Abstract
Aim:
This study aimed to validate the efficacy of hydrogen peroxide vapor (HPV) decontamination technology set up in a biosafety level 3 (BSL-3) laboratory on surrogates and hazard group 3 (HG3) agents.
Methods and Results:
The HPV decontamination system (Bioquell) was assessed with both qualitative and quantitative methods on (1) spore surrogates (Geobacillus stearothermophilus, Bacillus atrophaeus, and Bacillus thuringiensis) in the BSL-3 laboratory and in the material airlock and on (2) HG3 agents (Bacillus anthracis; SARS-CoV-2, Venezuelan equine encephalitis virus [VEE], and Vaccinia virus) in the BSL-3 laboratory. Other HG3 bacteria likely to be handled in the BSL-3 laboratory (Yersinia pestis, Burkholderia mallei, Brucella melitensis, and Francisella tularensis) were excluded from the HPV decontamination assays as preliminary viability tests demonstrated the total inactivation of these agents after 48 h drying on different materials.
Conclusions:
The efficacy of HPV decontamination was validated with a reduction in viability of 5–7 log10 for the spores (surrogates and B. anthracis), and for the enveloped RNA viruses. Vaccinia showed a higher resistance to the decontamination process, being dependent on the biological indicator location in the BSL-3 laboratory.
Introduction
Hydrogen peroxide vapor (HPV) decontamination is a « no-touch » automated technology that allows large-volume area treatments and ease of dispersion. It uses a generator to vaporize liquid hydrogen peroxide until reaching air saturation and HPV condensation on the surfaces to be decontaminated. The layer of microcondensation formed by HPV inactivates microorganisms it contacts. 1 Hydrogen peroxide is a potent oxidant and a highly active biocide, which forms hydroxyl-free radicals that penetrate the cell wall to attack lipids, proteins, or DNA. 2 Previous studies demonstrated the efficacy of vaporized hydrogen peroxide generating systems on bacteria,3–5 spores,6,7 or viruses.8–12 At the end of the HPV decontamination cycle (DC), the toxic vapor breaks down to water and oxygen alone, leaving no residues and requiring no postprocess neutralization.
This vapor technology is an effective alternative to the commonly used formaldehyde fumigation, and is less hazardous to human health. In the European Union (EU), formaldehyde is classified as a Class 1B carcinogen and a Class 2 mutagen by Commission Regulation (EU) No. 605/2014 of June 5, 2014. As its use is progressively banned and its supply becomes unavailable, safer and more environmentally friendly formaldehyde substitutions are urged.
HPV technology can be used for the decontamination of various areas, such as hospital rooms,13,14 laboratory chambers,3,15 microbiological safety cabinets (MSCs), 5 or buildings. 16 Moreover, in the current context of COVID-19 pandemic, hydrogen peroxide sterilization technologies have been tested in an extensive number of recent studies for the reuse of personal protective equipment (PPE), especially N95 respirators or coveralls.17–20
In this study, HPV technology (Bioquell) was set up in a biosafety level 3 (BSL-3) laboratory to replace the former formaldehyde decontamination system. Demonstration of the efficacy of this HPV disinfection technology is required by the French decree of June 11, 2013, 21 which imposes the validation of the decontamination methods used on all bacteria, spores, or viruses handled in confined laboratories.
In a first step, the HPV decontamination system was tested in the BSL-3 laboratory and in the material airlock using biological indicators (BIs) with endospore surrogates (Geobacillus stearothermophilus, Bacillus atrophaeus, and Bacillus thuringiensis). Sporulating bacteria are commonly used to validate fumigation efficacy studies due to their known high resistance in various environmental conditions. 22
In a second step, hazard group 3 (HG3) agents were exposed to HPV DCs in the BSL-3 laboratory. Hazardous biological agents are classified by the World Health Organization in different groups ranking from 1 (lowest risk) to 4 (highest risk), depending on the likelihood the agent will cause disease by infection or toxicity to humans, the likelihood the infection will spread to the community, and whether treatment or preventive measures are available for that agent. 23 HG3 agents used in the BSL-3 laboratory present a low community risk and effective treatment is available, but they represent a high individual risk and are potential bioterrorism agents (e.g., anthrax spores).
