Abstract
Although the cognitive benefits of estrogen therapies have been extensively studied in women, understanding of the cognitive benefits of androgen therapies in men has lagged behind. This review synthesizes current clinical research and animal research on the effects of testosterone and its major metabolites on spatial memory in young and aged males. Spatial memory and associated hippocampal function show age-related decline, and current research indicates that testosterone treatments may ameliorate some of the cognitive effects of aging. This review highlights testosterone dose, long-term versus working memory, place versus response strategies, and estrogen versus androgen pathways as key variables that influence the relative impact of testosterone on spatial memory. Despite inconsistencies, there is considerable evidence from rodent and clinical research that testosterone can improve spatial memory. The cognitive benefits are most consistent when testosterone is given to hypogonadal males for a prolonged period.
Introduction
A decline in some forms of memory is a well-known consequence of aging. 1 Normal aging is associated with deficits in episodic memory, attention, and working memory, with other cognitive domains remaining relatively intact.2,3 By age 50, the prevalence of memory impairment is more than three times greater than the general population, at 5.8% and 18.5%, respectively. 4 This decline in cognitive function may be partially due to decreasing levels of sex steroid hormones with aging, pointing to an important clinical role for testosterone.5,6 Aging also increases the risk for pathological memory loss, including mild cognitive impairment and dementia, and there is evidence that risk of both these conditions increases with reduced testosterone.6,7 This review will summarize evidence from human and rodent research that testosterone influences memory in both young and aged males.
Although aging leads to a general decline in learning and memory, spatial memory is particularly vulnerable.3,8 Spatial memory, broadly defined, is the ability to perceive, encode, store, visualize, retrieve, manipulate, transform, and integrate spatial information drawn from one or multiple environmental cues.9,10 This important cognitive ability is nurtured through an organism's typical exploration of its environment and is critical in acquiring new information to inform later navigational decisions. Similar to all forms of memory, spatial memory can be further subdivided into long-term, short-term, and working memory. 11 Long-term spatial memory is typically classified as a type of episodic memory: explicit memories about personal experiences that do not easily decay over time. 12 In the context of rodent research, long-term spatial memory is often referred to as “reference memory,” 13 but we will use “long-term memory” for this review to maintain consistency. Short-term memory is distinct from long-term memory in that it decays more rapidly over time and has limited capacity. 11 Working memory involves actively applying a memory to attend to a specific task for as long as it is useful to complete the task. 11 These distinctions between forms of spatial memory are important, as testosterone's effects vary depending on the type of memory. Among the forms of memory, short-term is the most difficult to characterize and has been rarely assessed in the context of testosterone studies, so our focus will be the distinction between working and long-term memory.
When an individual is navigating through space to reach a goal, one of two strategies can be employed: a place strategy or a response strategy. 14 A place strategy involves using knowledge about the positions of environmental cues relative to a goal and relative to one's own position to locate the goal,15,16 whereas a response strategy involves using stimulus–response associations to locate a goal.15,16 There is considerable evidence that the ability to effectively use a place strategy relies on the hippocampus, whereas use of a response strategy relies on the striatum.15,17–19 For example, acetylcholine release in the hippocampus predicted use of a place strategy for adult male rats in a T-maze, whereas acetylcholine release in the striatum predicted use of a response strategy. 20 The terms allocentric strategy and egocentric strategy are also sometimes used, 21 and these are roughly equivalent to place and response strategies, respectively.
When considering the effects of sex steroids on the various forms for spatial memory, the distinction is often made between organizational and activational effects. The steroidal conditions in which the brain develops have a profound effect on the long-term organization of the brain in terms of both neuroanatomy and neurochemistry.22,23 These organizational effects occur during critical periods of brain development (e.g., neonatal period and puberty), whereas the more transient effects of sex steroids on the adult brain are referred to as activational. Numerous experiments with humans and rats have demonstrated that testosterone has organizational effects on the brain early in development that lead to enhanced spatial learning and memory in adulthood.24–28 In one experiment, testosterone was given to male and female rats in the first week after birth. 28 Testosterone-treated females performed better than control females and comparable with control males on the Morris water maze (MWM), which assesses long-term spatial memory. In contrast, testosterone treatment during early development had minimal effect on male spatial memory. Although these organizational effects of testosterone are important developmentally, the focus of this review will be on the activational effects of testosterone. It should also be acknowledged that the adult brain is more plastic than was once believed, and the activational effects of sex steroids can influence many forms of neuroplasticity in the adult (e.g., synaptogenesis and neurogenesis), as described hereunder.
