Abstract
Introduction:
Carcasses from animal experiments with RG-3 pathogens should be decontaminated onsite in Austria.
Objective:
The aim of this study was to find out if the use of pass-through autoclaves for the decontamination of animal carcasses (up to 40 kg of weight) could serve as a routine method for smaller laboratories, as the installation of special carcass decontamination plants may be cost prohibitive.
Methods:
Biological indicators (BIs) were implanted into the carcasses of animals of different sizes and species with a novel method using stainless steel pipes. The bodies were placed in autoclavable plastic bags and equipped with thermal probes by insertion through the rectum. Subsequently a factory default autoclave cycle for liquids was performed, which holds a core temperature of 121°C for 20 min.
Results:
The weight of the carcasses ranged from 1 to 42 kg, the duration of the individual cycles reached from 2.2 to 17.23 h. Decontamination was successful every single time as shown by the BIs. The application through the natural orifices with the help of the application tools seems to offer a reliable alternative for implanting the BIs into the carcasses without creating new openings. Insulation properties did not pose substantial challenges to the process. Limitations on the packaging procedure were identified in carcasses larger than 30 kg.
Conclusion:
Based on the results of this study, using pass-through autoclaves represents an option as a routine method for the decontamination of animal carcasses up to at least 40 kg.
Introduction
The authors of this study are involved in the design and construction of a small biosafety level 3 (BSL-3) building, in addition to an existing containment facility. The unit will include laboratories as well as animal facilities for rodents and small farm animals up to a weight of around 40 kg, such as pigs or sheep. The animal rooms and laboratories are designed to be utmost adaptable and flexible in terms of study possibilities.
One of the challenges of animal research in a biosafety containment facility is the correct disposal of animal carcasses. To keep risks low, decontamination should happen onsite.
In reference to common literature and guidelines, incineration, alkaline hydrolysis, and rendering are the methods of choice concerning the decontamination of animal carcasses larger than laboratory rodents.1,2 The routine transport of nontreated carcasses containing RG-3 pathogens out of containment is not allowed by the competent Austrian authorities, so decontamination has to take place in the animal facility. 3 Incineration is the perfect solution to destroy infectious material, but these plants demand complex exhaust filter systems. 4
Alkaline hydrolysis is also capable of inactivating infectious agents, including prions, 5 but this method needs large and complex technical equipment. Rendering plants inactivate most biological organisms through processing and separating of carcass tissues into bone, protein, and fat,5,6 but the construction of a rendering plant is cost prohibitive for small facilities.1,7
Another option for carcass decontamination is the use of autoclaves. Steam autoclaves are a compulsory component of BSL-3 facilities and often installed in duplicate to offer some redundancy. Several studies considered autoclaving to be an adequate method in terms of successful inactivation of materials that may contain spore-forming organisms or other organisms that are difficult to decontaminate.8,9 Although some authors consider the use of autoclaves as a fallback position in case the main method for the decontamination of carcasses is not available, 2 they did not conclude whether autoclaves could serve as a routine method for decontamination of carcasses up to 40 kg in small BSL-3 or BSL-4 laboratories.
The aim of this study was to find out whether a factory default program of pass-through autoclaves could be validated to be the main tool for this purpose.
Methods
Autoclaves
For this project, two pass-through autoclaves of different sizes (Lautenschläger ZentraCERT Typ 2269, ZentraCERT 7269, Köln, Germany) were validated for the decontamination of animal carcasses. The small unit (no. 2269) has a useable chamber of 30 × 60 × 60 cm, whereas the chamber of the large autoclave (no. 7269) measures 60 × 60 × 120 cm. The autoclaves are mainly used for the decontamination of laboratory waste, which is intended to leave containment for final disposal. For the purpose of this study, the autoclaves were loaded and controlled from outside the laboratory containment.
For design reasons, only the main steam valve had to be operated from inside containment. On some occasions, the autoclaves had already run a short or long cycle before they were loaded with the carcasses. If they had not been used that day, Bowie-Dick tests were performed to check the functionality of the autoclaves and to preheat them.