HG3 agents chosen for this study are representative of the agents—or group of agents—likely to be handled in the BSL-3 laboratory: the bacteria Yersinia pestis, Burkholderia mallei, Brucella melitensis, and Francisella tularensis tularensis; the spores of Bacillus anthracis; and the viruses SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2, Coronaviruses), VEE (Venezuelan Equine Encephalitis virus, Alphaviruses), and Vaccinia (Orthopoxviruses). Preliminary viability tests were conducted to exclude from the HPV decontamination assays any of the HG3 agents that could not recover after 48 h drying on different materials (i.e., laps of time between the BIs preparation and the end of the DCs).
The HPV decontamination efficacy in the BSL-3 laboratory and in the material airlock was evaluated with both qualitative and quantitative methods. The expected decontamination performances, as required by the French Standardization Association guidelines NF T72-281, 24 were a minimum reduction in viability of 5 log10 for the agents (bacteria and viruses) and 3 log10 for the spores.
Materials and Methods
Biosafety
All experiments with HG3 agents were conducted in a BSL-3 laboratory in the facilities of the French chemical, biological, radiological, and nuclear defense expert center (DGA CBRN Defence), Vert-le-Petit (France). All staff entered the laboratory with a BSL-3–specific PPE (Tyvek suits, FFP3 respiratory protection, double gloves, protection glasses, overshoes, and overboots). Discharged air in the containment laboratory was filtered during the experiments with a high efficiency particulate air filter (20 vol/h). Waste was removed using pass-through autoclaves and transported offsite to biorisk waste treatment facilities. Experiments with bacterial surrogates (e.g., BIs preparation, culture, or incubation) were conducted in a BSL-1 laboratory.
HPV Decontamination Set-Up in the BSL-3 Laboratory and in the Material Airlock
The BSL-3 laboratory consists of an ∼200 m2 surface area divided into several zones: an entrance airlock, a corridor area, and six experimental rooms. A material airlock of 2.5 m2 adjoins the BSL-3 corridor and allows the transfer of bulky equipment between the inside and outside of the laboratory. In the BSL-3 laboratory, two Bioquell Clarus Z-2 generators were placed in the corridor area before starting a DC, each of them containing a 5-L bottle of a 35% hydrogen peroxide solution. The HPV was dispersed throughout the different rooms by airflows generated by 15 fans located in different areas. Locations and orientations of the fans, as well as the peroxide concentrations needed, were previously validated by a Bioquell technical expert. In the material airlock, the HPV decontamination is performed by a Bioquell SeQure generator fixed on the wall and connected to a module with two 950-mL bottles of a 35% hydrogen peroxide solution. DCs took place in four different phases: conditioning, HPV dispersion, contact, and aeration. All doors of the BSL-3 laboratory between the corridor and the experimental rooms and the entrance airlock remained open during the DCs, and all the MSCs were left open but turned off (to not disrupt air flows and to mimic a classic bench-top environment). The two doors of the material airlock were closed before starting the room decontamination.
