Abstract
Background:
Hypogonadism is characterized by low serum testosterone concentrations and the associated clinical manifestations (e.g., reduced libido, erectile dysfunction, and fatigue); testosterone therapy (TTh) is the primary treatment. This study compared treatment patterns and health outcomes in men using injection TTh with long-acting testosterone undecanoate (TU) injection versus short-acting testosterone cypionate (TC) injection.
Methods:
A U.S. electronic health records (EHR) database was retrospectively reviewed to identify adult men who had TTh of TU or TC (index treatment) between January 1, 2014, and December 31, 2018, and ≥1 EHR within the 12 months before and 6 months after index treatment.
Results:
There were 948 patients in the TU cohort and 121,852 in the TC cohort, of whom 419 and 86,219, respectively, were TTh-naive at index treatment. Hypogonadism diagnosis was documented in 81.1% and 58.8% of patients in the overall TU and TC cohorts, respectively. During the 1-year postindex period, higher adherence rates were observed in the TU versus TC cohort during months 7 through 12 (82.0% vs. 40.8%; p < 0.001), and a greater percentage of patients continued to receive TU (41.9%) versus TC (8.2%) for 12 months (p < 0.001). Variability in total testosterone levels was lower for TU versus TC in the TTh-naive population.
Conclusions:
In this retrospective EHR study, men who received TU demonstrated a two-fold higher adherence, a five-fold improved maintenance on index TTh, and more stable testosterone levels at 1-year follow-up versus men who received TC. More consistent TTh use observed with TU may result in improved health outcomes for men with hypogonadism.
Introduction
Hypogonadism is characterized by low serum testosterone concentrations and the associated clinical manifestations (e.g., reduced libido, erectile dysfunction, and fatigue) resulting from disruption of the hypothalamic/pituitary/gonadal axis.1,2 Testosterone therapy (TTh), the primary treatment for hypogonadism, is available in a variety of formulations including transdermal, oral, buccal, nasal, implantable, short-acting injectable, and long-acting injectable options.1–3 In some men, TTh has been shown to increase libido, improve erectile function, restore muscle mass, prevent bone loss, reduce depressive symptoms, and improve quality of life.4–9
As is the case for many long-term therapies, issues with treatment adherence (i.e., using the treatment as prescribed 10 ) and treatment persistence (i.e., continuing to use the prescribed treatment over time 10 ) have been observed with TTh.11–14 Low rates of medication adherence and treatment persistence have been observed across TTh formulations, including topical, transdermal, oral, and injectable products, with 50% to >80% of patients discontinuing prescribed TTh within 12 months.11–14
Whereas short-acting testosterone injections are usually given on a weekly or biweekly basis, long-acting injections are generally administered every 10 weeks. 3 Consistent with research demonstrating that reductions in dose frequency improved adherence for oral medications,15,16 preliminary evidence suggests that TTh persistence may be improved with the use of long-acting injections. 17
Although the efficacy and safety of the testosterone undecanoate (TU) intramuscular injection have been widely studied,6,18–23 the comparative characteristics of use and adherence for the TU 750-mg formulation available in the United States versus short-acting injectable testosterone formulations (e.g., testosterone cypionate [TC]) have not been examined in detail.
In particular, there is a lack of real-world differentiation regarding the clinical impact of these formulations of TTh. Both TU 24 and TC 25 are approved by the U.S. Food and Drug Administration (FDA) for testosterone replacement therapy in adult males with primary hypogonadism or hypogonadotropic hypogonadism. We hypothesized that the convenience of TU as a long-acting formulation would result in differences in use characteristics and possibly health outcomes compared with short-acting TC.
The objective of the current study was to use an electronic health records (EHR) patient database to evaluate and compare patient characteristics, treatment patterns, and health outcomes for adult men receiving TTh with TU or TC.
Materials and Methods
Data source and study population
This retrospective cohort study was conducted using patient-level data from Veradigm™ (Allscripts, Chicago, IL), a longitudinal, Health Insurance Portability and Accountability Act of 1996 (HIPAA)-compliant, U.S. patient database of EHR from ambulatory facilities. The clinical database used for this study contained information for ≈30 million patients and included demographics, diagnoses, medications, procedures, laboratory test results, and clinical observations from January 1, 2003, through July 31, 2019. This study was exempted from IRB review because research procedures, including data collection and analysis, were limited to the use of an anonymized, HIPAA-compliant database.