When it comes to the autoclaving of animal carcasses, factory default settings often do not seem suitable.7,8,10,11 We wanted to know whether our autoclaves would be capable of autoclaving carcasses by using one of the default settings available to us. For this study a standard cycle for open liquids was chosen, which was already programed on our autoclave, as it seemed most suitable to deal with the fluid content of the carcasses. 9 The heating-up time of this program is characterized by a chamber pressure of 2300 mbar that is maintained until the loaded materials reach the required temperature of 121°C without any prevacuum pulses.
After a sterilization time of 20 min at 2240–2260 mbar, a cooling cycle follows using jacketed cooling with a constant auxiliary chamber pressure of 2850 mbar until the temperature probes record a temperature of less than 80°C. 12 The duration of the heating time (h) as well as the cool down depends on the composition of the load, whereas the sterilization time of 20 min is constant. The controlled cooling prevents liquids from evaporating explosively out of the carcass because of delayed boiling. 9 The program protects the autoclave chamber from being contaminated and staff from being endangered by steam jets.5,13
Each autoclave has two thermal probes, which are connected to the machine control panel through cables and work independently. The sterilization phase does not start until both probes report a temperature of at least 121°C. When a program for liquids is used, the temperature probes are usually placed in a reference bottle filled with reference fluid, which corresponds to the volume and type of the liquids in the load. This ensures that the potentially infectious liquids in the bottles reach the required temperature.
However, as suggested by Schinköthe et al, 2 the temperature probes of the autoclaves were not placed in a reference liquid bottle but in the carcass itself. According to Schinköthe et al, the abdominal cavity seems to be the most difficult location to reach the required temperature.2,9 Therefore, it can be assumed that all other tissues are also exposed to that temperature. As we wanted to avoid opening the abdomen by cutting into its surface, we inserted the temperature probes through the rectum.
Preparation of the Carcasses
The carcasses used for this study were obtained from a local rendering plant. Intact animal bodies were chosen in a weight range from about 1 to 40 kg from species that could be used in animal experiments in our future facility. Rodents were not included in this study as autoclaving single rodent carcasses was not expected to present a challenge to the autoclave. We started with bodies of small animals such as cats and foxes.
In the further course, we proceeded to bodies of larger animals to determine to what extent the weight and size of the carcasses could be increased and still achieve a successful decontamination, always with the same autoclave program. The carcasses were stored at 2–8°C or frozen and then thawed at a cool storage temperature 1 day before autoclaving to represent the process planed for the new facility. Owing to different preparation procedures, the core temperatures varied at the beginning of the autoclaving process.
Packaging
As the carcasses of a BSL-3 animal facility would potentially contain infectious agents, leak-proof or even airtight packaging may be necessary for safety reasons. 2 To prevent any fluids from leaking into the autoclave chamber, we used a leak-proof stainless steel box in every experiment. The size of the box was dependent on the size of the carcass and the autoclave. In addition, we used packaging materials that are regularly used for autoclaving in our institute to create three different methods of packaging. The aim was to find out how little packaging would be sufficient to prevent the autoclaves from being contaminated by spattering.
For trial no. 1, the carcass was packed in an autoclavable bag with a capacity of 80 L (Sarstedt AG & Co. KG, Nümbrecht, Germany) and in a Bio-bin® (Econix, Sheffield, United Kingdom) with a capacity of 30 L or 8 kg, respectively. The Bio-bin was subsequently put in a stainless steel box.
For trial nos. 2–7 and 9–17, the carcasses were packed in an open bag in a stainless steel box.
For trial numbers 8, 18, and 19, the carcasses of larger pigs and calves were only covered with an autoclavable bag as they did not fit into the bags we used. This method would only be useable in facilities were the autoclave is directly accessible from the post-mortem room.
