Abstract
Background:
Klinefelter's syndrome (KS) is characterized by extra X chromosomes and features of primary hypogonadism including osteopenia and osteoporosis. Testosterone therapy (TTh) is widely used to treat men with KS and low serum testosterone/hypogonadal symptoms, though studies on its efficacy in improving bone density show varied outcomes.
Materials and Methods:
We studied the effects of TTh, bisphosphonates, and vitamin D/calcium in 38 men with KS and low testosterone, hypogonadal symptoms, and T-scores consistent with osteoporosis. Our aim was to investigate at the end of follow-up (median: 87 months, range: 27–147 months), associations between age, baseline total testosterone, and T-scores, and change in T-scores after treatment.
Results:
At final assessment, all men had T-score values outside the osteoporotic range (−1.1 standard deviation [SD], −1.8 SD). Baseline age but not median baseline testosterone appeared associated with change in T-score and T-score at final assessment. All men had dual-energy X-ray absorptiometry every 6 months and demonstrated continued improvement in T-scores after 3 months and up to 72 months. Baseline age and T-scores (stratified by median) were associated with change in T-score at final assessment. Compared with men ≥51 years, those aged <51 years showed significantly greater improvement in T-scores between 6 and 30 months. Men with worse T-score values (<3.7 SD) showed significantly greater improvement at every time point up to 36 months. Our results indicate that TTh, bisphosphonates, and vitamin D/calcium improve osteoporosis although there is a need to better understand the effects of the individual therapies, age, and baseline T-score on treatment efficacy.
Introduction
Klinefelter's syndrome (KS), a condition characterized by extra copies of the X chromosome, is associated with testicular hypotrophy, impaired spermatogenesis, and, even though 40–50% of affected men may have serum testosterone in the normal distribution, features of primary hypogonadism including metabolic syndrome, autoimmune disorders, and osteoporosis.1–9 Hypogonadism together with age >70 years, body mass index <21 kg/m2, >10% weight loss, physical inactivity, and prolonged corticosteroid use are recognized risk factors for male osteoporosis.10–14 Testosterone has direct (through the androgen receptor) and indirect (through aromatization to form estrogens) actions on bone metabolism. These promote periosteal bone formation during puberty and subsequently, decreasing resorption putatively explaining why hypogonadism, characterized by low testosterone levels and accompanying symptoms, is associated with both reduced bone mass and density.6,15,16
KS is associated with osteopenia, osteoporosis, and increased fracture risk.17–20 Reduction of bone density in men with KS is associated with postpubertal testosterone deficiency and although histological change is related to serum testosterone, some individuals with KS develop low bone mineral density even with testosterone levels in the normal distribution.6,19,21 This suggests a multifactorial etiology 21 with putative contributing factors including androgen receptor insensitivity16,22,23 and/or hypovitaminosisD. 24
Guidelines from the National Institute for Health and Care Excellence (https://bnf.nice.org.uk/treatment-summary/osteoporosis.html) and National Osteoporosis Guideline Group support lifestyle measures including increasing weight bearing activity and body mass index between 20 and 25 kg/m2 together with therapeutic interventions to prevent fractures in osteoporotic individuals. 25 Oral bisphosphonates inhibit bone resorption and are front-line agents in osteoporotic males with denosumab and teriparatide used in individuals intolerant to bisphosphonates. 25 Daily cholecalciferol supplements of 800 IU are recommended in men aged >50 years with confirmed osteoporosis. 25
There is debate whether men with serum testosterone in the normal distribution without hypogonadal symptoms benefit from testosterone therapy (TTh) and currently no international guidelines regarding TTh use in men with KS exists. 20 The clinical consensus appears to be that men with KS and low serum testosterone/hypogonadal symptoms require TTh. Ferlin et al. suggested a diagnostic and therapeutic flow pathway for KS patients based on bone mineral density and serum testosterone. 6 In men with low bone mineral density and low serum testosterone, TTh and bisphosphonates with possible vitamin D/calcium supplements and repeat dual-energy X ray absorptiometry (DEXA) monitoring at 24 monthly intervals are recommended. 6 Longitudinal studies on the efficacy of TTh in improving bone mineral density in men with hypogonadism show varied outcomes, possibly because of heterogeneity.20,26 A study of 72 hypogonadal patients (21 men with KS) showed TTh was associated with increased bone mineral density 27 whereas Ferlin et al. showed that bone mineral density improvement was associated with concurrent TTh and vitamin D supplementation in 14 men but with no improvement in 12 men given only TTh. 24 Haider et al. showed significant improvement in bone mineral density in 45 men with hypogonadism and osteoporosis given testosterone undecanoate (TU). 28
We describe further study of the impact of TTh on bone density (T-score) in an expanded version of the registry data base described by Haider et al. 28 The study group comprised 38 men with KS and osteoporosis with focus on those presenting with back pain and osteoporosis and on combination treatment with TTh, bisphosphonates, and vitamin D/calcium. Our aim was to document T-scores at the end of follow-up (median: 87 months, IQR: 57–117, range: 27–147 months) and T-score change at 3–6 monthly intervals up to 72 months. We examined the data for associations between age, baseline total testosterone, and T-scores, and change in T-scores after combination treatment.
