Abstract
Background:
Recent findings indicate that testosterone deficiency (TD) affects key metabolic processes, such as lipid and carbohydrate metabolism. Apolipoprotein E (APOE) is the natural ligand for the low-density lipoprotein receptor (LDLR). The natural APOE isoforms APOE3 and APOE4 have a high affinity for LDLR, while APOE2 exhibits impaired binding to the receptor and an inability to promote lipoprotein remnant clearance. Previous work indicated that LDLR modulates the effects of TD on adipose tissue mitochondrial metabolic activity. Here, we investigated how APOE2 or APOE4 expression influences the effects of TD on plasma lipid and glucose homeostasis and white adipose tissue and brown adipose tissue (BAT) mitochondrial metabolic activity.
Materials and Methods:
APOE2 knock-in (Apoe2knock-in) and APOE4 knock-in (Apoe4knock-in) mice were subjected to surgical castration or sham operation. Mice were fed western-type diet for a period of 12 weeks, and a series of analyses were performed on live animals or specimens collected at the end of the study.
Results:
TD results in body weight loss only in Apoe4knock-in mice. Castrated Apoe2knock-in mice show much reduced BAT mitochondrial uncoupling protein 1 (Ucp1) and CytC expression, which may explain the body weight difference between castrated Apoe2knock-in and Apoe4knock-in mice. The reduced body weight gain in castrated APOE4-expressing mice did not alleviate the already much impaired glucose tolerance of these mice or their hepatic triglyceride levels. Quite strikingly, in both mouse strains hypogonadism brings about negative effects on lipoprotein metabolism, manifested by different disturbances.
Conclusions:
TD is associated with distinct metabolic abnormalities, which depend on the APOE isoform expressed.
Introduction
Testosterone (T) is a sex steroid hormone produced and secreted into circulation by Leydig cells in a multistep process that utilizes cholesterol as the initial substrate. 1 It can also be produced by the adrenal gland, white adipose tissue (WAT), skin and brain, where it exerts numerous autocrine effects. 2 In addition to defining gender, T plays important roles in key metabolic processes such as carbohydrate, lipid, and protein homeostasis, oxidative stress, inflammation, endothelial dysfunction, 3 and adipose tissue and brain mitochondrial energy metabolism.4,5
The importance of T in human metabolism was documented in prostate cancer patients under androgen deprivation therapy, where testosterone deficiency (TD) was etiologically associated with a number of metabolic disorders, including obesity, metabolic syndrome (MetS), dyslipidemia, endothelial cell dysfunction, vascular disease, insulin resistance, and type 2 diabetes mellitus (T2DM).6–10 To the contrary, testosterone replenishment therapy (TRT) improves lipid and glucose metabolism, while it results in body mass index reduction. 11 It should be noted that excess WAT associated with morbid obesity also leads to decreased T levels. 12
Obesity is a pathological condition associated with a marked accumulation of visceral WAT as a result of an imbalance between energy intake and expenditure. 13 Sedentary lifestyle and excess fat and glucose accumulation contribute to the development of obesity. Adipose organ consists of different fat subtypes performing distinct functions. WAT, an endocrine organ secreting numerous adipokines that impact the physiology of many diseases, stores lipids in the form of triglycerides.
In contrast, brown adipose tissue (BAT), another type of fat with endocrine function, burns lipids through adenosine triphosphate (ATP) production and nonshivering thermogenesis. WAT may be metabolically activated into BRITE (BRown Into whiTE) acquiring BAT characteristics.14–17 Increased nonshivering heat production is a result of elevated uncoupling protein 1 (Ucp1) expression.14–17 Metabolic conversion of WAT into BRITE has been extensively investigated as a strategy for the treatment of morbid obesity, and different pharmacological targets have been proposed depending on the active substance tested.18–20
Previous work in our laboratory indicated that the lipid and lipoprotein transport system plays important role in the regulation of WAT and BAT mitochondrial metabolic activity and body weight gain.5,21–24 Of note, the effects of hypogonadism on WAT mitochondrial metabolic activity appear to be highly modulated by the low-density lipoprotein receptor (LDLR) since castrated LDLR-deficient (Ldlr−/−) mice were resistant to diet-induced obesity, in contrast to sham-operated animals, which showed significant weight gain in response to feeding western-type diet. When mitochondrial metabolic activity was assessed, mitochondrial cytochrome c (CytC) and Ucp1 expression were elevated in WAT of castrated Ldlr−/− mice, confirming increased mitochondrial metabolic activity due to oxidative phosphorylation and nonshivering thermogenesis. 5
More recently, in another study 21 we found that apolipoprotein E3 (APOE3) exerts a bimodal effect on visceral WAT mitochondrial metabolic activation that is highly dependent on its site of expression and independent of postprandial dietary lipid deposition. In brief, brain APOE3 expression inhibited visceral WAT mitochondrial oxidative phosphorylation, leading to increased body weight and obesity, when mice were fed western-type diet. In contrast, when APOE3 was expressed in the periphery, it triggered increased heat production in visceral WAT mitochondria, leading to resistance to obesity.
