Abstract
Background:
Life expectancy has increased dramatically worldwide, resulting in a sharp rise of age-associated diseases, including late-onset hypogonadism (LOH) in men.
Aim:
To assess the prevalence of LOH and to determine its relationships with cardiovascular risk factors in healthy middle-aged men.
Material and Methods:
A total of 200 men aged 44–55 (median 48.44, interquartile range 45.02–52.50) undergoing regular medical examinations were enrolled in a study. All participants were asked to complete the International Index of Erectile Dysfunction, Aging Male's Symptoms, and Beck Depression Inventory questionnaires. Sex and gonadotropin hormones were measured in all participants. Cardiovascular diseases (CVD) risk was assessed in all participants by using the Systematic Coronary Risk Evaluation scale, and additional risk factors were recommended in the 2019 European Society of Cardiology and European Atherosclerosis Society guidelines for the management of dyslipidemias: lipid modification to reduce cardiovascular risk.
Results:
Ninety-eight (49%) men suffered from LOH. Men with total testosterone (TT) of <12 nmol/L were considered testosterone-deficient. The mean TT level was 8.9 ± 2.4 nmol/L in the LOH group. Clinical signs and symptoms of LOH were associated with lower TT levels: 7.78 ± 2.49 and 10.15 ± 1.32 in the groups with and without LOH, respectively (p = 0.004). The prevalence of obesity, metabolic syndrome, and depression was significantly higher among men with LOH, compared with men without it. High cardiovascular risk was found only in those participants present with LOH (p = 0.003). After collecting other risk factors, we found that 94.9% of participants assigned to the LOH group had high cardiovascular risk.
Conclusion:
The prevalence of LOH is high among healthy middle-aged men. Moreover, it is associated with an increased cardiovascular risk. Early measurement of TT levels in this population group may predict the development of CVD and allow the introduction of timely primary prevention measures to reduce cardiovascular morbidity and mortality.
Introduction
Life expectancy has increased dramatically among the global population, resulting in a sharp rise of age-related diseases, including late-onset hypogonadism (LOH). LOH, being commonly an overlooked condition, has been recently associated with a higher risk of cardiovascular diseases (CVD) among men. 1 Despite the decline of testosterone levels in aging men being rather common, androgen deficiency is often associated with diabetes, abdominal obesity, and other related diseases. 1 The Massachusetts Male Aging Study has reported a 1.2% per year decline in testosterone levels in different age groups in the general population starting from 1987. 2 However, this trend cannot be explained by an increase in the incidence of obesity and metabolic syndrome, as well as a decrease in smoking in the population.
The LOH in men has been recognized as a growing clinical problem among aging men.3,4 The LOH is defined as a clinical and biochemical syndrome in middle-aged men with normal pubertal development and is primarily associated with a decrease in testosterone levels. Men with total testosterone (TT) levels of <12 nmol/L are considered testosterone-deficient, and TT levels of <8 nmol/L require hormone replacement therapy.5,6 Many patients with LOH are believed to go unrecognized, since the symptoms are non-specific and can occur even at normal androgen levels. Moreover, diagnostic criteria for LOH remain controversial (e.g., erectile dysfunction [ED]), being a key criterion for LOH, and they can be associated with other diseases, such as early atherosclerosis. 6
