Abstract
Introduction:
Although the COVID-19 pandemic began over 2 years ago, responses from institutions have not been thoroughly documented. Because of constantly evolving guidance from regulatory and public health agencies, the traditional evidence-based risk assessment process was disrupted. It took time to obtain enough actionable information about the novel SARS-CoV-2 virus and its transmission to be able to inform decision making and consistently make accurate recommendations.
Objectives:
The primary objective of this article is to detail the actions in chronological order taken by the biosafety team at Arizona State University in response to the COVID-19 pandemic.
Methods:
This article includes a chronicling of actual experiences and observations with a retrospective review and analysis of the actions taken to document and share lessons learned to guard against future pandemics. Specific infection control practices, such as measuring saliva droplets from dropped test tubes, are described and different COVID-19 testing and vaccination site designs are evaluated.
Results:
The biosafety team helped collect more than 304,000 saliva samples, deliver 15,528 vaccinations, safely manage more than 170,000 gallons of biohazardous waste, and oversee the pandemic response inventory. The team also conducted inspections, developed safety procedures, and reviewed research protocols.
Conclusions:
The COVID-19 pandemic required safety professionals to re-envision how they perform their work, not only at their place of employment, but also within their communities and at their homes. This presented a challenge to balance inquiry and the scientific process against the urgent need for understanding changing pandemic dynamics and information and developing new best practices.
Introduction
This article describes actions taken by Arizona State University during the COVID-19 pandemic. The goal of describing this response in detail is to share experiences and discuss lessons learned from the perspective of a biosafety program at a large research and teaching institution. There were several factors that were crucial for the institution to successfully respond to the pandemic, including the ability to perform infection control, implement timely mitigation measures, and leverage on-campus staffing and resources. This article is designed to be a resource to other institutions and safety professionals, and to serve as a possible framework to help prepare for and manage future large-scale public health crises.
Methods
January Through March 2020: The First 3 Months
In early January 2020, the biosafety team within the Environmental Health and Safety (EHS) department at Arizona State University began to closely follow and track news reports about a novel coronavirus (known as 2019-nCoV at the time) and its rapid spread. The World Health Organization (WHO) announced on January 5 that a coronavirus-related pneumonia was detected in Wuhan City within the Hubei Province of China. 1 There were 59 cases reported as of January 9, and discussions regarding travel precautions had begun in the United States. 2
The biosafety team, which has responsibility for infection control at the institution, initiated conversations with the university's health care organizations on January 20 after a Chinese health official reported, “14 cases in health care workers–a first–and a confirmed incident involving human-to-human spread of the new virus.” 3 After discussion with the biosafety team, the university's health care organizations promptly began to post signage in English, Chinese, and Pinyin, at entrances and reception areas to alert individuals who may have recently traveled to China to wear a face cover before entry and to notify the nurse triage station of any concerns. The first case of 2019-nCoV in the United States was reported on January 21, 2020, 4 although global cases were known at least 5 months earlier. 5
Arizona State University had previously created a Pandemic Committee (renamed to be the Emerging Infectious Diseases Committee in 2019) shortly after the Severe Acute Respiratory Syndrome outbreak of 2003. 6 The committee had an annual budget of $33,000 per year that was used to purchase supplies for point-of-distribution centers to give medication (e.g., antibiotics, antivirals) or vaccinations in the event of an outbreak.
The EHS staff began inventorying pandemic storage stocks on January 21. The inventory included tens of thousands of N95 respirators, surgical masks, nitrile gloves, disposable isolation gowns, shoe covers, tents, and other pandemic-related supplies. Table 1 provides a listing of contingency supplies and quantities on hand for essential staff at Arizona State University that would be required for 60 days of operation under pandemic conditions. Table 2 provides a list of equipment maintained by EHS to support medical care and critical infrastructure.
Contingency supplies and quantities during the COVID-19 pandemic
HEPA, high efficiency particulate air; PAPR, powered air purifying respirator; PPE, personal protective equipment.
Equipment maintained by Environmental Health and Safety during the COVID-19 pandemic
On January 22, the EHS team contacted senior leadership at the university to encourage all faculty, staff, and students to stay home if they were feeling sick and to seek medical attention if necessary. A day later, leadership began to query the university community to determine whether any individuals had recently traveled from Wuhan, China.
On January 24, the Emerging Infectious Diseases Committee held a 4-h tabletop exercise to assess the institution's readiness capabilities and to prepare for a possible future positive case at the institution. The scenario involved a 20-year-old suspect case who self-reported to a university health care organization with flu-like symptoms and a fever, with reported travel within the Wuhan boundary in the past 14 days. The tabletop was led by the university's emergency preparedness director, chief medical officer, and the head of EHS.
The exercise had 25 staff members with expertise from the institution's health care centers, emergency preparedness team, EHS, study abroad, international students' office, news and media, technology, dining, housing, facilities, finance, legal, research, transportation, and other key stakeholders. At the time of the tabletop, a U.S. State Department Level 4 Travel Advisory (i.e., do not travel) was in effect, but it was limited to Hubei Province, where all non-emergency U.S. government personnel and family members had been ordered to depart.
Two days later, the first case of 2019-nCoV reached Arizona State University. 7 The university opened the Emergency Operations Center, launched the institution's first coronavirus information website, and updated the essential personnel list on January 27. The next day, the university president banned all travel to China and requested that EHS distribute alcohol-based hand sanitizer and surgical masks from the previously established pandemic supply to campus health care facilities and building entrances.
At that time, EHS had more than 100,000 surgical masks available and worked closely with the institution's procurement division to order 26,000 four-ounce bottles of alcohol-based hand sanitizer. The EHS staff started to create “Coronavirus Safety Kits,” consisting of surgical masks, nitrile gloves, hand sanitizer, and disinfectant spray for distribution at designated drive-up locations for employees and students. The biosafety team issued its first safety document related to 2019-nCoV, titled, “Surface decontamination procedures for coronavirus and other respiratory viruses.”
This document was distributed to the institution's facilities department and posted on the EHS website as a reference document. On February 11, 2020, the WHO announced a new name for the disease sweeping the globe: COVID-19. 8
Over the course of February 2020, housekeeping staff were instructed to disinfect high touch surfaces once a day by using Environmental Protection Agency-registered disinfectants for coronaviruses. 9 The Emerging Infectious Diseases Committee met daily and held two additional tabletops to perform a dry-run of quarantine operations in campus housing and to determine how to manage large, university-related sporting events. The university president issued a proclamation about the need to be resilient, to practice positive community values, and to be supportive of the campus Asian community. Study abroad programs were deferred for the semester due to concerns from around the world.
In February, the biosafety team was asked to support a new COVID-19 clinical testing laboratory at the institution. Biosafety staff provided facility and equipment guidance, developed operating procedures, and conducted laboratory inspections. They also established personal protective equipment (PPE) criteria to safely handle and collect positive samples and reviewed sample inactivation protocols. They conducted site visits where nasal swabs were to be collected, reviewed transport and chain-of custody protocols, and created training materials for frontline health care workers and laboratory personnel.
For the clinical testing laboratory to launch, highly specific equipment and laboratory devices were required to process SARS-CoV-2 samples. The biosafety team evaluated different types of high-throughput machines to be used in different stages of the diagnostic sample processing for safety concerns, determined the appropriate locations for equipment to be placed in the laboratory, and recommended safety measures for personnel operating the equipment and those in the nearby vicinity. For high-throughput equipment generating large volumes of waste, the biosafety team determined safe and effective waste disposal methods to facilitate normal operations.