In another very elegant study, employing conditional Apoe deletion, animal models showed that deletion of WAT-expressed Apoe has no effect on morbid obesity, while deletion of hepatically expressed Apoe resulted in rather higher adiposity in mice, confirming the protective role of hepatically expressed APOE in obesity. 25 However, the precise mechanism by which hepatically or brain-expressed APOE impacts WAT metabolism remains elusive. These data supported a major “paradigm shift” from the existing view that APOE when expressed in periphery promotes obesity through receptor-mediated postprandial lipid delivery to WAT. 24
In light of these data, here we sought to investigate the potential functional crosstalk between TD and apolipoprotein E2 (APOE2) or apolipoprotein E4 (APOE4) expression in the regulation of plasma lipid and glucose homeostasis and WAT and BAT mitochondrial metabolic activity. Our studies were performed in APOE2 knock-in (Apoe2knock-in) and APOE4 knock-in (Apoe4knock-in) mice, which were subjected to surgical castration or sham operation. Our findings indicate that metabolic effects of TD are highly dependent on the isoform of APOE expressed.
Materials and Methods
Methods used in the present work are largely adopted from those used in Constantinou et al, 5 Zvintzou et al, 23 and Athanasopoulou et al. 4
Animal studies
The Apoe4knock-in and Apoe2knock-in mice used in our studies were purchased from Taconic Farms. Both strains were back crossed on the C57BL/6 genetic background for nine generations at least. Mice between 10 and 12 weeks of age were used in these studies, caged individually, and allowed unrestricted access to food and water under a 12 h light/dark cycle. At the beginning of the experiments, body weights, cholesterol and triglyceride levels of the individual mice were determined after 16 h of fasting. Mice were placed on standard western-type diet (Mucedola SRL, Milano, Italy), which contains 4.5 kcal/g (17.3% protein, 48.5% carbohydrate, 21.2% fat, 0.2% cholesterol [0.15% added, 0.05% from fat source]), while for the baseline experiments mice were fed the standard chow diet.
For the dietary studies, groups of mice (n = 5 per group) of the same genotype were selected to have similar body weights and biochemical characteristics such as cholesterol, triglycerides (TGs), and glucose levels. One group was surgically castrated and the other was sham operated to undergo similar stress levels. After a recovery period of 3 weeks on chow diet, all mice groups were switched into western-type diet (week 0) for an additional period of 12 weeks. At the end of our study, all mice were euthanatized to collect plasma and tissue samples.
All animal studies were regulated according to guidelines provided by the Protocol for the Protection and Welfare of Animals (EEC Directive of 1986, 86/609/EEC). Moreover, we took into consideration the 3Rs (reduce, refine, and replace). Optimal information size for each group was determined to be five mice (assuming that 95% of castrated mice will respond to lack of androgens at a desired power of estimate of 0.95), based on the online statistical tool https://www.stat.ubc.ca/~rollin/stats/ssize/b2.html. The work was authorized by the animal ethical committee of the Laboratory Animal Center of The University of Patras Medical School and the Veterinary Authority of the Prefecture of Western Greece (Approval No. 187563/629).
Surgical castration of mice
Surgical castration of mice was performed the way described previously. 26 Experimental mice were divided into two groups. One group (n = 5) was bilaterally castrated, and the other group (n = 5) was sham operated by executing only a skin incision. Both groups were under the influence of isoflurane (CP Pharma, Burgdorf, Germany) anesthesia. The animals left untreated to recover from the operation for a 3-week period. After this time, both groups switched to western-type diet for 12 weeks period as indicated previously. The same procedure was followed for both experimental groups Apoe4knock-in and Apoe2knock-in mice, and their control groups, respectively.
Determination of body weight and daily food consumption
Two weeks during the experiment, food intake was assessed during a 7-day period as described previously. 27 Specifically, all mice of each group (n = 5) for each mouse strain were tested. Every day, the daily average food consumption of each group was determined, along with the average daily food consumption per mouse, for each mouse strain. Results expressed as average food consumption per mouse per strain over 7-day period ± SEM.
Plasma lipid determination
After 16 h of fasting, plasma samples were isolated from the experimental mice. Plasma triglyceride and cholesterol levels were measured using the Triglyceride Determination Kit (DiaSys Diagnostic System GmbH, REF 1 5710 99 10 021) and the Cholesterol Determination Kit (DiaSys Diagnostic System GmbH, REF 1 1300 99 10 021), respectively, according to manufacturer's instructions and as described previously. 27
Measurement of hepatic triglyceride content
Hepatic triglyceride determination was performed as described previously. 27 Results are expressed as milligrams of triglycerides per gram of tissue ± SEM.
Fasting glucose determination
After 16 h of fasting, blood samples were drawn from tail vein of each mouse, and the glucose levels were determined using the Glucose Determination Kit (DiaSys Diagnostic System GmbH, REF 1 2500 99 10 021). For the glucose tolerance test (GTT) and insulin sensitivity test (IST), mice were fasted for 16 h. Dextrose (for GTT, 2 g/kg) or insulin (for IST, 0.5 U/kg Humulin; Eli-Lilly, Indianapolis, IN) was injected intraperitoneally; blood samples were collected at 0, 15, 30, 60, and 120 min postinjection through the tail vein, and glucose levels were measured as described previously. 5
Isolation of mitochondria
After 16 h of fasting, mice were euthanatized, and their BAT and WAT were collected to isolate mitochondria as described previously. 5 Both tissues were homogenized using a glass-Teflon homogenizer in ice-cold sucrose buffer (0.32 M Sucrose, 1 mM EDTA, 10 mM Tris-HCl, pH = 7.2 for BAT homogenization and 0.25 M Sucrose, 5 mM Tris-HCl, pH = 7.2 for WAT homogenization). After a centrifugation at 2700 rpm for 5 min at 4°C, the supernatant was transferred to another tube and centrifuged at 13,000 rpm for 10 min at 4°C; the pellet was resuspended with homogenization buffer for three consecutive rounds to purify. After the purification, the pellet containing crude mitochondria was resuspended in buffer containing 15 mM NaCl, 50 mM Tris-HCl (pH = 7.4), 0.5% sodium deoxycholate, 0.1% SDS, 1 mM EDTA, 1% Triton X-100. Determination of each sample protein concentration was performed by the Lowry assay using the DCTM Protein Assay Kit (cat no. 500-0016; Bio-Rad).
