Abstract
Neuroactive testosterone metabolites synthesized by enzymes 5α-reductase and 3α/3β-hydroxysteroid oxidoreductase are critical physiological modulators of the central nervous system (CNS) function. Indeed, as discussed in this review, they exert a variety of effects on myelination, brain maturation, neurotransmission, reproductive behavior, and the stress responses. The expression and synthesis of these enzymes as well as the levels of testosterone derivatives exhibit specific localized regional and sex differences in the CNS and are influenced under various physiological and neuropathological conditions. In addition, neuroactive metabolites of testosterone exert a variety of neuroprotective effects in several neuropathological animal models. Therefore, neuroactive testosterone metabolites might represent an interesting potential therapeutic option to treat some neurological disorders.
Introduction
Testosterone (T) and its various metabolites play an essential role in physiology and pathophysiology of the central nervous system (CNS). For example, it is well established that androgens play a seminal role in brain development and neurogenesis and in sexual differentiation. 1 Furthermore, androgens exerted a key role in CNS myelination and remyelination 2 and provide neuroprotection effects on the hypothalamus in conditions of metabolic syndrome. 3 Androgens are also thought to increase synaptic density in the gyrus dentatus 4 and improve regional brain perfusion 5 and enhances survival of new hippocampal neurons. 6 T also mediates rewarding properties in the nucleus accumbens 7 and its metabolite 3α-diol facilitates female sexual motivation when infused in the nucleus accumbens. 8 T metabolites were thought to serve as an anxiolytic and cognitive-enhancing modulator in the hippocampus 9 and T and its metabolites may modulate seizure susceptibility. 10
T appears to regulate Alzheimer-like neuropathology in male 3xTg-AD mice 11 and most recently it was demonstrated that blockade of 5α-reductase (5α-R) impedes cognitive performance, modifies dendritic morphology, and upregulates Tau phosphorylation in the hippocampus of male 3xTg-AD mice. 12 In this review, we provide a discussion of the biology, biochemistry, and regional distribution of key enzymes involved in T metabolism in the nervous system, such as 5α-Rs and 3α/3β-hydroxysteroid oxidoreductases (3α/3β-HSORs) (Fig. 1) and discuss the critical role of such biochemical pathways and potential clinical implication on brain function in health and disease.
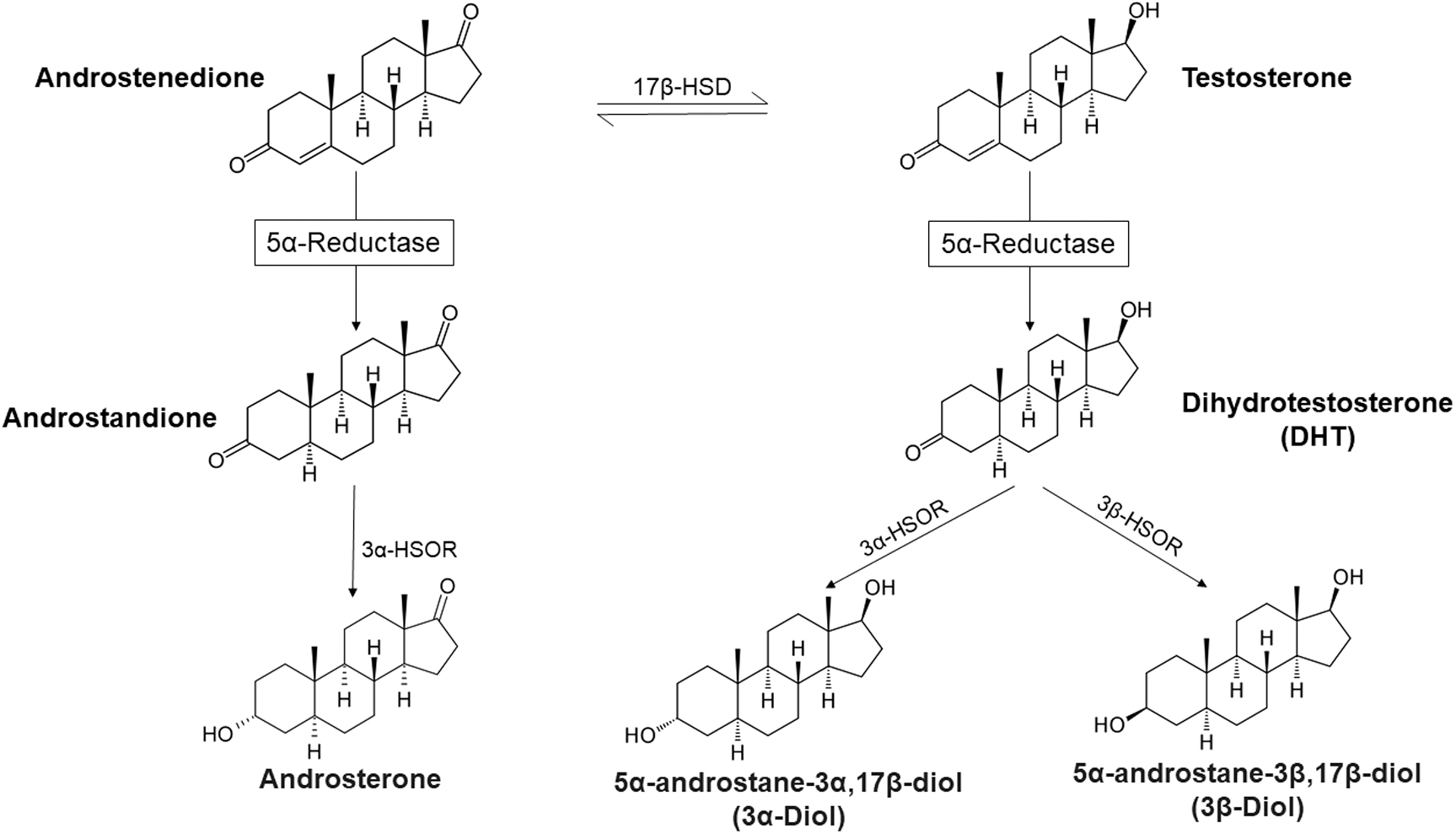
Androgen metabolism by the enzymes 5α-reductase and 3α/3β-hydroxysteroid oxidoreductase.
