Abstract
The coronavirus disease 2019 (COVID-19) that begun in December 2019 has spread worldwide and is caused by a novel coronavirus named severe acute respiratory syndrome coronavirus 2 (SARS-CoV2). In the Philippines, the first case of COVID-19 was reported on January 20, 2020. Early in the SARS-CoV2 outbreak, clinical samples from suspected COVID-19 patients had to be sent to a reference laboratory in Australia for confirmation. However, as of November 11, 2020, there are now 164 Department of Health (DOH) licensed COVID-19 testing laboratories in the country. The Department of Health-Research Institute for Tropical Medicine (DOH-RITM) is the National Reference Laboratory for emerging and re-emerging infectious diseases. The RITM follows the guidelines set forth by the World Health Organization (WHO) when responding to outbreaks. One of its functions is to conduct risk assessment and proficiency testing to ensure and maintain the safety and high-quality performance of independent laboratories. The majority of the COVID-19 testing centers use a real-time reverse transcription-polymerase chain reaction platform. As of November 14, 2020, there are >404,000 confirmed cases of COVID-19 in the Philippines of which 83% are mild cases. It is worthwhile to mention that before full-scale implementation and issuance of a license to operate a COVID-19 testing regimen, both hospital and nonhospital-based diagnostic laboratories undergo a multistage process for COVID-19 laboratory assessment. The requirements prescribed in the DOH assessment tool for licensing a COVID-19 testing laboratory include but are not limited to the availability of safety equipment and trained laboratory personnel, the facility must be a BSL-2 laboratory, and must have an updated protocol including a biosafety manual. In this article, the biosafety concerns associated with establishing a COVID-19 testing laboratory and running COVID-19 clinical samples will be highlighted. In addition, mitigation control measures that can be put into place for aerosol-generating procedures and key performance indicators will also be identified.
Introduction
On November 14, 2020, the Johns Hopkins Coronavirus Resource Center has reported that the number of coronavirus disease 2019 (COVID-19) cases worldwide has reached the 53 million mark with >1.3 million global deaths. 1 In Southeast Asia, the Philippines has the second highest number of COVID-19 cases with >404,000 confirmed cases, with >362,000 recoveries, and >7700 deaths as of November 14, 2020. 2 The first case of COVID-19 was reported in a 38-year-old female who had a travel history to Wuhan. The second case was a 44-year-old male; he was a close contact of the first case, and was the first COVID-19 death in the Philippines, and the first mortality outside of China.3–5 The modes of severe acute respiratory syndrome coronavirus 2 (SARS-CoV2) transmission include direct and indirect contact, droplet and aerosol transmission. 