Biological Indicators
To monitor the efficacy of the autoclaving procedure, the carcasses were implanted with biological indicators (BIs) containing Geobacillus stearothermophilus (ATCC 7953). There are numerous types of BIs available for the validation of autoclaving processes. In addition to spore strips, self-contained biological indicators (SCBIs) in the form of glass or plastic ampoules are available. In contrast to spore strips, SCBIs already contain culture medium that facilitates the handling after the autoclaving process. 14 One of the intentions of the initial run was to find out which type of BI is the most suitable for the use in animal carcasses. The following products were tested (Figure 1a):
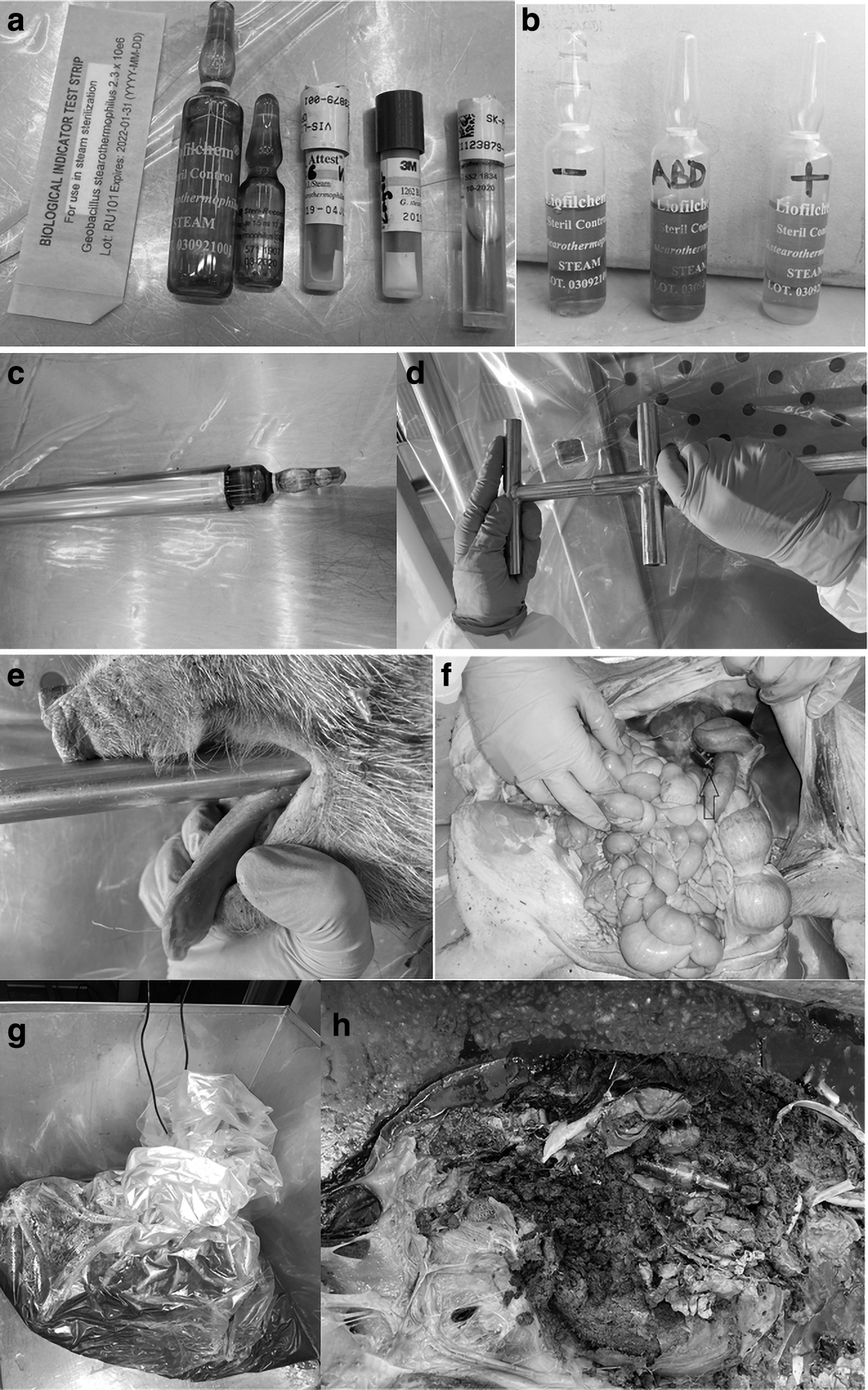
3M™ Attest™ Biological Indicator 1262 for Steam Sterilization, 100/BX, 4 BX/CTN
Crosstex Traditional Spore Strips—G. stearothermophilus (Cell Line 7953) BS-106
gke Steri-Record® Mini-Bio-Plus Standard
gke Steri-Record Stearo ampoules for liquid and waste sterilization B-S-AMP-10-6 (1.5 mL)
Mesa Labs ProSpore® ampoules PS-6-50 (4 mL)
STERIL CONTROL GST E6 ampoules, 1–5 × 106 CFU/ampoule (4 mL)
After autoclaving, the remains were carefully opened to find the BIs. The BIs were incubated according to the instructions. One BI of each type, which was not autoclaved, served as a positive control (1b).
