Abstract
Introduction:
Over the past decade, there have been outbreaks associated with high consequence infectious diseases such as Ebola virus disease, Lassa fever, and Monkeypox. The proper handling of clinical waste from patients infected with such pathogens is critical to ensure healthcare personnel and community safety.
Methods:
Mock clinical waste bags were created to simulate four distinct waste streams: personal protective equipment (PPE), dry linens, wet linens, and solidified liquids. Pressure and temperature data loggers were buried in the middle of simulated waste loads to record time at a sterilization temperature of 132°C (270°F) to reduce sterilization time. We also validated super rapid biological indicators (BIs) by embedding standard BIs (48 h), rapid BIs (3 h), and super rapid BIs (24 min) within each load. Cycles were validated over a 2-day period, using a total of 36 simulated waste bags (6 bags each for PPE, dry linen, and wet linen, and 18 bags for solidified liquids).
Results:
All waste bags achieved the target sterilization temperature, all BIs passed and cycle times were substantially decreased. For PPE waste processing, an estimated 15 h was saved for a 24-h period.
Discussion:
Default factory settings are inadequate to disinfect Category A clinical waste. Reliance on autoclave temperature readings may overestimate time at goal sterilization temperature for actual waste loads.
Conclusions:
The data provided by within bag data loggers and BIs allow for the optimization of autoclave parameters to increase throughput and enhance staff safety.
Introduction
Treating high consequence infectious diseases (HCIDs) presents a number of unique challenges. Prominent among them is the safe handling and inactivation of Department of Transportation (DOT) Category A classified infectious substances1,2 defined as “An infectious substance in a form capable of causing permanent disability or life-threatening or fatal disease in otherwise healthy humans or animals when exposure to it occurs.”3,4 Patients with HCIDs can produce significant amounts of this potentially infectious Category A waste over the course of their hospital stay.5,6
Given the high cost and logistical constraints of transporting Category A waste offsite for inactivation, the treatment of waste onsite with the use of a steam sterilizer or autoclave allows facilities to treat processed waste as nonhazardous material or regulated medical waste. 1 Centers for Disease Control and Prevention (CDC) guidance for the proper disinfection of waste from patients infected with the Ebola virus recommends treating at 121°C (250°FF) for at least 30 min. 7 Since there have been recent outbreaks of several Category A HCIDs (e.g., Nipah in India, 8 Lassa fever and Monkeypox in Nigeria,9,10 and Ebola in the Democratic Republic of Congo 11 ), this onsite capability is critical for developing a robust emergency waste management plan for use during the care of patients infected with HCIDs.
The Johns Hopkins Biocontainment Unit (JH BCU) is dedicated to the safe care of patients infected with HCIDs. Established in response to the 2014 Ebola virus disease outbreak in West Africa in 2014, the JH BCU is one of 10 federally funded Regional Emerging Special Pathogen Treatment Centers.2,12,13 The JH BCU houses two PRIMUS PSS-500 (Spire Integrated Solutions, Orlando, FL) pass-through autoclaves for decontamination of DOT Category A waste. During the development of initial standard operating procedures in 2015, the autoclave cycle settings were modified from factory settings using several types of biological indicators (BIs) to confirm decontamination of simulated waste bags. 1
To maintain the activation readiness of the JH BCU, autoclave cycles are validated quarterly to demonstrate effective decontamination has occurred inside and outside of simulated waste bags. The temperature within each autoclave bag is not currently measured as part of this validation process. During clinical activation, a single test pack with a steam indicator that changes color upon contact with steam, and a rapid BI, is placed in the middle bin underneath the packaged waste of every load and read out as negative (after 3 h of incubation) before final packaging and removal from JH BCU as regulated medical waste marked for incineration.
Since the initial development of the validation process, the JH BCU autoclaves have undergone a number of modifications to improve operational performance: (1) the addition of a larger air compressor, (2) the addition of a second reservoir with an auto-blowdown to remove residual moisture from the air system and increase the longevity of the compressor, and (3) the addition of fascia panel vents to reduce the external ambient temperature of the sterilizer controller.
