Abstract
Introduction:
Emergency preparedness is not a novel topic. What has been novel is the fast pace at which organizations, including academic institutions, have had to adapt to infectious disease outbreaks since 2000.
Objective:
The goal of this article is to highlight the various environmental health and safety (EHS) team activities during the coronavirus disease 2019 (COVID-19) pandemic to ensure that on-site personnel was safe, the research could be conducted, and critical business operations such as academics, laboratory animal care, environmental compliance, and routine healthcare functions could continue during the pandemic.
Methods:
The response framework is presented by discussing first the lessons learned in preparedness and emergency response during outbreaks that occurred since 2000, namely Influenza virus, Zika virus, and Ebola virus. Then, how the response to the COVID-19 pandemic was activated, and the effects of ramping down research and business activities.
Results:
Next, the contributions of each EHS unit are presented, namely, environmental, industrial hygiene and occupational safety, research safety and biosafety, radiation safety, supporting healthcare activities, disinfection, and communications and training.
Discussion:
Lastly, a few lessons learned are shared with the reader for moving toward normalcy.
Introduction
Emergency preparedness is not a novel topic. What has been novel is the fast pace at which organizations, including academic institutions, have had to adapt to infectious disease outbreaks since 2000. The U.S. Centers for Disease Control and Prevention (CDC) and the World Health Organization (WHO) assembled and published guidance documents in record times for outbreaks caused by pathogens such as Influenza virus, Zika virus (ZIKV), Ebola virus (EBOV), and most recently Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2).
The WHO published Emergency Response Framework in 2017 1 and, in 2021, the Guidance on Preparing for National Response to Health Emergencies and Disasters. 2 Similarly, the CDC 3 and the WHO 4 published clinical and laboratory biosafety guidance for handling samples potentially infected with SARS-CoV-2. Critically, during the coronavirus disease 2019 (COVID-19) pandemic, research studies were available online without completing the peer-review process. In the health and safety realm, earlier publications related to other outbreaks, fast-changing guidelines from regulatory agencies, and other publications helped to guide decisions made regarding risk assessment information associated with virion shedding, transmission, viral load in certain body fluids, reuse of respiratory protection, etc.
Emory University, an academic institution in the Southeastern United States, dealt with the COVID-19 pandemic by bringing together stakeholders and units across the university and health care to provide guidance. Units responding to the pandemic included emergency preparedness and response, academia, research, healthcare, Campus Life, Campus Services (CS), procurement services, and environmental health and safety (EHS). The goal of this article is to provide an account of the EHS activities at an academic medical institution, namely Emory University, to ensure that on-site personnel were safe, COVID-19-related research could be conducted, and critical business operations such as academics, laboratory animal care, environmental compliance, and routine health care functions could continue during the pandemic.
Unexpectedly, other skills became relevant and were frequently used by EHS and Emergency Response professionals. The skills will be presented in the context of participating as stakeholders in leadership activities to provide risk-based advice to address institutional needs.
Pre-Pandemic Preparedness: Lessons Learned from Previous Outbreaks
Vaccine development has accelerated over time due to advances in molecular biology and the experience acquired in prior outbreaks such as Influenza H1N1, Ebola virus, ZIKV, and SARS-CoV-2 as shown in Table 1. Emory University's decisive response to managing the COVID-19 outbreak was predicated on its experience in addressing other infectious disease outbreaks with global significance. The institution responded to infectious disease outbreaks in the context of research operations.
Infectious diseases outbreaks from 2009 to 2020
Travel associated.
EBOV, Ebola virus; EUA, emergency use authorization; FDA, Food and Drug Administration; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; PHEIC, Public Health Emergency of International Concern; WHO, World Health Organization; ZIKV, Zika virus.
The first response was to the Influenza Virus H1N1 outbreak in 2009. 5 During this outbreak, Emory University coordinated its response efforts through the Office of Critical Event Preparedness and Response (CEPAR). 5 Research activities were supported by the Environmental Health and Safety Office (EHSO). The lessons learned from the Influenza H1N1 outbreak included conducting rapid and comprehensive risk assessments to ensure appropriate use of personal protective equipment (PPE) and waste management, designing agent-specific training for different audiences (i.e., researchers, animal handlers), and designing medical alert cards.
