Abstract
Introduction:
Healthcare organizations are complex systems where healthcare professionals, patients, biological materials, and equipment constantly interact and provide feedback with highly consequential outcomes. These are the characteristics of a complex adaptive system. Healthcare delivery requires coordination but it necessarily relies on delegation of essential functions. It is thus essential to have an engaged workforce to ensure optimal outcomes for patients. Thus human performance factors play a key role in ensuring both the presence of excellent healthcare provision and the absence of outcomes that must be avoided—“never events.”
Methods:
The commitment of management was a precondition for the implementation of the high-reliability organization (HRO) principles. A team from middle management was engaged and provided with appropriate management tools for identifying, prioritizing, assessing, and applying solutions for the safety concern in their operating systems.
Results:
This article documents efforts at the National Institute of Health (NIH) to adapt the principles of HROs to diagnostic laboratories and vaccine production facilities at its campus in Islamabad, Pakistan, and seeks to draw some lessons for how this approach can be usefully replicated in such facilities elsewhere.
Conclusion:
Public health institutes such as NIH deliver vital products and services that are inherently risky to produce, where the consequence of failure can be catastrophic. Adopting the HRO principles is an approach to improving not just safety, but also the overall organizational performance in any setting, including low-resource settings, and can serve as an implementable process for other institutions.
Introduction
High-reliability organizations (HROs) have been studied for nearly 35 years. 1 These are organizations that operate in high-risk settings, such as airline companies, power grid stations, oil and gas exploration companies, and chemical industries where the slightest safety issue can lead to catastrophic effects and loss of human lives.2,3 However, this concept is now also being extended to healthcare systems, including hospitals, diagnostic and research biological laboratories, and vaccine production facilities, where error-free performance is obligatory.4,5 A key feature of HROs is the recognition that they are complex adaptive systems and hence it is impossible to predict all the ways in which the system can fail—despite the high consequences of failure.
The essence of HRO implementation lies in adoption of its five principles: preoccupation with failure, reluctance to simplify, sensitivity to operations, commitment to resilience, and deference to expertise. To achieve their results, HROs combine safety culture, workforce diversity, enabling organizational structures and management philosophy, and focus on developing key nontechnical interpersonal skills throughout the workforce. They deploy great effort to analyze processes and reduce the probability of errors and incidents and to prevent “never events” to promote patient care and safety in a hazard-free environment. 6 They also place greater emphasis on preparing for effective rapid response to the unexpected. This translates into HROs spending relatively more time building up the expertise of their personnel and relatively less time on planning and prevention than their non-HRO counterparts.7,8
Resilience for HROs means averting failure where possible, but ensuring that when it happens, failure is graceful (i.e., not catastrophic) and recovery is quick, keeping errors small and having business continuity plans in place. 7 They organize to immediately detect even the smallest variance from expectations, to identify, diagnose, and react appropriately to potential hazards before they can cascade into catastrophic failure. 9 This requires a highly delegated management structure to shorten learning feedback cycles, with the lowest level of operations having delegated authority to react to the unexpected and delegated responsibility for organizational learning. In turn, for this to work, the workforce must be armed with the nontechnical skills of situational awareness, effective decision making, communications, teamwork, and leadership. 10 Indeed, HROs organize to continually improve their processes. Thus accidents and failure are occasions for learning, not blame (unless the accident was the result of intentional sabotage or wilful negligence). Learning does not happen without reporting, and reporting will not happen if blame is attached to unintentional error.
The National Institute of Health (NIH), Pakistan, is an intricate organization with multidisciplinary public health activities. It is responsible for providing a wide range of preventive, diagnostics, and emergency healthcare services encompassing support for outbreak detection, investigations and response, vaccine and antisera production, food and drug testing, as well as research and development activities through standardized facilities and allied support. With such diverse operations, the institute has systems in place that are exceptionally consistent in achieving NIH's goals and avoiding systemic errors that can have direct impacts on public health. NIH's administration decided to instill HRO principles to further strengthen and improvise unerring operations, prioritize safety, and build a culture of reliability and the adoption of strategies for task completion.
Methodology
The principles of a HRO can operate in any organization by hardwiring certain habits, mindsets, and cultures into the fabric of the organization and then ensuring that everyone has the nontechnical skills required for effective implementation.