In addition, men may present with primary testosterone deficiency without any clinical manifestations: This could be considered as asymptomatic low testosterone. 7 The prevalence of LOH in men aged 40–70 years varies from 30% to 40%, whereas subclinical LOH occurs in 23–38% of patients.7–9 The incidence of androgen deficiency is significantly higher among men with CVD, obesity, diabetes, osteoporosis, and metabolic syndrome.10–13 Low testosterone level (>10 nmol/L) in older men has been reported to be associated with a higher risk of mortality, whereas TT level of >12 nmol/L may increase the life expectancy and improve the quality of life. 14 The relationship between low testosterone levels and the development of depression, hypertension, obesity, lipid metabolism disorders, insulin resistance, and diabetes mellitus is of particular concern for middle-aged men, as LOH may contribute to the early onset of atherosclerosis and a high risk of adverse events.10,15–17
The LOH can be considered as a marker and even an independent CVD risk factor.11,18 Testosterone plays an important role in lipid, carbohydrate, and protein metabolism. It has been reported to produce a significant impact on the cardiovascular system, thereby becoming a clinical concern for biomedical society.10,11,14,19,20 The LOH is commonly accompanied by abdominal obesity, elevated triglyceride levels, and elevated cholesterol and glucose levels, leading to the development of insulin resistance and diabetes. The LOH is associated with arterial hypertension, which is generally considered as the leading CVD, contributing to high cardiovascular morbidity and mortality.20–22 Thus, LOH has similar components than that of metabolic syndrome. A few researchers have considered metabolic syndrome as a main cause of testosterone deficiency and an independent CVD risk factor.8,23 Metabolic syndrome has been reported to increase the risk of thromboembolism due to the hemostatic alterations related to increased circulating levels of plasminogen activation inhibitor-1 (PAI-1). 24 Insulin resistance may cause an imbalance between the production of nitric oxide, a physiological vasodilator, and the production of endothelin-1 (ET-1), a physiological vasoconstrictor. Nitric oxide production decreases, whereas ET-1 production increases. 25 On the other hand, a higher risk of thromboembolism in men with hypogonadism may be associated with hypofibrinolysis caused by androgen deficiency. Thromboembolism and myocardial infarction in men with hypogonadism have been recently reported to be mediated by low fibrinolytic activity at baseline. Hypogonadism in men has been associated with increased fibrinolytic inhibition due to increased synthesis of the plasminogen activator inhibitor PAI-1. 26 The effects of testosterone replacement therapy in LOH on cardiovascular risk appear to be associated with secondary polycythemia and a higher risk of thromboembolism. 27
Our study is aimed at assessing the prevalence of LOH and determining the relationships with cardiovascular risk factors in healthy middle-aged men.
Methods
The study was conducted in accordance with the principles of the Declaration of Helsinki. The Local Ethics Committee approved the study design, protocol, and the form of the informed consent. All participants provided the written informed consent before being recruited to the study. A total of 200 participants aged 44–60 years (the median age 48.44 [45.02–52.5]) referred to the general medical examination at the Federal State Healthcare Institution “Kemerovo Region Primary Healthcare Unit of the Ministry of the Internal Affairs of the Russian Federation” were enrolled in the study.
All participants underwent electrocardiography (The Schiller Cardiovit AT-1 G2 ECG machine), fluoroscopy (X-ray diagnostic complex MRC—“OKO”), blood tests (Mindray Hematology Analyzer), urine protein test (Uni-Test-BM), lipid profiling, and glucose testing (automatic biochemical analyzer ABX Pentra). All participants were examined by neurologists, otorhinolaryngologists, ophthalmologists, surgeons, dentists, psychiatrists, and general physicians. Anthropometric measurements, including the height, weight, and waist circumference, were collected from each participant. We calculated body mass index by using a weight-to-height ratio and the World Health Organization classification. 1 All participants were stratified by CVD risk, received the recommendations on lifestyle changes and nutrition, and were prescribed medical therapy.
