Abstract
In 2005, we reported that, in the animal model, androgen deprivation resulted in significant changes in the structural elements of the penile corpus cavernosum (CC), including: (1) atrophy of the trabecular smooth muscle, (2) increased deposition of connective tissue, and (3) accumulation of adipocytes in the sub-tunical region. These structural alterations, together with reported decreases in the expression and activity of endothelial nitric oxide synthase (eNOS) and neural nitric oxide synthase (nNOS), are believed to contribute to reduced blood inflow and increased blood outflow, contributing to veno-occlusive dysfunction and erectile dysfunction (ED). More recently, several clinical studies have documented accumulation of adipocytes in the sub-tunical region of the corpora cavernosa from patients with ED, supporting findings reported in the animal models. Here, we summarize the observations from basic and clinical research and advance the hypothesis that testosterone deficiency contributes to significant changes in the structural integrity of the CC, resulting in atrophy of the trabecular smooth muscle and contributing to reduced vascular smooth muscle relaxation and diminished blood inflow into the penis, resulting in a failure to fill. In addition, atrophy of the trabecular smooth muscle, accumulation of adipocytes in the sub-tunical region, and increased deposition of connective tissue contribute to veno-occlusive dysfunction and a failure to maintain erection. It is postulated that testosterone treatment in men with testosterone deficiency may reverse the structural alterations in the CC and improve erectile function.
Introduction
Erectile function is a complex hemodynamic process and is largely dependent on vasodilation of the cavernosal artery and the helicine arterioles and the relaxation of the trabecular smooth muscle of the corpus cavernosum (CC). 1 Increased blood inflow into the CC concomitant with relaxation of the trabeculae results in trapping of blood in the lacunar spaces, under pressure, compressing the sub-tunical venules and impeding blood outflow, resulting in erection.
This hemodynamic process is termed “veno-occlusive mechanism.” 1 Veno-occlusive function is dependent on the integrity of various components of erectile tissue, including the vascular, neural, trabecular, endocrinological, and connective tissue fibroelastic components.2,3 Alterations in any of such components due to disease states or hormonal imbalances result in poor tissue compliance and veno-occlusive dysfunction due to reduced blood inflow (failure to fill) or increased blood outflow (failure to maintain), resulting in erectile dysfunction (ED).4,5
Considerable evidence exists, indicating that androgens play an important role in maintaining erectile physiology and androgen deficiency contributes to ED.6–23 Androgens regulate the expression of endothelial nitric oxide synthase (eNOS) and neural nitric oxide synthase (nNOS)24–28 and maintain penile corpora structural integrity by promoting trabecular smooth muscle growth and function, inhibiting adipogenesis, maintaining neural fiber network, and restoring the balance between trabecular smooth muscle and extracellular matrix, thus improving tissue compliance and erectile function.29–32 Androgen deficiency contributes to reduced eNOS and nNOS in the CC24–28 and reduced smooth muscle content, 18 concomitant with increased deposition of connective tissue,29–34 resulting in ED.
Animal models29–33 as well as clinical studies35–40 have demonstrated that testosterone (T) deficiency (TD) resulted in accumulation of adipocytes in the sub-tunica albuginea region of the corpora cavernosa. The accumulation of adipocytes in this region may contribute, in part, to venous leakage and veno-occlusive dysfunction, resulting in ED. Since compression of the sub-tunical venules is critical for impeding blood outflow and maintaining penile rigidity, it is possible that accumulation of adipocytes in the sub-tunical region of the corpora cavernosa compromises the veno-occlusive mechanism, leading to ED.
In this review, we summarize findings from the contemporary literature on the regulation of adipogenesis by androgens in the CC and advance a hypothesis that the accumulation of adipocytes in the sub-tunical region of the penile CC may, in part, be responsible for venous leakage and veno-occlusive dysfunction, resulting in ED.
Androgens Inhibit Differentiation of Preadipocytes to Adipocytes, and Androgen Deficiency Promotes Accumulation of Adipocytes
It is well established that androgens inhibit the differentiation of preadipocytes into mature adipocytes. 41 Using 3T3-L1 cells, Singh et al.42,43 demonstrated that the treatment of 3T3-L1 with T or 5α-dihydrotestosterone (5α-DHT) inhibited the conversion of these preadipocytes into adipocytes, in a dose-dependent manner. The addition of flutamide or bicalutamide, which inhibits the androgen receptor, promoted the differentiation of 3T3-L1 into adipocytes, suggesting an integral role of the androgen receptor in this mechanism.