Sex Differences in Spatial Memory
A sex difference in a physiological or behavioral trait suggests that it may be regulated by sex steroids, and the same is true for cognitive traits. A male advantage for spatial ability is one of the best characterized human sex differences in human cognition.29–32 This sex difference is most apparent for the mental rotation task, which tests participants' ability to mentally rotate a two- or three-dimensional object. 31 Brain-imaging studies indicate that this task relies heavily on cortical brain regions, with males relying more on the parietal cortex and females relying more on the frontal cortex.33–37 Males also outperform females on some other spatial tasks, including object location memory, 38 route-learning,39,40 and maze navigation.41–46 Based on functional magnetic resonance imaging (fMRI) studies, maze navigation tasks rely heavily on the hippocampus and parietal lobes, with males generally showing greater activation of the hippocampus than females.47,48
Among rats, males have better working and long-term spatial memory based on performance in the MWM,49–52 radial-arm maze (RAM),53,54 and object location memory task. 55 For example, one study demonstrated that female rats performed better than males on the novel object recognition task, whereas males performed better than females on the object location memory task. 56 The object location task relies more on the hippocampus than does the novel object task, 57 suggesting a sex difference in hippocampal function. A meta-analysis of experiments conducted with rats and mice also concluded that there is a male advantage on spatial tasks. 58 It is important to note, however, that human females consistently outperform males on tasks that involve remembering the relative position of objects in an array.59–61 Similarly, one experiment found that female rats had longer memory retention than males on a task that involved remembering the relative positions of two objects in an array of four objects. 55
When solving spatial tasks, men preferentially use a place strategy involving Euclidian cues, whereas women preferentially use a response strategy involving local landmarks,62–65 and some studies have found that men perform better than women on tasks that require a place strategy.66,67 For example, in a virtual reorientation task, females were more likely to use a response strategy to find a goal, whereas males were more likely to use a place strategy (i.e., create a mental map of the virtual environment). 68 Similar sex differences in spatial strategy use have been demonstrated with rodent experiments. In a classic study, Williams et al. 69 found that altering the geometry of the testing room impaired performance of male but not female rats on the RAM. In contrast, rearranging landmark cues impaired the performance of females but not males. 69 Thus, males relied on a place strategy and females relied on a response strategy. Similarly, female rats show a strong bias for using local landmarks to find the escape platform in water maze tasks,49,70,71 whereas males rely more heavily on extra-maze cues.72,73 Male rats outperformed females in the water maze when a place strategy was required to solve the task, but no sex difference was observed when response strategies (local cues or a turn bias) could be used to solve the task. 74 Similarly, female rats made more errors than males when they had to switch from a response strategy to a place strategy. 16
There is considerable evidence that the activational effects of sex steroids play a causal role in sex differences in spatial strategy preferences as well as spatial learning and memory more broadly. Meta-analyses indicate that sex differences in various spatial abilities become more apparent after puberty in humans.29,31 However, a female bias for locating and using landmarks for navigation seems to arise before puberty, 75 and some studies have found a sex difference in route learning in children as young as 6 years old.76,77 No sex differences were observed on the water maze among juvenile rats (18–24 days old), 78 and one study found that unlike adult rats, juvenile rats did not show a sex difference in spatial strategy preference. 71 Thus, at least some sex differences in spatial memory seem to be due to the activational effects of sex steroids, whereas others may be established early in development.
Human Research with Younger Males
Some reviews have concluded that elevated circulating testosterone levels do not consistently improve spatial memory in men.79,80 Despite inconsistencies, however, past research indicates that testosterone levels in males correlate with improved mental rotation ability,81–83 route-learning, 84 and performance on a block design task. 85 Discrepancies among studies seem to be at least partially due to dose-dependent effects of testosterone. The relationship between circulating testosterone levels and spatial memory has often been described as an “inverted U” or negative parabolic relationship. Shute et al. 86 conducted two studies on free circulating testosterone in healthy adults. They found that males with relatively low levels of circulating testosterone performed better on spatial tests than males with higher levels. This trend was reversed for females—those with relatively high levels of circulating testosterone performed better on spatial tests than females with lower levels. This relationship has been replicated several times,87,88 suggesting that relatively high circulating testosterone levels improve spatial abilities in females but impair spatial abilities in males. In support of this conclusion, females given a single administration of testosterone made fewer errors on a mental rotation task than did those given a placebo.89,90 Interestingly, there is evidence for a similar negative parabolic relationship between perinatal androgen exposure and masculine sexual behavior from both human and rodent research. 91
There are also some studies with all-male populations that have supported the optimum-testosterone hypothesis.44,92 For example, one study observed the effects of testosterone on spatial memory among eugonadal and hypogonadal males of approximately the same age. 93 In this study, supraphysiological testosterone levels were induced by injecting eugonadal males with testosterone. Spatial memory performance decreased in the testosterone-supplemented group relative to a eugonadal placebo group, whereas the hypogonadal group performed worse than either eugonadal group. 93 Some studies have demonstrated that long-term use of anabolic-androgenic steroids impairs spatial memory,94,95 supporting the conclusion that supraphysiological levels of androgens have negative effects on memory. The dose-dependent effects of testosterone were also demonstrated by a study that analyzed both circulating testosterone and androgen receptor genotype. 44 The androgen receptor gene has a trinucleotide (cytosine-adenine-guanine [CAG]) sequence repeat on exon 1, and longer repeats are associated with weaker receptor activation. 96 Among males with the genotype for lower androgen receptor activity, higher testosterone levels predicted poorer performance on a virtual water maze task, whereas no relationship between testosterone level and performance was observed among males with high androgen receptor activity. 44 A more recent study with younger males found a positive relationship between number of CAG repeats and performance on a spatial visualization task. 97 Thus, although testosterone dose is clearly a critical variable, it may interact with other variables such as sex and genotype to determine the effects of testosterone on spatial memory.
Experimental manipulation of testosterone levels in younger men has primarily involved populations undergoing treatment for prostate cancer. Androgen deprivation therapy is a common treatment for prostate cancer because it reduces the rate that the cancer spreads. 98 Androgen deprivation therapy is sometimes followed up by testosterone treatment, but the therapeutic benefits of testosterone therapy remain controversial.99,100 Among patients with prostate cancer, androgen deprivation therapy caused significant cognitive impairments, including impaired spatial memory, which were associated with changes in the right parietal–occipital regions of the brain.101–105 However, in an experiment in which testosterone was given to hypogonadal young to middle-aged males (20–59 years old), no changes in spatial ability were observed. 106 Similarly, a short-term androgen deprivation treatment was found to have no effect on spatial cognition in both healthy younger (25–35 years old) and healthy older (60–80 years old) males. 107 In a study in which 2 years of testosterone treatment was given to hypogonadal young to middle-aged males without prostate cancer, regular testosterone injections resulted in improvements in attention, visual scanning ability, executive function, and psychomotor speed. 108 The opposite effect was found in eugonadal males, with subjects who received testosterone biweekly for 8 weeks performing worse than the placebo group. 93 In summary, studies in which testosterone levels were manipulated in young males provide only mixed evidence to indicate an activational role for androgens on spatial memory, with testosterone dose, duration of treatment, and possibly health status (i.e., with or without cancer) being key variables.
Few human studies have tested the effects of testosterone on the use of specific spatial strategies. A study of male and female college students found that higher levels of circulating testosterone in males, but not females, were associated with more use of a place strategy. 84 However, for a judgment of line orientation task, the addition of landmarks, which facilitates the use of a response strategy, helped males to solve the task if they also had relatively high circulating testosterone levels. 66 Given that the results of these two experiments are somewhat contradictory, further study is needed in this area.