The Biology of 5α-R
5α-Rs and 3α/3β-HSORs are families of enzymes expressed in the CNS and in the periphery, and are responsible for transformation of androgens, progestins, glucocorticoids, and mineralocorticoids into neuroactive signaling steroid molecules.13–28 5α-Rs catalyze the formation of 5α derivatives of neuroactive steroids with second-order kinetics representing the rate-limiting step19,26,29–31 (Fig. 1). One critical pathway in the CNS physiological function is the transformation of T or Δ 4 androstenedione into neuroactive androgen derivatives by 5α-Rs concomitant with 3α/3β-HSORs.19,26,31–34 As is discussed in detail in this review, these neuroactive androgens metabolites modulate a host of physiological processes including myelination,2,35 neurotransmission,36–39 response to stress,40–43 and serve as anti-inflammatory agents.44–47 The expression of 5α-Rs and 3α/3β- HSORs exhibit sex-specific differences and regional differences within the CNS and are modified by various physiological modulators as well as pathological conditions.
Traish 25 reviewed the distribution of 5α-R types 1, 2, and 3 and reported on localization of these enzymes in various tissues including liver, prostate, epididymis, seminal vesicle, and spinal cord, as well as the white and gray matter. 5α-R type 2 is expressed in glial cells of the white matter and in oligodendrocytes, dorsal horn, and ventral horn in the gray matter and motoneuronal cells of the anterior horn. Type 1 activity predominates in other peripheral tissues. 5α-R type 1 activity is localized in many regions of the CNS encompassing mixed glia, type 1 astrocytes, oligodendrocytes, and neurons, and is localized in white matter of the spinal cord. Type 1 activity is detected in many other tissues such as adrenal glands, liver, kidney, skin, and scalp. 5α-R type 3 is distributed in many tissues including the hippocampus, cerebellum retina, heart, skin, kidney, prostate, liver, skeletal, muscle, myometrium, and pancreas.
Expression and regional distribution of 5α-Rs in the CNS
5α-Rs catalyze a key biochemical reaction in the regulation of development and physiology of male sexual differentiation and metabolism.26,27,48 The family of 5α-Rs is currently thought to encompass five different enzymes, based on differences in the primary DNA sequences. The members of this family include three well-characterized 5α-R isotypes (types 1, 2, and 3; SRD5A1, SRD5A2, SRD5A3). In addition, two trans-2,3 enoyl-CoA reductases (TECR and TECR-like) have been identified and partially characterized.18,27,28,49–51
5α-R type 1 activity is found in high levels in various regions of the CNS, more specifically those rich in white matter, such as midbrain, corpus callosum, anterior commissure, optic chiasm, pons, and spinal cord.14,18,52 Similarly, 5α-R type 1 activity is elevated in samples of purified myelin preparations isolated from rat brain13,14,53 and is detected in oligodendrocytes, the myelin forming cells of the CNS, and in neurons.54–56 Interestingly, 5α-R type 1 activity was also detected in the myelinating cells of the peripheral nervous system (PNS) (i.e., Schwann cells).57–60 Further observations indicate the presence of 5α-R type 1 in microglia determined by reverse transcriptase-polymerase chain reaction 61 and in astrocytes by functional enzymatic activity.54,55 Interestingly, as reported in cell cultures, type 2 astrocytes, which probably correspond to fibrous astrocytes in vivo, but not type 1 astrocytes, which probably correspond to protoplasmatic astrocytes, express 5α-R type 1, assessed by functional enzymatic activity.54,55 However, the 5α-R activity detected in type 1 astrocytes is enhanced by the concomitant presence of neurons. 62 5α-R type 1 mRNA has also been detected in cortical, hippocampal, and olfactory bulb glutamatergic neurons and in some output neurons of the amygdala and thalamus. 63
5α-R type 1 enzyme genetic deficiency has not been reported to date and has not been related to any human disorder. In contrast, congenital 5α-R type 2 deficiencies are well characterized with adverse clinical consequences such as significant changes in sexual differentiation.48,50,64–68 5α-R type 3 deficiencies due to mutations have been described and were thought to be associated with visual disturbances and various mental retardation.69–72 Recently, TECR mutations were also thought to be associated with mental retardation 73 even though the exact functions of these enzymes (as well as TECRL) remain poorly characterized, and information on its relevance in the process of steroidogenesis is currently not available.