Chazenbalk et al. 44 described several androgen-dependent molecular targets that facilitated the mechanism underlying differentiation from preadipocytes to mature adipocytes in the absence of androgens. More recently, Sebo and Rodeheffer 40 reported that androgen deprivation stimulates adipogenesis in vivo and administration of T or 5α-DHT to orchiectomized animals attenuated the hyperproliferative response of preadipocytes to a high-fat diet in orchiectomized animals, indicating a role for androgens in the suppression of adipogenesis in vivo.
Accumulation of Adipocytes in the Sub-Tunical Region of the Corpora Cavernosa in Castrated Animals
Traish et al. 19 and Rogers et al. 20 introduced the hypothesis that T deficiency produces significant alterations in the corpora cavenosa structural integrity, including reduced trabecular smooth muscle cell content, reduced myelination of the nerve fiber network, and increased deposition of extracellular connective tissue components, resulting in venous leakage and veno-occlusive dysfunction.19,20 The treatment of castrated animals with T restored smooth muscle content and erectile function in the animal model,19,20 suggesting the reversibility of this physiological mechanism.
More recent studies in the animal model demonstrated that T deficiency also contributes to the accumulation of adipocytes in the sub-tunical region of the corpora cavernosa (Figs. 1 and 2). 29 Okumu et al. 45 also reported that the treatment of immature animals with gonadotropin releasing hormone-A resulted in loss of CC smooth muscle cells and loss of lacunar spaces, concomitant with accumulation of adipocytes. Interestingly, no significant alterations were noted in the corpus spongiosum.

Hematoxylin and eosin staining of penile tissue. Male rabbits (4.5–5 kg) were subjected to sham surgery (left panel) or bilateral orchiectomy (right panel). After 2 weeks, rabbits were euthanized; penile cross-sections were removed, fixed in buffered 10% formalin, embedded in paraffin, sectioned (6 mm), and stained with hematoxylin and eosin. Note the numerous empty cellular structures adipocytes from which fat is washed out under the tunica albuginea in the orchiectomized rabbit (arrows).
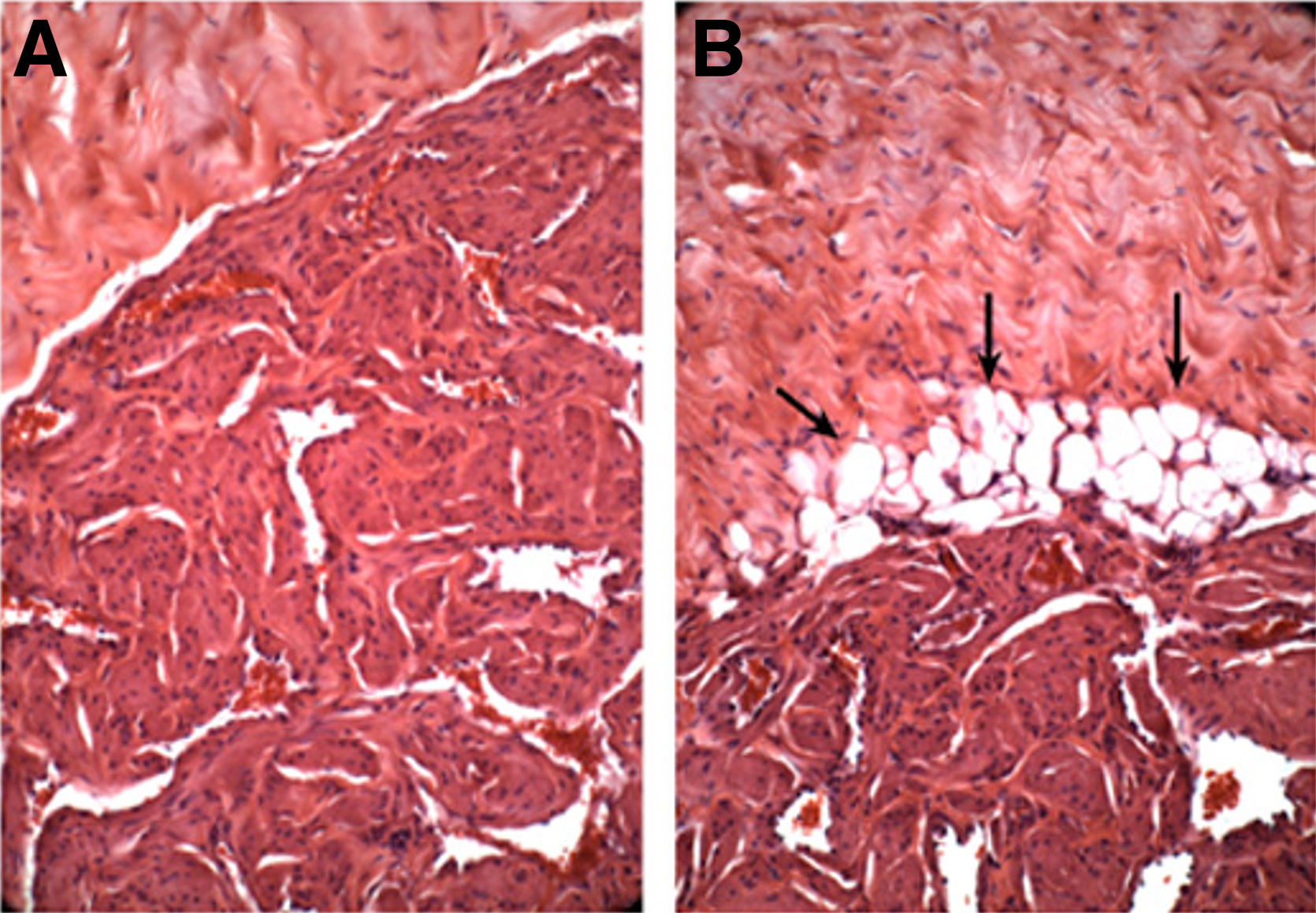
Hematoxylin and eosin staining of penile tissue. Male rabbits (4.5–5 kg) were subjected to sham surgery
