Abstract
Introduction:
This article will review the processes utilized to develop simple effective containment engineering controls. Short-Term Use Biocontainment Bubbles-Yale (STUBB-Ys), as Yale refers to them, were designed, built, tested, and implemented to protect members of the Yale University community from exposure to SARS-CoV-2 aerosols. STUBB-Ys were designed and created in conjunction with end users, constructed by Environmental Health and Safety (EHS) or partner groups, and tested onsite after installation to verify effective operation and containment.
Methods:
A wide variety of devices in different settings were developed and installed. STUBB-Ys were used at COVID-19 indoor test centers, laboratories, and clinics. The devices were pursued to create infection prevention measures where existing processes could not be utilized or were inadequate. Each STUBB-Y was tested with a C-Breeze Condensed Moisture Airflow Visualizer to generate smoke and a Fluke 985 Particle Counter, which gives the particle counts from 0.3 to 10 μm to measure particle escape visually and quantitatively. Airflow rates were also tested where applicable with a TSI VelociCalc 9525 Air Velocity Meter.
Results:
Students and faculty were able to safely continue vital research or clinical study in the targeted areas with the addition of these simple containment devices to confine aerosols.
Conclusion:
From a biorisk management point of view, EHS was able to confine aerosols at their potential source using simple designs and equipment and adhering to the hierarchy of controls. This article demonstrates how a straightforward design process can be used to enhance worker protection during a pandemic.
Background
On January 13, 2020, the Yale New Haven Health system issued a Situation, Background, Assessment and Recommendations (SBAR) reporting an outbreak of pneumonia in China related to a novel coronavirus. 1 The report identified an acute respiratory tract infection and required airborne precautions and a negative pressure room for any patients meeting the SBAR case description. This SBAR and preexisting knowledge of aerosols, including creation, terminal settling velocity, deposition in the respiratory tract, led to Environmental Health and Safety's (EHS) decision to explore the development of additional barriers to protect workers during the pandemic.2–4
In addition, on July 9, 2020, the WHO released a scientific brief that SARS-CoV-2 could be transmitted through aerosols, “particularly in specific indoor locations, such as crowded and inadequately ventilated spaces.” 5 Three days prior on July 6, an article published in Clinical Infectious Diseases appealed to the medical community to recognize the importance of SARS-CoV-2 airborne transmission. 6
Indoor and outdoor airborne infections from SARS-CoV-2 were confirmed by several outbreaks throughout the United States, including large super spreader events.7–9
Introduction
Yale EHS Interventions
There were significant reasons for EHS to help identify engineering control options. From the industrial hygiene hierarchy of controls, ventilation controls are favored over PPE controls, which is the lowest rung of control. From the start of the pandemic, N95 respirators, similar PPE supplies, glove boxes, and biosafety cabinets were in short supply, unavailable, or had long lead times. Respirators were also reserved for frontline healthcare staff. Many of the traditional controls were not a good match for the situations where researchers, clinicians, and healthcare staff were at risk. The unknowns associated with the virus created significant concerns among essential personnel who faced potential exposure. There was a palpable anxiety among some Yale University staff who were returning to work with patients, research subjects in clinical settings, or unfixed human specimens, such as blood or salvia.
Stakeholders and essential staff reached out to the Yale EHS office, asking if their work in a variety of settings was safe to conduct with the potential presence of SARS-CoV-2. Owing to the increasing requests for evaluations of potential risks, Yale EHS established a team to identify potential engineering control solutions to minimize exposures and protect workers from SARS-CoV-2 aerosols where applicable. The team was assembled at the onset of the pandemic in March 2020. The work was established before the availability of widespread COVID-19 testing or immunizations.