Application Tools
As creating new openings in the carcasses could possibly influence the validation procedure adversely, finding other methods for the placement of the BIs into carcasses is currently a subject of research. 8 Until now, standard practice is placement of the BIs by opening the abdominal cavity with a scalpel, placing the BIs inside it and suturing the abdomen afterward. 7 When using this method, one has to consider that steam could get access into the abdominal cavity easier as it would at an unopened carcass.
Thus, the validation could possibly underestimate the heating requirements. Suturing is also time consuming and stitches may be corrupted by the steam treatment, thus failing to keep the incision closed. Besides, if validation is performed with potentially contaminated carcasses, the contact with these carcasses should be minimized when preparing the carcass for the autoclaving process.
To avoid these problems, two application tools were designed that allow the positioning and placement of the BIs into the carcass (Figure 1c, d, e) while leaving the carcass intact. The tools consist of two stainless steel pipes (CrNiMo-Stahl 1.4401 OENORM EN 10088) of different sizes (18 × 1 and 15 × 1 mm, respectively). The diameter of the outer larger pipe matches the diameter of the BIs. The smaller pipe just fits into the larger pipe. For a more comfortable use, handles were attached to the rear end of both pipes (Figure 1d). One version of the tool has a blunt front end, this tool is used to insert BIs through natural orifices (Figure 1c). Another version of the tool has a sharp front end and is used to insert BIs into body parts that are not accessible through natural openings, such as large muscle parts or the brain.
To position the BIs in the most central anatomical region, such as through the rectum into the colon close to the diaphragm or through esophagus into the stomach, the necessary distance the tool has to go into a certain body is measured roughly by holding the tool along the side of the body.
To place the BIs into the carcass, the smaller pipe is drawn back as much as required to load the larger pipe with the BI. The loaded tool is then inserted into the natural openings of the carcass to the required position of the BIs. As the tool is rather stiff, its position could be made out by palpating it through the abdominal wall. By holding the smaller pipe in place, while withdrawing the larger pipe, the BI is left at place without coming under pressure when the tool is extracted. After withdrawing both pipes together from the carcass, they can be cleaned and disinfected as per normal dissecting instruments.
Orientation is easier when marks are made on the tool to know when the required penetration depth is reached and to hold the inner pipe in the right position, respectively. As the application tool usually slides along the esophagus or rectum easily, it can be noticed whether the tool unintentionally penetrates the intestinal or esophageal wall. To verify the process of positioning, a pig carcass with a weight of 17.0 kg was dissected immediately after the BIs were inserted with the application tools (Figure 1f). Thus, the position of the BIs could be inspected without a possible distortion caused by the manipulation or the autoclaving process.
Results
Autoclaves
Overall, 19 autoclave cycles were performed (Table 1). Total run time ranged from 2.27 to 17.23 h. Shorter runs were loaded and unloaded on the same day, whereas longer cycles ran overnight. Disregarding trial numbers 6, 7, and 9, wherein the data recording of the autoclaves was incomplete, no other adverse effects were reported. Both autoclaves remained visibly clean and suffered no damage.
