Abstract
Purpose:
Oral and injectable testosterone replacement therapies have been reported to increase blood pressure (BP) in hypogonadal men. Less is known about the effects of topical testosterone formulations on BP.
Materials and Methods:
We studied the effects of a new oral testosterone undecanoate (TU) (Kyzatrex™) and a topical testosterone gel (Androgel™) on BP and heart rate, in a 2:1 randomized trial of men with hypogonadism. Digital BPs and clinical assessments were obtained at baseline and at 90, 180 and 365 days.
Results:
There were 314 men randomized (mean age 49.7 years, 82% white, and 35% with treated hypertension). The mean changes from baseline in systolic BP were 2.2 mmHg (95% confidence interval [CI] 0.6–3.7) on the oral TU and 3.1 mmHg (95% CI 1.0–5.1) on the topical testosterone at 90 days. Similar findings were seen after 180 and 365 days. Heart rate increased by 4.3 and 2.7 beats/min at 90 days on oral and topical testosterone, respectively. For those men with treated hypertension, changes in systolic BP in the oral TU group were higher compared with those without hypertension, whereas those on topical testosterone had similar increases in systolic BP regardless of hypertension status. Outlier analyses for systolic BP at 90 days demonstrated a higher proportions of subjects with increases of 5 and 10 mmHg postbaseline on topical testosterone gel versus this oral TU.
Conclusions:
These data demonstrate that the overall effects of the oral TU on BP were comparable with a topical testosterone. In men without hypertension, the oral TU induced smaller changes in BP compared with topical testosterone.
Clinical Trial Registration
(https://www.clinicaltrials.gov/)
Introduction
The most common testosterone therapies for male hypogonadism (also known as testosterone deficiency) include injections, transdermal gels, and now oral formulations. 1 Testosterone itself has limited oral bioavailability. Long-chain fatty acid esterification of testosterone to create testosterone undecanoate (TU) allows for absorption through the intestinal lymphatic system and bypasses first-pass metabolism in the liver. 2 Testosterone is then liberated from TU by endogenous nonspecific esterases. 2
Oral formulations of TU have been developed to provide average serum testosterone levels in the eugonadal range (typically 300–1000 ng/dL or 10.4–34.7 nmol/L) and avoid peak concentrations above 1500 ng/dL. Some observational studies and meta-analyses have demonstrated no change or small reductions in blood pressure (BP) over time.3,4 However, randomized clinical trials focused on BP as a primary endpoint have demonstrated that testosterone formulations have increased BP,5-8 but the mechanism of these increases have not been entirely elucidated. Previously there has also only been one clinical trial evaluating the effects of an oral TU versus a topical gel on BP. 5 Hence, we compared the effects of a new oral TU therapy (Kyzatrex™) versus the testosterone gel (Androgel™) in a long-term randomized trial that assessed BP along with standard clinical and laboratory parameters.
Materials and Methods
The study, which followed guidance by the U.S. Food and Drug Administration (FDA), was a randomized open-label multicenter active-controlled trial with an untreated screening period, baseline visits to assess BP and heart rate before administration of study medication, and postrandomization visits over 12 months after initiating the oral TU or topical testosterone gel. Eligible study participants were randomized 2:1 to oral TU or topical testosterone and stratified by study center. Seated clinic BP measurements were performed in duplicate at all study visits.
For those randomized to oral TU, the initial dose was 600 mg (400 mg with the morning meal and 200 mg with the evening meal). Plasma (oral TU) and serum (AndroGel) testosterone was measured using validated liquid chromatography with tandem mass spectrometry (LC-MS/MS); plasma samples were collected in NaF/EDTA tubes, which were chilled immediately after collection through processing. The LC-MS/MS methods were validated over the range of 10–3000 ng/dL of testosterone; the coefficient of variation (%CV) for the plasma assay ranged from 5.4% to 1.3% and the %CV for the serum assay ranged from 7.2% to 1.6%.