Non-inactivated diagnostic samples were handled in Biosafety Level (BSL)-2+ laboratories. BSL-2+ procedures included handling all diagnostic samples inside a biological safety cabinet and using the following PPE: facility dedicated clothing, scrubs and shoes, disposable lab coat, safety glasses, N95 respirator, and double gloves. Wearing Tyvek or plastic sleeves over disposable laboratory coats was optional. The biosafety team developed a train-the-trainer procedure to demonstrate the proper donning and doffing of PPE when entering and exiting the BSL-2+ laboratories.
The biosafety team developed an N95 respirator reuse protocol that was implemented when supplies were very limited and when long supply chain delays were experienced. This protocol was analogous to those used at hospitals around the country, following Centers for Disease Control (CDC) and Food and Drug Administration guidance. All laboratory personnel working with diagnostic specimens were trained in the N95 respirator reuse procedures. Laboratory personnel who did not pass a fit-test in the available N95 respirators were provided with half-face respirators.
Laboratory personnel were provided with the required PPE to work safely with diagnostic samples, and location-specific, step-by-step instructions on how to don and doff PPE. Special instructions were also provided before the personnel could enter BSL-2+ spaces. For example, the procedure for the laboratory stated, “Do not open the door or enter the laboratory if samples are being manipulated in the biosafety cabinet.” Procedures also included the steps to follow in case of a spill or personal exposure. Instructions describing the order for PPE donning and doffing and N95 reuse instructions were printed and posted in laboratories.
As the clinical testing laboratory expanded operations, an increase in space was needed. The biosafety team inspected locations to process non-inactivated and heat-inactivated samples and areas to store waste. The biosafety team conducted pre- and post-inspections for all proposed laboratories to determine the best locations for BSL-2+ and BSL-2 room additions and the proper placement of biological safety cabinets and other equipment within the rooms.
February also marked the beginning of research projects at Arizona State University involving positive SARS-CoV-2 samples handled at BSL-2 containment with Biosafety Level 3 (BSL-3) practices and procedures (BSL-2+). 10 For SARS-CoV-2 laboratories working at BSL-2+, enhanced PPE was required as previously mentioned. All personnel working in these laboratories were informed of the risk and disease symptoms of the agent, enrolled in the occupational health program, required to self-monitor for symptoms, and trained in the procedures to take in the event of a possible exposure.
Laboratory access was removed for all personnel, except those individuals working on approved projects. In addition, signage was posted at the entrance to these laboratories stating, “Do not enter. Authorized personnel only.” The signage also indicated the type of samples being processed, required PPE to enter, emergency contact information and a biohazard symbol.
The biosafety team reviewed Institutional Biosafety Committee disclosures related to these projects, determined required PPE, established the occupational health requirements in consultation with university medical providers and current CDC and WHO guidance, and developed medical surveillance protocols, post-exposure protocols, and additional safety measures for each project.
The biosafety team was tasked with supporting the conversion of a select agent registered BSL-3 containment laboratory into a SARS-CoV-2 research laboratory. The biosafety team, select agent principal investigator, and high-containment facility manager conducted a call with Arizona State University's select agent program file manager to obtain guidance on how to successfully transfer the select agent laboratory to a different location in the high-containment facility.
After the select agent lab had been moved, the biosafety team reviewed and provided input on the plans to remodel this laboratory for SARS-CoV-2 work based on the 6th edition of the CDC Biosafety in Microbiological and Biomedical Laboratories 11 manual and the National Institutes of Health (NIH) Design Requirements Manual. 12 Once the construction was completed, the biosafety team actively participated in a commissioning process for this space in accordance with the NIH BSL-3 Laboratory Certification Requirements, 13 since this space was renovated outside of the normal recommissioning cycle for the BSL-3 facilities.
The biosafety team also assisted with the development of the procedures for the new SARS-CoV-2 laboratory, including requirements for personnel to be enrolled in the occupational health program. The team also updated the BSL-3 facility procedures and provided SARS-CoV-2 training to all personnel who worked in the BSL-3 suite where this new laboratory was housed.
On March 2, the EHS team assisted in the development of a remote work operations plan for essential and non-essential staff. The plan identified which staff would be considered essential personnel based on job description, how and at what frequency Arizona State University would communicate and update staff throughout the pandemic, as well as technology solutions for those working remotely. Individuals deemed essential included those with specific job functions in support of critical infrastructure, such as health care, fire, police, construction, maintenance, energy, and communications. The majority of EHS and biosafety personnel were deemed to be essential and were required to remain on campus.
On March 3, a second positive case occurred in Maricopa County, where the main campus for Arizona State University is located. 14 New travel and self-isolation guidelines were released, and senior leadership met to discuss business interruption and insurance concerns in the event of a ramp-down of services. The biosafety team introduced enhanced cleaning and disinfection protocols for campus shuttles and began COVID-19 site safety assessments for onsite employees.
Supplies were centralized through the university's purchasing division to foster rapid and timely access to critical supplies that had not been easily accounted for earlier, such as disposable gloves in teaching laboratories, disinfectants in janitorial closets, and respirators in other warehouse locations. On March 6, university travel restrictions were implemented, with almost all air travel prohibited.
On March 11, the WHO announced that COVID-19 was officially a pandemic: “There are now more than 118,000 cases in 114 countries, and 4,291 people have lost their lives.” 15 The Governor for the State of Arizona declared a state of emergency, Arizona State University made the decision to transition in-person classes to online education, and university events were canceled. Shortly thereafter, the governor ordered public schools to be closed. 16
The EHS and the biosafety team quickly implemented ramp-down procedures for laboratories, developed procedures for self-screening health checks for on-campus personnel, and installed new signage for campus shuttles (Figure 1).

Arizona State University's campus shuttle signage. Color images are available online.
In light of the university slowdown, EHS developed ramp-down and continuity of operations procedures for research laboratories. Examples of ramp-down procedures included: updating labels on all temperature-controlled units with emergency contact information, posting signage on restricted areas, limiting the number of onsite personnel, communicating responsibilities clearly within the research teams, postponing animal orders and delaying animal studies, and recalling all field researchers.
EHS provided recommendations for increased security of experiments, research spaces and supplies, and encouraged the locking of cabinets and freezers. Ramp-down guidance included taking steps to ensure reagents were placed into long-term storage or disposed of properly (e.g., requesting a pick-up for all waste to prevent an accident), leaving the laboratory spaces clean and preparing for a time when ramp-up of research was permitted. Researchers, technicians, and animal care staff (especially those providing the ongoing care of the vertebrate colonies on campus) performing limited on-campus activities during the ramp-down were encouraged to work in pairs or follow a buddy system for personal safety.
The first COVID-related death in Arizona occurred on March 17, 2020, 17 and the Governor issued stay-at-home orders for the public on March 30. 18
In response to the PPE shortage that was being experienced nationwide, engineering students and staff at Arizona State University began producing single-use face shields for health care and testing operations on March 27, 2020. The biosafety team worked with the engineers to validate the design, source products for the generation of the face shields and develop procedures to ensure quality and proper disinfection post-production. At the height of the need for disposable face shields, there was also a significant global supply chain deficit and retail impact in which many supplies needed for the face shield constructions were not available. As a solution, the clear face shield portion was made from empty lamination sheets when clear plastic was not available, forehead cushion from window foam, and the headband was made from Coban—a water-vapor permeable, non-woven polyester fabric—when elastic was not available (Figure 2).

Disposable face shields produced for health care and COVID-19 testing operations. Color images are available online.
