Abstract
Testosterone (T) is the most abundant biologically active hormone in women. It has a direct effect at the androgen receptor in every major organ system. Local aromatization of T is a major source of bioavailable estradiol. Adequate amounts of bioavailable T are essential for optimal health, immune function, and disease prevention. More than 80% of bioavailable T in women is from the local intracrine production of T from the adrenal precursor steroids androstenedione and dehydroepiandrosterone (sulfate). Serum T levels reflect <20% of the total androgen pool in women, which limits its usefulness in diagnosing or treating androgen deficiency. The gradual decline of androgens associated with aging is responsible for many of the adverse signs and symptoms of aging, including mental and physical deterioration. Decades of evidence support the safety and efficacy of T therapy in women. We have found that subcutaneous T implant therapy relieves symptoms of hormone deficiency in women with and without breast cancer, improves their quality of life, and maintains overall health and well-being. T does not increase and may lower the risk of breast cancer. The combination of T with an aromatase inhibitor prevents the conversion of androgens to estrogens, limiting their stimulatory effect in estrogen-sensitive diseases, including breast cancer. Adequate doses of T therapy should provide adequate levels of bioavailable T in the target organs—determined by clinical response (benefits) versus adverse side effects (risks). Pharmacological dosing of T implants in women is safe and necessary for physiological effect.
Introduction
Androgens are critical for immune function and overall health in both sexes. Androgens decline with age, adversely affecting mental and physical health. Replacing (declining) androgens with the consistent and continuous release of testosterone (T) from the subcutaneous implant significantly improves women's health, sexuality, and quality of life (QoL).1–6
Many controversies surround the use of T therapy in women. Recent pharmaceutical sponsored studies have focused on topical T formulations and recent narratives have argued against the use of T pellet implants. 7 However, T implant therapy has been (safely) used in female patients since 1937 in doses of 50–400 mg without excessive androgenic effects.3–6,8,9 In addition, significantly higher doses (500–1800 mg) have been safely used to treat breast cancer patients.3,10
Understanding the physiology of androgens in women is the foundation for understanding the extent of T's clinical effects; the rationale behind ‘T dosing’ and ‘serum levels on therapy’; the significance of local aromatase production and its role in estrogen-sensitive diseases; and the therapeutic effects of T alone (no estrogen). In a series of studies, we provide decades of experience and evidence supporting the safety and efficacy of T implant therapy in women, including breast cancer patients.1,11–25 The therapeutic potential of T combined with an aromatase inhibitor (AI) is discussed and supported by clinical evidence.
Androgens in Women
T is the most abundant biologically active hormone in women. It is produced in the ovaries, adrenal gland, and locally at the cellular level in target organs from androgen precursors. The major portion of serum T is bound to albumin and sex hormone-binding globulin. T has a direct effect at the androgen receptor (AR). It is metabolized through the enzyme 5α-reductase to the more potent androgen, dihydrotestosterone. T is also aromatized to estradiol (E2) in the ovaries and locally in all peripheral tissues, thereby having a secondary effect through the estrogen receptor (ER). Many physicians are not aware that serum T levels are markedly (10- to more than 15-fold) higher than E2 levels throughout the female lifespan, barring pregnancy (Fig. 1). 26

Serum T levels compared with E2 levels in women. E2, estradiol.
The major source of androgenic activity in both pre- and postmenopausal women is the local intracrine production of T from the adrenal precursor steroids dehydroepiandrosterone-sulfate (DHEAS), dehydroepiandrosterone (DHEA), and androstenedione (Fig. 2).

Steroid synthesis pathway (permission for use granted by ZRT Laboratory, Beaverton, OR).