Fractionation of plasma lipoproteins by density gradient ultracentrifugation
After 16 h of fasting, plasma samples were isolated from the mice of each group. Ultracentrifugation was used to separate plasma lipoproteins. About 0.5 mL of pooled plasma from each mice group, separately, was fractionated by KBr-density gradient in a Beckman-Coulter ultracentrifuge using a SW41 rotor, as described previously, 28 over a 4 mL KBr (Sigma-Aldrich, St. Louis, MO) gradient (1.23 g/mL over 1.21 g/mL over 1.063 g/mL over 1.019 g/mL over saline).
Western blot analysis
Western blotting of plasma APOE2, APOE4, murine apolipoprotein A1 (ApoA1), murine apolipoprotein C1 (ApoC1), murine apolipoprotein C2 (ApoC2), and murine apolipoprotein C3 (ApoC3) was performed as described previously.29–31 For the detection of APOE2 and APOE4, Rabbit anti-APOE (cat no. 13366) antibody from Cell Signaling (Danvers, MA) was used as primary and a goat antirabbit antibody (cat no. 7074; Cell Signaling) as secondary. For the detection of ApoaI, ApocI, ApocII, and ApocIII, a goat anti-Apoa-Ι (cat no. K45252G; Meridian Life Science), a goat anti-Apoc-I (cat no. 31H-G1b; Academy Biomedical Company, TX), a goat anti-Apoc-II (cat no. K59600R; Biodesign International), a goat anti-Apoc-III antibody (cat no. K74140G; Biodesign International), respectively, were used.
To investigate the role of APOE2 and APOE4 in the metabolic activation of BAT and WAT after surgical castration, pure visceral WAT and BAT mitochondria proteins were isolated from Apoe2knock-in and Apoe4knock-in mice on week 12 of the experiment. Then, mitochondrial Cytc and Ucp1 were measured as biochemical markers of metabolic activity and expressed in relation to Cox4 protein, a constitutively expressed structural protein used as an internal control. In addition, Ucp1/Cytc ratio as a measure of the extent of uncoupling of oxidative phosphorylation from ATP production was determined.
Cytochrome c, Ucp1, and cytochrome c oxidase subunit 4 (Cox4) detection was performed using IgG primary rabbit antirabbit antibodies (cat no. 4272, Cell Signaling; cat no. 14670, Cell Signaling; cat no. 4844, Cell Signaling, respectively). Concisely, mitochondrial samples of BAT (8 μg/lane) and WAT (15 μg/lane) were resolved by SDS-PAGE (12.5% acrylamide and 0.51% N,N′-diallyltartardiamide). Semiquantitative determination of the relative protein amounts was performed by ImageJ (Fiji) free software and Cytc, and Ucp1 levels were normalized to Cox4 levels, a constitutively expressed mitochondrial protein whose expression was unaffected by the conditions.
Statistical analysis
All data sets were tested using the Kolmogorov–Smirnov and Shapiro–Wilk tests, and were treated with parametric (p > 0.1) or nonparametric tests (p < 0.1) according to their deviation from normality. Comparison of data from two groups of mice was performed using the t-test. All data were found to follow normal distribution. Statistical tests were performed using the Prism software (GraphPad Prism Software, Version 8). Data are reported as mean ± S.E.M. *Indicates p < 0.05, **indicates p < 0.01, ***p < 0.001, ****indicates p < 0.0001.
Results
Effects of hypogonadism on body weight gain, plasma cholesterol and glucose levels, and hepatic lipid deposition of Apoe2knock-in and Apoe4knock-inmice
After a period of 3 weeks on chow diet to allow for elimination of residual testosterone from circulation of castrated mice, animals were placed for an additional 12 weeks on western-type diet, and body weight (Fig. 1A, B) was determined at the indicated time points (Fig. 1A, B). Mean food consumption during the course of the experiment was also measured (Fig. 1C). All groups showed an elevation in body weight in response to feeding western-type diet. No statistically significant difference could be found in the body weights of castrated and sham-operated Apoe2knock-in mice.

Body weight gain of sham-operated and castrated Apoe2knock-in mice
Specifically, both Apoe2knock-in mouse groups had similar starting body weights (27.1 ± 0.3 g for sham-operated and 23.1 ± 0.3 g for castrated, p > 0.05) at week 3. Both groups gained weight progressively and at week 12, average body weight reached 32 ± 0.6 g for sham-operated mice and 30.5 ± 0.99 g for castrated animals (p > 0.05). However, in mice expressing APOE4, castrated animals gained much lower body weight compared with their sham-operated counterparts. Indeed, castrated Apoe4knock-in mice changed from 25.7 ± 0.9 g at week 3 to 26.6 ± 0.7 g at week 12, while sham-operated Apoe4knock-in mice showed a much higher increase from 27.4 ± 0.4 g at week 3 to 32.6 ± 0.8 g at week 12 (p < 0.001). Interestingly, sham-operated APOE4-expressing mice gained similar weight to sham-operated or castrated APOE2-expressing mice. No difference in food consumption was noted between castrated and sham-operated mice of both genotypes (Fig. 1C).