To do this, NIH with its partners, the Pakistan Biological Safety Association (PBSA), Fogarty International Centre (FIC), and Chrome Biorisk Management LLC, adopted a multistage approach in instilling these factors within NIH:
An initial kick-off meeting with senior executives at NIH that identified organizational objectives in launching this initiative, and that developed vision, values, and mission statements for the organization. The aim of this meeting was twofold: to obtain senior management buy-in and engagement in the initiative and to identify the members of the implementation team who would lead the subsequent work. A 1-week training workshop for the implementation team. The team was trained in the following: management systems, the principles of HROs, the organizational cultures required to implement high-reliability principles, the mindsets required to enable these cultures to take root and flourish, the nontechnical and interpersonal skills required to be effective in creating and maintaining a new culture of safety excellence, change management principles, and the design of appropriate metrics to measure the implementation and impact. Implementation team members were introduced to a suite of management tools, for modeling their processes (process mapping), hazard and risk identification (job hazard assessment forms, a risk analysis algorithm), prioritizing (Pareto analysis), planning change management (A3 Forms, Gantt charts), implementing change (RACIOS metrics), and measuring change (metrics). Furthermore, they were introduced to methods for engaging the workforce in the process, such as active listening. An implementation period led by the implementation team. During this period, bi-weekly conference calls were held with the partners to discuss both successes and find answers to any challenges encountered, to ensure that the process was kept on track. A wrap-up meeting to assess the impact of the initiative and to discuss ways forward.
Implementation Process at NIH
NIH in coordination with PBSA implemented the HRO approach from May 2018 to December 2018. NIH staff from each division participated in an initial workshop describing the HRO approach and later used the principles of HRO, implementing from May 2018 onward. The four divisions selected for implementation process utilizing the developed tools included the Biological Productions Division (BPD), Drugs Control and Traditional Medicine Division (DCTMD), Nutrition Division (ND), and Public Health Laboratories Division (PHLD). The operational activities of each division involve laboratory setup of biosafety levels 1 and 2, however, these laboratories are unique and different from each other in terms of functionality.
Biological Productions Division
BPD is responsible for the production of safe, potent, and effective quality biological products (vaccines and sera), keeping highest standards of integrity at all levels of production through environment protection agency and government hospitals. BPD is the largest division of NIH and is the sole public sector producer of quality bacterial and viral vaccines against tetanus, measles, rabies, typhoid & cholera, and allergy. It also manufactures therapeutic antisnake venom and antirabies serums. Moreover, oral rehydration salt is produced in this division. Each product is being manufactured in a separate building with specific staff. These products are tested in a quality control laboratory that has three subsections, namely, bacterial vaccines testing, viral vaccine testing laboratory, and one laboratory dedicated for chemical analysis.
Vaccine manufacturing is a complex system that generates several cognitive and physical challenges for the individuals working in the system. The purpose of HRO in BPD was not just to find the few potential problems that still exist in this journey of consistent quality production but also to deal with shortcomings in the system. 11 To ensure quality production, the division excelled in organization and management, documentation, data information ensuring adequate and competent personnel, quality control laboratory testing performance, training of new staff, supply chain management, infrastructure improvement, and improvement in interdepartmental communication. These quality assurance measures enhanced the core capacity and competency of the division; however, they were having issues in implementation and operations of biorisk management.
Drugs Control and Traditional Medicine Division
DCTMD is responsible for providing quality and innovation in test/analysis and research in the field of pharmaceutical, narcotics, psychotropic drugs, and complementary and alternative medicine. It is also responsible for providing advice to implement governmental policies, regulatory programs, and strategic objectives. This division includes the sole appellate laboratory of Pakistan doing drug testing. This marks its significance in terms of biosafety since testing is mandated and the decisions/results are authoritative.
Nutrition Division
ND conducts research on applied nutrition such as clinical nutrition and food analysis. It also undertakes nutrition education programs. ND is also responsible to advise the Federal government in developing food standards/laws in collaboration with the international agencies such as WHO/Food and Agriculture Organization and others.
Public Health Laboratories Division
PHLD is responsible to provide diagnostic services to the general public while taking into account the highest possible quality assurance norms and to function as a research and training hub for laboratory professionals. It is the second major division of NIH and performs public health functions in addition to diagnosis of infectious and noncommunicable diseases and provides grounds for major surveillance programs that exist in the country. It houses eight different laboratories namely cytogenetic, hematology, endocrinology, special chemistry, microbiology, water testing laboratory (WTL), immunology, and virology to complement its function.