As the disruptions in the global supply chain increased, the Arizona State University purchasing department was challenged with finding reputable sources for disinfectant, cleaning supplies, PPE, and other in-demand items. Purchasing staff would locate a particular product in the marketplace and would be told by the vendor that they had less than 5 min to make a decision. After obtaining a few inferior and counterfeit items, such as KN95 respirators with large holes in the breathing zone, single-ply face covers, and disinfectant not approved for coronaviruses, EHS and the biosafety team was required to make the final decision to purchase the item or not, always within a very short period of time. Even with these time crunches during the early months of the pandemic, the university acquired more than:
800,000 pairs of nitrile gloves. 300,000 face masks. 10,000 gallons of alcohol-based hand sanitizer. 56 tons of Community of Care kits. 1 million pipette tips, used in laboratories for COVID-19 testing and research. 3,000 laptops to distribute to faculty and students for the shift to remote learning.
April Through June 2020: From Ramp-Down to Ramp-Up
On April 6, EHS and the biosafety team began the live delivery of “COVID-19 Safety Training for On-Site Personnel,” which included virtual presentations for onsite staff about SARS-CoV-2 based on the latest guidance from the CDC, WHO, State of Arizona Department of Health Services and Maricopa County Department of Public Health.19,20
The purpose of the training was fourfold: (1) Discuss personal wellness, conduct self-health assessments, and report COVID-19 exposure; (2) Provide information on the institution's efforts to prepare the campus for return; (3) Provide steps that could be taken by individuals to stay safe, including social distancing and wearing face covers; and (4) Provide information on personal hygiene, cleaning, and disinfection. The training covered specific information about the virus, general infection prevention and control, hygiene and live demonstration of donning and doffing of safety glasses, surgical masks, and gloves. The majority of the onsite staff represented professions that had no prior experience with wearing or using PPE.
On April 7, the CDC issued guidance that everyone should wear a cloth face cover in public settings, in addition to practicing social distancing measures, especially when performing essential activities such as going to grocery stores and pharmacies. The biosafety team consistently monitored CDC guidance, which changed frequently. As guidance evolved, the training was revised, often several times a day. The training became a trusted resource for the university community to get the latest guidance in one online location. More than 3,000 employees took the training within the first 2 weeks; more than 25,000 took it in the first 6 months.
Although there were rapid changes to guidance at the local, national, and global level, Arizona State University continued to post updates on guidance and commonly asked questions on a central COVID-19 information webpage. Throughout the course of the pandemic, employees, students, contractors, and visitors were directed to this resource to stay informed of changes. This website was critical in effectively communicating with individuals onsite as well as those working remotely.
On April 30, university leadership decided to resume in-person classes for the forthcoming fall semester, even though many other colleges and universities elected to remain virtual. 21 On May 15, the institution announced a phased campus return. During the summer of 2020, the EHS and biosafety team was tasked with assisting in the development of the health and safety requirements for the return to campus.
Throughout the month of May 2020, a cross-functional team wrote a “COVID-19 Safety Plan for Fall 2020 On-Campus Instruction” that consisted of seven elements: facility spacing and set-up guidelines, disinfection and cleaning protocols, health and wellbeing standards, PPE and face coverings, building access restrictions, materials and supplies, and communication protocols.
The plan provided recommendations for common areas (including lunchrooms, break rooms, and outdoor furniture), traffic control spaces (including hallways, entryways, stairwells, and elevators), and learning locations (including classrooms, study rooms, and computer laboratories).
Cleaning protocols were developed to ensure that the university had universal cleanliness standards applied consistently across all campuses and buildings. Social distancing requirements and health standards were based on current CDC guidelines. Procurement and distribution of PPE was centralized to ensure the most urgent operational needs (such as animal care, dining, facilities maintenance, health care centers, housing, police, clinical and research SARS-CoV-2 laboratories, and COVID-19 testing sites) were met first and that no individual unit was left without the appropriate safety material to meet its mission.
Building access restrictions were created to increase both awareness and control of who was entering the campus facilities and when. Employees, including EHS and biosafety personnel, were issued unique university identification badges that were required to access campus facilities during the reduced operations. Buildings that were placed into hibernation (e.g., due to no one needing to enter them) on campus had exterior locks changed to prevent the building from being left unsecure.
Communication protocols were implemented to guarantee that there was only one message being conveyed across all media channels informing the university community of what actions were being taken to ensure their safety and well-being. The biosafety team also worked with the university's communication team to develop return-to-campus graphics (Figure 3) and vinyl floor stickers with directional arrows and distancing markers to communicate how to stay safe during the ongoing outbreak.
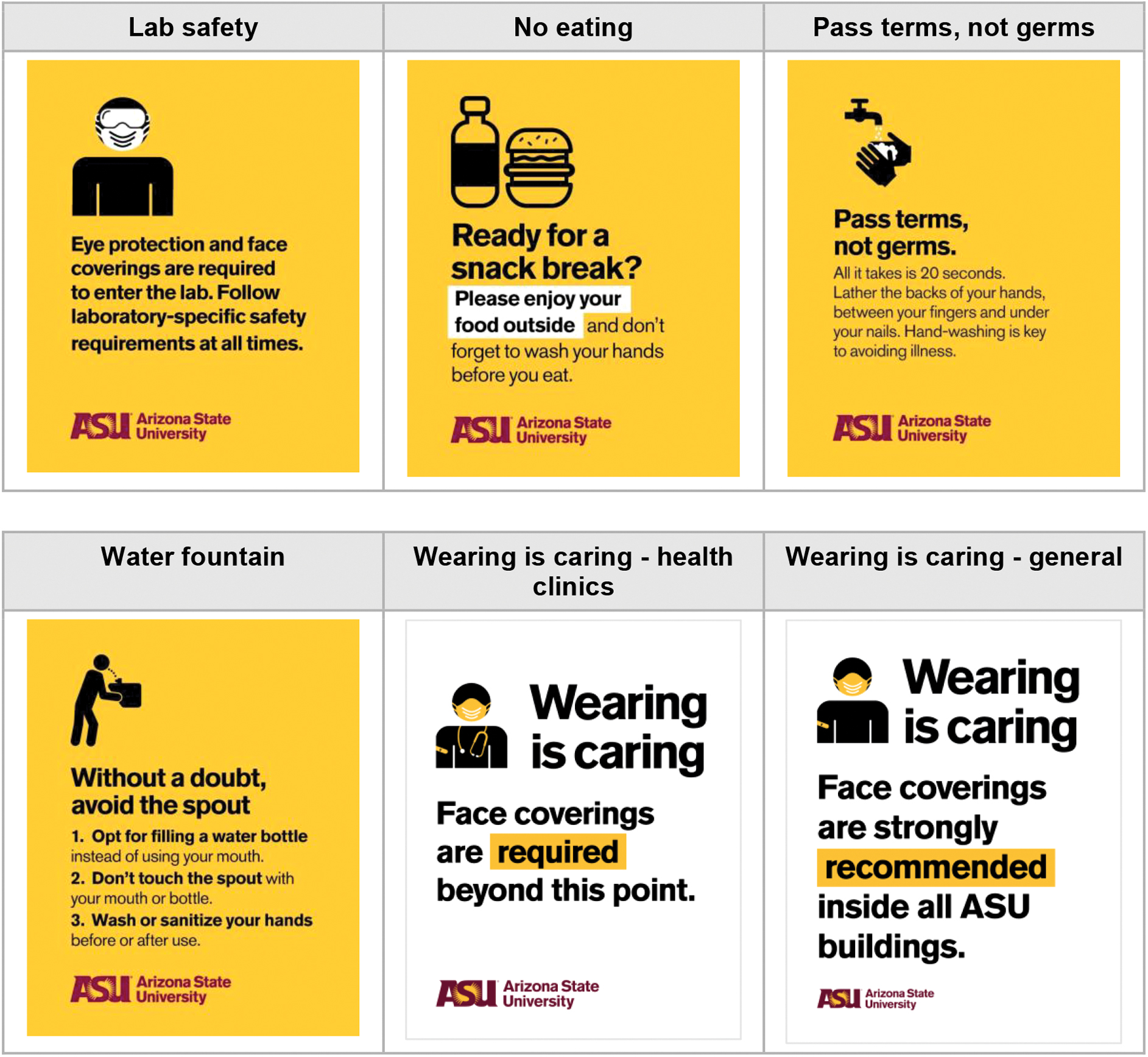
Examples of Arizona State University's Fall 2020 return-to-campus graphics. Color images are available online.
