Abstract
Background:
With increased rates of laboratory-acquired infections from clinical and research laboratories globally, efforts have been made to improve awareness of modern practices and pursue innovations in biosafety to manage risks and laboratory exposures arising from infectious agents and other hazards.
Objectives:
This article demonstrates a sustainable biosafety training model developed to enhance laboratory quality and support accreditation in health facilities in Kenya.
Methods:
A biosafety technical working group was formed, and sensitization meetings held with health managers. Trainings were then conducted for training of trainers (TOTs) who then cascaded trainings in health facilities. This was followed by mentorships and monitoring for implementation.
Results:
Five sensitization meetings were carried out for 264 health managers. TOTs was done for 48 trained trainers and 1044 laboratory workers in 216 facilities covering 44 counties. Site visits were done in 51 facilities, with biosafety achievements measured in 21 (41%), respectively. Achievements in 21 facilities included the following: improvised eye wash stations in 16 facilities (76%), biological spill kits in 17 (81%), buckets of sand in 15 (71%), fire extinguishers in 12 (57%), hepatitis B vaccination in 14 (66%), establishment of phlebotomy areas in 18 facilities (85%), material safety data sheets in 18 (85%), documentation of incidents and exposures in 16 (76%), and proper waste segregation in 17 (81%).
Conclusion:
This model ensured rapid scale-up to multiple counties and enabled learners to understand biosafety principles. Due to management buy-in, resources were availed to implement interventions, and this was demonstrated by remarkable achievements across all assessed facilities.
Introduction
With increased rates of laboratory-acquired infections from clinical and research laboratories globally, 1 occupational exposure among healthcare workers (HCWs) has led to the recognition that biosafety remains an important element within the greater framework of strengthening global health and security. Globally, it is estimated that 1 in 10 HCWs experiences a sharp injury every year. 2 While all HCWs are exposed to occupational hazards, those in sub-Saharan Africa have higher rates of exposure to infectious diseases than those in developed countries due to the high burden of such diseases in Africa. 3 A 2015 WHO report on HCW's infections found that they are between 21 and 32 times more likely to be infected with Ebola than people in the general population. 4 A case in point was the 2014 West Africa Ebola outbreak, during which the case fatality ratio of Ebola-infected HCWs was reported to be 57%, with all eight HCWs who had been infected in Congo dying. 5 Recently, the COVID-19 global pandemic of 2019–2020 and the Ebola outbreak in Congo in 2018–2019 have further seen an increase in infections among HCWs.
Laboratory workers are exposed to pathogens while providing services for diagnosis and monitoring of cases, an integral part of the response to disease outbreaks and patient management. Biosafety and biosecurity are key components in biorisk management and entail implementation of laboratory practices and procedures, specific construction features of laboratory facilities, safety equipment, and appropriate occupational health programs to prevent personnel exposure when working with potentially infectious microorganisms and other biological hazards. 6 Rapid scale-up of HIV/AIDS testing, care, and treatment, as well as services for prevention of mother-to-child transmission, has resulted in high demand for laboratory procedures, with inadvertent occupational exposure and risk of biohazards against a backdrop of nonexistent biosafety mechanisms and standard operating procedures. Human error and poor technique can compromise the best of safeguards to protect the laboratory worker.
Training can be a powerful tool for improving biosafety practices in the laboratory, just as it is in many other areas. 7 Laboratory staff must be effectively trained and asked to demonstrate their competence and practice critical thinking to increase the likelihood of safe laboratory behavior. Factors that contribute to gaps in biosafety practices include the following: poorly defined occupational health and emergency response plans, poor infrastructure and engineering mechanisms; inadequate availability or use of personal protective equipment (PPE); inadequate and unsustainable training programs; and lack of systematic reporting mechanisms.8,9 According to Heckert et al., any country operating laboratories that provide some combination of diagnostic, clinical, or research activities involving human and/or animal pathogens should have laboratory, biosafety and biosecurity capabilities. 10
Over the past several years, policymakers and the public have become increasingly aware of the potential risk of HCW exposure to biological hazards and the possibility of environmental release, due to the expansion of high- and maximum-containment research and diagnostic facilities. 11 In Kenya, the National Biosafety Office within the Ministry of Health (MOH) and the US President's Emergency Plan for AIDS Relief (PEPFAR), through the Centers for Disease Control and Prevention (CDC), worked with the Management Sciences for Health (MSH) to provide resources, technical support, and monitoring of program activities for the biosafety program. Specifically, the PEPFAR- and CDC-funded Strengthening Public Health Laboratory Systems project managed by the MSH in Kenya was implemented between 2009 and 2015, with the aim of strengthening the capacity of the MOH to effectively manage a country-wide, high-performing, sustainable, public health-oriented laboratory system, hence improving patient care and saving lives.
Biosafety is an important element of a quality management system (QMS), because it is a measuring tool for compliance with accreditation and certification standards. 11 In leading its support for scaling up awareness of biosafety, the project recognized that a well-functioning laboratory system should strive to implement an effective QMS, which entailed developing a sustainable biosafety training program. The implementation of the biosafety training model continued through the devolution of health services from one national government to 47 county governments after enactment of a new constitution in Kenya. High staff turnover was observed during this transition as HCWs moved from one county to another, compromising the continuation of interventions that might have been made following the trainings. Infrastructure in most facilities in Kenya does not favor the implementation of good biosafety practices and this is further decremented by erratic supply of safety commodities. 9
The purpose of this study was to demonstrate the process undertaken to conduct sustainable biosafety trainings among laboratory workers to enhance laboratory quality and support accreditation in health facilities with limited resources in Kenya. This article discusses improvements achieved after the longitudinal follow-up, shares lessons learnt, and makes recommendations for the future.