Androstenedione, the direct precursor to T, is found in more than fivefold higher concentrations than serum T in women.27,28 Circulating DHEA and DHEAS are present in 20- to 1000-fold higher concentrations than T. Interestingly, men and women produce similar amounts of adrenal androgens. The preandrogens contribute >75–80% of biologically active T to the AR in premenopausal women and near 100% in postmenopausal women—versus 50% in men.29–31
Serum levels of T are not a valid marker of tissue exposure in women, reflecting <20% of the total androgen activity. Accordingly, serum T levels would not be expected to correlate with androgen deficiency symptoms or clinical conditions caused by androgen deficiency. 30 This concept is extremely important to comprehend. Serum T levels should not be relied on to diagnose T deficiency or manage T dosing in women.5–9
It is well recognized that T has a profound effect on lean muscle mass, bone density, and confidence as well as sex drive and performance in both sexes. It is beyond the scope of this article to provide a detailed review of the physiological effects of androgens. Excellent reviews have been previously published on the clinical significance of T in women. 32
It is important to recognize that there are active ARs located in every major organ system throughout the body.33–38 Adequate amounts of (local) bioavailable T at the AR are critical for overall health, immune function, and preventing inflammation, as well as cardiovascular, neurological, gastrointestinal, pulmonary, endocrine, breast, and genitourinary health (Supplementary Table S1).32–42 Thus, clinical indications for T therapy include many signs and symptoms caused by T deficiency (Table 1).1,43
Signs and Symptoms of Aging Related to Androgen Deficiency
T is the direct precursor for E2 in every major organ system, including the ovaries. The enzyme aromatase (P450) catalyzes the biosynthesis of estrogens from androgens. Tissue-specific aromatase and other steroidogenic enzymes are located in every organ system—supplying estrogens locally to the ER from T and androgen precursors (Fig. 2).44–46 The main source of estrogen in postmenopausal women is the local conversion (paracrine/intracrine) of T to biologically active E2. Unlike adipose tissue, which can contribute to the circulating pool of estrogens, E2 from local aromatization would not be measurable in serum.31,46,47 Therefore, similar to serum T levels, serum levels of E2 should be interpreted with caution and taken into context with clinical evaluation.
Obesity, medications, xenoestrogens, certain disease states (endometriosis and fibroids) and cancers (breast and endometrial) upregulate aromatase resulting in excess intracellular (local) E2 production. Increased aromatase activity and excess estrogen relative to T can stimulate breast and uterine tissues and remains an underappreciated cofactor in the etiology of endometriosis, dysfunctional uterine bleeding, uterine fibroids, as well as breast and uterine cancers.44–53
A marked decline of T and the adrenal precursor steroids (DHEA and androstenedione) occurs in women between their late 20s and 50s, which has a significant impact on their health, sexuality, and QoL.32,54,55 Symptoms of androgen deficiency can occur before menopause and are not related to estrogen levels.1,3–6,43 In fact, many premenopausal women have symptoms of estrogen excess in addition to androgen deficiency.1,3–6 As evidenced earlier, serum T testing would not be reliable in diagnosing androgen deficiency. The gradual decline of (all) androgens is associated with signs, symptoms, and disease states associated with aging (Supplementary Table S1). In addition, T deficiency has a negative impact on cardiovascular and neurological health in women.32,56
T implant therapy in women and treatment controversies
T implant therapy has been safely used in women since 19373–6,8,9—which may be a reason there are a limited number of recent controlled studies, which can be overwhelmingly costly—yet necessary for Food and Drug Administration (FDA) approval. Well-designed randomized controlled trials (RCTs) are valuable in assessing the effectiveness of drug treatments. However, by nature they obscure individual variability seen in clinical practice. Evidence-based personalized medicine promotes integrating the best research evidence, the physician's clinical expertise, and the patient's values, preferences, and expectations. Decades of clinical experience and evidence (original data) support the long-term safety and efficacy of T therapy in women.1,11–25
Since 2005, >2500 women have been treated with subcutaneous T implants, including >230 breast cancer patients. In 2020, 3331 T pellet insertions were performed in 1022 female patients, 105 of whom had a diagnosis of breast cancer. All patient's initial severity of symptoms and subsequent hormone-related changes are evaluated using the validated Health-Related QoL questionnaire, Menopause Rating Scale (MRS) (Fig. 3). Additional symptom-specific validated questionnaires are administered if clinically applicable. T implants are not regulated.

Health-related QoL, MRS-validated questionnaire: indications for T therapy in pre- and postmenopausal women. MRS, Menopause Rating Scale; QoL, quality of life.
All patients are required to sign a consent informing them of the “off-label” use, benefits, and risks of T implants in women (Supplementary Data S1). Patients are informed of (expected) elevated serum T levels on therapy and the stimulation of red blood cell production. Patients are monitored for secondary polycythemia.
In the United States, androgens are listed as a “class X” teratogen and premenopausal patients are instructed that they “must use birth control” (listed on the consent) with the “warning” that T could masculinize a female fetus.
However, there are no reports in the literature evidencing that T delivered by subcutaneous implants (i.e., a daily dose/release rate of 1–3 mg per day)2,11 has any adverse effect on a fetus—even in animal studies. 57 Although 400–800 mg of danazol (a potent synthetic androgen) results in clitoromegaly and fused labia in some female fetuses, 58 animal studies have shown that virilization of a female fetus requires >30 times normal maternal levels or >50–500 times human T doses.57,59 In addition, the placenta buffers hormone diffusion and is a significant source of aromatase, which metabolizes maternal T to E2.
