Abstract
Introduction:
The biosafety level-3 (BSL-3) core facility (CF) at Yong Loo Lin School of Medicine (NUS Medicine) in National University of Singapore (NUS) has adopted international standards and guidelines to establish a biorisk management (BRM) system that helps to improve its BRM system and consistently minimize the risks to employees, the public, and the environment to an acceptable level while working with SARS-CoV-2.
Methods:
When the NUS Medicine BSL-3 CF started its operations, the Occupational Health and Safety Assessment Series 18001:2007 and the CEN Workshop Agreement 15793:2011 guidelines were used to establish its first BRM framework. The BRM framework provided the roadmap of how to organize, systematically manage, and structure the various biorisk programs that was then modified according to International Organization for Standardization 35001:2019 during the coronavirus disease 2019 pandemic in 2020 to address the specific circumstances.
Results:
Adopting a management system approach allowed BSL-3 CF to efficiently manage its BRM even during unpredicted emerging pandemic situations. It resulted in integrating a risk management process into daily laboratory operations and ongoing identification of hazards, prioritization of risks, and the establishment of risk mitigation measures specific to SARS-CoV-2. In addition, the implementation of a BRM system in the BSL-3 CF has increased biorisk awareness among BSL-3 CF users and encouraged every stakeholder to take ownership of their activities, and continual improvements in mitigation of biorisks.
Discussion:
This article summarizes the systematic approaches and major elements of the BRM systems adopted by NUS Medicine BSL-3 CF for the implementation of biosafety and biosecurity precautions, and control measures to minimize the risk of research activities using various RG3 biological agents including SARS-CoV-2.
Introduction
There is an increasing need for a harmonized approach in managing biosafety and biosecurity (biorisk) in laboratories due to emerging potential high-risk pathogens and increasing global public health issues. The current public health emergency surrounding the coronavirus disease 2019 (COVID-19) pandemic has resulted in setting up or expanding many research and diagnostic laboratories around the world to handle SARS-CoV-2. International standards and guidelines have been developed to assist laboratories in establishing biorisk management (BRM) programs. The International Organization for Standardization (ISO) 35001:2019 standard is the first such international standard specifically designed for BRM, published in November 2019. 1
A BRM program established based on a management system approach should provide tools to effectively identify, assess, control, and evaluate the laboratory biosafety and biosecurity risks associated with hazardous biological agents (BAs) and laboratory activities. 2 This article describes how a management system based on plan-do-check-act (PDCA) framework enabled the biosafety level-3 (BSL-3) core facility (CF) at Yong Loo Lin School of Medicine (NUS Medicine) in National University of Singapore (NUS) to establish the BRM program, build up a strong organizational BRM culture, improve the biorisk measures, and overall performance by implementing various policies, processes, and procedures.
The BSL-3 CF, a state-of-the-art facility, was opened in 2014. It supports the research, including animal research, and diagnostic work involving SARS-CoV-2 and a range of risk group (RG)3 BAs. The CF developed a system and processes to control the overall safety and security risks associated with the handling, storage, and disposal of BAs and other hazards in the facility. The BSL-3 CF's BRM framework was established since the start of its operations by adopting two well-known international documents (1) Occupational Health and Safety Assessment Series (OHSAS) 18001:2007 and the (2) European Committee for Standardization (CEN) Workshop Agreement (CWA) 15793:2011 Laboratory Biorisk Management.3,4 The CWA document (predecessor of ISO 35001:2019 standard) enabled the CF to effectively identify hazards, monitor, and control the laboratory biosafety and biosecurity risks.
The existing BRM program, including all necessary processes and procedures, was modified using the experience of CF team, to correctly identify the specific hazards and systematically manage the risks associated with SARS-CoV-2 work in BSL-3 CF, within in a short period of time during the recent COVID-19 pandemic. Some of the processes and risk mitigation measures were modified or relaxed after 2 months, when more information on SARS-CoV-2 was available. Various elements of the BRM program and how it was applied to minimize the risks while working with SARS-CoV-2 during the recent COVID-19 pandemic are briefly described in this article.
The Singapore legislation named “Biological Agents and Toxins Act” (BATA) and its subsidiary legislations/regulations provided some of the specific safety practices and security controls for handling of high-risk BAs and toxins listed under the BATA. 5 Adopting the risk-based and performance-oriented standards resulted in more efficient use of resources, more targeted controls to improve risk management, and the protection of people and environment. It also helped to clearly define the roles and responsibilities, and set clear expectations for CF staff and users, ensure compliance to legal obligations, and deliver consistent, harmonized, and improved services. The implementation of a sustainable BRM program allowed the BSL-3 CF to have better employee awareness and commitment to safety through a continuous cycle of performance evaluation, correction, and continual improvement of operations and processes to ensure that risks are consistently minimized.