BIs Preparation and HPV Decontamination on Surrogates
The HPV decontamination efficacy was first assessed on spore surrogates before proceeding with hazardous agents. Six different BIs were used: two commercial and ready-to-use BIs at 106 spores (Geobacillus stearothermophilus [Bioquell] and Bacillus atrophaeus [Mesalab]), and two “self-prepared” BIs at 105 and 107 spores (Bacillus atrophaeus [ATCC 9372], and Bacillus thuringiensis produced from the biopesticide Delfin®). Commercial BIs at 106 spores per disk and prepared BIs at 105 spores per disk were used to obtain a qualitative response; prepared BIs at 107 spores per disk were used for both quantitative and qualitative responses. A BI consists of a 1 cm stainless steel disk containing a defined amount of dried agents/spores sealed in Tyvek pouches. In brief, the noncommercial BIs were prepared as follows: stainless steel disks, collected from commercial BIs, were autoclaved (20 min at 120°C) and disposed with sterile tweezers on an empty Petri dish in an MSC. A 10 μL drop of 105 or 107 spores was spread on each disk and left to dry one night in an hermetic plastic box in the dark, at ambient temperature (AT). Disks were then introduced in Tyvek pouches and closed with a clip. Prior the DCs, BIs were taped in 33 different positions in the BSL-3 laboratory (e.g., inside the MSC, behind freezers, in open incubators, or centrifuges) and in 10 different positions in the material airlock (e.g., on walls, inside and outside a cabinet introduced in the airlock). After the HPV DC, BIs were collected and cultured. For qualitative response, disks were introduced in a 9 mL tryptic soy broth tube (TSB; Biomérieux) and incubated at 37°C for B. atrophaeus and B. thuringiensis, and at 56°C for G. stearothermophilus. Culture development in the broths was followed at +24 h, +48 h, and +7 days. For the quantitative response, disks of the prepared BIs at 107 spores were vortexed 2 min in 2 mL of sterile water in a 15 mL conic tube. After 1 h at AT, 100 μL and 1 mL of the suspension were plated on tryptic soy agar (TSA; Biomérieux). A qualitative response was also obtained with the BIs at 107 spores by adding 9 mL of TSB in the remaining bacterial suspension. Plates and tubes were incubated as described at 37 or 56°C and observations for bacterial development conducted at +24 h and +48 h for the plates and the tubes, and at +7 days for the broths only. Qualitative responses of positive controls (not exposed to HPV) for commercial BIs and prepared BIs at 105 and 107 spores were obtained under the same conditions as the treated samples. Positive controls of the BIs at 107 spores were cultured for numerations: a 2 mL bacterial suspension in sterile water was obtained as previously described; the bacterial suspension was further diluted to 10−4, 10−5, and 10−6 and plated on TSA for 24 h at 37°C. HPV decontamination assays on bacterial surrogates in the BSL-3 laboratory and in the material airlock were reproduced three times each.
Viability of HG3 Bacteria and Viruses Without HPV Decontamination
We have conducted preliminary viability tests to exclude from the HPV decontamination assays the HG3 agents that could not recover after a 2-day period at AT in the dark. Indeed, the time needed between the preparation of the “self-prepared” BIs and the end of the HPV decontamination is ∼48 h (24 h of drying and 24 h for the decontamination process).
For HG3 bacteria, the viability was assessed for strains of Y. pestis (CO92), B. mallei (ATCC 23344T), B. melitensis (ATCC 23456), F. t. tularensis (FSC 041), and spores of B. anthracis (FERRARA) as a positive control. Bacterial strains were preserved at −80°C in high-security cryopreservation straws (Cryobiosystem) and spore suspension of B. anthracis was kept at +4°C. Cryopreserved strains were suspended in a brain heart infusion broth (Biomérieux) before being isolated on agar plates. Yersinia pestis, B. mallei, and B. anthracis were cultured on TSA at 28°C, B. melitensis and F. t. tularensis were cultured at 37°C + 5% CO2 on TSA and PVX (chocolate agar+polyViteX; Biomérieux), respectively. After 3–4 days of incubation, several isolated colonies per plate were suspended in 5 mL of sterile water with an inoculation loop and further diluted to 10−1. Numerations on appropriate agar medium were conducted to determine the initial concentration of the bacterial suspensions. A 10 μL drop of the suspensions was spread on an autoclaved stainless steel disk (20 min, 120°C) or on autoclaved plastic flooring coupons (30 min, 115°C). Inoculated disks and plastic coupons were left to dry in an MSC for 2 days and then vortexed 2 min in 2 mL (disks) or 5 mL (plastic coupons) of sterile water. After 1 h at AT, 100 μL of the bacterial suspension was further diluted to 10−4, 10−5, and 10−6 for numerations, and 9 mL of TSB was added in the remained suspension for a qualitative response. Plates and broths were incubated 11 days in the conditions previously described and observed daily for growth.