In our clinic, a 38-year-old nulliparous patient treated with T pellets (two insertions) became pregnant after a decade of not having menstrual cycles and not using birth control for 4 years; she subsequently delivered a healthy baby girl. The author is aware of several other unexpected pregnancies with similar results (RL Glaser, personal communications). Nevertheless, contraception should be mandated.
Original data
Glaser and Dimitrakakis have shown that T implant therapy successfully relieves symptoms of hormone deficiency improving QoL in both pre- and postmenopausal patients. 1 Three hundred female patients were evaluated; 36% were premenopausal and 64% were postmenopausal. Pre- and postmenopausal women had similar baseline T levels.
As expected, there was no relationship between baseline T levels and presenting symptoms (other than sexual complaints) or response to therapy. Premenopausal women reported a higher incidence of psychological complaints (depressive mood, anxiety, and irritability), which may be contributed to by higher—or fluctuating—levels of estrogen relative to declining T levels.3,60 Postmenopausal women reported more hot flashes, vaginal dryness, and urological symptoms, which may be contributed to by lower levels of estrogen. T alone (no estrogen) delivered subcutaneously resulted in statistically significant improvement (p < 0.0001) in all 11 MRS symptom categories (Fig. 3).
Both groups demonstrated similar improvement in the total score, as well as psychological, somatic, and urogenital subscale scores. Higher doses of T correlated with greater improvement in symptoms. There were no adverse drug events reported in 285 patients treated for >1 year (mean 28.1 ± 10.4 months). These benefits are consistently seen in clinical practice (Supplementary Table S1).
T therapy in premenopausal women has not been evaluated in controlled trials. However, clinical studies have reported positive effects in conditions caused by excess estrogen, including hypermenorrhea, uterine fibroids, endometriosis, premenstrual tension, dysmenorrhea, breast pain, and chronic mastitis.3,5,6 We have also published a case report on T implant therapy during breastfeeding—a 100-mg subcutaneous T pellet was effective in relieving maternal symptoms of depression, anxiety, fatigue, decreased libido, memory problems, and pain—T was not measurably increased in breast milk or infant serum. 61
Evidence supports that T is neuroprotective (Supplementary Table S1).32,41,42 T's neuroprotective effect is consistent with our experience in clinical practice, where “self-reported” memory issues are improved on therapy, returning toward the end of the T implant cycle. Essential tremors are also improved on T therapy (Fig. 4).

Left: a 65-year-old patient's attempted signature (top) and initials (middle) before her first T pellet implant and 2 h post-160-mg T implants (bottom). The patient also reported being able to drink from a water bottle without spilling, blow dry her hair, eat soup in a restaurant for the first time in 4 years, and use automated teller machines. Right: the same patient overdue for implants. Two attempted signatures before T implant therapy (top) and 24 h post-300-mg T implants (bottom). T, testosterone.
A significant finding noted in the past 15 years is the consistent relief of migraine headaches in pre- and postmenopausal women, which we documented in a small pilot study. 15 Hormonal stabilization may improve headaches and other conditions, including epilepsy.62–64
Currently in clinical practice, premenopausal women with migraine headaches, seizures, dysfunctional uterine bleeding, and endometriosis—are treated with an AI (anastrozole [A]) combined with T in the implant—as these conditions are affected by excess or fluctuating estrogens.44–53,62–64 Since serum levels of E2 do not reflect the local production of estrogen, the clinical signs and symptoms of excess estrogen should be monitored, including breast pain, fluid retention, anxiety, emotional disturbances, irritability, aggression, and lack of effect from T therapy.1,18–20
Some women discontinue T therapy for cosmetic and skin side effects, including facial hair growth, acne, mild clitoromegaly, and hirsutism. Some women choose to lower their T dose, whereas others prefer the benefits of higher T doses and choose to treat the side effects.
We have previously addressed some common myths and misconceptions surrounding T therapy in women. 12 In a questionnaire study on 285 patients, 48 of 76 (63%) patients who complained of age-related hair loss before therapy reported hair regrowth on T pellet therapy. 13 Interestingly, baseline serum T levels were lower in women who reported age-related hair thinning compared with women who reported “no hair thinning.” 13 T does increase red blood cell production, which can lower iron levels and contribute to iron deficiency—indirectly affecting hair. Thyroid, iron, and ferritin levels are monitored.