Collected data during the trials
Carcasses
The weight of the carcasses ranged from 1.0 to 42.8 kg, which entails correspondingly different durations of the individual cycles. Starting temperature of the carcasses reported by the thermal probes of the autoclaves ranged from 2.2°C to 18.1°C. The measured and calculated parameters of the individual trials are listed in Table 1.
Heating time per kilogram weight for the carcasses that were packaged comparably (trial nos. 2–19) ranged from 0.2 h/kg (42.8 kg calf) to 1.10 h/kg (4.9 kg cat). The plot of the observed heating time versus the carcass weight (Figure 2) is indicating rather a logarithmic than a linear relation, but there are too few data sets available to accurately determine the functional form of the relationship.
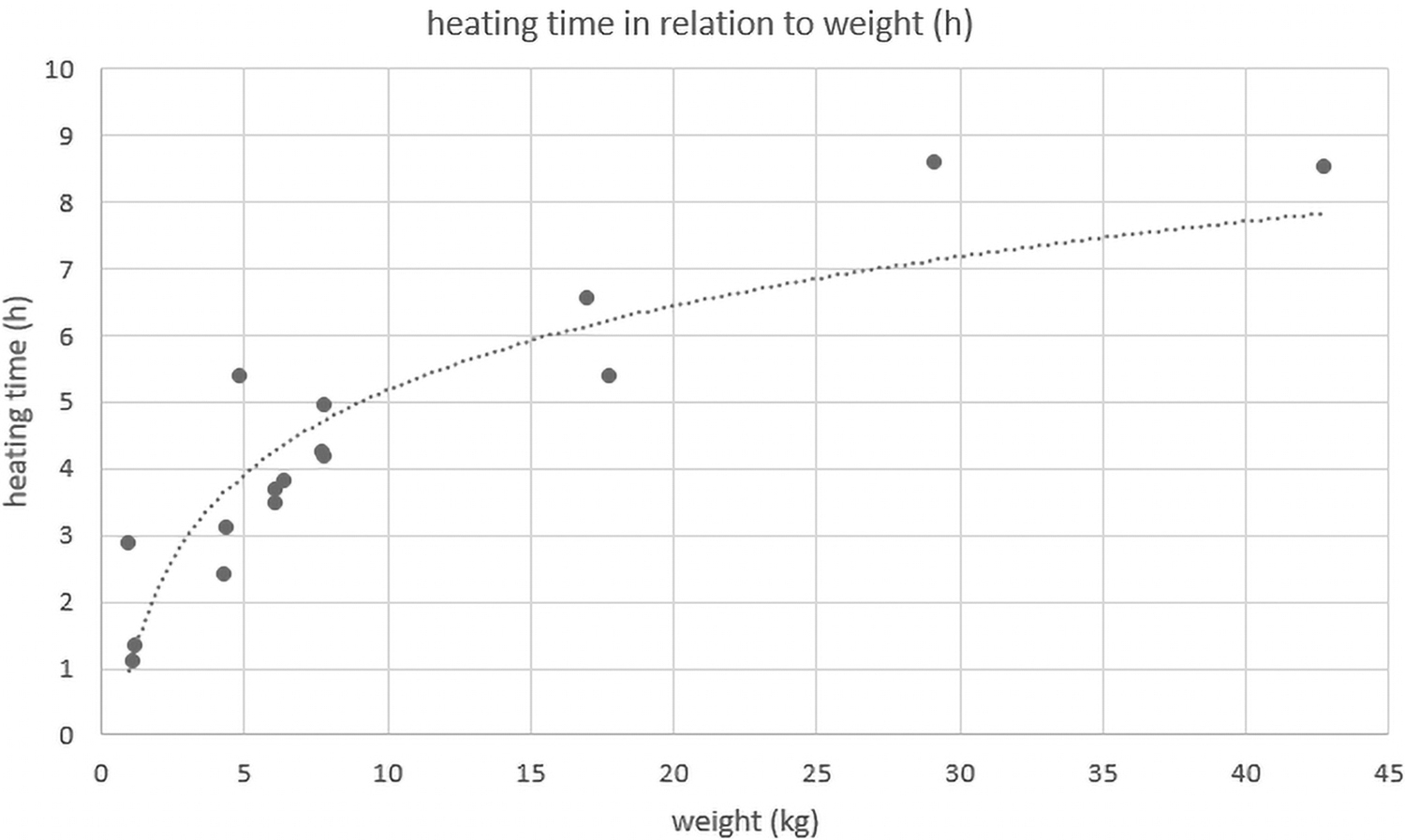
Heating time in relation to carcass weight.
