Abstract
Background:
Burkholderia pseudomallei is a Tier 1 overlap select agent and subject to the select agent regulations (42 CFR §73 and 9 CFR §121). It is a gram-negative, motile, soil saprophyte, and the etiologic agent of melioidosis. B. pseudomallei infection can produce systemic illness and can be fatal in the absence of appropriate treatment. Laboratory exposures involving this organism may occur when appropriate containment measures are not employed. Current disease treatment inadequacies and the risk factors associated with melioidosis make this an agent of primary concern in research, commercial, and clinical laboratory environments.
Results:
This study presents data reported to Centers for Disease Control and Prevention (CDC), Division of Select Agents and Toxins for releases involving B. pseudomallei in 2017–2019 that occurred in Biosafety Level (BSL)-2 and BSL-3 laboratories. Fifty-one Animal and Plant Health Inspection Service (APHIS)/CDC Form 3 release reports led to the medical surveillance of 275 individuals. Entities offered post-exposure prophylaxis to ∼76% of the individuals impacted in the presented events.
Summary:
Laboratory safety can be improved by implementing appropriate safety precautions to minimize exposures. Most of the incidents discussed in this evidence-based report occurred during work conducted in the absence of primary containment. None of the releases resulted in illness, death, or transmission to or among workers, nor was there transmission outside of a laboratory into the surrounding environment or community. Effective risk assessment and management strategies coupled with standard and special microbiological policies and procedures can result in reduced exposures and improved safety at facilities.
Introduction
Burkholderia pseudomallei is a gram-negative, rod-shaped, oxidase-positive bacterium that is naturally found in soil and water of tropical and subtropical regions, including, but not limited to, Southeast Asia, northern Australia, and Central or South America. 1 The Southwestern region of the United States is also proposed as a potential endemic region, evidenced by incidences of melioidosis that occurred in a Texas resident in 2018 and in a 2019 fish tank case with no recent international travel history.2,3
Also, the need for the awareness of this organism is substantiated by a recent outbreak involving B. pseudomallei in four cases (two of which were fatal) in Minnesota, Kansas, Texas, and Georgia between March and July 2021 related to contaminated products imported from endemic countries.4,5 Further, ecological niche modeling suggests the potential for B. pseudomallei to become endemic to Gulf Coast states.6,7 Melioidosis is not currently on the national notifiable disease surveillance list. Therefore, reporting of disease outbreak is completed on a voluntary basis.
Melioidosis was suspected in about 47 case reports to the Centers for Disease Control and Prevention (CDC) Bacterial Special Pathogens Branch between 2008 and 2013. 8 The approximated annual global impact of melioidosis is estimated at 165,000 cases, including 89,000 deaths.7,9,10
B. pseudomallei is the causal agent of melioidosis, or Whitmore's disease, characterized by a variable clinical manifestation. 11 Potential symptoms of melioidosis include fever, localized pain with abscesses, a pneumonia-like presentation, and life-threatening septicemia. Symptoms can persist for more than two months in chronic cases. Its wide-ranging symptomatology contributes to a challenging diagnostic process.11–13 B. pseudomallei is transmitted to humans or animals via direct contact with infected animals, soil or water, the inhalation of infected dust, or the ingestion of contaminated water.
Although there have been a few cases of person-to-person transmission, this is rare. Its incubation period is from 1 to 21 days. However, infection can resurface after up to 29 years of latency, suggesting either an inability to produce a sufficient immune response to B. pseudomallei or other influencing factors, such as underlying health conditions.14–16 This organism can be isolated from several specimen types, including blood, skin abscesses or lesions, urine, or sputum. Laboratory-acquired melioidosis has been reported in several instances.17–21
Exposures can potentially occur in the laboratory while performing aerosol-producing procedures with cultures of this organism outside of primary containment. As evidenced by the number of Animal and Plant Health Inspection Service (APHIS)/CDC Form 3s received by Division of Select Agents and Toxins (DSAT) from clinical diagnostic laboratories as compared with non-clinical laboratories, these exposures often occur in clinical diagnostic laboratory environments in which little is usually known about the specimens received for analyses.
The Federal Select Agent Program (FSAP), administered by the U.S. Department of Health and Human Services (HHS), CDC, DSAT and the U.S. Department of Agriculture (USDA), APHIS, Division of Agricultural Select Agents and Toxins (DASAT), regulates the possession, use, or transfer of biological select agents and toxins (BSAT) through the following select agent regulations (SAR): 7 CFR §331, 9 CFR §121, and 42 CFR §73.