Levels of TT were measured with enzyme-linked immunosorbent assay (ELISA) by using the automatic Evolis Twin Plus System analyzer. Reference values 12.1 nmol/L are higher. The average values of the optical density of the calibration samples were 1, 3, 1, 30, and 60 nmol/L. The laboratory normal range was 4.5–35.4 nmol/L for men. If TT level was <12.1 nmol/L, the measurement was repeated. Free testosterone (FT) was calculated by using an open-access software (www.csm4you.ru/kalkuljatory) according to the Vermuelen Formula. To calculate FT, sex hormone-binding globulin (SHBG) was measured at a fixed albumin of 4.3 g/dL. Serum levels of follicle-stimulating hormone (FSH) and luteinizing hormone (LH) were measured with ELISA by using the automatic Evolis Twin Plus System analyzer. A cut-off of 12.1 nmol/L for TT and 0.243 nmol/L for FT was used as laboratory criteria for LOH.5,7
All participants were asked to complete the Aging Male's Symptoms (AMS) questionnaire to measure health-related quality of life and symptoms in aging males.7,28 The AMS rating scale consists of 17 symptoms evaluated on a 5-score scale. The LOH symptom severity was defined as “no/little” (17–26 scores), “mild” (27–36 scores), “moderate” (37–49 scores), and “severe” (≥50 scores). The ED was assessed by the International Index of Erectile Dysfunction (IIEF-5). IIEF-5 ranges from 5 to 25 points, classifying ED into severe (5–7 points), moderate (8–11), mild to moderate (12–16), mild (17–21), and absence of ED (22–25). 29 Participants with a total AMS or ED score of at least 27 were considered to have LOH symptoms. The Beck Depression Inventory was used to evaluate depression and its severity. An index score of ≤9 is considered to be within normal range, whereas a score of 10 suggests the presence of depressive symptomatology from minimal to severe.
The LOH symptoms without a laboratory confirmed reduction in TT or FT were considered as manifestations of general somatic pathology.
All subjects were examined by a urologist to confirm LOH. We used the cut-off of 12.1 nmol/L for TT in diagnosing male hypogonadism. The LOH was diagnosed in case of positive laboratory criteria combined with associated clinical symptoms after excluding other causes. Asymptomatic low testosterone was diagnosed in the absence of clinical symptoms. 6
Statistical analysis was performed by using the commercially available IBM SPSS Statistics 25 software. The Shapiro–Wilk test was used to examine the data distribution. If variables were normally distributed, the Student's t-test was used to compare two sets of quantitative data. One-way analysis of variance was used to compare three or more independent groups. The Scheffé method was used to perform a one-step multiple comparison procedure. The Mann–Whitney U test was used to compare differences between two independent groups when the variables were not normally distributed. The Kruskal–Wallis test was used to compare three or more groups. In case statistically significant differences were detected, the Mann–Whitney U test was used for pairwise comparison. Contingency tables were used to compare qualitative variables in two independent groups. The Fisher's exact test was used for 2 × 2 tables, whereas the likelihood ratio was used for multiple columns. In both cases, the Cramer's V criterion was used to assess the strength of the relationship between the selected variables. The interpretation of the obtained values of statistical criteria was performed in accordance with the recommendations of Rea and Parker. 30 A p-value of 0.05 was considered statistically significant. Quantitative variables are presented as the mean and standard deviation (M ± σ).
Results
Ninety-eight participants (49%) reported TT levels below 12.1 nmol/L. After being consulted with an endocrinologist and excluding other causes of androgen deficiency, participants with confirmed LOH were assigned to the LOH group. Of them, 72 (73.5%) cases reported decreased levels of TT and FT. Twenty-six (26,5%) participants had an isolated reduction of TT levels, but FT levels were within the reference range. There were no cases of isolated reduction of FT levels. The mean TT level was significantly lower in the LOH group than in the group without LOH (8.9 ± 2.4 nmol/L vs. 22.5 ± 2.8 nmol/L, p = 0.000).
One hundred ten participants (55%) in the study population more often had ED and other symptoms measured with the AMS questionnaire than a decrease in testosterone levels. The LOH and asymptomatic low testosterone was confirmed by signs and symptoms, laboratory tests, and specialists' examinations in 42 (21%) and 56 participants (28%), respectively.