A prospective study specifically designed to investigate the effect of T implant therapy on the female voice demonstrated that therapeutic doses of T—resulting in “supraphysiological” T levels—had no adverse effect on the female voice, including lowering or deepening of the voice. 14 Interestingly, two of three patients with “lower than expected” fundamental frequencies at baseline improved on T therapy, which may be due to T's anti-inflammatory effects.14,39
Androgens and breast cancer
Although some epidemiological studies have shown an “association” between endogenous T levels and breast cancer risk, there is no evidence that T treatment causes breast cancer.7,17–19,32
Almost two decades ago, it was surmised that it is the balance (ratio) of T to E2 that prevents breast tissue from oncogenesis. 26 Subsequently, in an experimental in vivo primate model, we showed that the addition of T to “conventional” hormone replacement therapy attenuated the proliferative effects of estrogens on breast tissue. 65 The same effect was reported in women from Australia: “The addition of testosterone to conventional hormone therapy for postmenopausal women does not increase and may reduce the hormone therapy-associated breast cancer risk, thereby returning the incidence to the normal rates observed in the general untreated population.” 66
We measured salivary hormone levels in 357 newly diagnosed breast cancer patients and compared them with a matched “control” group. Steroid concentrations measured in saliva represent bioavailable hormone levels, excluding the fraction tightly bound to serum proteins (i.e., unavailable for biological action) and thus more accurately reflect steroid (androgenic) activity. 67 We found that breast cancer patients had lower T levels and a lower ratio of T to estrone, suggesting that higher bioavailable T counters the proliferative effects of estrogen in the breast. 67
In March 2008, a prospective Institutional Review Board-approved cohort study was initiated, which was specifically designed to investigate the incidence of breast cancer in women (n = 1267) treated with T implant therapy. Ten-year results revealed a reduced incidence of invasive breast cancer in women treated with T therapy.19,20 A total of 11 (vs. 18 expected) cases of infiltrating breast cancer were diagnosed in patients on T pellet therapy equating to an incidence rate of 165/100,000 person-years (p-y), which was significantly less than the age-matched “Surveillance, Epidemiology, and End Results” expected incidence rate of 271/100,000 p-y (p < 0.001) and historical controls.
Withdrawal of T therapy led to an increasing trend toward diagnosis of clinically active tumors over time (Fig. 5)—suggesting that T may reduce the progression of undetected cancers. 19 Data reported at year 5 showed that—unlike adherence to estrogen/progestin therapy (increased events) 68 —adherence to T therapy decreased the incidence of breast cancer, signifying a protective effect. 18 The reduced incidence of breast cancer in our cohort of women treated with T implants continues into 2021 (13 years).

Bootstrap results confirm a significant reduction in the incidence of invasive breast cancers on T therapy (≤120 day) compared with SEER incidence rates—with an increasing incidence after withdrawal of T therapy—number of days since last insert. 19 . SEER, Surveillance, Epidemiology, and End Results.
The innovative yet obvious use of an AI combined with T in a solitary pellet implant (T+AI) has revolutionized the use of T therapy in breast cancer patients. The combination T+AI subcutaneous implant enables the simultaneous and continuous delivery of both pharmaceutical active ingredients while avoiding the first pass effect. 69 The combined use of T and an AI provides women with the beneficial effects of T without compromising these results with the conversion of T to estrogens and their possible adverse effects in estrogen-dependent diseases, for example, hormone receptor-positive breast cancer.
Subcutaneous delivery (T+AI) has also proven useful in patients unable to tolerate oral AI therapy. There are no gastrointestinal side effects, including nausea and gastritis, abdominal or stomach pain, and vomiting. Subcutaneous delivery also bypasses the liver avoiding the enterohepatic circulation and hepatic metabolism, which is significant in patients with mild or moderate liver impairment or on oral medications that have a high “hepatic adverse drug reaction” potential. 70
Data presented at the American Society of Clinical Oncology conference demonstrated the beneficial effects of T on the relief of severe hormone deficiency symptoms in breast cancer survivors (stage 0–4) using the validated MRS questionnaire (Fig. 3). 23 Survivors were treated with the combination of T with A combined in the pellet implant. T doses and levels on therapy were followed. E2 levels were monitored and remained low. Statistically significant (p < 0.0001) improvement in all 11 symptom categories was reported (Fig. 6), supporting the direct effect of T at the AR in the relief of symptoms. In addition, there was (and continues to be) a reduced incidence of breast cancer recurrence in patients treated with subcutaneous T + A implants. 23

Summary of the distribution of severity scores in each of the 11 symptom categories at baseline (pretherapy) and post-T+A implant therapy. 41 A, anastrozole.