Measuring Safety Metrics
HROs provide safety at the highest level and apply a particular method to achieve their results. To measure the efficiency and effectiveness of the HRO process, key ingredients for safety were identified and included for the evaluation. 8 In each section of respective divisions (PHLD, BPD, DCTMD, and ND), safety metric forms (SMFs) were distributed (Supplementary Data S1) for evaluating improvement in achieving the goal of becoming an HRO. These SMFs were customized according to the safety concerns of each section based on hazard and risk identification of the processes, and officers in charge were made responsible to carry out the tasks. Utilizing these forms, the methodology adopted included collection of real-time observational data on safety metrics specific to different laboratories for a period of 2 months for July and August 2018. To remove behavioral bias (such as compliance in front of authority), daily observations with no specific timings were recorded by laboratory supervisors or by senior technologists (compliance vs. noncompliance) based on the discrepancies observed in following standard operating procedures (SOPs). The SMF stipulates workforce responsibilities and general behavioral safety issues such as not using personal protective equipment (PPE) during procedures, not mouth pipetting, use of mobiles during bench work, eating and drinking habits in laboratories, and some general safety priorities such as laboratory design and improvements in workflow.
Pareto Analysis
To pinpoint the most probable safety issues or those that needed urgent attention among the multiple safety problems identified by each section, a statistical analysis known as Pareto analysis was done to help in decision making. The observations/number of counts was analyzed using the 80/20 rule for identifying safety issues. Pareto analysis or 80/20 rule is a technique for identifying 80% of the problems by addressing 20% of the mistakes or the “trivial many.” t-Test was performed to determine the inferential differences within the pre- and post-Pareto analyses.
A3 Forms
Another tool named the A3 form (Supplementary Data S2) was utilized for structured thinking about a problem. This serves as a communication tool for the workforce to report problems and improvement suggestions to the management. It is a well thought-out form with six steps to include: stating a problem, process mapping, root cause analysis, future state, implementation plan with time- bound activities, and audits of the expected results. Weekly meetings at the departmental level were arranged to explain the activities to be conducted and the issues faced by the team members. The management and implementation team then took steps for execution of the solutions. Officers in charge of their respective department conducted internal audits to ensure implementation.
Bimonthly Meetings
From August to December 2018, after every 2 weeks, Webex calls were conducted with the international consultant for discussing the issues faced by the workforce or at implementation steps and providing feedback on the progress made.
Resources
At implementation stage of HRO, some issues of resources were faced by almost all divisions that were supported by some parallel activities such as ISO certification. Quality Management System ISO-9001-2015 certification was achieved first time at NIH, Pakistan, in August 2019. The purpose of the quality management system in NIH was to ensure systematization, information as a basis for the continuous enhancement of quality, customer participation in system, involvement of leadership at all levels for the steering of quality, and risk assessment of all procedures for prevention of mistakes. It is during ISO 9001-2015 certification that all of NIH worked as one team to achieve this significant accreditation and those departments that had very basic level of documentation were also brought into the main stream.
The documentation system was practically implemented in each section. Trainings by the section heads were provided to their subordinates at the grassroot level for better awareness and implementation of the system. Workforce participation was also ensured by each department in form of worker feedback performas. During this implementation, leadership provided waste segregation equipment such as color-coded bins, color-coded bin liners, and sharp containers; laboratory coats, gloves, and goggles were also provided to departments. Eye wash stations and spill kits were ensured in each block. A system to report nonconformance was made mandatory in each section.
Results and Discussion
NIH implemented HRO principles in four of its divisions. While the functions of each division are different, all included the laboratory component with similar safety concerns. The new leadership of the institute realized that the true potential of the system was affected due to shortage of staff. After hiring new staff, there was a huge generation gap and wariness among employees due to poor communication, limited resources, and ambitious and energetic attitude of the newcomers.
Initially the junior staff faced recalcitrant behavior from the existing staff since they were overconfident about the safety issues. Staff assumed that nothing went wrong over the decades they did not follow SOPs or wear PPE. To build the relationship of trust and congeniality among the long-term and new staff, HRO tactics played an important role in prioritizing safety and building a safety culture.