Plasma cholesterol levels in both Apoe2knock-in mouse groups increased as a function of time although castrated animals had significantly higher levels than sham-operated mice (from 160 ± 16.7 mg/dL for both groups on week 3, to 306 ± 19.9 mg/dL for sham-operated and 545 ± 15.2 mg/dL [p < 0.0001] for castrated mice on week 12) (Fig. 2A). However, plasma cholesterol levels of Apoe4knock-in mice (Fig. 2D) remained physiological (<200 mg/dL), although castrated animals had slightly lower levels than their sham-operated counterparts (from 70.7 ± 8.5 mg/dL for both groups at week 3, to 128.2 ± 9.5 mg/dL for the sham-operated and 104.1 ± 20.6 mg/dL for the castrated group, p > 0.05).

Total cholesterol
Notably, analysis of hepatic TG content revealed that expression of APOE4 resulted in a much more profound TG accumulation in the liver, compared with APOE2 (Fig. 2B, E). Specifically, sham-operated Apoe4knock-in mice and Apoe2knock-in mice had 107.8 ± 21 mg/g of tissue and 8.3 ± 1.5 mg/g of tissue, respectively (p < 0.001) (Fig. 2B, E). Similarly, castrated Apoe4knock-in mice and Apoe2knock-in mice had 96.4 ± 17.2 mg/g of tissue and 21.3 ± 2 mg/g of tissue, respectively (p < 0.001) (Fig. 2B, E).
Interestingly, TD promoted a higher hepatic lipid accumulation only in Apoe2knock-in mice (8.3 ± 1.5 mg/g of tissue for sham-operated and 21.3 ± 2 mg/g of tissue for castrated, p < 0.001) (Fig. 2B). TD in Apoe4knock-in mice did not affect significantly hepatic lipid levels (107.8 ± 21 mg/g of tissue for sham-operated and 96.4 ± 17.2 mg/g of tissue for castrated mice, p > 0.05) (Fig. 2E).
Plasma glucose levels were deteriorated as a function of time only in Apoe4knock-in mice reaching 161 ± 4.7 mg/dL in sham-operated and 167.3 ± 8.5 mg/dL (p > 0.05) in castrated animals (Fig. 2F) (p > 0.05). To the contrary, sham-operated and castrated Apoe2knock-in mice maintained physiological plasma glucose levels (<100 mg/dL) throughout the study, and no statistical differences were found between the two Apoe2knock-in groups (all p > 0.05) (Fig. 2C).
Effects of hypogonadism on lipoprotein and apolipoprotein profiles of Apoe2knock-in and Apoe4knock-in mice
Fractionation of plasma samples by KBr-density gradient ultracentrifugation showed that TGs were found increased in chylomicron/very low-density lipoprotein (CM/VLDL), intermediate density lipoprotein (IDL), and low-density lipoprotein (LDL) fractions in both Apoe2knock-in groups (TG levels in CM/VLDL fraction were 83.2 mg/dL in sham-operated vs. 115.8 mg/dL in castrated mice) (Fig. 3A). Accordingly, in Apoe2knock-in mice there were elevated cholesterol levels present in CM/VLDL and LDL fractions (Fig. 3B), and castration further exacerbated the hypercholesterolemia of these mice (cholesterol levels in the CM/VLDL fraction were 44.9 mg/dL in sham-operated vs. 55.1 mg/dL in castrated mice) (Fig. 3A, B).

Triglyceride
In Apoe4knock-in groups, lipoprotein triglycerides were much lower than Apoe2knock-in groups (TG levels in the CM/VLDL fraction of sham-operated Apoe4knock-in mice were 17.7 mg/dL vs. 83.2 mg/dL for sham-operated Apoe2knock-in mice), while in Apoe4knock-in mice cholesterol was primarily present in high-density lipoprotein (HDL) fractions and to a lesser extent in CM/VLDL and LDL fractions (Fig. 3D). Castration of Apoe4knock-in mice did not affect triglyceride distribution among different lipoprotein fractions although it resulted in a significant reduction in HDL-C levels (46.4 ± 0.3 mg/dL for sham-operated vs. 31.3 ± 0.1 mg/dL for castrated mice, p < 0.05) (Fig. 3C, D).
Western blot analysis for APOE2, APOE4, Apoa1, Apoa2, Apoc1, Apoc2, and Apoc3 on isolated lipoprotein fractions revealed significant levels of APOE2 in VLDL, LDL, and HDL particles in sham-operated Apoe2knock-in mice (Fig. 3E). In their castrated counterparts, trace amounts of APOE2 were found only in HDL fractions (Fig. 3F). Castration resulted in a massive increase in Apoa1 levels of these mice in all lipoprotein fractions (CM/VLDL, IDL/LDL, and HDL) (Fig. 3F).
This is in sharp contrast with sham-operated Apoe2knock-in mice where Apoa1 was detectable in trace amounts only in CM/VLDL fraction (Fig. 3E). No Apoc2 and Apoc3 could be detected in either mouse group, although significant levels of Apoc1 could be found in the CM/VLDL/IDL/LDL fractions of both Apoe2knock-in groups, with higher amounts present in castrated animals (Fig. 3E, F).
Similar analysis in Apoe4knock-in mice showed that APOE4 was present mainly in CM/VLDL, IDL/LDL and the lighter fractions of HDL in the sham-operated mice (Fig. 3G), while castrated animals had their APOE4 distributed in LDL and HDL fractions. In both sham-operated and castrated Apoe4knock-in mice, Apoa1 was detected in all lipoprotein fractions (Fig. 3G, H). Apoc1, Apoc2, and Apoc3 were present primarily in HDL fractions of sham-operated Apoe4knock-in mice (Fig. 3G). Apoc1, Apoc2, and Apoc3 content of castrated mice was much lower with trace amounts present in CM/VLDL particles (Fig. 3H).