5α-R type 2 is also widely detected in most key regions of the adult rat brain using immunofluorescent staining. 74 Based on detection by immunoreactivity, 5α-R type 2 is present in higher concentrations in the spinal cord (i.e., dorsal and ventral horn of the gray matter) and particularly in oligodendrocytes. 52 It is also present in motoneuronal cells of the anterior horn, with greater levels in the L1 to L3 tract of the lumbar spinal cord. 75 It is worth noting that not only differentiated CNS cells but also stem cells originating from the mouse striatum express 5α-R. 76 We should also emphasize that 5α-R type 3 expression and tissue distribution encompass the hippocampus and cerebellum of human brain.72,77 5α-R type 3 is thought to be more responsible in N-linked protein glycosylation through a mechanism that involves polyprenol reductase 69 and may not be critical in steroid metabolism. 78 SRD5A3, a gene encoding an enzyme responsible for dolichol synthesis, and when defective, leading to impaired N-glycan biosynthesis in the endoplasmic reticulum. 72 Only few reports suggesting that it is expressed in liver and prostate were reported. Nikolaou et al. 79 suggested that the role of 5α-R type 3 is not entirely clear. This isoform is found in a variety of human tissues, including liver, skin, kidney, skeletal muscle, pancreas, and testis, and it is the mostly highly expressed in human abdominal adipose. A number of studies have tried to elucidate the role of this enzyme in steroid hormone metabolism, but data have been conflicting. 79 A role for this enzyme in the nervous system remains unclear, at present.
Biochemistry of 5α-Rs
5α-Rs catalyze the chemical reduction of the double bond in the A ring at the 4,5 positions in C19 and C21 steroids. 5α-Rs transfer a hydride from nicotine amide adenine dinucleotide phosphate (NADPH) to the 5α position of the steroid nucleus to produce its 5α-reduced derivative.29,80 The precursors for 5α-Rs include T, progesterone (PROG), deoxycorticosterone, corticosterone, cortisol, and aldosterone. The derivatives produced from these reactions result in formation of 5α-dihydro-derivatives such as 5α-dihydrotestosterone (5α-DHT), 5α-dihydroprogesterone (5α-DHP), 5α-dihydrodeoxycorticosterone, 5α-dihydrocorticosterone, 5α-dihydrocortisol, and 5α-dihydroxyaldosterone. More importantly, 5α-dihydrocortisol and 5α-dihydroxyaldosterone may serve as a potential active mineralocorticoid. The activity of 5α-Rs determines the rate limiting step in the synthesis of 3α, 5α steroid derivatives.26,27 Thus, 5α-Rs reactions generate products that serve as substrates for 3α-HSOR) and 3β-HSOR) enzymes. 31 3α-HSOR transforms DHT to 3α-diol, DHP to tetrahydroprogesterone (THP; also known as allopregnanolone), and DHDOC to tetrahydrodeoxycorticosterone (THDOC). 3β-HSOR transforms DHT to 3β-diol and DHP to isoallopregnanolone. We have previously addressed the role of 5α-Rs and 3α/3β-HSOR in the metabolism of other steroids, like for instance PROG.26,31 Therefore, in this review we focus our attention on T metabolites.
Molecular mechanisms of 5α-R catalysis and inhibition
5α-Rs are NADPH-dependent enzymes that catalyze the reduction of 3-keto, Δ 4 , steroids. The binding of T and NADPH to the enzyme results in the formation of an enzyme–substrate complex that undergoes activation into the transition state complex (Fig. 2). The pyridine group from NADPH initiates a nucleophilic attack that results in isomerization of the three keto group into the enol group and reduction of the C-4 double bond. In a subsequent reaction, and acid–base catalysis step results in enol to keto formation and abstraction of the pair of the electrons, resulting in dissociation of NADP+ from the intermediate and dissociation of DHT from the complex and eventually the enzyme active site. As reported by Bull et al. 29 at standard conditions of pH 7.20 and 37°C, the catalytic constants for conversion of T to DHT are Km = 24.6 ± 0.7 nM for T; Km = ∼1 μM for NADPH; kcat = 0.075 ± 0.006/S. From the kinetic constants reported by Bull et al., 29 the second-order rate constant for reduction of T to DHT equivalent to kcat/Km = (3.0 ± 0.3) × 106 /(M S). These catalytic parameters point to an efficient enzymatic reaction with high affinity for the substrate (T). Finasteride: (17β–(N-tert-butylcarbamoyl)-4-aza-5α-androst-1-en-3-one (MK-906) is a potent inhibitor of 5α-R types 1 and 2. Bull et al. 29 proposed a novel mechanism related to catalysis, in which finasteride acts through a mechanism-based inhibitor (suicide substrate). 