Fitting an ordinary least square regression model to heating time including the factors species, logarithmic weight, and starting temperature of carcass revealed a significant effect of carcass weight on heating time (Table 2). For each additional unit of log (carcass weight [kg]), it is estimated that the heating takes 2.4 h longer. The 95% confidence interval ranges from 1.59 to 3.25 h. The observed data do not indicate a significant effect of starting temperature (confidence interval −0.16 to 0.33).
Results of ordinary least square regression for heating time (h)
Except for the pig (n = 7), only one to three heating times were recorded per species (sheep [n = 3], fox [n = 2], cat [n = 1], cattle [n = 1], swan [n = 1]). Hence, there is not enough data to conclude in detail on the impact of differences neither on species itself nor in features such as fur, feathers, or fat content on the heating time.
Statistical analyses and estimation of the multiple regression model were done in software R. 15
After autoclaving the pig carcasses, it was noticed that the gluteal muscles and the intestinal wall of some parts of the gastrointestinal tract showed a firmer texture than other tissues of the carcass. In contrast, bony structures nearly disintegrated completely. Therefore, certain tissues seem more robust when exposed to a combination of steam, heat, and pressure than others.
Placement of Temperature Probes
Insertion of the temperature probes into the rectum worked well. Both probes reported independent temperatures and stayed at the given position throughout the autoclaving. The probes could be cleaned afterward and suffered no damage.
Packaging
Packing the carcasses in a bag and a Bio-bin in the initial trial turned out to prolong the total time of the autoclaving considerably. In addition, the bottom of the Bio-bin got stuck to the steel box, so we decided to not use the Bio-bin for the following trials. The autoclavable bags, in contrast, turned out to be eminently robust and withstood even long cycles and weights up to 31 kg. Nevertheless, we assumed that sealing it would also prolong the heating process. As a result, we decided to proceed in the study with the carcasses packed in a loosely folded autoclavable bag and placed in a stainless steel box (Figure 1g).
Biological Indicators
During the first trial, we gained considerable experience about the differences of the different types of BIs, which facilitated the decision on a specific type to use for the rest of the study.
When used in carcasses, the SCBI plastic ampoules did not perform as well as the glass ampoules. As there is an opening on top of the plastic ampoules, body fluid got inside the SCBI. Thus, the color changes and the results could be falsified. 7 The spore strips, in contrast, stuck together and were hard to retrieve and to remove from the cavity. Besides, the spore strips had to be transferred from the packaging with sterile forceps into a culture medium before the incubation could be started, which entails an additional step of procedure and a source of possible contamination.
Some of the BIs showed the widely described caramelization effect.2,7,8,16 This applies to those BIs whose culture medium contains glucose. As the culture medium in the ampoules and SCBI is also exposed to the autoclaving process, the glucose starts to discolor if the heating process is long and hot enough. 16 This color change does not interact with the color change caused by the persistence of spores of G. stearothermophilus, however (Figure 1b). Therefore, the BIs that showed this caramelizing effect could still be used for validating the decontamination process.7,8
The gke Steri-Record Stearo ampoules for liquid and waste sterilization B-S-AMP-10-6 (1.5 mL) were the only ampoules that did not show a caramelization effect. However, it could be difficult to retrieve small ampoules like these from the abdominal cavity of larger carcasses.
After evaluating the results of the initial trial, ampoules with a volume of 4 mL were considered to be the most suitable BIs for the use in carcasses in terms of handling, readability, and usability. The supplier chosen (STERIL CONTROL, Roseto degli Abruzzi, Italy) was a matter of price and availability of the BIs.