The team operated under the assumption that SARS-CoV-2 could be an airborne transmitted pathogen, posed a risk to others 2 days before the onset of symptoms, the disease could be transmitted by individuals with an asymptomatic infection, and there was limited information on which human specimens harbored the virus in significant titers. Staff were also instructed to disinfect high-touch surfaces with an appropriate disinfectant noted by the United States Environmental Protection Agency. 10 These factors reinforced the need for considerable additional controls. It was later confirmed that asymptomatic people accounted for 40–45% of SARS-CoV-2 infections, which could be transmitted over approximately a 2-week period. 11
Risk Assessment
The requests to assist in enhancing the safety of staff work environments led to the start of industrial hygiene and biorisk management challenges within EHS. As each client group posed a question on the safety of their work or workplace, EHS would send an email to technical staff from all disciplines in the EHS office. A risk assessment questionnaire found in Appendix A1 was utilized to brainstorm if a Short-Term Use Biocontainment Bubble-Yale (STUBB-Y) was needed. EHS then asked the client for a detailed list of all work procedures or interactions to get a clear understanding of what was done, when, and where in the workspace procedures were done. The EHS team also worked closely with Yale's Facilities Department to identify the key heating, ventilation, and air conditioning (HVAC) parameters of the space, such as air changes per hour (ACH), outdoor air exchange, supply air filtration, and the negative pressure relative to adjoining spaces.
This risk assessment process became the road map for potential engineering controls to minimize exposure in clinics, laboratories, or other settings. If the assessment identified a potential airborne exposure risk with insufficient ventilation, a STUBB-Y would be considered for the human subject or human sample research. EHS was faced with a critical decision on the use of high-efficiency particulate air filter (HEPA)-filtered devices, as testing of commercially available devices by Yale's biosafety cabinet certification vendor identified that very few of these devices passed the Institute of Environmental Sciences and Technology (IEST-RP-CC002.4) Contamination Control Division Recommended Practice 002.4 Unidirectional-Flow, Clean-Air Devices testing. 12
Only HEPA filters that were 99.97% effective when challenged with 0.3 μm particles by Yale's IEST-RP-CC002.4 certified biosafety cabinet vendor were used if known COVID-19 patients or subjects were involved. This air could also then be recirculated into the room. Exhaust fans without IEST-RP-CC002.4 testing certification were exhausted outdoors. 13 HEPA filtering fans were utilized as they are effective for removing certain airborne infectious diseases, including Mycobacterium tuberculosis and SARS-CoV-2.14–16
Trial STUBB-Ys for human subject research or human samples were built by EHS, Yale Facilities, Yale machine shops, or a contractor. If the client was satisfied with the trial device, the device met visual and quantitative smoke testing, and necessary flow rates, it would remain in place for their utilization with any training necessary. Each enclosure STUBB-Y achieved a minimum inward airflow of 75 feet per minute (fpm), mimicking a Class I biosafety cabinet. 17
If the device contained a tested and certified HEPA filter, it was added to the Yale EHS records for annual retesting by an outside vendor.
The goal of these STUBB-Y projects was based on the principle of isolating the hazard from potentially exposed personnel. Enclosures, the use of directional airflow, and devices that provided filtration were the three principles used by the EHS team for biohazard isolation.
Methods
STUBB-Y Projects
COVID-19 test centers
Multiple indoor test centers were opened on campus in 2020 to facilitate COVID-19 testing multiple times each week. Some of the utilized locations in dormitories did not have significant HVAC controls or accessible windows.
Before the availability of vaccines, confining any potentially infectious aerosols was a priority for the team. It was important to test asymptomatic students and staff given the high risk of spreading from people without symptoms. 18 Once the locations were determined, a decision was made that the best way to confine aerosols was to develop local exhaust ventilation (LEV). Three such controls were evaluated.
The first was a flexible pop-up personal stand-up tent designed to protect photographers from the elements when taking pictures outdoors. This tent was commercially available, but the first trial identified a significant trip hazard. It was also not very sturdy, staff had to spend a lot of time opening and closing the very long zippered door, and it was difficult to connect to an exhaust fan.
The next containment test device was a barrier that was a solid three-walled chamber with a center view screen developed by Yale Engineering. It had a roof where an exhaust port was positioned to collect aerosols at the top. The design was very sturdy, but particle containment testing demonstrated that it was subject to loss of containment with turbulence from foot-traffic in the room. The large opening in the rear of the barrier represented too significant a risk for particle release in locations that may not have strong exhaust capacity.