Based on thresholds of morning plasma testosterone between 3 and 5 h post-morning dose [<235 ng/dL (8.2 nmol/L) to titrate upward or >1120 ng/dL (38.9 nmol/L) to titrate downward], dose decreases to 200 mg once daily or increases to a maximum of 600 mg in the morning and 400 mg in the evening took place at the 28- and 56-day visits during the first 90 days of the trial. The testosterone gel was applied at a starting dose of 40.5 mg once daily. Serum testosterone levels at days 14 and 42 were used to determine the need to titrate at 28 and 56 days, respectively, per the label of Androgel. If the serum testosterone level was <350 ng/mL, the daily dose was increased by 20.25 mg (1 additional pump). If the serum testosterone was >750 ng/dL the daily dose was decreased by 20.25 mg. After the initial 90-day treatment period, changes in dose were also allowed on days 180 and 270 for both treatment groups.
Study participants
All participants were men between 18 and 65 years of age, inclusive, with documented hypogonadism as defined by a below normal serum testosterone and at least one sign or symptom of testosterone deficiency. The total serum testosterone level was required to be ≤281 ng/dL (lower limit of normal range for the immunoassay method used for screening, as required by the U.S. FDA) on two consecutive blood samples obtained between 7 and 10 am on separate days, at least 3 days apart either in individuals naive to testosterone therapy or after at least 8 weeks of washout of current testosterone therapy (washout periods of 6 months were required for testosterone implants). Also required was that there was no change in medications, including antihypertensive agents, within 3 months before enrollment.
Subjects with uncontrolled hypertension (clinic BP >150/90 mmHg) were excluded from participation based on FDA guidance. Other key exclusion criteria were the use of any medications or clinical conditions that could affect absorption or levels of TU; hemoglobin A1c >8%; hemoglobin <11.0 or >16.0 g/dL; serum transaminases more than two times the upper limits of normal; estimated glomerular filtration rate of <60 mL/min/1.73 m, or prostate-specific antigen >2.5 ng/mL, and/or an abnormal prostate gland on palpation. Study participants using any drug that could interfere with the measurement or assessment of serum androgen levels or had antiandrogenic properties must have been stopped for at least 1 month before study entry (6 months in the case of dutasteride).
The trial was conducted in accordance with Good Clinical Practice requirements, as described in the current revision of International Conference on Harmonization of Technical Requirements of Pharmaceuticals for Human Use (ICH) guidelines and the Declaration of Helsinki. The study protocol and informed consent forms were reviewed and approved by the Copernicus Group Institutional Review Board (Cary, NC). Before any study procedures could occur, a written informed consent was obtained from each study participant.
Safety assessments
Clinical evaluation and vital signs were assessed at baseline and at each scheduled clinical trial visit. At each clinic visit after the screening visit, all study participants were queried about adverse events using nonleading questions and a symptom-directed physical examination was performed as indicated clinically. Laboratory tests, including average plasma (for Kyzatrex) and serum (AndroGel) testosterone concentrations measured by mass spectrometry, were also performed.
BP monitoring
BP was monitored manually in the clinic at the baseline and post-treatment study visits. The clinic measurements were made in the seated position in duplicate after 5 min of rest and using appropriately sized cuff and bladder with a digital recorder. The same cuff was used for all visits for each subject. Any study participant with a baseline clinic average BP >150/90 mmHg was withdrawn from participation in the trial. At the 90-day visit, subjects in both treatment groups participated in an overnight stay. Seated BPs and heart rates were recorded within 60 min before dosing and at 4-h intervals (4, 8, 12, 16, 20, and 24 h postdose). To avoid the influence of a meal upon BP, predose BP was assessed in the fasting state.
Statistical analyses
Systolic and diastolic BPs were summarized with means and 95% confidence intervals (CIs). The change from baseline for the average BP was a key safety end-point of the study. Key secondary end-points were the changes from baseline to days 180 and 365 for the average systolic BP. Other assessments included changes from baseline in the diastolic BP and heart rate at days 90, 180, and 365. In addition, the BP and heart rate changes were evaluated in subgroups of study participants with and without treated hypertension at baseline. Categorical distributions of changes in systolic and diastolic BP on each treatment group were calculated for bins of ≤−20, −20 to ≤−15, −15 to ≤−10, −10 to ≤−5, −5 to <0, 0 to <5, 5 to <10, 10 to <15, 15 to <20, and ≥20 mmHg. The incidence of adverse events was tabulated in all participants who received at least one dose of study drug (safety population).
Comparisons of Kyzatrex with Androgel were performed using a mixed model for repeated measures (MMRM) approach as subjects were assessed across multiple visits. The model included all postbaseline visits where fasting BP assessments were taken predose within the clinic. All changes from baseline were calculated using least square (LS) mean differences and 95% CIs. Hypertension status and treatment were included as binary factors and baseline BP was included as a continuous covariate.