BRM Structure of BSL-3 CF
The BRM system of NUS Medicine BSL-3 CF described in this article is built on the PDCA framework and it was initially adopted from OHSAS 18001:2007 (replaced by ISO 45001:2018), the CWA guidelines, and other Singapore regulations and guidelines,3–6 then modified based on the ISO 35001:2019 standard. 1 The PDCA model is an iterate process used to achieve continual improvement of BRM system for laboratories and its requirements through a cycle of planning, implementing, reviewing, and improving the processes. Figure 1 illustrates the PDCA framework of ISO 35001:2019 (which is a reorganized version of PDCA framework of CWA guideline and OHSAS 18001 standard with same concept) and how it relates to other elements of the BRM system of BSL-3 CF and this article (adapted from ISO 35001:2019). The elements of modified ISO 35001:2019-based BRM system aimed for continual improvement and it is similar to the CWA document except for the new component named “support” that connects the PDCA cycle elements (Figure 1).
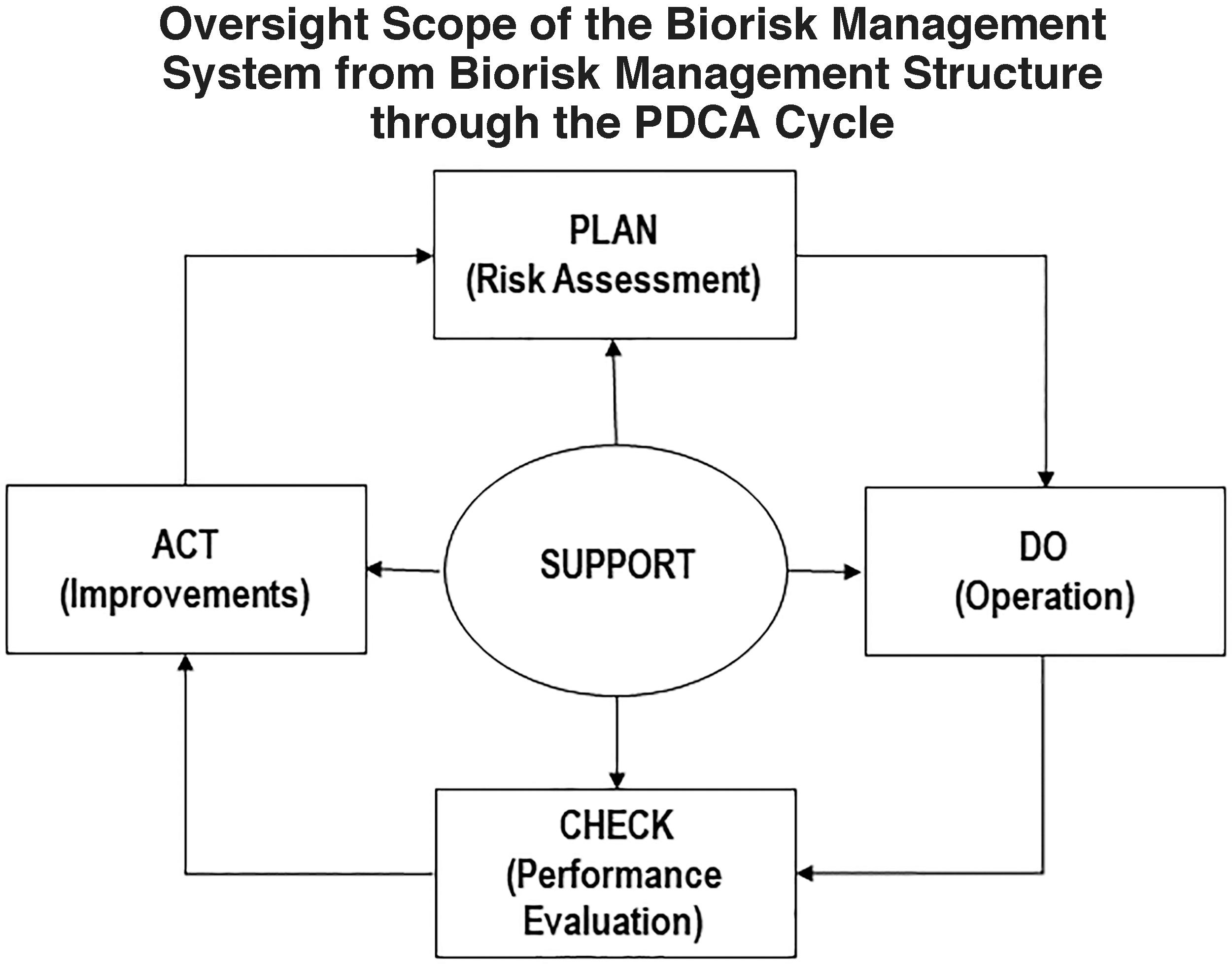
The PDCA framework of BSL-3 CF. BSL-3, biosafety level-3; CF, core facility; PDCA, plan-do-check-act.