To prepare Arizona State University for return-to-campus in the fall of 2020, “Community of Care” kits (Figure 4) were provided free of charge to all students and employees. The kits contained two face coverings, alcohol-based hand sanitizer, disinfectant wipes, and a personal thermometer. These kits were available at campus stores for pick-up. Kits were also distributed to employees by interoffice mail, and students living in the housing facilities received the kits during move-in. Community of Care kits were also distributed at the beginning of the 2021 spring semester.

Arizona State University's Community of care kits. Color images are available online.
In August 2020, the biosafety team provided input into a new university-wide, mandatory, daily health check for students and employees to complete before coming to the university. The health check was a short questionnaire that asked individuals whether they had COVID-19 symptoms. The specific questions were:
Are you performing any work on behalf of Arizona State University today? Yes or no.
Will you be onsite at a university location or interact with anyone from the university community in person while you are working today? Yes or no.
Are you currently under isolation or quarantine restrictions? Yes or no.
Are you experiencing new or worsening of any of the following? Answer choices included fever, cough, shortness of breath, or none.
Are you experiencing new or worsening of any of the following? Choices included chills, diarrhea, vomiting, loss of smell or taste, sore throat, muscle pain, or none.
After all responses were submitted, the user was provided with the following message: “If you know you were exposed to COVID-19 by someone who tested positive in the last 14 days, you should not come to work. Please notify your supervisor and contact your healthcare provider to help determine if testing is right for you.” 22 Noncompliance could result in a loss of access to university computer systems until the health check was completed. There were three ways to complete the daily health check: a mobile app, a university web page, or by calling a dedicated phone number.
Plans to successfully transition the previous select agent BSL-3 laboratory to a SARS-CoV-2 laboratory and move the select agent laboratory to another space in the facility were finalized and approved by the entity's select agent file manager. Between May and August 2020, both laboratories were renovated and commissioned for BSL-3 research. The CDC conducted a virtual select agent inspection of the new laboratory in September 2020 to ensure compliance with Federal regulations, and the space was approved for select agent activities.
Due to the large number of SARS-CoV-2 projects and the limited number of personnel with BSL-3 experience, additional researchers were identified to receive training to work in BSL-3 spaces. The biosafety team spearheaded the creation of a new BSL-3 Requirements and Practices Policy 23 that outlined the requirements and steps to complete for personnel to be approved to work at BSL-3. This policy includes the minimum requirements to participate in the BSL-3 training certification program and detailed step-by-step training plans for individuals with varying levels of experience to complete. The policy was approved by ASU's Institutional Biosafety Committee. The biosafety team also assisted with training new personnel to work in the SARS-CoV-2 laboratories.
Core Biosafety Support of Research
The biosafety program at Arizona State University is responsible for conducting biosafety research reviews to ensure compliance with Federal, state, and local regulations, institutional policies, funding requirements, and best management practices. Throughout the COVID-19 pandemic, the biosafety team continued to be responsible for conducting biosafety inspections for the institution:
ABSL-3, BSL-3, and Select Agent laboratories inspected every 6 months.
BSL-2 laboratories inspected every 12 months.
BSL-1 laboratories inspected every 18 months.
All new laboratories working with biological material are inspected before approval from the Institutional Biosafety Committee.
All laboratories undergo a lab-close-out inspection process when someone leaves or moves locations.
During the pandemic, there was an overall increase in the amount of research with SARS-CoV-2, which correlated to an increase in the number of ABSL-3 and BSL-3 users. The ABSL-3 facility and BSL-3 laboratory witnessed a quadrupling of research staff in the space, and the number of principal investigators increased from 2 to 9.
The biosafety team reviewed new Institutional Biosafety Committee (IBC) disclosures and Institutional Animal Care and Use Committee protocols for SARS-CoV-2 research, determined PPE and occupational health requirements, developed inactivation protocols for SARS-CoV-2 samples—including the steps required to bring inactivated materials from the BSL-3 to BSL-2 laboratories, wrote procedures for the transport of materials between ABSL-3 and BSL-3, created SARS-CoV-2 agent-specific training, registered to receive SARS-CoV-2 permitted materials, reviewed material transfer agreements to receive and send materials, and developed procedures for shipping, receiving, and unpacking of SARS-CoV-2-containing materials. The IBC also increased the frequency of their meetings to accommodate the escalation of COVID-19 research.
Beyond high-containment research, the biosafety team reviewed research protocols for SARS-CoV-2 detection in wastewater. The biosafety team obtained a CDC import permit for wastewater samples containing SARS-CoV-2, had a virtual inspection of laboratory spaces with the CDC Import Permit Program, developed procedures for loading and unloading of safety cups within the biological safety cabinet, developed a training specific to SARS-CoV-2 for staff and visitors, and developed signage to be posted in laboratories where wastewater samples were used or stored.
SARS-CoV-2 Testing Site Safety Procedures
In April 2020, researchers at Arizona State University's Biodesign Institute determined that a less-invasive, rapid SARS-CoV-2 test was needed to replace nasopharyngeal swabbing. The research team observed that the testing options were not adequate or sufficient to test essential personnel, let alone anyone with symptoms who might be concerned that they had COVID-19. There was also a realization that the institution needed to rapidly and correctly identify all contagious individuals to implement quarantine measures. Because the SARS-CoV-2 virus is most infectious pre-symptomatically, focusing on individuals with symptoms was considered too late.
In May 2020, the institution developed a clinical saliva test to quickly test for SARS-CoV-2. The test used TaqPath COVID-19 combination kit reagents, manufactured by ThermoFisher Scientific and approved through a Food and Drug Administration Emergency Use Authorization, to test for SARS-CoV-2 in saliva specimens. 24 The saliva test assessed three unique SARS-CoV-2 RNA fragments to improve accuracy. The test allowed individuals on campus the ability to perform a diagnostic assay without the need for a clinician, thereby decreasing barriers to access and increasing the clinical testing capacity at the institution.
The biosafety team worked closely with a newly formed Employee Health unit to review locations and develop safety procedures for employee and student saliva-based COVID-19 testing and vaccination sites. The required PPE for sample collection included N95 respirators, disposable isolation gowns, face shields, and double gloves. The biosafety team performed risk assessments and developed the PPE requirements for the in-person COVID-19 saliva testing sites.
The EHS and the biosafety team conducted more than 1,000 N95 respirator fit tests for personnel working at COVID-19 saliva testing sites. In addition to N95 respirator fit testing, the biosafety team conducted in-person training and audits of PPE donning and doffing procedures over the course of the 18 months when the in-person COVID-19 saliva testing sites were operational.