The HHS and USDA jointly oversee BSAT that have the potential to pose a severe threat to both public health and safety, as well as to animal health or to animal products, referred to as overlap BSAT. In 2012, B. pseudomallei was designated as an overlap Tier 1 select agent because it is an agent that presents a great risk for deliberate misuse with significant potential for mass casualties or devastating effects to the economy, critical infrastructure, or public confidence.22–24
As defined in the SAR, an entity is any government agency (Federal, State, or Local), academic institution, corporation, company, partnership, society, association, firm, sole proprietorship, or other legal facility. Entities are required by the SAR to immediately notify FSAP upon the identification of B. pseudomallei. The FSAP categorizes entities as either registered entities (REs) or non-registered entities (NREs), depending on whether they have a current approved APHIS/CDC Form 1 registration on file with the FSAP.
The REs are permitted to possess, use, and transfer BSAT. The NREs include clinical diagnostic laboratories that meet select agent regulatory exemption requirements outlined in Sections 5 and 6 in the SAR. The NREs may encounter BSAT for diagnostic, verification, or proficiency testing purposes. Regardless of their registration status, on discovery of release of BSAT causing occupational exposure or release of BSAT outside of the primary barriers of the biocontainment area, an individual or entity must immediately notify CDC or APHIS. 24
Entities must submit a completed copy of the APHIS/CDC Form 3, Incident Notification and Reporting (Theft, Loss, Release), within 7 days of discovering the release event to comply with Section 19 of the SAR.
The APHIS/CDC Form 3 release reports include details on incidents involving BSAT. Collected information includes: (1) the number of individuals exposed, (2) confirmed laboratory-acquired infections (LAIs), if any, (3) details about contamination beyond primary containment, (4) the measures implemented to protect health and security, (5) the measures implemented to decontaminate the BSAT, and (6) internal investigation results to establish the root cause and the implementation of corrective actions to curtail future incidents.
In a clinical diagnostic environment, it is important to consider that unknown patient specimens can contain BSAT. Specimen processing and laboratory safety policies must be developed to protect staff who can encounter BSAT in the laboratory environment. The deleterious effects of this organism along with the implementation of the SAR highlight the significance of awareness of BSAT and the associated hazards. Further, applying safeguards is essential to ensuring the protection of laboratorians, the environment, and the public.
Materials and Methods
Electronic Federal Select Agent Program Information System
The secure information system, Electronic Federal Select Agent Program (eFSAP), is a two-way electronic communication portal that was used by REs to self-report release events involving BSAT. Data were obtained from REs via the electronic APHIS/CDC Form 3 in the eFSAP information system. This information was critically reviewed by the FSAP to determine whether additional information or clarification was required. The FSAP submitted information requests to REs via the eFSAP information system to REs, as necessary, to finalize recommended improvements for the entity.
The NREs did not have access to eFSAP and self-reported these types of incidents using the paper and/or the electronic version of the APHIS/CDC Form 3 via fax or email, which was subsequently documented by the FSAP in the eFSAP information system. Review procedures for release reports received from NREs were conducted as described above. The FSAP communicated with NREs via email, as necessary, to conduct follow-up reviews and to finalize recommended improvements to the NREs. If an incident was deemed to not be a “true” release, the incident was not included in this case study. In some instances, the FSAP conducted on-site compliance inspections to obtain additional information and address safety concerns identified after an incident occurred.
APHIS/CDC Form 3 Reports
The APHIS/CDC Form 3 report was used by entities to report thefts, losses, or releases of BSAT and is referenced in this case study as a release report when not submitted to report a theft or loss. Releases involving BSAT outside of primary containment were self-reported by entities to FSAP using the APHIS/CDC Form 3. Entities provided entity and incident information, and, when applicable, report of release information, including, but not limited to, the incident type, incident date and location, organism(s) involved, the biosafety level (BSL), personal protective equipment (PPE), corrective actions, whether medical surveillance follow-up was performed, etc.
Medical surveillance (e.g., temperature and/or serological monitoring, post-exposure prophylaxis, etc.) included any post-exposure follow-up procedures that entities reported to the FSAP. Release report data were compiled and sorted by the following categories: year, organism, registration status (RE or NRE), entity type (Federal Government, State or Local Government, Academic, Commercial or Private), and BSL-2, BSL-3/animal biosafety level (ABSL)-3.