With these two evaluations, it was clear that a containment enclosure with either HEPA-filtered exhaust back to the room, or ducted exhaust to the outdoors were the targeted specifications for asymptomatic and symptomatic testing locations.
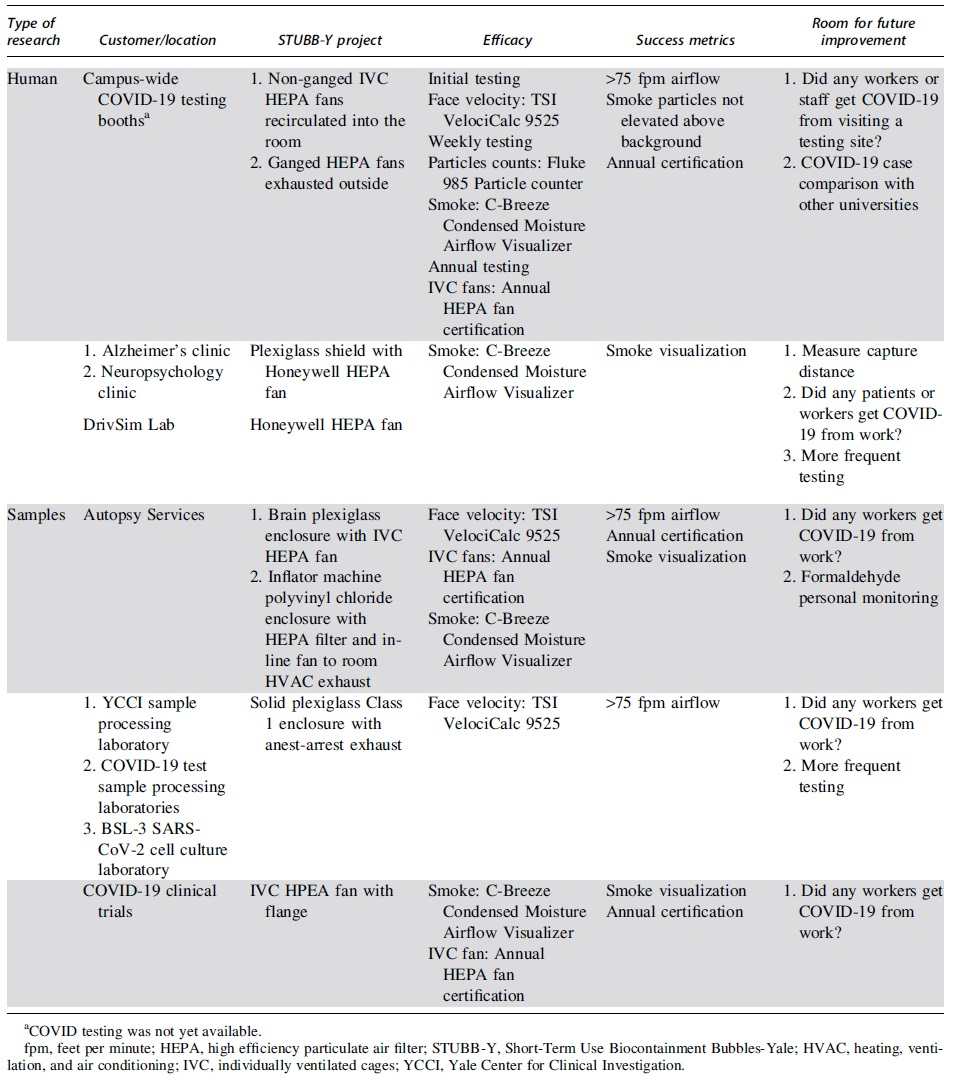
Yale Facilities then built a 3′ × 3′ × 7′ phone booth style, completely enclosed, test chamber that had a storm door for entry and exit as seen in Figure 1. (Larger 3′ × 4′ × 7′ booths were also constructed to meet accessibility requirements). Figure 2 shows a foot handle placed at the bottom of the door to allow for hands-free entry and exit. The storm door could be pushed open to leave with an arm or other body part. The storm door had a sliding screen and window, which provided a way to modify the airflow into each test booth. The screen was set to only have the bottom 1/4 open. Each door was equipped with a spring to serve as a self-closing mechanism. HEPA-filtered exhaust fans or exhaust ductwork (if exhausting to the outside) were placed at the top of the booth based on the room configuration.