In clinical practice, T+AI implants (anastrozole or letrozole) used to treat symptoms of hormone deficiency in breast cancer patients, have also significantly reduced tumor size, including complete clinical and complete radiological responses (Fig. 7). Multiple case reports on the in vivo tumor responses to T+AI therapy have been published demonstrating the unarguable direct beneficial effect of T on invasive breast cancers.17,22,23
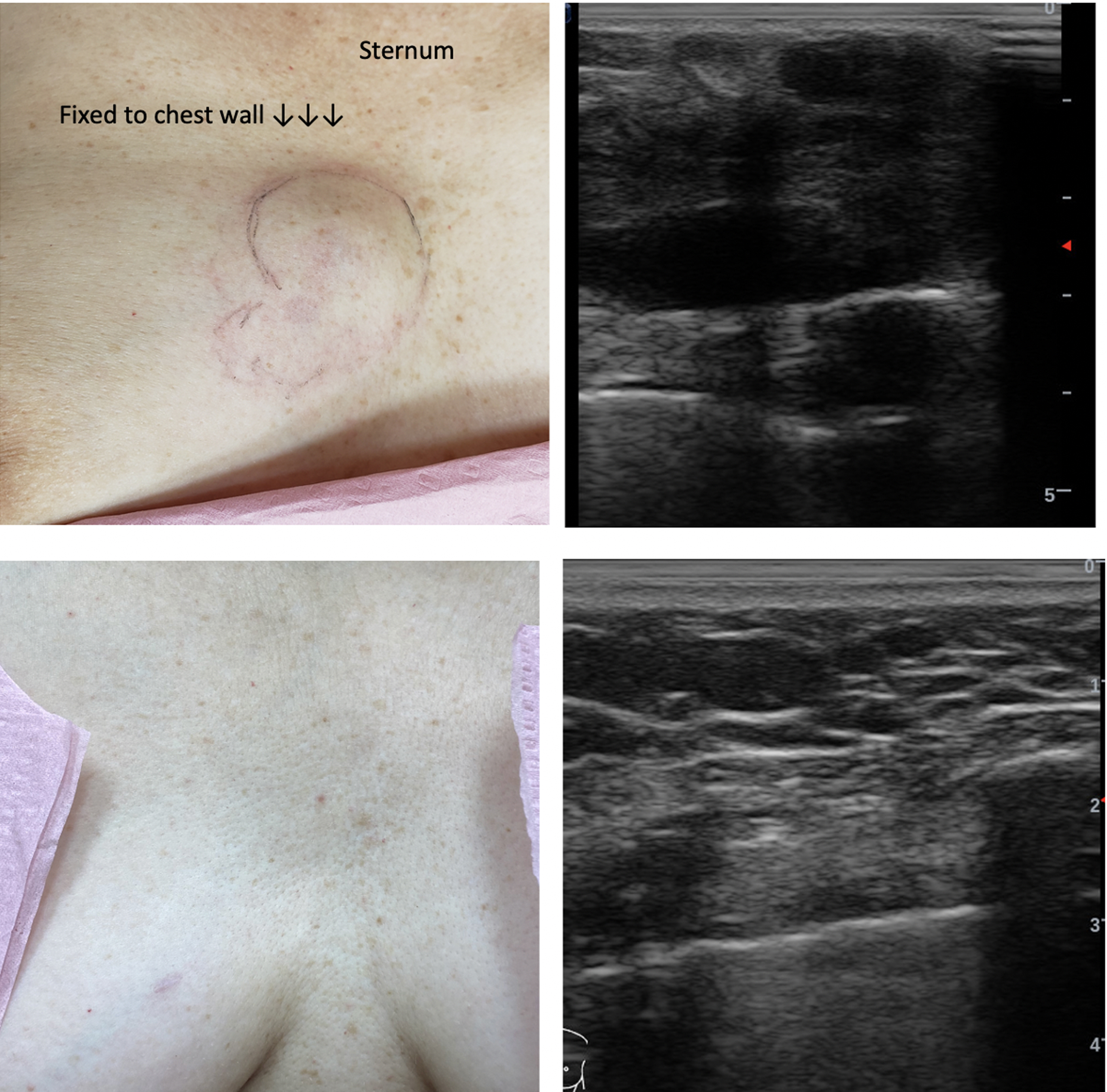
Fifty-eight-year-old patient referred with large immobile breast cancer fixed to sternum. Refused conventional therapy. She was treated with T 180–240 mg +12 mg letrozole combination pellet implants at baseline, weeks 6, 14, and 26. She also implemented dietary changes. Top left: baseline, 6-cm tumor fixed to chest wall (sternum) UIQ R breast, skin discoloration. Top right: baseline ultrasound, tumor invading periosteum (sternum) and skin—too large to be measured (extends off screen). Bottom left: week 14, complete clinical response, mass no longer palpable. Note indentation/shadow where tumor had stretched skin. Bottom right: week 26, complete radiographic response confirmed on ultrasound. Patient continues on T + A pellets and remains healthy and disease free at 2 years. UIQ, upper inner quadrant.