Facilities/individuals who were sensitized or trained were longitudinally followed up with the following specific objectives: to evaluate the outcome of sensitization meetings conducted to the county/facility top managers on biosafety and biosecurity; to evaluate the outcome of biosafety training on the individual trainee; to compare the assessment (audit) score on safety section before and after the biosafety trainings; to assess health facility safety improvements after staff have been trained on biosafety; and to assess the efficiency of training more trainers on biosafety and biosecurity.
Methodology
Planning
Consultative and planning meetings were conducted between the National Biosafety Office, CDC, MSH, and the Biosafety Technical Working Group (TWG). The TWG formed in 2012 and composed of the MOH, implementing and developing partners, research institutions, and major hospitals selected clinical laboratories to benefit from the biosafety training program. Those selected were clinical laboratories enrolled in the ISO 15189:2012 accreditation process or those due to be enrolled. ISO 15189 standard, medical laboratories—requirements for quality and competence, hereafter referred to as ISO 15189:2012 is an international standard that specifies the QMS requirements particular to medical laboratories. 12 The goals of the ISO standard include continual improvement of laboratory operations; systems and the accompanying accreditation can benefit biorisk reduction efforts. Public, private, and nongovernmental health institutions were included to foster private–public partnership, enhance universal health coverage while factoring in high HIV/AIDS burden and hard-to-reach areas. MSH developed a concept note and biosafety facts sheet on the biosafety training program, which provided an outline of the program, target group, and approaches to be used. The concept notes and fact sheets were approved by the MOH at the national level through the National Biosafety Office. They were then sent to the County Health Management Teams (CHMTs) in the regions targeted. The program adopted a curriculum that was developed by the MOH in collaboration with the University of Maryland-Baltimore and utilized master trainers trained previously. The county teams then identified the laboratory professionals to be trained and provided overall coordination and leadership of the training program. The biosafety improvement approach is summarized in Figure 1.
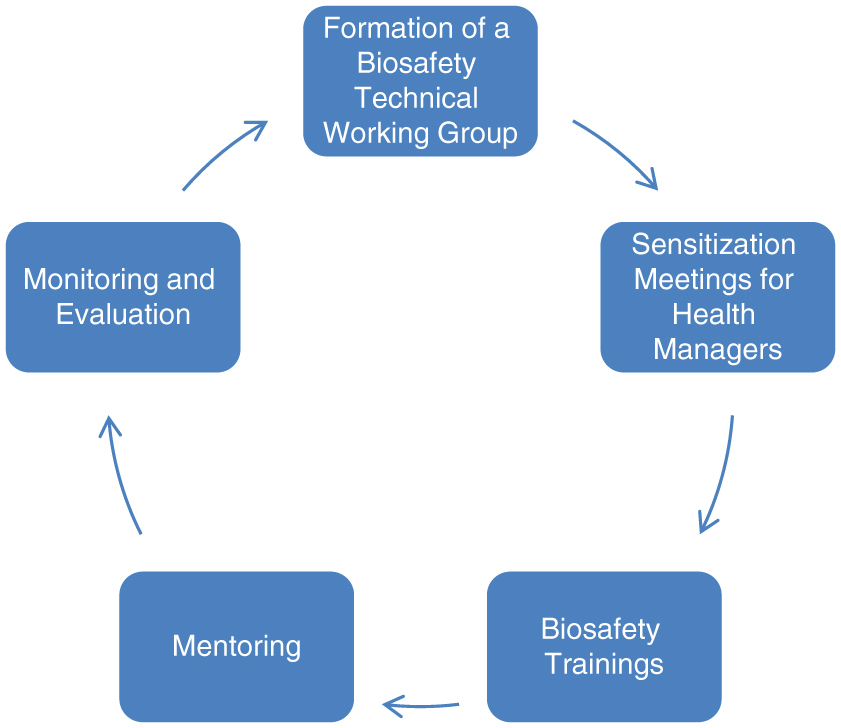
Summary of biosafety training model. Color images are available online.
Sensitization and Engagement
One-day sensitization meetings were held for county and facility managers (facility in-charges, county laboratory coordinators, facility laboratory managers, and nursing in-charges) between 2012 and 2014. The Managers were sensitized from all the regions in Kenya as shown in Figure 2. The meetings aimed to familiarize health managers on biosafety and biosecurity in healthcare settings and to showcase biosafety as an important aspect of quality in the laboratory. The managers then developed a work plan based on their respective counties, which included the dates when the training would be conducted, the venue, the participants who would benefit from the program, and their management role during the training and implementation period. In addition, the managers supported health facilities in acquiring the materials and equipment needed for continuing medical education, including projectors and flip chart stands. They also lobbied the county medical directors and partners to improve the infrastructure and layout of laboratories for safer operation of laboratory activities. The county and facility health managers, with the trainers and MSH, conducted the mentorship visits, prepared reports, and maintained the biosafety training database in their counties. During the sensitization meetings, the MSH provided participants with information, education and communication materials in the form of biosafety handbooks, guidelines, signages, and job aids. Figure 3 summarizes the guidelines and communication materials disseminated.