Sample size calculation
The sample size of this trial was based on requirements for demonstrating efficacy as the percentage of subjects within the normal range for their 24-h average testosterone concentration after 90 days. The FDA required that at least 100 subjects in the oral TU treatment arm reach 12 months of study drug exposure. Assuming a dropout rate of 50% over the course of the 12-month treatment period, 200 subjects were required to be randomized to the oral TU group.
Results
Subject disposition and baseline characteristics
A total of 314 subjects were randomized into the trial: 214 subjects in the oral TU group and 100 in the testosterone gel group. A total of 187 (87.4%) subjects in the oral TU group and 90 (90%) subjects in the testosterone gel group completed the day 90 visit. The distribution of doses in the oral TU group on day 90 (after titration) was as follows: 61.0% at the starting dose; 7.9% up-titrated; 31.0% down-titrated. The distribution of doses in the testosterone gel group at day 90 was 24.4% at the starting dose, 68.8% up-titrated and 6.7% down-titrated. Furthermore, 162 (75.5%) subjects in the oral TU group and 76 (76%) subjects in the testosterone gel group completed the day 365 visit. The most common reasons for early termination were loss to follow-up/withdrawal by the subject (18.7% in oral TU and 17% in testosterone gel) and adverse events (4.7% in the oral TU group and 5.0% in the testosterone gel group).
Demographic and baseline characteristics of study participants are shown in Table 1. These characteristics, including baseline BPs, were comparable for each treatment group. The mean age at baseline was 49.7 years, 82% were white, and 18% were non-white. Thirty-five percent (110 of 314) of the study participants were treated hypertensive patients. Thirty-two (10.2%) subjects were initiated on new antihypertensive therapy or with a dose increase in antihypertensive therapy over the 365 days of treatment (25 (11.7%) in the oral TU group and 7 (7.0%) in the testosterone gel group). Fourteen percent (44 of 314) of the study cohort had type 2 diabetes mellitus. The average clinic BP was 126/79 mmHg.
Characteristics of the Patient Population at Baseline (n = 314)
BP, blood pressure; TU, testosterone undecanoate.
Signs and symptoms of androgen deficiency included reduced sexual desire (libido) in 258 (82.2%) subjects, decreased spontaneous erections in 221 (70.4%) subjects, and decreased energy, motivation, initiative, and self-confidence in 269 (85.7%) subjects. At least one symptom of androgen deficiency was required for enrollment. Half (n = 157) of the subjects had received prior testosterone replacement therapy.
In the oral TU group, the percentage of study participants achieving a normal testosterone after 90 days of treatment was 90% [plasma Cavg0–24 = 378.5 ng/dL (13.1 nmol/L)]. The mean maximal testosterone concentration was 946.5 ng/dL in the oral TU group, as determined from 24-h pharmacokinetic measurements at day 90. In the topical testosterone gel group, the percentage of subjects achieving a normal testosterone after 90 days of treatment was 71.3% [plasma Cavg0–24 = 502.7 ng/dL (17.4 nmol/L)].
The mean maximal testosterone concentration in the topical testosterone group was 788.6 ng/dL (27.4 nmol/L) The average dihydrotestosterone after 90 days of treatment for oral TU and testosterone gel was 77.1 and 72.5 ng/dL (2.7 and 2.5 nmol/L), respectively.
BP and heart rate
Change from baseline in the systolic BP in the oral TU and testosterone gel groups is shown in Table 2. At day 90, the clinic systolic BP rose 2.2 mmHg in the oral TU group and 3.1 mmHg in the testosterone gel group (both results significantly higher than baseline, p < 0.01 but not between groups). There were similar trends observed at 180 and 365 days of the treatment period. Lesser effects were seen for the diastolic BP in both treatment groups although there were statistically significant increases observed in the topical testosterone gel group at 180 and 365 days (Table 2).
Blood Pressures and Heart Rates After Oral Testosterone Undecanoate and Testosterone Gel
Based on analysis of variance with treatment as fixed effect. Censored for changes in BP medications.
p ≤ 0.01.
p ≤ 0.05 for within group change from baseline.
CI, confidence interval.
