Abstract
Background:
As the pharmaceutical industry advances its understanding of biological processes and how they relate to (the causes and treatments of) disease, many new modalities such as protein therapeutics (PTs) are emerging as breakthrough therapies to treat both rare and common diseases. As PTs become more prevalent, occupational health and safety professionals are challenged with identifying potential occupational exposure risks, health hazards, and assessing best practice recommendations for workers who develop, manufacture, and administer PTs.
Methods:
To characterize airborne exposures to PTs, we conducted a retrospective analysis of industrial hygiene (IH) data for PTs spanning >15 years. This information was used to support the development of an occupational exposure control banding system designed for and applicable to biologically derived PTs (produced in living cells). Overall, 403 IH samples were evaluated that included exposure data for monoclonal antibodies, fusion proteins, PEGylated proteins, and surrogates.
Results:
Our evaluation of historical IH PT sample data indicated low exposure potential across manufacturing activities with >99% (400/403) being below an airborne concentration of 1 μg/m3. Processes with the highest potential for airborne exposure included high-energy operations (e.g., homogenization) and maintenance activities (e.g., cleaning and repairs).
Conclusion:
The observed low exposure potential is expected given that many biological manufacturing activities are closed to maintain product sterility. This evaluation indicated that the banding systems historically utilized for small molecules could benefit from being revisited for PTs.
Introduction
Protein therapeutics (PTs) have become a growing market segment in the pharmaceutical industry. PTs comprise chains of amino acid monomers linked by peptide (amide) bonds, where the covalent chemical bonds form when the carboxyl group of one amino acid reacts with the amino group of another. PTs can be native, recombinant or fusion proteins, or monoclonal antibodies. They are generally considered to be macromolecules in the size range of >10 kDa with monoclonal antibodies typically being >140 kDa. As the market and pharmaceutical company pipelines swell with PTs, questions have arisen regarding the safe handling and banding of these compounds.
Employees have the right to a safe and healthy workplace and this includes ensuring any occupational exposures to pharmaceuticals are controlled to safe levels. Active pharmaceutical ingredients (APIs) are specifically designed to modify biological function and can place employees at risk of experiencing pharmacological and/or toxicological effects if exposures are not adequately controlled. 1 Although these effects are considered desirable or acceptable in patients (based on a risk–benefit assessment), any clinically significant pharmacological or toxicological effect occurring as a result of an occupational exposure, whether considered beneficial or not, is unacceptable. Gathuru et al. (2015) reviewed the occupational health research regarding adverse health outcomes in pharmaceutical manufacturing workers and reported evidence that some pharmaceutical manufacturing workers experienced an increased risk of adverse health outcomes including cancer, endocrine dysfunction, and liver disease. 2 It is imperative that pharmaceutical companies and pharmaceutical contract manufacturers protect workers, and utilizing occupational exposure control banding paradigms is one such way this can be accomplished.3–5
In the pharmaceutical industry, proprietary compounds (including proteins) are generally assigned to occupational exposure control bands (ECBs). ECBs define airborne concentration ranges (e.g., μg/m3) of substances and represent conditions under which nearly all workers can be exposed without adverse health effects. The exposure concentrations are commonly referred to as time-weighted average (TWA) concentrations that are set for a normal 8-h workday and a 40-h workweek. Each “band” is associated with a set of handling practices designed to maintain exposure to the compound within (or below) the designated range. ECB systems (Table 1) are commonplace in the pharmaceutical industry and play an integral part in the communication of hazards and safety precautions.3–6 The ECB system was designed to give guidance, based on historical experience, on safe handling practices for compounds with limited data as a stopgap until additional relevant data could be generated. For pharmaceutical compounds with robust data sets, compound-specific occupational exposure limits (OELs) are established to protect employees; however, when there are limited data for a compound, especially in early drug development, ECBs are normally employed to establish occupational exposure constraints.
Example of a small molecule occupational exposure control banding system
Historically, ECB systems were designed with small molecules in mind. As PTs are generally large molecules manufactured in closed processes, the inhalation exposure risk is often perceived as being lower than that of traditional small molecule therapeutics. Therefore, applying the ECB system historically designed and utilized for small molecules is called into question with regard to whether the personal protective equipment (PPE) and associated engineering and administrative controls are needed for PTs (e.g., whether we are overengineering exposure constraints unnecessarily). Occupational exposures, including both airborne concentrations and the potential for systemic bioavailability (BA), are expected to be lower for most PTs in comparison with that of small molecules. Typical small molecule synthesis consists of a series of chemical reactions with isolated intermediates (generally crystallized solids) enroute to the API, and the physical characteristics and hazards associated with each isolated intermediate can vary from reaction to reaction. Biologics, however, are produced from living cells or biological processes and are generally thousands of times larger and more complex than small molecules. Owing to their large size, PTs are expected to have very limited dermal and inhalation BA, as well as very limited oral BA due to their degradation in the digestive tract. Consequently, the BA of PTs through these routes of exposure is expected to be low.7–10 In addition, biological manufacturing through cell culture or fermentation generally occurs in closed processes subject to sterile or near sterile conditions to protect the quality of the product, subsequently minimizing the potential for occupational exposures. PTs also tend to be handled in solution and are unstable under normal environmental conditions (e.g., sensitive to temperature/light). Owing to these differences in their inherent properties and manufacturing processes, the occupational exposures to PTs and small molecules are expected to differ.