Another aspect of not using PPE came forth in this process when leadership stressed the use of PPE, not just for process accountability but also for safety of the employees themselves. The workforce learned through repetitive trainings that PPE is for their own safety, however, low availability of PPE posed a challenge in most of the sections of almost all divisions. Gloves and laboratory coats were then provided to each section by the management. Encouraging the proper use of PPE was the next task for which different sections adopted various strategies for self-accountability to make it habitual before starting laboratory work. They placed daily charts upon which each laboratory worker was supposed to tick before donning and after doffing proper PPE. This is a perfect example of two-way communication and leadership involvement in solving the safety problems and providing what is necessary to adopt safe practices.
To assess the biorisk management, safety metrics performas were circulated in 10 laboratories of BPD and checklist of all laboratory workers was placed for accountability. Section supervisors were empowered to look for the problems and come up with ways to deal with them. Each section supervisor has provided training on general safety metrics performas, the reason why it was being implemented in sections, why safety is important for the individuals working in the laboratory, and the repercussion for unsafe behavior for the employees and for their families. Each section came up with its own observations, and after monitoring the staff for two consecutive months one major problem was selected for mitigation.
To build a safety culture, all section supervisors took different approaches. For example, in the cell culture rabies vaccine production laboratory, supervisors placed many rules to promote biosafety and ensured that the workers showed compliance. The strategies included a small tea time meal, held for all staff, only if they complied with the biosafety rules. Later it was implemented as a positive reinforcement for the workers. Anyone who showed noncompliance of biosafety rules would be responsible for organizing the tea time meal for the entire staff.
During this process, employees learned that HRO provides “Just Culture” with balance and accountability where discipline is equally applied to the seniors and departmental heads. 12 One such example came from measles vaccine production laboratory. Before entering into the laboratory, it is compulsory for all staff members to use laboratory shoes/shoe covers. To ensure compliance, a list of all laboratory workers was made and entry was only allowed after the checklist was maintained against their names on the list to document compliance. This acts as a scrutinizing step for the nonfollowers. On one instance, the head of the department in a hurry forgot to switch into normal shoes and was reminded by laboratory workers.
The promotion of trust, and not being punished encouraged workers to speak up. This helped in highlighting the incidences that were otherwise not reported. For example, in the bacteriology section of QCL, needle prick injuries were never reported before. Three incidences were reported and mitigation measures were taken. This communication system resulted in better incident reporting. 13
Figure 1 shows pre- and post-Pareto analyses of all 10 sections of BPD. In the first round of observations in BPD, waste management had a cumulative value of 80% and after intervention it was reduced. The figure shows that techniques and efforts applied by the officer in charges did bring positive change in the behavior of workers, and repeated refresher trainings regarding safety enhanced their skills set. The t-test score was 3.99, obtained after comparison of pre- and post-Pareto values of 11 indicators to further test the interventions (Table 1). p Value, p ≤ 0.03, was significant among the two groups. Implementation of HRO also results in an organization with skilled and knowledgeable workforce that is engaged in solutions to make their practices safe. 14 Another noteworthy role that HRO played in BPD was building a high level of communication and adaptability that mitigates the consequences of human errors. Additionally, safe techniques do not hinder the performance of the worker or the department but make them more prepared not only for prevention of adverse occurrences in future but also made them more efficient in monitoring each other and assisting those team members who need help.