Case Study Description
In this study, DSAT reviewed B. pseudomallei release reports that were received from REs and NREs in 2017–2019. DSAT determined the number of true release events. The study reports the number of true releases that led to post-exposure medical surveillance. The geographical locations of B. pseudomallei releases were identified. The root causes of B. pseudomallei releases were also examined.
Results
There were a total of 51 B. pseudomallei releases reported to DSAT in 2017–2019. Forty-three (84%) occurred at NREs, and 8 (16%) were reported by REs. For all U.S. states with release reports involving B. pseudomallei during this period, follow-up medical surveillance was offered to 275 individuals (Figure 1A, B). The APHIS/CDC Form 3 reports involving B. pseudomallei in this case study represent ∼8% of the total DSAT release reports received in 2017–2019. The reported releases impacted a greater number of individuals at NREs (n = 211) as compared with REs (n = 64).
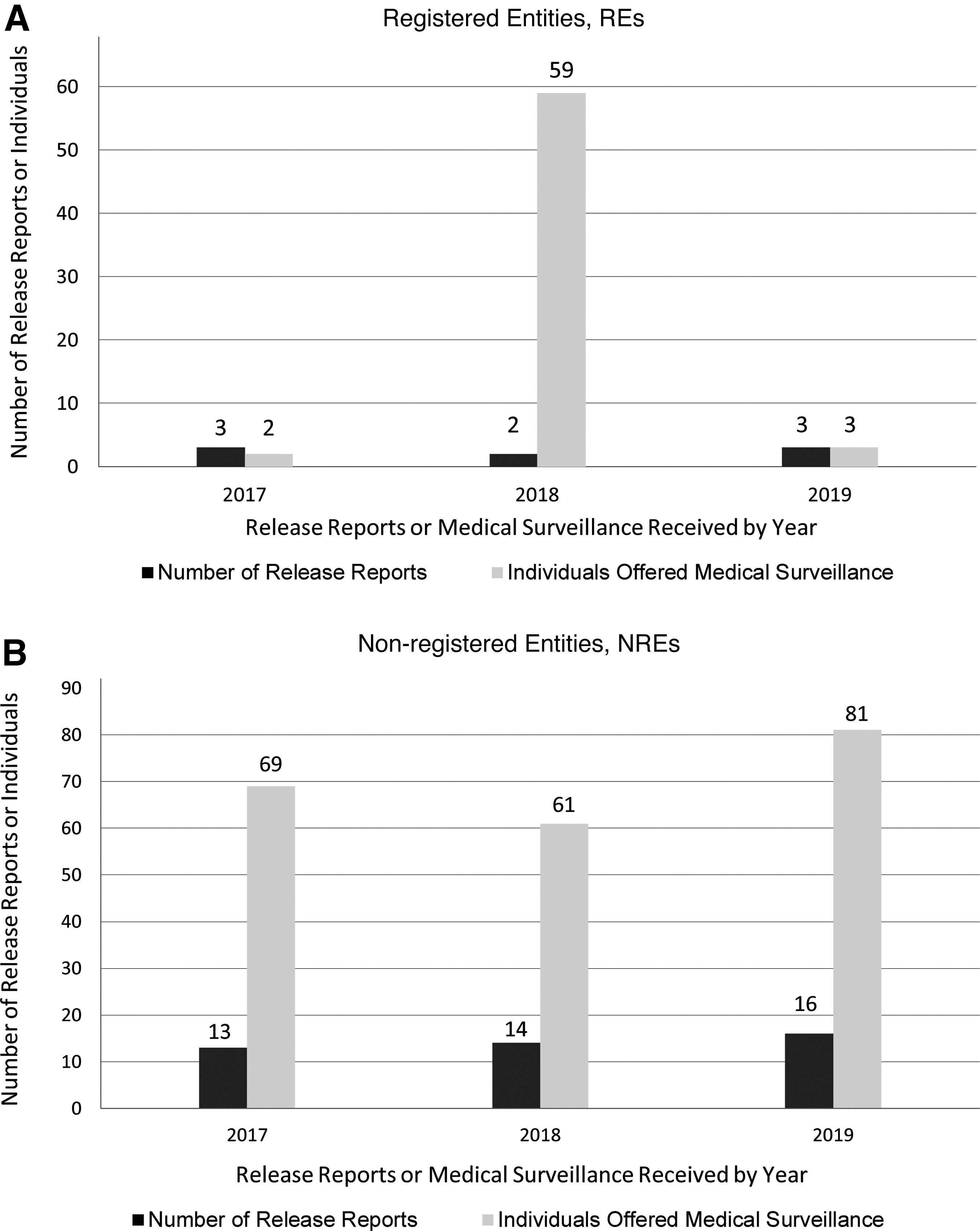
Prophylaxis was recommended by the entities in 47% of the releases reported, and there were no confirmed LAIs resulting from the exposures (data not shown). The releases reported by REs in 2018 represented only two entities and resulted in 59 individuals on medical surveillance. These were unique occurrences since, although they represent a small number of incidents, they impacted a significant number of facilities and other support staff at the entities (Figure 1A). Overall, 86% (n = 44) occurred in a BSL-2 environment as compared with 14% (n = 7) of the releases that occurred in BSL-3 laboratories.