Small increases in the heart rate (0.4–4.3 beats/min) were observed in both testosterone treatment groups (Table 2). The analysis (MMRM) after 365 days demonstrated a statistically significant difference (LS mean difference = −1.4 mmHg, p-value = 0.0011) when comparing diastolic BP changes with oral TU compared with testosterone gel. Comparison of mean changes in systolic BP between TU and topical groups was not statistically significant (LS mean difference = −0.5 mmHg, p-value = 0.44) (Supplementary Table S1).
The results for systolic and diastolic BPs are shown in Figure 1. These demonstrate small, but consistently higher systolic and diastolic BP in the subjects randomized to the topical testosterone gel compared with the oral TU at all time points. An analysis of the categorical changes from baseline in the BP at 90 days of therapy show that there were a greater proportion of subjects with increases of 5, 10, and 15 mmHg systolic and 5 and 10 mmHg diastolic in those subjects randomized to the topical testosterone gel than to those randomized to the oral TU (Fig. 2). Cumulative distribution curves of changes from baseline in 24-h mean in-clinic systolic and diastolic BP are shown in Supplementary Figure S1. For systolic BP, there was an increased frequency of increases in the ranges of 1–15 mmHg for the topical testosterone gel compared with the oral TU. For diastolic BP, there was increased frequency of increases in the ranges of 2–10 mmHg for the topical testosterone gel compared with the oral TU.

BP postrandomization on oral testosterone decanoate (TU) and testosterone gel over 1 year. The Upper panel
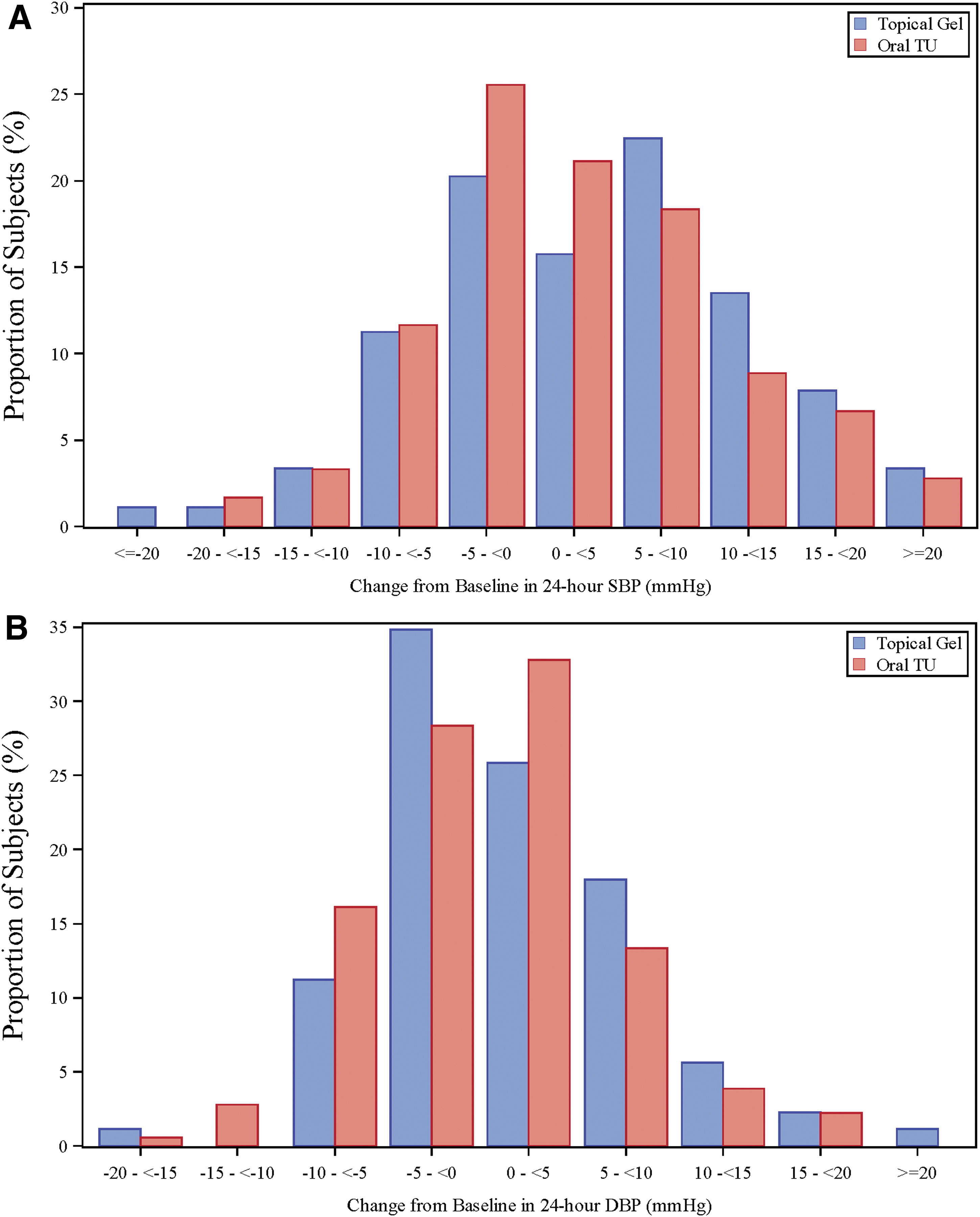
Categorical changes in BP in each treatment group after 90 days of therapy. At 90 days, subjects were in-house and BP were collected at several time intervals and averaged for 24 h and compared with two baseline visits. The upper panel
BP changes in subgroups with treated hypertension
Changes from baseline in BP and heart rate at 90, 180, and 365 days in study participants with and without treated hypertension are shown in Table 3. At day 90, changes in subjects with hypertension were 4.3/0.4 mmHg in the oral TU treatment group and 3.3/1.9 mmHg in the topical testosterone gel treatment group. In those subjects without hypertension, changes in BP in the oral TU group were 1.1/0.7 and 2.9/0.6 mmHg in the topical testosterone gel treatment group. At day 180 or day 365 versus 90 days, the changes were slightly less for systolic BP in those patients with hypertension, and were similar for diastolic BP in both treatment groups. In subjects without hypertension, changes were similar at days 90, 180, and 365 for those treated with oral TU and mixed for those treated with testosterone gel (lower systolic BP and higher diastolic BP as shown in Table 3).