Men ≥18 years of age with a TU or TC injection administered between January 1, 2014, and December 31, 2018, were eligible for inclusion. Patients were classified into treatment cohorts using the following algorithm: patients with a prescription for TU were included in the TU cohort; among the remaining patients, those with a prescription for TC were included in the TC cohort.
The index treatment in each cohort was defined as the first TU or TC administration between January 1, 2014, and December 31, 2018. Patients were required to have ≥1 EHR in the database within 12 months (360 days) before the index treatment date and ≥1 EHR within the first 6 months (180 days) of the index treatment date. Patients were considered to be TTh-naive if they did not receive TTh within 12 months before the index treatment date. Patient information was available for a minimum of 6 months after the index date, with data collected in 6-month increments through the end of the data window (July 31, 2019).
For laboratory or other physiologic data, the baseline period was defined as 3 months (90 days) before, or on, the index treatment date. Comorbidities and prior medication use were reported using all information available in the database.
Adherence and outcome assessment
Patients were classified into postindex categories reflecting patterns of TTh use (i.e., maintained, switched, or discontinued). Maintenance on index treatment was defined as a gap of no more than twice the recommended dosing interval (i.e., 20 weeks [140 days] for TU or 4 weeks [28 days] for TC) between the end date of one prescription and the date of the next prescription. Switch to a nonindex TTh was defined as cessation of TU and initiation of non-TU TTh within 20 weeks (140 days) from the end date of the last TU prescription, or cessation of TC and initiation of non-TC TTh within 4 weeks (28 days) from the end date of the last TC prescription.
Discontinuation of index treatment was defined as cessation of TU index treatment and no initiation of non-TU TTh within 20 weeks (140 days) from the end date of the last TU prescription, or cessation of TC index treatment and no initiation of non-TC TTh within 4 weeks (28 days) from the end date of the last TC prescription.
For the index treatment (TU or TC), treatment adherence was assessed from the index date until an identified discontinuation or switch date or, for patients maintained on index treatment, until the end date of the last prescription. The assessment of treatment adherence was based on gaps between index therapy prescriptions. The end date for each prescription was calculated from the administration start date plus the length of supply in days. When the supply duration was zero or missing, a value was assigned based on the recommended dosing interval: for TU prescriptions, a value of 28 days (4 weeks) was assigned for the initial dose and a value of 70 days (10 weeks) was assigned for all other doses; for TC prescriptions, a value of 14 days (2 weeks) was assigned.
The end-to-start gap was defined as the time between the end date of an index prescription and the date of the next index prescription. Patients in the TU cohort were considered nonadherent if there was a gap >42 days (6 weeks) between the initial TU prescription end date and the second TU prescription start date or, for all prescriptions after the second, an end-to-start gap >105 days (15 weeks). Patients in the TC cohort were considered nonadherent when there was an end-to-start gap >21 days (3 weeks).
Outcome assessments included change from baseline in body weight, body mass index (BMI), blood pressure levels, total and free testosterone levels, and the incidence of newly diagnosed cardiovascular events and risk factors (e.g., cerebrovascular disease/stroke, congestive heart failure, erectile dysfunction, hyperlipidemia/dyslipidemia, hypertension, myocardial infarction, obesity, and type 2 diabetes). “Newly diagnosed” conditions were defined as those for which a diagnosis code was present in the follow-up period (12 months after the index treatment date) and absent in the baseline period (3 months preindex).
Statistical analyses
Statistical analyses were performed using SAS version 9.4 software (SAS Institute, Inc., Cary, NC). Patient characteristics, patterns of TTh use, and health outcomes were compared for the overall TU versus TC cohorts and for subcohorts of TTh-naive patients using t-tests for continuous variables and chi-square tests for categorical variables. All comparisons were conducted using a two-tailed test of significance and a type I error rate of 0.05.