Eighty-four percent (n = 43) of the releases occurred when work was conducted outside of primary containment. The geographical distribution of the APHIS/CDC Form 3 report submitters referenced in this case study indicates a widespread area in which incidents that involved B. pseudomallei occurred and does not support restriction to specific areas as previously suggested (Figure 2). DSAT received reports from a total of seventeen states (Figure 2; shown in gray). The greatest number of reports were received from entities in California (n = 13) and Maryland (n = 8).
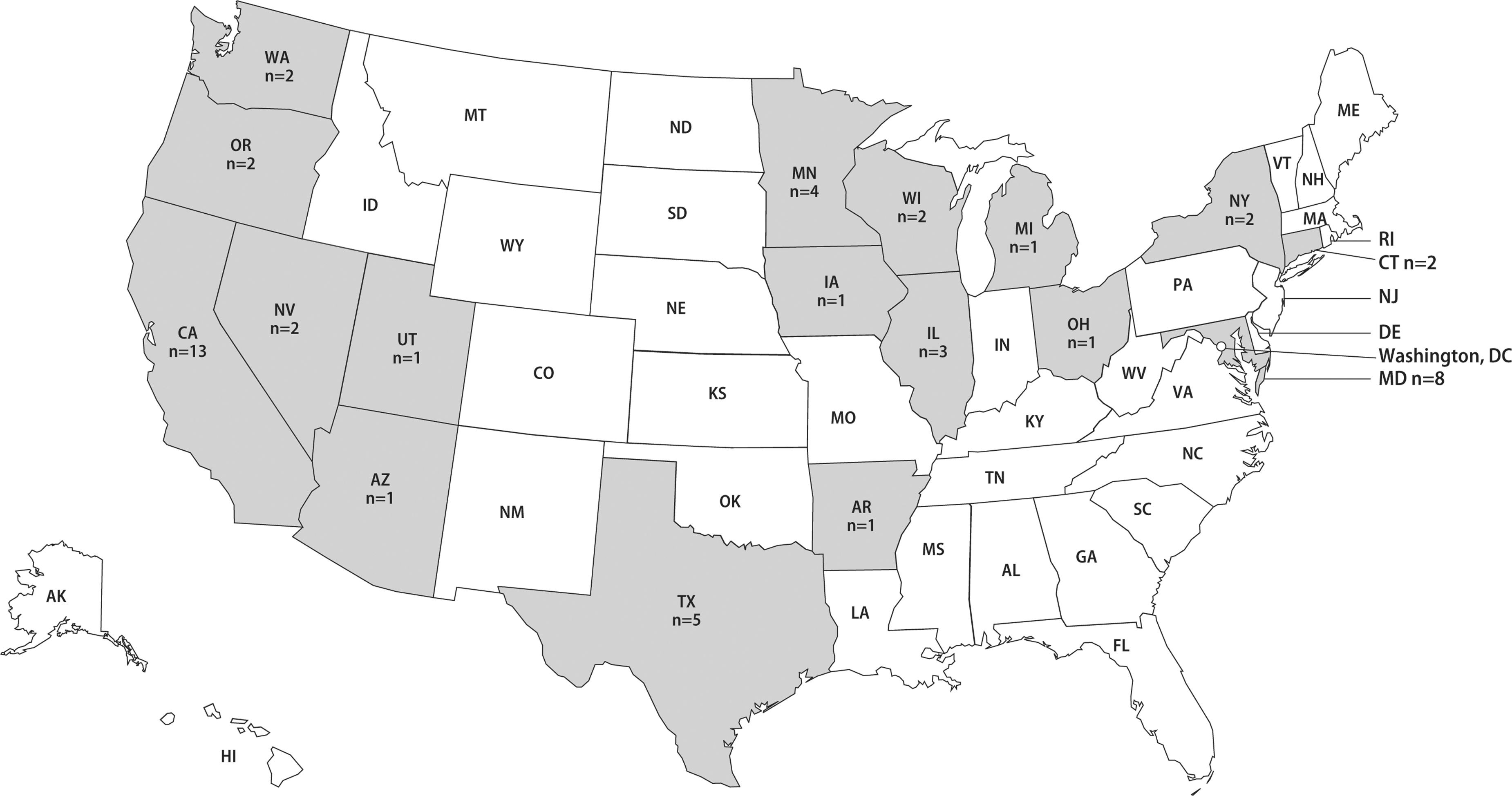
The geographic distribution of Burkholderia pseudomallei release reports received by CDC DSAT in 2017–2019 (shown in gray). The states shown in white did not report B. pseudomallei release incidents in 2017–2019.