Recognizing the difference in hazards, exposures, and risks associated with PTs, we sought to determine whether historically utilized small-molecule approaches to banding were a good fit for PTs or whether the bands could benefit from being redesigned. In an effort to better understand and characterize worker inhalation exposure potential during the development and manufacturing of PTs, we evaluated historical industrial hygiene (IH) data consisting of 403 IH samples (342 during PT activities; 61 during surrogate activities) collected and analyzed over a 15-year period across manufacturing activities.
PT Biological Manufacturing Activities
The activities that comprise PT development and manufacturing typically include inoculation, cell culture and fermentation, harvest and recovery, purification, formulation, and fill–finish operations (Figure 1). In general, the production of a biologic begins with a cell or organism (e.g., mammalian cell) that is modified in such a way (e.g., transfection with RNA) to enable it to produce the substance of interest (typically a protein, peptide, or antibody) directly without any further open chemical reactions or processes. That modified organism is then cultured in a seed reactor under closed and sterile processes, where it grows and multiplies. When the organisms reach a specific production point, the process is halted, and the substance of interest is harvested. Harvesting activities include processes that release the substance of interest from the cell/organism. Once harvested, the substance is purified through various closed processes and prepared for fill–finish activities. Once production is completed, systems are cleaned and steamed in place before opening the process equipment, or single use technology containment is disposed.
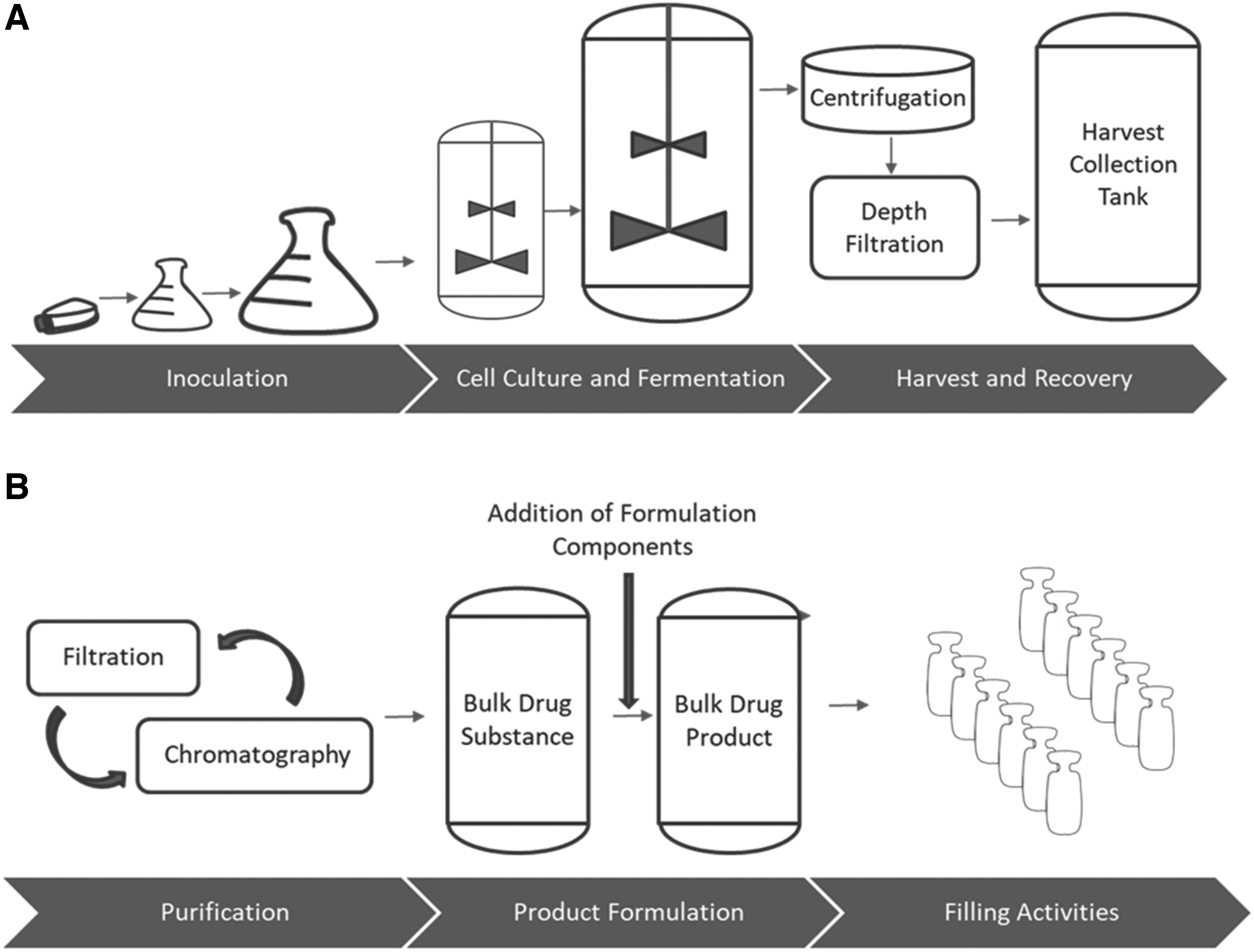
Overview of the biologics manufacturing process.