Ganged student testing booths.
Non-ganged student testing booth airflow diagram.
If there was access to the outdoors through a window or an accessible wall, a high-flow HEPA-certified fan unit with a manifold system that connects multiple booths was used. A 6″ diameter hole was cut at the top of the booth to connect to the exhaust fan. The enclosed booth outperformed the three-sided barrier on particle release testing. Preliminary velocity testing measurements across the duct opening with a TSI VelociCalc demonstrated that 80–90 ACH could be achieved. Once booths were set up in testing locations, air exchange rates improved to between 100 ACH (for small individually ventilated cages [IVC] rack exhaust HEPA-filtered fans exhausted back to the room) to 200 ACH (for the larger ducted exhaust fan systems). This far exceeded even the standard ACH for BSL-3 laboratories, which is 15 ACH. 17 The IVC fan was tested annually under IEST-RP-CC002.4 guidelines by an outside vendor.
Initially, a total of 72 test booths were constructed and installed at 17 test sites. As there were multiple booths in each location, a 6-min wait period was also used in between individuals using each booth for self-swabbing or to be swabbed by a healthcare provider.
The booths were, and still are, tested weekly by EHS staff. EHS used a C-Breeze Condensed Moisture Airflow Visualizer to generate smoke (simulating unmasked sneezing) within the booth to test the airflow both visually and quantitatively. Escaped particles were measured with a Fluke 985 Particle Counter, which gives the particle counts normalized to concentration from 0.3 to 10 μm, appropriate for SARS-CoV-2 particle size. 19
To test each booth, one member of EHS would stand inside a booth and release smoke for ∼10 s, while another member stood outside the booth with the Fluke. Particle counts were checked at the inflow portion of the door, along the edge of the door, and when the patient exited to ensure particles were not escaping. If the particle counts remained at background levels, the booth was performing as designed. Nonexpensive alternatives could include dry ice or baby powder to visualize airflow. Seventy percent ethanol was provided to the test centers for booth decontamination when needed. 10
The booths provided containment for nonsymptomatic users to self-swab with masks removed. The booths allowed a downgrade to only the use of surgical masks and gloves for workers operating the asymptomatic test centers. Healthcare workers and staff working at the symptomatic test centers were provided with N95 respirators as a precaution. Booths at the symptomatic test sites included an operational window so a healthcare professional could swab symptomatic subjects.
Clinical research protocols: Alzheimer's clinic
The Alzheimer's clinic was concerned about potential risks from asymptomatic unmasked subjects. Staff reported that study subjects in the clinic could remove their masks at any time while interviews were conducted. The clinic's plan was to only hold interviews in very large rooms and use the largest possible physical distance between the interviewer and the interviewee. Distances of 15–20 ft were planned due to super emission of aerosols with increased human speech.20,21 However, this extensive physical distancing made hearing each other very difficult during interviews.
EHS designed a STUBB-Y barrier that consisted of two pieces of plexiglass positioned at a 90° angle with a HEPA air purifier fan to the side of the plexiglass nearest the interviewee to confine and strategically direct aerosols.
Clinic managers were also concerned about protecting research subjects from interviewers in the event of an asymptomatic staff member. This was a concern during interviews where documents had to be shared at close range. For this STUBB-Y, a single plexiglass barrier was designed with a slot to pass documents back and forth. The Honeywell HPA300 HEPA air purifier was placed in between both the interviewer and interviewee for aerosol containment from both sides (Figure 3).

Plexiglass barrier shield perpendicular to Honeywell HEPA fan. HEPA, high-efficiency particulate air filter.