Effects of hypogonadism on glucose metabolism of Apoe2knock-in and Apoe4knock-in mice
An intraperitoneal GTT performed in sham-operated and castrated APOE2- and APOE4-expressing mice showed that both sham-operated and castrated APOE2-expressing mice maintained intact their ability to handle glucose since they showed similar AUC values at weeks 3 and 12, with no statistically significant differences between the two groups (30.678 ± 4234 vs. 33.071 ± 1303; p > 0.05 for castrated and sham-operated APOE2-expressing mice, respectively) (Fig. 4A–C).
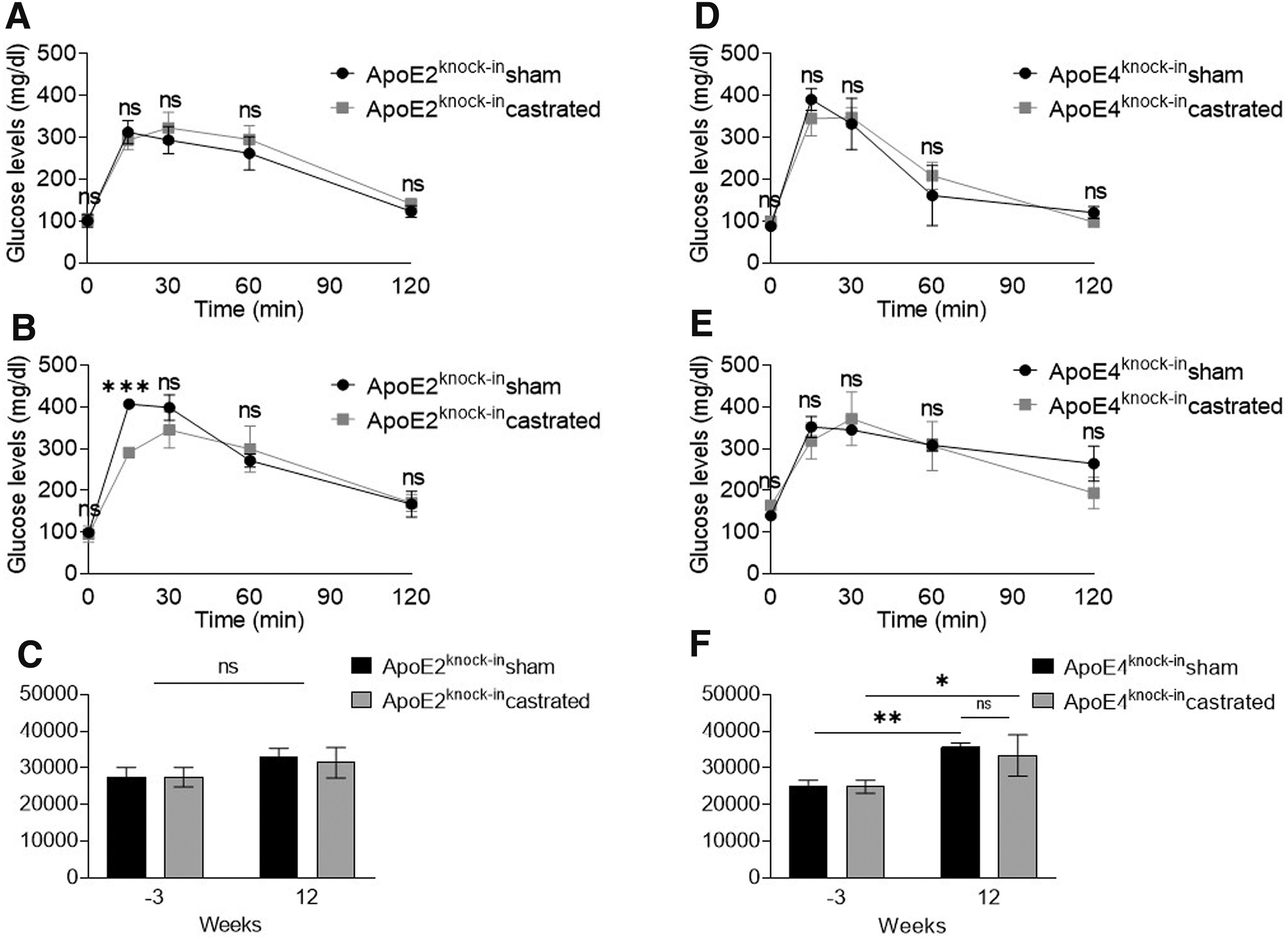
GTT of sham-operated and castrated Apoe2knock-in
To the contrary, both sham-operated and castrated Apoe4knock-in mice showed a deterioration of their ability to handle glucose since their AUC values increased significantly between 3 and 12 weeks although no statistically significant difference was again noted between the two groups (Fig. 4D–F) (35.593 ± 1235 for sham-operated and 33.394 ± 5641 for castrated APOE4-expressing mice at week 12, p > 0.05). Our findings indicate that APOE4 expression predisposes to glucose intolerance although TD does not contribute to further deterioration of this condition.