Survival tests on stainless steel disks were also conducted with three HG3 viruses: VEE TC 83 (Venezuelan Equine Encephalitis virus), Lister strain of vaccinia virus (VVLister) (both strains from DGA MNRBC collection), and SARS-CoV-2 (strain BetaCoV/France/IDF0372/2020 from Pasteur Institute). Pure viral suspensions at 4 × 109 pfu/mL for VEE TC 83, 6 × 106 pfu/mL for VVLister, and 2 × 104 pfu/mL for SARS-CoV-2 were preserved at −80°C. A 10 μL drop of pure viral suspension was spread on the disk and left to dry 1 or 4 days in an hermetic plastic box at AT, in the dark. For each survival time assessed, three replicates were prepared. Disks were immersed in a tube with 2 mL of culture medium and vortexed for 2 min. Culture medium was composed of M199 (Gibco) +2% fetal bovine serum (FBS; Gibco) +1X antibiotic/antimycotic (ATB; Gibco) for VEE TC 83 and VVLister, and of Dulbecco's modified Eagle's medium (DMEM; Gibco) +2% FBS +1X ATB for SARS-CoV-2. Viral suspension in each tube was transferred in a well of a six-well microplate containing VERO cells for VEE TC 83 and VVLister, or VERO E6 cells for SARS-CoV-2. The tube was rinsed with 1 mL of the culture medium, which was then added in the well with the viral suspension. Culture plates were prepared 1 day before the experiment with VERO cells in M199 + 10% FBS +1X ATB for VEE TC 83 and VVLister, and with VERO E6 cells in DMEM +10% FBS +1X ATB for SARS-CoV-2. Prior to the transfer of the viral suspension in the culture plate, the host cell medium was removed and wells were rinsed twice with 1 mL of phosphate-buffered saline. Negative controls with VERO or VERO E6 cells were prepared with culture medium only. Plates were incubated at 37°C + 5% CO2 and cytopathic effects on the cell layer were observed under light microscope for a 3-day period. The observation of a cytopathic effect corresponds to the presence of at least one viral particle in the well and provides a qualitative answer; it did not allow determining the number of particles in the well.
Survival assays were repeated three times, with triplicates for each support (stainless steel disk and/or plastic coupons) and for each bacterial or viral strain.
HPV DCs on HG3 Bacteria and Viruses in the BSL-3 Laboratory
For bacteria, BIs with B. anthracis at 105 and 107 spores per disk and commercial BIs with B. atrophaeus at 106 spores per disk were prepared and placed in eight different locations in the BSL-3 laboratory. The HPV decontamination efficacy was evaluated with qualitative responses (B. atrophaeus and B. anthracis at 105 and 107 spores per disk) and quantitative responses (B. anthracis at 107 spores per disk). Positive controls, not exposed to HPV, were prepared and cultured as previously described.
For viruses, BIs were prepared by spreading a 10 μL drop of pure viral suspensions on stainless steel disk. The pure viral suspensions previously described were used for VEE TC 83 and VVLister. A pure viral suspension at 1.6 × 107 pfu/mL, preserved at −80°C, was used for SARS-CoV-2. Disks were left to dry one night in the dark at AT in an hermetic plastic box before being introduced in Tyvek pouches. BIs with viral particles were placed in three different locations of the BSL-3 laboratory. After the DC, disks were inoculated in 2 mL of culture medium to be vortexed for 2 min, and 13 mL of culture medium was further added to the viral suspension. This 1/15 dilution allowed preventing the cytotoxic effects of HPV residues on VERO and VERO E6 cells (data not shown). Five wells of a six-well culture plate were filled with 3 mL of the 15 mL viral suspension. Culture plates, prepared as previously described, were incubated at 37°C + 5% CO2 and observed under light microscope for cytopathic effects at +3 days, +4 days, and +7 days (qualitative response). Negative controls, exposed to HPV, were prepared to verify the absence of cytotoxicity on VERO and VERO E6 cells. Positive controls, not exposed to HPV, were cultured by successive dilutions from pure suspension in a 96-well culture plate to quantify the viral particles reduction after a 48 h period at AT. The Reed and Muench calculation method was used for virus titration (Reed and Muench, 1938). 25
HPV DCs with the HG3 bacteria and viruses were reproduced three times.