Adverse Health Effects of PTs
Importantly, proteins and their metabolic products, amino acids, are endogenous to all living things and PTs are cleared through the same catabolic pathways utilized to eliminate endogenous and dietary proteins. 10 There are, however, still concerns with occupational exposures to PTs as these molecules are pharmacologically active compounds with high potency and specificity to their therapeutic target(s). Notably, PTs have different pharmacokinetic (PK) profiles than conventional small molecule drugs. PTs are designed to have long half-lives (once in the body) that can fall within the range of days or weeks (supporting intermittent administration) in comparison with hours as is generally the case with small molecules, and this information needs to be considered when deriving OELs and assigning occupational ECBs to PTs. 11 Although some PTs lead to the death of targeted cells, they are not considered to be conventional cytotoxic agents because they do not directly or indirectly damage DNA or RNA. PTs as a class are generally not considered to be carcinogenic, mutagenic, or teratogenic to patients or those handling them. 12 However, for PTs that activate the immune system, developmental toxicity is a potential hazard.13–15 Immunogenicity is also a concern with exposure to biologics, with immunogenic potential being expected to increase with the proportion of foreign protein. 16 For additional information on the potential adverse health effects of PTs, broken out by modality (both occupational and therapeutic use related), see Graham et al. 17
Materials and Methods
IH Samples and Biological Processes Monitored
In total, 403 IH samples consisting of 194 area and 209 personal samples were collected and analyzed by Bristol Myers Squibb (BMS) professionals across biologics manufacturing and development activities for ∼15 years (2002–2017) at BMS sites worldwide. The biological processes monitored included inoculation, cell culture, fermentation, harvest and recovery, purification, and product formulation (Figure 2). Of the 403 samples, 342 were obtained during routine operations with PTs and 61 were obtained during surrogate monitoring activities (Table 2). Both area (194) and personal (209) samples were included in this evaluation. The PTs sampled for included monoclonal antibodies, fusion proteins, PEGylated proteins, and protein surrogates (mannitol or lactose in solution) (Supplementary Figure S1).
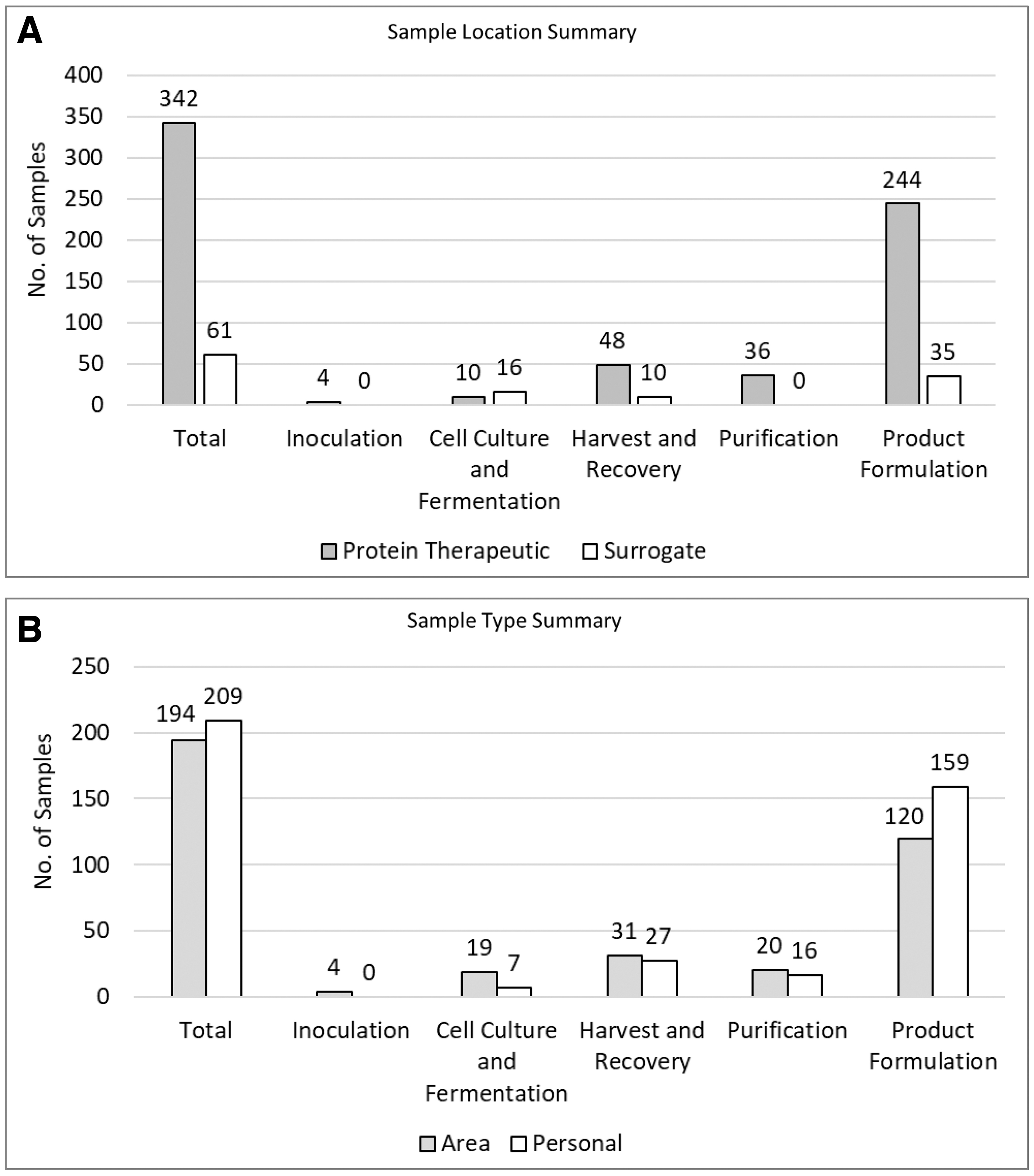
IH sample summary.