In 2014, during the EBOV outbreak, Emory's response evolved in three phases: preparedness, medical care, and training. The preparedness phase involved the maintenance of the Serious Communicable Disease Unit (SCDU) in collaboration with the CDC since its inception in 2002.16,17 The lessons learned from the EHS perspective were drawn from the second and third phase. These activities involved day-to-day risk assessment for donning and doffing of PPE, training, environmental services, waste management, facilities, transportation, occupational health monitoring, preparation for other medical specialties such as pediatrics, and obstetrics, walk-throughs of medical spaces, emergency response to spills, and just-in-time training, and designed easy-to-follow donning and doffing checklists.
In late 2015, ZIKV cases were reported in the Americas. The first cases of ZIKV were reported in the U.S. Territories with warmer temperatures such as Puerto Rico, and the cases in continental United States were associated with travelers to areas where ZIKV was ramping. 18 From the research perspective, samples were transferred to multiple laboratories on campus to conduct immunological analyses. The Emory EHSO supported the research community by conducting risk assessments for in vitro and in vivo research. The biosafety team established standard operating procedures, and an occupational health review process for screening of personnel assigned to handle samples potentially infected with ZIKV. In addition, the risk assessment dictated the PPE to be used, transfer of samples, disinfection, waste management, and housing of animals. 19
Activating the Emergency Response to COVID-19
Emory University's Office of CEPAR is charged with planning and coordinating emergency response to catastrophic events affecting the Emory community and its surroundings. Accordingly, in December of 2019, CEPAR began monitoring the U.S. CDC and WHO alerts regarding cases of respiratory illnesses occurring in China. The CEPAR Emergency Support Function Group was activated after the first case of COVID-19 was detected in the United States.
The CEPAR leaders delivered a formal presentation, including what was known at the time to university executive leadership and the President's Leadership Team in early March of 2020. The formal Incident Command structure was activated on March 19, 2020. The overall objectives for the University's response included:
Work with State and Local Public Health to develop mitigation strategies to protect the Emory community to maintain the health and wellness of our community Work with business units that provide essential functions to ensure continuity plans are in place that will ensure the delivery of their function Develop and implement contingency planning for academic affairs Manage supply chain disruption to support essential functions Maintain financial control strategies and track costs of the COVID-19 response Develop internal and external communication strategies to inform the broader Emory community Develop and implement contingency planning for critical research activities Preserve the essential business functions of our community Preserve the academic mission of our community
The CEPAR Incident Command (CEPAR IC) collaborated with State and Local Public Health officials to implement risk mitigation strategies to protect the Emory community. In addition, they supported all internal and external communications. During the activation of the response, the CEPAR IC supported the schools' business units, ensuring that continuity plans were activated for the delivery of their core functions, and supported the Provost's Office activities related to academic continuity. Logistically, CEPAR IC managed supply chain disruptions to support essential functions, maintained financial control strategies, tracked costs of the COVID-19 response, and supported the development and implementation of contingency planning for critical research activities. A matrix was developed considering key indicators for the purpose of defining safe activities that supported the objectives and the essential operations of the university. The key indicators included:
Manageable community point prevalence and real-time transmission of COVID-19
Demand for health care services
Testing capacity using reverse transcriptase-polymerase chain reaction with a rapid turnaround time
Adequate isolation and quarantine capacity
Capacity to conduct robust contact tracing
Adherence to infection control measures
Availability of critical supplies
Key indicators were reviewed to inform the alert levels for the university, which include four levels.
Level 1: New Normal
Level 2: Moderate Alert
Level 3: High Alert
Level 4: Very High Alert
The highest alert that the university operated in was Level 3, High Alert.