Changes from Baseline in Blood Pressure at Day 90, 180, and 365 in Study Participants With and Without Treated Hypertension
Values are least square mean based on a mixed model repeated measures analysis with visit, prior treatment, baseline antihypertensive treatment status as fixed effects and study participant as a random effect. Censored for changes in antihypertensive medication. Changes shown are changes from baseline.
Defined as a medical history of hypertension and on antihypertensive medications at baseline.
p ≤ 0.01 versus baseline.
At baseline, the serum hemoglobin values were similar in each treatment group (Supplementary Table S2): 14.4 ± 0.9 g/dL for oral TU and 14.2 ± 1.0 g/dL for the topical testosterone group. We found that hematocrit values were unstable when measured at a central laboratory and baseline and, therefore, were not able to pursue an analysis of hematocrit in the trial. There were modest increases in the serum hemoglobin levels in each of the treatment groups (Supplementary Table S2). To determine whether BP changes were related to hemoglobin levels, subjects were divided into tertiles at 90 days based on hemoglobin concentrations. No relationship was observed in either drug treatment group for systolic BP changes from baseline among hemoglobin tertiles during the 24-h in-clinic visit (Supplementary Table S3).
Discussion
The results of this randomized trial of testosterone therapies demonstrated that both the oral TU formulation Kyzatrex and the topical testosterone gel Androgel were associated with small (2–3 mmHg) increases in systolic BP during a 1-year period of testosterone therapy in hypogonadal men. For subjects randomized to oral TU the change in mean diastolic BP were minimal (≤1 mmHg), whereas those randomized to testosterone gel showed increases of 1–3 mmHg. The impact of the two testosterone formulations on BP reached a plateau by 90 days and showed little change between 90 and 365 days of daily therapy. In both treatment groups, there were clinically insignificant increases in the heart rate of 1–4 beats/min. Small changes in serum hemoglobin were observed in both the oral TU and topical testosterone treatment groups, but there was no association between increases in hemoglobin and increases in BP in either treatment group.
For subjects randomized to the oral TU, a larger increase from baseline BP was observed in men taking antihypertensive drug therapy compared with those without hypertension, consistent with previous reports with other testosterone therapies.5,7,8 In the topical testosterone group the mean changes from baseline in systolic BP were similar for those with and without hypertension at day 90.