6 The mechanism for direct contact is that infective particles are transferred directly from one person to a susceptible host. For indirect contact, infective particles are transferred from one person to another through a contaminated surface or object.7–9 For respiratory droplets (>5–10 μm in diameter) and aerosols (<5 μm in diameter), the infective particles settle on the mucosa (e.g., mouth, nose, and eyes) of a susceptible host.6,10
Multistage Process for COVID-19 Laboratory Assessment
It is well recognized that one of the risk mitigation strategies in COVID-19 containment includes routine diagnostic testing of clinical samples for COVID-19.11,12 The Department of Health-Research Institute for Tropical Medicine (DOH-RITM), the National Reference Laboratory for COVID-19 testing, had urged both hospitals and nonhospital-based diagnostic laboratories to adhere to the multistage process for COVID-19 laboratory assessment. The composite team responsible for evaluating the various laboratories is made up of authorized representatives from the RITM, Health Facilities and Services Regulatory Bureau (HFSRB), the Centers for Health Development-Regulation, Licensing, and Enforcement Division (CHD-RLED), and the World Health Organization (WHO). 13
The first step in this multistage process is self-assessment by the laboratory and application for a license to operate (Figure 1). This serves as an initial evaluation to determine the readiness of the laboratory to conduct COVID-19 testing. The requirements prescribed in the DOH assessment tool for licensing a COVID-19 testing laboratory include availability of safety equipment and trained personnel, appropriate design and layout of facilities, logistics and supplies, management system, safety controls, and streamlined workflow. The next step is onsite assessment. Here, a DOH assessment team will use the self-assessment tool designed by the RITM and provide the laboratory with an assessment of gaps in the technical and safety protocols. The laboratory will then implement responses that will address any identified gaps in the technical and safety protocols. Now, once the recommendations have been provided and addressed, the laboratory must submit evidence of their compliance before the next stage, which involves proficiency testing. At this point, the DOH-RITM will provide a test panel that consists of unknown samples. The results of the testing will be the basis for determining whether the laboratory is capable of testing for COVID-19. The final step is the full-scale implementation of COVID-19 testing with periodic evaluation and monitoring by DOH-RITM, HFSRB, and CHD-RLED.13–16
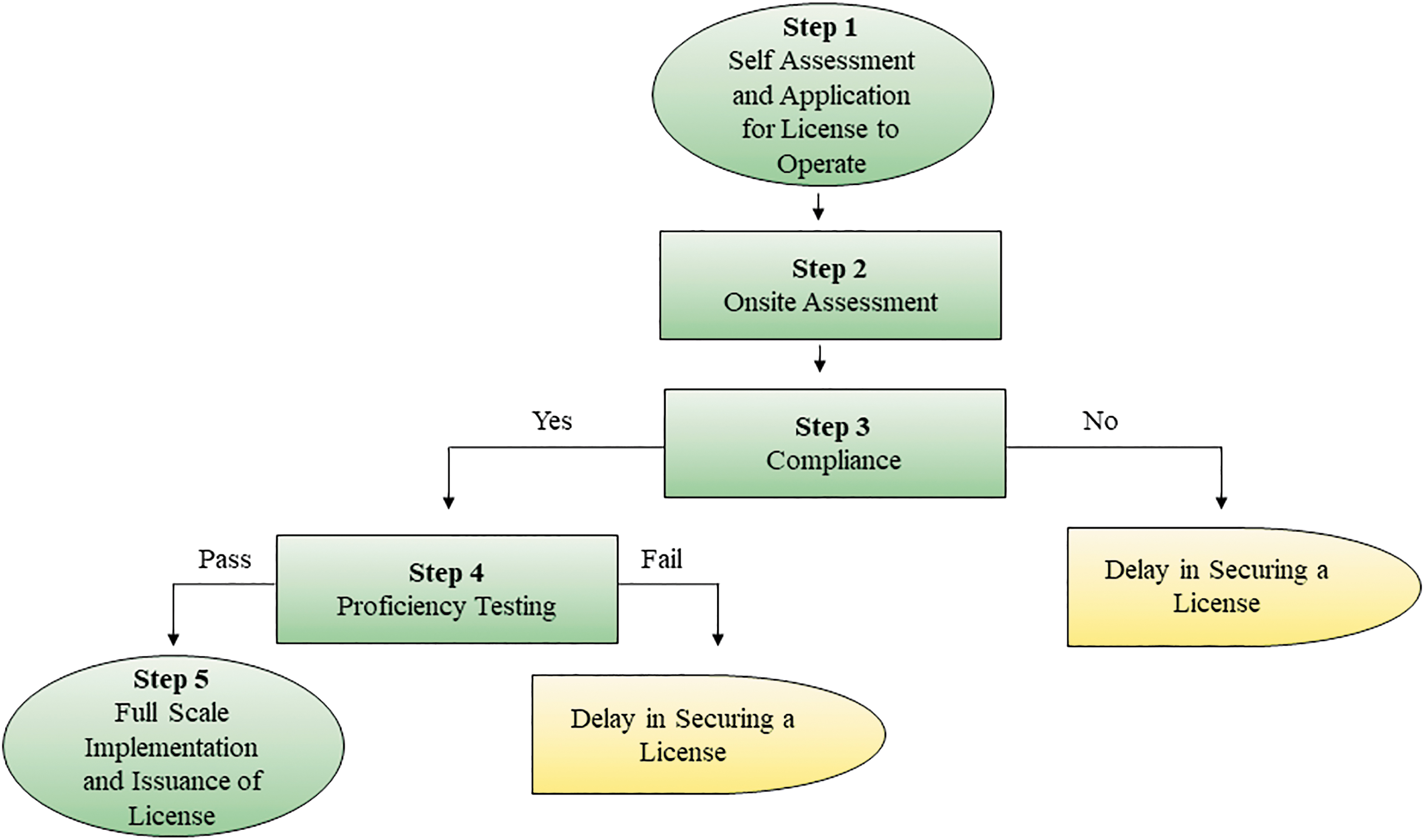
Multistage process for COVID-19 laboratory assessment by the Department of Health-Research Institute for Tropical Medicine (DOH-RITM).