To optimize the activation readiness of the JH BCU and to ensure that waste is adequately decontaminated, the autoclave cycles and quarterly validation process were re-evaluated with four objectives: (1) to standardize the contents and packaging of simulated waste used during validation processes to better reflect the actual waste that would be produced during unit activation, (2) to validate autoclave cycle settings using National Institute of Standards and Technology traceable pressure and temperature data loggers within waste bags, (3) to decrease overall cycle times required for inactivation by using higher pressure set points to achieve a temperature goal of 132°C (270°F), and (4) to validate the use of super rapid (24 min) BIs and test packs to optimize waste throughput during activation.
Materials and Methods
General Autoclave Characteristics
The JH BCU autoclaves are PRIMUS PSS5-D-MSDD Effluent Pass through sterilizers. Each sterilizer chamber is 26″ × 26″ × 67″ (width, height, and length) with a double door that allows loading of contaminated material on one side of the autoclave and unloading of treated waste on the other side. Chamber steam, condensate, and vapor are held inside the chamber throughout the sterilization cycle as part of an effluent decontamination configuration. Steam enters the chamber from the bottom to ensure that the condensate is kept at the sterilization temperature throughout the cycle. The vacuum is pulled from the top of the chamber through a 0.2-μm filter to ensure adequate filtration before the drain.
Standardized Simulated Waste Bag Contents and Transport
Simulated waste bag contents were standardized for all cycles to reflect realistic waste products with characteristics similar to those produced during an actual unit activation. To take into account the variability that occurs with waste handling during clinical activation, extreme scenarios were considered in terms of selecting bag contents, and compactness of simulated waste loads. A summary of simulated waste bag contents is provided in Table 1. For example, wet linens were packaged to simulate a linen change after an incontinence event. Wet linens are packaged in a tight wad of saturated linens to contain infectious bodily fluids and reduce healthcare worker contamination.
Contents of simulated waste bags for each type of clinical waste
BI, biological indicator; DSFS, Dual Semi-Flex Sensors; HCW; PAPR; PPE, personal protective equipment; PTS, Pressure + Temperature Sensor.
During unit activation, garbage bins in the patient room (Rubbermaid Streamline, 24-gallon step-on resin front step-on container, Huntersville NC) are prelined with a single autoclavable biohazard waste bag (Medegen Medical Products, Gallaway, TN). When the bin is filled with the appropriate waste (e.g., personal protective equipment [PPE]), the in-room provider loosely gathers the neck of the autoclave bag and secures it with a single rubber band (pale crepe gold, item no. 909713; Alliance Rubber Company, Hot Spring, AR).
A red garbage can on wheels (Rubbermaid Brute, 32-gallon plastic red round trash can, Huntersville, NC) is prelined with an autoclave bag in the doffing room and wheeled to the exit door of the patient room. The in-room provider deposits the loosely secured autoclave bag into the red garbage can by reaching across the exit threshold. The doffing room attendant then clips the top of the outer bag with a 2-inch binder clip (Item no. 308957; Office Depot, Boca Raton, FL) to limit spillage during transport. The double-bagged waste is deposited into a trolley on the clean side of the doffing room.
A third team member then transports the trolley to the autoclave room for loading into the autoclaves (Figure 1). When the waste is transferred from the trolley into an autoclave bin, the binder clip on the outer bag is removed to allow steam to enter the inner bag during the autoclave cycle. A small strip of steam tape (3M Comply™ Steam Indicator Tape, 1355-24MM; 3M Company, St. Paul, MN) that changes colors upon exposure to steam is also placed on the outside of each bag as a process indicator.

Transport of waste from patient room to autoclave room.
The maximum capacity of each JH BCU autoclave is three bins. Each bin can fit either one large bag (38″ height and 47″ width) used in PPE, dry linen, and wet linen cycles, or three medium bags (31″ height and 38″ width) used in the solidified liquid cycles. As a result, the maximum capacity within each autoclave for PPE, dry linen, and wet linen cycles is three simulated waste bags, and the maximum capacity for the solidified liquid cycle is nine simulated waste bags (Figure 2).

Layout of autoclave bins, BIs, and waste bags.