The research subjects were inside of the clinic space for roughly 6 h per day and needed a place to eat during visits with their families. To provide a safe location for research subjects and their families to eat, a 6-foot-high barrier wall with two pieces of plexiglass at 90° angles was placed in the main break area to segregate research subjects and their families for lunch. 22 The Honeywell HPA300 HEPA fan was positioned on the table to draw exhaled air from the unmasked test subjects while they dined and filter it before recirculating these particles back to the room. The location of the HEPA fan for each setup was determined with smoke testing using the C-Breeze machine, not capture distance.
DrivSim Lab, developmental neurocognitive driving simulation research center, Department of Emergency Medicine
The DrivSim Lab needed a control to address aerosols that could be generated during the use of a breathalyzer. EHS worked with one of the physicians at this clinic to set up the short-term procedure in front of a Honeywell HEPA air purifier. Smoke tests with the C-Breeze machine were conducted to visualize aerosol capture and the device was deployed within the test laboratory. This clinic had access to N95 respirators for other exposures to nonmasked patients but wanted to confine what they believed was the greatest potential aerosol risk for their subjects. This engineering control helped to lower potential risks if a positive asymptomatic subject participated in their studies.
Neuropsychology clinics
Similar to the Alzheimer's clinic, the neuropsychology department was worried about potential risks from asymptomatic subjects due to prolonged proximity interviews with masked subjects. A plexiglass barrier plexiglass shield was constructed and placed between the patient and the doctor or study administrator. Figure 4 shows a HEPA fan placed perpendicular to the plexiglass shield to direct the airflow from interviewees. A pass-through window in the plexiglass shield was constructed by a contracted vendor. The dimensions of the plexiglass window largely depended on the size of the desk. A Honeywell HPA300 fan provided about 5 ACH for a 465 ft 2 room. 23 Smoke testing with the C-Breeze machine was conducted to visualize and confirm the movement of particles from the patient away from the physician.

Smoke test for neuropsychology plexiglass setup.
Autopsy services containment
With the increase in deaths related to COVID-19, there was also an elevated risk during autopsies to the Yale School of Medicine Pathology-Autopsy Services staff, specifically for brain extractions and lung inflations. A glovebox enclosure was created for removal of the brain from COVID-19 patients. Figure 5 shows how this design covered the head and upper torso of the patient and provided a negative airspace for the procedure. A removable 4-inch flexible duct was connected to an IVC rack HEPA filter fan to a common toilet flange mounted on the side of the enclosure.

Brain autopsy glovebox enclosure on top of downdraft table.
Two holes were cut into the plexiglass box for arm access for the operator to perform the removal. To help achieve higher airflow, iris ports were constructed from stainless steel rings and silicone sheets and added over the two precut holes, to further seal the enclosure. In addition, two holes were cut into the sides of the plexiglass enclosure for saw and cord access. Rotating plexiglass covers were made to open and close access to the ports on the sides. A large cut was made on the backside of the enclosure, opposite the iris ports, for the patient's torso. Strips of clear vinyl sheeting were attached at the top of the backside torso cut to help slow air infiltration between the patient and the enclosure. The IVC fan was tested annually under IEST-RP-CC002.4 guidelines by an outside vendor.
The lungs were also removed from the chest cavity of the patient and placed in a lung inflator machine. Owing to risk of SARS-CoV-2 and formaldehyde gas exposure, a plastic enclosure was constructed. Ducting was attached to the enclosure tent with an in-line fan. The ducting was then connected to the general room's hazard exhaust vent diffuser. Smoke testing with the C-Breeze machine and flowrates with the TSI VelociCalc 9525 Air Velocity Meter were performed to demonstrate containment for both enclosures, to ensure that there was no re-entrainment of exhausted aerosols, and to verify the flowrate was at least 75 fpm.