To ensure the safety of staff and participants at the COVID-19 testing sites, the biosafety team worked in collaboration with an aerosol scientist at Arizona State University, to estimate the distance saliva traveled when the sample vials were dropped, and the resulting particle sizes that were created. For in-person testing sites, the distance saliva would travel when dropped from a tube was important to determine the size of the area for a biological spill cleanup. This was critical, because the size of the testing sites varied.
In some cases, a spill would require the full testing site to close during the biological spill cleanup. For larger testing sites, only an area or wing needed to be closed during the spill cleanup and the rest of the site could remain in operation. During the height of the pandemic, thousands of samples were processed every day, so it was therefore crucial for testing sites to remain open.
To estimate the distance the saliva traveled, the biosafety team used red-colored liquid, made by adding red food coloring to commercially available semi-viscous, multi-surface disinfectant, filled to the maximum level for the saliva samples, and dropped them from different heights, angles, and locations within an individual testing station to evaluate the maximum area impacted by a dropped sample tube. One limitation to the drop test was the lack of access to a saliva simulant. The results indicated that splashes could reach up to a 10 ft radius from the location of the spill when dropped. This distance was used at the in-person testing sites for cleanup after a spill. Individuals providing a sample at the COVID-19 testing sites were 6 ft apart and separated by solid dividers. However, to ensure the safety of all staff and individuals at the test site, it was determined that a 10-ft distance from the spill would require disinfection.
To validate the distance the saliva traveled, the aerosol scientists performed independent tests by dropping artificial saliva-filled sampling tubes in a confined space and measuring size-resolved particle number concentrations in the air above the fall point. In the particle size range tested from 20 nm to 20 μm by using a Scanning Mobility Particle Sizer and an Aerodynamic Particle Sizer (0.5–20 μm), no significant increase in particle concentrations relative from background were detected. Although these tests cannot totally exclude any small aerosol formation, if this happens, it is minor. More likely is the generation of larger (>20 μm) droplets that would settle more readily by gravity.
The experiments showed that any small particles generated would likely be diluted rapidly in the circulation of air in the room. Based on these experiments, the team concluded that the dropping of vials was not a significant source of small aerosol particles and, therefore, not a significant hazard.
SARS-CoV-2 Testing Site Designs
Indoor
The EHS and biosafety teams were tasked with designing the in-person testing sites for the institution. The sites were designed to share key traits, including a single entrance and exit with a traffic pattern to prevent individuals from crossing paths, while maintaining a physical separation of 6 ft at all times. All testing sites had three primary attributes: (1) an entry point where individuals received instructions for the saliva collection and supplies (noted as 1 in Figure 5); (2) a station for sample collection that was separated from other individuals by at least 6 ft (noted as 2 in Figure 5); and (3) opaque barriers to provide privacy and a station for sample collection (noted as 3 in Figure 5).
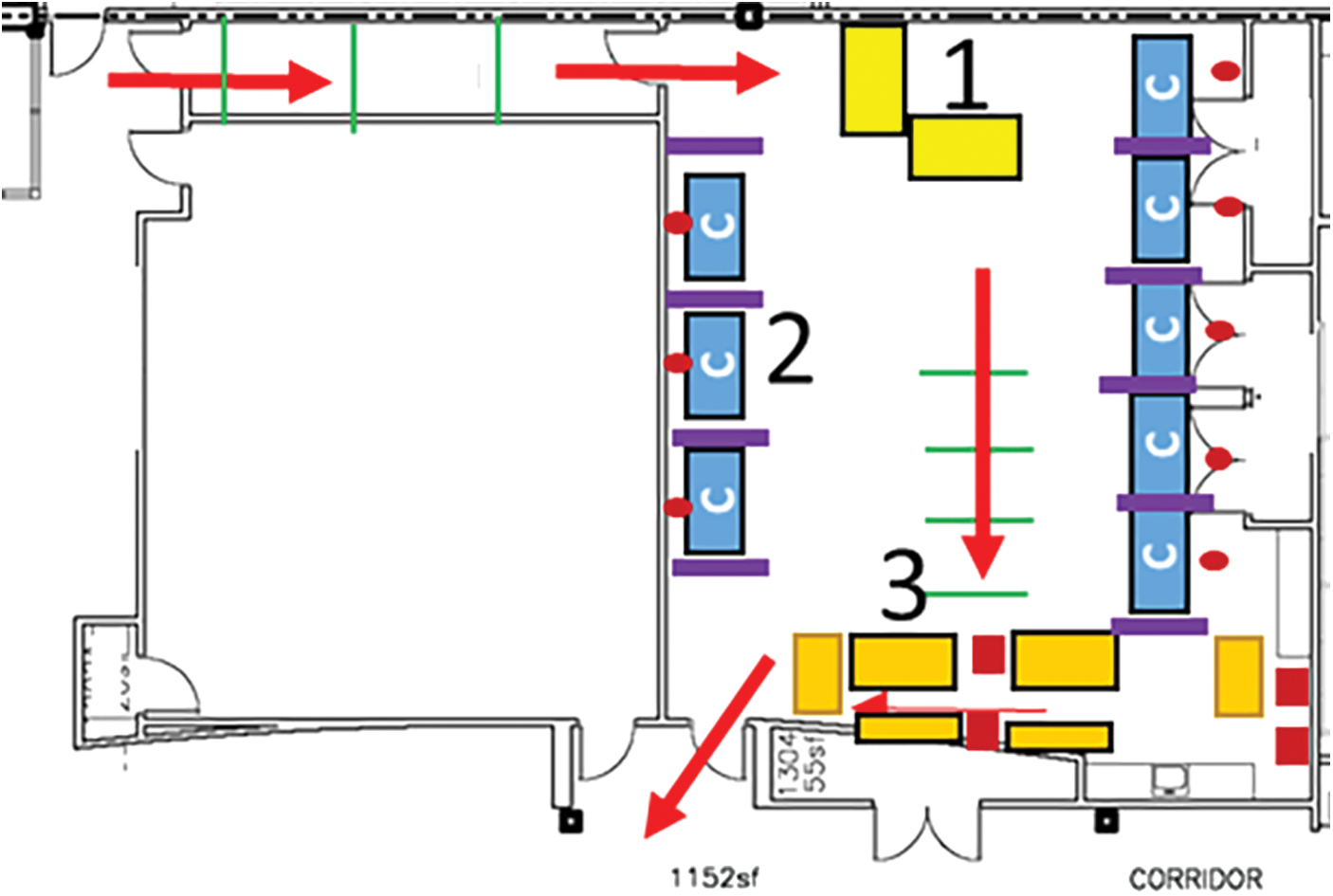
Diagram of the first in-person COVID-19 testing location. Color images are available online.
The biosafety team was tasked with implementing an onsite SARS-CoV-2 testing location for employees on June 8, 2020. Ten days later, the first saliva samples from employees were collected by using the ASU Biodesign Institute's homegrown qPCR saliva test. The samples were transported to the clinical testing laboratory for analysis. The first indoor site (Figure 6) collected a maximum of 24 samples per hour and was open to onsite personnel that did not have symptoms of SARS-CoV-2 infection.