81 The enzyme binds finasteride and reduces finasteride to dihydrofinasteride through NADPH as a cosubstrate and the NADP-dihydrofinasteride adduct remains very tightly bound to the enzyme and does not dissociate fast, keeping the enzyme locked up in an inactive state. This NADP-dihydrofinasteride adduct serves as a bisubstrate analog inhibitor, and ranks among the most potent noncovalently bound enzyme inhibitors known for this enzyme. The mechanism-based inhibition of 5α-R by finasteride proceeds through closely related enol(ate) intermediates, but then diverges significantly from the natural substrate because finasteride redirects the carbanion center to a position where it escapes the proton transfer that normally completes the reduction. The adduct phosphor-adenosine diphosphate-dihydro-finasteride remains bound to the enzyme and does not dissociate easily, resulting in a dead end (suicide substrate) and is lethal to enzyme function. Although the enzyme is bound to phosphor-adenosine diphosphate-dihydro-finasteride, it is not available to perform its anticipated cellular activity of transforming T to DHT. Over a longer period of time, the adduct is hydrolyzed either through enzyme catalyzed or nonenzymatic reaction releasing the dihydrofinasteride. However, because of these pharmacokinetic parameters, the finasteride adduct may remain attached to the enzyme for days, weeks, or perhaps months, thus rendering the enzyme incapable of its regular biological function. For this reason, finasteride is considered a suicide substrate for the enzyme. Bull et al. 29 suggested that the kinetic data demonstrate that the rate constants for the forward and backward reactions of the enzyme–inhibitor complex show that the (steady-state) dissociation constant of finasteride is Ki* = koff/kon ≤ 3 × 10−13 M.

Mechanism of action of 5α-R. The action of the enzyme 5α-R on testosterone and other substrates is divided into three main steps: the first step (1) shows a nucleophilic attack of pyridine group from NADPH to testosterone leading to the binding to substrate with enzyme. The second step (2) consists in isomerization of the three keto group into the enole group and an abstraction of the pair of the electrons resulting in dissociation of NADP+ from the intermediate; the third step (3) shows the dissociation of 5α-DHT from the enzyme–substrate complex and eventually the enzyme active site. 5α-DHT, 5α-dihydrotestosterone; 5α-R, 5α-reductase; NADPH, nicotine amide adenine dinucleotide phosphate.
It is important to emphasize that human 5α-R type 2 isozyme has apparent Ki values for finasteride and dutasteride in the nanomolar range (69 and 7 nM, respectively), suggesting that both inhibitors significantly reduce DHT biosynthesis. 82 The apparent Ki values for type of 5α-R type 1 isozyme for finasteride and dutasteride (360 and 6 nM, respectively), suggest that finasteride is not highly selective for 5α-R type 1 and may not significantly inhibit DHT. 82 This is not the case in the animal model studies in which finasteride and dutasteride inhibit both 5α-R types 1 and 2. Finally, it should be pointed out that 5α-R type 2 isozyme has apparent K values in the nM range for steroid substrates, whereas the type 1 isozyme has apparent K values in the μM range. Rat type 2 enzyme is maximally active over a narrow acidic pH range, whereas the rat type 1 enzyme expressed in these cells has a broad neutral basic pH optimum.83,84
The Biology of 3α/3β-HSORs
3α- and 3β-HSORs are members of the aldo-keto reductase superfamily that includes aldehyde reductase, aldoreductase, and dihydrodiol dehydrogenase.85–91 This group of enzymes catalyzes conversion between aldehydes or ketones and alcohols, using NAD(H) or NADPH(H) as cofactors. In humans, multiple cDNAs encode proteins related to 3α-HSOR,92–96 and four isozymes of 3α-HSOR were identified.90,96 In contrast, in rats only one type of enzyme was identified in both brain and liver.97–100 The Δ 4 -3-ketosteroids, which are reduced by 5α-R, are metabolized consecutively to 3α,5α-tetrahydrosteroids by 3α- HSOR.90,101 The enzyme activity of 3α- HSOR is also widely distributed in the CNS (e.g., in rat cerebral cortex and the cerebellum, with lower expression in the latter structure) 102 and is especially high in the olfactory bulb.103,104 Agis-Balboa et al. 63 reported that 5α-R type 1 and 3α- HSOR colocalize in cortical, hippocampal, and olfactory bulb glutamatergic principal neurons and in some output neurons of the amygdala and thalamus of mice. Neither 5α-R type 1 nor 3α- HSOR mRNAs are expressed in S100β- or glial fibrillary acidic protein-positive glial cells. 5α-R type 1 and 3α-HSOR were not detected in cortical and hippocampal GABAergic interneurons. However, 5α-R type 1 and 3α- HSOR are significantly expressed in principal GABAergic output neurons, such as striatal medium spiny, reticular thalamic nucleus, and cerebellar Purkinje neurons. A similar distribution and cellular location of neurosteroidogenic enzymes were observed in rat brain. These findings demonstrate that THP and THDOC, which may be synthesized in principal output neurons, modulate gamma-aminobutyric acid (GABA) function at GABA-A receptors, through an autocrine mechanism or through a paracrine mechanism or by allosteric interaction with GABA-A receptor intracellular sites through lateral membrane diffusion.