Yale Center for Clinical Investigation sample processing laboratory
Yale Center for Clinical Investigation (YCCI) needed benchtop containment for COVID-19 clinical laboratory samples. The laboratory had done work on the open bench, but they were concerned about the potential aerosol exposures from their prior operations. EHS created two bench top anest-arrest STUBB-Y plexiglass enclosures with >100 fpm inflow, which was verified with the TSI VelociCalc. Anest-arrest was a name provided by a Yale industrial hygienist for a similar containment strategy for exhausting waste anesthetic gases through a laboratory's hazardous exhaust diffuser in emergency situations until appropriate devices could be identified. In this configuration, the exhaust from the enclosure was connected to flexible ducting, which was connected to an in-line fan connected to more flexible ducting and then connected to the room's exhaust vent.
The experiments conducted in the laboratory could not be transferred to another space and there was no biosafety cabinet available in the room. As the laboratory could not wait to process samples, the plexiglass Class I enclosure provided a quick and safe solution. Later, the genetics laboratory utilized a similar anest-arrest enclosure for viable cells under microscopes.
COVID-19 test sample processing laboratories
Several laboratories on campus shifted to processing and testing COVID-19 samples due to the increase in demand for testing. A pathology laboratory purchased a robotic sample processing machine. EHS performed testing using fluorescent beads with Cyclex D filter cassettes at a 20 fpm flow rate as a surrogate clinical specimen. 24 Aerosols were not observed during sampling, but gross contamination within the enclosure was identified during the ejection of pipette tips. The group was interested in pursuing an enclosure at this location as they were set up to conduct testing for symptomatic subjects. The enclosure was also recommended as their laboratory was located off a public access corridor. Figure 6 shows how EHS constructed a negative pressure plexiglass enclosure that was exhausted through the laboratory's hazardous exhaust duct. Airflows at the face of the enclosure were verified with the TSI VelociCalc.

Class I plexiglass enclosure over robotic sampler with black in-line fan ducted to general HVAC system. HVAC, heating, ventilation, and air conditioning.
BSL-3 SARS-CoV-2 cell culture laboratory
A plexiglass Class I Hood was constructed for the BSL-3 facility at Yale. Figure 7 shows the enclosure was placed over a plate reader that was being used for high-throughput screening of SARS-CoV-2 infected cells. As this represented work outside a biosafety cabinet, there was a need for a primary containment enclosure. An anest-arrest enclosure was constructed. Rigid ductwork was placed up through the IVC rack connections that were placed in each BSL-3 room for flexibility of transitioning from laboratory to animal rooms. The exhaust went directly into the HEPA filters from the enclosure. The objective of this enclosure was to also provide inward directional airflow at ≥75 fpm across the front opening, which was achieved and tested with the TSI VelociCalc. The front sash was only opened for loading and closed while the instrument was in operation.

BSL-3 facility plate reader plexiglass enclosure with rigid ducting exhausted to HEPA-filtered HVAC system.
COVID-19 clinical trials
The clinical trials team needed LEV for containment of aerosols from temporarily unmasked symptomatic subjects for COVID-19 testing. The LEV needed to be HEPA filtered for the swabbing performed by healthcare staff as there was not a capacity to exhaust the air outdoors. The COVID-19 patients were seen between day 3 and 10, and were tested indoors at the clinic sites. 25 The proposed initial clinical locations included a gym and a temporary trailer, both of which had very poor HVAC controls.
EHS purchased an existing device used to control aerosols created in dental settings, but the cost of the device was problematic for other clinic sites. EHS then made an equivalent containment STUBB-Y. Figure 8 shows how EHS utilized an IVC Rack HEPA-filtered blower that was modified with a containment cone that could be positioned adjacent to the patient's face. Smoke testing with the C-Breeze machine and training identified the most appropriate location and configuration of the containment device. The IVC fan was tested annually IEST-RP-CC002.4 guidelines by an outside vendor.