Materials and Methods
We used the database from an ongoing observational, prospective, and cumulative registry study that included data from 47 men with KS confirmed by karyotyping. DEXA identified osteoporosis in 39 men with KS and this study presents data from the 38 men treated with TU, bisphosphonates (all the men were on oral alendronate 70 mg once weekly), and vitamin D (colecalciferol 20,000 IU per week)/calcium during the entire or part of follow-up. Alendronate was discontinued in 4, 19, 22, 23, 24, and 26 men (cumulative numbers) by 12, 24, 36, 48, 60, and 72 months of follow-up, respectively, whereas 12 men continued with the treatment after 72 months. The one man treated only with TTh was excluded. The complete registry database currently comprises 823 men who presented with urological symptoms and low testosterone levels (≤12.1 nmol/L) and hypogonadal symptoms; 737 and 39 men were diagnosed with adult onset testosterone deficiency and non-KS primary hypogonadism, respectively. Gonadotropin and estrogen levels were not measured either at baseline or during follow-up. The median age of the cohort was 51 (IQR: 44/55) years at presentation and TTh commencement. Some of the 38 men in the study cohort were on antihypertensive agents (2 men), oral hypoglycamic agents (1 man), statins (3 men), and phosphodiesterase-5 inhibitors (7 men); the dates of initiation of these agents were not available.
Osteoporosis was identified using DEXA to measure bone mineral density in the spine and/or hip. 10 Results were compared with those of a healthy young adult and a T-score provided as standard deviations (SD); T-scores between +1 and −1 SD, −1 SD and −2.5 SD, and <2.5 SD indicate normal bone density, osteopenia, and osteoporosis, respectively.11–13 After diagnosis of osteoporosis, TTh (1000 mg TU, interval between first and second administration: 6 weeks, thereafter: 12 weekly) was initiated. Of this cohort, 35 men were commenced on bisphosphonates at baseline (1 man was started at 6 months and the remaining 2 men after 9 months) and all were on vitamin D/calcium supplements at baseline. Data including DEXA were entered into the database at least 6 monthly (many had 3 monthly DEXA) during follow-up. As the study is longitudinal, number of patients decreased with follow-up; data were available on 36, 35, 35, 29, 23, and 16 (of the 38) men at 12, 24, 36, 48, 60, and 72 months, respectively. None of the men suffered from fractures during the follow-up period. The German Medical Association's ethical guidelines for observational studies were followed and each participant consented to be included in the register and their data were analyzed after being provided study details. Ethics committees (Germany and England) reviewed the study and stated that formal approval was not required. Keele University Staffordshire and University Hospitals Birmingham NHS Foundation Trust reviewed the manuscript/study and provided Institutional Review Board Statements.
Study Measurements
Serum testosterone was measured using the Abbott Architect immunoassay. Bone mineral density was measured by using a whole body dual-energy X-ray densitometer (Norland XR-800), with calculations performed in line with the standardized procedures issued by the manufacturer. The daily quality assurance calibration procedures were in accordance with the manufacturer's recommendation using a QA Calibration Standard and a QC Spine Phantom. The accuracy of AP spine scans and hip scans was within 1.0% of industry standard. The in vivo precision of AP spine scan and hip scan is 0.84% (bone mineral density lumbar vertebra 2–4 coefficient of variation) and 1.4% (bone mineral density femoral neck coefficient of variation), respectively. Bone mineral density is expressed in gram per square centimeter. The individual bone mineral density variation was expressed as T-score measurements of the spine (lumbar vertebra 2–4) and femoral neck.
Statistical analysis
Nonparametric tests were carried out to compare between- (rank-sum) and within-(sign-rank) cohort changes in T-scores during the follow-up. All analyses were performed on Stata 14 (StataCorp LLC, TX).