Overview of industrial hygiene samples on protein therapeutics and surrogates
Bold values are the total of the “product” and “surrogate” rows below.
Includes samples below the LOQ.
LOQ, limit of quantitation; OEL, occupational exposure limit.
IH Sample Methods
Proprietary IH analytical methods were developed and utilized by the BMS American Industrial Hygiene Association (AIHA) accredited laboratory to determine exposure levels. Air samples were collected on 0.45 μm polytetrafluoroethylene membranes in polypropylene cassettes with a typical air flow of 2 L/min. Air samples were extracted with appropriate solvents and analyzed by high performance liquid chromatography with ultraviolet or fluorescence detectors. All methods were compound specific and had limits of quantitation (LOQ) that ensured the OELs could be quantified with the given sampling times or volumes of air. The sample results were reported as 8-h TWAs by the BMS AIHA accredited laboratory. In cases wherein PT operations and associated samples did not last an entire shift, the assumption was made that additional PT exposures were not occurring during the remaining portion of the shift.
Results
Biological Processes and Corresponding Exposure Monitoring Results
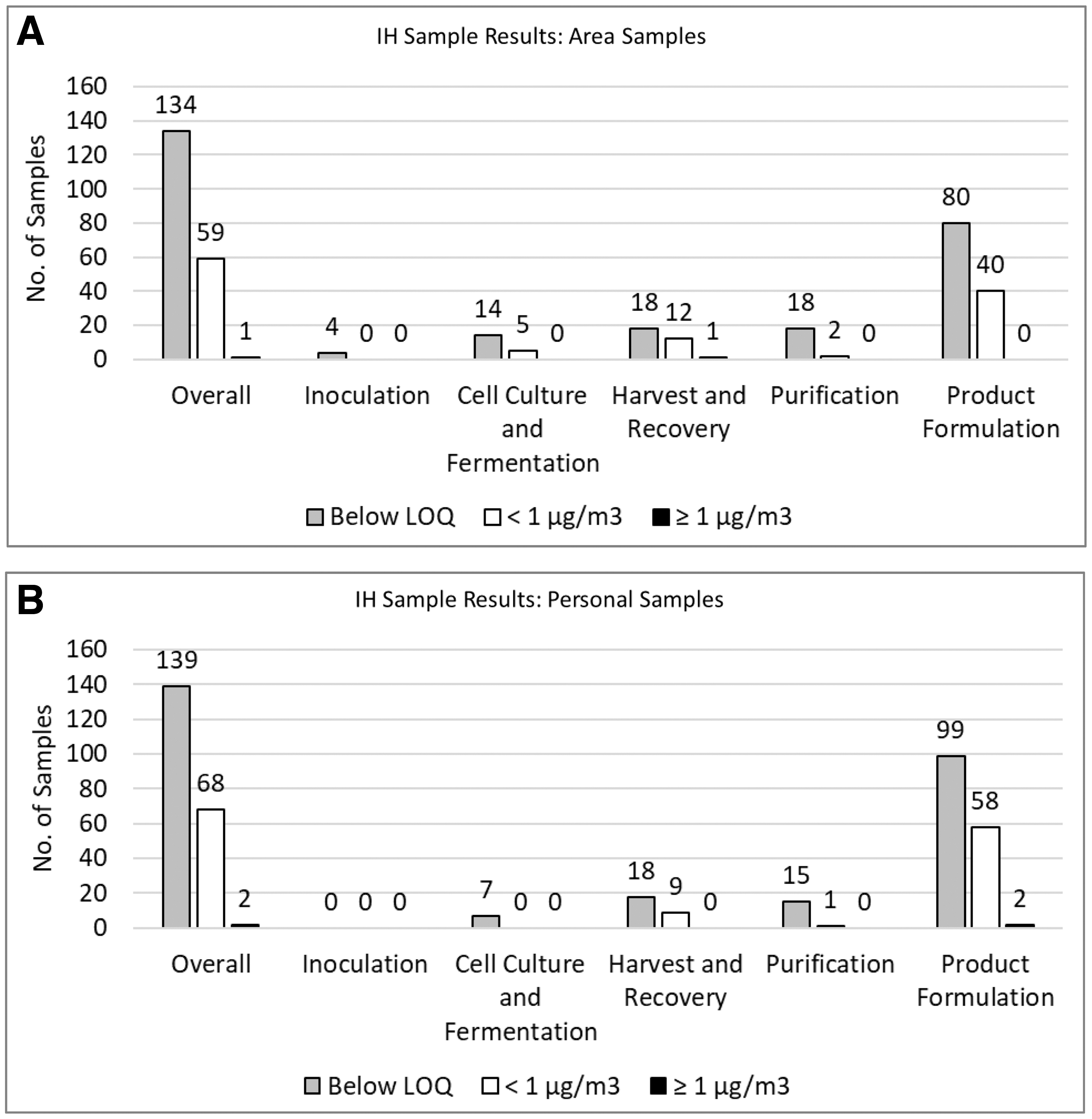
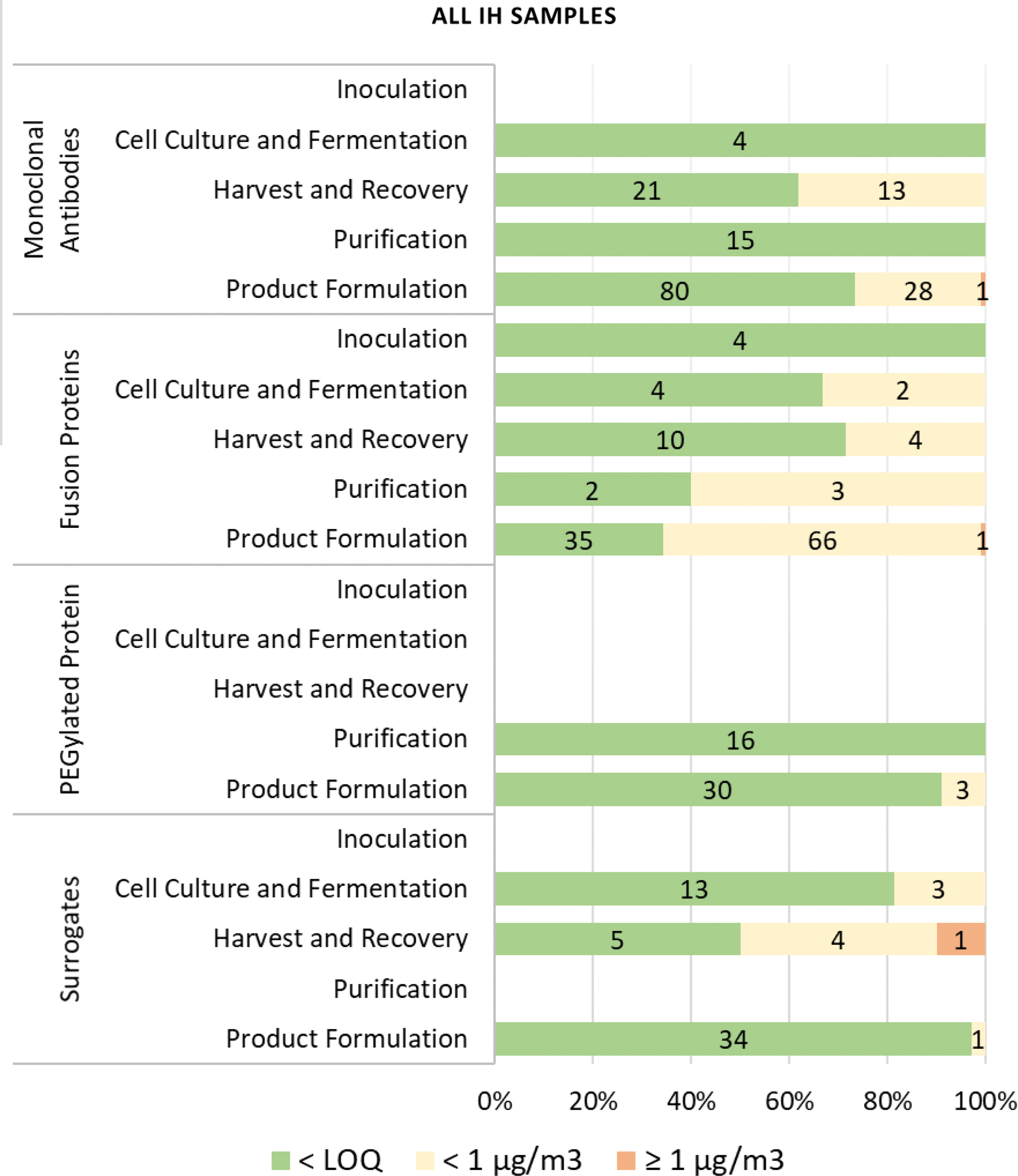
The evaluation of IH monitoring results for the inoculation, cell culture and fermentation, harvest and recovery, purification, formulation, and fill–finish operations revealed an overall low to negligible potential for exposure risk (Figures 3 and 4; Supplementary Figures S2 and S3).
Maximum concentrations observed across protein therapeutic manufacturing and development activities.
IH sample results. Airborne concentrations observed across protein therapeutic manufacturing and development activities are presented (including results from surrogate monitoring activities). Data parsed out for area samples and personal samples are presented in Supplementary Figure S3. Color images are available online.
Inoculation
Cell culture and fermentation
Harvest and recovery
Analysis of yield samples
Purification
Product formulation and fill–finish operations
Areas Warranting IH Priority Evaluation
Overall, 400/403 samples from manufacturing activities with PTs and surrogates were <1 μg/m3. Three samples were >1 μg/m3 consisting of one (46 μg/m3) recorded near a microfluidizer during harvest/recovery activities, and two that occurred during product formulation activities and were 1.003 μg/m3 (during equipment cleaning) and 27 μg/m3 (due to a faulty pump). High-energy operations such as homogenization and microfluidization as well as maintenance and cleaning activities appear to be areas where focus is warranted to ensure that employee exposures are minimized.