Texas (n = 5), Minnesota (n = 4), and Illinois (n = 3) also reported releases during this period. Two reports per state were received from entities in Connecticut, Nevada, New York, Oregon, Washington, and Wisconsin. The remaining states (shown in gray) reported one B. pseudomallei release incident, including Arkansas, Arizona, Iowa, Michigan, Ohio, and Utah. The states with no cases are shown in white. All releases reported by NREs occurred in clinical diagnostic laboratories (n = 43; Table 1). While the types of releases associated with REs were variable, the releases reported by NREs all involved the absence of primary containment (Table 1).
Data retrieved from the information system, electronic Federal Select Agent Program (eFSAP) information system, retrieved May 1, 2020.
ABSL, animal biosafety level; BSL, biosafety level; NRE, non-registered entity; PPE, personal protective equipment; RE, registered entity.
Although requested during information collection, entities infrequently provided root cause information. The REs provided root cause information in 75% of the release reports, while NREs reported root causes in 37% of the release reports (Figures 3A, B). The dominant root cause reported or inferred by REs was human error. Human error was further characterized at REs by releases related to container mishandling (25%), failure to follow established laboratory policies and procedures (25%), and inappropriate PPE doffing procedure incidents (12.5%).
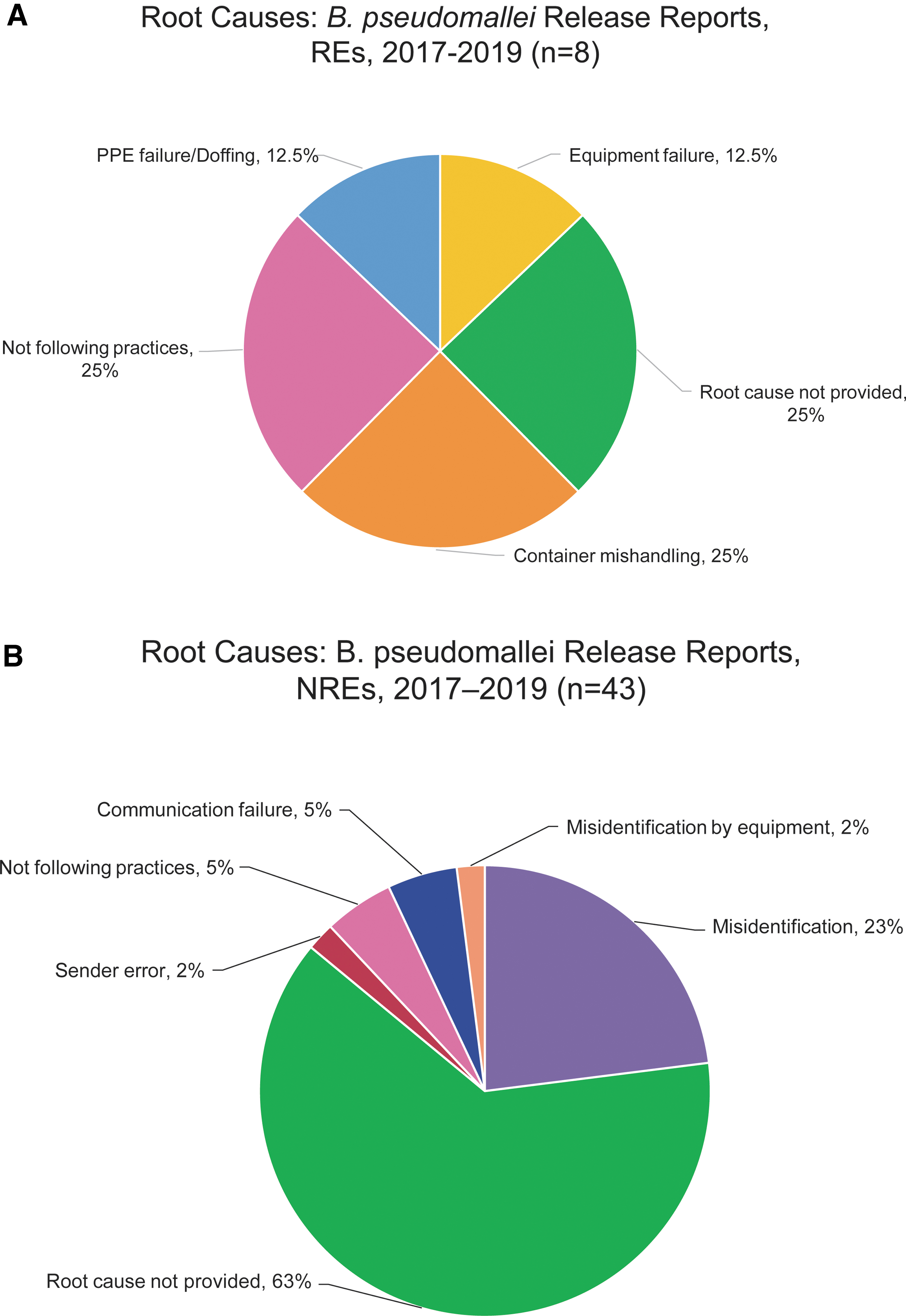
The REs also indicated the failure of equipment designed to contain the select agent (denoted as equipment failure; Figure 3A) as the root cause in 12.5% of the reported release events. The NREs also reported human error as the dominant root cause (Figure 3B). At NREs, human error occurrences involved misidentification of BSAT by laboratory staff (23%), failure to follow established laboratory policies and procedures (5%), communication failure (5%), and sender error (2%). The report of communication failure indicated that the laboratory staff failed to review information about suspected BSAT associated with the patient specimen, although it was communicated by the requesting physician.
In the incident related to sender error, the client provided known BSAT to an entity against the receiving laboratory's policy. The NREs also reported misidentification of B. pseudomallei by the equipment or analyzer (2%). The misidentifications of B. pseudomallei led to releases when work was conducted outside of primary containment (laboratory bench top).