Packaging Simulated Waste with BIs and Data Loggers
The BIs used in the validation cycles all contained Geobacillus stearothermophilus. G. stearothermophilus spores have a high moist-heat resistance and are among the most commonly used BIs used to monitor the effectiveness of steam sterilization protocols. 14 They are more heat resistant than the Category A pathogens that might be encountered clinically and provide a conservative estimate for the likelihood of decontamination of those pathogens.
For dry linen and PPE cycles, standard (3M Attest™ Biological Indicators 1262/1262P; Geobacillus stearothermophilus ATCC 7953, Population [mean/strip] 2.4 × 105 C.F.U, kill-time 19.70 min at 121°C [250°F], 48-h incubation time), rapid (3M Attest™ Rapid Biological Indicator 1292; Geobacillus stearothermophilus ATCC 7953, Population [mean/strip] 4.7 × 105 C.F.U, kill-time 16.44 min at 121°C [250°F], 3-h incubation time), and super rapid (3M Attest™ Super Rapid Biological Indicator 1492V; Geobacillus stearothermophilus ATCC 7953, Population [mean/strip] 1.5 × 106 C.F.U, kill-time 4.88 min at 132°C [270°F], kill-time 4.07 min at 135°C [275°F], 24-min incubation time). BIs were tied to a length of twine with a metal washer for ease of retrieval and to ensure BIs were anchored in the middle of the simulated waste bags (Figure 3A).
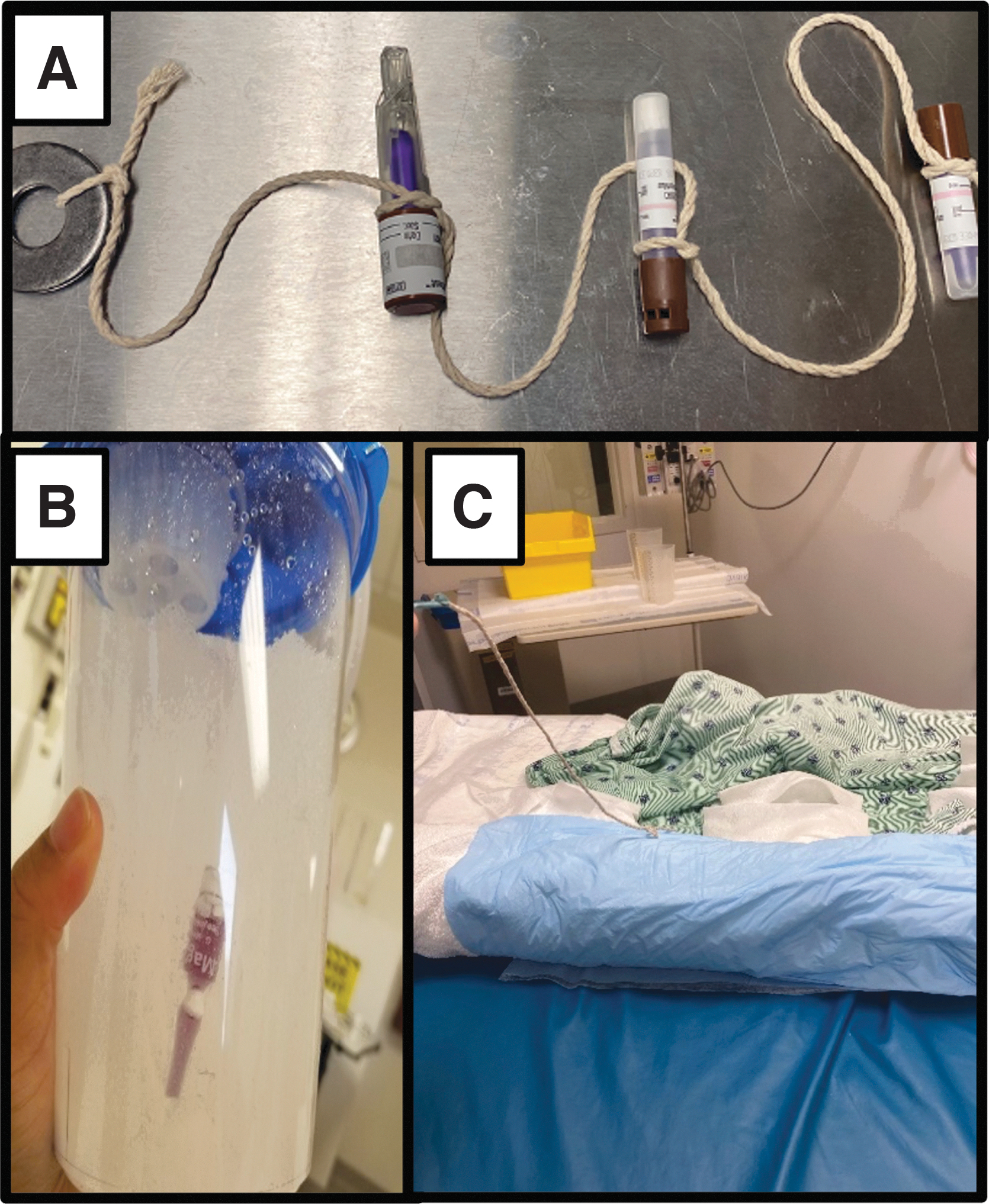
Examples of buried BIs for simulated waste.