Leadership
Strong leadership support is vital for ensuring the success of a BRM system in any organization. The leadership must provide the directions, empowerment, and resources needed for the establishment, implementation, maintenance, and continual improvement of the BRM system to control biosafety and biosecurity risks. The NUS Medicine's leadership has provided necessary resources and empowerment to CF staff to support a safe working atmosphere in BSL-3 CF. The senior management of NUS Medicine has taken up the role of “Facility Operator (FO)” (Table 1) of the BSL-3 and they had delegated some of their roles to the director (BSL-3 director reports to the dean of NUS Medicine) and operation team of the BSL-3 CF. The BSL-3 CF policy is endorsed by the director and it is displayed at the facility entrance. The operation team updates the senior management of all major activities and milestones of CF on a regular basis.
The key roles, responsibilities, and accountabilities of various personnel/team/committees of biosafety level-3 core facility are listed as per Biological Agents and Toxins Act and core facility policy
BA, biological agent; BATA, Biological Agents and Toxins Act; BC, biosafety committee; BCO, biosafety coordinator; BRM, biorisk management; BSL-3, biosafety level-3; CF, core facility; FO, facility operator; NUS, National University of Singapore; OH, occupational health; PI, principal investigator; PIN, personal identification number; PPE, personal protective equipment; SOP, standard operating procedure.
The commitment and support from the NUS Medicine senior management were evident during the COVID-19 pandemic. After consulting the BSL-3 CF operation team, the senior management had a meeting with all BSL-3 users to discuss and inform them the decision to stop all ongoing non-COVID-19 research work in the BSL-3 to allow research and diagnostic work with COVID-19 samples. In addition to funding the additional expenses of BSL-3 CF due to the COVID-19 pandemic, the researchers who were working with COVID-19 samples were provided with dedicated accommodations in the campus to protect them from potential community infections. Funding was also provided to BSL-3 CF to form a dedicated COVID-19 research team to support the scientists and clinicians. The operation team was empowered to take decisions when necessary to deal with operational crisis and emergency situations related to COVID-19/SARS-CoV-2 work in BSL-3 CF. Employees who are empowered by the leadership feel personally responsible for safety and security, and this helps the entire organization to identify and mitigate the risks proactively. They also learn what the accepted practices are in an organization by following the examples set by their leaders (e.g., clearly articulating the safety norms and visibly demonstrating that the safety concerns of the CF users are addressed).
Policy
The safety and health management policy for NUS Medicine BSL-3 CF was developed and documented with the intension of proactively controlling the risks, ensuring occupational health (OH) and safety of personnel and continuously improving the overall BRM system and its performance. It highlights the need for effective BRM that integrates all operational requirements, to effectively inform and ensure safety and health of all BSL-3 CF users, staff, and relevant third parties, and communicate individual obligations with regard to biorisk to those groups. It also articulates the scope and a framework for setting up the BSL-3–specific objectives for the implementation of the BRM system. The BSL-3 CF policy is committed to comply with all institutional and national requirements, protect laboratory personnel, community, and environment from biological hazards. The policy is also aligned with safety policies of NUS and the NUS Medicine. This policy also reflects the commitment of NUS Medicine's leadership to the BSL-3 CF, and roles and responsibilities of various personnel involved.
Roles, Responsibilities, and Authorities
The leadership of NUS Medicine has delegated specific roles and responsibilities to various personnel or committees as deemed appropriate. The responsibilities for relevant roles are also communicated within the facility including those who manage the facility day-to-day operation, perform research (users of the facility), implement biorisk measures, and verify the work associated with control of biological materials (biosafety coordinator [BCO]). The key roles and responsibilities of the personnel/committees (FO, biosafety committee [BC], and BCO) for BSL-3 CF are defined by BATA 5 and CF policy, and are summarized in Table 1.
Laboratory BRM is a collaborative effort and, to be successful, should have commitment and participation of all stakeholders (internal and external) involved. The appropriate breadth of collaborations will depend on the type of hazards, nature of work activities, and the complexity of the laboratory facility. External collaborators may include contractors, regulatory agencies, first responders (e.g., fire, police, and emergency medical personnel), and hospitals or other healthcare facilities (OH clinic).
There are a number of national legislations/regulations that also play a major role in influencing the BRM practices. The legal framework in Singapore guides the FOs, BC, laboratory managers, and ultimately laboratory workers to take responsibility in developing and implementing the necessary safeguards. Aside from relevant rules and regulations, biorisk control measures have been implemented as part of NUS requirement or BSL-3 CF requirements. Some of the most relevant national legislations and guidelines are listed in Table 2.