There is pre-clinical evidence suggesting that T may attenuate some side effects from chemotherapy, which is consistent with T's protective effects (neurological and cardiac) and with what we have experienced in clinical practice. 22 Subcutaneous T+AI therapy has been “life-changing” in the palliative treatment of women with metastatic breast cancer. T therapy significantly improves patient's QoL while simultaneously controlling disease—alone or along with conventional therapy (Figs. 8 and 9).

Significant improvement in QoL documented by MRS in two patients with metastatic breast cancer. Left side, baseline. Right side, on T+AI implant therapy. Top: 58-year-old patient with metastatic breast cancer, 4.5-cm palpable right breast mass, severe abdominal pain, weight loss, incontinence, malnutrition, unable to tolerate oral letrozole therapy. Refused chemotherapy. Patient treated with T + A alone. Breast tumor is responding to therapy. Bottom: 60-year-old patient presented (2015) with metastatic breast cancer (on conventional therapy), extremely severe bone pain, severe menopausal symptoms, and required assistance to walk. Currently 5-years out (65-year-old), alive, and well (thriving) on T+AI therapy. Continues conventional therapy. AI, aromatase inhibitor.

A 67-year-old female presented with acute respiratory failure. Baseline CT scan (left column) of the chest showed multiple noncalcified pulmonary nodules—bilateral and throughout the lungs—compatible with metastatic disease. Core biopsy (breast mass) revealed ER+ invasive ductal carcinoma. The patient refused conventional therapy and was treated with T+letrozole pellet implants (320 mg T + 24 mg letrozole every 9 weeks). She also began a “whole food” low glycemic diet. One year later, CT scan (right column) showed considerable improvement in the size and number of nodules throughout the lungs. The patient lost 13.6 kg (note significant decrease in fatty tissue on CT), remains asymptomatic, and “feels amazing.” The large 8-cm breast mass has markedly decreased in size and axillary nodes are no longer palpable. CT, computed tomography; ER, estrogen receptor.
One of us (R.G.) was a clinical consultant to the Mayo Clinic for the “Alliance Trial A221102, a Randomized Double-Blind Placebo Controlled Study of Subcutaneous Testosterone (pellets) in the Adjuvant Treatment of Postmenopausal Women with Aromatase Inhibitor Induced Arthralgias.” 71 Patients receiving subcutaneous T + A implants reported statistically significant improvements in hot flashes, fatigue, mood swings, urinary incontinence, and skin appearance, tone, and texture. 71
However, the 120-mg T implant dose (with 8 mg anastrozole) did not (significantly) relieve arthralgias in patients on oral AIs. The investigators surmised that the dose of subcutaneous T was too low. Consistent with T's dose-dependent effects,1,11,72,73 a previous observational study using higher doses of subcutaneous T (169 ± 32 mg) in breast cancer survivors (not on oral AI therapy) reported significant improvements in somatic symptoms, including joint pain and muscular discomfort. 23 Unfortunately no RCTs using higher doses of subcutaneous T have been performed.
Pharmacological dosing for a physiological effect
Controversial topics in treating women with T include the following: the diagnosis of androgen deficiency, T dosing, and T levels on therapy.7,43,74
Some guidelines recommend against treating women with T because serum levels do not correlate with symptoms.7,43,74 We have shown that neither symptoms of androgen deficiency (with the exception of sexual complaints) nor response to therapy correlate with baseline T levels, which is consistent with other studies and the physiology of androgens in women.1,11
The decision to initiate T therapy is a clinical decision between the doctor and patient based on the patient's symptomatology.1,11 This assessment is in agreement with the American College of Obstetricians and Gynecologists (ACOG) Committee Opinion, which states that “Individualized testing is only indicated when a narrow therapeutic window exists for a drug or drug class. Steroid hormones do not meet these criteria and do not require individualized testing” and “If treatment is initiated for symptom control, subjective improvement in symptoms is the therapeutic end-point, and there is no need to assess hormone levels. Hormone therapy should not be titrated to hormone levels.” 75 This opinion differs from statements and guidelines that recommend baseline testing and monitoring hormone therapy with serum levels.7,74
Recent guidelines use the terms “physiological dosing” and “physiological levels” when making recommendations for T therapy. 7 However, this is counterintuitive to physiology—the major source of bioavailable T in women is unmeasurable and not reflected in serum T levels. “Physiological dosing” may be why T therapy—effectively raising T levels into the mid to high physiological range—has proven clinically ineffective in some studies. 76 T's effect is dose dependent, and there is no evidence (i.e., drug concentration in blood studies), or documented adverse events, supporting the “opinion” that serum T levels on therapy should remain within endogenous or “physiological” ranges—concentration/dose–response studies support the opposite.1,11,72,73
True “physiological” T dosing must deliver adequate amounts of T to the AR (tissue level) to replace the minor contribution (<20%) from circulating T (measurable in serum)—and most importantly—to replace the major contribution (>80%) to T from the preandrogens, which also decline with age.27–31 Severe T deficiency occurs in conditions that affect production of adrenal precursor steroids—further supporting the major contribution of DHEAS, DHEA, and androstenedione to the peripheral production of T in target organs. 77
Doses and therapeutic ranges for exogenous therapy with T pellet implants have been published in the peer-reviewed literature (Table 2).11,14,20,23 Trough (nadir) levels, measured when symptoms returned, 11 support that T levels on exogenous therapy cannot be compared with, monitored by, or dosed based on endogenous T ranges in serum.8,9,77 In addition, there is a significant interindividual variation (coefficient of variation [CV] >40%) as well an intraindividual variation (CV 25%) in T levels on therapy suggesting that a single T level is extremely variable and of little or no value in clinical decision making, further supporting ACOG's position on hormone testing.11,75
Testosterone Doses and Serum Levels on Therapy
Trough/end levels were drawn when patient symptoms returned. Of note, trough levels were several-fold higher than endogenous T ranges.