In the spinal cord, 3α-HSOR presence, as detected by immunostaining, is mainly distributed in both white and gray matters, even if the greatest density is found in sensory regions of the dorsal horn. 52 The 3α-HSOR activity is little in neurons and high in astrocytes, especially in type 1 cells54,55 and oligodendrocytes,52,105 although this activity has not been observed in myelin membranes of the CNS.13,14
Physiological Effects of Testosterone Metabolites
The metabolites of T as a result of the action of the enzymatic complex 5α-R and 3α- or 3β-HSOR exert critical physiological action in the CNS, since such metabolites serve as chemical messengers (ligands) for a host of neuronal and glial receptors that are not known to be directly modulated by T. Indeed, although the 5α-reduced metabolite of T (i.e., DHT) is still able to bind to the androgen receptor (AR), showing higher affinity for this receptor than its precursor T, 106 the further 3α,5α-reduced metabolites interact with different receptor molecules. Indeed, 3α-diol and 3β-diol are known to act as agonists of GABA-A receptors, as well as ERβ.107–112
Activation of AR by DHT initiates a cascade of molecular mechanisms that regulate neuronal, glial, and synaptic differentiation and in the coordination of the appearance of typical male features in specific brain regions.113,114 Interestingly, DHT regulates synaptic density and transmission in male, but not in female rodent hippocampal cultures. In this experimental model, finasteride, blocking DHT synthesis, diminishes dendritic spine maturation and long-term potentiation. 115 In addition, this T metabolite affects plasticity of CA1 hippocampal pyramidal neurons in adolescent male rats, 116 it is necessary for inducing long-term depression of synaptic transmission, 117 and plays a role in the regulation of spine plasticity in circuits related with motivated behaviors. 118 DHT mediates also the age-dependent change in GnRH neuron firing in adult prenatally androgenized female mice 119 and facilitates sexual behavior in gonadectomized female rats primed with estradiol. 120 Finally, activation of AR-dependent signaling pathway promotes neurogenesis, 121 increasing the neural progenitor cells in the developing mouse cortex. 122
3α-diol, by the interaction with GABA-A receptors, regulates behavior.123,124 3β-diol seems to modulate the activity exerted by 3α-diol on the stress response mediated by HPA axis. 107 In particular, 3β-diol is effective in attenuating corticosterone and ACTH increases in response to restraint stress.125,126 These effects are inhibited by administration of tamoxifen (i.e., an estrogen receptor [ER] antagonist) suggesting an involvement of ERβ.125–127
5α-reduced derivatives of T exert physiological effects in the PNS. In mature male animals, gonadectomy significantly reduces gene expression of myelin proteins such as P0 and PMP22 in the sciatic nerve, whereas subsequent treatment with 5α-DHT or 3α-diol restores P0 mRNA to precastration levels.128,129 Only 3α-diol treatment is effective on PMP22 levels. 128 These results demonstrate that 5α-DHT and 3α-diol increase P0 and PMP22 mRNA levels, respectively, in cultures of rat Schwann cells, in agreement with previous observations.129,130 These results seem to suggest that P0 and PMP22 are regulated by the action of AR and GABA-A receptor, respectively. Indeed, inhibition of AR action by flutamide (i.e., an antagonist of AR) or with bicuculline (i.e., an antagonist of GABA-A receptor) reduces the synthesis of these two myelin proteins.128,129 The regulation of P0 expression by classical steroid receptors is further inferred from findings that putative androgen responsive elements have been shown in the promoter region of P0 gene. 129
Levels of Testosterone Metabolites Under Physiological Conditions
Data based on analyses of metabolites of T using liquid chromatography-tandem mass spectrometry demonstrated that the levels of reduced T metabolites are significantly different in the nervous system, plasma, and cerebrospinal fluid (CSF), especially, in the CNS versus PNS and in male versus female rat. 131
The sex differences in the levels of 5α-reduced T metabolites may be related to sex dimorphism of the steroidogenic enzymes responsible for their synthesis. Indeed, as reported in the brain of green anole lizards, the gene expression of 5α-R type 2 is higher in females than in males. 132 Sex differences in the brain expression of 5α-R have also been reported in rats. Thus, the gene expression of this enzyme is significantly higher in the cerebellum of males, whereas that of 3α-HSOR is significantly higher in the cerebellum of proestrus females. It is important to highlight, that this sex dimorphism is brain area specific; indeed, does not occur in the cerebral cortex. 102
Interestingly, the levels of 5α-reduced metabolites of T in the nervous system are influenced by their circulating levels. Thus, gonadectomy affects the levels of these molecules in the CNS and the PNS. 133 As reported in rodents, the changes induced by gonadectomy are different depending on the nervous regions and sex considered, as well as by the duration of gonadal hormone deprivation. 133 For instance, 3α-diol levels present in the cerebellum, cerebral cortex, and spinal cord are decreased, by long-term gonadectomy, only in male animals, whereas in the sciatic nerve, they occurred only in female animals. 133 On the contrary, short-term gonadectomy does not show any effects in the cerebellum, cerebral cortex, and sciatic nerve of both sexes, but it induces an increase of 3α-diol levels in spinal cord of female animals. 133 A sex-specific effect of long-term gonadectomy on 3α-HSOR expression has also been recently reported. 102 Similar alterations in the levels of T metabolites have been recently reported in quail brain of control and gonadectomized animals. 134 In summary, these findings demonstrated that the levels of 5α-reduced metabolites of T detected in the nervous system, even if modulated by the sex steroid hormone milieu, point to a specific pattern depending on sex and brain region.