In the solidified liquid and wet linen cycles, submersible BIs (Mesa Labs MagnaAmp II™ Biological Indicators, Bozeman, MT; Geobacillus stearothermophilus ATCC 7953, Population [spores/unit] 2.6 × 106, kill-time 21.51 min at 121°C [250°F], 48-h incubation time) were used in place of the rapid, super rapid, and standard BIs. One submersible BI was placed inside each of the water and solidifier-filled suction canisters (Figure 3B) or rolled in the middle of each wet linen package (Figure 3C).
In all cycles, one super rapid test pack (3M Attest™ Super Rapid Steam-Plus Challenge Pack, Catalog Number 41482V) and one rapid test pack (3M Attest™ Rapid Readout Test Pack, Catalog Number 1292) were placed in each of the three bins under the packaged waste. In addition to BIs and test packs, a data logger was also buried in the middle bag of each load. Dual Semi-Flex Sensors were used in the solidified liquid cycles and Pressure + Temperature Sensors (Ellab, Inc., Denver, CO) were used in all other cycles (Figure 4). Third Generation Pro Loggers recorded data from the sensors using ValSuite Pro 6.0.16.0 software (Ellab, Inc.).
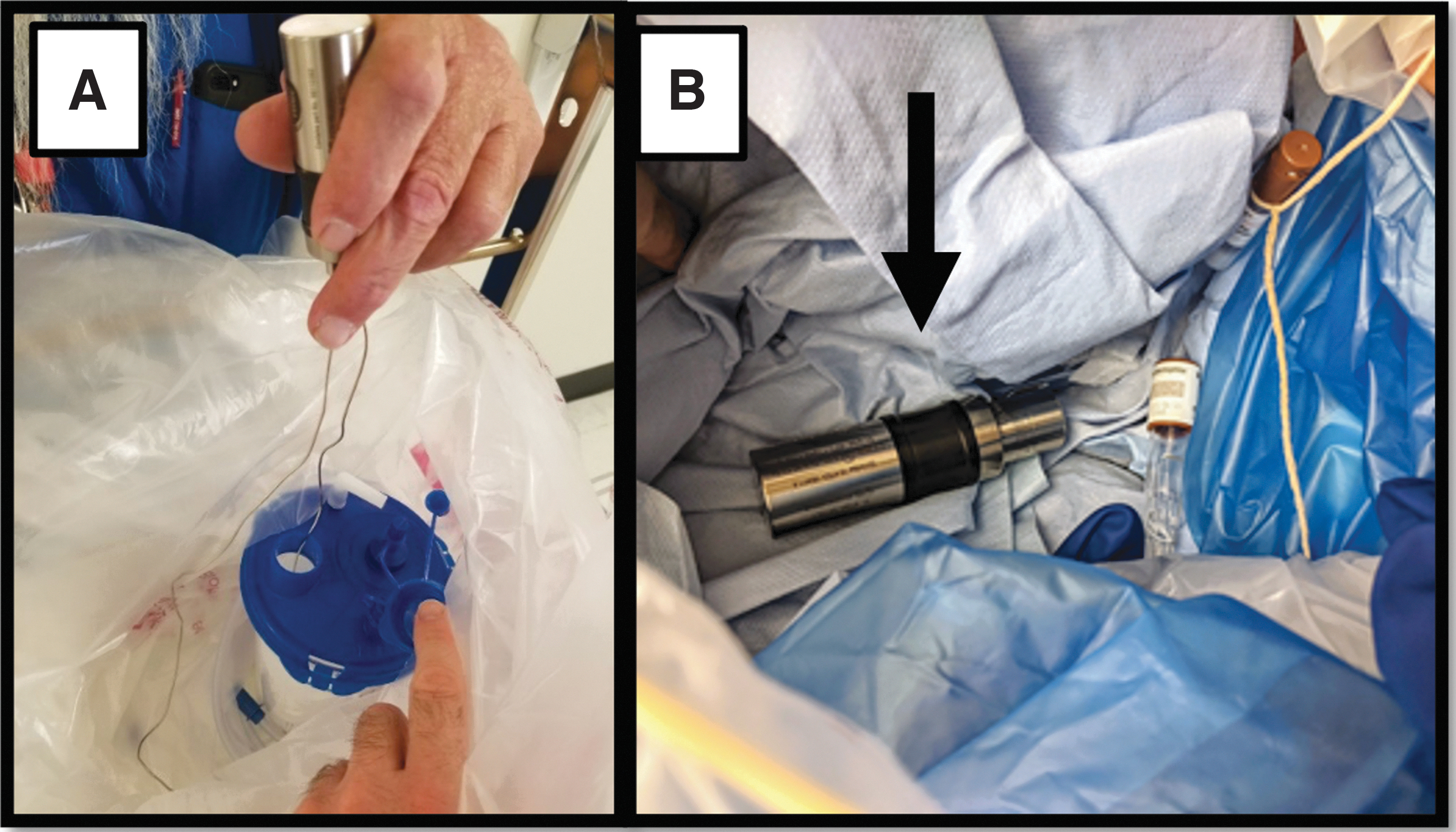
Examples of buried data loggers for simulated waste.
Operating Parameters
The CDC recommends that waste from patients infected with Ebola virus be sterilized for at least 30 min at 121°C (250°F). 7 JH BCU autoclave cycles were set to a higher temperature goal of 132°C (270°F) with sterilization times as short as 20 min in an effort to reduce cycle time and increase waste throughput. Over the course of 62 trial loads with BIs and 122 trial loads with data loggers, new autoclave cycles were configured for each of four waste streams: PPE, dry linen, wet linen, and solidified liquid. These parameters were designed using the principles outlined by John J. Perkins for use in autoclave sterilization 15 (Table 2).