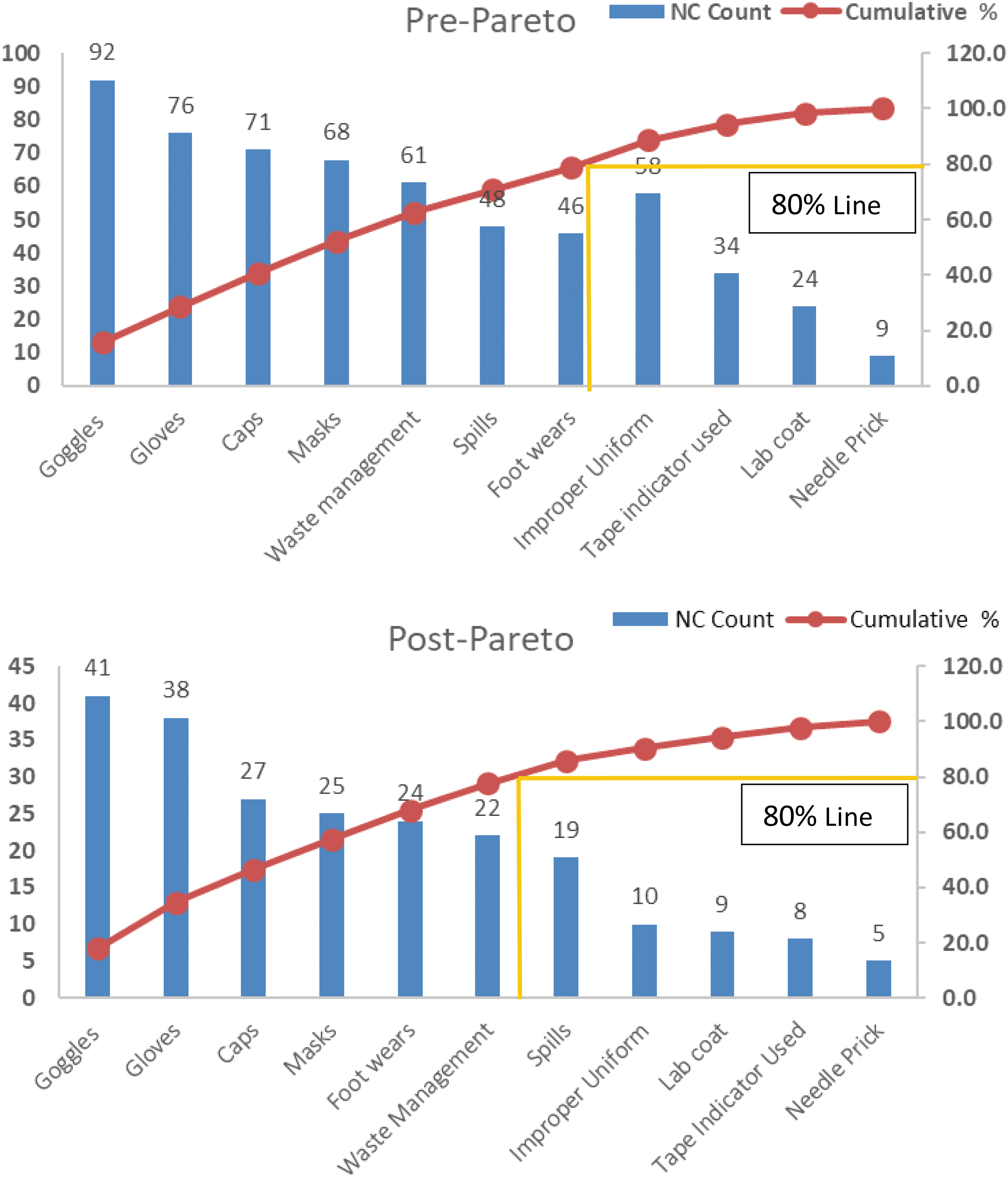
Pre- and post-Pareto analyses of BPD. The numbers in counts on y-axis represent the NC identified during the observational period. BPD, Biological Production Division; NC, nonconformity. Color images are available online.
t-Test for Pareto analysis for Biological Production Division
BPD, biological productions division; CL, confidence interval; LL, lower limit of confidence interval; M, mean; SD, standard deviation; UL, upper limit of confidence interval.
Drugs Control and Traditional Medicine Division
Three laboratories within the division were assessed and a gap analysis showed issues of organization and management, improper management of waste, sample management, equipment maintenance, lack of chemical inventory, and lack of compliance with biosafety practices. The microbiology section of DCTMD pointed out expired certification of a biosafety cabinet (BSC) as a safety hazard, which was then certified for operating in a safe manner. The chemistry section identified the irregular placement of chemicals and reagents, and that the chemical inventory was not maintained. Lack of training, carelessness of staff, and lack of space were the underlying causes of the problem. Provision of space, training of staff, placement of chemicals in alphabetic order, and segregation of volatile and nonvolatile acids/bases were some of the activities done to address the problem. Equipment maintenance and proper use were safety concerns of some sections. In the narcotics section, eating (in the lab) during working hours was regularly done and posed a huge safety concern. After doing the in-depth analysis, it was evident the workforce was unaware of the consequences, indicating a complete lack of training. They were provided with appropriate training and also given a separate room and refrigerator for storing food items. A very important factor of developing a safety culture is not just empowering the employees with resources, it is also empowering them with knowledge by providing opportunities for training. It boosts employees' satisfaction, their ability to perform challenging roles, improves performance, and develops a culture of learning and teaching. 15 Alerting employees to problems and engaging them in coming up with appropriate solutions enhances implementation in a timely efficient manner. Improvement in organization and management, provision of new equipment, training of human resources, management of samples, biorisk implementation, and documentation upgraded the overall functions of the division.