Overall EHS-Related Pandemic Response
Early in the COVID-19 pandemic, Emory University developed a color-coded status compass that provided the operational expectations for students, faculty, and staff. The operating conditions status could be found in the University designated communications portal, and it was updated daily. 20
The EHSO supported the response to COVID-19 by providing expertise in occupational safety, industrial hygiene, infectious agents, and biosafety. Examples of immediate pandemic response included providing recommendations for reducing density on campus, actively disinfecting spaces, and preparing laboratories to conduct critical COVID-19 research. Subsequently, EHSO participated in research continuity activities such as respirator fit testing and training, preparation of high-containment laboratories, and prioritizing the approval of COVID-19 research protocols.
As the university prepared to reopen, EHSO worked closely with the Office of Human Resources (HR) to provide COVID-19-specific training for all faculty, staff, and students. The research community received COVID-19 training tailored specifically for their work environment. Examples of the different training documents created by EHS can be found in Supplementary Materials S1. This training was fundamental in providing strategies for safe operations, including disinfection procedures for faculty, staff, and students.
Additional written guidance was created to assist faculty and staff with implementing disinfection strategies. The EHSO assisted in the redesign of spaces for academia, performing arts, and athletics regarding density, distancing, PPE, and general health and safety practices.
The EHSO collaborated closely with other units supporting the COVID-19 response, as shown in Figure 1. These units included CS, HR, CEPAR, Student Health Services, and Procurement Services. The CS includes Facilities Management, Emory Police, Finance and Business Operations, Customer Experience, and Planning, Design, and Construction. During the COVID-19 pandemic, CS provided services to address the needs of the Emory Community, including Zero Waste Training to include details on reducing and diverting waste from COVID-19 disposable PPEs, Fresh Air/heating, ventilation and air conditioning (HVAC) Strategy and Campus Sanitization Plan, among others. 21 Our partnership with Procurement was crucial in ensuring that necessary supplies, such as disinfection products, supplies such as plexiglass screens, and PPE, were readily available. This required daily collaboration to review supply stocks and sourcing alternatives.
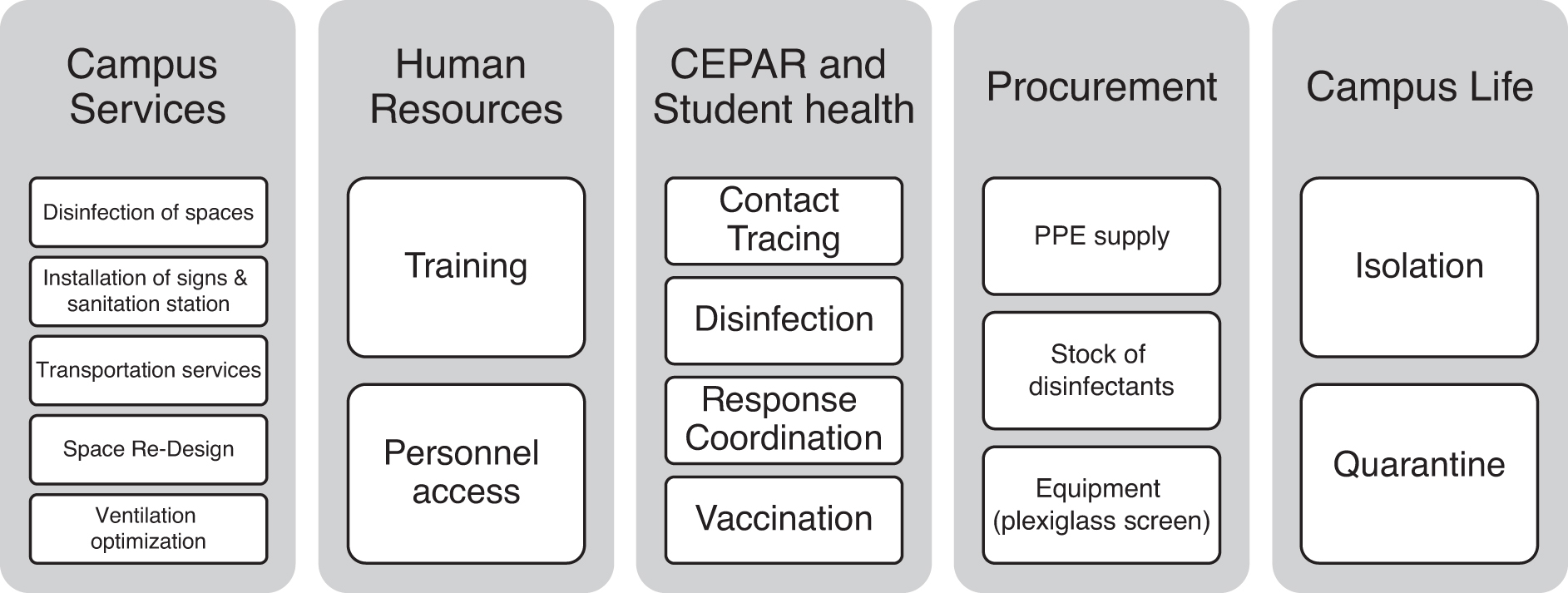
Emory University EHSO interacted with key units and departments during the COVID-19 pandemic. COVID-19, coronavirus disease 2019; EHSO, Environmental Health and Safety Office.