The list of relevant national regulations/guidelines that impact the biosafety level-3 core facility for the implementation and maintenance of the biorisk management system
COVID-19, coronavirus disease 2019; GMOs, genetically modified organisms; IPA, Infrastructure Protection Act; NACLAR, National Advisory Committee for Laboratory Animal Research; WSHA, Workplace Safety and Health Act.
Planning (the Plan Phase in PDCA)
In this first step of the PDCA cycle, the BRM system of BSL-3 CF identified various hazards through risk assessment (RA), risk mitigation, and communication strategies, and determined the legal and other requirements (internal/institutional and external) to operate the BSL-3 facility and work with RG3 BAs. Much emphasis was placed on RA as mandated by the Singapore Ministry of Manpower's Workplace Safety and Health Regulatory Framework and BATA.5,6,16 The risk management has been integrated into daily operations of the CF, and the process of identification of hazards and risk mitigation of protocols are tailored and prioritized to address any new/specific situations. For the work involving highly pathogenic or contagious BAs (e.g., highly pathogenic avian influenza virus), dedicated suites with additional controls such as showering upon exit and monitoring of the daily body temperature of the users are used.
RA and Risk Communication
The RA process includes identification of various types of hazards associated with the work in BSL-3 facility with emphasis placed on biological hazards associated with SARS-CoV-2. Various international guidance documents and the ministry of health (MOH) guidelines with standard precautions or good laboratory practices for handling of COVID-19 samples9,17–26 are referred to during the RA. RA is performed by a team consisting of facility personnel, scientists, and biosafety professionals. The RA team lists the various types of routine and nonroutine activities and the hazards associated with each activity are identified and documented. The RA includes the following steps: (1) identifying the hazards associated with the infectious agent or material, (2) identifying the category of hazards including physical situation and work activities, equipment, other types of materials (e.g., chemicals), or people involved (e.g., immunocompromised). For evaluating the risk, a quantitative method using a 3 × 3 matrix that the NUS uses has been adopted. 27
The RA for BSL-3 CF is mainly sorted into four categories. (1) RA for routine activities led by the researchers, (2) RA for routine activities performed by the facility operation team (e.g., regular maintenance of personal protective equipment [PPE]), (3) RA for nonroutine activities led by the facility operation team (e.g., equipment maintenance work by a contractor), and (4) RA for specific research protocols led by the researchers (e.g., administration of SARS-CoV-2 into animals). The institutional and regulatory requirements and biosecurity aspects of activities involving SARS-CoV-2 and other RG3 BAs are also considered in the RA.5,7,12
The RAs are subjected to BC's review, deliberation, and approval at the regular BC meetings. The outcome of the RA and risk indicators including incidents and near misses are effectively communicated to all relevant personnel by the BCO. The principal investigators (PIs)/supervisors are responsible for communicating specific hazards and risks associated with their work to their staff/students. A regular review of the RAs is required by the BATA and Workplace Safety and Health Act. The RAs of the BSL-3 CF are reviewed every 2 years, or earlier when there are new activities or laboratory incidents, change/introduction of new regulations, and/or significant alterations to standard operating procedures (SOPs). The RAs are reviewed by the BC, operation team, the PIs and their team members to ensure the sustainability and effectiveness of biorisk control measures implemented in CF.
Legal and Other Requirements Relevant to BSL-3 CF
In addition to the international standards and guidelines, national legislations and guidelines also play a major role in influencing the BRM practices for handling this novel virus at laboratory level.5,12,17 The BSL-3 CF has adopted all applicable national legal framework supporting BSL-3 laboratories; involved the regulatory agencies (e.g., MOH biosafety branch who is responsible for administering the BATA) in discussions; and set the goal of managing potential biorisks; and appropriately addresses, manages, and minimizes the risks associated with BAs such as SARS-CoV-2.
The MOH has established/amended the legislation and guidance documents9,17 that stipulate the control measures that must be implemented by each laboratory for storage and handling of SARS-CoV-2. The MOH has classified and grouped pathogenic microorganisms into different Schedules and this novel coronavirus is placed together with
Risk Mitigation
The risk mitigation strategies for biorisks associated with SARS-CoV-2 work in BSL-3 CF are based on RA. The RA process at planning stage recognized a hierarchy of controls, beginning with the elimination or reduction of hazards, then progressed to implement the appropriate engineering and/or administrative controls along with other operational controls to address residual risks, and identified as well as implemented appropriate PPE to protect the users. As mentioned in the above paragraph, the SARS-CoV-2 work involving live virus is performed in the BSL-3 CF that is a Protected Place with additional physical and administrative security measures.
