Abstract
Introduction:
The applications of fumigation and the challenges that high-containment facilities face in achieving effective large volume decontamination are well understood. The Biosecurity Research Institute at Kansas State University sought to evaluate a novel system within their biosafety level 3 (BSL-3) and animal biosafety level 3 agriculture (ABSL-3Ag) facility.
Methods:
The system chosen for this study is the CURIS® Hybrid Hydrogen PeroxideTM (HHPTM) system, comprising a mobile 36-pound (16 kg) device delivering a proprietary 7% hydrogen peroxide (H2O2) solution. To examine the system's efficacy in multiple laboratory settings, two BSL-3 laboratories (2,281 [65 m3] and 4,668 ft3 [132 m3]) with dropped ceiling interstitial spaces and an ABSL-3Ag necropsy suite (44,212 ft3 [1,252 m3]) with 21-foot (6.4 m) ceilings were selected. Biological indicators (BIs) of
Results:
After cycle optimization, the smaller laboratory had a total of 60 BIs over two treatments that demonstrated a greater than 6-log reduction of bacterial spores. The larger laboratory (192 BIs) and the necropsy suite (206 BIs) had no BIs positive for spore growth when incubated at 60°C for 24 h per manufacturer's specifications.
Conclusion:
Overall successful results through multiple components of this study demonstrate that the HHP device, paired with the pulsed 7% H2O2 solution, achieved efficacy regardless of variables in laboratory size and layout. Perceived challenges such as 21-ft (6.4 m) ceiling heights, active equipment, and difficult to access ceiling interstitial spaces proved unfounded. Given the successful sterilization of all challenged BIs, the HHP system presents a useful alternative for high level decontamination within BSL-3 and ABSL-3Ag facilities.
Background
Biosafety Level 3 (BSL-3) facilities and Animal Biosafety Level 3 Agriculture (ABSL-3Ag) facilities are designed to allow for the study and handling of high-consequence pathogens that affect plants, animals, and food products, including zoonotic pathogens that can infect humans. Guided by Biosafety in Microbiological and Biomedical Laboratories, 6th Edition, principles for biosafety are focused on practices of containment and risk assessment. 1 Decontamination practices reduce potential for cross contamination between research experiments and ensure that new studies start with a pathogen-free space. These laboratories are required to use a reliable and validated process for whole space decontamination. 2 It is also of utmost importance that decontamination methods do not harm sensitive and complex laboratory equipment. 2
Understanding the challenges of various spaces within BSL-3 and ABSL-3Ag facilities is key to achieving proper decontamination and ensuring the microbiological security of the facility. Research performed at BSL-3 and ABSL-3Ag facilities requires the ability to house both plants and livestock of various sizes, while also encompassing the equipment necessary to study microscopic organisms. Among this range of laboratory spaces are interstitial spaces that are normally located adjacent to or above an adjoining laboratory, with varying degrees of separation from the laboratory. One particularly challenging interstitial space is located above the drop ceiling tile grid. The area above the ceiling grid, as well as the laboratory, is subject to contamination risk either from equipment use or procedures that may generate aerosols.
ABSL-3Ag facilities also contain a necropsy laboratory that is designed to facilitate the postmortem examination of infected livestock. Necropsy investigations make these spaces particularly susceptible to exposure to pathogens due to the possible aerosolization and overall presence of bodily fluids. The necropsy suite, due to the nature of the work performed there, is the largest space within a facility. This laboratory can have ceiling heights reaching 21 ft (6.4 m) and contain large laboratory equipment. The equipment present within the laboratory can create additional surface locations where contamination can occur. Effective decontamination of these two unique environments can be difficult to achieve, and as such an ideal decontamination system for these facilities would be able to handle both of these different applications.
The goal for BSL-3 and ABSL-3Ag facilities is to achieve a complete decontamination of research spaces. Indicators containing
Given the wide range of spaces needing decontamination, it is difficult to find a method that is both highly effective and extremely versatile to serve multiple areas within the facility. BSL-3 and ABSL-3Ag laboratories have historically used a range of decontamination techniques to address contamination concerns such as fumigated formaldehyde, chlorine dioxide gas, or, most recently, vaporized 35–59% hydrogen peroxide (H2O2).2,4 Although these methods are efficacious, they may also be damaging to equipment after many cycles, pose extreme exposure risks for staff, and in some cases require lengthy clean-up of residues left behind.2,4 Owing to the caustic and hazardous nature of these chemicals, their application must be constantly monitored, which can be both labor intensive and create additional risk for staff.