Results
HPV DC Parameters
Six DCs were conducted in the BSL-3 laboratory: three cycles to validate the HPV decontamination on surrogates and three cycles on HG3 agents. The duration of each cycle was 7 h 20 ± 7 min, the hygrometry varied between 52.1 ± 1.3% relative humidity (RH) at the beginning of the conditioning phase and peaked at 99.5 – 0.5% RH during the vaporization phase. HPV concentration reached 527.9 ± 15.6 ppm during the contact phase and the temperature was maintained at 21.4 ± 0.5°C for the duration of the DCs. In the material airlock, the three DCs lasted 2 h 45 ± 0 min, the hygrometry varied between 63.5 ± 1.1% RH and 99.9 ± 0% RH, and the HPV concentration peaked at 799 ± 38 ppm. Temperature in the airlock was maintained at 21.8 ± 2.3°C.
HPV Decontamination Efficacy on Surrogates in the BSL-3 Laboratory and in the Material Airlock
In the BSL-3 laboratory, results showed the complete inactivation of spore surrogates in the 33 different positions after the HPV DCs for the commercial BIs, the noncommercial B. atrophaeus BIs at 105 and 107 spores, and B. thuringiensis at 105 spores (Table 1). Only one BI of B. thuringiensis at 107 spores was not fully decontaminated after each DC, at different locations, as growth was observed in broths (the growth of B. thuringiensis in these broths was confirmed by isolations on TSA). However, the absence of colony on plates after DC1 and DC2 indicated a decontamination efficacy of 7 log10, and the colonies counted on one plate after DC3 indicated a reduction of 5.3 log10 for the positive BI. Results in the BSL-3 laboratory validated the HPV decontamination efficacy on bacterial surrogates as well as the optimal dispersion of the decontaminating product through the different rooms.
Hydrogen peroxide vapor decontamination cycles efficacy on surrogates in the biosafety level 3 laboratory and in the material airlock
Commercial BIs.
BI, biological indicator; BSL-3, biosafety level 3; CFU, colony forming unit; nb, number.
In the material airlock, several B. atrophaeus BIs at 107 spores were not completely inactivated after DC2 and DC3, but the mean log10 reduction calculated were, respectively, 6.3 ± 0.8 log10 and 5.9 ± 0.3 log10 (Table 1). The other BIs in the material airlock were fully inactivated (Table 1).
The HPV decontamination efficacy was higher than the expected 3 log10 reduction in both the BSL-3 laboratory and the material airlock, and for all the surrogates tested.
Viability Assays on HG3 Bacteria and Viruses After 48 h Drying Without HPV Decontamination
Preliminary viability assays on HG3 bacteria demonstrated the total inactivation of F. t. tularensis, B. mallei, and B. melitensis after 48 h drying on stainless steel disks and plastic coupons (Table 2). Data for F. t. tularensis were obtained for two trials only as no growth was obtained for the positive control in the third trial. In the third trial with Y. pestis, two of three broths inoculated with the disk were positive, and one of three broths inoculated with the plastic coupon was positive, whereas no colony grew on plates. The number of remaining viable bacteria was estimated at <10 per disk or coupon. Out of the six HG3 bacteria tested, it appeared relevant from these results to evaluate only the HPV decontamination on B. anthracis.
Colony forming unit recovered of hazard group 3 bacteria after 48 h drying on different materials
HG3, hazard group 3; nd, not determined; SD, standard deviation.