Updated autoclave parameters compared with previous validated settings
In previous publication, saturated linens were tested but no cycle was ever designed to specifically address inactivation of saturated linens.
N/A, not applicable.
Cycles run at original settings while using data loggers showed that the desired temperature was not achieved even though the BIs were effectively killed. This was thought to be due to the fact that the 15 pounds per square inch gauge (psig) prevacuum (prevac) pressure pulses were not sufficient to heat the loads. Adjustment to pressure pulses from 15 psig to 32 psig (determined by using standard steam tables) was made to achieve the desired temperature as required. PPE and linen cycles used three prevac pulses at 15 psig at 273°F (134°C). We increased the prevac pressure to 32 psig and the temperature was then achieved and confirmed using the data loggers with the original sterilization time. We ran several cycles and occasionally would fall short of the designated sterilization time.
To ensure we achieved our designated time settings at 273°F (134°C), the temperature was increased to 276°F (135°C). We then continued to decrease the sterilization time to increase waste throughput. The liquid cycle initially used water in the suction canisters and was considered to be a “Hybrid Liquid” cycle. This cycle used prevac pulses for air removal and used a slow exhaust as the last phase of the cycle to allow for cooling of the water to prevent a burn hazard upon unloading. This cycle took 4 h to complete.
Looking across the medical facility at how normal waste was collected, we decided to incorporate the process of using a solidifier in conjunction with the suction canisters. Using a solidifier made the liquid more of a porous substance and changed the cycle from a Hybrid Liquid to a standard Prevac Cycle. This decreased the cycle time to 3 h.