The authors postulated that androgens are necessary for the differentiation of stem cells that reside in the CC into trabecular smooth muscle and androgen deficiency results in reprogramming stem cells to differentiate into adipocytes instead of differentiation into CC smooth muscle cells. Since androgen deprivation not only reduces penile trabecular smooth muscle content but also introduces fat cells in the sub-tunical region, it is possible that such alterations in the structural integrity of the CC may result not only in diminished blood inflow and failure to fill but also in increased blood outflow and failure to maintain erection with the outcome of ED.
It was postulated that such accumulation of adipocytes may interfere with tissue compliance and impeding erectile physiology, contributing to a failure to maintain erections and veno-occlusive dysfunction.
Accumulation of Adipocytes in the Corpora Cavernosa of Patients with Erectile Dysfunction
Palma et al. 35 investigated the accumulation of adipocytes in penile corpora cavernosa of men with severe ED who were deemed refractory to phosphodiesterase type 5 (PDE5) inhibitors treatment or intracavernous or intraurethral prostaglandin E1 (PGE1) or vacuum-assisted therapy and were considered appropriate candidates for penile implant surgery. As a control group, men with stable Peyronie's disease who were potent but were refractory to medical therapy underwent penile curvature correction surgery.
Penile corpora cavernosa tissue specimens from all patients were obtained; sections of the corpora cavernosa were fixed and embedded in paraffin for staining with hematoxylin and eosin and examined histologically for the presence of adipocytes in the sub-tunical region analogous to those histological data reported in the animal model of androgen deprivation. 29 Out of the nine patients, seven exhibited accumulation of cavernous fat cells (78%).
Only a single patient in the potent group demonstrated adipocytes accumulation (9%). A follow-up study by Vinay et al. 36 compared the histology of surgical specimens derived from patients who underwent surgical procedures for penile prosthesis implantation to treat drug-refractory erectile dysfunction with surgical specimens from potent patients, who were undergoing surgical procedures for Peyroni's disease to correct penile curvature. Interestingly, 11 of the 17 patients with drug-refractory ED demonstrated adipocytes accumulation in the penile sub-albuginea region. However, only 1 patient out of 14 surgically treated for Peyronie's disease showed accumulation of adipocytes in the sub-albuginea region.