Effects of hypogonadism on brown and WAT mitochondrial metabolic activity of Apoe2knock-in and Apoe4knock-in mice
In Apoe2knock-in mice, castration resulted in lower levels of BAT mitochondrial Ucp1 (Fig. 5C) and CytC (Fig. 5D) (p < 0.001 and p < 0.01, respectively), suggesting reduced nonshivering thermogenesis and oxidative phosphorylation in BAT mitochondria of this mouse strain (Fig. 5C, D). Castration had no statistically significant effect on the extent of uncoupling of oxidative phosphorylation from ATP production (Fig. 5E). In contrast, when WAT mitochondria were analyzed, castration resulted in significantly elevated Ucp1 (Fig. 5F) levels although no effect of castration on CytC expression was evident (Fig. 5G). A notable uncoupling of oxidative phosphorylation from ATP production was evident (Fig. 5H).
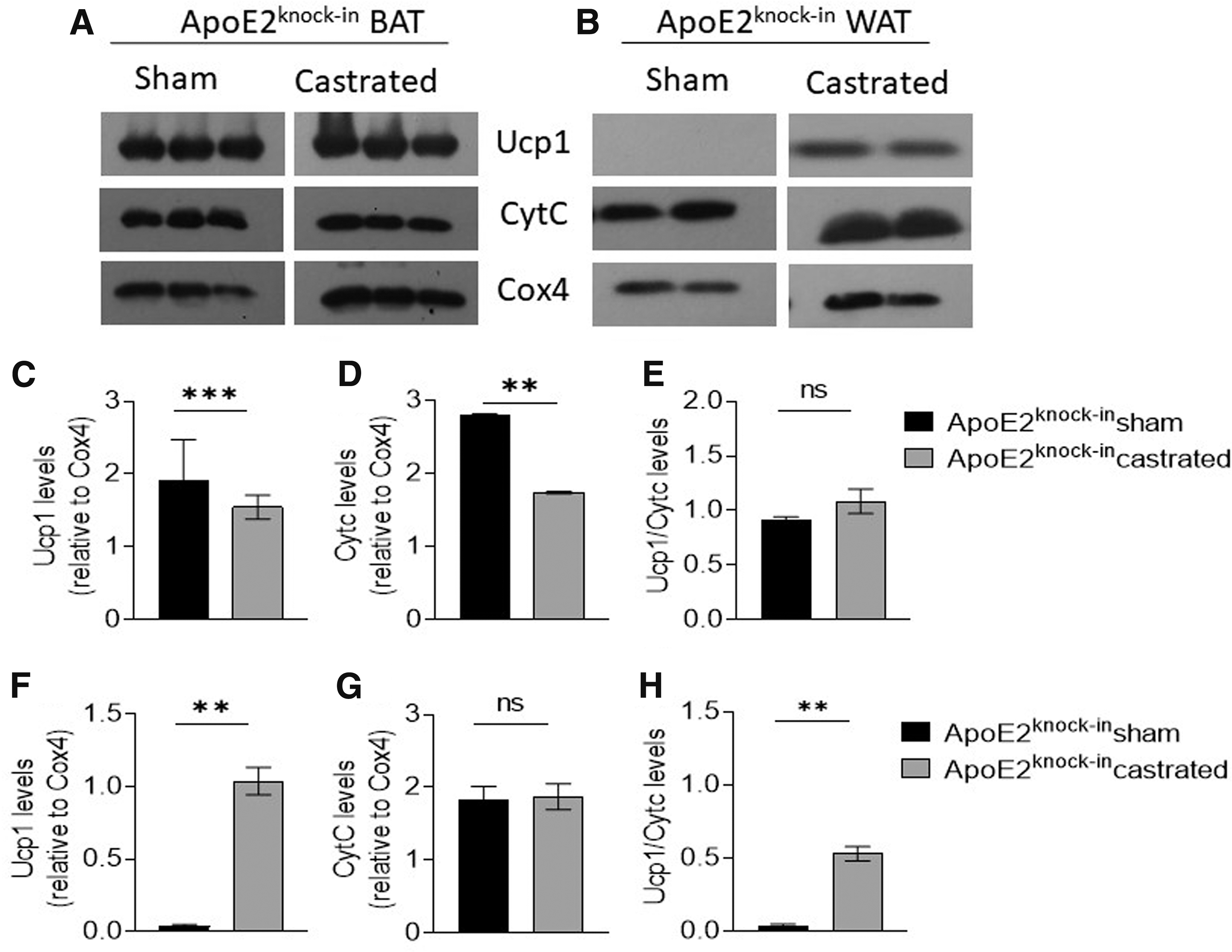
Representative Western blot analysis for Ucp1, CytC, and Cox4 of mitochondrial extracts from BAT
In BAT of Apoe4knock-in mice, castration did not have any measurable effect on Ucp1 (Fig. 6C) and CytC expression levels (Fig. 6D) or the extent of uncoupling of oxidative phosphorylation from ATP production (Fig. 6E) (all p > 0.05). However, when WAT mitochondrial protein extracts were analyzed, it was found that castration triggered a quite substantial increase in Ucp1 expression (p < 0.05) (Fig. 6F). No effect of castration on WAT CytC expression was found (Fig. 6G) although it resulted in a significant uncoupling of oxidative phosphorylation from ATP production (Fig. 6H).

Representative Western blot analysis for Ucp1, CytC, and Cox4 of mitochondrial extracts from BAT
Discussion
The data presented here indicate that the natural human APOE isoforms APOE2 and APOE4 modulate distinctly the effects that hypogonadism may exert on plasma lipoprotein and glucose metabolism, hepatic lipid deposition as well as WAT and BAT mitochondrial metabolic activation.