Nutrition Division
The microbiology laboratory, water chemical laboratory, and food analytical testing laboratory implemented HRO principles. Microbiology and water testing sections pointed out the noncompliance of PPE use, particularly during reagent preparation, sample analysis, media preparation, and filtration steps. In-depth analysis showed that personnel were not very aware of laboratory safety. There was casual behavior, lack of supervision by a designated person, improper record keeping of PPE, and scarcity of PPE. In-house training on biosafety was provided to improve the general perception of safety, ample PPE were made available by gaining the commitment from the chief of the division. Compliance was initially assured by designating a person for monitoring staff, and later peer to peer accountability was observed among the workforce. Record keeping and inventory maintenance of consumable supplies other than reagents were also emphasized to prevent misuse of PPE. Food analytical testing laboratory highlighted the issue of nonavailability of reagents/standards at the time of analysis. During process mapping, it was noticed that the standards were not prepared on time, their concentration and dates of preparation were not recorded and were not organized properly. The in-house biosafety training assisted in this regard by emphasizing on the adoption of SOPs and quality maintenance during procedures. A laboratory technician was assigned the responsibility of timely preparation of standard reagents, labeling the concentration and date of preparation and placing the solutions in a designated area. Regular monitoring by the officer in charge also encouraged the workforce to do their duties on time and efficiently. Improvement in internal communication, budget, biorisk implementation and documentation, record keeping, sample receiving and management, training of human resource, waste management process, and availability of PPE, upgraded the overall functions of the division.
Public Health Laboratory Division
Safety metrics forms were distributed to all departments/sections of PHLD to collect data regarding their safety concerns and hazard identification. After thorough evaluation, the laboratories of PHLD were strong in their public health function such as surveillance and response infrastructure and testing facilities, equipment, specimen collection, handling and transport, and finally reporting. The aspects that needed marked improvement were documentation and biorisk management. The reporting system was simplified and made more transparent by shifting to laboratory information management system from a manual system. Figure 2 shows the pre-Pareto analysis of the safety issues highlighted by different laboratories. Major safety concerns identified were mobile phone use in the laboratory, not using PPE for laboratory work, incorrect donning and doffing sequence of PPE, issues in waste segregation and disposal in some sections, logbooks maintenance, and access control issues. These matters were dealt separately and started off with providing basic biosafety training on PPE use. The sequence of donning and doffing of PPE was printed and pasted on notice boards as reminders. To encourage hand hygiene, the seven steps of hand washing were demonstrated and pictorial representation was placed besides the hand washing sinks. Clean areas were designated for mobiles placement during bench work, behavioral cues were also used but still the issues were not completely rectified. The biggest challenge in this regard was modeling behavior and providing an alternative. Having a schedule with “cell phone break,” restricted the workers from bring mobile phones to the laboratories rather than limiting its use to their desk areas only. Properly turning off equipment was also a concern in the virology department of PHLD and putting a sign on the equipment as a visual cue helped a great deal.