Results
Table 1 gives median age, total testosterone, and T-score values at baseline that were 51 years, 9.7 nmol/L, and −3.7 SD, respectively, in the 38 men with KS treated with TTh, bisphosphonates, and vitamin D/calcium. The footnote of Table 1 shows other baseline characteristics. One man had type 2 diabetes at baseline with no further cases diagnosed during follow-up. Three men were prescribed statins. At baseline, all the men had T-score values consistent with osteoporosis (between −2.6 and −4.2 SD). At final assessment, all the men had T-scores outside the osteoporotic range (between −1.1 and −1.8 SD), the difference between baseline and final assessment values was significant (p < 0.0001). Median trough total testosterone levels increased from 9.7 to 18.4 nmol/L after TTh.
T-Score at Baseline and Final Assessment in the Total Cohort and Subgroups Stratified by Median Age, Total Testosterone Concentrations, and T-Score at Baseline
Demography [Median (IQR) for continuous variables, % for discrete variables].
Baseline—waist circumference: 100 (98/105) cm, weight: 91.5 (85/103) kg, Hb: 14.8 (14.5/15.2) g/dL, hematocrit: 46 (45/46), PSA: 0.35 (0.12/0.69), HbA1c: 5.4 (5.1/5.7)%, systolic blood pressure: 132.5 (128/136) mmHg, diastolic blood pressure: 77 (74/82) mmHg, IIEF-EF score: 15.5 (11/22), cholesterol: 7.2 (6.8/7.5) mmol/L, triglycerides: 2.6 (2.5/2.9) mmol/L, HDL-cholesterol: 0.93 (0.70/1.11) mmol/L, diabetes: 2.6%, smoking: 47.4%, statin treatment: 7.9%, vitamin D treatment: 100%, bisphosphonates treatment: 92.1%. Final assessment—diabetes: 2.6%, smoking: 47.4%, statin treatment: 7.9%, vitamin D and bisphosphonate treatment: 100%, mortality: 0%, cardiovascular disease: 0%.
SD, standard deviation; TTh, testosterone therapy.
Table 1 also gives the effects of TTh, bisphosphonates, and vitamin D/calcium in subgroups stratified by median values of age, baseline testosterone, and baseline T-scores. T-scores and follow-up values together with within- (sign-rank tests) and between (rank-sum)-group differences are presented for each subgroup. Regardless of stratification, all subgroups showed significantly (within-group sign-rank test) improved T-scores at the end of follow-up. Age and baseline total testosterone levels (stratified by median) were not associated with baseline T-scores or duration of follow-up. However, baseline age appeared associated with change in T-score and T-score at final assessment after treatment, whereas no difference was observed between the men when stratified by median baseline total testosterone. Table 1 also gives data for the men stratified by median baseline T-score; follow-up significantly varied (p < 0.0001) between the two groups. Despite that men with a baseline T-score <−3.7 SD demonstrated greater improvement, although not statistically significant (p = 0.096).
Change in T-scores at 3 monthly time points for 72 months
Table 2 and Figure 1 show median T-score values at 3 monthly intervals, all men having had DEXA every 6 months and many every 3 months. Using fixed time points removed impact of treatment duration (follow-up) as a confounder. Table 2 presents the number of men showing improvement, no change or deterioration in T-score values, compared with values obtained at baseline and the DEXA scan carried out 3 months previously. Table 2 demonstrates continued improvement up to 72 months with T-scores significantly better than baseline values after only 3 months treatment (no man had a T-score worse than his baseline value at any time point). The T-score range at each time is presented and this shows that after 9 months treatment, every man had improved values. After 45 months treatment, every man (30/36 men had follow-up ≥45 months) had a T-score above the osteoporosis cutoff of −2.5 SD. A worsening of T-scores compared with that obtained 3 months previously was evident in only one individual between 36 and 39 months and another between 60 and 63 months.
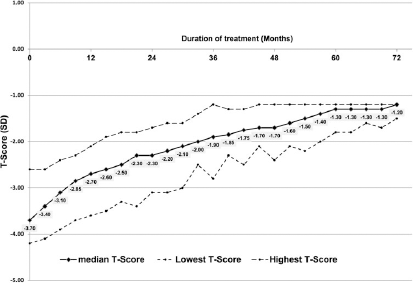
Median T-score values at 3 monthly intervals in the total cohort after treatment.