Introduction
The applications of fumigation as well as the challenges BSL-3 and ABSL-3Ag facilities face in achieving safe, consistent, and effective decontamination are well understood within the industry.2,4,5 Even as technologies for decontamination advance, the overall goals remain constant. Joslyn describes the ideal fumigant as one that should leave no residues or should be capable of rapid removal to safe levels after fumigation. 5 H2O2 disinfection technologies as a whole have demonstrated an efficacy equal to that of traditionally employed methods, 2 paired with advancements in their delivery systems.
The use of H2O2 systems in laboratories has been the subject of numerous studies and comparisons.2,4 Ample research demonstrates the efficacy of high concentration H2O2, using a variety of delivery devices. That prior research has highlighted both efficacy and ease of use of equipment as important measurements of a desirable system. 4 As a result, it becomes important to review both use and versatility of a system, relative to those methods that have been commonly applied, when considering its value to a laboratory facility. As H2O2-based technologies become increasingly widespread, questions remain as to the applicability and efficacy at varying concentrations. What has not yet been seen is whether or not advancements in lower concentration solutions paired with manufacturing that favors versatility can improve upon historically applied disinfection methods.
With this in mind, this study sought to challenge an advanced design Hybrid Hydrogen Peroxide™ (HHP™) system within the BSL-3 and ABSL-3Ag facilities at the Kansas State University Biosecurity Research Institute (BRI). The HHP system comprises a mobile 36-pound (16 kg) device paired with a 7% solution (CURoxide™). This solution is delivered by the device into the air as a hybrid mixture of vaporous and aerosolized particles. The device functions through pulse technology, which, after the primary injection of HHP into the space, accounts for the natural decomposition of H2O2 by periodically replenishing the HHP fog thus enabling an enhanced contact time. Although any fogged chemical requires some degree of attention to safety, this 7% solution falls below the hazardous chemical range of 8% H2O2 and well below the industrial use range of solutions >28% H2O2 common to traditional systems. 6 In addition, the optimal operating condition of the HHP system is ∼139 parts per million (ppm), in contrast to higher concentration systems that operate between 300 and 2,000 ppm. 7
As a safety measure, the system operates manually with a delayed start or remotely from an electronic tablet. Use of the tablet enables documentation of cycle parameters through the manufacturer software application (CURIS® App). Both control options contain emergency stop capabilities. For validation, commercially available carriers of
Overall, this study sought to determine whether one system could conquer a range of challenges. One specific interest was the ability of the device to deliver a solution capable of migrating around equipment, into adjacent interstitial spaces and attached rooms, as well as the ability to capably treat large open spaces with ceiling heights up to 21 ft (6.4 m). The HHP system was implemented in a manner consistent with the laboratory's existing protocols for gaseous fumigation systems, which includes the use of fans to disperse the fumigant. Although normally not required, the HHP system was operated under similar conditions as these other systems so as not to present additional variables when comparing the different technologies. Within the facility, two particularly challenging areas of the laboratory were chosen for this investigation.
The first study sought to decontaminate two laboratories along with their affiliated ceiling interstitial spaces. In addition to the challenges posed by access around equipment and into small spaces, these laboratories contained working biological safety cabinets (BSCs) that actively filtered the air during treatment. As the HHP system had previously demonstrated efficacy in this application, 8 this further analysis was intended to expand upon that study by adding this challenge to the overall goal of decontaminating the laboratory and its ceiling interstitial space.
The second study took place within the necropsy suite, one of the largest spaces within the facility. This study sought to discover whether the small low-profile devices were powerful enough to be used in a space with 21-ft (6.4 m) ceiling heights, walk-in cooler, a soft sided anteroom, two shower block change rooms, and additional equipment. This study also sought to determine the usefulness of this particular technology's ability to pair multiple devices for automated synchronized decontamination. In an effort to establish the range of operation, this study challenged the technology by using one fewer device than the manufacturer-recommended number for the 44,212 ft 3 (1,252 m3) space.
Collectively, these study components were designed to aggressively test the efficacy and versatility of the HHP system with the intention of discovering the system's ability to match or exceed traditional methods with high-concentration solutions in safety and efficacy to successfully decontaminate laboratories and their contents.