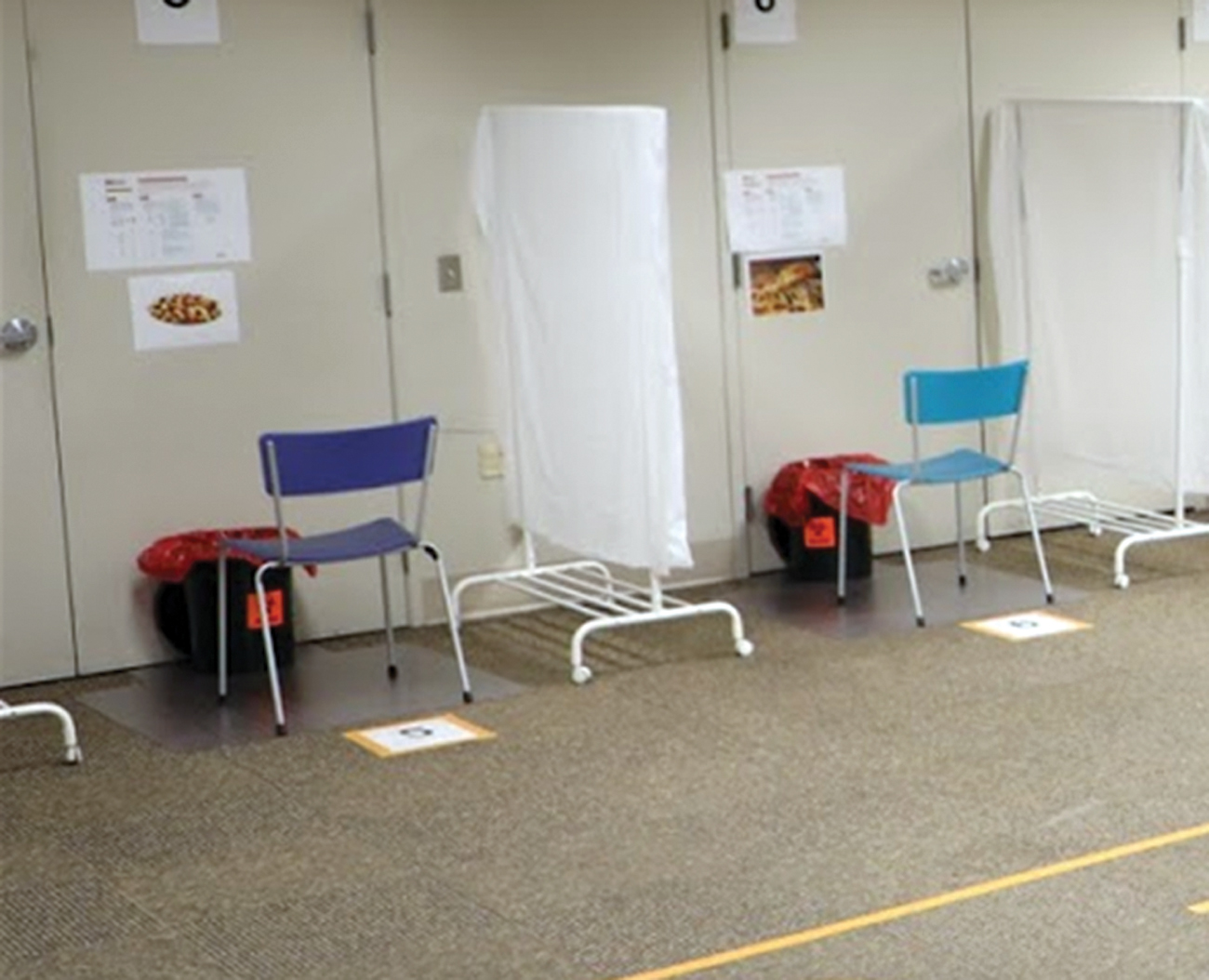
Photograph of the first in-person COVID-19 testing site at Arizona State University. Color images are available online.
The number of samples per hour was calculated by the number of stations used for sample collection multiplied by four samples per hour, which assumed that each sample collection would take 15 min (6 stations × 4 collections per hour). This assumption was later modified based on additional data, which determined that each sample collection took ∼10 min, which allowed for six collections per hour for each station.
The construction of the COVID-19 saliva testing sites had unique challenges, including carpet flooring, an absence of structures to divide the space, and a lack of appropriate heating, ventilation, and air conditioning (HVAC) systems. The team devised a plan to find commercially available solutions: clothing racks sourced locally, covered with opaque plastic sheeting that could be disinfected easily, and disinfectant that was available and effective against SARS-CoV-2. Also lacking were biohazard waste containers in large numbers.
Carpeted floors were covered with commercial grade painters' plastic sheeting and plastic desk chair mats. Five-gallon buckets with lids lined with biohazard waste bags were used for biohazard waste containers for each sample collection station. At one of the larger sites with limited air flow, a large trailer with a mobile HVAC unit was rented, and plenum ducts were installed to increase air circulation, lower temperatures, and improve overall safety.
Drive-up
In an effort to provide testing for individuals with symptoms of COVID-19, an on-campus drive-through testing site was opened on June 25, 2020. The drive-through site had a sample collection capacity of 120 samples per hour from individuals in vehicles. The process for sample collection was similar to the first in-person site, in that individuals were provided with supplies for the saliva sample collection and then directed to a location to provide a sample in their vehicles in a parking lot. The samples were then driven to a collection location, staffed by trained personnel.
Capacity at the drive-through location was greatly increased, as each individual was enclosed in a vehicle and the parking lot in use was large. Every minute, two vehicles were provided the sample collection supplies, allowing for 120 samples per hour to be processed. Personnel were required to have biosafety training, bloodborne pathogens training, health care protections training, and cybersecurity training. The drive-through location presented additional hazards from that of the indoor, in-person COVID-19 testing sites, from multiple perspectives, with the greatest hazard for site staff in PPE being high temperatures. The temperature reached 108°F on the first day of the drive-through site. Subsequent cooling options and training on heat stress and heat stroke were provided to personnel at the site.
Simultaneously, additional indoor testing site locations were established at all satellite campus locations, with sample collection capacities from 48 to 72 samples per hour (8–12 sample collection stations with 6 samples per station collected per hour), with five COVID-19 testing sites operational by June 29, 2020. There was a 50-mile distance across the satellite locations, with the main campus location serving as the central supply location.
The return of students to campus for the Fall 2020 semester necessitated the expansion of the indoor COVID-19 testing location on the main campus to 300 samples per hour (50 sample collection stations, 6 samples per station collected per hour; 12 stations were held in reserve for sample repeats and flow of individuals). The only locations available on campus large enough to accommodate up to 2,400 individuals per day were large, multiple court gymnasiums (Figures 7 and 8). Less than 1 month after the first in-person testing at Arizona State University, a large, 300-sample per hour site was opened and remained open and functioning for a year as the primary location for the institution's COVID-19 testing.