Median T-Score Values at 3 Monthly Intervals in the Total Cohort and the Number of Men Showing Improvement, No Change, and Deterioration in T-Score Values Compared with Baseline and DEXA 3 Months Previously
DEXA, dual-energy X ray absorptiometry.
Change in T-score at 3 monthly time points for 36 weeks in groups stratified by baseline median age and T-scores
Data in Table 1 indicate baseline age and T-scores (stratified by median) are associated with change in T-score at final assessment after treatment. Age (stratified by median) was not associated with T-scores at baseline (Table 1, p = 0.81) or baseline total testosterone (p = 0.64). We now studied the associations between age, baseline T-score (both stratified by median), and change in T-scores (compared with baseline) at 3 monthly time points up to 36 months (number of patients decreased with follow-up). Compared with men aged ≥51 years, those aged <51 years showed significantly greater improvement in T-scores between 6 and 30 months (Table 3 and Fig. 2). Between-group difference in the change in T-score was maximal at 27 months (0.75 SD) and then decreased (30 months: 0.55, 36 months: 0.35, values from the same 35 men). Table 3 and Figure 3 show that men with worse T-score values (<3.7 SD) showed significantly greater improvement at every time point up to 36 months. There did not appear to be any convergence in the T-score change.

Median values of change in T-score at 3 monthly intervals in the cohort stratified by median age at baseline (51 years).

Median values of change in T-score at 3 monthly intervals in the cohort stratified by median baseline T-score (−3.7 SD). SD, standard deviation.
Median T-Score Change After Treatment in the Cohort Stratified by Median Age and T-Scores at Baseline
Discussion
We studied changes in T-scores in 38 men with KS after treatment with TTh, bisphosphonates, and vitamin D/calcium and then determined whether baseline age, testosterone, and T-scores predicted change in T-scores. Baseline T-scores of <−2.5 SD (range: −4.2/−2.6) indicated all 38 men had osteoporosis. Improvement in T-scores was noted in some after 3 months treatment and in all men after 9 months. After 45 weeks, no patient had a T-score in the osteoporotic range. Treatment in men <51 years or with a baseline T-score <−3.7 effected significantly greater improvement in score after 6–30 months and 3–36 months, respectively.
Currently there appears consensus that TTh is beneficial for most men and adolescents with KS with elevated serum luteinizing hormone and low (or low-normal) testosterone levels. 20 As the men we studied had total testosterone <12.1 nmol/L and symptoms of hypogonadism, TTh was appropriate treatment and every man in our cohort demonstrated improved T-scores after 9 months. Indeed, a previous study showed that although combined TTh and vitamin D treatment improved bone mineral density in KS, TTh without vitamin D supplementation was not beneficial. 24 Furthermore, Stepan et al. studied the effects of intravenous ibandronate on bone density and biochemical bone markers in 14 men with KS (mean aged 55.2 years, 5.9 years follow-up) and found hypovitaminosis D adversely affected the efficacy of bisphosphonate therapy. 19
Our analyses for 36 months of treatment showed men <51 years enjoyed greater benefit (statistically significant during 6–30 months treatment) than their older counterparts. Men with T-score <3.7 SD also demonstrated greater benefit during the entire 36 weeks studied than men with T-scores between −3.6 and −2.6 SD. Limited number of patients did not allow study of interactions between these predictive baseline factors, though importantly age was not related to baseline T-score and, therefore, does not appear related to the etiology of osteoporosis in KS. If our findings are validated, treatment with one or more of the therapeutic agents we used may be more efficacious in younger men. The relationship between baseline T-score and change in values after treatment is in accordance with the Wilder principle that states that the “direction of response of body function to any agent depends to a large degree on the initial value of that function.” 29 The lack of a significant association between baseline testosterone and T-scores perhaps does not suggest that TTh alone reverses the low T-scores associated with hypogonadism. This suggests that there may be other factors that we have not identified that could be part of a multifactorial etiology. Furthermore, as all the men had serum total testosterone levels <12.1 nmol/L at baseline, a possible association with T-scores could have been lost due to compression of the baseline serum testosterone level. However, the observed association is reassuring as men with the lowest T-scores gained greatest benefit, enabling each man to have a T-score above the osteoporosis threshold at final assessment. Currently repeat DEXA is recommended after a minimum of 2 years though we found benefit after 9 months of treatment, suggesting the time period for repeating DEXA could be less frequent for men given TTh, bisphosphonates, and vitamin D/calcium. 6
Bone mineral density (spine and hip) measurements were significantly improved in the 211 men (≥65 years) participating in the bone trial arm of the T-trials randomized to either testosterone or placebo gel for 12 months. 30 Ng Tang Fui et al. recently reported that 2 years of TU treatment of 92 men with hypogonadism, compared with 85 men on placebo, was associated with increased tibial and spinal bone mineral density in a double-blind RCT (T4Bone substudy). 31 Porcelli et al. reviewed the trials of male osteoporosis treatment with bisphosphonates, denosumab, and teriparatide, and suggested that earlier diagnosis of the condition and secondary causes together with improved treatment were essential. 32 Thus, it is our intention to carry out a similar analysis of the combination of TTh, bisphonates, and vitamin D treatments as well as the effects of the individual treatments, data permitting, on men with adult onset TD treated similarly to study T-score changes and predictors of change. We avoided pooling both cohorts in view of the varying etiologies.