Results
Study population
The analysis included 948 men in the TU cohort and 121,852 men in the TC cohort (Fig. 1). Baseline characteristics were similar in terms of body weight, BMI, and most medical comorbidities. Comparing the TU cohort with the TC cohort at baseline, a small but statistically significant difference was observed for mean systolic blood pressure (129.6 mmHg vs. 130.7 mmHg, respectively; p = 0.04) and mean diastolic blood pressure (78.5 mmHg vs. 79.6 mmHg, respectively; p = 0.001). Before receiving the index treatment, a significantly greater percentage of patients in the TU cohort had established diagnoses of hypogonadism, erectile dysfunction, obesity, and polycythemia/erythrocytosis, whereas congestive heart failure was significantly more common in the TC cohort (Table 1).
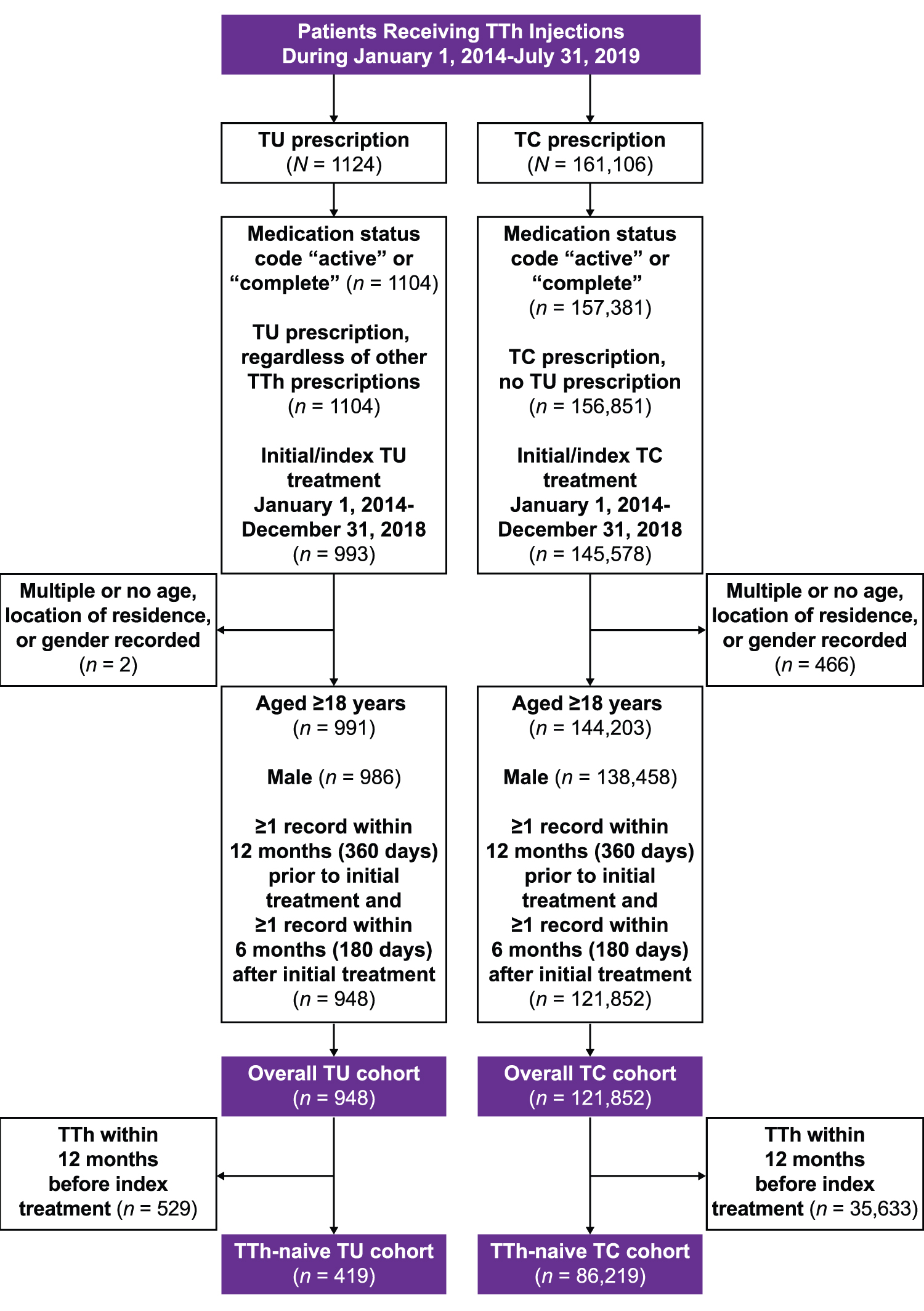
Study disposition. TC, testosterone cypionate; TTh, testosterone therapy; TU, testosterone undecanoate.