Engineering controls
Facility infrastructure, primary barriers, and other engineering controls were identified during the design phase and implemented during the construction stage of the CF to ensure alignment with international 1 and national standards/regulations.5,12,28 All facility barriers are maintained and monitored through a building automation system without any disruptions. The facility is fitted with inward airflow from the cleanest areas to areas where BAs are handled, with additional engineering controls and precautions such as high-efficiency particulate air (HEPA) filtration on the exhaust air to mitigate the potential risks to personnel, surrounding community, and the environment. Engineering controls are adopted for all work involving handling of infectious agents/materials including SARS-CoV-2, and are performed inside Class II biological safety cabinets (BSCs). Other biosafety controls such as enclosed leakproof containers for sample movement within the facility, biocontainment cages fitted on animal racks with HEPA filter system for housing infected animals, and centrifuges with sealed rotors and safety cups are used depending on the RA.
Administrative controls
There are a number of common SOPs and a comprehensive laboratory safety manual that include all administrative controls for the CF. Examples of information covered include but are not limited to entry and exit procedure, access control, facility emergency response plans, incident management, implementation of procedures, and overall oversight of the facility. Administrative controls also include research-specific controls and facility maintenance program. For SARS-CoV-2 research work, the BSL-3 CF relies on a combination of engineering controls, additional administrative and work practice controls, and PPE for their safety mitigation measures. Some of the additional risk controls for those who are working with SARS-CoV-2 were showering before exiting the CF after their work, getting the influenza vaccination, (before the COVID-19 vaccine was available), and wearing a pair of disposable waterproof sleeve covers over the disposable gown for working inside BSCs.
Biosecurity risk controls
The most effective control measures are implemented for mitigating the risks identified by the RA for the BAs possessed and used in the facility to prevent unauthorized access and deliberate release of those agents. Additional administrative controls and policies are established for a minimum of two personnel to be present in the containment area at all times (i.e., a buddy system) for biosafety and biosecurity purposes. The biosecurity control measures of the BSL-3 facility include information control, personnel control, physical security, transport control, pathogen accountability, and dual-use research concern.5,12,13,29 Under the BATA, any BSL-3 facility possessing high security risk (first schedule) BAs including SARS-CoV-2 in Singapore needs to be gazetted as a Protected Place and ensures compliance with security requirements under the Infrastructure Protection Act. 12 All BSL-3 CF users are required to clear the preplacement security/background check and attend the mandatory biosecurity training conducted by the BSL-3 CF team. All information regarding the Protected Place, inventory of BAs, and other information is kept with controlled access. The BSL-3 CF is also supported with security cameras and video surveillance system.
Support
Human Factors
Behavioral factors
It is believed that majority of work-related accidents are due to employee/laboratory personnel behavior. 30 There are many reasons why employees face accidents at work including but not limited to the procedures, health factors, and misunderstanding/miscommunication of risks. Fatigue, complacency, stress, and high work demand at work also may contribute to laboratory incidents and accidents. Leadership behavior and organization culture also have an impact on what the behaviors laboratory personnel adopt and prioritize. 31 The BSL-3 CF operations team closely monitors individual users and acknowledges their best practices and put in effort to remove or reduce unsafe acts by limiting the working hours and having a buddy system. The behavior of each user is monitored by observing unsafe conditions and compliance to the BSL-3 procedures during inspections, the training process, and interactions with the staff and other users of the CF. All newly trained and certified users are required to work with a mentor for the first 10 times before they are allowed to work independently together with their buddy. In addition, all users need to attend quarterly refresher trainings conducted by the CF operation team.
Personal heath factors
Personnel with special health conditions such as immunosuppression are considered in the RA. They are required to inform NUS OH physician about the condition. As required by the BATA and NUS OH program, all personnel working with BAs in the BSL-3 CF including SARS-CoV-2 are enrolled under NUS BSL-3 OH program. The OH program includes undergoing medical surveillance and vaccination (if available) and seeking medical advice. The medical surveillance program comprises a baseline medical evaluation before BSL-3 work and thereafter on an annual basis. When the researcher stops working in the BSL-3 CF, they are advised to undergo an “exit” medical evaluation to ensure that he/she has not developed a medical condition associated with working in the BSL-3 laboratory. In the event of any potential exposure to BAs, the exposed personnel will be sent to the emergency medicine department (EMD) in the nearest hospital and subsequently followed up with the NUS OH clinic. The EMD offers emergency and inpatient medical services to the personnel for the list of BAs that are previously made known to them by the BSL-3 CF.