Another scenario warranting additional IH consideration is PTs with OELs <1 μg/m3. Although all PTs with an OEL ≥1 μg/m3 were well below their respective OELs (most <10% of their OELs), one personal sample obtained during a filling operation for a PT with a low OEL of 0.01 μg/m3 was elevated with a TWA concentration of 0.019 μg/m3 (Figure 5). In addition, precaution is warranted in scenarios with potential for sharps exposure and accidental injection.
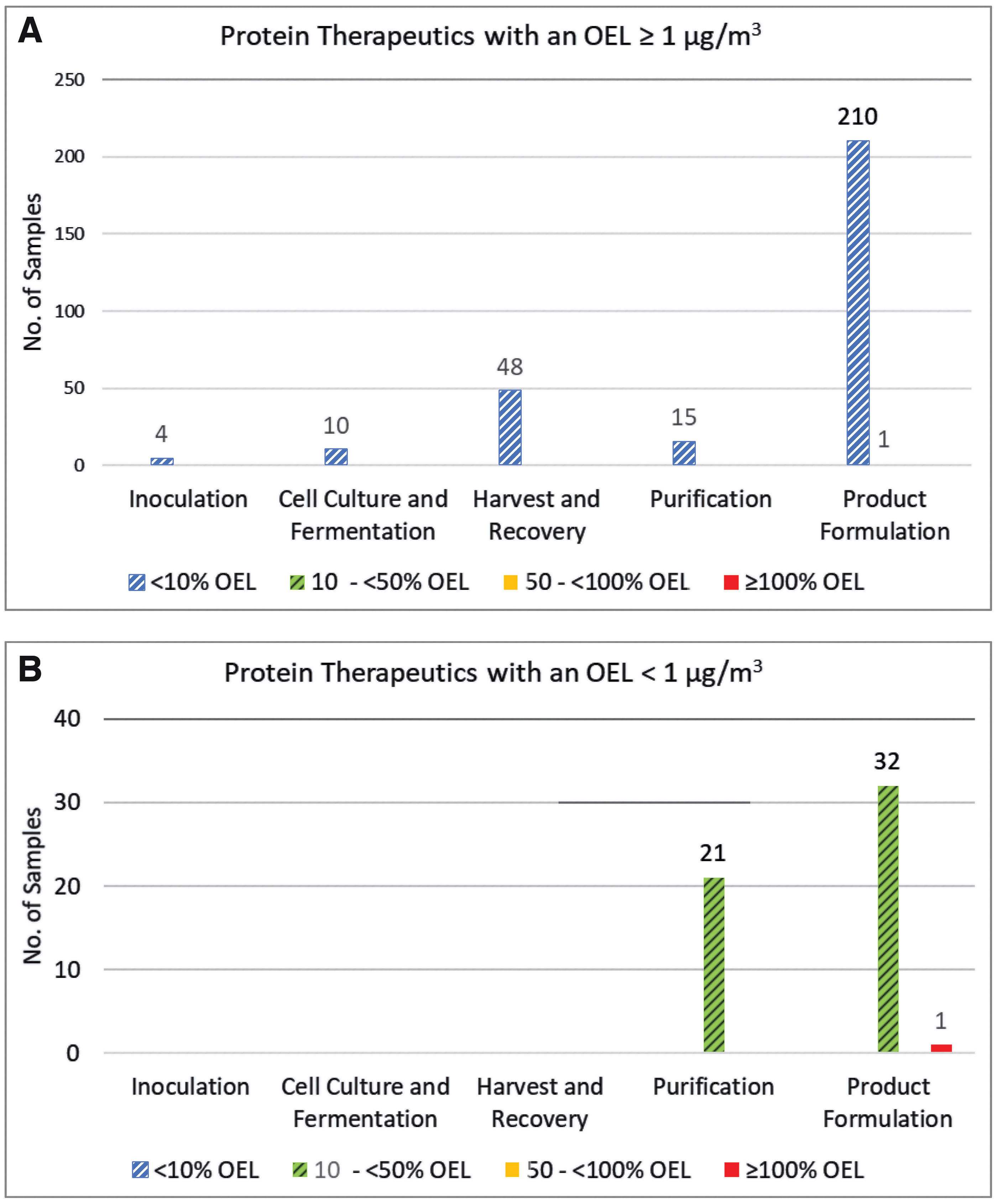
Industrial hygiene sample results for protein therapeutics expressed as the percent of the protein therapeutic's respective OEL.
Discussion
To understand potential exposure to PTs during their development and manufacturing and to inform the development of a biologics banding system, 403 IH samples (area and personal samples) taken across biological handling and production processes were evaluated (Figure 2). This retrospective evaluation was conducted to characterize the occupational exposures expected and elucidate any areas of concern. The IH samples were focused on airborne concentrations as the PT in the air is expected to be in its active form, while PT present on a surface may no longer have pharmacological activity due to the general instability of biologics under normal environmental conditions. 18
Occupational Exposures to PTs
The potential routes of exposure in the workplace include inhalation, ingestion (through mucocilliary transport from the lung, hand to mouth contact, etc.), dermal contact/absorption, and accidental injection (e.g., needle stick). When establishing OELs, the potential for systemic exposure is also taken into account.