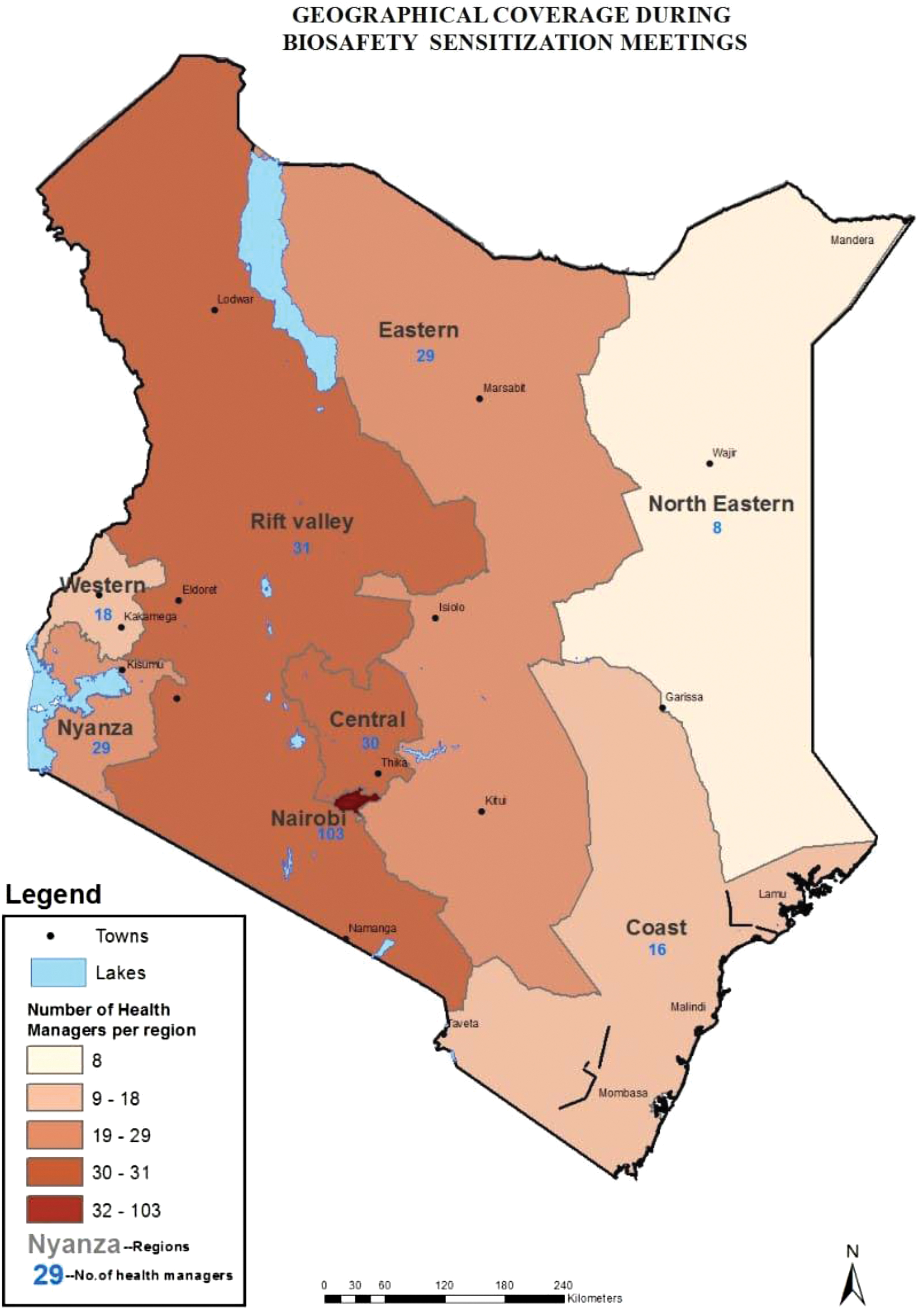
Managers were sensitized from all the regions in Kenya. Geographical coverage of regions covered during the biosafety sensitization meetings targeting health managers. Color images are available online.

Resource materials used during sensitization of managers and during trainings and TOT. TOT, training of trainer. Color images are available online.
Biosafety Training of Trainers
Previously, the MOH was using biosafety trainers who had been trained outside the country and others trained in-country by the Kenya Medical Research Institute (KEMRI) in collaboration with the CDC to conduct biosafety trainings. Since the trainers were inadequate in number to scale up the trainings, a decision was made to train more trainers in each region of implementation. Figure 4 shows the regions covered during the TOT. The additional trainers were trained by qualified master trainers between 2014 and 2015. The purpose of the training was to equip trainees with adequate knowledge and skills to deliver biosafety trainings, as outlined in the National Biosafety Training Curriculum. To ensure that that the right laboratory personnel were selected to benefit from the training, the following process was followed: training advertisement was shared through the National biosafety office requesting for applications from interested candidates; formal minimum criteria were developed; all the applications were reviewed by the TWG against the set criteria and a score was awarded; the applicants were ranked according to their score and a cutoff score was determined for those who were to be admitted in the biosafety training of trainers (TOTs) class; a communication was made for both the successful and unsuccessful candidates. The selected participants were sent for a preworkshop assignment 2 weeks in advance. Prerequisites for participants were that they must be qualified medical laboratory professionals working in a clinical laboratory; have attended the standard five-day basic Biosafety Training for Health Care Workers; have shown interest and commitment by filling out a TOT application form; and have completed and submitted a preworkshop assignment.