Between March 2020 and June 2021, all EHSO programs contributed to ensuring business continuity as described in Figure 2. Some programs included the number of events associated with activities. 22 Offices such as Radiation Safety continued to offer services during the pandemic to ensure business continuity, especially associated with health care. These programs are expanded upon later. The EHSO continues to be vigilant in providing guidance and support to protect our community from COVID-19.

Activities conducted by the EHS team between March 2020 and June 2021. Text in gold refers to COVID-19 specific activities; text in gray involves COVID-19 and regular business operations activities. This figure was presented in Poster #14 during the ABSA International Conference in October 2021.
Environmental Response
Because of the nature of the Environmental team's duties, the option to work “remotely” was rare. The EHS specialists were on-site daily throughout the entire pandemic because of the continuous research and health care operations. Functions related to hazardous waste management and receipt and tracking of radioactive materials remained active during the pandemic.
The Environmental team maintained the Spill Response phone line, which was available 24/7 to provide a resource for staff, students, and faculty who needed to request EHS services such as response to and cleaning of chemical spills, reporting potential exposures, requesting, responding, and decontaminating dorm rooms, office spaces, bathrooms, health care facilities, and vehicles that would have been used by suspected cases.
Industrial Hygiene and Occupational Safety Response
The expertise in industrial hygiene and occupational safety was paramount for ensuring that those who needed to use an N95 respirator had access to it. Personnel fit testing early in the pandemic included Emory's first responders, such as police and emergency medical services. In addition, fit tests had to be conducted multiple times on the same individual due to the varying models of N95s available because of the critically low supply chain. Risk communication was a critical component of this effort, as many faculty and staff requested N95 respirators who did not require them based on their occupational exposure risk. The industrial hygienists on the team worked and shared resources and best practices with colleagues outside of Emory. For example, early in the pandemic, a team from EHSO was deployed to fit test military personnel at a local Air Force base.
Industrial Hygiene Challenges During the Pandemic
The main industrial hygiene challenge during the pandemic was the respirator shortage and counterfeit respirators. The EHSO collaborated with the University's Procurement unit to verify potential sources of N95, KN95, and triple layered-surgical masks. Another challenge was the management of routine occupational safety and health programs when most resources were diverted to the COVID-19 response.
Radiation Safety Response
With COVID-19 restrictions on campus, Radiation Safety was unable to conduct various tasks to satisfy regulatory requirements. Communication with the State of Georgia was critical in assessing the risks, functions, and responsibilities. One example of tasks suspended (in accordance with conversations with State officials) include quarterly inspections and 6-month leak tests. Functions that continued during the pandemic included supporting radiotherapy patients.
A case in point was the conversion of in-patient I-131 therapy rooms to COVID-19 wards after clearance given by Radiation Safety. Other activities that continued during the pandemic also included radiological emergencies, collection, and distribution of dosimetry badges, and prioritization of a protocol review of studies that involved COVID-19 and radiation and/or radiation producing machines.