Levels of Testosterone Metabolites Under Pathological Conditions
The levels of T-reduced metabolites in the nervous system are also altered by psychiatric and neurodegenerative diseases (Fig. 3). In addition, many neurodegenerative and psychiatric disorders exhibit a sex difference with regard to incidence and/or manifestations of the pathology. Interestingly, in some of these pathologies, a sex difference is also reported in the magnitude of 5α-reduced metabolites of T in the nervous system.

Levels of testosterone metabolites in experimental models of neuropathological conditions. The arrows indicate the increase or decrease of testosterone metabolites, DHT, and 3α-diol. ALS, amyotrophic lateral sclerosis; CMT, Charcot-Marie-Tooth; CSF, cerebrospinal fluid; DHT, dihydrotestosterone; EAE, experimental autoimmune encephalomyelitis; MS, multiple sclerosis; PD, Parkinson's disease; TBI, traumatic brain injury.
For instance, 3α-diol levels are decreased in association with increased depression and anxiety symptoms in the plasma of anorexic and overweight/obese women. 135 As shown in prefrontal cortex Brodmann's area of depressed patients, 5α-R type 1 enzyme is also downregulated. 136 Changes in expression of 5α-R type 1 as well as of types 2 and 3 were reported in the prefrontal cortex of adolescent male rats after ethanol treatment. 137 Polymorphism in 5α-R type 1 and 3α-HSOR has also been related to elevated risk of alcohol dependence. 138
Changes in T metabolism have also been reported in autism. At postnatal day 5, a decrease of DHT levels was reported in male, but not in female cerebellum of the reeler mouse (an experimental model of autism). 139 Interestingly, a significant association between single nucleotide polymorphisms rs523349 (Leu89Val) located on the gene encoding 5α-R type 2 and autism has also been reported. 140 Moreover, knockout mice for 5α-R type 2 enzyme exhibited reduced dominance-related behaviors, as well as deficits of novelty-seeking and risk-taking responses. 141
In experimental model of traumatic brain injury (TBI), levels of DHT are significantly altered in the brain.142–144 In particular, in agreement with the reported sex-dimorphic incidence and outcome of TBI, 145 sex differences in T derivative levels were also observed in animal models. Indeed, a decrease of DHT levels was observed after TBI in the male, but not in the female brain. 143
Changes in T metabolism were also observed in neurodegenerative disorders. For instance, DHT levels are decreased in the CSF of male and female patients affected by amyotrophic lateral sclerosis. 146 In Parkinson's disease (PD) patients, the expression of the 5α-R type 1 enzyme is downregulated in the substantia nigra, whereas that of 3α-HSOR type 3 is upregulated in the caudate nucleus. 147 The levels of the 5α-reduced metabolites of T are modified in PD rodent models. For instance, injection with 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP; i.e., an experimental model of PD) increases the levels of DHT in the brain of male mice. 148
Observations performed in patients affected by multiple sclerosis (MS) show altered levels of T metabolites. In the CSF of relapsing-remitting MS male adult patients, a decrease in the levels of DHT associated with an increase in those of 3α-diol was reported. 149 In agreement, in the hippocampus and cerebral cortex of the mouse cuprizone demyelination model, a decrease of the 5α-R expression was reported. 150 In addition, observations performed in experimental autoimmune encephalomyelitis (EAE) rat MS model showed alterations of T-reduced metabolite levels in CNS.151,152 Indeed, a decrease in the levels of DHT and 3α-diol was reported in the spinal cord at the chronic phase of the disease. 152
In agreement with the higher incidence of MS in females153–155 and the existence of sex differences in MS symptomatology (i.e., course of the disease, age of onset, and pathological alterations), 156 different levels of T-reduced metabolites have been detected in male and female EAE animals.