Competence of the personnel
All personnel working in the BSL-3 CF are required to undergo a mandatory BSL-3 training program and be certified to ensure competency before starting their work. The supervisor, the BC, and the operation team review and agree on the training needs for each individual. The training includes the policies, regulatory requirements, risk management, operational and research SOPs (e.g., use and maintenance of PPE and transfer of BAs), and emergency response procedures (ERPs). The main routes of exposure of the BAs (e.g., SARS-CoV-2) handled in the facility, potential route of transmission to humans, common signs and symptoms of the infection, and other information are also included in the training. For those who are certified as competent, there is a follow-on training on specific laboratory procedures, practices, and techniques, and on various SOPs to ensure that they have achieved the expected work performance requirements, and thorough understanding and knowledge in biosafety and biosecurity aspects of their work.
Regular drills and exercises are also conducted for ERPs with the BSL-3 CF users and other relevant parties. Some of the drills are conducted together with NUS rapid response team (RRT), NUS campus security, MOH officers, and Singapore civil defense force (SCDF). A typical example of such drill will include a causality due to a fire in the building or a major accident in the CF and another scenario involving spillage or exposure to BAs by a BSL-3 CF user. The responders of this emergency drill should inform relevant parties (RRT, campus security, ambulance, and SCDF), rescue the casualty, and bring them to the ambulance. At the same time, other responders should accompany the exposed personnel to the nearest EMD following the CF SOP. At the end of the drill, all stakeholders will meet up for a debriefing and to discuss and document areas for improvement.
Operation/Implementation (the Do Phase in PDCA)
The implementation of a BRM system integrates both biosafety and biosecurity practices. The risk control measures and operational practices identified by the RA have been implemented in the BSL-3 CF. Some of the examples of operational controls to address biosafety and biosecurity aspects that are relevant for SARS-CoV-2 work are listed hereunder.
Transfer, Storage, and Tracking of BAs
Samples are packed and placed in secondary containers for transfer within the facility. Trolleys and transfer baskets are also used if more than one sample is transferred at a time. For local transfer and transportation of BAs (transfer between buildings by walking and for samples that are sent out through vehicles), the BATA (transportation) regulation is followed. 5 The BAs are contained in a watertight and leakproof primary receptacle. Each receptacle is individually wrapped in absorbent material of sufficient quantity so that in the event of a breakage of or leakage from the primary receptacle, all fluids are absorbed. Primary receptacles are then packed in a secondary receptacle and transferred/transported in an upright position. The name and the emergency contact number of the person who is responsible and has the knowledge of the BAs are displayed on the outer package containing the BAs. All BAs including COVID-19/SARS-CoV-2 samples are stored in dedicated fridges/freezers that can be locked and with restricted access inside the BSL-3 CF. The BA inventory with description of the BAs, its quantities, storage location and use, the person responsible, documentation of every use or disposition, and internal and external transfers is kept up to date. The inventory of BAs is secured with controlled access by authorized BSL-3 CF personnel.
Disinfection/Cleaning Procedures for Equipment and Instruments
Based on the RA and the BAs handled inside the BSL-3 CF, appropriate disinfectants are used for the respective BAs. For example, PREempt and 4% Melsept® SF32,33 are used for most of the BAs including SARS-CoV-2 for decontamination because of their broad spectrum of antimicrobial activity. Ethanol (70%) is also used for disinfecting the workspace, equipment surfaces (e.g., BSC, microscope), reusable PPE, and other small equipment or tools. The efficacy of the mentioned disinfectants for decontaminating SARS-CoV-2 was tested and confirmed in the BSL-3 CF.
Maintenance, Calibration, and Certification of the Facility and Equipment
The facility has a program in place to ensure all equipment and other facility engineering systems are maintained, calibrated, and certified according to the regulatory/institutional requirements. All BSL-3 facilities in Singapore need to be certified by the MOH on annual basis. The certification process is done by an independent facility certifier registered with the MOH, whose certification criterion is based on the MOH checklist that was derived from the third edition of WHO Laboratory Biosafety Manual Certification checklist and BATA requirements. 5 Before the certification, the facility is thoroughly cleaned and fully decontaminated using vaporized hydrogen peroxide. Maintenance and calibration work occur before certifiers entering the laboratory to conduct the certification inspection and testing. Some of the CF equipment maintained, calibrated, or certified annually are centrifuges, BSCs, autoclaves, animal holding racks, and ultralow-temperature freezers.
Waste Management
There are different types of wastes generated in the BSL-3 CF. Contaminated biological waste materials are mainly segregated as solid and liquid wastes. They are packaged and decontaminated with the appropriate method that is approved by the BSL-3 BC. All solid wastes and liquid wastes (except hazardous chemical wastes that cannot be autoclaved) are autoclaved and then sent out for disposal through licensed hospital waste collecting agencies. 13 Some of the liquid wastes may be autoclaved and others are treated with appropriate chemical disinfectants before disposal in the sewer system. Each autoclave cycle for various types and quality of wastes, and a maximum load were validated and verified regularly using a biological indicator at different frequencies (at least twice a year) depending on the need.