Rodent Research with Younger Males
Given the lack of consistent results from human studies, it is useful to look to rodent research, which allows for better control of variables and easier use of testosterone manipulation. Multiple tasks are commonly used to test spatial learning and memory in rodents, varying in the type of memory tested and the motivator. The MWM requires rodents to escape a cold pool of water by navigating toward a hidden platform in opaque water using only external visual cues, with testing typically occurring over multiple days to measure long-term memory. 109 The Barnes maze is another aversively motivated task, which involves training subjects to find an escape hole among many holes around the edge of a circular platform, and this task is also most frequently used to measure long-term memory. 110 The RAM is another commonly used spatial task, consisting of straight arms (8, 12, or 16) radiating out from a single central platform, with food rewards placed at the ends of specific arms. Arm entries can be used to score both working memory and long-term memory. 13 Finally, the object location memory task relies on a rodent's natural tendency to explore novelty. Subjects are first allowed to explore two objects, and then one of the objects is moved and the subjects are re-exposed to the objects after a retention interval. Increased exploration of the moved object relative to the unmoved object is indicative of good spatial memory. 111 Various T-maze and Y-maze tasks have also been devised, with exploration of novelty and food reward being used as motivators.
The effects of testosterone on spatial memory among rodents are most clear for spatial working memory. In various versions of the RAM, T-maze tasks, and Barnes maze, castration impaired spatial working memory.53,112–117 Furthermore, testosterone given to castrated male rats improved spatial working memory on the RAM and T-maze.117–119 Thus, there is considerable evidence that testosterone improves spatial working memory, corroborating some findings from human research.
The effects of testosterone on long-term spatial memory among male rodents have been less consistent than the effects on spatial working memory. Experiments using the RAM suggest that testosterone either impairs long-term spatial memory53,113 or has no effect. 120 Studies employing the MWM have shown that testosterone improves,118,121–123 impairs,124–126 or has no effect113,127–130 on long-term spatial memory. Both the MWM and the RAM typically involve a 24-h retention interval. Experiments with the object location memory task have consistently shown that testosterone improves long-term spatial memory,120,131,132 with the key distinction compared with the MWM being that relatively short (30–120 min) retention periods were used. Thus, testosterone may act primarily on working memory and long-term memories of shorter duration, but there are exceptions to this conclusion. Hawley et al. 133 studied the role of testosterone on long-term memory using a version of the Y-maze that is comparable to the object location task in that it relies on a rat's interest in exploring a novel location. They found that castration impaired performance after a 48-h retention interval and that performance was restored by testosterone implants. Similarly, one experiment using the Barnes maze demonstrated that castration impaired and testosterone implants restored long-term spatial memory (7-day interval). 115 Procedural differences among tasks, possibly involving relative stress levels, may explain the inconsistent effects, but there is growing evidence that testosterone can enhance long-term spatial memory.
Supporting the previously mentioned results of human studies, experiments with rodents have shown that the effects of testosterone on spatial memory vary with dose. A number of studies using the MWM demonstrating a negative effect of testosterone or no effect on long-term spatial memory have involved supraphysiological doses.125,127,130,134–136 In contrast, Spritzer et al. 118 found that testosterone doses ranging from low physiological to slightly supraphysiological resulted in better long-term spatial memory on the MWM relative to castrated controls. A subsequent experiment showed that both high and low physiological doses of testosterone improved spatial working memory (RAM) and long-term spatial memory (object location memory) in castrated male rats, but an intermediate dose did not. 120 These results suggest that the dose–response relationship for testosterone and spatial memory may be even more complex than the negative parabolic relationship that has been suggested by human research. Other experiments with male rats provide some support for this conclusion. Hawley et al. 133 gave castrated rats two doses of testosterone followed by testing on a Y-maze, and they found that the higher dose of testosterone, but not the lower dose, restored long-term spatial memory. This suggests that doses of testosterone that elevate serum testosterone levels slightly above a physiological baseline are sufficient to restore long-term spatial memory in castrated males. Jia et al. 122 documented a negative parabolic relationship between testosterone dose and long-term spatial memory using the MWM. Thus, dose influences the effects of testosterone on both spatial working memory and long-term spatial memory, but the nature of the dose–response relationship is still being clarified.