A, anastrozole; ASCO, American Society of Clinical Oncology, CV, coefficient of variation, PK, pharmacokinetic; QoL, quality of life; SD, standard deviation; T, testosterone.
Of note, salivary T levels may be more accurate than serum for assessing bioavailable T in patients treated with T implant therapy (Supplementary Data S2). However, serum is readily available and commonly used in clinical practice.
Additional data
Between March 2017 and January 2021, we collected 1106 data points on 667 female patients treated with T implants. These data points do not represent all patients receiving T pellets, only those who had laboratory data collected during this time frame. Of note, breast cancer patients are monitored (E2 levels) more frequently (month 1 and nadir)—and may represent a higher proportion of patients in this series.
Patient's age, body mass index (BMI), weight, T dose, interval of insertion, and blood count on therapy are listed in Table 3. T stimulates erythropoietin and increases red blood cell production. Patients sign a consent informing them that T therapy can raise “red blood counts” and that “high blood counts have been associated with blood clots.” Hemoglobin (Hb) and hematocrit (Hct) are monitored in patients treated with T implant therapy. Four of 667 female patients had Hb >17 g/dL and two patients had a Hct >52%. One patient had a Hb of 18.8 and a Hct of 53.4%. It was recommended that she must consult with a hematologist and discontinue T therapy.
Patient Demographics
BMI, body mass index.
In the past 15 years, no female or male patient in our clinical practice has had any adverse events (cardiac or thrombotic) due to secondary polycythemia.1,11,25,78 Although some studies show an association between elevated Hct and thrombosis, thrombosis does not accompany most types of erythrocytosis. 79 True erythrocytosis is defined as a packed red blood cell volume >125% of predicted for an individual's height and weight or a Hct >56% in a female. 80
Secondary polycythemia from T therapy or other nonmedical conditions (e.g., high altitude) does not have the same risk of thrombotic events compared with polycythemia due to medical conditions (e.g., chronic lung disease/hypoxia), which are associated with many confounding health problems.79,81,82 This lack of adverse events is consistent with the lack of adverse thrombotic and cardiovascular events reported in transgender men and male patients treated with T therapy.78,82–89
T levels over time (days since implantation) are shown in Figure 10. In this particular data set, we were interested in T levels at week 1. Note the cluster of levels at week 1 and at month 1, which is a frequent collection time frame. T levels peak at insertion. There was a gradual decline of serum T levels, at a rate of 3.86 ng/dL per day. The intercept was 572.5 (ng/dL): week 1 levels (collected between day 4 and 10) were 518.6 ng/dL ±215.2 (CV 0.41) (Fig. 10).

T levels on subcutaneous implants throughout the implant cycle. n = 1106 data points. Intercept 572.49 ng/dL. Slope −3.86 (ng/dL per day).
Trough (nadir) serum T levels were drawn when “symptoms returned” and obtained within ≤7 days before the patients subsequent T insertion procedure. Mean serum T levels at the end of the implant cycle, that is, when symptoms returned, was 236.9 ± 108.0 ng/dL (CV 45.6). However, in some circumstances (e.g., metastatic breast cancer, multiple sclerosis, and memory loss) patients return before the onset of clinical symptoms for optimal disease control.