Materials and Methods
Study 1: BSL-3 Laboratories with Dropped Ceiling Interstitial Spaces
Study 1 tested the HHP disinfection system (CURIS, Oviedo, FL) in two inactive clean BSL-3 laboratories measuring 2,281 (65 m3) and 4,668 ft 3 (132 m3). Decontamination runs were performed separately for each laboratory. Both laboratories had standard ceilings and a lower dropped ceiling that consists of ceiling tiles in a metal grid that creates an interstitial space between the two. Environmental conditions ranged from 66.3°F to 75°F (19°C–24°C) and from 20.7% to 52% relative humidity, no conditioning occurred. Doors were sealed and HVAC bioseal dampers were closed during the decontamination process. At least four ceiling tiles were removed in each laboratory to allow for air flow into the interstitial space.
After initial testing and calibration runs, fog and pulse rates were adjusted to ensure H2O2 reached all areas of the laboratory. Consistent with routine BRI protocol for gaseous fumigation, at least two oscillating fans were added to the laboratories to create air movement, along with two or more blower fans directed toward the interstitial openings.
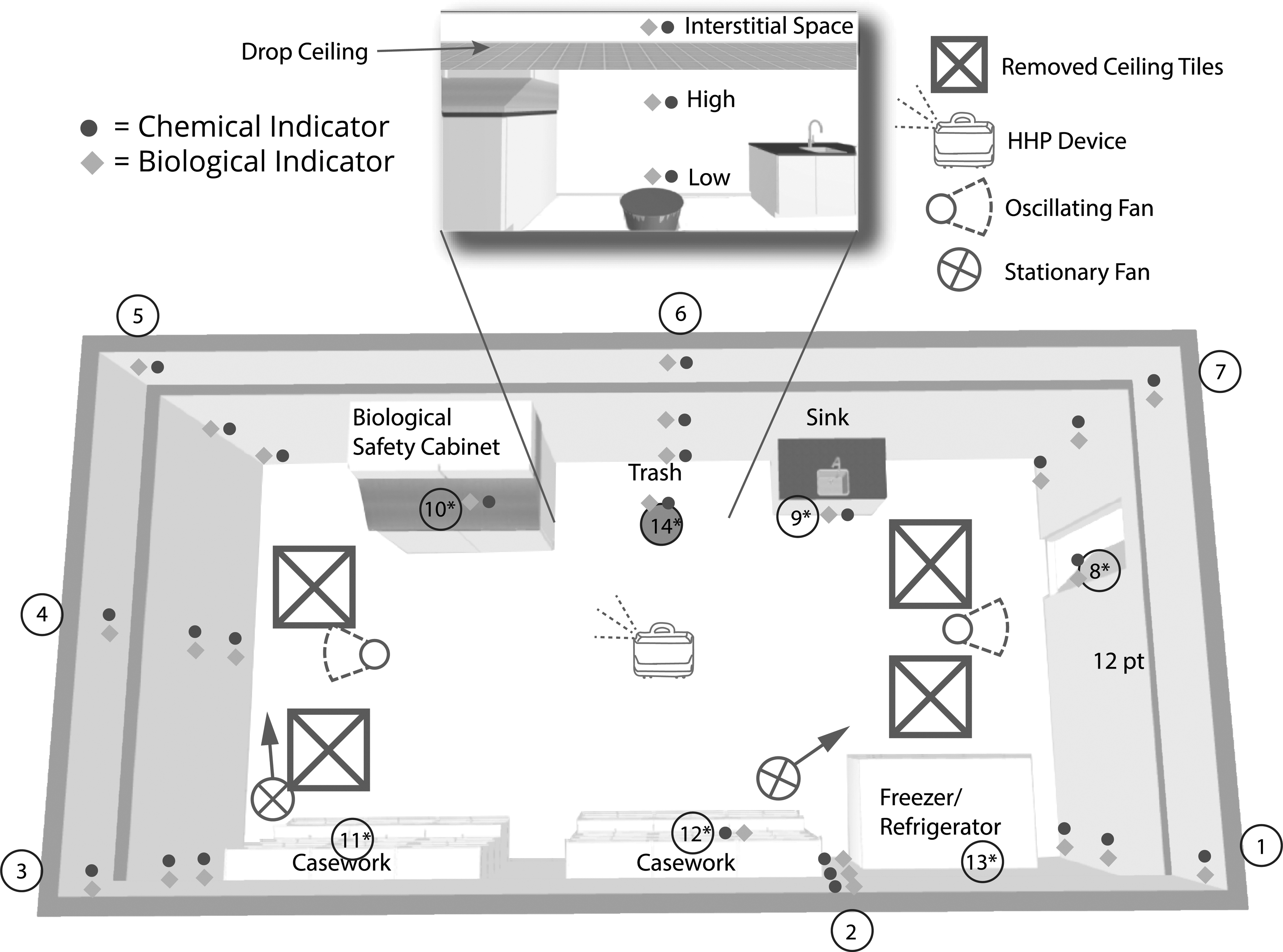
Diagram of Laboratory 1 (2,281 ft
3
[65 m3]). BIs of
One BSC in laboratory 1 and two BSCs in laboratory 2 were on and running for the duration. The HHP device was placed on the floor in laboratory 1 initially, with subsequent runs elevated, and elevated ∼36″ in laboratory 2 for all runs. Room dimensions, including the interstitial area, were preprogrammed into the HHP system's online data management system. Cycle parameters were determined by room volumes and accounted for the operational BSCs (Table 1). The HHP devices were operated remotely on a tablet through the accompanying CURIS App.