Another critical variable that has not been considered in most previous studies with rodents is whether a spatial task is testing place memory, response memory, or some combination of the two. Place and response memory are assessed in rodents using various versions of a T-maze (Fig. 1), with a dual-solution task used to assess strategy preference and specific place and response tasks used to assess ability to use a particular strategy. The effects of estrogens on spatial strategies have been extensively tested using female rats. 137 Ovariectomized females given estradiol performed fewer errors than did ovariectomized females without estradiol on tasks requiring a place strategy, whereas for tasks requiring a response strategy, females without estradiol performed better than did females given estradiol.138–140 In addition, intrahippocampal infusions of estradiol improved place learning in females rats 141 and intrastriatal infusions of estradiol impaired response learning.141,142 Testosterone has also been shown to mediate the bias between place and response strategies in a dose-dependent manner. In particular, a low dose of testosterone biased castrated male rats toward using a response strategy and a high dose biased them toward using a place strategy. 143 When the task required the use of a particular strategy, male rats given a low dose of testosterone made fewer errors on a response task, whereas a high dose of testosterone resulted in fewer errors on a place task. 144 Another study also found that male rats administered a low dose of testosterone performed worse compared with intact males when they were unable to use a response strategy on a working memory version of the RAM. 53 Interestingly, an intermediate dose provided no benefit for performance of either strategy, 144 which may explain why rats receiving this dose perform poorly on spatial tasks where they could make use of either strategy (Fig. 2). 143 Specifically, whereas a low dose of testosterone may activate the striatum and a high dose may activate the hippocampus, the intermediate dose is ineffective in activating either brain region, possibly due to a conflict between memory systems.15,145 It could be that the striatum is more sensitive to testosterone than the hippocampus. 144 There are more androgen receptors in the striatum than in the hippocampus of male rats, 146 and low levels of circulating testosterone have been shown to downregulate androgen receptor expression in the hippocampus of male rats. 123 Failure to consider the interplay between testosterone dose and spatial strategy helps to explain why broad reviews of human research have concluded that testosterone has no effect on spatial memory.79,80
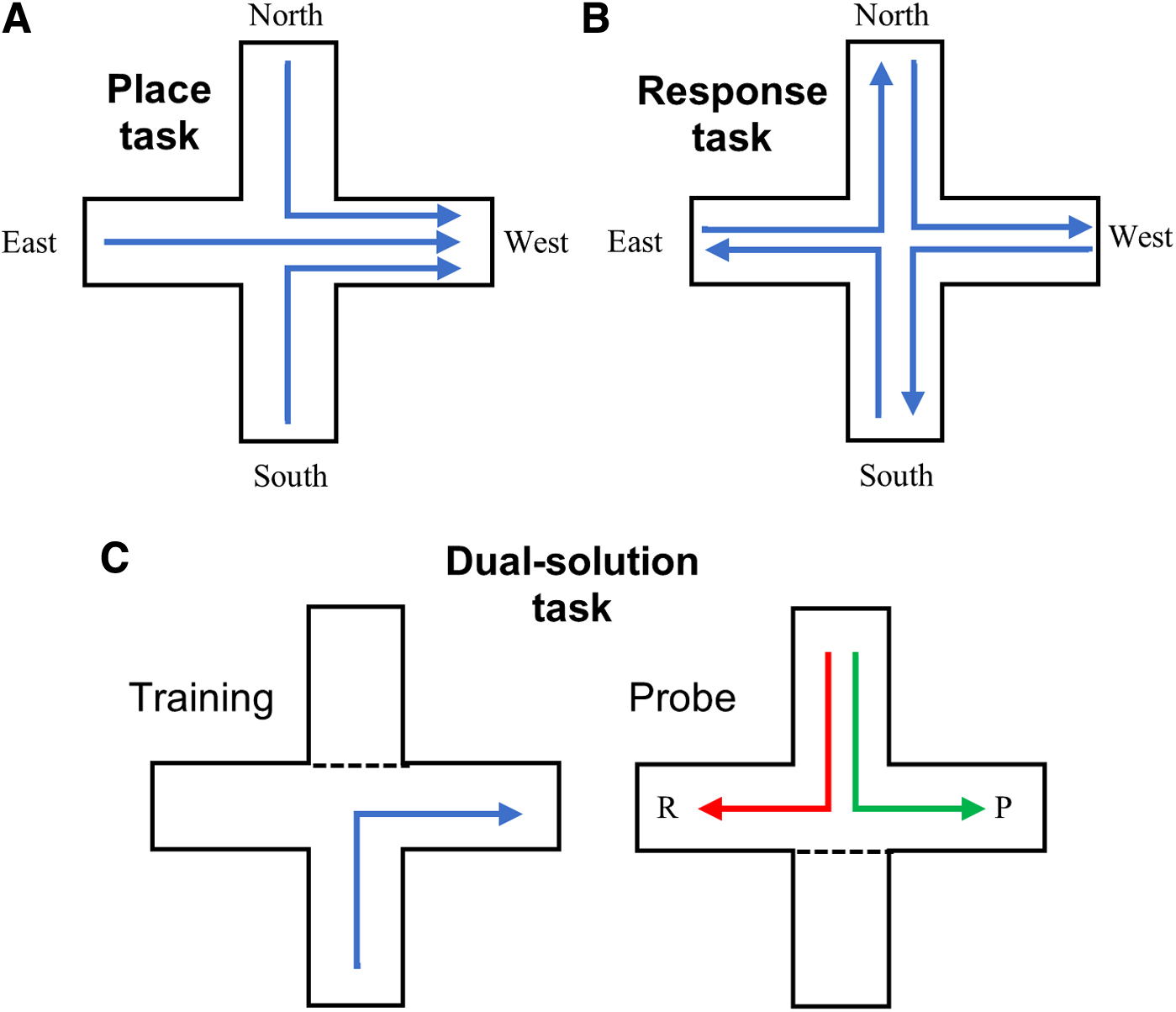
Examples of plus-maze protocols used to test place and response memory. Arrows indicate correct choices for each version of the task.

Graphical model of the relationship between circulating testosterone levels and spatial working memory. Based on experiments with male rats,120,143,144,203 low and high serum testosterone leads to good performance on spatial tasks that require the use of a response strategy and a place strategy, respectively. Intermediate levels of testosterone are associated with poor use of both place and response strategies.
Human Research with Older Males
In men, testosterone levels begin to decline by age 30 at a rate of about 2% per year.147,148 This decline leads to hypogonadism (serum testosterone levels consistently <325 ng/dL), which occurs in about 20% of men 60–69 years old and in about 50% of men >80 years old. 148 Andropause refers to this age-associated hypogonadism and related symptoms in males 149 and is thought to be due to impaired feedforward and feedback functions of the hypothalamic–pituitary–gonadal (HPG) axis. 150 Specifically, age is associated with a disruption of either the production of testosterone within the testes or impaired hypothalamic secretion of gonadotropin-releasing hormone (GnRH). Impaired GnRH regulation results in inadequate release of luteinizing hormone (LH) from the anterior pituitary, which in turn reduces the conversion of cholesterol to testosterone by Leydig cells of the testes. 151 There is also evidence that a lack of negative feedback inhibition from testosterone by the testes in aged males can lead to unusually high levels of circulating LH, 152 which may be a causative factor in the progression of Alzheimer's disease (AD).153,154 In addition, longer CAG repeat sequences in the androgen receptor were shown to predispose males to hypogonadism in older age. 155 Thus, andropause involves dysfunction of the entire HPG axis.