Biosafety Response
Early in the pandemic, it was clear that biological risk assessment would take front and center in research activities because the pathogen was novel. Recall that the first name associated with this Coronavirus was nCOV-19 for novel Coronavirus. The NIH Office of Science Policy issued guidance regarding the risk group and Institutional Biosafety Committee requirements for handling SARS-CoV-2 soon after the pandemic started. 23 The risk assessments were structured using the known and unknown factors presented in Table 2.
Known and unknown severe acute respiratory syndrome coronavirus-2 and coronavirus disease 2019 biosafety-related factors considered in risk assessment
Controls and risk mitigation practices were implemented to handle the virus and samples potentially infected with SARS-CoV-2 to ensure the safety of the laboratorians and the community, as shown in Table 3. Also included in Table 3 are the controls used for work with large and small laboratory animals, non-human primates, and hamsters.
Risk mitigation strategies used by the biosafety professional for handling severe acute respiratory syndrome coronavirus 2 and coronavirus disease 2019-related samples
ABSL, animal biosafety level; BSL, biosafety level; COVID-19, coronavirus disease 2019; PAPR, powered air purifying respirator.
Factors That Facilitated the Start of Work with SARS-CoV-2
Emory University already had trained personnel (researchers, veterinary staff, and animal care) working in biosafety level 3 (BSL-3) and animal biosafety level 3 (ABSL-3) facilities. The just-in-time preparation for initiating work with SARS-CoV-2 included completing agent-specific training, updating the Occupational Health Services (OHS) screening questionnaire, ensuring availability of PPE, and reviewing competency for donning and doffing of PPE (Table 3). The BSL-3 and ABSL-3 facilities were verified before initiating work with SARS-CoV-2 based on the CDC BSL-3/ABSL-3 HVAC and Facility verification checklist. 24
Specific Risk Assessment Decisions Made by the Biosafety Professionals
For work in BSL-2 facilities, the risk assessment included what sample types could be handled. Criteria used included types of samples (lower respiratory sample, swabs, upper respiratory samples), procedures conducted (high risk procedures were filtering, shearing), and potential viral load (known positive, unknown, negative). Occasionally, seasonal coronaviruses were used as controls. Dedicated biosafety cabinets were recommended for handling blood samples and upper respiratory swabs with minimal processing (centrifugation, aliquoting, and freezing or shipping). BSL-3 practices needed to be followed. This included all work to be done inside the biosafety cabinet, use of a secondary container for transfer of samples outside the biosafety cabinet, disposal of waste in biohazard bags, collection by an approved vendor, donning and doffing stations had storage space for PPE, posted instructions, disinfection bottles (avoiding the use of spray nozzles), and limited use of sharps.
Challenges for Conducting COVID-19 Related Research During the Pandemic
The evolving CDC's guidelines for handling samples, use of medical-grade masks, or other respiratory protection types had to be reflected in existing practices, thus requiring updating of training and practices provided to the researchers.
Because of the shortage of PPE, specifically medical-grade masks, N95 masks, and solid front gowns, the risk assessment for what PPE to use was based on the specific proposed work for each research team. For example, medical-grade masks were used for processing blood samples and upper respiratory samples. All personnel working in BSL-3 and ABSL-3 ultimately switched to using Powered Air-Purifying Respirators while the N95 supply was re-directed for health care use. Also related to PPE supply, counterfeit medical-grade masks, N95, or KN95, was a common finding.
The OHS screening for researchers assigned to work with COVID-19 related samples or the virus was an added burden to the efforts of the OHS team that was dedicated to conduct contact tracing and staff assessment.
In general, laboratory supplies were affected by supply-chain disruptions. Because of the ramp-down of all research activities, many researchers shared their laboratory supplies for ongoing COVID-19 research. Several scientists, already trained to handle human source materials, joined COVID-19 research teams. In these cases, the risk assessment considered the experience of the individual to handle such specimens. Bloodborne Pathogens training and Hepatitis B antibody titer check is always required at our institution for handling human source materials. Lastly, there was an increase in biosafety protocol submissions, including the handling of COVID-19 related samples in BSL-2 facilities following BSL-3 practices and procedures or in BSL-3 facilities.