We divided the study population into two groups depending on the presence and absence of LOH. One hundred two men with a mean age of 46.43 ± 1.7 years with normal testosterone levels were assigned to Group 1 (non-LOH group). Ninety-eight men with a mean age of 46.26 ± 2.44 years and confirmed LOH were assigned to Group 2 (LOH group). Group 2 was then divided into Subgroup 1 with 42 enrolled participants presenting with LOH and clinical signs, and Subgroup 2 with 56 enrolled participants and confirmed asymptomatic low testosterone.
Clinical signs of LOH were associated with lower TT levels (Group 2 TT level of 7.78 ± 2.49 nmol/L vs. Group 1 TT level of 10.15 ± 1.32 nmol/L, p = 0.004).
Both groups reported significant differences in the mean levels of TT and FT. However, no differences were found in the mean levels of FSH and LH. Clinical signs of LOH were associated with the lowest mean levels of TT and FT according to the pairwise comparison in the subgroups (Table 1).
Mean Levels of Sex and Gonadotropin Hormones in the Study Subgroups
FSH, follicle-stimulating hormone; FT, free testosterone; TT, total testosterone.
Figure 1 reports the results of the AMS scale depending on the presence of LOH. The major proportion of participants in Group 1 reported no clinical symptoms or had mild to moderate symptoms. Alternatively, more than half of the participants in Group 2 suffered from LOH of varying severity. A relationship of good strength between the severity of clinical signs of LOH and reduced TT levels had been reported (Cramer's V criterion 0.301; p < 0.05).
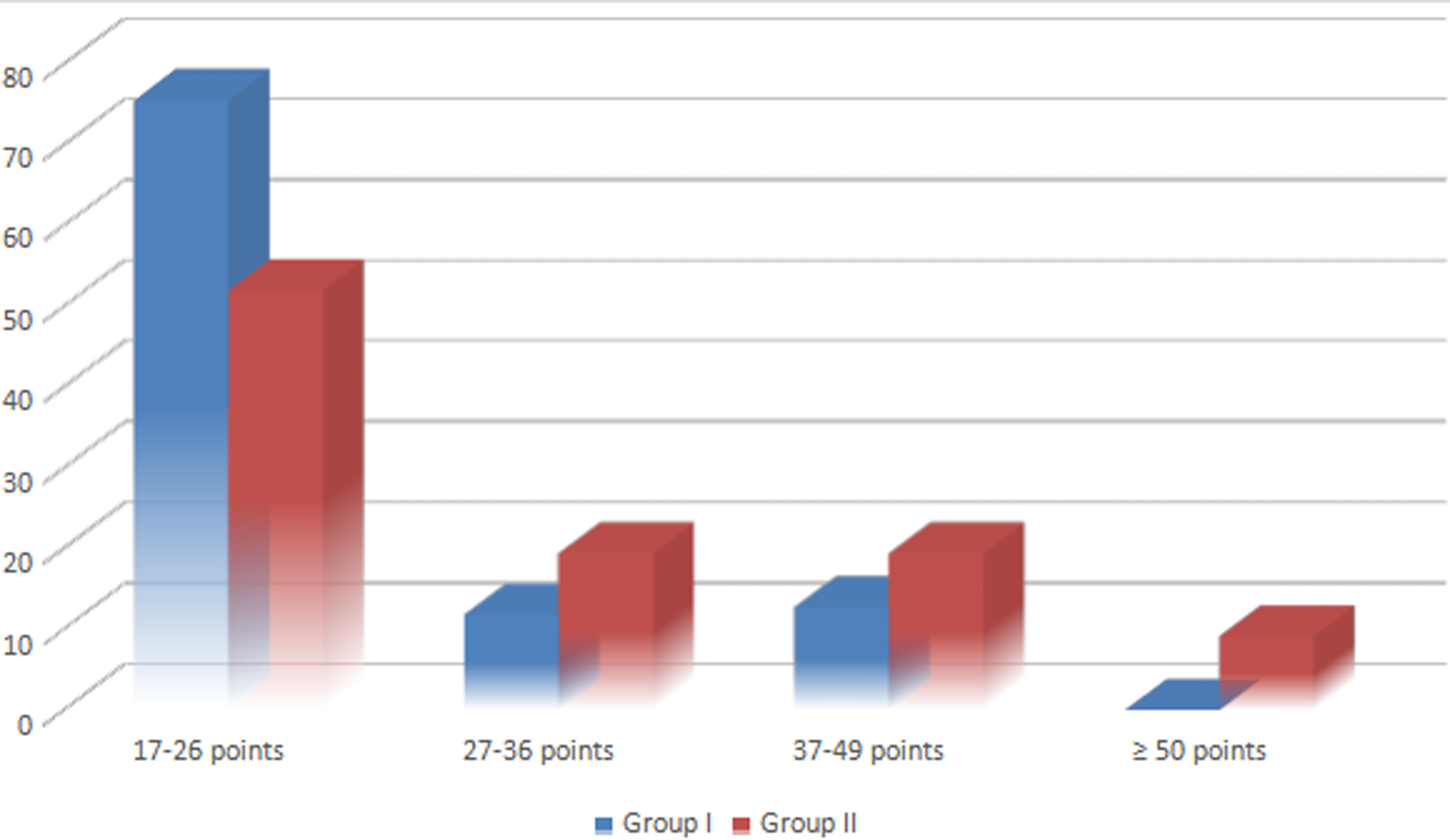
Results of the Aging Male's Symptoms scale.
Table 2 demonstrates the prevalence of ED and its severity among the study population. The mean IIEF-5 score was lower in the LOH group as compared with the non-LOH group (19.8 ± 2.4 vs. 21.5 ± 1.9, p = 0.012, respectively).
Results of the International Index of Erectile Dysfunction
IIEF-5, International Index of Erectile Dysfunction.