Discussion
To increase laboratorian safety while working with BSAT, it is important to employ practical methods to address releases. This analysis identified human error as the primary root cause in release reports involving B. pseudomallei by entities, irrespective of their registration status. The root cause information emphasized the necessity for research and diagnostic laboratories to expand policies and procedures to further improve their effectiveness in the mitigation of BSAT releases.
In REs, laboratorians must implement exposure prevention strategies, such as the development of improved risk assessments that consider potential safety hazards for all laboratory procedures. Additional precautions to minimize exposures include standard and special microbiological practices described in the Biosafety in Microbiological and Biomedical Laboratories, 6th edition, such as increased training on safety policies and procedures, including, but not limited to, donning and doffing protocols. Also, the development and implementation of effective exposure evaluation and corrective action measures will help to ensure effective management of such occurrences. 25
Atypically, in REs the equipment failure root cause was attributed to an Effluent Decontamination System (EDS) failure. As a result of this single incident, many individuals were offered medical surveillance. It is important to note that REs are more likely to possess waste treatment systems that manage large volumes of potentially contaminated effluents. Waste treatment systems, such as EDSs, are primarily maintained by non-laboratory support staff who must be trained on the safe handling of microbiological hazards that are present.
Entities must ensure that training sufficiently addresses the needs of the staff, the procedures that they perform, and the risks of select agents and toxins that they may encounter. Improved maintenance and decontamination practices will help to limit releases caused by equipment failures. Additional measures to positively impact facility safety include effective site-specific risk assessment and management as well as recurrent review of laboratory safety policies and procedures by laboratory and support staff.