None of the BIs that were autoclaved changed color from violet to yellow during the subsequent incubation time, which indicates that the spores of G. stearothermophilus in the BIs were successfully inactivated during the autoclaving procedure. In contrast, the positive controls showed the expected color change from violet to yellow (Figure 1b).
Application Tools
In the first trial, the abdominal and thoracic cavities were opened surgically to insert different types of BIs. Afterward, the carcass was stitched closed using catgut and Polyglactin. After the autoclaving process, it was not possible for us to retrieve the suture material and it remains unclear to what time of the process these materials had lost their functionality. Other suture materials were not tested as the placement of the BIs in the following trials was performed with the help of the application tools. The use of the application tools to insert and place the BIs worked well. In some cases, the application tools penetrated the intestinal wall of the rectum or esophagus, when a little more effort was applied, so a certain amount of sensitivity is needed to ensure walls are only penetrated when intended to.
In the dissection after autoclaving, all ampoules except for one were found in the position where they were originally placed (Figure 1h). Even after thorough examination of the remnants of the carcass, one BI could not be retrieved and it remained unclear why and where it disappeared. All retrieved BIs except one remained intact. One BI was destroyed within the applicator when the tool was accidently moved laterally while the BI was still partly inside. This event was noticed and the BI was replaced. The dissection of a pig immediately after insertion of the BIs using the application tools showed that the tools enable the user to place the BIs in the required position precisely. The location of a BI changed only in the event that a second BI was inserted into the same orifice that causes the shift of the first BI.
Discussion
Our data demonstrate that using a default autoclaving program may be a suitable method to routinely decontaminate animal carcasses up to a weight of at least 40 kg per animal in containment facilities where the installation of an incinerator or alkaline hydrolysis plant is not feasible. Redundancy could be gained by having two autoclaves, which would be desirable for the everyday maintenance duties of an animal facility anyway.
During our validation, we had to realize that the time to heat a carcass depends on several factors.
Some authors9,11 mentioned that the temperature of the carcasses before autoclaving affects the duration of the autoclaving process. The data of this study suggest the influence to be small as long as the material is not frozen. Owing to the enthalpy of fusion, it takes the same amount of energy to melt 1 kg of frozen water as it takes to heat 1 kg of water from 1°C to 80°C. Therefore, carcasses stored frozen have to be defrosted to a temperature of 2–8°C before autoclaving.
The heating time was notably prolonged by certain types of multilayered packaging, which prevented steam from penetrating through to the carcass. If packaging is needed, it should be kept to a minimum.
This study did not measure the influence of preheating the autoclaves in relation to energy consumption and total running time. However, variability in the decontamination time could be caused by the previous use of the autoclaves that day, whether it had been used for a long cycle program or just for a Bowie-Dick Test.
The impact of the insulating layers of the carcasses themselves (fur, fat, or feathers) on the heating time was surprisingly low throughout this study.
Using application tools for inserting the BIs into the carcasses turned out to be a satisfactory method. As the ampoules are very robust and give a distinct crunching signal to the user when breaking, there only remains a minor risk of actually inserting a broken BI without noticing it.
Regarding the packaging of the carcass, it should be taken into account that considerable amounts of liquid in the form of body fluid from the carcass and condensed water are to be expected. If the steel box is sufficiently sized, contamination of the autoclaves can be extensively avoided, even if the primary packaging would fail.
We also noticed odor pollution when opening the door of the autoclave. A possible way to deal with this would be to ensure that the air coming out of the autoclaves can be aspirated by the ventilation system immediately.
Regarding the loss of data in some trials, the service provider suspected that due to prolonged total process times, there would be a restriction by the data storage device, but it is still unclear why the data recording failed three times but worked properly in others experiments.
One of the downsides of using a gravity displacement program is that the steam does not readily penetrate packaging material or the carcass itself, which results in long cycles and enormous consumption and disposal of mains water. Most of the mains water consumed during the cycles in a jacketed autoclave is used for cooling. In the setting of this study, the water ends up in the containment facility effluent tank and has to be heat treated before discharging it into the public sewer. As the cooling water is not contaminated, it would be possible to directly release it to the sewer.