Andropause can lead to many negative clinical implications: decreased muscle tone and strength, increased fat mass, decreased bone mineral density contributing to osteoporosis, decreased insulin sensitivity, impaired glucose tolerance, and negatively affected mood.156–158 Given that this decline in testosterone parallels the age-related decline in cognitive ability, there has been much speculation regarding a causal link between the two.5,32 Both human and rodent studies indicate that memory tasks involving the hippocampus and the prefrontal cortex are most severely affected during both normal aging and by pathological aging.3,159–161 Spatial ability, in particular, is negatively affected, with older males exhibiting impaired learning and memory on a variety of spatial tasks.3,79,162
Cross-sectional studies have shown a positive correlation between testosterone levels and cognitive ability in older men.163,164 In a longitudinal study involving 407 nondemented older men followed for 10 years, higher testosterone levels were associated with better spatial ability and a reduced rate of decline in visual memory. 165 Testosterone-supplementation studies with healthy older males have produced mixed results, with some results showing improved spatial ability in males receiving testosterone,166,167 and others showing no effect of testosterone on cognition.168,169 One month of testosterone supplementation was shown to significantly improve working memory in healthy older men (61–75 years old) to a level comparable with that of younger men (23–34 years old), 170 but this experiment did not specifically test spatial memory. Similar to studies with younger males, androgen deprivation therapy for prostate cancer has been shown to impair memory in older males.171,172 Discrepancies among studies may be due to differences in the duration of hormone exposure and the specific doses used. Cherrier et al. 173 found that a moderate dose of testosterone (100 mg) injected weekly for 6 weeks caused healthy older males to perform better on spatial memory tasks than those injected with either low or high testosterone doses (50 or 300 mg). Thus, moderate testosterone supplementation may be best at improving memory in older males.
A nonquantitative review of four clinical trials (conducted 2003–2006) involving testosterone treatment in older males concluded that testosterone supplementation had moderate positive effects on selective cognitive domains, including spatial abilities. 6 However, a more recent review of clinical trials concluded that testosterone treatment had minimal effect on cognitive abilities and the authors suggested that prolonged, rather than acute, treatments with testosterone may be needed to see positive effects. 174 Two recent meta-analyses of clinical trials involving testosterone supplementation came to opposite conclusions. Buskbjerg et al. 175 synthesized 23 randomized clinical trials, mainly involving older subjects, and concluded that testosterone supplementation had no effect on cognitive functioning among eugonadal males. In contrast, Tan et al. 7 synthesized 14 randomized clinical trials and concluded that testosterone supplementation resulted in improved executive function and improvement on a cognitive composite score. Key differences between the two meta-analyses were that: (1) Tan et al. used only trials involving healthy older males, whereas Buskbjerg et al. included studies with a wider age range and included studies on males with dementia at the time of treatment; (2) the Tan et al. study only included clinical trials with a documented increase in circulating testosterone due to treatment, whereas the Buskbjerg et al. study did not. Thus, testosterone supplementation may be most beneficial when given for prolonged periods to elderly males before the onset of any cognitive impairments and it should result in a quantifiable increase in circulating testosterone. Somewhat surprisingly, Tan et al. did not find that baseline testosterone was predictive of post-treatment cognitive abilities; rather, final circulating testosterone levels after treatment was the key predictor. 7 It should also be added that both of the meta-analyses included studies that assessed a broad range of cognitive functions, although some form of visuospatial memory was commonly included in testing batteries.
Aging has been shown to lead to increased use of a striatum-dependent response strategy rather than a hippocampus-dependent place strategy in males and females,21,176,177 which suggests that neurodegeneration within the hippocampus, specifically, plays an important role in age-related memory loss. A study in which both sexes navigated through a virtual 8-arm maze showed that 84% of children (8 years old), 46% of young adults (19–40 years old), and 39% of older adults (53–85 years old) employed a place strategy to solve the task. 177 Another study showed a similar decrease in the use of a place strategy by both sexes with increasing age. 178 Some studies have shown that this increased preference for a response strategy among aged individuals is due to an impaired ability to employ a place strategy.176,179,180 For example, when participants were asked to explain changes in the position of an object in a virtual room, younger subjects performed better than older subjects when distal cues were changed (place learning), whereas there was no age effect when only the relative position of the observer was changed (response learning). 181 One study examined the neurological correlates of the age-related shift in spatial strategy using fMRI and demonstrated that younger adults showed more activation of the hippocampus than did older adults when solving a spatial navigation task and, as expected, older adults showed greater activation of the striatum. 182 In combination with the studies demonstrating age-related hippocampal atrophy,2,159,183 these results suggest that hippocampal deficits force elderly individuals to rely more heavily on the striatum for spatial memory.
AD shows similar incidence between the sexes up to age 80, when women show greater incidence than men. 184 In addition, men show earlier mortality than women after developing AD.184,185 These results suggest that sex steroids may play a role in the progression of AD. Deficits in spatial memory are symptomatic of AD, and decreased spatial working memory seems to be particularly useful in diagnosis. 186 Two cross-sectional studies have shown that males with AD have significantly lower serum testosterone levels than nonsymptomatic control subjects.187,188 A long-term longitudinal study (subjects followed 4–37 years) demonstrated that baseline testosterone levels were lower in males who developed AD later in life. 189 Another large longitudinal study followed 4069 males over an average of 10.5 years and found that low baseline testosterone levels were a significant predictor for developing dementia later in life. 190 A shorter (1-year) study also found the low baseline testosterone levels predicted increased risk of developing AD. 191
Studies involving males with AD or mild cognitive impairment showed that testosterone supplementation improved cognitive abilities in general and improved spatial memory in particular.166,192 In contrast, one study found no effect of testosterone on males with mild cognitive impairment, 193 and another found that testosterone improved “quality of life” in AD patients but had no effect on cognition. 194 Some of these varied results may be due whether or not individuals are carriers for the APOE4 allele of the APOE gene, which codes for the lipid-transport protein apolipoprotein E. In healthy older males without the APOE4 allele, circulating free testosterone levels were positively correlated with cognitive function, whereas in males with the APOE4 allele, testosterone level was negatively correlated with cognitive function. 195 One short-term study found that androgen deprivation increased levels of anxiety and depression as well as circulating amyloid-β levels. 196 In a study that encompassed 9272 males with prostate cancer, it was found that androgen deprivation therapy led to a significant increase in the likelihood of developing AD. 197 The absolute increase was only 4.4%, but this involved a doubling of risk from 3.5% in patients not receiving therapy to 7.9% among those receiving androgen deprivation therapy (hazard ratio = 2.17). 197 Two even larger studies (23,651 and 154,089 subjects) also concluded that androgen deprivation therapy increases risk of developing AD and other forms of dementia, although with smaller hazard ratios (1.34 and 1.14, respectively).198,199 A recent meta-analysis of 13 studies concluded that androgen deprivation therapy, regardless of method, leads to a 21% increased risk of dementia in patients with prostate cancer. 200 Thus, hypogonadism is associated with increased risk of AD, and androgen therapies hold promise for treating and possibly preventing the development of AD and other forms of dementia.