Baseline Demographics and Clinical Characteristics
At initial treatment date.
p = 0.005 versus. TC.
Up to a 3-month baseline time frame.
p = 0.04 versus. TC.
p < 0.001 versus. TC.
p = 0.03 versus. TC.
Based on ICD codes in the database and not mutually exclusive; no documented hypogonadism for 18.9% of TU patients and 41.2% of TC patients in the overall population.
ICD code on/before initial treatment date.
p = 0.004 versus. TC.
p values not available.
Before initial treatment date (on or before for “other testosterone medications”).
BMI, body mass index; DBP, diastolic blood pressure; ICD, International Classification of Diseases; SBP, systolic blood pressure; TC, testosterone cypionate; TE, testosterone enanthate; TTh, testosterone therapy; TU, testosterone undecanoate.
In the TU and TC cohorts, 18.9% and 41.2% of patients, respectively, did not have a documented diagnosis of hypogonadism recorded in the database. The mean [SD] free testosterone level at baseline (within 90 days before index treatment) was higher in the TU cohort (65.2 [77.1] pg/mL) versus the TC cohort (38.8 [57.8] pg/mL; p < 0.001), whereas the mean [SD] total testosterone level was similar (322.2 [237.6] ng/dL vs. 310.1 [251.9] ng/dL). Previous TTh use was more common in the TU cohort compared with the TC cohort (Table 1), and the percentage of TTh-naive patients (defined as those with no TTh within 12 months before the index treatment date) was lower in the TU (44.2% [419/948]) versus TC (70.8% [86,219/121,852]) cohort.
Treatment patterns
During the 12 months of follow-up postindex treatment, rates of adherence to the prescribed TTh were similar between the two cohorts during the first half of the year (months 1–6), but were significantly higher in the TU cohort versus the TC cohort during the latter 6 months (82.0% vs. 40.8%; p < 0.001; Fig. 2).

Adherence to prescribed TTh during 12 months of follow-up. *p < 0.001 versus TC cohort.
In addition, a significantly greater percentage of patients were maintained on their index TTh in the TU cohort (41.9% [n = 397]) compared with the TC cohort (8.2% [n = 10,036]; p < 0.001; Fig. 3), and a significantly smaller percentage of patients in the TU cohort had discontinued all forms of TTh compared with the TC cohort (33.8% [n = 320] vs. 89.9% [n = 109,528], respectively; p < 0.001; Fig. 3). Thus, the rate of TTh treatment persistence was 10.1% in the TC cohort compared with 66.2% in the TU cohort.