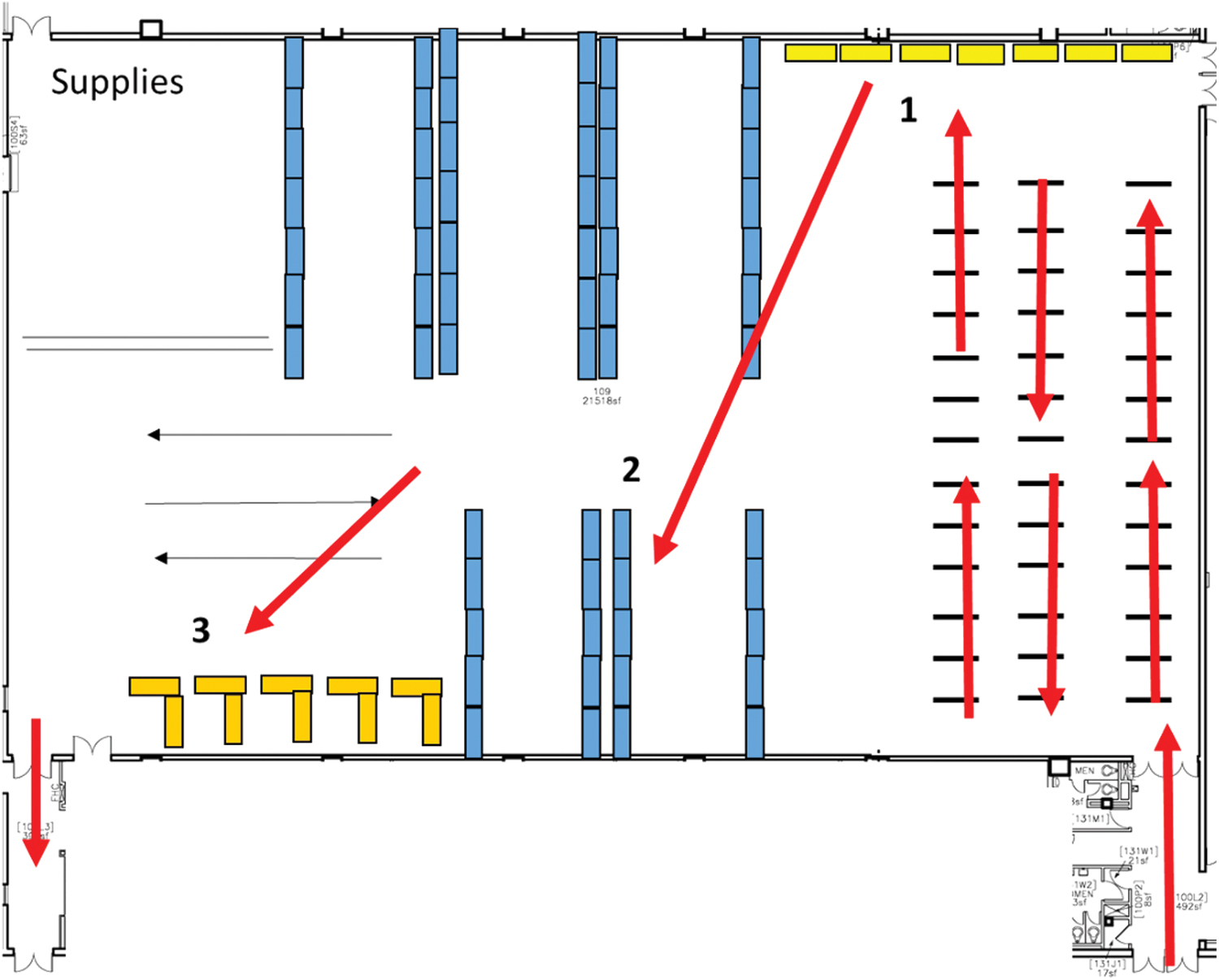
Diagram of the first high-capacity, in-person COVID-19 testing location (Note: The space was 21,518 sq ft). Color images are available online.

Photograph of the first high-capacity, indoor COVID-19 testing location at Arizona State University. Color images are available online.
Pop-up sites
In an effort to promote surveillance COVID-19 testing for students in campus housing, pop-up saliva collection sites were set-up at housing locations where students were living so they could drop off a completed saliva sample for testing. These sites were set-up and disassembled quickly, typically within 48 h of use, with the same characteristics of the in-person testing, except that the samples were not collected onsite. There were no appointments issued for these locations, and the number of students providing samples was not predictable.
On several occasions, the number of students providing samples overwhelmed the pop-up sites and students were directed to the larger, in-person testing locations to collect the samples. These sites were discontinued after 2 weeks due to the logistical challenges and irregular staffing times required to manage them.
Self-administered
On January 21, 2021, Arizona State University began to transition from in-person COVID-19 sample collection sites to an unsupervised, self-administered saliva sample collection model for all campus locations. The transition was completed on June 11, 2021 with the closure of all five in-person COVID-19 testing locations to 35 unsupervised collection sites.
These sites are completely unsupervised, the sample tubes with unique barcodes are registered by the individual in the online COVID-19 testing portal and then dropped off at locations housing locked sample collection bins. These bins are secured to the location by either a bike chain lock or a secure room, and saliva samples are placed in sealed biohazard specimen bags before being placed into the collection bin. Collection bins are picked-up daily and transported to a central location where samples are prepared for transport to the clinical testing laboratory. This transition decreased the daily staffing requirements by 83% as compared with in-person COVID-19 testing sites.
The first 12 months of COVID-19 testing efforts at Arizona State University culminated in the processing of more than 304,000 saliva samples.
Creating and Operating COVID-19 Vaccination Sites
In January 2021, the State of Arizona announced the ability to vaccinate individuals older than age 65, health care workers, law enforcement officers, and higher education employees prioritized for COVID-19 vaccines. 25 In response to this order and to aid in the vaccination of employees, Arizona State University received Moderna COVID-19 vaccines to vaccinate employees who qualified for vaccination. 26 The transition from COVID-19 testing to COVID-19 mass vaccination sites was modeled after experience gained from operating the large COVID-19 saliva testing sites and observations at mass vaccination sites in the state.
On January 17, 2021, the first COVID-19 vaccine point of distribution (POD) for Arizona State University began. Again, the mass sites were set-up in multiple court gymnasium spaces, ensuring unidirectional traffic flow patterns, utilizing the majority of the space for post-vaccination monitoring while maintaining physical distance. There were no privacy panels between individuals to allow for the monitoring of large numbers of post-vaccinated individuals simultaneously by onsite Emergency Medical Technician (EMT) personnel.
The site was designed and staffed to support the vaccination of 1,200 individuals per day (5 individuals vaccinated every 2 min for an 8-h day) (Figure 9). As with the COVID-19 testing sites, the vaccination site layout, patient flow, and infection control protocols were evaluated and approved by the biosafety team. All personnel performing vaccinations were required to be enrolled in the university bloodborne pathogen program and take training (verified by the biosafety team).