Disinfection Response
The disinfection response can be divided into two phases. First, the disinfection during the ramp down started a few weeks before the university switched to virtual operations. At that time, the coronavirus was deemed to be an emerging virus. The selection of disinfecting solution was based on the availability of disinfectants kept in stock by CS and the spectrum of those disinfectants. In addition, the U.S. Environmental Protection Agency (EPA) published List N Disinfectants for Coronavirus (COVID-19). 25
This selected list included less than 100 products in 2020, and currently there are more than 600 products. Although disinfectants included in this list are not endorsed by EPA, it was a useful guide to determine what solutions could be used. The selection of PPE was conducted more conservatively, as it was still being determined whether this respiratory virus was transmitted through large or small droplets. The EHSO coordinated the institution-wide effort to ensure that the work and residential spaces were disinfected in a timely manner.
Weekly communications between EHSO, CS, Campus Life, and the University's leadership point of contacts allowed prompt risk assessments to determine what level of disinfection was needed, what PPE should be used by the technicians, and the duration the space should remain unoccupied after disinfection. During phase 2, the portion of the pandemic when most spaces were unoccupied, EHSO's role shifted to assessing the risks to guide personnel from CS in determining the disinfection needed as shown in Table 4. To maximize resources, the risk assessment included a no disinfection option when spaces remained unoccupied for a minimum of 72 h after which normal cleaning would occur.
Example matrix for disinfection of residential spaces
ASAP, as soon as possible; BRS, Building and Residential Services; PUI, person under investigation.
As the status of the university shifted according to prevalence rates, classrooms were assessed to determine what disinfection schedule was needed. An isolation/quarantine facility on campus was identified and individuals who were on campus and were symptomatic or had been identified as a contact in a contact tracing cohort would be moved to this facility for further evaluation and care, if needed.
Supporting Our Healthcare Partners
Before the pandemic, in addition to providing hands-on waste management and radiopharmaceutical support, Emory EHSO provided environmental health and occupational safety assistance to our healthcare partners. The EHSO serves as a consultant to Emory Healthcare on topics, including occupational safety, industrial hygiene, biosafety, radiation safety and environmental management. Our support during the Ebola outbreak enhanced our contributions to health care, which, in turn, led to health care consulting EHSO for support when the COVID-19 pandemic began in March of 2022.
As non-health care employees, EHSO was able to use our expertise to assist in mitigating risk when they became overwhelmed with patient support. Initially, EHSO engaged with the SCDU and Infection Prevention in conducting Just in Time PPE training marathons and developing PPE donning and doffing protocols. When nursing and Infection Prevention resources were directed toward patient support, EHSO led this training at several hospitals. We also began supporting fit testing in health care, which had previously only been performed by health care employees. The EHSO consulted with health care leadership on safety and transportation issues surrounding bulk purchases of hand sanitizer.
We also served as their regulatory liaison. As the pandemic progressed, EHSO coordinated with Infection Prevention on developing the COVID-19 Plan as required by the Occupational Safety and Health Administration's Emergency Temporary Standard for COVID-19 in health care. Similarly, EHSO coordinated the development of the Response Plan for COVID-19 for non-health care workplaces. 26 Today, EHSO continues to provide respirator fit testing.
Assessments and Communications
The EHSO professionals conducted in-person COVID-19 risk assessments for operational and academic continuity. Stakeholders included business units, departments, and colleges. See Supplementary Materials S2 for a full list of stakeholders.
Recommendations were provided to Emory leadership, including suggestions for how to provide instruction by course, classroom, and or program by evaluating proposals submitted by faculty planning in-person instruction in coordination with Emory University Deans. Additional considerations and recommendations are presented next:
Addressed unique needs of various campus business units ensuring the health and safety of employees and students present in-person on campus during the COVID-19 pandemic
Recommended solutions to business unit leadership and facility managers relating to physical distancing, COVID-19 PPE, and administrative practices that can be put in place to reduce transmission of the SARS-CoV-2 virus
Communicated best practices and explained the theory or reasoning supporting these practices in a straightforward manner that was easy to follow and implement
Conducted space and facility maximum occupancy/density assessments
Answered questions from the Emory community relating to applicability and alignment of Emory policies and guidance to public health directives and scientific studies pertaining to aerosol/droplet transmission of viral particles as well as justification for guidance developed in response to COVID-19 pandemic
As participants in multiple COVID-19 task forces and working groups established by the University, EHSO personnel provided guidance to leadership for planning and responding to the pandemic (see Table 5 for a list of Committees and Task Forces). Subject matter expertise was provided for the development of risk mitigation strategies, and campus COVID-related policies and operational processes (e.g., to include COVID-related PPE recommendations and administrative work practices, accommodations for faculty, staff, and students).