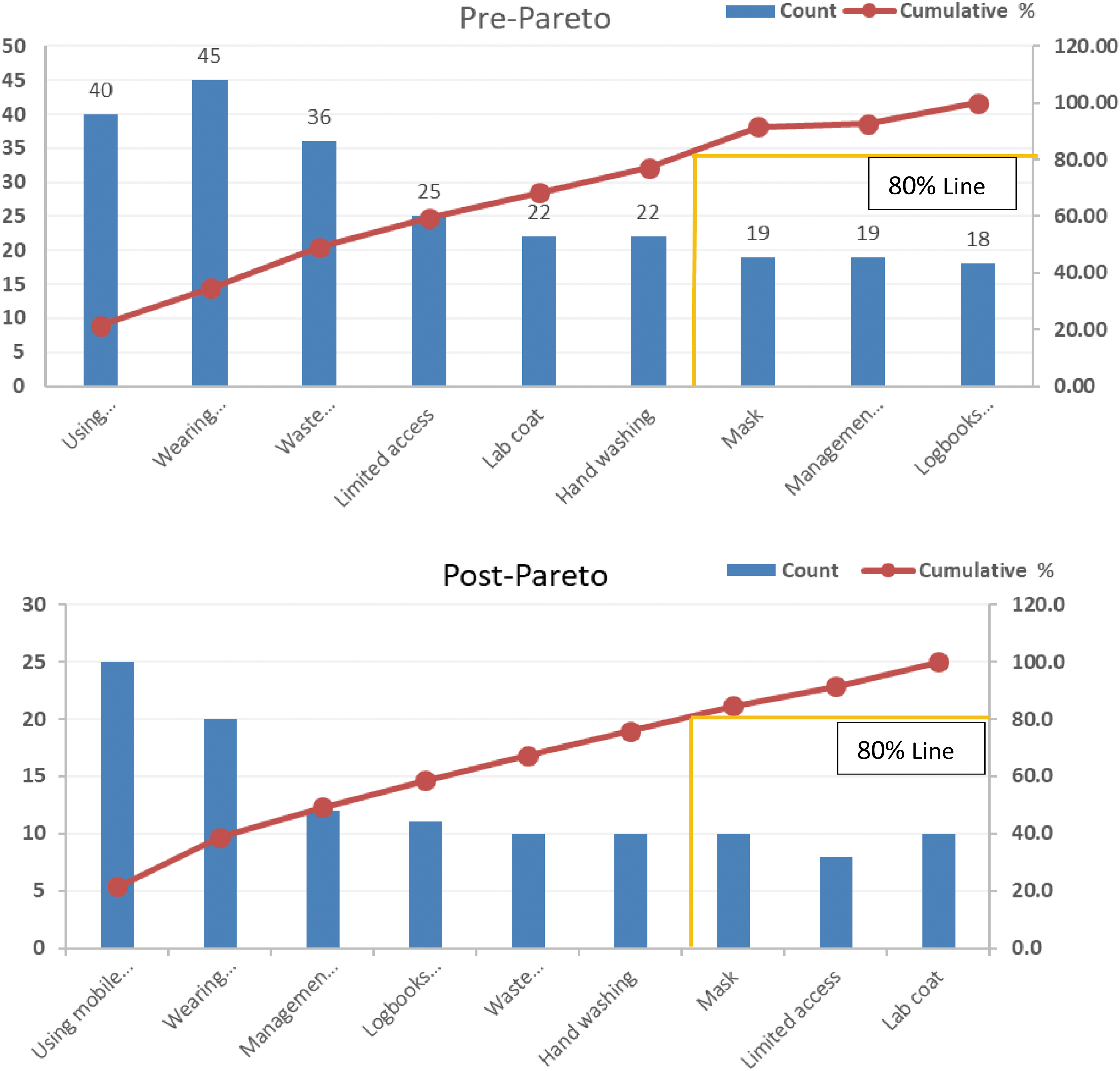
Pre- and post-Pareto analyses of PHLD. The numbers in counts on y-axis represent the NC identified during the observational period. PHLD, Public Health Laboratories Division. Color images are available online.
Figure 1 shows the improved counts in the safety concerns identified in different laboratories. Some concerns required approval from the head of the department for allocation of resources for certain activities. Examples include provision of PPE, color-coded bins for waste disposal, and placement of sterilized water bottles for sampling at reception of PHLD to minimize uncontrolled access. Duties were assigned to personnel and they were held accountable in case the duties are not performed in a timely manner.