Validation of the cycles occurred on November 4 and November 5, 2021. Each cycle was validated once on each autoclave using three bins per cycle (i.e., 18 simulated waste bags were run for the solidified liquid cycle and 6 simulated waste bags were run for the other waste streams for a total of 36 validation waste bags). A comprehensive time-stamped view of temperature and pressure trends from data loggers was obtained from the center bag in each validation cycle and matched with the corresponding BIs from that cycle.
Results
All eight validation cycles reached the goal sterilizing time of at least 20 min at or above 132°C (270°F). The dry linen, wet linen, and solidified liquid cycles also achieved at least 30 min of sterilizing time at or above 121°C (250°F). Figure 5 shows the pressure and temperature measurements from a sample dry linen cycle and demonstrates that the autoclave temperature effluent reaches goal sterilization temperature several minutes before that temperature is recorded in the middle of the waste bag. In addition to achieving target sterilizing times at 132°C (270°F), all BIs within the simulated waste loads as well as all BIs in the test packs outside of the waste loads passed (Table 3).
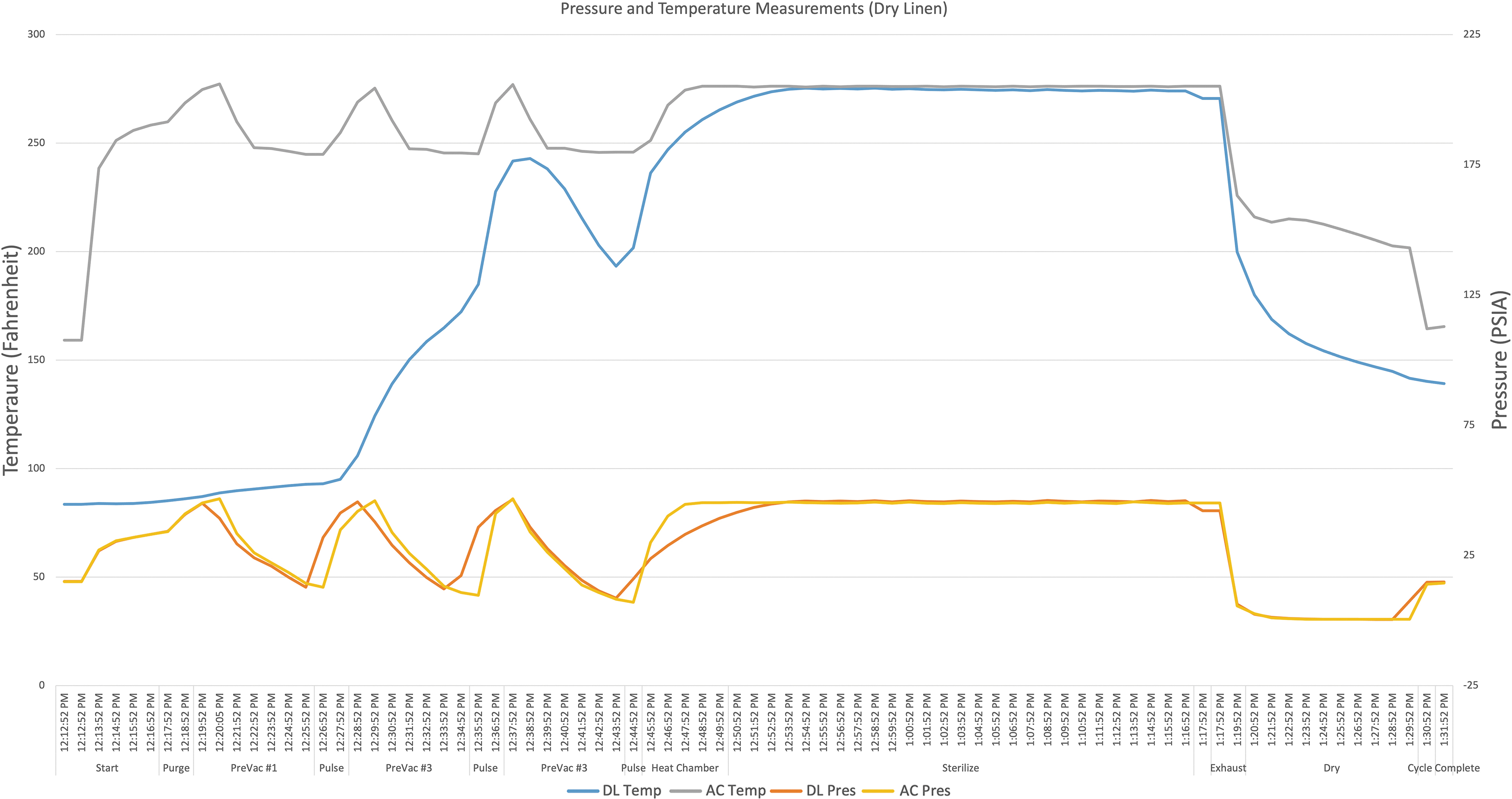
Example of a dry linen cycle comparing pressure and temperature readings from the factory autoclave sensors to the within bag data loggers. AC, autoclave; DL, data logger; Pres, pressure; PSIA, pounds per square inch absolute; Temp, temperature.