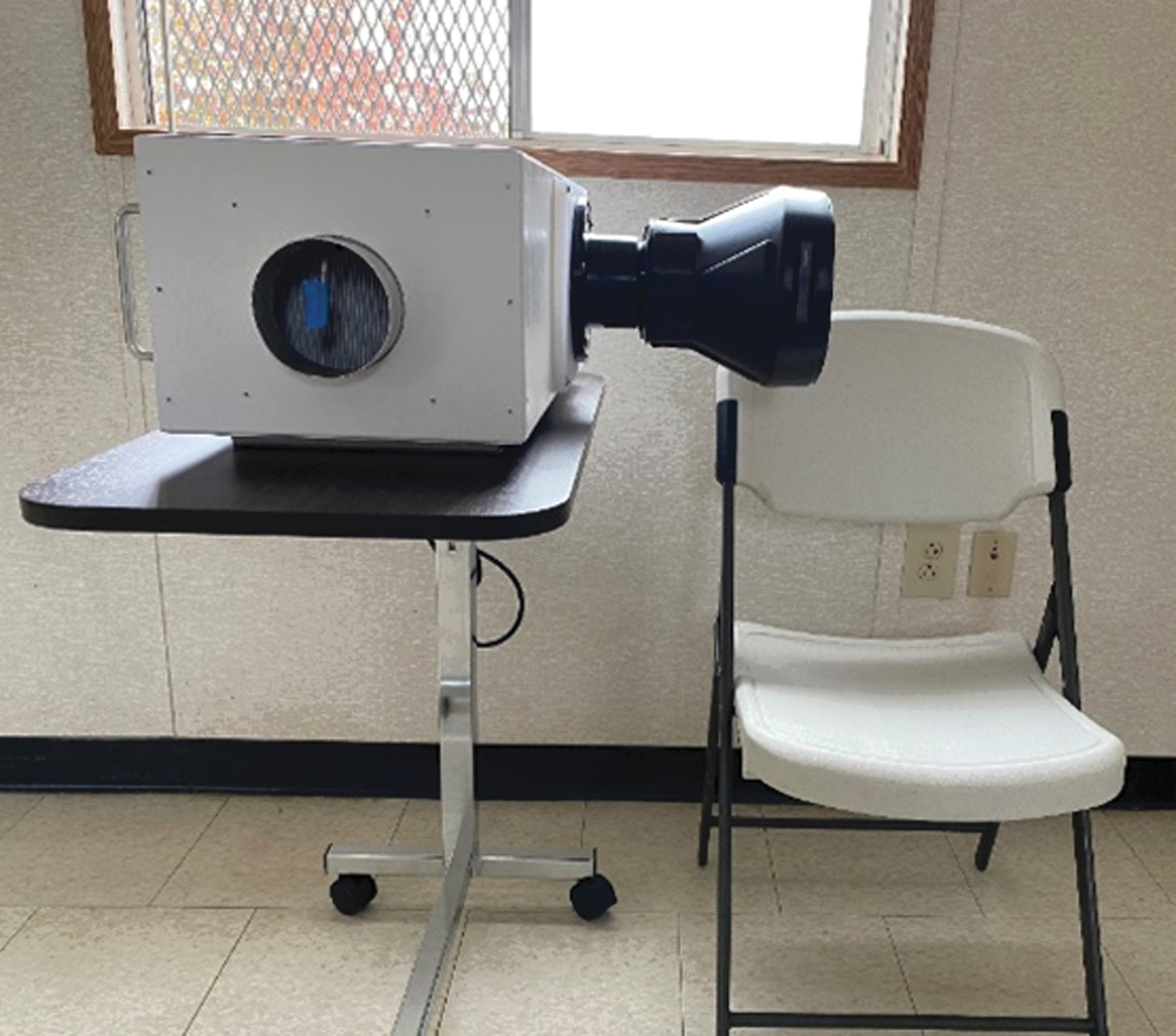
LEV system with IVC fan for self-administered COVID-19 swabs (aerosol extractor). LEV, local exhaust ventilation; IVC, individually ventilated cages.
This portable STUBB-Y was also successfully used as a local exhaust HEPA-filtered aerosol capture device for the Yale Positron Emission Tomography Center and multiple clinics within the Yale Health Center where tests had to be done with unmasked symptomatic patients. PPE such as N95s were available for these groups, but there was limited down time in these rooms and the need for additional decontamination of the room by custodial staff was time consuming.