Clothing and PPE
Before entering the containment area of the facility, all personnel are required to change from street clothes to laboratory scrub suit and put on other BSL-3 PPE that includes two layers of gloves, socks, shoes, head cover, powered air purifying respirator (PAPR), a long sleeved disposable surgical gown, and shoe covers (knee length). All these PPEs are stored in the facility clean changing area. Additional pair of disposable sleeve covers is worn to ensure coverage of the wrist to minimize contamination while working inside the BSCs with certain pathogens (e.g., SARS-CoV-2). When exiting the BSL-3 suites, the disposable gown, shoe covers, and outer layer of gloves are disinfected by spraying PREempt and removed after 5 minutes in the anteroom following the CF SOP. The PAPR is also sprayed with PREempt and thoroughly cleaned before exiting the anteroom. Shoes are taken off at the clean corridor, just before entering the dirty side of the changing room, and the PAPR, hair cover, socks, and the scrubs are removed in the dirty changing room. The PAPRs that are already sprayed with PREempt are stored in the pass-through PAPR cabinet that can also be opened from the clean changing room. The scrubs are then placed in biohazard bags for it to be autoclaved and sent for laundry. All personnel working with SARS-CoV-2 are also required to shower after removing the PPE, before leaving the facility.
Emergency Response Procedure
The BSL-3 CF has implemented and maintained ERPs to address unintentional or intentional biosafety and biosecurity incidents or emergencies. All incidents including near misses are reported using the NUS online system. The facility team investigates and records the outcome of each incident. Some of the incidents/accidents (e.g., exposure to BA in the laboratory, incidents leading to medical leave) may need to be reported to MOH and also ministry of manpower depending on the nature and severity/classification of the incident. All personnel working in the BSL-3 facility are trained in ERP SOPs depending on the nature and scope of their work and the incident reporting processes.
There is a BSL-3 emergency response team (ERT) consisting of trained and experienced personnel to support the users during any adverse situations. In case of emergency situations in the facility, the ERT member on duty will initiate the necessary actions and respond to incidents and emergency situations accordingly. Personnel are trained to inform the BSL-3 ERT and evacuate the laboratory in the event of an emergency or if any personnel are exposed to the BAs (e.g., if there is a major spill outside the BSCs, a fire in the facility). If there is a causality, the ERT will inform the EMD and send the causality to EMD accompanied by a responsible person. Emergency drills and awareness trainings are also conducted to ensure careful coordination between facility staff and first responders to handle medical emergencies and other building/facility emergencies.
Documentation and Document Control
BSL-3 CF maintains all records and documentations associated with its policies, plans, procedures, operations of the CF, and performance monitoring up to 5 years. The facility has developed a BSL-3 laboratory safety manual (which covers the entire BRM elements) and several operational aspects that include facility and pathogen-related procedures. There are a few new SOPs developed specifically for working with SARS-CoV-2 and various types of COVID-19 samples. Examples of SARS-CoV-2–specific SOPs include but are not limited to culturing of SARS-CoV-2 from COVID-19 samples, heat inactivation of SARS-CoV-2, RNA extraction from SARS-CoV-2 samples, and virus quantification assay for SARS-CoV-2. All documentation of BSL-3 CF (both hard copies and soft copies) is kept securely with controlled access.
Performance Monitoring and Evaluation (the Check Phase in PDCA)
All processes in BSL-3 CF and components of BRM system are monitored, measured, and evaluated regularly by external and internal parties. Examples of performance monitoring indicators include effectiveness of operational controls, outcome of safety and security emergency drills, outcome of audits/inspections, incident trends, any other nonconformities, and customer satisfaction on safety, security, and service quality. The customer satisfaction survey is done annually by sending a form to all the BSL-3 users with a set of questions. The summary of the survey is presented to the BSL-3 management and the BC for their review and suggestions. Any areas that can be improved further will be addressed by the BSL-3 CF operation team and communicated to the users.
In BSL-3 CF, all incidents including accidents and near misses are reported, root and contributing causes are identified, promptly addressed, and corrected based on the revised RA. The CF operation team works closely with the users and encourages them to report and openly discuss near-miss incidents and errors internally. The RA and SOP may be revised to implement additional mitigation measures if needed. If any specific procedures/areas need more attention, the stakeholders will also be retrained. The outcome of performance monitoring and evaluation is brought up to the management during the annual management review (MR) exercise. If there are serious incidents or deficiencies noted, it will be brought up to the senior management, BC, NUS corporate safety office, or MOH biosafety branch depending on the cases as per BSL-3 reporting procedures.