Performance of new autoclave parameters during validation
For the PPE, dry linen and wet linen cycles, each bin contained one autoclave bag (three bags per cycle). For the solidified liquid cycle, each bin contained three autoclave bags (nine bags per cycle).
BIs on the outside of the waste bags are included as part of a test pack that includes a temperature indicator, steam indicator, and the BI.
The new parameters decreased overall waste processing time by reducing the time of each autoclave cycle. Cycle time was decreased by 10 min for PPE, 50 min for dry linens, and 25 min for solidified liquids. We did not previously have a dedicated wet linen cycle so no time savings could be recorded. The validation of the super rapid BIs also reduced processing time since they are read in 24 min as opposed to the 3-h incubation time for the rapid BIs. Table 4 gives the estimated time saved based on anticipated trash volumes for a viral hemorrhagic fever patient over a 24-h period.
Estimated time saved using new cycle parameters and rapid biological indicators
Based on these estimates, overall processing time in a 24-h period was reduced by 996 min for PPE, 206 min for dry linens, and 181 min for solidified liquids. The time saved is most notable for PPE cycles since PPE is expected to generate the largest volume of waste over a 24-h period given the need for multiple healthcare providers to enter and exit the clinical care space several times during a shift.
Discussion
The management of DOT Category A infectious waste is one of the biggest challenges in providing care for patients infected with HCIDs.16–18 In this study, we used data loggers embedded in simulated waste loads to optimize a previously validated decontamination process for such clinical waste. Data gathered from the loggers confirmed that target decontamination temperatures were achieved inside simulated waste loads. The use of data loggers in combination with super rapid BIs also led to a substantial reduction in waste processing time by confirming the effectiveness of higher pressure and temperature targets and reducing incubation time for BIs. These innovations will enhance staff safety and increase efficiency when handling this clinical waste.
Our study reiterates the important point that the content and packaging of clinical waste impact the cycle parameters needed to ensure adequate decontamination. It also redemonstrates that preset factory cycles are likely inadequate for clinical waste disinfection. 1 We tested over 100 different cycles before selecting individual parameters to accommodate each of four unique waste streams: PPE, dry linen, wet linen, and solidified liquid waste. Health systems looking to use autoclaves to decontaminate HCID clinical waste need to optimize and validate their cycle parameters using the products and materials from their clinical environment to ensure the safety and efficacy of disinfection.
The data loggers within the bags reached goal sterilization temperature several minutes after the autoclave sensor recorded goal temperature in the autoclave effluent demonstrating the phenomenon of “load lag.” 15 It is essential that autoclave cycles are validated with data loggers buried in mock waste loads to account for this delay in achieving target load temperatures. This validation is critical to ensuring staff safety when handling waste after it has been processed and thought to be disinfected. We were able to potentially shorten cycle times by using a higher target temperature of 132°C (270°F) and confirming a minimum sterilization time of at least 20 min at this temperature.