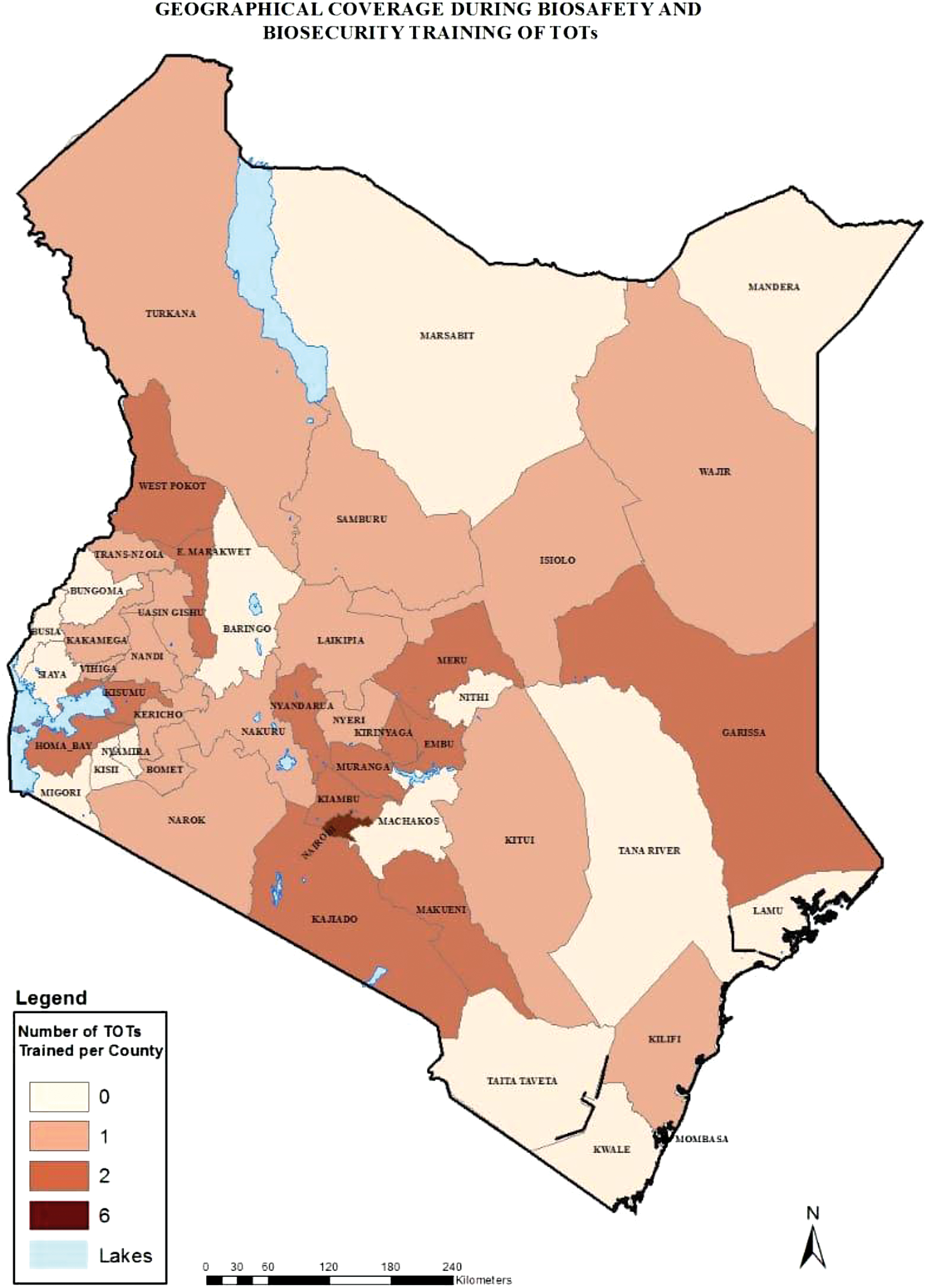
Geographical coverage during biosafety and biosecurity TOT. Geographical coverage of regions covered during TOTs. Color images are available online.
During the 5-day TOT, lectures, plenary sessions, “teach-back” sessions, and site visits to selected health facilities during practical sessions were conducted, with additional evening preparation sessions by appointment. The plenary sessions included descriptions, exercises, and information about training basics and how to facilitate an interactive lecture, activity, or discussion. The teach-back sessions entailed participants delivering a presentation and putting into practice the training basics acquired during the plenary session. This would enhance interactive and adult learning approach to enhance learning. The participants were also expected to provide a plenary teach-back session. The trainers then completed individual assessments and provided feedback to the trainees. To be a certified biosafety trained trainer, one must have attended all the training sessions including the teach-back session and must have passed the pre- and post-test. A self-structured questionnaire was used to access if the participants exhibited clear understanding of the training content, demonstrated being prepared to train, exhibited effective communication skills, applied adult learning principles, managed the training effectively, and ability to coordinate discussions/activities in an effective manner. An individual feedback was provided to each of the participants with the individual strengths and recommendations highlighted. Table 1 summarizes the steps in the TOT program.
HCW, healthcare worker; MSH, Management Sciences for Health; TOT, training of trainer; TWG, Technical Working Group.