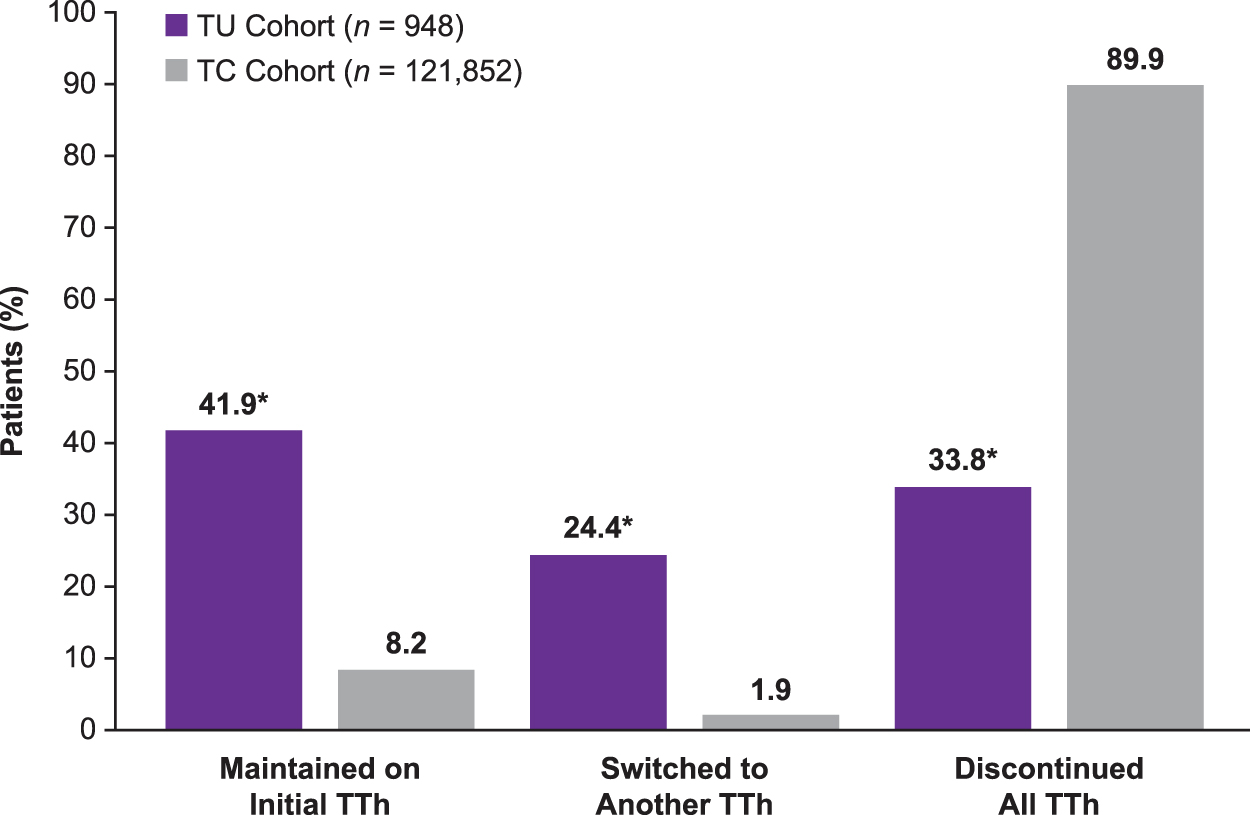
Treatment persistence during 12 months of follow-up. *p < 0.001 versus TC cohort for the overall analysis of TTh use (maintained, switched, discontinued).
Testosterone levels
The values for mean change in total testosterone (171.7 ng/dL vs. 59.6 ng/dL; p < 0.001) and free testosterone (25.4 pg/mL vs. 3.7 pg/mL; p = 0.001) levels at the 12-month follow-up were significantly greater in the overall TC cohort versus overall TU cohort (Table 2). Variability in testosterone levels was assessed in patients naive to TTh, based on the range of observed values for each patient during the 12-month follow-up period. Variability in total testosterone levels was significantly lower in the TU versus the TC cohort for both the overall TTh-naive population (mean of the observed range [maximum-minimum], 187.3 ± 225.6 ng/dL vs. 242.2 ± 313.3 ng/dL; p = 0.04) and those within this population who were maintained on their index therapy (126.9 ± 147.4 ng/dL [TU] vs. 275.5 ± 319.2 ng/dL [TC]; p = 0.03) (Table 3).
Cardiometabolic-Related Outcomes and Risk Factors at 12-Month Follow-Up in the Overall Patient Cohorts
n = 566.
n = 57,973.
n = 582.
n = 58,588.
n = 690.
n = 70,856.
n = 317.
n = 19,480.
n = 129.
n = 5788.
ICD code not present in the 3-month baseline time frame and present in the 12-month follow-up period.
Cardiometabolic-Related Outcomes and Risk Factors at 12-Month Follow-Up in TTh-Naive Patients
ICD code not present in the 3-month baseline time frame and present in the 12-month follow-up period.
The difference between the maximum and minimum values during the 12-month follow-up period was calculated for each patient and the mean (SD) was determined for each cohort.
Cardiometabolic-related outcomes
The mean change from baseline in weight, BMI, systolic blood pressure, and diastolic blood pressure at the 12-month follow-up was similar between patients receiving TU or TC (Table 2). Newly diagnosed erectile dysfunction and obesity were significantly more common in the TU cohort, whereas newly diagnosed hypertension, congestive heart failure, and chronic pain were more common in the TC cohort (Table 2).