Some of the causes for application delays in securing a license to operate a COVID-19 testing laboratory include inappropriate workflows, noncompliance with facility and equipment standards, and noncompliance related to biosafety practices. One of the biosafety concerns raised by the DOH assessment team is for the laboratory personnel to have appropriate trainings prescribed by the DOH-RITM. In particular, the laboratory personnel should undergo the following training modules:
Fundamentals of biosafety and biosecurity. Biological risk assessment. Mitigation controls (e.g., engineering, practices and procedures, administrative). Personal protective equipment. Specimen transport. Waste management (e.g., biosolid, sharps, liquid, and chemical). Decontamination and disposal. Emergency responses (e.g., biological spill).
The laboratory personnel involved in COVID-19 testing should likewise undergo fit testing for respirators such as N95 or better. The COVID-19 testing laboratories should have a manual of operations that include but are not limited to the standard operating procedures (SOPs) being implemented in the facility, policies and procedures on biosafety and biosecurity, infection prevention and control, records management, and preventive maintenance of equipment. 17 As part of the emergency contingency plans to address the shortage and ensure continuous availability of N95 masks, reprocessing and reuse of N95 are strictly observed.
COVID-19 Testing Laboratories
The requirements prescribed by DOH include that the facility must be a BSL-2 laboratory with controlled and adequate ventilation and with the prescribed air changes per hour. The biological safety cabinets (BSCs) should be properly maintained and certified annually by a third-party service professional. The SOPs on handling, processing, storing, and transport of clinical samples should be in place. 16 In addition, the laboratory personnel should undergo training on molecular diagnostics by a board-certified clinical pathologist. 17
Meanwhile, in an article by the Philippine Society of Pathologists, Inc. (PSP), they proposed to simplify licensing requirements for COVID-19 testing laboratories. In the position article, it was mentioned that the PSP is willing to assist DOH-RITM in conducting training modules in molecular pathology, biosafety and biosecurity for pathologists, medical technologists, and other relevant health care workers (HCWs). 18
As of November 11, 2020, there are 164 DOH-licensed COVID-19 testing laboratories nationwide, both government and private. 19 Of these, 42% (70/164) came from the National Capital Region where the number of cases had reached >191,000. 2 The majority of the COVID-19 testing laboratories utilized real-time reverse transcription-polymerase chain reaction (PCR) platform or cartridge-based PCR. It is noteworthy to mention that as of October 30, 2020, the Philippine Food and Drug Administration (FDA) has released a total of 303 COVID-19 test kits for commercial use. 20 Note that COVID-19 testing shall only be done in a DOH-licensed laboratory and shall only use FDA registered testing kits, reagents, and devices. 17
Health Care Workers
On April 28, 2020, the DOH had reported that 1336 HCWs had contracted COVID-19. Of these, 4% (47/1336) were medical technologists or laboratory scientists. 21 As of August 11, 2020, there have been 5551 HCWs in the Philippines infected with COVID-19, of whom 610 are active cases. 22 Recently, the DOH recorded >11,600 HCWs positive for COVID-19. Of these, 97.2% (11,340) have recovered, 0.6% (73) have died, and 2.2% (257) were active cases. Out of the 257 active cases, 43.1% (111) were mild cases, 43.6% (112) were asymptomatic, 0.8% (2) were in moderate condition, 8.2% (21) were in severe condition, and 4.3% (11) were in critical condition. 23 Wang et al. suggest that the considerable number of early health care infections and deaths may have been due to a combination of inadequate personal protective equipment (PPE) due to lack of awareness early in the epidemic, large-scale exposure to infected patients, shortage of PPE, and inadequate infection prevention training. 24 It is likely that the causes of workplace-acquired infections are similar in China and the Philippines. However, the possibility of community-acquired infection cannot be totally ruled out.