Rodent Research with Older Males
As with humans, male rats show an age-related decline in testosterone201–203 and aging impairs spatial memory. In a mouse model (SAMP8) that shows early memory decline (8 months old), circulating testosterone levels were found to drop significantly more with aging relative to wild-type mice. 204 Older male rats performed worse than young rats on the Barnes maze.205,206 Similarly, increased age has been shown to impair both long-term spatial memory and spatial working memory on a variety of tasks, including the MWM,124,160,207 water-escape RAM, 208 and the traditional RAM.203,207 Aging also impaired long-term spatial memory on the object location memory task. 203
Castration alone has been shown to have no effect on spatial memory in aged male rats tested using a variety of tasks,203,206 but this is not unexpected as testosterone levels are already quite low in aged males before castration. 203 Testosterone supplementation, however, improves spatial memory in aged males. Using a water-escape RAM, Bimonte-Nelson et al. 208 found that testosterone supplementation given to intact aged males improved spatial working memory but not long-term spatial memory. An experiment with a traditional food-reward RAM showed that testosterone injections caused dose-dependent improvement in spatial working memory among aged male rats, with both high and low doses of testosterone improving memory in a manner similar to that demonstrated for young males. 203 In contrast, an intermediate dose of testosterone was optimal for improving long-term spatial memory on an object-location memory task, suggesting differences in the optimal dose for working and long-term memory. 203 A study with mice showed that testosterone injections improved spatial working memory (RAM) similarly in both aged (18 months old) and young (29 days old) mice. 209 Thus, growing evidence indicates that testosterone supplementation can improve multiple forms of spatial memory in aged male rodents.
As with humans, aging influences spatial strategy use among rodents. Young male rats are significantly more likely than aged males to use a place strategy rather than a response strategy on a dual-solution task.110,206 A study using various water maze tasks showed that aged male rats performed significantly worse than young males on a place task and that there was no effect of age on performance on a response task. 210 These results parallel the general findings with aged human subjects. An interesting experiment divided aged male rats into those with impaired or unimpaired spatial memory based on preliminary performance on the MWM, and then aged and young subjects were tested on place and response tasks. 211 Memory-impaired aged males made fewer errors on a response task and young males made fewer errors on a place task. Interestingly, memory-unimpaired aged males performed well on both place and response tasks, and the authors suggest that this may be due to reduced competition between the hippocampus and striatum within this group. 211 As all the subjects in this experiment were males, it is possible that the memory-impaired and memory-unimpaired groups had differences in circulating testosterone levels, but this was not measured. The potential role of testosterone in regulating the age-related transition from a place strategy to a response strategy should be explored further.
Some studies have tested the therapeutic benefits of testosterone in rodent models of AD. An androgen receptor antagonist (hydroxyflutamide) given to a transgenic male mouse model of AD (NSE-apoE) caused significant memory deficits on the MWM, suggesting a neuroprotective role for androgens. 212 This conclusion was supported by a follow-up experiment in which the NSE-apoE mice also lacking androgen receptors were found to have impaired memory relative to mice with normal androgen receptor levels. 42 In a triple-transgenic mouse model of AD, castration increased the rate of amyloid-β accumulation in the brain and treatment with the androgen-receptor agonist dihydrotestosterone (DHT) reversed this effect. 213 Jia et al. 122 gave rats intrahippocampal injections of amyloid-β to model AD and found a negative parabolic relationship between long-term spatial memory (MWM) and testosterone dose. In a follow-up experiment using the same rat model, testosterone injections improved long-term spatial memory and flutamide (androgen-receptor antagonist) blocked this effect. 214 However, one study found that neither testosterone nor DHT supplementation had an effect on amyloid-β levels in aged male rats, 208 suggesting that the memory-restoring effects of testosterone may not involve direct changes in amyloid-β. Overall, however, rodent experiments indicated memory-enhancing benefits of testosterone treatments for AD.
Metabolites of Testosterone: DHT and Estradiol
As alluded to previously, testosterone either binds directly to androgen receptors or is converted to DHT or estradiol to bind androgen or estrogen receptors, respectively. The enzyme 5α-reductase catalyzes the conversion of testosterone into DHT, 215 which has been shown to have about twice the binding affinity to androgen receptors and a fivefold slower dissociation constant from these receptors compared with testosterone. 216 The classic androgen receptor is a 110-kDa ligand-activated protein, which binds to DNA to act as a transcription factor. 217 Although poorly characterized, there are also membrane-bound androgen receptors that can have more rapid (nongenomic) effects on cellular functions. 218 Alternatively, testosterone can be converted into estradiol in the brain through P450 aromatase.219,220 Estradiol acts through two known intracellular receptors (ERα and ERβ) to have genomic effects as well as through a G-protein-coupled estrogen receptor to have more rapid nongenomic effects. 221 Estrogens have been shown to regulate spatial memory in women, and these effects have been extensively reviewed.32,222,223 Although steroidal mechanisms of action in the male brain have not been fully characterized, both estrogen receptors and androgen receptors have been localized to the hippocampus and striatum among male rodents.123,224–227 Thus, testosterone may influence male spatial memory through an androgen-dependent pathway, an estrogen-dependent pathway, or some combination of the two.