Oral exposure
Owing to the large size of PTs and degradation in the digestive tract, the BA of PTs through the dermal, oral, and inhalation routes of exposure (with the exception of accidental injection) is expected to be low. Although oral exposure can occur in the workplace, PTs are expected to be broken down quickly by digestive tract acids and enzymes before a substantial systemic impact could occur.10,19 Therefore, systemic exposure to the active PT through ingestion is expected to be negligible.
Dermal exposure
The dermal BA of a PT depends on properties such as its molecular weight (MW), lipophilicity, and whether there are target receptors present in the skin (potential for direct skin effects). Owing to their large size, PTs are expected to have low to negligible dermal BA as compounds with a MW of >500 Da are generally too large to penetrate the skin. 7 However, dermal absorption may be a concern for an individual with nonintact skin. 19
Inhalation exposure
Importantly, the airborne concentration of a PT does not directly correlate with the amount systemically absorbed upon inhalation. As PTs are macromolecules (>10 kDa), their large size helps to limit their inhalation BA. Protein inhalation studies have suggested an inverse relationship between MW and BA and demonstrated low systemic BA (e.g., <1% to 5%), which may be much lower than that of small molecules.8,9 Systemic exposure resulting from the inhalation of PTs is expected to be limited; however, this may not be the case if there are target receptors present in the lung or a concern for other direct effects on the lung. 20
Although it is known that PTs generally do have health hazards when used therapeutically and in nonclinical studies, it is important to note that these hazards are generally not expected to occur from occupational exposures due to limited systemic exposure potential through the inhalation, ingestion, and dermal routes based upon the study data that were reviewed. Additional characteristics of biologics expected to limit occupational exposure potential include the following:
PTs are typically handled as dilute mixtures with concentrations ranging from 0.2% to 25% and the remaining percentages consisting of excipients and stabilizers. In addition, exposure potential is lower for compounds handled in solution, due to a reduced potential for becoming airborne than when handling a solid/powder. PTs are typically not stable long term or outside recommended storage conditions and degrade under normal environmental conditions (e.g., sensitive to light/temperature); therefore, the likelihood that exposure to pharmacologically active PTs can occur due to residual PTs on surfaces is limited.
18
PT activities are typically conducted in closed systems.
Sampling Results and Occupational Exposure Control Banding Considerations for PTs
Owing to the paucity of occupational exposure data for biologics as well as the pharmacological and toxicological effects of PTs upon therapeutic administration being considered as potential effects resulting from occupational exposures, concerns have arisen around handling PTs in the workplace. 19 Owing to the limited potential for systemic exposure, it is important to note that adverse effects observed during therapeutic use and/or in PT nonclinical studies are generally not expected to occur from occupational exposures. In addition, the IH sampling results demonstrated that >99% of samples were <1 μg/m3 across manufacturing operations, indicating that inhalation exposure may be lower for PTs than historically observed and assumed for small molecule pharmaceutical compounds (Figures 3 and 4; Table 2). This, coupled with the low systemic BA through occupationally relevant routes (generally closed manufacturing activities, and handling of PTs in dilute solutions), limits the potential for airborne material and subsequent exposure as compared with typical small molecule solids. In addition, although confirmation is needed for specific molecules, PTs are generally unstable in the environment, minimizing recirculation of, and exposure to, previously released materials.
Based on IH monitoring data and the considerations already presented, PT exposure potential is expected to be limited or negligible with the exception of high-energy processes, maintenance activities and sharps exposures. Evaluation of IH monitoring results and exposure assessments indicates that inhalation occupational exposures to PTs under normal working conditions are low, with 99% (400/403) being <1 μg/m3 (Figures 3 and 4). Of the samples taken during operations with PTs, 99% (340/342) were <1 μg/m3 consisting of 100% (135/135) of area and 99% (205/207) of personal samples. PT modalities above the LOQ were highest for fusion proteins (76/131), followed by monoclonal antibodies (42/162), and PEGylated proteins (3/49) (Figure 4; Supplementary Figure S3). Of the samples taken during surrogate monitoring activities, 99% (60/61) were <1 μg/m3 consisting of 98% of area samples (58/59) and 100% (2/2) of personal samples. Based on the low airborne concentrations observed across activities, the closed manufacturing processes employed with PTs, and the occupational exposure assessment (Occupational Exposures to PTs section), it was determined that a different banding system than that employed for small molecules could be successfully employed for PTs. Based on this conclusion, a biological-specific banding system was developed consisting of a different set of exposure controls and handling practices, from those associated with the small molecule ECB system.
A Biological-Specific Banding System
Activities to develop a banding system specific to biologics were undertaken using the BMS small molecule control practices as a framework. Biosafety levels established by the National Institutes of Health/Centers for Disease Control and Prevention (NIH/CDC) and the World Health Organization (WHO) for pathogens were considered, but not seen as a good fit as they characterize controls needed for the host organism and do not define controls needed for the resulting PTs. 21 A review of the IH monitoring data obtained from PT development and manufacturing activities supported a limited banding system, such as a two-band system versus the typical four or more band systems used for small molecules (Table 1). 3 Historical IH data demonstrated that ∼100% of samples (286/287) for PTs with an OEL of ≥1 μg/m3 had exposures <10% of their respective OELs and were well controlled by the process. This was compared to PTs with an OEL of <1 μg/m3, where IH data demonstrated that 98% (53/54) of exposures were controlled to between 10% and 50% of their OEL with 2% (1/54) exceeding their OELs. This observation indicated the need for more rigorous risk assessments and evaluations of controls for PTs with OELs <1 μg/m3 in development and manufacturing settings (Table 2 and Figure 3). It was, therefore, decided to utilize a two-band system with a cutoff between them of 1 μg/m3. This decision was based on the historical IH exposure data, occupational exposure potential, as well as an understanding of where highly hazardous and potent compounds should be controlled. Using this system, PTs and other active biologically derived materials could be categorized into one of two biologic control categories (BCCs): BCC-A with an OEL ≥1 μg/m3 or BCC-B with an OEL <1 μg/m3 (Table 3).