Emory University coronavirus disease 2019 committees, task forces, or working group, and the expertise provided by Environmental Health and Safety Office
The participation of EHSO personnel in these committees required keeping up with CDC's guidance and best practices issued by professional entities such as the American Industrial Hygiene Association. Data sources were researched and benchmarked for guidelines and evolving best practices, to update and revise Emory published COVID-19 related campus guidance and operational processes. The types of data evaluated included the following broad categories: COVID-19 and related scientific studies, established best practices published by industry groups, regulatory bodies, academic organizations, and peer institutions.
The goal was ensuring that Emory policies, protocols, and processes for campus activities aligned with Federal, state, and local public health directives and recommendations. As government and local public health directives and recommendations were updated, these would be brought to the attention of Emory leadership and suggestions were offered for updating existing documents.
Moving Toward Normalcy
After the first COVID-19 vaccine became available in December of 2020, the guidelines for returning to campus were modified to align with the CDC's recommendations. University personnel were prioritized to receive the vaccine according to their job functions, for example those working in health care, high-containment, those coming to campus daily due to their functions (essential workers), and other criteria.
Emory University also established an Operating Condition Status visual compass, including four alert levels going from red (only essential activities are permitted), Orange (The prevalence of COVID-19 transmission in the community is elevated or trending unfavorably), Yellow (The underlying threat of outbreak remains, but prevalence of the virus is lower and trending favorably), and Green (Restrictions on size, density, and duration are lifted for on- and off-campus events and gatherings). 20 During the first months of the pandemic, the Operating Status in September 2020, when the Operating Condition Status compass was launched, was yellow. In April 2021, the Operating Conditions Status moved from Orange to Yellow and Modified Yellow (later in September 2021) to include the response related to Flu. The University is currently operating at Yellow since January 2022. 27
The option to continue to work remotely continued through 2022 as the different variants of SARS-CoV-2 made their journey through populations. Emory University adopted a hybrid work model that has created unique challenges for EHSO, including ergonomics for home offices, and injury and/or illnesses occurring at home while working.
Emory University continues to monitor daily cases by maintaining the COVID-19 dashboard, 28 which shows numbers of vaccinated personnel, isolations, screenings, and confirmed cases. In perspective, this dashboard resembles the dashboard maintained by the U.S. CDC for Flu 29 except that the Flu dashboard reports positive tests.
In summary, the lessons learned from this pandemic can be summarized according to the following categories:
Communications: Verbal and writing skills were critical when drafting documents. In addition, the ability to conduct internet searches, followed by analytical skills to interpret constantly changing guidance, was important to support the recommendations provided to leadership.
Teamwork: Since the biosafety office had established a good working relationship with the researchers, when the COVID-19 research started, it facilitated the review of the proposals and preparing researchers to initiate their work in a shorter time frame.
Flexibility: The EHS personnel were assigned to various tasks to meet the institutional demands related to the COVID-19 response.
Business continuity: Each unit continued with their normal activities virtually, or in-person, as needed. This required either following existing Continuity plans or adjusting existing plans to virtual settings or modified in-person activities.
Footnotes
Acknowledgments
The authors would like to thank members of the Emory Community who collaborated with the Environmental Health and Safety Office team and the Office of Critical Event Preparedness Response during the COVID-19 pandemic. The long hours and brainstorming sessions are forever engrained in our memory.
Authors' Disclosure Statement
No competing financial interests exist.
Funding Information
No funding was received for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