Cycle parameters
Parameters of the HHP system for each laboratory decontamination cycle.
BI, biological indicator; HHP, Hybrid Hydrogen Peroxide.
After cycle completion and aeration, all indicators were collected. CIs were analyzed for exposure. BIs were transferred into tryptic soy broth with pH indicator (Crosstex). BIs were incubated for 24 h at 60°C (140°F) per manufacturer's specifications and results were recorded (Table 2). Positive BI controls were used accompanying every run to confirm viability of the BIs.
Study 1 biological indicator results
BIs of
Study 2: ABSL-3Ag Necropsy Suite
Study 2 tested the HHP system in a clean inactive ABSL-3Ag necropsy suite, including a walk-in cooler, and a soft sided anteroom with two connected change rooms, for a total volume of 44,212 ft 3 (1,252 m3). Four synchronized HHP devices were utilized, each device accounting for 11,053 ft 3 . Environmental conditions ranged from 68.7°F (20°C) to 72.9°F (23°C) and from 28.8% to 29.3% relative humidity, no additional conditioning occurred. Exterior pressure-resistant doors and HVAC bioseal dampers were closed. Change room and soft sided anteroom doors were propped open.
BI and CI pairs were hung at three heights: one just above the floor, one midway up the wall, and one near the ceiling in multiple locations around the rooms, including three points on hanging outlet wires in the middle of the room. BIs were also placed on equipment and countertops (Figure 2). Seven fans were placed in the room and four HHP devices were positioned on tables ∼36 in (91 cm) from the floor. One fan was an industrial 36 in (91 cm) drum unit, placed in the center of the room pointing upward. Fans, fan placement, and HHP system elevation were included as part of routine BRI protocol. Dimensions for the entire laboratory suite were preprogrammed into the HHP system's online data management system.
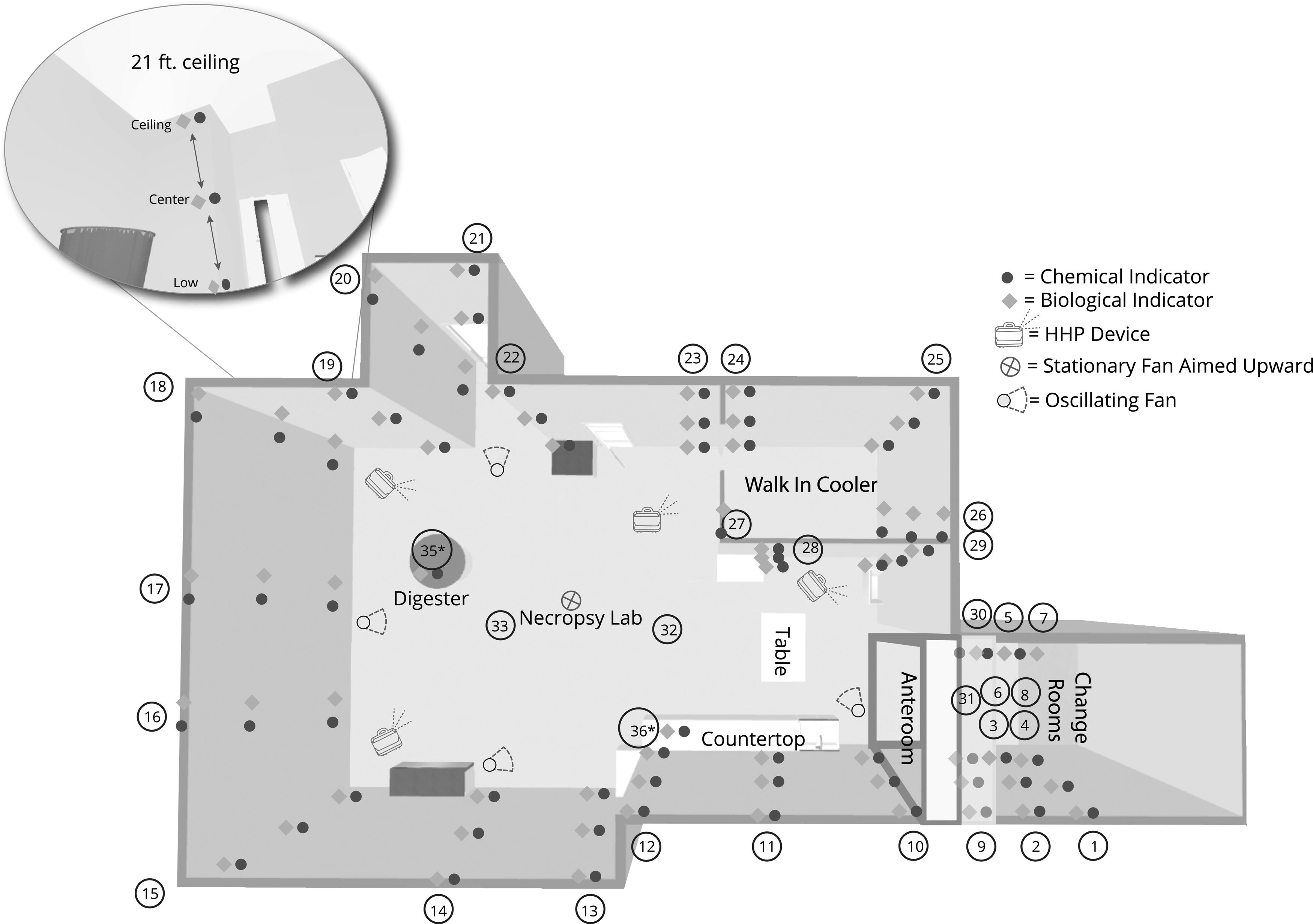
Diagram of the Necropsy Suite (44,212 ft
3
[1,252 m3]). BIs of