Rodriguez et al. 37 reported that 63 consecutive patients who underwent surgery for squamous cell carcinoma tissue sections from partial penectomy specimens were evaluated histologically. The authors noted that adipose tissue was present in 33 cases (52%). The fat cells were peripherally located at the junction of the tunica albuginea with the corpora (Fig. 3).

Alwaal et al. 38 have examined lipid accumulation in the CC of a case series comprising nine patients with erectile dysfunction who underwent penile prosthesis implantation using Oil Red O staining. The authors demonstrated lipid accumulation in all nine CC tissue samples. The authors further postulated that the lipid accumulation may be attributed, in part, to androgen deficiency or dedifferentiation of corpus smooth muscle cells into other phenotypes.
Discussion
Venous leakage is a common condition among erectile dysfunction patients who are nonresponders to medical management. Venous leakages may be the result of vascular disease or increased penile fibrosis due to reduced smooth muscle content and increased connective tissue deposition, due to hormonal imbalance or other disease conditions that promote trabecular smooth muscle atrophy and increase extracellular matrix deposition and fibrosis. It is well established that androgens maintain the trabecular smooth muscle content and function via facilitating the differentiation of progenitor cells into smooth muscle cells and inhibit preadipocyte differentiation into mature adipocytes.
We have previously postulated that T deficiency may contribute to venous leakage and veno-occlusive dysfunction due to a host of metabolic, neural, and structural alternations in the cavernosum body.18,19,23,29–32 T therapy in men with ED and venous leakage may restore erectile function. 46
Erectile physiology is dependent on the integrity of penile tissue structural and vascular components, including an intact arterial inflow into the CC, normal sinusoidal smooth muscle relaxation, tunic tissue elasticity and compliance, and an adequate reduction in venous outflow during erection.1,4,5,47–49 Thus, changes in the CC structure because of TD, including reduced trabecular smooth muscle content, increased connective tissue deposition, and accumulation of adipocytes, may contribute to venous leakage and veno-occlusive dysfunction leading to ED.
Venous leakage is often associated with trabecular tissue structural alterations and abnormal venous outflow resistance because of impaired expandability of the trabecular erectile tissue and inability to compress and elongate the sub-tunical venules against the tunica, thus permitting blood outflow and inhibiting erectile function.
Since vasculogenic ED is attributed to either arteriogenic causes derived from arterial stenosis/occlusion in the hypogastric-cavernous arterial system or venogenic ED due to venous leakage and failure to occlude the subtunical venules sufficiently to maintain intracavernous pressure,1,4,5,47–49 it would be important to determine whether the accumulation of adipocytes in the interface of the CC with the tunica albugenia contributes to the pathophysiological mechanism of veno-occlusive dysfunction.
A host of techniques have been utilized in the evaluation of ED, including dynamic infusion cavernosometry and cavernosography (DICC), duplex doppler ultrasonography, and magnetic resonance imaging.50–59 The DICC is often utilized to assess flow to maintain and venous outflow resistance to better evaluate the veno-occlusive function. This approach defines the hemodynamic parameters related to trabecular smooth muscle relaxation, tissue compliance, and the contribution of venous outflow and arterial inflow to the veno-occlusive function.1,4,5,47–49
The DICC permits examination of the integrity of veno-occlusion function during pharmacological erection and allows assessment of the role of the trabecular smooth muscle relaxation changes in the corpora cavernosa structure in maintaining this mechanism. Maximum equilibrium corporeal body pressure is assessed in DICC by the infusion of vasoactive agents such as papaverine and phentolamine into one corpus to relax corporeal smooth muscle. Intracavernosal pressure at equilibrium represents cavernosal arteries pressure minus loss of pressure from venous drainage. The corporeal body pressure is raised by the infusion of heparinized saline to raise intracorporeal pressure.
Cavernosography is carried out by the infusion of contrast material to raise intracorporeal pressure. These experimental and clinical approaches permit the evaluation of whether the veno-occlusive mechanism is intact or not.1,4,5,47–49 If the corporeal capacitance is very small, then any increase in arterial inflow during erection would bring about marked enhancement of the intracavernosal pressure.1,4,5,47–49 This elevation in pressure results in a very significant state of penile rigidity, indicating potency.