Example of a biologic-specific banding system
BCC, biological control category.
The categorization of a PT is based on its intrinsic pharmacology, toxicology, and PKs to enable an appropriate margin of safety between the dose expected to cause a clinically significant effect and the potential occupational exposure. BCC-A is expected to be protective for the handling of biologics with low to moderate toxicity or potency while BCC-B exposure controls and corresponding handling practices should be considered for especially potent or toxic PTs (see Graham et al. 17 for more detail with regard to the characteristics of biologics that fall into each BCC).
The recommended handling guidance for compounds assigned to BCC-A and BCC-B is presented in Tables 4 and 5 and is expected to be protective for workers handling the PT across development and manufacturing operations. The recommended exposure controls presented in Tables 4 and 5 were derived based on BMS internal sampling efforts (data not shown) of activities, equipment, and unit operations. The statistical evaluations of the sampling data were conducted according to guidance in EN 689 (data not shown). 22
Biologic control category A handling guidance for protein therapeutics with occupational exposure limits ≥1.0 μg/m3
Biologic control category B handling guidance for protein therapeutics with occupational exposure limits <1.0 μg/m3
Note that the cutoff for BCC B powders is lower than that recommended for BCC A powders, although the cutoffs for solutions remain the same. The cutoff of 5 g of powders is recommended for BCC B PTs in an effort to be conservative with these potent compounds. Also, as BCC B compounds are more potent, they tend to be handled in smaller quantities than BCC A PTs. The cutoffs for solutions are based on the general volumes called for in unit operations. In addition, there is more of a concern with a dry solid becoming airborne than with the generation of microaerosols of the active PT from a solution. For these reasons, the volumes are consistent between BCCs.
BSC, biological safety cabinet; HEPA, high-efficiency particulate air; PTs, protein therapeutics.
The proposed two-BCC system simplifies and clarifies the process for selecting workplace controls. Given the controls already inherent to the good manufacturing practices (GMPs) and procedures, the nature of PT manufacturing activities, and the occupational exposure potential to active PTs, two levels of controls or bands were deemed appropriate for PTs. PTs assigned to BCC-B require a more rigorous evaluation of containment along with a thorough review of any potential sharps exposures while also ensuring accidental release control procedures are in place. The utilization of the BCC system presented in Table 3 with the recommended handling presented in Tables 4 and 5 is expected to be protective for biologically derived PTs. Note that compounds with limited data are assigned to a BCC and once a sufficient data package is available to establish an OEL, the handling guidelines utilized should correspond to ensuring exposures remain below the respective OEL.
As with any process evaluation, a risk assessment should be conducted on the entire process with a focus on areas for exposure potential. In general, PT manufacturing is performed utilizing closed systems to maintain sterility and GMP compliance. As a PT is developed and moved from research to development and then into manufacturing, the process controls and containment increase. For this reason, IH evaluations should be conducted at all stages of the process with a focus on high-energy operations as well as cleaning and maintenance activities. Development activities should also be thoroughly evaluated since controls might not be as rigorous as in a GMP setting.
Conclusion
The general processes and procedures involved in the development and manufacturing of PTs are expected to be associated with low occupational exposures, which was confirmed by the retrospective evaluation of IH monitoring presented herein. In addition, these monitoring activities elucidated areas for IH focus such as high aerosol potential operations, cleaning/maintenance operations, and when working with PTs that have low OELs. Owing to the general instability of PTs under normal environmental conditions, limited exposure during routine activities, and the limited BA through occupationally relevant routes, it can be argued that a simplified banding system (in comparison with the one utilized for small molecules) could be employed for PTs and be protective for employees when working with biologics as compared with small molecules. Equally important is that the health hazards observed for PTs upon therapeutic dosing do not always directly correlate with occupational health hazards due to the limited potential for systemic exposure to PTs in the workplace. The newly developed BCC system was designed with PT manufacturing and handling practices/procedures in mind, is anticipated to be protective, and proposes to utilize two bands with associated exposure controls (encompassing engineering and administrative controls along with PPE use). More data are welcomed in this field to support the two BCC model. The proposed BCCs present a straightforward cost-effective means to categorize PTs based on occupational exposure hazards and assign handling controls to ensure worker safety.
Footnotes
Authors' Contributions
E.F. conceived of the presented theory and identified the relevant IH sampling data. H.Y. assisted with obtaining the IH sampling data. J.G. compiled and analyzed the IH sampling data and wrote the article with support from E.F. and H.Y. All authors provided critical feedback and helped shape the analysis and article.
Acknowledgments
The authors thank Nancy DeMaio and John Trela for initial contributions to the article as well as Danielle Menture, Gene Schulze, and Michael Skoglund for their support of this study and reviews of the article.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This study was funded by Bristol Myers Squibb.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