The HHP devices were synced and activated remotely through a tablet and the CURIS App. Figure 3 shows the HHP device at startup. Data generated from the HHP devices uploaded to the online data management system, and treatment reports were generated from that system. After the treatment, bioseal dampers were opened and laboratory doors unsealed for aeration through the building HVAC system. BIs and CIs were collected and processed as in Study 1.

Synchronized HHP devices at primary injection in the necropsy suite.
Results
Study 1: BSL-3 Laboratory with Interstitial Space
BIs were analyzed after incubation. All positive controls confirmed viability of the BIs. In the first laboratory, 30 BIs and CIs per treatment were used and demonstrated no bacterial growth for all 30 BIs. Over two treatments, all 60 BIs indicated a >6-log reduction of bacterial spores. CIs were analyzed after cycle aeration, and demonstrated a change in color, verifying the migration of HHP throughout the room (Table 2).
In the second laboratory space, 64 BIs were used per treatment. Over three treatments, no bacterial growth was observed for any of the 192 total BIs, indicating a >6-log sterilization of
Study 2: ABSL-3Ag Necropsy Suite
All CIs showed a change in color after decontamination, indicating adequate migration of the HHP fog throughout the laboratory space. After incubation, all 103 BIs were negative for bacterial growth, indicating a >6-log sterilization of challenged BIs. Testing was repeated a second time according to the described methods with the same results observed for a combined total of 206 indicators of
Necropsy suite biological indicator results
BIs of
CI, chemical indicator; H2O2, hydrogen peroxide.
Discussion
In decontaminating BSL-3 and ABSL-3Ag facilities, an ideal method is one that is highly efficacious, leaves no residuals behind, and lowers exposure risks for staff. As the internal rooms of laboratory facilities are made up of spaces of all sizes with a variety of equipment, the ideal system would meet these criteria while remaining powerful enough to overcome the inherent challenges. This study was undertaken to determine whether a highly portable HHP device would be capable of addressing the challenges of efficacy and usability while mitigating safety concerns by applying a lower concentration H2O2 solution with resulting low ppm.