In patients who were TTh-naive, systolic blood pressure decreased significantly from baseline to 12-month follow-up in the TU cohort (mean Δ, −2.3 mmHg) versus the TC cohort (mean Δ, 0.0 mmHg; p = 0.02; Table 3). In TTh-naive patients who were maintained on index treatment, a significantly lower rate of newly diagnosed hypertension was observed after 12 months in men treated with TU versus TC (28.6% vs. 43.7%; p = 0.03; Table 3). Comparing TTh-naive patients in the TU versus TC cohort, incident rates were significantly lower for hyperlipidemia/dyslipidemia (30.4% vs. 44.4%, respectively; p = 0.04), fatigue (5.4% vs. 16.4%, respectively; p = 0.003), and chronic pain (16.1% vs. 46.0%, respectively; p < 0.001) (Table 3).
Discussion
TTh is widely used in the treatment of men with hypogonadism 1 ; however, overall adherence and persistence are suboptimal.11–14 The use of medications that reduce dosing frequency may improve persistence with treatment and, thereby, help improve clinical outcomes. 26 This retrospective cohort study was conducted to address the need for real-world data on the potential benefits of different formulations of TTh and the clinical impact on health-related outcomes. In the current study, patient characteristics, patterns of use, and health outcomes were compared for men treated with TU versus TC.
There were two major findings in this study. First, in the analysis of TTh use patterns, it was observed that patients who received TU (index treatment) had notably higher persistence during the 12-month follow-up period versus patients who received TC (index treatment). In the TU cohort, 41.9% of patients continued treatment with TU and another 24.4% switched to a different TTh, for an overall TTh persistence rate of 66.2%. In the TC cohort, the overall TTh persistence rate was only 10.1% (8.2% continued TC and another 1.9% switched to a different TTh). The significantly higher TTh persistence with TU versus TC (p < 0.001) may be related to the less frequent dosing schedule recommended for TU, a long-acting formulation, in comparison with TC, a short-acting formulation.
The second major finding was that patients treated with TU were significantly more adherent to their prescribed TTh regimen than patients treated with TC: 82.0% of patients were adherent to TU, whereas 40.8% were adherent to TC during months 7 to 12 of treatment (p < 0.001). These differences support our initial hypothesis that the convenience of long-acting TU would result in better real-world use characteristics than short-acting formulations such as TC.
For patients requiring long-term treatment for chronic medical conditions, the importance of medication adherence for improving health outcomes and reducing health care costs is well known.26,27 The benefits of TTh in men with hypogonadism, not only for improving sexual function but also for mitigating the detrimental effects of comorbidities such as obesity and type 2 diabetes, have been clearly established.1,2,28 The substantially higher rates of medication adherence and treatment persistence observed in the current study for TU relative to TC indicated that men in the TU cohort were more likely to be receiving adequate treatment for their hypogonadism and associated comorbidities.
Thus, the findings of the current study suggest that the use of a long-acting testosterone injection may improve patient adherence with prescribed TTh and increase the likelihood that patients will follow health care provider recommendations regarding continued treatment to maximize TTh benefits on clinical outcomes. Additional research is warranted to investigate the potential benefits of improved adherence and persistence with TTh for men with hypogonadism and should include longer term follow-up to evaluate effects on health outcomes.
In the current study, variability in total testosterone levels during 1 year of follow-up was significantly lower in the TU versus TC cohort, which may be attributed to the potentially more favorable pharmacokinetic properties of TU. Long-acting depot medications such as TU have been shown to produce more stable physiologic levels of testosterone, which may ameliorate the fluctuations in clinical symptom improvement (e.g., mood, energy, and sexual function) that have been reported with short-acting TTh.18,20,29 It is notable that the increases in mean testosterone levels observed at 1-year postindex treatment were significantly greater in patients in the TC versus TU cohort.
We interpret this finding with caution because most patients in the TU cohort had a previous history of TTh use, which may have impacted baseline testosterone levels observed before initiation of TU (index treatment). In addition, in the TC cohort, a substantial percentage of patients did not have a confirmed diagnosis of hypogonadism at baseline, which is suggestive of greater off-label use of short-acting injectable TTh.
The incidences of newly diagnosed hypertension, hyperlipidemia/dyslipidemia, fatigue, and chronic pain within the 1-year postindex period were significantly lower in patients naive to TTh who initiated and maintained use of TU compared with those who initiated and maintained use of TC. The incidences of newly diagnosed congestive heart failure, peripheral vascular disease, and type 2 diabetes were numerically lower in previously TTh-naive patients who were maintained on TU versus TC for 1 year; the lack of statistical significance was likely due to the small sample size in the TU cohort and the limited duration of follow-up.