APOE2-expressing mice had profoundly higher plasma cholesterol levels compared with APOE4-expressing mice whose cholesterol levels were maintained <200 mg/dL for the duration of the experiment. This was expected given that lipoprotein-associated APOE2 is a poor substrate for the LDLR, 32 leading to reduced hepatic clearance and excess accumulation of CM/VLDL in the circulation of these mice. On the contrary, APOE4 binds LDLR with a much higher affinity, and as such, it promotes the efficient clearance of these lipoproteins from circulation.
Interestingly, TD resulted in a modest decrease in plasma cholesterol levels in mice expressing APOE4 compared with their sham-operated counterparts (Fig. 2D). Notably however, this was not the case in mice expressing APOE2, where hypogonadism resulted in a quite significant exacerbation of the hypercholesterolemia (Fig. 2A).
Analysis of cholesterol and triglyceride lipoprotein profiles after purification of lipoproteins by KBr-density gradient ultracentrifugation indicated that in both mouse strains hypogonadism exerts proatherogenic effects on plasma lipoprotein profile, which are highly dependent on the APOE isoform expressed. Specifically, in APOE2-expressing mice hypogonadism was associated with elevated CM/VLDL and IDL/LDL (Fig. 3A, B), while in Apoe4knock-in mice it resulted in a substantial reduction in HDL-C levels (Fig. 3C, D).
It is important to remember that total plasma cholesterol levels are the sum of cholesterol present in proatherogenic lipoproteins such as VLDL, IDL, and LDL and the cholesterol present in the antiatherogenic HDL lipoproteins. Therefore, the different effects of hypogonadism on total plasma cholesterol levels of Apoe2knock-in and Apoe4knock-in mice could be the result of distinct changes in the levels of any of these lipoproteins. Indeed, in a recent study, TD resulted in increased PCSK9 expression and reduced LDLR expression in castrated male pigs. 33 If a similar mechanism exists in our animal models, one hypothesis could be that this mechanism drives the sharp increase in plasma cholesterol levels observed in castrated Apoe2knock-in mice.
These mice have an already reduced capacity of LDLR-mediated hepatic clearance of VLDL/IDL/LDL, which could be further impaired even by a subtle reduction in LDLR expression levels. On the contrary, in Apoe4knock-in mice where lipoprotein-bound APOE4 has a high affinity for the LDLR, small changes in LDLR expression may leave the levels of atherogenic lipoproteins unaffected. In that case, VLDL/IDL/LDL levels will be unchanged (as they appear in our study), and the reduction in plasma cholesterol levels of these mice can be explained by the observed reduction in HDL-C levels, as documented by the UCF analysis.
It is remarkable that in Apoe2knock-in mice castration resulted in a sharp elevation of ApoA1 levels in all lipoproteins and APOC1 levels in CM/VLDL/IDL/LDL, with a concomitant large reduction in plasma APOE levels. Given that APOC1 is a potent inhibitor of Lpl, 34 this increase in APOC1 levels may explain the further elevation in the levels of triglyceride-rich lipoproteins in response to castration. In the case of sham-operated Apoe4knock-in mice, APOE is distributed among all lipoprotein fractions, while in castrated Apoe4knock-in mice APOE is no longer present in CM/VLDL fractions. Although APOA1 distribution was largely unaffected, a significant reduction in the levels of APOC1, APOC2, and APOC3 was found in the HDL fractions of castrated APOE4-expressing mice, explaining the observed reduction in HDL-C levels in these animals.
Analysis of hepatic triglyceride content indicated that Apoe2knock-in mice had much lower levels compared with Apoe4knock-in mice. Since our study involved dietary intervention of long-term feeding western-type diet, this could be explained again by the reduced affinity of APOE2 for LDLR, which could result in much reduced uptake of dietary lipid-laden CM by the liver. However, other mechanisms, such as reduced de novo triglyceride biosynthesis, in the liver of Apoe2knock-in mice cannot be excluded. TD had no effect on hepatic triglyceride deposition in Apoe4knock-in mice, while it resulted in a modest increase in Apoe2knock-in mice although hepatic TG content of these mice was still much lower than that observed in sham-operated and castrated APOE4-expressing animals.
Even though TD has no apparent effect on body weight gain in Apoe2knock-in mice, it results in significantly reduced weight gain in Apoe4knock-in mice. Analysis of WAT and BAT mitochondrial Ucp1 and CytC levels indicated that in Apoe2knock-in mice castration resulted in lower nonshivering thermogenesis (indicated by the reduced Ucp1 levels) and oxidative phosphorylation (indicated by the reduced CytC levels). Under these conditions, one would expect that TD would result in reduced fat consumption toward energy production and increased body weight gain. However, castration also resulted in significantly elevated energy consumption for nonshivering thermogenesis in WAT mitochondria of these mice, which apparently suffices to counterbalance the reduced energy consumption in BAT. As a result, TD does not impact weight gain in Apoe2knock-in mice.
In contrast, in Apoe4knock-in mice, castration had no impact in mitochondrial energy metabolism in BAT, although a quite substantial increase in nonshivering thermogenesis was noted in WAT. This increased energy consumption toward heat production in WAT of castrated Apoe4knock-in mice explains the observed reduction in body weight gain of these mice.
Sham-operated APOE4-expressing animals showed increased body weight gain compared with sham-operated APOE2-expressing mice in response to feeding western-type diet. This increase in body weight was accompanied with a marked deterioration in glucose tolerance of Apoe4knock-in mice, which was not apparent in Apoe2knock-in mice. Castration did not exert any further effect on glucose tolerance of these mice.