In support of a role for an androgen-dependent pathway, male rats with nonfunctional androgen receptors exhibited poorer spatial memory, as assessed by the MWM. 228 Systemic DHT administration also restored long-term spatial memory among castrated male mice. 229 Furthermore, DHT, but not estradiol, has been shown to enhance synaptic density and neurogenesis within the hippocampus of adult male rats,230,231 and intrahippocampal injections of DHT improved long-term spatial memory among male rats. 232 In contrast, a study with aged male rats found no effect of DHT on spatial memory (neither working nor long-term), suggesting that testosterone improves memory through an estrogen-dependent pathway. 208
In support of an estrogen-dependent pathway, two studies found that estradiol implants improved spatial working memory in castrated young male rats.119,233 However, estradiol implants given to aged males had little effect on working memory but improved memory when there was a 2- to 3-h delay between choices, 233 suggesting a role for estrogens in long-term spatial memory among aged males. Similarly, intrahippocampal injections of estradiol enhanced long-term spatial memory among young male rats, 234 and young males given estradiol injections showed increased hippocampal spine density within 30–60 min of injection and improved performance on an object location memory task. 132 Alejandre-Gomez et al. 235 used letrozole, an aromatase inhibitor, to block the conversion of testosterone into estradiol in adult male rats. They found that rats given letrozole exhibited fewer errors on a spatial working memory task (plus maze) than did either untreated controls or rats administered a letrozole vehicle control. This suggests that estradiol may actually interfere with spatial working memory in some cases, potentially providing insights regarding the dose-dependent effects of testosterone. However, letrozole injections impaired performance by male mice on the water maze, 129 leading to the interesting possibility that androgens facilitate working memory and estrogens facilitate long-term memory. In summary, there is evidence that testosterone acts through both estrogen- and androgen-dependent pathways to improve spatial memory in males, and the relative importance of these pathways likely depends on the form of spatial memory and may change with age.
Lakshman et al. 236 compared the kinetics of testosterone conversion with its major metabolites in older and younger human males given weekly doses of testosterone for 5 months. Interestingly, they found no effects of age on DHT levels, but estradiol levels were elevated in the older males relative to the younger males. The authors suggest that this effect may be due to higher percent body fat in older males, as aromatase is highly expressed in adipose tissue. 236 It is possible, therefore, that the differences between older and younger men in the effects of testosterone on memory may be due to increased conversion of testosterone to estradiol in older men. However, Laskshman et al. measured hormone levels peripherally, and the kinetics of steroidogenesis in the brain, particularly in humans, is much more poorly understood than the kinetics in blood. Indeed, there is evidence from rodent studies that the rate of estradiol production in the hippocampus depends on the level of neural activity. 237
Neuroplasticity and Neuroprotection
Numerous forms of neuroplasticity are modified by testosterone, and these may be the cellular substrate for memory formation. For example, castrated male mice exhibited a reduction in dendritic spine density among CA1 cells of the hippocampus, and testosterone treatment reversed this effect.129,238 Similar results have been obtained using male rats,132,230,239 and this effect was replicated with DHT,230,240 suggesting an androgen-dependent pathway. Surprisingly, DHT also enhanced CA1 spine density among testicular-feminization mutant rats that have nonfunctional androgen receptors, 241 which indicates that DHT is either acting on membrane-bound androgen receptors to increase spine density or is acting through another molecular pathway involving one of its metabolites. 242 Both castration and letrozole (aromatase blocker) were shown to reduce a variety of molecular markers of synaptic plasticity in male mice, 129 suggesting that estrogens also enhance neuroplasticity in males. The therapeutic value of testosterone was supported in an experiment in which testosterone treatment given to an AD rat model caused both an increase in CA1 spine density and improved spatial memory. 214 Within the CA3 region of the hippocampus, castration caused a similar reduction synaptic spine density but, interestingly, this was accompanied by an increase mossy fiber density in the hippocampus of male rats, suggesting a neurological trade-off. 239 There is also considerable evidence that testosterone upregulates adult neurogenesis within the hippocampus, specifically increasing the survival of new neurons, while having minimal effect on cell proliferation.231,243 Finally, electrophysiology studies with rat hippocampus slices (CA1 region) have produced somewhat divergent results: castration impaired excitatory postsynaptic field potentials in temporal slices, 244 but testosterone administration impaired long-term potentiation in the dorsal hippocampus. 245 In summary, there is evidence that testosterone influences many forms of neuroplasticity through both estrogen- and androgen-dependent pathways.
Although there are many unexplored downstream effects of activated testosterone receptors, 246 brain-derived neurotrophic factor (BDNF) has become a molecule of particular interest because of its importance in regulating neuroplasticity.247,248 A number of studies have shown that testosterone upregulates the expression of BDNF in the spinal cord and the peripheral nervous system.249,250 Hill et al. 251 showed that surges of testosterone early in development among male mice increased BDNF expression in both the PFC and the striatum, but not the hippocampus. A number of studies have also shown that castration decreases total BDNF protein levels in the hippocampus of adult male rats.252–254 Similarly, castration caused a decrease in BDNF mRNA (as well as other mRNA markers of neuroplasticity) in the hippocampus of male rats. 253 More refined analyses have shown that castration increased BDNF levels specifically within the mossy fibers extending from the dentate gyrus to the CA3 layer of the hippocampus,255,256 but castration decreased BDNF production within CA1 layer of the hippocampus, again indicating a trade-off. 238 Zhang et al. 144 recently demonstrated that a low dose of testosterone increased BDNF levels in the striatum of male rats and that a high dose increased BDNF levels in the hippocampus. The same experiment demonstrated that the low dose improved response learning and the high dose improved place learning, implying a functional significance for these changes. 144 Testosterone implants increased both BDNF and CA1 spine density in SAMP8 mice, and flutamide blocked this effect, suggesting that testosterone enhances BDNF through an androgen-dependent pathway. 257 Although many details need to be clarified, considerable evidence indicates that testosterone enhances BDNF levels, which may in turn improve memory through increased neuroplasticity.