Both at the acute phase (i.e., 14 days postimmunization) 151 and chronic phase of the disease (i.e., 40 days postimmunization), 152 DHT and 3α-diol levels are decreased, in the spinal cord of male, but not of female animals. In the cerebellum, only a significant decrease of 3α-diol levels was reported in males.151,152 In the male cerebral cortex, there is a decrease in the levels of DHT and 3α-diol at the acute phase of the disease, 151 however, at the chronic phase, only the levels of DHT remain decreased. 152 The levels of 5α-reduced metabolites of T were unaffected in these nervous areas of female animals.151,152
Diabetic encephalopathy (i.e., the damage in the CNS induced by diabetes mellitus) also induces changes in the levels of T-reduced metabolites.157–162 As reported in an experimental model (i.e., rats raised diabetic by streptozotocin injection), the specific alterations in the levels of these T metabolites depend on the duration of pathology. For instance, the levels of 3α-diol are decreased in the spinal cord of male rats after long- but not after short-term diabetes.159,160 In agreement with sex dimorphic features of diabetic encephalopathy, 163 changes in the nervous levels of T-reduced metabolites depending on the sex were also reported. For instance, in diabetic animals the levels of DHT and 3α-diol are decreased in the cerebral cortex, cerebellum, and spinal cord of males but not of females. 159 Diabetes mellitus also induces damage in the PNS (i.e., diabetic peripheral neuropathy). Also, in this case the specific alterations in the levels of 3α-diol depend on the duration of pathology. Indeed, the levels of this T metabolite are decreased in the sciatic nerve of male rats after long- but not after short-term diabetes.159,164 Sex-specific changes in the levels of T metabolites also occur in diabetic peripheral neuropathy as well as other peripheral neuropathies. For instance, in the sciatic nerve of male but not in female rats, DHT levels are decreased after short-term diabetes. 164 After longer time of diabetes, a decrease not only of DHT but also of 3α-diol levels is observed in the male rat sciatic nerve. 159 In another model of peripheral neuropathy, such as the crush injury of the sciatic nerve, a decrease in the expression of enzyme 5α-R has been observed in the distal portion of the injured nerve in male rats. 165 In addition, in an animal model Charcot-Marie-Tooth disease type 1A (i.e., an inherited peripheral neuropathy), the levels of 3α-diol are decreased in the peripheral nerves of male but not of female animals. 166 Altogether these observations clearly show that CNS and PNS damage is also associated with altered nervous levels of 5α-reduced metabolites of T. On this basis, the neuroprotective actions of these molecules have been explored in several experimental models of psychiatric and neurodegenerative disorders.
Neuroprotective Effects of Testosterone Metabolites
Some of the 5α-reduced metabolites of T have been reported to exert effects in mood and anxiety disorders. 167 In particular, sex-specific features are an important aspect of these actions. For instance, 3β-diol normalized the HPA axis responses to interleukin-1β only in adult male prenatally stressed rats. 168
T-reduced metabolites also exert protective effects in a variety of in vitro and in vivo models of neurodegeneration (Fig. 4). Indeed, DHT is neuroprotective in experimental model of PD,169,170 in EAE models,171–174 as well as in an experimental model of TBI. 175 This T metabolite is also able to protect hippocampal neurons from damages, like for instance kainic acid injection, 176 apoptosis,177,178 and serum deprivation. 179 In addition, it is also a protective agent for motoneurons 180 and also after spinal cord injury. 181 On stroke, both protective and deleterious effects have been reported. 182

Neuroprotective effects of testosterone metabolites. The main protective effects of DHT and 3α-diol are shown. 5α-DHT exerts neuroprotective effect on
Also the further metabolite of DHT, 3α-diol, exerts neuroprotective effects. In SH-SY5Y neuronal cells and in primary cortical neurons, this metabolite, by both GABA-A receptor-dependent and independent mechanisms, inhibits the phosphorylation of extracellular signal-regulated kinase induced by amyloid β peptide 1–42183 as well as modulates mitogen-activated protein kinase phosphatase 3/dual specificity phosphatase 6. 184 Some of the protective effects exerted by the metabolites of T show sex-specific features. In cultured hippocampal neurons, DHT shows protective effects for apoptosis induced by glutamate in males, but not in females. 185
5α-reduced metabolites of T are also protective in the PNS. For example, it has been reported that DHT induces a faster regeneration and functional recovery of injured nerves.180,186–189 In the streptozotocin (STZ) experimental diabetic rat model, this metabolite stimulates the activity of Na+, K+-ATPase in the sciatic nerve and counteracts the impairment of nerve conduction velocity, thermal sensitivity, and skin innervation density. 190 Also 3α-diol is effective; indeed, it reduces morphological alterations in the sciatic nerve 191 and improves nerve conduction velocity, thermal sensitivity, and skin innervation density observed in STZ experimental diabetic rat model. 190
Pain is an important component of the peripheral neuropathy. Interestingly, 5α-R and 3α-HSOR are expressed in pain information processing centers of the CNS, such as the dorsal root ganglia and the dorsal horn of the spinal cord.192,193 In agreement, T-reduced metabolites decrease neuropathic pain associated with diabetes mellitus. In particular, DHT and 3α-diol treatments are effective on alterations of mechanical nociceptive threshold and tactile allodynia induced by diabetes, respectively. 160 3α-diol is also able to exert beneficial effects on painful symptom- occurring in paclitaxel-induced peripheral neuropathy. 194
Myelin compartment is another target of the neuroprotective effects of T-reduced metabolites. Indeed, in cerebellar organotypic cultures, DHT protects against acute demyelination, 195 and in the STZ experimental model, DHT stimulates the expression of P0 in the sciatic nerve. 190 In addition, 3α-diol reduces the accumulation of saturated fatty acids in the myelin of sciatic nerve reported in STZ experimental diabetic rat model. 191 Neuroinflammation, a common aspect among neurodegenerative and psychiatric diseases,196–201 may be regulated by T metabolites. In the EAE model, DHT reduces proinflammatory IFN-γ expression and gliosis in the spinal cord 174 and increases the expression of anti-inflammatory IL-10 by autoantigen-specific T lymphocytes. 173 In addition, this T metabolite inhibits microglia inflammatory responses by suppressing tool-like receptor 4/nuclear factor-kappa B signaling.45,46 Altogether these observations indicate that the treatment with T-reduced metabolites exert a variety of protective actions in the nervous system.