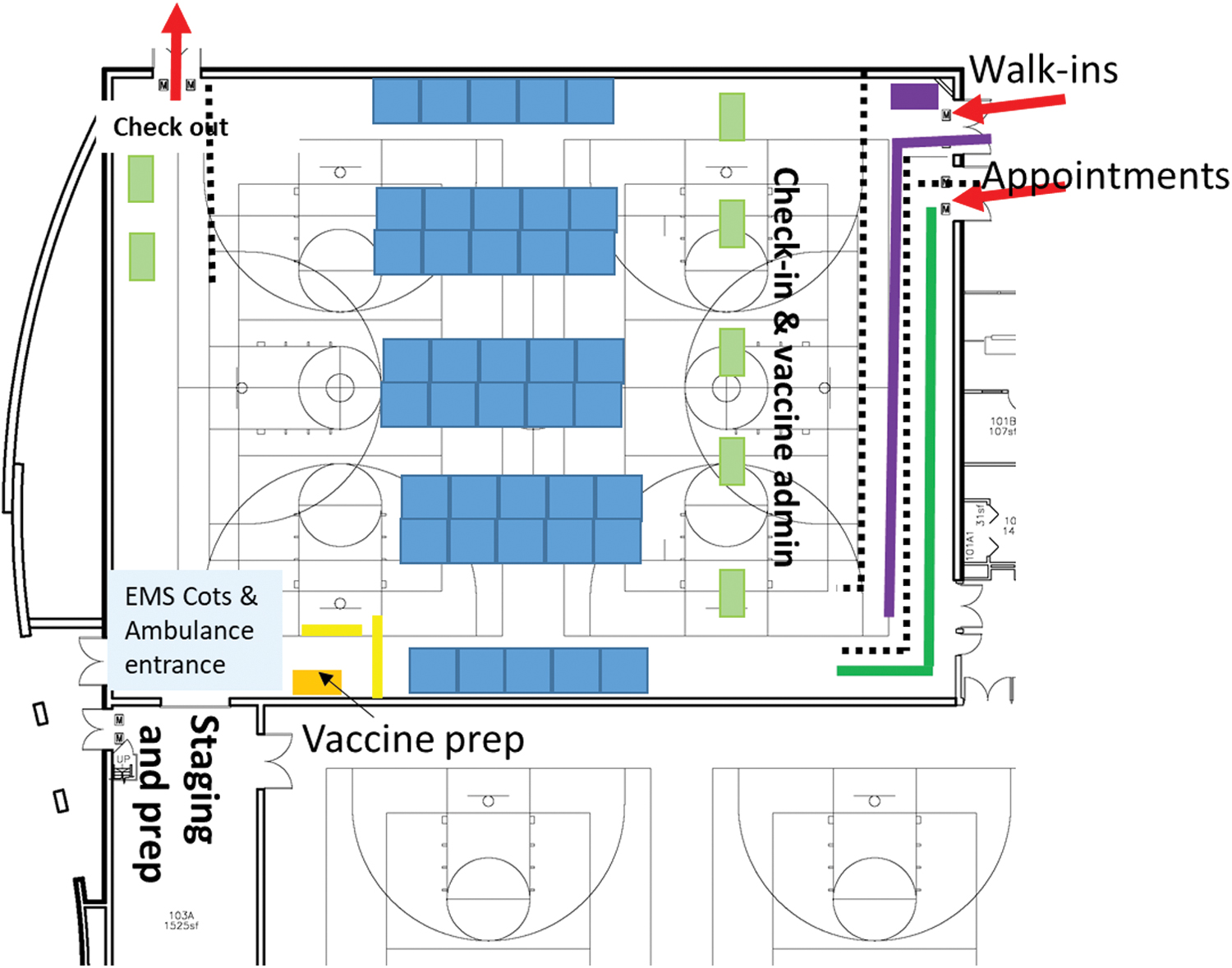
Diagram of the first high-capacity, indoor COVID-19 vaccination site at Arizona State University (Note: The space was 10,983 sq ft). Color images are available online.
All vaccination site-related waste, including disinfection materials, sharps, and medical waste, were managed by EHS. At the height of the mass vaccine sites, up to 258 gallons (43-gallon containers × 6 containers) per day of biohazardous waste were generated with a total of nearly 5,000 gallons generated over the course of the mass vaccination site operations. In addition, more than 170,000 gallons of biohazardous waste was safely managed during the pandemic for all aspects of COVID-19 response, including testing, vaccination, research and healthcare.
The most significant limitations of the POD were the availability of medical professionals available to administer vaccines, COVID-19 vaccine availability, the space required for post-vaccination monitoring, and the unpredictable number of individuals who required a 30-min monitoring period versus a 15-min monitoring period. The Arizona State University Employee Health department leveraged an emergency order for EMTs to perform vaccination, with training from the National Guard, to allow 20 EMTs to be able to administer vaccines. Arizona State University Employee Health also partnered with the institution's nursing school to help staff registered nurses and students to administer vaccinations. The mass vaccination sites operated until May 7, 2021, with a total of 15,528 COVID-19 vaccine doses administered to employees.
Conclusion
A major benefit at Arizona State University was the development and roll-out of an accurate SARS-CoV-2 saliva-based test. This allowed the institution to rapidly identify COVID-19 positive individuals to help prevent the spread of outbreaks and allow business to continue. The transition to an unsupervised, self-administered saliva test method streamlined operations and efficiencies and decreased overall costs associated with in-person testing.
Although there were many successes, there were several challenges, including: having outdoor testing sites during extreme weather conditions (e.g., extreme hot, extreme cold, microbursts), not having timely and accurate weather reports, having outdoor collection sites without access to shade and cooling, having “pop-up” testing sites at dormitories, and having more than one team managing COVID-19 test sites. Another challenge was not having adequate numbers of vehicles or trained personnel to meet the U.S. Department of Transportation UN 3373 shipping and transporting requirements.
To prepare for future pandemics, epidemics, and outbreaks, it is crucial that entities have a stockpile of supplies to enable their institutions to continue to operate, even in a limited capacity, when facing public health crises. A recommended list of supplies for large, research, and teaching institutions is included in Tables 1 and 2. Having these emergency and medical supplies available will allow institutions to keep essential personnel onsite to support critical infrastructure and research projects (if applicable) and help prepare the institution for a full return to operations.
To successfully respond to the COVID-19 pandemic, Arizona State University's leadership, faculty, staff, students, and volunteers effectively mobilized to keep the on-campus and off-campus community safe. An enormous advantage in the pandemic response was the willingness of employees, students, and volunteers to step-up and assist during the pandemic. Thousands of individuals performed different roles at the COVID-19 saliva testing and vaccination sites for months on end. There were also many people who helped deliver food to students in isolation and quarantine in housing facilities.
Every institution should have at least one individual who continuously prepares for emergency situations and develops continuity of operations plans for the organization. Larger institutions should have a committee of experts from across the institution meeting on a regular basis to discuss pandemic preparedness. The committee should have input from EHS, biosafety, dining, facilities, finance, health care, housing, legal, media, research, technology, transportation, and other key stakeholders.
The experiences at Arizona State University were successful because of established internal communication networks, as well as strong relationships with community partners, including the state and county public health agencies. For example, there were 28 internal departments that collaborated on COVID-19 saliva-based testing at the institution. Because of all the efforts outlined in this article, the institution's employee COVID-19 positivity rate remained less than 1% for most of the pandemic.
Although COVID-19 remains a challenge, institutions should continue those activities that helped to create healthier working, learning, and living environments. Solutions, such as a voluntary daily health check, can serve as a mechanism to remind people to think about their health as well as the health of others before engaging in daily activities. Technological solutions that detect outbreaks and help to avoid exposing those who are healthy to those who are sick will help slow or stop the spread of infectious diseases. Technology may also be helpful to determine when additional cleaning or air filtration measures may be required, or when contact tracing should be implemented to alert an individual to a particular health care issue.
The COVID-19 pandemic not only created many challenges for biosafety professionals but also presented many opportunities for them to learn, collaborate, and, most importantly, to bring attention to the biosafety field. The pandemic continues to highlight the need for more biosafety professionals to help prepare organizations and train others to successfully face future public health crises. Throughout the pandemic, there was a shortage of PPE, disinfectants, and supplies, but the most extreme shortage was a deficit of qualified, trained staff. This included health care providers (especially nurses), biosafety personnel, preparedness coordinators, supply chain experts, and many others who helped manage the disparate aspects of the pandemic.
Footnotes
Acknowledgments
The authors would like to thank everyone who stepped-up to provide support at Arizona State University and the surrounding communities during the COVID-19 pandemic response.
Authors' Contributions
D.G.: Conceptualization, methodology, writing—original draft, writing—review and editing, visualization, supervision, project administration. A.R.: Writing—original draft, writing—review and editing. I.M.: Writing—original draft, writing—review and editing.
Disclaimer
ASU and the Arizona State University “sunburst logo” are registered trademarks and service marks of the Arizona Board of Regents, for and on behalf of Arizona State University and may not be used or reproduced without permission from Arizona State University. This work is licensed under a Creative Commons Attribution 4.0 International License. Created by Arizona State University, 2022.
Authors' Disclosure Statement
D.G. is employed by Arizona State University and is the Past-President of ABSA International and an Associate Editor of Applied Biosafety. A.R. is employed by Arizona State University. I.M. is employed by Arizona State University.
Funding Information
This work was supported by Arizona State University.