Changes from baseline in diastolic BP were small and similar for those with and without hypertension in the oral TU group but mixed for those treated with topical testosterone gel. For example, at 90 days, the average change in BP in those men on antihypertensive therapy in the oral TU group was 4.3/0.4 mmHg, whereas for those not taking antihypertensive therapy a substantially smaller and clinically insignificant change of 1.1/0.7 mmHg was observed. This finding was also observed in a single-arm study of this oral TU using ambulatory BP monitoring. 6 For the topical testosterone gel group, the average change in BP in those men on antihypertensive therapy was 3.3/1.9 mmHg, whereas for those not taking antihypertensive therapy, the increase was similar, 2.9/0.6 mmHg.
The biological mechanism for changes in BP with testosterone therapy is not well understood, particularly the observed differences between men with treated hypertension versus those with normotension.5,7,8 Data from pre-clinical studies have shown that testosterone may result in upregulation of a number of pressor substances, including norepinephrine, angiotensin II, and endothelin-1 along with some attenuation of vasodilator action (e.g., adenosine). 9 It is possible that these effects may be more prominent in men with underlying hypertension than those without hypertension. It has also been reported by Dalmasso et al 10 that there are differential effects of testosterone on BP in the spontaneously hypertensive rat (SHR) depending on the age of the SHR—an effect that may be mediated in part by the renin–angiotensin system. In contrast, a clinical study by Yaron et al 4 demonstrated that BP drifted downward after testosterone treatment and was associated with a small decline in the pulse wave velocity, a marker of arterial stiffness.
The increases in BP were modest in both treatment arms and showed no cumulative effect over the course of 1 year of therapy. Previous studies with two other oral TUs5,7 and testosterone enanthate (subcutaneous injection) 6 reported larger numerical increases in BP than the changes seen in this study. For example, change in the clinic BP on the oral TU formulation Tlando™ was 4.8/1.7 mmHg; 7 increases in systolic BP for subjects receiving antihypertensive medication was 5.5 mmHg. In a clinical trial that compared the oral TU Jatenzo™ with the topical testosterone gel Axiron™, changes in 24-h mean ambulatory systolic BP were 4.9 mmHg versus 0.2 mmHg, respectively; changes in men with hypertension were also found to be higher than in the men without hypertension. 5
In prospective studies of large populations, a systolic BP increase of 3 mmHg or higher has been shown to have strong relationships with adverse cardiovascular events, particularly heart failure and stroke. 11 The clinical importance of small increases in BP in hypogonadal men observed in this study is less clear since men with testosterone deficiencies have increases in cardiovascular risk 12 and there are data that suggest that normalizing testosterone levels in hypogonadal men may be associated with lower rates of cardiovascular morbidity than men who stay at low testosterone levels. 13 In addition, large analyses over intermediate to long periods of time3,14 have shown no detrimental effects of testosterone therapy from a cardiovascular safety perspective.
Nevertheless, in hypogonadal men who require testosterone therapy, particularly those with a history of hypertension, careful clinical assessment for possible increases in BP remains important in clinical practice.
Conclusions
The new oral TU, Kyzatrex, induced small increases in BP by 90 days of administration that did not increase further over the remainder of the 1-year study period. These changes were similar to those observed with a topical testosterone formulation. There were also small clinically unimportant increases in heart rate (<5 beats/min) in both treatment groups. Study participants with a history of hypertension taking antihypertensive therapy had larger increases in BP after chronic oral TU therapy than those without hypertension. In the oral TU group and in contrast to the topical testosterone gel group, hypogonadal men who were not receiving antihypertensive medication had negligible changes in BP.
Footnotes
Acknowledgments
All data were available to all authors for review and analysis. Biostatistics consultation, clinical pharmacokinetics, and statistical programming services were provided by Apex Biostatistics and PharStat, Inc., located in Research Triangle Park, NC.
Disclaimer
The authors are solely responsible for the design of the study, all study analyses, and the drafting and editing of the article and its final contents.
Author Disclosure Statement
Dr. White has received consulting fees from Marius Pharmaceuticals, Inc; Dr. Morgentaler has received consulting fees from Marius Pharmaceuticals, Acerus Pharmaceuticals, and speaking honoraria from Bayer; Drs. Bernstein, and Dhingra are employees of Marius Pharmaceuticals, Inc.
Funding Information
The clinical study was sponsored by Marius Pharmaceuticals, Inc., Raleigh, NC.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