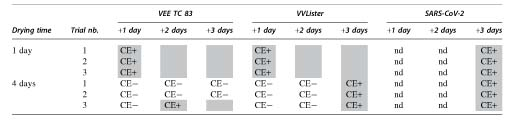
Assays on HG3 viruses demonstrated that viable particles of VEE TC 83, VVLister, and SARS-CoV-2 remained on disks after 4 days of drying (Table 3). Considering the viability of the viral particles, HPV decontamination in the BSL-3 laboratory had to be evaluated on these three viruses.
Cytopathic effect observations over a 3-day period of culture cells exposed to virus suspensions dried 1 or 4 days on stainless steel disks
CE, cytopathic effect; CE−, no CE observed; CE+ and gray shade, observation of CE; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; VEE TC 83, Venezuelan equine encephalitis virus.
HPV Decontamination of HG3 Bacteria and Viruses in the BSL-3 Laboratory
HPV decontamination totally inactivated B. anthracis spores in all the eight positions tested in the BSL-3 laboratory (Table 4).
Hydrogen peroxide vapor decontamination efficacy on Bacillus anthracis in the biosafety level 3 laboratory
Commercial BI.
DC, decontamination cycle.
For viruses, VEE TC 83 and SARS-CoV-2 were inactivated in all the three positions tested (Table 5). VEE TC 83 positive control could not be quantified due to a too low concentration remaining on the disk, but cytopathic effects observed in the culture wells testified the presence of viable viral particles. For VVLister it appeared that the decontamination efficacy was dependent on the BI position. Indeed, the virus was totally inactivated in position 3 (P3), whereas decontamination proved ineffective in P1 and P2 (Table 5).
Hydrogen peroxide vapor decontamination efficacy on hazard group 3 viruses in the biosafety level 3 laboratory
P1, position 1; P2, position 2; P3, position 3; PFU, particle forming unit.
Discussion
To validate the decontamination efficacy in the BSL-3 laboratory, BIs with dried agents on stainless steel disks were exposed to HPV. This desiccating process can be harmful for some agents and can lead to a significant loss of viability to a total inactivation. Previous studies demonstrated the reduction in recoverable number of B. pseudomallei, F. tularensis, or Y. pestis after 1 to 12 h of drying on different materials. 3 A 1 log10 reduction in viability of Y. pestis dried in its growing medium onto stainless steel was also witnessed by Pottage et al. (2019) after only 90 min. 5 In this study, we assessed the viability of several HG3 agents dried for 48 h on stainless steel disks, which is the approximate time between the drop deposit of the agent on the disk and the end of the DC. As the bacterial strains of B. melitensis, F. tularensis, Y. pestis, and B. mallei were totally inactivated by this 2-day desiccation, we considered irrelevant to include these agents in the HPV decontamination assays. In contrast, the resistance to the drying process of the three viruses VEE TC 83, SARS-CoV-2, and VVLister testified the need to confirm their inactivation by hydrogen peroxide. Spores of B. anthracis, well known for their persistence, 26 were expected to recover from the desiccation process and were also included in the HPV decontamination assays in the BSL-3 laboratory.
In a first step, we have demonstrated the Bioquell HPV decontamination efficacy in both the BSL-3 laboratory and the material airlock on surrogates with a log reduction in recovery of 5–7 log10 for all the indicators tested, all tests combined. These results also testified that the HPV was optimally dispersed throughout the different rooms of the BSL-3 laboratory, even in hard-to-reach areas.
Second, our assays with HG3 agents in the BSL-3 laboratory confirmed the efficacy of the Bioquell technology, with the total inactivation of the B. anthracis BIs at an initial load of 106 or 107 spores per disk. These results are in accordance with previous study, which demonstrated the total inactivation of B. anthracis at an initial load of 108 spores per BI by the Bioquell HPV technology in an MSC. 5 HPV decontamination was also previously proved effective to inactivate various viruses,10,11 and our assays confirmed the disinfection efficacy of this technology on the RNA enveloped viruses VEE TC 83 and SARS-CoV-2, with a minimum of 5 log10 reduction in recovery.