The findings from a number of studies is that changes in the corpora cavernosa structure such as a reduction in the trabecular smooth muscle content, increased connective tissue deposition, or other vascular impediments contribute to veno-occlusive dysfunction.1,4,5,47–49 Thus, it is possible that in patients with TD such changes in penile corpora structure including reduced trabecular smooth muscle mass, increased adipocytes accumulation, and increased connective tissue deposition concomitant with reduced eNOS and nNOS could result in veno-occlusive dysfunction and ED.1,4,5,47–49
It is also well established that androgens regulate the nitric oxide/cyclic guanosine monophosphate (NO/cGMP) pathways, which play a critical role in promoting vascular and trabecular smooth muscle relaxation and increasing blood flow into penile corpora cavernosa. Other androgen-dependent mechanisms involve maintenance of the structural integrity of the cavernosal body and the fibroelastic properties that are responsible for tissue compliance and maintaining erectile physiology.
Thus, it is reasonable to propose that T deficiency may contribute to venous leakage and veno-occlusive dysfunction by multiple mechanisms that encompass the disruption of corpora cavernosa structural integrity, such as the accumulation of adipocytes in the sub-tunical region. Indeed, T deficiency reduces trabecular smooth muscle content and increases extracellular matrix deposition and accumulation of adipocytes.18,19,23,29–32 These observations may account for the observed venous leakage not only in the animal model of T deficiency 20 but also in patients with ED. 46
The accumulation of adipocytes in the sub-tunical region represents a pathophysiological state that was not previously reported; however, it remains to be established whether such accumulation of fat cells does contribute to venous leakage and veno-occlusive dysfunction. Although in the animal model, venous leakage was demonstrated by DICC in castrated rats and testosterone treatment restored erectile function in vivo 20 it remains uncertain as to what degree adipocytes accumulation in the androgen deficiency state contributed to venous leakage and veno-occlusive dysfunction.
We should point out that the exact origin of the adipocytes detected in the CC of the androgen deficient-animal models remains unknown. However, studies in the immature animal model suggest that these adipocytes were differentiated from resident stem cells/pluripotent cells. 45 Further, histological studies of the intact adult rabbit and rat CC do not show the presence of adipocytes. The presence of adipocytes in the CC in the castrated animal model would suggest that the differentiation of preadipocytes (stem cells or other pluripotent cells) into adipocytes was stimulated by androgen deficiency.
As previously discussed, 30 it is possible that trans-differentiation of the trabecular smooth muscle into preadipocytes and finally to adipocytes may occur in the state of androgen deficiency. Currently, there is no evidence in the literature to suggest that trans-differentiation or de-differentiation of the trabecular smooth muscle of the CC takes place during androgen deficiency. Moreover, there are no data in the literature to suggest that adipocytes found in the CC may have migrated from other tissue sources. However, such a possibility may exist, although very remotely. Clearly, more studies are needed to fully examine the contributions of the lipid-containing cells that accumulate in the CC of the penile tissue to the pathophysiology of ED.
In summary, testosterone deficiency results in significant changes in the structural elements of the CC, contributing to trabecular smooth muscle death and atrophy, increased deposition of collagen, and reduced elasticity concomitant with tissue fibrosis and accumulation of adipocytes in the sub-tunical region, resulting in reduced tissue compliance. These structural alterations together with reduced expression of eNOS and nNOS contribute to reduced vascular smooth muscle relaxation and impeding blood inflow into the penis, resulting in a failure to fill.
In addition, the changes in the extracellular matrix and the accumulation of adipocytes may contribute to increased blood outflow, thus contributing to the veno-occlusive dysfunction and resulting in erectile dysfunction. It is postulated that men with testosterone deficiency and erectile dysfunction may benefit from testosterone therapy to improve erectile function.
Footnotes
Authors' Contributions
This work is solely the intellectual effort of the author. The article was conceived, drafted, revised, and finalized by the authors listed in the article.
Acknowledgment
The author would like to thank Dr. Abraham Morgentaler for his careful reading and critical discussion of this article.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This work was not supported by any governmental agency or any other organization. It is simply the genuine effort of the author