According to the service provider, the autoclaves would also be able to cool down without forced cooling, but this would result in even longer cycle times.
The total duration of the individual trials could be shortened by adding prevacuum pulses to the program. Using several low prevacuum pulses has already been shown to be safe and improves the decontamination process. Only the pressure support during cooling would be essential to prevent the liquid from evaporating explosively.
During our study, we considered that inserting the thermal probes into the rectum immediately before autoclaving is not always possible, as the carcasses may have to be packaged in a sealed container or bag that must not be opened again preautoclaving.
Placing the temperature probes in reference bottles filled with water alone does not seem to be suitable to control the process of autoclaving carcasses.2,7 Even when the bottles contain liquids the same weight as the carcass being autoclaved, this is inadequate to control the heating up phase of the cycle, as the water heats up faster than the carcass.2,7
Several other options for the type and placement of thermal probes are conceivable. Thermal probes of our autoclave could be detached, packed to the carcass in the postmortem room and connected to the autoclave again later, but this would include a lot of technical work and it would increase the risk to allow contaminated air out of the package. Alternatively wireless thermal probes could be placed inside the carcass. However, by doing so, the carcass needs to be cut open or manipulated in other ways, which means an increased risk of contamination for staff.
Another method would be to find a suitable substance, which mimics the heat distribution of the carcass representatively or heats up even slower than the carcass. The substance could be placed in the autoclave next to the carcass and equipped with the thermal probes.
As the options already mentioned are either not applicable for our purposes or not available yet, development and validation of cycles are desirable. Some of these cycles have already been published.7,8 The data of this study show that it is basically possible to find a relation between any given weight of a carcass and the required heating time. This knowledge supports efforts to forgo the application of thermal probes into the carcasses at all and to develop either a series of programs, each for a specific weight, or to create one program that allows a heating time to be defined according to the weight of a given carcass.
More trials are necessary to validate the settings for a reliable decontamination of carcasses packaged into a sealed bag or box. To shorten heating time in such a setting, a packaging system could be chosen that melts down partly or even completely in an early phase of the heating. This would allow steam to get direct access to the carcass and would shorten the process. We tested one carcass in a melting bag in a stainless steel box. The bag melted to a lump and could be easily removed after autoclaving.
Conclusion
It could be shown that pass-through autoclaves using a default, temperature controlled, liquid cycle are able to successfully decontaminate animal carcasses of different species, sizes, and insulation properties up to a weight of at least 40 kg. The use of a liquid cycle with pressure support during cool down allows decontamination of the carcasses in reasonable time and without any visible contamination or damage to the autoclaves.
Despite the caramelization effect, 4 mL SCBI glass ampoules are suitable for validating this process.
The use of a simple tool made from stainless steel pipes turned out to be reliable and timesaving for placing BIs into carcasses for validation purposes. As no artificial openings had to be created, the risk of contamination for staff when working with potentially infectious tissues was reduced. To keep the heating time low, packaging of the carcasses should be avoided or minimized.
Footnotes
Authors' Contributions
S.L. contributed to conceptualization, investigation, methodology, validation, visualization, writing—original draft, and writing—review and editing. W.S. was involved in data curation and writing—review and editing. K.W. carried out data curation, formal analysis, software, and visualization. T.S. was involved in formal analysis, validation, and writing—review and editing. F.S. was in charge of funding acquisition, resources, supervision, and writing—review and editing. H.S. was in charge of conceptualization, funding acquisition, investigation, methodology, project administration, validation, and writing—review and editing
Ethical Approval Statement
Not applicable.
Statement of Human and Animal Rights
No live animals or human subjects were involved in this study.
Authors' Disclosure Statement
No competing financial interests exist.
Funding Information
This FEMtech project “Lab3plan” was co-financed by the Austrian Research Promotion Agency (FFG) (grant number: 888211).