The trained trainers then scaled up the biosafety trainings to facility laboratory HCWs in 2014 − 2015, under the technical guidance and coordination of the project in collaboration with the National Biosafety Office, biosafety TWG, and County medical Laboratories Coordinator (CMLC). Figure 5 shows the geographical coverage of counties reached with biosafety training of laboratory HCWs. The trainees were selected by the facility laboratory managers and submitted to the CMLC for verification, and finally submitted to the project. The training approach entailed didactic lectures using an interactive adult learning approach, with practical sessions during site visits in clinical laboratories to enhance skills acquisition. The participants developed work plans that set forth biosafety activities to be implemented in their facilities, which enabled them to identify biosafety gaps and establish improvement measures. The participants' knowledge of biosafety was tested before and after the training through pre- and post-test examinations. During the training, the project disseminated the biosafety guidelines, flyers, and the safety signages. Figure 3 shows a summary of the documents disseminated. The content in the biosafety curriculum would act as reference materials in the facilities.
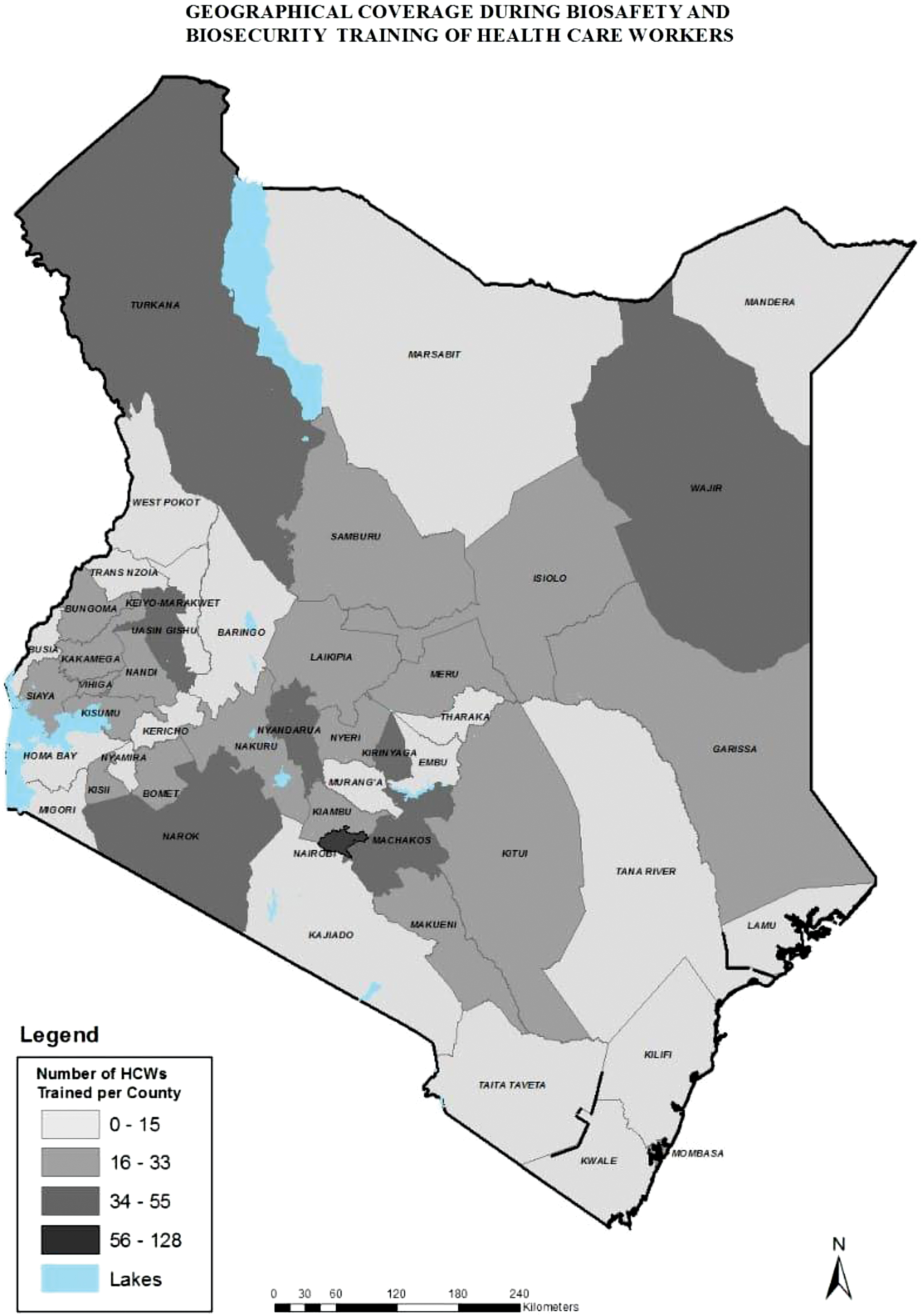
Geographical coverage of counties reached with biosafety training of laboratory HCWs. Geographical coverage of regions covered during biosafety trainings for HCWs. HCW, healthcare worker. Color images are available online.
The CMLCs also benefited from the biosafety trainings as it was envisioned that as members of the county and subcounty senior health management teams, they would play a crucial advocacy and linkage role between the HCWs and the CHMTs because they are strategically positioned to influence county resources allocation, especially purchasing materials required to support the adoption and maintenance of appropriate biosafety practices in the laboratories. The CMLCs would provide mentoring to facility staff during supportive supervision and include safety elements currently lacking in the laboratory safety section of the integrated supportive supervision tool.
