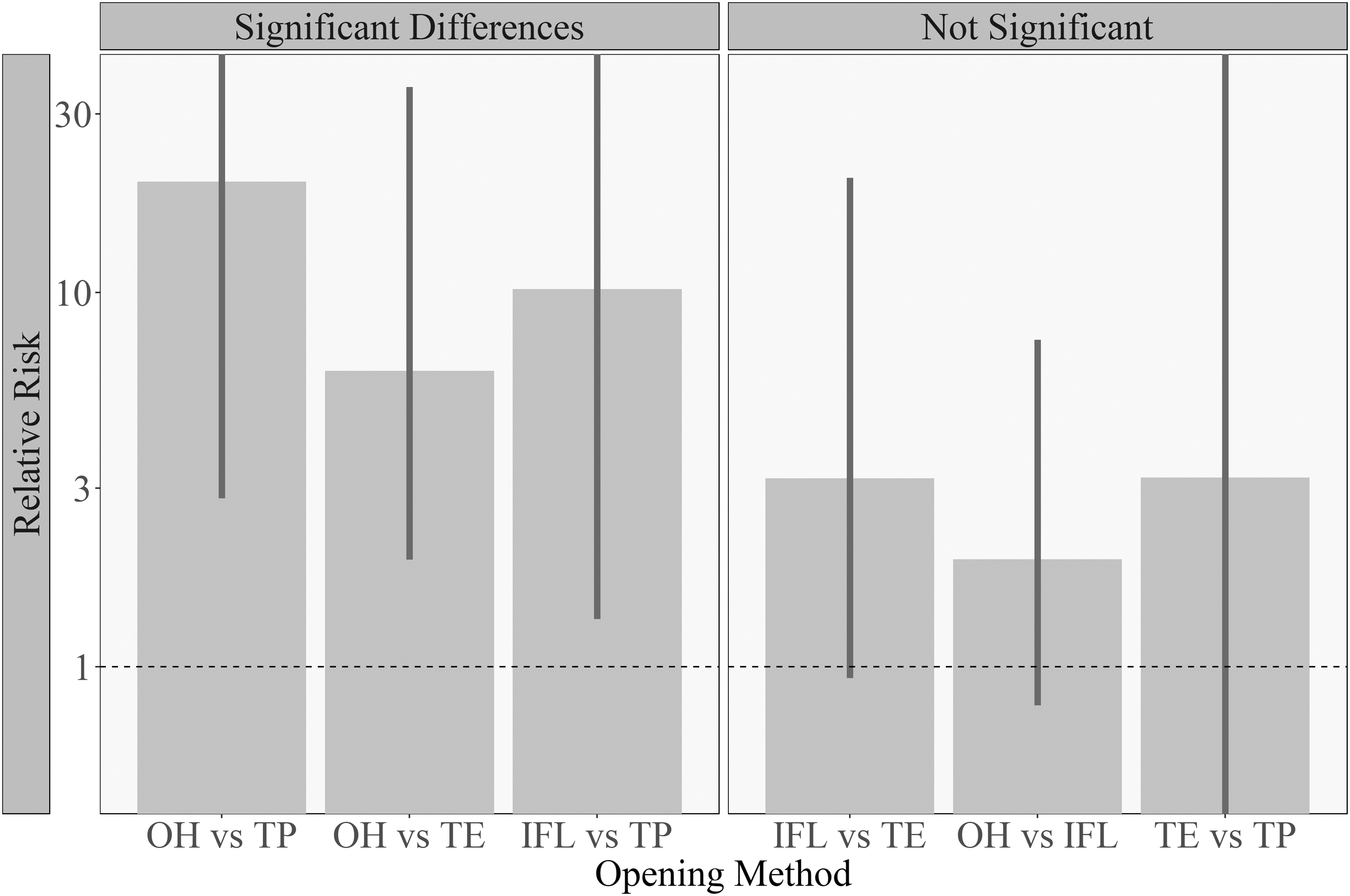
Testing for significant differences in splash rates between methods for drops on bench. Inferential results for the rate of splashes on the benchtop surface, showing the increase in risk as a ratio of the splash rates for pairwise method comparisons. The bars represent confidence intervals for the relative risk. Solid vertical bars are confidence intervals for the relative risk that keep the FWER below 5%. Bars that reach the edge of the plot were cut off for readability. When the error bars overlap 1 (dotted line), a difference between the opening methods cannot be corroborated. For example, the relative risk of the OH versus thumb-pulling methods is the ratio of their EMMs: 0.40/0.02, which is ∼20. The lower and upper bound of the confidence interval for this pairwise comparison are 2.8 and 368. For the interpretation and justification of these values, please refer to the Materials and Methods section and the statistical supplement.
Last, the model that best fit the splash rate on the tube openers' smocks was a logistic regression model with no other covariates or random effects. Overall, splashes on the tube opener's smock were observed far less frequently than on the opener's gloves or benchtop, with zero total splashes being reported using the two-handed thumb pulling method. The OH opening remained the most error-prone method, with 7% of microfuge tube openings resulting in splashes on the experimenter's smock. However, no statistically significant differences could be established between any of the methods (not shown).
Discussion
Snap-cap tubes are ubiquitous in microbiology and other laboratories, and they are regularly used for routine laboratory procedures and sample transport or storage. However, although some biosafety recommendations and guidance documents advise that snap-cap tubes are generally less safe and more prone to creating splashes or aerosols compared with screw-cap tubes, very little data are identified to quantify this risk or reinforce this guidance. These data are needed to further inform biorisk management for procedures that involve the use of these tubes.