Laboratory Biosafety
The WHO and the US Centers for Disease Control (CDC) have provided interim guidances for laboratory biosafety related to COVID-19 on May 13, 2020, and August 15, 2020, respectively. The purpose of the interim guidance is to ensure that laboratory personnel involved in COVID-19 testing adhere to appropriate biosafety practices. This is in keeping with the goal that only properly trained and competent laboratory personnel should be authorized to handle and process clinical samples for COVID-19 testing.25,26
According to the WHO, nonpropagative diagnostic laboratory work such as nucleic acid amplification and sequencing should be conducted in a facility using procedures equivalent to those used in a BSL-2 laboratory. It is important to note that WHO recommended that for propagative work such as virus isolation, culturing, and neutralization, testing should be conducted in a BSL-3 laboratory. 25 Although the WHO interim guidance recommended a BSL-2 laboratory to conduct routine diagnostic testing for COVID-19, the DOH-RITM further increased its mitigation measures by recommending a BSL-2 laboratory with enhancements based on local risk assessment. The BSL-2 enhancements include a certified BSC, onsite autoclave, anteroom, directional airflow, negative pressure room, clean to dirty workflow, special practices (e.g., measurement of body temperature and health declaration), training and proficiency, restricted work areas, use of fit-tested National Institute for Occupational Safety and Health (NIOSH)-approved filtering facepiece respirator, and the use of dedicated laboratory shoes, shoe cover, and hair net. 27 In this way, the DOH ensured that the safety of the laboratory workers will be further protected.
As we all know, the backbone of the practice of biosafety is risk assessment.28–30 Thus, each hospital and nonhospital-based diagnostic laboratories should conduct site-specific and activity-specific risk assessments to identify and evaluate the risk of performing COVID-19 testing in their own institutions, to determine specific mitigation control measures to implement, and to review risks and control measures. 31
Some of the common laboratory activities that may expose laboratory personnel to potential hazards in relation to COVID-19 testing include the following:
Receiving clinical samples.
Routine analysis of blood or other body fluids.
Inactivation of respiratory samples during the process of cell lysis.
Extraction of viral RNA from clinical samples.
Disinfection of surface or equipment.
Disposal of wastes generated from COVID-19 testing.
Receiving Clinical Samples
The potential hazards associated with receiving clinical samples are contact with infected aerosols and droplets, splashes, or spills. Engineering control measures include the use of high efficiency air filters, protective barriers, pass box, and sealed specimen containers. For administrative control and practices, this must include staff training, good microbiological practices and procedures (GMPPs), and hand hygiene. 25 The use of proper PPE cannot be over emphasized, and this includes the use of face shield, gloves, goggles, gown or cover-all suit, and a fit-tested NIOSH-approved respirator or better.
Routine Analysis of Blood or Other Body Fluids
For routine analysis of blood or other body fluids, there are many potential hazards associated with aerosol-generating procedures such as pipetting, shaking, uncapping specimen containers, and vortex mixing. These exposure hazards include aerosols, droplets, splashes, and spills. Engineering strategies to control these factors include the use of safety equipment such as a BSC, sealed centrifuge rotors, and sealed safety cups. Administrative controls that can be put into place include proper labeling, signages, SOPs, and staff training. Again, proper donning and doffing of PPE cannot be overemphasized.
Inactivation of Respiratory Samples
The potential hazards associated with inactivation of respiratory samples during the process of cell lysis are aerosols, droplets, splashes, and spills. Given this, the engineering controls that can be placed include BSC class II A1 or A2, safety centrifuge buckets, pass box, filtered tips, and sharp containers. The laboratory design must include an anteroom, inward directional airflow, sink, and an emergency eyewash station within 10 s of travel. Administrative controls should include signages, SOPs, and staff training. GMPPs as well as hand hygiene mitigate the risk of exposure.