To enhance infection prevention and control (IPC), the project used the training program as an avenue for distribution of PPE donated by USAID through CDC's Risk Communication and Emergency Response program. The PPE was distributed during the training and an opportunity to put new skills into practice on the job was created, since trainees had learned what the equipment was, and how and when to use it.
Mentorship Visits to Selected Health Facilities Following the Biosafety Trainings
Baseline assessments were conducted before the implementation of the trainings in each of the facilities, with follow-up visits done 3–6 months after the training. Figure 6 summarizes the steps in facility performance during the baseline and follow up assessments. The Stepwise Laboratory Quality Improvement Process Towards Accreditation (SLIPTA) checklist (Section 12) was used during the visits. The Kenya National Safety checklist, which had been used by the HCWs during the training for identifying their respective strengths and weaknesses at the facility level, was also utilized to guide discussions with laboratory staff. Data on facility characteristics and implementation of biosafety measures were collected using the structured questionnaires.
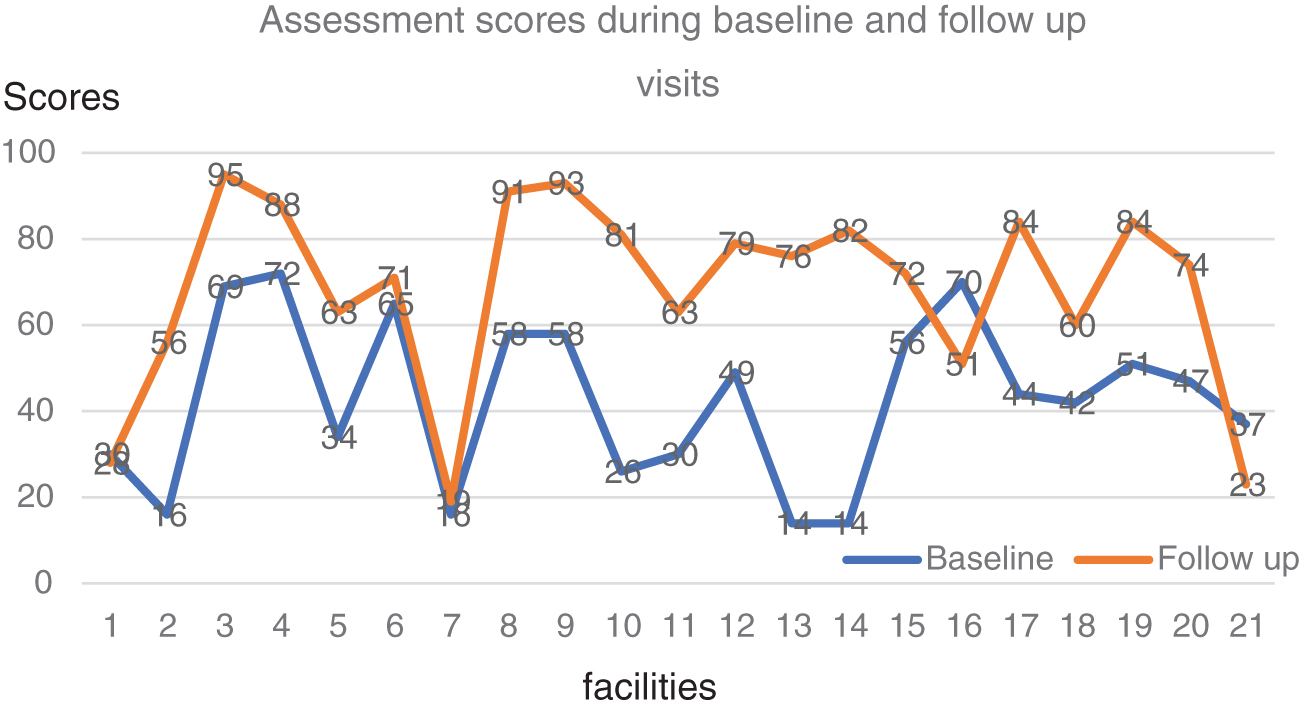
Facility performance during baseline and follow-up visits to health facilities. Assessment scores following biosafety baseline assessments and follow-up visits. Color images are available online.
To support and review the implementation of biosafety practices, the project team, CMLC, and the biosafety trainers conducted joint site visits in selected health facilities after the biosafety trainings. During the visit, the team used the work plans developed during the training to assess the extent to which the biosafety practices had been implemented. The team then mentored and guided the HCWs on areas of improvements and worked together with the facility to develop other action items for the next 6 months.
Monitoring the Impact of Biosafety Trainings
CMLCs and biosafety trainers conducted monitoring visits at least quarterly in selected facilities whose laboratory staff had been trained in biosafety. The purpose of the monitoring was to assess the impact of implementation of biosafety practices during follow-up site visits following the biosafety trainings. The site visits were conducted with technical guidance from MSH's Biosafety Technical Advisor. Primary data were collected using structured questionnaires and were analyzed descriptively and presented using charts.
The study was reviewed and approved by the Management Sciences for Health and US Centers for Disease Control and Prevention Associate Director for Science and categorized as nonresearch.
Results
Sensitization of Health Managers
Five sensitization meetings were carried out for a total of 264 health managers drawn from the MOH, staff of partner organizations, and educational and research institutions, between 2012 and 2014. The health managers sensitized included top management who contributed to the development and implementation of work plans for each of the facilities trained. They took part in the mentorship visit and lobbied for staff support from the facility in charges and administrators within the facilities that would contribute to the availability of resources needed for implementation of biosafety practices. The outcome of the sensitization meetings was evidenced by procurement of safety equipment and supplies such as spill kit contents, fire extinguishers, waste bins, and bin liners with support from hospital management. The project fostered equity by engaging relevant health managers in all regions in the country.