The results from this study showed that splashes happen very frequently when opening snap-cap microcentrifuge tubes, for all opening methods tested. Snap-cap tubes are often held and opened with one hand, as the lid can be easily flicked open by extending the thumb. However, this OH opening method was the most likely to cause splashes, with splashes observed on the experimenter's gloves in 97% of trials, on the benchtop in 40% of trials, and on the experimenter's smock in 7% of trials.
Although two-handed opening methods were observed to cause fewer splashes than the OH method, possibly because the tube can be gripped more securely and stabilized, or the lid may be opened with less force, similarly high splash rates were seen with the three two-handed methods tested. This rate of splashing was particularly noticeable on the experimenter's gloves, where splashes were observed 70–83% of the time across all two-handed opening methods.
Therefore, although opening snap-cap tubes OH is the most error-prone method, no two-handed method was clearly superior and all tube opening methods frequently produced splashes. The two-handed thumb pulling method could be considered as the tentatively safest choice, given that it had a significantly lower splash rate than the OH and index finger leverage methods on the bench. It is also not yet shown to be statistically inferior to the index finger leverage method for splashes on gloves.
In addition to frequently causing splashes, Pottage et al 3 found that OH opening of snap-cap tubes containing Bacillus atrophaeus spore suspensions (107–109 CFU/mL) generated high concentration aerosols (up to 104 CFU/m3), which we did not measure. The spore concentrations in the aerosols produced from opening snap-cap tubes were higher than those measured in a range of other microbiological procedures and accidents, including from accidental spill tests that involved knocking over tubes and dropping them from a height of 18–20 cm.
Taken together with the results from this study, snap-cap microfuge tubes appear to be frequent and significant potential sources of inadvertent splashes, spills, and aerosols in the laboratory. Therefore, alternatives to snap-cap tubes should be considered when working with extremely hazardous materials or secondary measures should be implemented to further mitigate the risk associated with splashes from these tubes.
If they must be used, they should be opened carefully using the two-handed with thumb pulling method until more data are available showing superiority or equivalence with other methods. Moreover, if snap-cap tubes must be used, methods should be modified to reduce contact of fluid with the lid (e.g., opening only after a brief centrifugation). These results also highlight the value of secondary containment, personal protective equipment, and regular and thorough decontamination of the workspace.
Given the frequency that material is ejected from the tubes and the amount that is ejected (according to data in Pottage et al), our combined results suggest that snap-cap tubes should be avoided when the results or repeatability of the results is highly dependent on the volume retained in the vessel. If most tubes lose an unpredictable volume of their contained liquid each time they are mixed and opened, predicting the final concentration of any reagent added after the tube is opened will be impossible to determine precisely.
Our study necessitated complex statistical models, because we used several experimenters to ensure that this research was accessible to undergraduates, to minimize tedium and not bias the study by the skill level of a particular experimenter. The statistical model selection step could be made easier by narrowing the range of possible models. More focused comparisons of human operator roles could reduce possible confounders.
For example, the same study could be tried with a single opening method, and a factorial design of fillers and openers. This approach could establish whether there is a significant effect on splash rate from the fillers or openers alone, or if there are positive or negative synergies from some teams.
Another important potential improvement is to allow for some crossing between experimenters and day of the week. We hypothesized that day of week could influence splash rates but were unable to test that association using our design. It could be that experimenters pay less attention to their work on a Friday afternoon compared with a Monday morning, and this could lead to higher splash rates on Friday afternoon. It is possible that this day of the week effect could be very context dependent and even reversed for some experimenters.
Therefore, we recommend using a two-way block design with day of week and experimenter as blocks. This design need not be complete but should allow for estimates of the effect of both factors. Finally, we recommend that future studies improve on this work by randomizing the run order of sets to prevent possible biases from experimenters improving their reliability over time.
This study is just one of many that should be conducted to understand and minimize risks when working with microcentrifuge tubes. Further experiments could explore how the volume contained influences risks of splashes (ours were filled with 1 mL only) and could test other methods for opening the tubes (such as opening with one hand while the tube is immobilized in a rack, or opening with a plastic opener), for mixing the fluid inside (such as pipetting up and down or vortexing) and other mitigations (a quick centrifuge or allowing time for the liquid to settle).
The rate of spills and splashes could be directly compared with a variety of other types of small tubes with stoppered and screw capped lids (with or without additional seals). Given the ubiquity of these tubes in the modern biology laboratory, further empirical exploration into these questions would certainly benefit biorisk management.
Moreover, this study is an example of one of many that could be conducted in small, liberal arts colleges because it is accessible to undergraduate-level researchers and laboratories with limited budgets. Although simple, studies of this type can fill critical evidence gaps in biorisk management.
Footnotes
Acknowledgments
The authors acknowledge the generous funding of the Open Philanthropy Project that supported this work. The authors would like to thank Barbara Johnson, Ryan Ritterson, Erin Lauer, Amanda Lee Skarlupka, and Jim Nolan for their review and commentary on this work. They thank Laurel MacMillan for her review of the statistical methods.
Authors' Contributions
H.W.: Formal analysis, writing (original draft, review and editing); A.A.C.: Writing (original draft, review and editing); K.K.: Project administration, writing (review and editing); C.H.: Investigation; methodology; E.J.R., C.M., K.C., S.D., L.G., I.I., C.W., A.M., and A.V.: Investigation; A.F.: Methodology; R.R.: Project administration, methodology, supervision, writing (review and editing); R.C.: Conceptualization, methodology, writing (review and editing).
Authors' Disclosure Statement
No competing financial interests exist.
Funding Information
This study was funded by an Empirical Biosafety Research grant from Open Philanthropy.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