Extraction of Viral RNA from Clinical Samples
For viral RNA extraction from clinical samples, the potential hazards associated with aerosol-generating procedures are aerosols, droplets, splashes, and spills. Mitigation controls include the use of safety equipment, well-lighted and adequate spaces, compliance with administrative controls and standard precautions, and proper use of PPE.
Disinfection of Surface or Equipment
For disinfection of surface or equipment, the engineering controls include the use of high efficiency air filters and other PPE as appropriate. Note that appropriate disinfectants with proven activity against enveloped viruses (e.g., SARS-CoV2) should be used for the recommended contact time at the correct dilution. 25
Disposal of Wastes Generated from COVID-19 Testing
On disposal of wastes generated from COVID-19 testing, the WHO mentioned that there is no evidence to suggest that laboratory wastes related to COVID-19 testing need any additional packaging or disinfection procedures. 25 Locally, infectious wastes that include COVID-19–related wastes are not mixed with other wastes during handling and collection. 32 Biohazardous wastes are autoclave at 121°C, 15 psi for 30 min before disposal. Autoclave tape and/or biological indicators are used to monitor performance of the autoclave.
The other activities that may expose personnel to biohazards include sharps, centrifugation, mixing by vortex, and type of sample (e.g., blood, lower respiratory samples, and upper respiratory samples).
Moreover, regarding the hierarchy of controls and the types of measures that can be used to protect laboratory personnel, note that a combination of engineering controls, administrative controls, practices, and PPE mitigates the risk of exposure.27,33,34 It is worthwhile to mention that among these, the most effective mitigation measure is engineering controls, because it serves as the primary barrier against infection for laboratory personnel, work environment, and the community.
Performance
It is best that the following regular performance evaluation must be conducted:
Auditing (e.g., annually) and inspection visit (e.g., announced or unannounced) by safety officers, infection control personnel, and pathologists.
Coaching and counseling by the immediate supervisor and trained counselors.
Strict compliance to institutional policies on infection control standards and laboratory workflows.
Health checks of the laboratory personnel and other HCWs, which include measurement of body temperature, health declaration, and COVID-19 screening.
Practicing other proven risk mitigation strategies such as physical distancing, limiting social engagements in the community, occupational health program, alert cards, procedures for reporting near misses, incidents, and accidents.
The findings of auditing activities, inspections, and personnel health status must be reported to the immediate supervisor or head of the laboratory, so that timely intervention can be provided in the area where the safety link is the weakest. In this way, a culture of safety will be embodied by each member in the institution.
Summary
Considering the COVID-19 pandemic, more hospitals and nonhospital-based diagnostic laboratories had set up their own testing center after the multistage process for COVID-19 laboratory assessment by DOH-RITM. Each institution had conducted site-specific and activity-specific risk assessment to identify the potential hazards in the workplace. A combination of engineering controls such as the use of safety equipment that serves as a primary barrier, administrative controls such as policies and SOPs, GMPPs, and proper donning and doffing of PPE mitigate the risk of exposure.
Conclusion and Recommendations
Given that we cannot lock up SARS-CoV2 and quarantine HCWs, however, we can:
Foster a culture of safety by providing training to laboratory personnel and other HCWs, encouraging open communication, developing positive reporting process, and leading by example;
Review risks and control measures to make room for improvement; and
Run more samples to quickly isolate infected patients and control transmission.
In agreement with the interim guidance for laboratory biosafety by the WHO and the CDC, it is recommended that:
BSCs should be properly maintained and certified when running COVID-19 clinical samples;
Policies and protocols including a biosafety manual should be regularly updated;
Laboratory personnel should be properly trained in biosafety, biosecurity, and molecular diagnostics by a safety officer and pathologist;
Droplet protection, GMPPs, and waste management should be strictly implemented; and
Donning and doffing of proper PPE should be carefully observed.
Ethics Approval
Considering the article type (Brief Review Article), ethical approval was not sought for this article.
Footnotes
Author Disclosure Statement
No competing financial interests exist.
Funding Information
No funding was received for this article.