In our previous validations, we were unable to successfully achieve kill of BIs embedded in solidified liquid waste. 1 The use of submersible BIs coupled with data loggers with flexible sensor probes that could be threaded through the opening of the suction canister allowed us to find settings that adequately achieved sterilization temperatures, confirmed by multiple BIs. This is an important update to our current protocols as solidifying liquid waste greatly reduces the length of time required for decontamination and significantly reduces the risk of spills and exposure to healthcare personnel handling liquid waste.
The time saved using these updated protocols is substantial and is mostly due to the use of the super rapid BIs. For example, PPE waste processing over a 24-h period was reduced by over 15 h due to the estimated six autoclave runs required to handle the large volume of PPE used in clinical care. Since staffing ratios are higher when caring for patients infected with Category A pathogens, 19 time saved in waste processing can free staff to assist in other essential unit operations.
There are limitations to this study. Our validation processes reflect the experience of a single institution. As illustrated by the number of loads tested to select our autoclave parameters, each facility needs to optimize and validate their own protocols using facility-specific simulated waste to ensure effective decontamination. Our temperature and pressure monitors only recorded data from the center bag or canister of each autoclave load.
It is possible that temperature variation within the autoclave could lead to areas that did not achieve target temperature for the set period of time in each cycle. It is also possible that there were areas within each bag where steam did not penetrate. Although the use of BIs in each bag and cannister indicated effective decontamination, future experiments will utilize additional data loggers and BIs to better map the temperature profile of each autoclave and ensure adequate steam penetration throughout each waste bag.
The final validated cycle parameters require the autoclaves to operate at a higher overall pressure than our previous cycles. There is a possibility that this could lead to faster wear on the seat of the valves (Burkert 2000 A2, two-way piston operated angle seat valve, Burkert Fluid Control Systems, Ingelfingen, Germany). However, the valves are replaced on a yearly basis as part of our existing service agreement. We have not experienced an increase in mechanical problems as a result of the new parameters.
We used a higher target temperature of 132°C (270°F) to reduce overall decontamination time. The dry linen, wet linen, and solidified liquid cycles still achieved at least 30 min of sterilization time at 121°C (250°F) as recommended by the CDC for Ebola waste processing, but the PPE cycle did not achieve 30 min at 121°C (250°F). Since the biggest decrease in processing time was achieved using the super rapid BIs, the PPE cycle could be adjusted to increase sterilization time at 121°C (250°F) without greatly increasing overall time.
Current CDC guidance was developed to create a buffer to ensure decontamination since pathogens such as Ebola can be inactivated by heating to 72°C (162°F) to 80°C (176°F) for 30 min. 7 Heating for 20 min at 132°C (270°F) should be adequate to sterilize Category A waste as supported by the successful inactivation of multiple BIs in each PPE cycle with kill temperatures ranging from 121°C (250°F) to 135°C (275°F) and kill times ranging from 4.07 to 19.70 min.
Our validated cycles for wet linens and solidified liquid used water. It is possible that the viscosity and density of bodily fluids such as blood would require additional sterilization time. Simulated waste bags used in the validation process are packaged to ideal volumes and contents. Variability in waste packaging and unanticipated additional items during actual clinical care might lead to variation in decontamination performance. Future validations will target a wider variability in the contents and packaging of mock waste loads to account for heterogeneity of clinical waste during unit activation.
Conclusions
This study demonstrates the value of using data loggers and BIs embedded inside simulated waste loads to ensure adequate decontamination of Category A clinical waste. The data provided by such devices allow for the optimization of autoclave parameters to increase throughput and enhance staff safety when handling processed waste loads.
Footnotes
Authors' Contributions
J.F. contributed to conceptualization, methodology, investigation, writing—original draft, and writing—review and editing. A.M. and L.B. carried out methodology, investigation, writing—original draft, and writing—review and editing. N.A.H. was involved in conceptualization, writing—original draft, and writing—review and editing. D.P. was involved in conceptualization, methodology, software, visualization, investigation, writing—original draft, and writing—review and editing. B.T.G. carried out conceptualization, methodology, visualization, investigation, writing—original draft, writing—review and editing, data curation, and funding.
Authors' Disclosure Statement
The authors have no conflicts of interest to report.
Funding Information
This study was supported by the Office of the Assistant Secretary for Preparedness and Response through the Hospital Preparedness Program in partnership with the Maryland Department of Health.