This longitudinal registry study has strengths and weaknesses. As TU was injected in clinic, compliance was absolute. Follow-up was long and we had a complete data set for all men at 6 monthly and most at 3 monthly intervals. Although clinical guidelines do not advocate 3 monthly DEXA, this was in accordance with the local orthopedic scientific practice with an interest in short-term changes in bone density and performed at no additional cost to the patient. 28 We recognize that this frequency of testing is neither needed nor feasible in routine clinical practice, but the availability of the data allowed us to define the change in bone density over time with confidence. The number of patients was limited and hence, we could not study interactions between age, baseline T-scores, and effect of treatment. The declining number of patients during follow-up is not due to drop-out rates, but a result of cumulative ongoing recruitment (there was follow-up data in 35 of the 38 men at 36 weeks). As the 38 men were on TTh, bisphosphonates, and vitamin D, study outcomes are only applicable to this combination therapy as opposed to single agents. Interestingly, although the action of bisphosphonates is mainly antiosteoclastic, 25 TTh has been demonstrated to increase osteocalcin levels, suggestive of osteoblastic stimulation. 33 Furthermore, we did not have serum vitamin D levels at baseline or during follow-up. The German Health Interview and Examination Survey for Adults analyzed vitamin D status (serum vitamin D concentration ≤50 nmol/L considered low and suitable for supplementation) in German adults and found the mean serum vitamin D levels to be 45.6 nmol/L (61.6% and 30.2% of the individuals had levels <50 and <30 nmol/L, respectively) with no gender differences. 34 Subgroup analysis showed that the 759 men residing in northern Germany at a latitude between 52° and 54° (Bremerhaven is situated at an approximate latitude of 53.5°) had a mean vitamin D concentration of 45.1 nmol/L. 34
Conclusions
We studied 38 men with KS and low total testosterone, hypogonadal symptoms, and T-scores consistent with osteoporosis who were treated with TTh, bisphosphonates, and vitamin D/calcium supplements. Beneficial change in T-scores was seen in all patients after only 9 months. Men with the lowest T-scores improved most. Our results indicate the need to better understand the effects of the individual therapies and the effects of age and baseline T-score on treatment efficacy. Defining patient subgroups and studying their outcomes should improve therapeutic efficacy. A further factor in considerations of TTh use are studies showing improvements in muscle mass preservation in healthy men and preserved knee extensor muscle mechanical function in men with T2DM.35,36 Thus, TTh may in at least some men offer improvements to both bone density and muscle function.
Footnotes
Authors' Contributions
R.C.S. contributed to design of study, data analysis, and preparation of article; C.S.K. contributed to design of study and preparation of article; A.P. contributed to design of study and preparation of article; A.R. contributed to data analysis and preparation of article; G.H. contributed to design of study and preparation of article; A.H. and K.S.H. contributed to patient recruitment, data collection, and preparation of article; P.D. contributed to transposing the data, data analysis, and maintaining the database; F.S. contributed to maintaining the database, design of study, and preparation of article; S.R. contributed to design of study, data analysis, and preparation of article.
Author Disclosure Statement
S.R. has received research grants, travel grants, and speakers' honoraria from Besins Healthcare. F.S. works as a consultant for Bayer AG and is stockholder. G.I.H. has been an occasional speaker for Bayer AG and Besins Healthcare. A.H. and K.S.H. have received research grants, travel grants, and speakers' honoraria from Bayer AG. C.S.K., A.P., A.R., P.D., and R.C.S. have no disclosures.
Funding Information
No funding was received for this article.