Importantly, the ability of testosterone to upregulate BDNF seems to be lost in aged males. Two studies have shown that testosterone supplementation has no effect on BDNF levels in the hippocampus or striatum of aged male rats.203,208 This suggests that the molecular effects of testosterone may differ between the young and aged male brain, and perhaps a disconnect between testosterone and BDNF is partially responsible for age-related memory loss.
In addition to enhancing various forms of neuroplasticity, the neuroprotective effects of testosterone are well established. Testosterone prevents the apoptotic effects induced by oxidative stress and a variety of neurotoxins.258,259 This effect seems to be dose dependent, as relatively high doses of testosterone induce apoptosis.260–262 Although some of the neuroprotective properties of testosterone may occur through aromatization to estradiol, 263 evidence suggests that neuroprotection occurs mainly through androgen-dependent pathways.262,264 For example, DHT implants caused a significant reduction in neuronal loss in the hippocampus after kainate lesions among castrated male rats. 265 Similarly, DHT decreased apoptosis among cultured rat hippocampal neurons. 259 The neuroprotective properties of androgens involve activation of the mitogen-activated protein kinase (MAPK) pathway.261,266 Specifically, Gatson et al. 261 found that DHT induced phosphorylation of MAPK through a genomic pathway and activated the phosphatide 3-kinase pathway through membrane-bound receptors. In addition, testosterone implants given to castrated male rats were found to upregulate MAPK1 (one protein in the pathway) within the hippocampus. 267 Androgens also rapidly phosphorylate cyclic-AMP response element binding protein (CREB) in cultured hippocampal neurons, which can have neuroprotective effects, but this effect was found to involve the protein kinase C pathway rather than the MAPK pathway. 268
Testosterone application also reduces β-amyloid production in neurons269,270 and blocks formation of neurofibrillary tangles, 271 both distinctive symptoms of AD. There is evidence that this neuroprotective effect against Aβ neurotoxicity occurs through both the MAPK pathway and by upregulating the enzyme neprilysin. 272 Caspase proteins have a well-established role in inducing apoptosis,273,274 and increased caspase activity has been identified as having a causal role in AD pathology. 275 There is a tentative link between testosterone and caspase-3 activity in the hippocampus: castration increased the number of caspase-3 positive cells within the dentate gyrus region of the hippocampus, while simultaneously downregulating both BDNF and its primary receptor (tyrosine receptor kinase B). 252 In summary, multiple intracellular pathways are activated by androgens that lead to enhanced neuroprotection.
One way that testosterone may act as a neuroprotectant is by blocking the neurotoxic effects of the hormonal stress response, and interactions between the HPG axis and the hypothalamic–pituitary–adrenal axes have been extensively documented using rodent experiments.276,277 Chronic stress, acting through glucocorticoids (i.e., cortisol and corticosterone), impairs spatial memory and hippocampal plasticity.278–280 For example, castration resulted in a reduction in hippocampal neurons among rats that were also exposed to an acute stressor (restraint and water immersion). 281 Furthermore, intrahippocampal injections of an androgen-receptor antagonist caused an increase in anxiety-like behavior in male rats, 242 suggesting that testosterone reduces the stress response by acting directly on the brain. Interestingly, higher testosterone doses were found to reduce anxiety on a burying task, whereas a lower dose did not, 282 which indicates that the dose-dependent effects of testosterone may involve suppression of the stress response. Direct experimental tests of the relative impact of stress and testosterone on spatial memory would be a useful line of research.
Conclusion
Sex differences on some spatial tasks piqued interest in studying the activational effects of sex steroids on spatial memory. Despite variable results from both clinical trials and animal experiments, current research indicates an activational role for testosterone in sustaining spatial memory in young males and potentially restoring memory in aging males. Discrepancies among studies are likely due to uncontrolled variables. Dose of testosterone used for androgen therapies is of particular importance, as there is considerable evidence that the relationship between circulating testosterone levels and memory is nonlinear. Human studies indicate an inverted-U relationship, 93 and some research with rats suggests an even more complex relationship (Fig. 2).120,203 Testosterone dose has been shown to influence which strategy male rats use to solve spatial tasks, with low doses improving response learning and high doses improving place learning.143,144 Considering that aging leads to increased use of a striatum-dependent response strategy rather than a hippocampus-dependent place strategy in both humans and rodents,177,211 it is reasonable to hypothesize that impaired androgen activity within the hippocampus, specifically, is responsible for some cognitive deficits associated with aging. Human studies have demonstrated the most consistent beneficial effects of testosterone when given to hypogonadal rather than eugonadal males, likely due to low physiological doses being more beneficial than supraphysiological doses.86,93 This may explain why the cognitive benefits of testosterone therapies seem to be more consistent for older males, as they are more likely to be hypogonadal. 7 Similarly, prolonged testosterone deprivation has been shown to consistently increase risk of developing dementia. 200 This demonstrates a need to carefully consider the costs and benefits of prolonged androgen deprivation for the treatment of prostate cancer. Rodent research suggests that testosterone has a stronger effect on spatial working memory than long-term memory, 118 but this distinction has not been systematically tested in human research. Another important variable is whether testosterone is influencing cognition through an androgen- or estrogen-dependent pathway. Current evidence indicates that both estrogens and androgens can enhance spatial memory in males, but increased aromatization of testosterone to estradiol in aged males is an intriguing effect that should be researched further. 236 The molecular mechanisms by which testosterone influences spatial memory are likely diverse, but current evidence points to enhancement of neuroplasticity through elevated BDNF levels and neuroprotection by blocking apoptotic pathways.
Particularly important is a potential role for testosterone therapies in preventing or treating AD, as supported by both clinical and animal research.7,200,214 In 2021, an estimated 6.2 million people aged ≥65 years in the United States were living with AD, which accounts for one in nine Americans in this age range. 283 Globally, an estimated 50 million people have dementia. 284 As this number is expected to increase with aging global populations, there is urgent need for new therapies, and androgens provide a promising direction for further research.
Footnotes
Authors' Contributions
Conception and design by M.D.S.; drafting and approval of the article by all authors.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
No funding was received for this article.