Sometimes a solution lies in simplifying things. Changing the placement of equipment from clean to dirty work flow, can have a great effect on workflow and separating infectious and non-infectious processes. This approach was adopted in the immunology section of PHLD, where the same BSC was being used for multiple procedures such as cell culture and extraction of infectious material. Having separate dedicated BSCs for cell culture and extraction of infectious substances proved to be helpful and mitigated the risk of cross-contamination. Placing an additional table to reduce overcrowding of equipment on a single bench reduced potential electrical hazards. In the hematology laboratory of PHLD, assigning clear-cut job descriptions to different personnel reduced the chances of contamination, and all necessary tasks were completed on time. Initially the same person was responsible for bench work and data entry. For convenience and to save time, the technician used to do data entry without removing gloves. By assigning a person only for data entry, this problem was solved and the chances of contamination reduced by many folds. To elucidate our interventions, the daily noncompliance of safety concerns by laboratory staff was measured as counts on the safety matrix forms by the concerned officers in different sections of PHLD. These observations in counts were converted into meaningful data by pre- and post-Pareto analyses. Table 2 gives the comparison of pre- and post-Pareto analyses of nine variable indicators concerning safety issues determined in PHLD. Significant difference in scores was found (p ≤ 0.03) with 95% CL and a mean difference of 14.45. t-Test score for PHLD was 3.7.
t-Test for Pareto analysis of Public Health Laboratories Division
PHLD, Public Health Laboratories Division.
These are some of the perfect examples of how HROs operate with a robust process improvement and positive safety culture and aim for zero harm goals by recognizing early signs of failure. Safety is not limited to technical aspects of engineering, equipment, work processes, or safety system, but it roots down to nontechnical aspects such as enabling the organizational growth by providing equal opportunities for learning, situation awareness, assertiveness/conflict resolution, coordination, and cooperation. 10
Implementation of HRO methodology in different divisions of NIH allowed identifying the key variances necessary for situational analysis, recognizing the issues as they exist and self-educating how they can be resolved by learning from others' experiences to bring in line the planned level of safety. These undertakings run in parallel with the biorisk management system and ISO certification for gap analysis of the current operating system. However, certifications are more focused on documentations that need to be evaluated annually or biannually. The HRO approach is about changing behaviors and developing mindsets related to safety concerns and finding their own solutions. It enhances human performance and induces behaviors to avoid penalties by positive reinforcement making them very personal, the development of new behaviors meaningful, and more fluent and habitual.
Conclusion
Implementation of HRO characteristics at NIH was significant in revamping the organization's reputation and credibility and revitalizing a culture of safety and equity. It identified the vital features and crucial characteristics that required focus, to achieve the positive safety culture and HROs objectives. Through this process, we were able to implement the HRO principals effectively in 30 different departments of NIH. Investment on resources, training of staff on safety concerns, involvement of frontline employees, encouraging reporting without fear of blame, empowering the employees to take ownership of their achievements, acknowledging good efforts at management level, embracing failures and weak points by enhancing interactive sessions to build bottom to up communication, and simplified the reporting system were some of the key areas that required deliberation. Apart from evaluation, this process also allowed the team to recommend the improvement strategies and solutions to higher management as a result transition from an unmindful culture to a generative and maturity culture was reflected in the overall decorum of an institute and satisfaction of the employees.
It is the view of the authors that this is an approach to improving not just safety, but also overall organizational performance in any setting, including low-resource settings, and can serve as implementable design and approach for other institutes. The authors encourage other institutions to adopt the HRO approach to biosafety and also the development of a network of such institutions to facilitate cross-learning about effective implementation of HRO approaches in healthcare settings and to generate further improvements.
Footnotes
Acknowledgments
We acknowledge FIC, Bethesda, USA and Chrome Biorisk Management for designing the methodology for HRO implementation. We are also grateful to chiefs of PHLD, BPD, DCTMD, and ND and their respective teams for coordinating the activity in the respective facilities.
Authors' Contributions
S.T., S.S.A., T.T., and A.I. contributed to the conception or design of the study or the acquisition, analysis, or interpretation of data for the study; N.J., A.Z., R.M., Q.M.A., A.M., M.S., M.N., S.H., and I.S. drafted the study and revised it critically for important intellectual content; T.T. and A.I. involved in final approval of the version to be published; S.T. and S.S.A. agreed to be accountable for all aspects of the study in ensuring that questions related to the accuracy or integrity of any part of the study are appropriately investigated and resolved.
Ethical Approval
This study was approved by the institutional review committee of NIH, Islamabad, Pakistan, to evaluate the appropriate conduct of the research.
Authors' Disclosure Statement
No competing financial interests exist.
Funding Information
No funding was received for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