MR of the BRM System
MR of the BRM system is conducted annually at the end of every year. The MR committee is appointed by the director of the BSL-3 CF and it consists of the director, representatives from BC, NUS Medicine representatives, BCO, and BSL-3 operation team. The committee reviews all elements of the BRM program to ensure its continuing suitability, adequacy, and effectiveness. The outcome of the review is documented and followed up by the responsible parties. If the issue is biosafety related, the BCO will follow up with BC and the PIs. If it is related to policy level concerns, it will be discussed at the BC meeting and the recommendation will be brought up to the NUS Medicine senior management for final approval. MR also includes opportunities for improvement of the BRM; any changes to the system, policies, programs, and objectives to determine any modifications needed; or to establish new ones for the next years.
Continual Improvement Process (the Act Phase in PDCA)
Based on the outcome of the MR, the BSL-3 CF operation team makes necessary changes and improvements for existing programs and policies if necessary. For the SARS-CoV-2 work, when the first batch of COVID-19 clinical samples reached the CF in January 2020, many additional mitigations were included during the initial RA in consultation with MOH, OH physician, infectious disease specialists, biosafety experts, virologists, and senior management. Since there was uncertainty about the risk nature of SARS-CoV-2, more precautions and stringent measures were implemented. After 2 months, when more scientific information on this virus was available, some of these additional controls were removed or relaxed. For example, personnel working with SARS-CoV-2 are no longer required to stay in the dedicated accommodations provided in the university campus and are no longer required to report their temperatures twice a day to the OH physician. Also, based on the re-evaluation of the RA, “batch validation” of inactivated samples is not required before inactivated samples can be removed from BSL-3 CF and worked with in BSL-2 facilities. Any changes or improvements to the program are communicated to all relevant parties including the BSL-3 CF staff and laboratory personnel, and its timely implementation is ensured by the BCO and the BSL-3 BC. The outcome of the MR and the implementation of any improvement program will also be updated and reviewed at the next annual MR.
Conclusion
Adopting a management system allows an organization to efficiently manage the biorisk even during unpredicted emerging pandemic situations. It was very evident during the COVID-19 pandemic that a robust BRM system was needed for the rapid and efficient identification of hazards, prioritization of risks, and the establishment of risk mitigation measures specific to SARS-CoV-2 in laboratories. By promoting a positive safety culture and integrating a systematic risk management process into daily laboratory operations, the NUS Medicine BSL-3 CF has consistently minimized the risks to employees, the public, and the environment. However, the CF team faced the issue of a high volume of requests for COVID-19 research; a limited number of virologists to support the work; a limited number of trainers; and concern on availability of disinfectants, PPE, and other supplies during the early stage of COVID-19 pandemic. These challenges were managed with tremendous support from the NUS Medicine leadership, by carefully analyzing the situation, and coordinating and collaborating with various stakeholders by the CF team. The existing BRM program and its common goal of mitigating biosafety and biosecurity risks through the support of regulatory framework, allocating resources appropriately, prioritizing training needs, and risk management were useful to address these challenges. The BSL-3 CF was also provided with a dedicated and BSL-3–trained research team to support the COVID-19 research.
A well-established and sustainable BRM system for CF was achieved through:
commitment and support from the leadership; implementation and communication of biorisk policy to establish roles and responsibilities, and performance expectations for all BSL-3 CF personnel as well as the stakeholders; dedicated and well-experienced BSL-3 CF team and their relationship and trust with the CF users; providing necessary resources, appropriate education, and continuous training; integrating biosafety and biosecurity management throughout all the BSL-3 work processes and compliance to all legal and institutional requirements; establishing risk criteria and biorisk objectives, promoting preventive measures, and conducting safety and security drills; using the annual MR process to identify deficiencies for continual improvement and prevention; and encouraging open feedback and recognizing improvements and best practices.
Implementing and maintaining a BRM system require an organizational culture that ensures biosafety and biosecurity, in addition to complying with relevant legal requirements and policies, and most importantly strong leadership support. 34 Rules and procedures have no worth if people do not follow them; a culture of responsibility is also essential for ensuring that people follow safety and security procedures and that they act responsibly even in new or unfamiliar scenarios. 34 With the support of efficient and knowledgeable personnel and using the well-established BRM system, the cycle of continual improvement can be well maintained in containment laboratories. A laboratory that has implemented a robust BRM system is prepared to address risks associated with emerging pathogens, and its personnel will have necessary knowledge, skills, and abilities required to deal with unknown risks.
Footnotes
Acknowledgments
The BRM program of NUS Medicine BSL-3 CF is supported by the NUS Medicine's senior management, BSL-3 biosafety committee, and the BSL-3 CF's operations team and research team.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
No funding was received for this article.