The extent to which baseline differences between the treatment cohorts contributed to differences in health outcomes is unknown. The impact of TTh on cardiovascular risk remains an open question.30,31
The FDA requires a class-wide warning for prescription testosterone products, noting that some studies have reported an increased risk of myocardial infarction and stroke associated with the use of testosterone replacement therapy, while others did not. However, there is evidence suggesting that TTh is not associated with increased cardiovascular risk and may even be cardioprotective.2,32,33 Additional data are needed from adequately powered, long-term, placebo-controlled trials.
This was a retrospective study using EHR, and the limitations inherent in this type of database apply to the current study. Data availability was limited to information captured in the EHR database, which may not fully represent treatment patterns and medical outcomes.
For example, many patients treated with TTh did not have a diagnosis of hypogonadism documented in the EHR database, and it is unclear whether the lack of a diagnostic code could be due to the code being entered before the defined preindex observation; the patient not having the diagnosis; or the patient having the diagnosis but the information not being entered into the database (e.g., error). The database does not provide time-of-day (time stamp) values, and thus, timing of assessments (e.g., testosterone levels) was possible only with day-level granularity.
A future analysis including a patient cohort with a confirmed diagnosis code for hypogonadism and time-of-day data for blood samples (given diurnal variations in testosterone levels) may provide additional information about differences between long-acting and short-acting TTh in a more homogeneous population. Inclusion of additional TTh formulations (e.g., pellets for subcutaneous implantation, oral tablet, and topical gel) also would be informative; however, restricted availability of data on products with specialty pharmacy distribution, limited data tracking on topical formulation use, and limited uptake for the oral formulation since approval may be a substantial challenge.
Another study limitation was that patterns of use and treatment adherence and persistence were evaluated only until the end of the study time frame, with a restricted (12-month) duration of follow-up. Also, for analyses of TTh use, patients may not have had contiguous data during the study period. Data were not available on patient resource utilization not serviced by the EHR systems; therefore, any patient who may have continued TTh outside of the EHR database was classified as having discontinued therapy. It should also be noted that this analysis focused on a TC dosing frequency of once every 2 weeks; however, this medication may be prescribed at a frequency of once every 2–4 weeks.
It has been challenging to identify a U.S. database with an adequate number of patients taking TU; this database included a smaller number of patients treated with TU versus TC, and the sample size of TU-treated patients was even smaller in subgroup analyses of TTh-naive patients. Thus, some smaller magnitude differences in health outcomes (e.g., systolic and diastolic blood pressure) may not represent generalized findings, due to the relatively small sample size in the TU cohort, and require replication in a study with a larger sample size. Findings are limited by the lack of a propensity-score matching analysis to control for baseline differences between the treatment cohorts, also due to a small sample size.
Conclusions
This retrospective study is the first comparative analysis between long- and short-acting injectable TTh using an EHR database. At 1 year of follow-up, men who received TU demonstrated a two-fold higher adherence, a five-fold improved maintenance on index TTh, nearly a two-thirds lower TTh discontinuation rate, and more stable testosterone levels compared with men who received TC. Further research is needed to determine whether increased adherence with the use of long-acting TU results in long-term improved health outcomes for men with hypogonadism.
Footnotes
Authors' Contributions
Y.H. and K.L. conceived and designed the article. K.L., S.T., and D.K.H. acquired the data, and all authors performed data analysis. A.M. and Y.H. drafted the article. All authors revised the article and approved the final article.
Acknowledgments
Technical editorial and medical writing assistance were provided, under the direction of the authors, by Mary Beth Moncrief, PhD, and Nancy Holland, PhD, Synchrony Medical Communications, LLC, West Chester, PA.
Author Disclosure Statement
A.M. reports receiving a research grant from Endo Pharmaceuticals, Inc., and serving as a consultant to Acerus Pharmaceuticals, Bayer, and Clarus Therapeutics, Inc. Y.H. is an employee of Endo Pharmaceuticals, Inc. K.L., S.T., and D.K.H. are employees of Boston Strategic Partners, which conducted this analysis with funding from Endo Pharmaceuticals, Inc.
Funding Information
Funding for this study was provided by Endo Pharmaceuticals, Inc., Malvern, PA.