Biosafety TOTs
Two biosafety TOTs were conducted for 48 laboratory workers from 45 facilities and institutions in 37 of Kenya's 47 counties, attaing an 82% country coverage. The rest of the counties were not trained due to factors such as insecurity and poor roads. Of those workers, the majority, or 44 (92%) of the attendees, were from the MOH facilities, while the other 4 (8%) came from religious and nongovernmental organizations. Of the total participants, 98% qualified to be biosafety trainers after the TOT, and 1 trainee did not qualify to be a trainer because he or she did not attend all the sessions.
Training of Laboratory Health Workers on Biosafety
During the biosafety training period between 2012 and 2015, 40 training workshops were conducted for 1044 laboratory workers from 216 health facilities in 44 counties. The rest of the counties were covered by other partners. Of the total number of trainings, 14 (34%) were conducted at the facility level, while the rest were conducted at a central location with facilities clustered according to regional proximity. The participants trained were drawn from a variety of affiliations: MOH 941 (90%); faith-based organizations 65 (6.2%); non-governmental organizations 21 (2.0%); private facilities 15 (1.4%); and training institutions 2 (0.2%). To assess the knowledge gained, a pretest and post-test were conducted at the beginning and end of the training, respectively. The average pre- and post-test scores for the 40 workshops were 55% and 79%, respectively, with a mean gain of 24%.
Before the training, biosafety fact sheets were distributed to the potential participants and CHMTs in 11 counties to provide information on the training entailed. During the trainings, 87 boxes of PPE were distributed to 35 counties, and 600 laboratory safety signs were distributed to 31 facilities in 9 counties.
The CMLCs and National Biosafety Office provided oversight and coordination during the training under the technical guidance of the project. The MSH generated a biosafety training database with information about all the officers trained, contacts, facility, and when and where the training took place. The training database was shared with the National Biosafety Office, CDC, and implementing partners.
Mentorship and Monitoring in Selected Health Facilities Following the Biosafety Trainings
During the mentorship and mentoring visits, biosafety trainers conducted site visits in 51 selected health facilities after the biosafety trainings. Of those facilities, 21 (41%) were then followed up 6 months after the training. Performance in 18 of 21 (86%) facilities improved, while it declined in 3 facilities. This was due to high staff turnover caused by devolution of health services leading to shortage of staff, increased workload, and inability to implement safety procedures. In addition, one trainee did not qualify to be a biosafety trainer, due to weak training delivery skills, despite having been coached during the training. The trainee was encouraged to continue implementing and advocating for good biosafety practices at the facility level as he continued to improve his skills.
Biosafety improvements included improvised eye wash stations in 16 facilities (76%), availability of improvised biological/chemical spill kits in 17 (81%), buckets of sand in 15 (71%), fire extinguishers in 12 (57%), and provision of hepatitis B vaccination in 14 (66%); improved management support; and purchase of waste bin liners and counter books. Separation of patient reception and testing areas was observed in 18 facilities (85%), development of material safety data sheets in 18 (81%), documentation of postexposure prophylaxis procedures in 16 (76%), and proper waste segregation in 17 (81%).
Discussion
The sensitization of health managers before the training encouraged their commitment to the biosafety training program, which enabled them to purchase supplies and equipment needed for implementation of the biosafety practices, as evidenced by availability of waste bins and liners, registers for documenting incidence and occurrence exposures, and safety equipment following the training, which in turn served as a source of motivation to the trained staff.
“My motivation came from the support provided by the management and administration. When I asked for materials and supplies to be able to implement the activities in my work plan, the management supported us and that's why you see these changes today.”
“My colleagues are also very supportive, and we work as a team, all in agreement that safety is everyone's responsibility.”
Bota D, Mwangi J, Championing medical laboratory biosafety standards at facility level in Kenya. 29 Jan 2014. Cited 16 Apr 2019. Available from: http://www.msh.org/news-events/stories/championing-medical-laboratory-biosafety-standards-at-facility-level-in-kenya
Use of the ISO 15189 standard, which offers an overarching structure to laboratory operations, 12 was useful in implementation of structured safety standards including training HCWs on biosafety and biosecurity, components under section 12. Building the capacity of biosafety trainers complemented the existing workforce of trainers and fostered saturation of training in lower level facilities. The development of the biosafety TOT application form as part of the selection criteria enabled the selection committee to identify and train committed trainers who were able to impart knowledge and skills to the trainees during the laboratory worker trainings and mentorship and monitoring site visits. These findings mirror a study in Kenya that noted developing expert training for staff via a train-the-trainer model is an efficient way to initiate and maintain long-term change in biosafety practices in low-resource settings and regions. 9
Notable improvements in biosafety practices during the mentoring and monitoring visits, including innovations, for example, improvised eye wash stations and spill kits indicate that the training approach enabled the learners to understand biosafety principles and motivated them to apply the principles to address safety challenges. This mirrors a study in Argentina, 7 where it was observed that training in good laboratory practices promoted several aspects of biosafety, ranging from a change in work practices, to the use of standard protocols that promote motivation, and to working in a safe environment that minimizes opportunities for the spread of diseases. Mentorship following trainings is key to enhance application of the knowledge and skills learnt.