To this date, it is unclear how APOE, a protein expressed at ∼75% by the liver, influences events in adipose tissue.21,25 The current data further add to this complexity, indicating that such APOE-mediated effects on mitochondrial metabolic activity of BAT and WAT are highly dependent on the APOE isoform expressed. One possibility that needs further exploration is that APOE and obviously its different natural isoforms impact the delivery and uptake of lipids to and from plasma membranes.
In turn, this lipid exchange affects lipid composition of cellular membranes and to a great extent their fluidity, which is a key factor for the responsiveness of cell-surface receptors, such as adrenergic β3-receptors, to their stimuli. Recently, we demonstrated that in the absence of APOA1, pancreatic β-islets acquire more rigid membranes through a significant shift of cell membrane fluidity toward a more gel-like structure, a physicochemical property that is associated with reduced stimulation of ion channels. 22 Therefore, it is possible that in the mouse model, APOE2 and/or APOE4 expression has different effects on WAT and BAT membrane fluidity, which in turn affect receptor-mediated cellular pathways responsible for the regulation of WAT and BAT mitochondrial metabolic activity. This hypothesis is currently under investigation in our laboratory.
Our findings provide further support for the action of testosterone on carbohydrate and lipid metabolism. However, some areas that could explain mechanistically the differential role of APOE2 and APOE4 isoforms in TD remain unexplored. For example, the role of Liver X Receptor α (LXRα) should be further investigated. Recent data indicate that T acts through androgen receptor-dependent pathways to stimulate LXRα and downstream targets to induce cholesterol clearance in human macrophages. 35
These molecular targets include APOE and the lipid transporter ABCA1. Similarly, in the testicular feminized mouse, TD was associated with suppressed LXRα levels and reduced hepatic APOE and ABCA1 expression. 36 In addition, it resulted in reduced GLUT4 and glycolytic enzymes in skeletal muscle. 36 Importantly, the relation between TD, APOE isoforms, and dysfunctional mitochondria warrants further investigation, as in men it is tightly associated with insulin resistance. 37
Conclusions
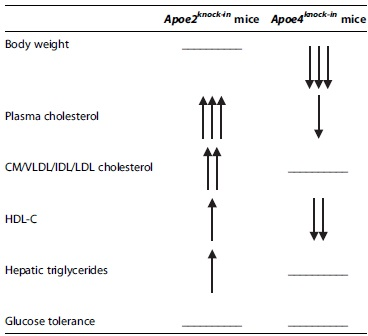
In this study, we identified a functional crosstalk between TD and APOE, which is highly modulated by the isoform expressed. Table 1 provides a qualitative summary of the distinct effects of castration on APOE2- and APOE4-expressing mice compared with their sham-operated counterparts. It is quite striking that in both mouse strains, hypogonadism brings about negative effects on lipoprotein metabolism, which are manifested by different disturbances. Moreover, the reduced body weight gain in castrated APOE4-expressing mice did not alleviate the already much impaired glucose tolerance of these mice or their hepatic triglyceride levels.
Qualitative Depiction of the Effects of Castration on APOE2- and APOE4-Expressing Mice Compared with Their Sham-Operated Counterparts
Flat line indicates no change, one arrow indicates minor change, two arrows indicate medium change, and three arrows indicate major change.
CM/VLDL/IDL/LDL, chylomicron/very low-density lipoprotein/intermediate density lipoprotein/low-density lipoprotein; HDL-C, high-density lipoprotein-cholesterol.
Epidemiological data suggest that the ApoE2 isoform is most favorable for protection against cardiovascular disease, whereas the ApoE4 is much less favorable. 38 Our data show that under the stress conditions generated by hypogonadism, APOE2 expression results in serious adverse effects associated with elevated VLDL/IDL/LDL cholesterol and triglyceride levels and increased body weight gain. APOE4 expression on the contrary results in reduced HDL-C levels and impaired glucose tolerance, despite a very modest reduction in body weight. Therefore, with respect to lipoprotein profile and body weight disturbances, it appears that the effects of TD may be more severe in the presence of APOE2 than APOE4.
Extrapolating our findings, it is expected that correcting hypogonadism by TRT may benefit more the APOE2 phenotype. Nevertheless, the benefit that TRT may have on HDL-C levels and glucose homeostasis of the APOE4 phenotype should not be discounted as these conditions are also important risk factors for cardiovascular disease.
By translating our findings in the clinical setup, it is important to note that changes in hypogonadal individuals, which could be perceived as beneficial (e.g., reduced weight gain), may not necessarily reflect a better health state. Rather, pathological conditions such as dyslipidemia, nonalcoholic fatty liver disease, and type II diabetes mellitus should not be ruled out, and they should be carefully evaluated in relation to the APOE isoform(s) expressed.
Footnotes
Authors' Contributions
D.-S.K., P.C.G., A.N., N.M., and E.Z. participated in the execution of experiments, evaluation of data, drafting of the article, and approving its final submitted version. K.E.K. participated in the design of the studies, execution of experiments, evaluation of data, drafting of the article, and approving its final submitted version.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
We acknowledge partial support of this work by the project “INSPIRED” (MIS 5002550), under the Action “Reinforcement of the Research and Innovation Infrastructure,” funded by the Operational Program “Competitiveness, Entrepreneurship and Innovation” (NSRF 2014-2020). P.C.G. is funded by a research fellowship through the operational Programme “Human Resources Development Education and Lifelong Learning” in the context of the project “Strengthening Human Resources Research Potential through Doctorate Research” (MIS 5000432), implemented by the State Scholarship Foundation (IKY).