Results
Owing to the success of these implemented projects throughout Yale's campus, many are still being utilized. Five additional aerosol extraction fans were requested in March 2022 for containment of aerosols during COVID-19 testing of suspect patients at Yale Health Center. The fact that these devices are still being requested from EHS 2 years into the pandemic is a testament to their success. This highlights the strength of the thorough design process for these simple innovative and inexpensive biocontainment enclosures.
Students, faculty, and clinicians were able to continue vital work and research during the pandemic. Seventy-two test booths were built and strategically placed around the campus to facilitate self-testing for COVID-19. Their design and ease of use was lauded as facilitating the mandatory testing requirements for students and affiliated campus personnel during the pandemic. The testing booths were also able to identify COVID-19 clusters on campus quickly and subsequently quarantine individual(s) involved and decrease the number of outbreaks, as were seen around the country in schools and at conferences.26,27
Discussion
One of the lessons learned from the STUBB-Ys was there were many HEPA fans that failed to contain 99.97% of particulates or meet the IEST-RP-CC002.4 test. 12 This was initially detrimental to the project as early plans for the self-swab COVID-19 test booths were to use large HEPA fans and exhaust air back to the room. Only one large commercially advertised HEPA-filtered exhaust fan was capable of being certified, but the technician had to use caulking around the perimeter of the large HEPA filter housing to ensure proper sealing.
The fans that did not pass were relegated as control devices where the exhaust was directed outdoors and away from any individuals or air intakes to prevent re-entrainment.
The project also ran into a problem with the size of the certified HEPA filter blower units. They were too large and there were too few. Many IVC Rack HEPA-filtered exhaust blower fans were repurposed for STUBB-Y projects from metal recycling bins. These units passed HEPA certification testing under the IEST-RP-CC002.4 test and were used for test center booths to provide negative pressure and were positioned near the breathing zone of the individual removing their mask for self-swabbing. New HEPA filters had to be installed in many of the units due to the characteristic smell associated with animal facilities.
The effectiveness of these projects was verified with annual fan certification by the outside vendor, smoke tests, flowrate checks, and particle counts. It is recommended that more in-depth verification measures be implemented if these projects were utilized as long-term solutions. This could include capture distance measurements, particle count measurements in a room during the specific medical evaluation or procedure, and COVID-19 cases comparison across multiple similar universities. The STUBB-Y projects, efficacy, and future use improvement ideas are summarized in Table 1.
Short-Term Use Biocontainment Bubbles-Yale summary table
Conclusion
The STUBB-Y program was an excellent team-building exercise for Yale EHS, linking experts from all disciplines within the EHS office: biosafety, industrial hygiene, physical safety, and environmental affairs. The challenges presented tapped into EHS's collective and diverse knowledge and expertise, which was greatly needed for innovative approaches to help protect workers at Yale in the early stages of the pandemic.
From a biorisk management point of view, the lessons EHS learned refer to the “Golden Age of Biosafety” (1949–1979: when the bulk of the research that cemented our knowledge of our profession and elucidated these basic control measures), were validated as we used them in each of our projects. Dr. Arnold Wedum's “confine aerosols as close as possible to their point of generation” was the starting point of each discussion and was immediately followed by a simple diagram to illustrate how to contain the potential source of biohazard risk. 28 An adherence to the hierarchy of controls was also critical, to ensure that all measures that could be applied were executed.
These opportunities reinforced the central mission of our vocations and provided another example of how biorisk management can be utilized to control hazards in the workplace. These STUBB-Ys demonstrated how a straightforward design process can be used to enhance worker protection during a pandemic or future potential high-risk situation.
Footnotes
Acknowledgments
EHS spent considerable time with representatives from Yale Facilities Department, Custodial Services, Yale specialized machine shops, vendors, Animal Resources Center, Yale Health Center, and Yale New Haven Hospital.
Authors' Contributions
All listed authors contributed significantly to this study.
Authors' Disclosure Statement
All authors are employees of Yale University's Environmental Health and Safety.
Funding Information
No funding was received for this article.