Correlations between patient demographics and T doses and levels on therapy are presented in Table 4. T dosing is weight based and there was a moderate positive correlation between weight/BMI and T dose. There was also a small positive correlation between age and T dose. The total androgen pool declines with age and we have found that some women benefit from increasing doses of T as they age.
Pearson Correlations
Hb, hemoglobin; Hct, hematocrit.
Serum T levels were collected throughout the implant cycle, which makes correlations with T levels on therapy difficult to interpret. However, there was a small negative correlation between BMI/weight and T levels on therapy. This is consistent with what we reported in male patients: men with lower BMI had higher serum T levels on therapy (all time frames) despite lower dosing. 25 There were small (<0.29) positive correlations between Hb/Hct and body weight, BMI, T dose, and T level on therapy—the strongest correlation with T dose (Table 4).
Adequate dosing of T is critical for optimal therapeutic effect. Symptom response and control of disease should guide therapy rather than arbitrary serum T levels. T therapy is continued or adjusted based on the patient's response to therapy (benefits) versus side effects, not on a single T level, which is inherently unreliable.1,11 Pellets are (re) inserted when symptoms return. The T dose and/or the interval of insertion are adjusted based on an individual's disease state, response to therapy, goals, and preferences.
Additional data supporting the safety of pharmacological T dosing include long-term studies on transgender men, which have shown that significantly higher (male) doses of T do not increase the risk of cardiovascular events, stroke, cancer—and increase insulin sensitivity.84–89 The increase in insulin sensitivity may be surprising as women with polycystic ovarian syndrome and insulin resistance also have high T levels. However, evidence suggests that hyperandrogenism is secondary to hyperinsulinemia—insulin stimulates the production of androgens and treating hyperinsulinemia ameliorates hyperandrogenism.90,91 The absence of significant “therapy-related” adverse health events is consistent with our long-term experience.
Discussion
We have provided clinical evidence supporting indications for, safety of, and benefits of androgen therapy in women, which extends beyond hypoactive sexual desire disorder/dysfunction. This is in contrast to the Global Consensus Position Statement on the Use of Testosterone Therapy for Women—which was widely publicized by specialty societies—and lists hypoactive sexual desire disorder/dysfunction as the “only evidence-based” indication for T therapy. 7 However, relying solely on industry-sponsored RCTs (which many of the authors have been involved with) 7 as the only “evidence,” could lead to biased recommendations (Supplementary Data S3).92,93
Although we recognize that other studies on T and T implant therapy often include heterogeneous populations, have variable dosing, and have different outcome measurements, they should not be totally omitted or disparaged by guideline authors.92,93 Although guidelines are a valuable source of information, there are “concerns” regarding their integrity and, in particular, recommendations based on “opinion” as the level of evidence (Supplementary Data S3).7,92–96
Although unregulated, T pellets have been safely used for >80 years in clinical practices around the world. We presented original data supporting the efficacy and safety of T implant therapy in clinical practice and detailed an unmet and urgent need in breast cancer patients. Understanding the physiology of androgens in women enables one to comprehend the rationale behind T implant therapy (indications and dosing), and counters disingenuous arguments against the use of “T preparations that result in supraphysiological concentrations.” 7
Conclusion
T is not a “new” drug. It is an endogenous steroid hormone with a wide margin of safety—high therapeutic index. a Adequate amounts of bioavailable T at the AR are critical for optimal physical and mental health. T therapy in women should be dosed based on clinical efficacy and response to therapy. There is no evidence that T therapy should be monitored by serum T levels or managed based on arbitrary ranges.
In >15 years of clinical experience, T implant therapy has proven invaluable, dramatically improving the health, sexuality, and QoL of thousands of women. T combined with an AI should be considered in patients with symptoms of estrogen excess or a history of ER-positive breast cancer.
The values and goals of the individual patient should be paramount in decision making. Decades of evidence support both the safety and efficacy of T therapy in women, including breast cancer patients. Withholding adequate doses of T therapy could be detrimental to an individual's health, QoL, and survival.
Footnotes
Authors' Contributions
Both authors contributed to the conception and design of the article, critical revisions, and final approval of the article. Both authors agree to be accountable for all aspects of the study.
Acknowledgments
We thank Jennifer Dichito, MA, and Shayna Smith, BS, for data collection and diligent patient follow-up, and Michael Glaser-Garbrick, BS, MS, for his technical assistance and statistical analysis.
Author Disclosure Statement
C.D. has no stocks or shares in a company or financial ties to an organization, funding, employment, or other competing interest. R.G. has no stocks or shares in a company or financial ties to an organization funding, or employment. R.G. does have a patent on the testosterone/AI patent.
Funding Information
No funding was received for this article.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