The decontamination performances obtained in the material airlock and in the BSL-3 laboratory met the requirements of the French Standardization Association guidelines NF T72-281, 24 which demands a minimum reduction of 5 log10 for the agents (bacteria and viruses) and 3 log10 for the spores. However, the DNA enveloped virus VVLister showed resistance to the HPV decontamination process for BIs located in two of the three positions tested. These two BIs were located under two different MSCs of the same room (dedicated to virology experiments), whereas the fully decontaminated BI was located under an MSC of another experimental room, proving that total decontamination of VVLister by hydrogen peroxide may be effective in areas under optimized decontamination parameters.
The HPV decontamination in the virology room was nevertheless efficient as the spore surrogates and the viruses VEE TC83 and SARS-CoV-2 were totally inactivated, but it might not have been fully optimized for agents that are particularly resistant to decontamination processes, such as VVLister. 27 To test that hypothesis, we placed another resistant agent, B. anthracis, at an initial load of 107 spores per BI in the virology room before starting a DC (data not shown). We observed bacterial growth in the broth, testifying that B. anthracis was not fully inactivated in the virology room, whereas our results showed that B. anthracis was totally decontaminated in the eight other locations tested. Therefore, to achieve total decontamination of VVLister (and Orthopoxviruses in general) by hydrogen peroxide in the virology room, it would be necessary to make adjustments such as changing the position of the generators and/or of the fans in the room; performing an additional decontamination in this room only; or changing the cycle parameters (e.g., increasing the decontamination time).
In any cases, HPV decontamination remains a terminal decontamination step, and a careful surface disinfection using decontaminant products must always be performed beforehand. Indeed, the degree of soiling may reduce the effectiveness of the vapor decontamination.8,28 Therefore, the use of BIs with dried agents in their growth medium in this study, instead of washed suspensions, is more challenging for the HPV technology as the organic matter and salts provide protection from the contact with hydrogen peroxide. 5 In addition, these nonwashed BIs are more representative of contamination incidents that may occur in laboratories. Moreover, BIs with dried viruses as those used in this study present a greater challenge for HPV decontamination in comparison with viruses in suspension. 29
Despite its decontamination efficacy, the HPV technology presents several drawbacks, especially related to the laboratory preparation before the decontamination (disinfection of surfaces and removal of absorbent materials), and to the oxidative properties of hydrogen peroxide. Indeed, it had been necessary to change the coating of the walls and the paint of the doors that were incompatible with the HPV decontamination in the BSL-3 laboratory. Several devices, which were starting to rust or blister after a few HPV DCs, had also to be replaced (e.g., MSCs, incubators, stools). Moreover, to validate each DC, it is required to culture in broth commercial BIs exposed to HPV and observe for growth for 7 days, preventing the entry in the laboratory during this entire period.
The Bioquell HPV decontamination system set up in the BSL-3 laboratory nevertheless has advantages in comparison with its formaldehyde predecessor as, unlike the later, hydrogen peroxide is not a carcinogenic, mutagenic, and reprotoxic classified substance, and it leaves no odor or residues on treated surfaces.
Conclusion
This study demonstrated the efficacy of the Bioquell HPV decontamination technology on surrogates and on HG3 bacteria and viruses in an entire BSL-3 laboratory of several hundred square meters. Running such decontamination validation assays require a large number of consumables (e.g., culture medium, BIs, and PPE) and is time consuming, but it ensures the proper disinfection of the premises, and guarantees the safety of the personnel entering the BSL-3 laboratory after the DCs.
Footnotes
Acknowledgment
We thank Pr. Sylvie van Der Werf for providing us the SARS-CoV-2 BetaCov/France IDF0372/2020 viral strain.
Authors' Contributions
C.F., C.B., V.L., L.B., A.M., and V.V-L. carried out the experiments and contributed to the interpretation of results; M.A., L.B., and F.R. conceived and planned the experiments; C.F. wrote the article with support from C.B., G.L., and F.R.; and F.R. and L.T. supervised the project. All authors provided critical feedback, discussed the results, and contributed to the final article.
Authors' Disclosure Statement
No competing financial interests exist.
Funding Information
No funding was received for this article.