Poor personnel training increases the risk of a laboratory-acquired infection or other biological accidents in the laboratory, and may also contribute to improper pathogen accounting, storage, and transportation, which in turn could contribute to the illicit acquisition of biological agents by terrorists or would-be biocriminals. 4 In recent years, biosafety and biosecurity have received international recognition as key pillars of high-quality health services, as evidenced in the International Health Regulations, 2005 checklist of indicators for annual reporting to the World Health Assembly. QMS tools are being used to improve practices, with accreditation being a means to demonstrate that standards are met. 11 Implementation of laboratory standards is verified through the process of accreditation. All testing areas and processes are included, from preanalytical through postanalytical procedures, as are laboratory biosafety and ethics.
Challenges
This study happened when the country had just started devolution of health services from one national government to 47 county governments after enactment of a new constitution in Kenya. This led to high staff turnover and lack of implementation and sustainability of good biosafety practices initiated following the trainings in some facilities. The infrastructure in most facilities did not favor implementation of good biosafety practices such as installation of eye wash and emergency wash stations. The supply of safety commodities was also erratic. Chemical hygiene and biosecurity policies were lacking or inadequate. Fire management guidelines and materials were not widely available. Biosafety policy guidelines had not been disseminated to lower level facilities with need for a biosafety refresher training program in the country.
Recommendations
Biosafety and biosecurity are key pillars in achieving standards for driving laboratories toward accreditation and are crucial aspects of IPC in all health facilities. The biosafety curriculum should be introduced in preservice training of HCWs. Instilling these principles into future biosafety professionals and scientists before they enter the workforce will help establish an integrated culture of safety in health facilities. 6 Biosafety professionals and their professional societies should strongly advocate for inclusion of biosafety principles and practices as part of the life-sciences curriculum at the undergraduate and graduate university levels. 7
A biosafety refresher guide for in-service HCWs is needed, with refresher trainings offered at least annually, to reacquaint staff with skills and processes while introducing the new skills and processes required to improve operations. The guide should be made available online for easy accessibility. To complement this, continuous mentorships of trained personnel at the facility level and evaluation of biosafety programs are key for effective implementation of biosafety practices in resource-limited settings. The country also needs to develop a chemical hygiene strategy to provide a framework for handling expired chemicals. Dissemination of the existing biosafety policy guidelines at all levels will facilitate progress in this area.
In summary, laboratory staff must be effectively trained and supported by managers for them to demonstrate the skills needed to ensure a safer laboratory culture.
Conclusion
The study shows that biosafety trainings equip HCWs with adequate knowledge and skills to demonstrate improved IPC practices in the laboratory. For a successful training program, involvement of health managers at the national, county, and facility levels, with their coordination and administrative roles clearly defined, facilitated commitment to and ownership of the program and fostered the provision of materials and equipment required during implementation. Managers need to ensure that workers in clinical laboratories work in a safe and healthy environment that will promote the maximum utilization of the vital facilities. 8
Biosafety training should entail an interactive and adult learning approach to enhance skills acquisition. Biosafety mentorship and monitoring will support implementation of safety practices, and safety equipment can be improvised where resources are limited. This article has illustrated that the model can be adopted for conducting sustainable biosafety trainings to complement the accreditation process in Kenya and beyond.
Summary
Notable improvements, including innovations such as improvised safety equipment including eye wash stations, emergency stations, and biological spill kits, indicated that the training approach enabled the learners to understand biosafety principles and motivated them to apply those principles to address biosafety challenges.
Footnotes
Acknowledgments
We thank the MOH team led by Albert Bunyasi and facilities in the regions involved for their cooperation and collaboration. We also acknowledge the staff of the Strengthening Public Health Laboratory Services project for their teamwork and support: Catherine Gichimu, Lucy Oywaya, Ernest Ruto, Jackline Mboya, Mary John, Mary Kiwoi, Millicent Opiyo, Susan Wendot, Eric Wakaria, and Jane Muikamba. Gratitude goes to the CDC team for technical guidance and support during the project: Dr. Muthoni Junghae, Dr. Jane Mwangi, Dr. Daniel Kimani, Mboya Franklin, and Mercy Njeru. This article would not have been completed without the technical guidance, editing, and kind support from the MSH team: Spencer Ochieng, Judy Mwangi, Dr. Alaine Umubyeyi Nyaruhirira, and Dr. Barbara K. Timmons.
Ethical Considerations
No data used in this article represent personally identifiable information about a patient, staff, or other use of health services. As part of research ethics, the coauthors sought permission from the researchers who took part in the study. The researchers, in turn, sought clearance from the CDC and MSH. The researchers also assured the MOH of privacy and confidentiality. The findings of the article will be shared with the institutions concerned to guide them in planning and implementation.
Authors' Contributions
D.B. conceived and drafted the article. A.A. made a substantial contribution to the conception and design. J.W.W. critically revised the article for important intellectual content. J.O.O. made a substantial contribution in the analysis and interpretation of data. A.B. contributed to the conception and design.
Disclaimer
The views, thoughts, and opinions expressed in this publication belong solely to the author, and not necessarily to the authors' employer, organization, committee, or other groups or individuals. The findings and conclusions in this report are those of the author(s) and do not necessarily represent the official position of the funding agencies.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This project has been supported [in part] by the President's Emergency Plan for AIDS Relief (PEPFAR) through the Centers for Disease Control and Prevention (CDC) [under the terms of 5U2GPS001862-04].