These processes can collectively facilitate the identification and implementation of vital improvements that can strengthen safety programs and improve the effectiveness of current facility biosafety measures. Regular drills or exercises are additional measures that could also reduce BSAT release incidents. Frequent testing of the written safety policies and procedures enable entities to identify methods to enhance safety programs.
The proper use of primary containment devices, such as biological safety cabinets (BSCs) and appropriate PPE, can also ensure that work with BSAT is conducted safely. Further, evaluation of facility training programs is an important factor that can ensure that improved behaviors and intended safety practices are achieved.
Clinical diagnostic laboratories are primarily NREs that involve a high throughput of specimens and minimal clinical history information. Additionally, laboratory space, design, and resource constraints often limit the availability of BSCs for all analyses. It is, therefore, important for clinicians to communicate patient travel history to laboratory staff if melioidosis is suspected. Also, laboratory staff should perform a thorough review of submission forms and optimize the testing algorithm before processing specimens that may contain BSAT.
Automated equipment is commonly used in clinical diagnostic laboratories since there is pressure to improve cost efficiency and productivity with a decreasing labor force. 26 However, some of the analytical instrumentation may present a major issue to laboratory operations due to the misidentification of an agent and the increased potential for exposure of laboratory staff to BSAT. When automated instrumentation is used in release events involving BSAT, many individuals can be impacted.
Laboratory release incidents can add a considerable burden to local, state, and federal laboratory and epidemiology resources who are tasked with conducting immediate analyses and investigations. 24 Also, costs associated with employee health, increased absences, and medical surveillance follow-up due to potential exposures to BSAT should also be considered.26–28 A key to achieving a decrease in laboratory releases and exposures is expanding training efforts, including BSAT growth characteristics, colony morphology, and following rule-out algorithms, such as guidelines outlined in the American Society for Microbiology (ASM) Laboratory Response Network Sentinel Level Clinical Laboratory Protocols.
Following these procedures could help to minimize safety risk and provide recommendations for laboratory workflow and alert staff regarding when to move the analyses from the open bench into a BSC. Knowledge of these processes and implementation of available sentinel clinical laboratory resources, such as Association of Public Health Laboratories bench cards, could also enhance training, awareness, and safety improvement efforts. When BSAT is suspected, a BSC should be used for subsequent assays until BSAT is ruled out.
Expanding training efforts, such as via the ASM protocols for applicable NRE staff, can significantly develop laboratory entity biosafety. 29 It is critically important to use primary containment when working with unknown specimens that may later be identified as BSAT to enhance laboratory safety.
Conclusion
The FSAP requires all entities to self-report releases involving BSAT. The timely reporting of these types of incidents to the FSAP promotes laboratory safety and security. No LAIs or transmissions were identified (data not shown) in the 51 reports of laboratory releases involving B. pseudomallei. While B. pseudomallei does not present a great LAI risk, the absence of LAIs is likely attributed to the amount of pathogen present in the release events, immediate engagement of impacted individuals to determine the risk of infection, quick communication to exposed staff, prompt surveillance activities, and quick post-exposure prophylaxis measures when recommended.
There were ∼69% more B. pseudomallei release events in NREs (n = 43) than REs (n = 8) in the period examined. This is a common finding in overall NRE and RE release reports. The REs work with known select agents within primary containment. Most NREs work with unknown diagnostic samples and are unaware of when patient specimens contain BSAT. Further, the NREs reported in this study primarily operate at a BSL-2 level, in which laboratory practices allow for work on an open benchtop.
In contrast to NRE BSL-2 laboratories, primary containment devices are required in BSL-3 laboratory environments commonly used by REs. An increase in the awareness of the potential for the presence of BSAT in patient specimens will assist in efforts to mitigate similar release events. The successful implementation of effective risk assessment and other improvement strategies discussed can drastically reduce safety risks while working with BSAT.
Limitations
This study consisted of CDC DSAT data only and includes reports to CDC DSAT of releases of one select agent that occurred in BSL-2, BSL-3, ABSL-2, and ABSL-3 laboratories. It does not include reports made to USDA APHIS DASAT.
Footnotes
Disclaimer
The findings and conclusions in this article are those of the authors and do not necessarily represent the official position of the (U.S.) Centers for Disease Control and Prevention.
Authors' Disclosure Statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding Information
The authors received no financial support for the research, authorship, and/or publication of this article.