First, two laboratories measuring 2,281 (65 m3) and 4,668 ft 3 (132 m3) with ceiling interstitial spaces including operating BSCs were treated. Testing and calibration runs were performed with minimal displacement of ceiling tiles to establish the decontamination cycle parameters. Fans were used consistent with the laboratory's existing protocol for gaseous fumigation. After this testing, subsequent cycles reported here adjusted primary injection and pulse timing and removed additional ceiling tiles to enhance HHP movement and account for the overhead interstitial space. Full exposure was then achieved by increasing access between the spaces.
As an additional challenge, the BSCs were turned on with the front sash open. As BSCs contain their own high efficiency particulate air filtration systems, this filtration had to be accounted for to not present a variable to disinfection. This was achieved using options within the HHP system's programming that increased the delivered dosage of solution into the space. This study expands on that of Ghidoni et al. that demonstrated simultaneous room and BSC decontamination. 8 The overall success of the BIs points to a successful decontamination and to the capability of the HHP system to achieve decontamination of laboratories and interstitial spaces, as well as within operating BSCs.
The second part of this study treated the 44,212 ft 3 (1,252 m3) necropsy suite with 21-ft (6.4 m) ceilings, walk-in cooler, soft sided anteroom, and adjacent change rooms. This large space required the use of four synced HHP devices, which operated through the CURIS App. Individually, each device accounted for 11,053 ft 3 (313 m3). Fans were used to disperse the HHP in a manner consistent with BRI protocols (Figure 2).
For both studies, confirmation of a >6-log reduction was achieved by means of commercially available
Along with efficacy, it is equally important to enable ease of use of the decontamination process without introducing risks in the form of chemical exposure. Historically, formaldehyde is used in concentrations ranging from 600 to 1,400 ppm, 4 and vaporized H2O2 systems with solution ranges of 35–59% have been shown to operate as high as 2,000 ppm 7 . Although these methods are applied following full safety protocols, there is still an inherent potential danger to an operating ppm that greatly exceeds the occupational safety and health administration permissible exposure limits of 0.1 11 and 1 ppm, respectively. 12 At 7% H2O2, the HHP system's lower operating concentration of ∼139 ppm 13 may reduce the risks associated with accidental exposure.
Owing to the fortified construction, maintaining connectivity with remote operated devices proved to be challenging in certain areas of the facility. The manual start option is one solution for this, however, creating a job report must then be done through the online data management system. For facilities that must maintain meticulous documentation for all decontamination procedures, the remote start through the CURIS App will be an important operation method if the device is to provide cycle data in an automated reportable format. Should the tablet lose connection during the treatment, connection must be re-established with that device to download the data.
Within the large necropsy suite, the decontamination process was enhanced by the ability of the four synchronized HHP devices to work simultaneously as a network, delivering the optimal amount of HHP fog needed. This particular design element enhances the versatility of the system when the goal is to achieve a >6-log reduction within large volume laboratory spaces.
Conclusion
This study demonstrated the HHP system's capabilities within the challenging environment of a BSL-3 and ABSL-3Ag facility. Building on these results, it would be of interest to investigate the system's ability to achieve success in more sensitive areas of the laboratory or as part of a response procedure for laboratory spills and accident protocols. As the HHP system has shown efficacy against Norovirus,10,14 SARS-CoV-2,
9
Overall successful results through multiple components of this study demonstrate that the lower concentration 7% CURoxide solution paired with the portable HHP device achieved efficacy regardless of variables in laboratory size and layout. Perceived challenges such as 21-ft (6.4 m) ceilings, active equipment, and difficult to access ceiling interstitial spaces were unfounded, even when using one fewer device than the manufacturer-recommended number, through incorporation of BRI protocols. As a result, the challenge of decontaminating large volume spaces coupled with high ceilings may not be as insurmountable as previously perceived. In light of the necessity of an efficacious system, without residues and with improved safety for staff, the results of this study demonstrate that decontamination with the HHP system is a viable option for BSL-3 and ABSL-3Ag laboratory decontamination in spaces of all sizes.
Footnotes
Acknowledgments
The authors thank Jodi Woodson and Tabor Grinrod for their contributions to designing the accompanying figures and tables.
Authors' Contributions
R.R.S., J.D., E.A.M., and A.T. carried out the decontamination setup, cycles, and processing of biological indicators. J.R.H. and R.R.S. conceived, planned, and supervised the decontamination testing. J.R.H. and M.H. wrote the article with support and feedback from all authors to create the final document. All listed authors contributed significantly to this study.
Authors' Disclosure Statement
M.H. is the senior research scientist for CURIS System. All other authors are employees of the Biosecurity Research Institute, Kansas State University.
Funding Information
No funding was received for this article.