An alternative to the systemic treatment with these molecules, it is to enhance their endogenous synthesis. One option could be represented by the pharmacological activation of steroidogenesis with ligands of translocator protein of 18kDa (TSPO) (i.e., a molecule involved in the transfer of cholesterol into mitochondria). 202 Indeed, some TSPO ligands stimulating steroidogenesis and, therefore, the synthesis of T-reduced metabolites,203–205 exert neuroprotective actions in animal models. Examples of that are provided by the etifoxine 206 and XBD173207 in EAE mice, midazolam 208 and YL-IPA08 in rat models of post-traumatic stress disorder (PTSD), 209 chronic stress-related depression 210 and anxiety disorders, 211 PK11195, in a rat ex vivo glaucoma model, 212 and Ro5–4864 and AC-5216 in diabetic rats.213,214
Activation of liver X receptors (LXRs), it has also been demonstrated to increase the levels of T-reduced metabolites in the nervous system as well as to exert neuroprotective effects in animal models of cerebral ischemia, 215 MS, AD, and PD.216,217 In particular, the treatment with an LXR ligand, such as GW3965, increases the levels of 3α-diol in the spinal cord, cerebral cortex, and sciatic nerve of STZ experimental model.213,218 Interestingly, at least in the sciatic nerve, the protective effect of LXR ligands is associated with an increase in the expression of 5α-R, among other steroidogenic molecules. 218 The modulation of 5α-R activity has been proposed as a therapeutic treatment for disorders of the nervous system. In observational studies, dutasteride in adult males was shown to be associated with reduced self-reported sedative effects of a moderate dose of alcohol in a laboratory setting and reduced alcohol self-administration for 1–2 weeks after a single 4-mg dose in the natural environment. 219 Moreover, a clinical study performed in women with premenstrual dysphoric disorder showed that this 5α-R inhibitor reduces irritability, sadness, anxiety, food cravings, and bloating. 220 We should point out that limited studies in the animal model, as well as in human, have investigated the effects of dutasteride in the CNS. Nevertheless, it was postulated that dutasteride elicits a neuroprotective activity, in the animal model of PD. Dutasteride but not finasteride protected dopamine (DA) neurons against MPTP-induced toxicity. MPTP mice treated with dutasteride (5 mg/kg and 12.5 mg/kg) showed higher striatal DA and metabolites (i.e., 3,4-dihydroxyphenylacetic acid and homovanillic acid) with a decrease of metabolites/DA ratios compared with saline-treated MPTP mice.221,222 Dutasteride has been proposed for therapy of human glioblastoma.219,223 In addition, another 5α-R inhibitor, such as the finasteride, is protective in ischemic brain injury induced in aged rats. 221 Finally, in adolescent male rats, this 5α-R inhibitor inhibits dopaminergic system and, therefore, has been proposed for neuropsychiatric disorders associated with hyperactivity of this neurotransmitter system. 222
In contrast, it is critical to emphasize that endocrine and neuropsychiatric side effects have been reported in a subset of male patients treated with 5α-R inhibitors for androgenetic alopecia (i.e., erectile and ejaculatory dysfunctions, loss of libido, depression, anxiety, suicidal thoughts, and sleep problems). It is significant that such effects may persist despite treatment discontinuation,26,224–227 leading to new clinical disorder referred to as “postfinasteride syndrome.” Interestingly, changes in the methylation pattern of the gene encoding for 5α-R type 2 and in the levels of the T-reduced metabolites were detected in the CSF of patients with this newly defined syndrome228,229 associated with alterations in the gut microbiota population 230 as well as major depressive disorders, erectile dysfunction, and peripheral neuropathy.227,228
Conclusions
Significant literature indicating that 5α-Rs and 3α/3β-HSORs are widely expressed and distributed in various regions of the nervous system suggests a critical function of these enzymes in the effects exerted by testosterone and its metabolites. Indeed, 5α-Rs catalyze a key rate-limiting step in formation of 5α-reduced neuroactive steroids and, therefore, is critical in maintaining the physiological function of the nervous system. Indeed, T and its metabolites are implicated in processes of neurogenesis, myelination and remyelination, neuroprotection, and attenuation of neuroinflammation. Furthermore, these metabolites play an important role in the reduction of stress responses and modulation of behavior. Significant changes in the concentrations of testosterone and its various metabolites have been noted in the nervous system under various pathological conditions, such as neurodegenerative and psychiatric disorders. Altogether, observations reported in this review suggest that a better understanding of the biochemical actions of testosterone and its metabolites in the nervous system will have important clinical implications in understanding of and in treatment of neurological disorders.
Footnotes
Author Contributions
R.C.M. and A.M.T. carried out data collection and analyses. L.C. and S.D. prepared the figures. R.C.M., L.C., S.D., and A.M.T. were involved in writing, review, and editing the article.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
No funding was received for this article.