The highest proportion of participants in the non-LOH group had either no symptoms of ED, or mild ones. However, most of the participants enrolled in the LOH group had symptoms of ED, including moderate ones. There were no cases of severe ED in both groups. A relationship of moderate strength between the severity of ED and low testosterone levels had been reported (Cramer's V criterion 0.352; p < 0.05).
Thus, participants with ED and other symptoms measured by the AMS scale prevailed in Group 2. Men with either ED, or other AMS symptoms were less commonly found in Group 2. There were cases of asymptomatic low testosterone among men enrolled in Group 2.
To estimate the relationships between LOH with CVD risk, we analyzed the prevalence of CVD risk factors in Groups 1 and 2. Table 3 demonstrates the prevalence of CVD risk factors among the study population.
Prevalence of Cardiovascular Risk Factors in the Study Population
ABI, ankle-brachial index; GFR, glomerular filtration rate; IMT, intima-media thickness measured by duplex scanning of brachiocephalic arteries; LVH, left ventricular hypertrophy; MAU, microalbuminuria; MDRD, Modification of Diet in Renal Disease.
We found a high proportion of participants without positive CVD history who had high prevalence of CVD risk factors. Among these factors, lipid metabolism disorders and markers of subclinical lesions prevailed.
To estimate the relationships between LOH and CVD risk, the prevalence of CVD risk factors was assessed in both groups, with and without LOH (Table 4).
Prevalence of Cardiovascular Risk Factors in the Non-Late-Onset Hypogonadism (LOH) Group and LOH Group
Both groups had a similar prevalence of dyslipidemia and hypertension. Obesity, metabolic syndrome, and depression prevailed in Group 2 (Table 4). Relationships of good strength had been found between LOH and obesity, LOH and metabolic syndrome, and LOH and depression (Cramer's V criterion 0.269, p = 0.005; 0.316, p = 0.001 and 0.348, p = 0.0001, respectively).
In the present study, the study of insulin concentrations in blood plasma was not performed; however, when analyzing the level of fasting glycemia in Groups 1 and 2, no differences were found: 6.05 ± 1.53 versus 5.95 ± 1.39 mmol/L, p = 0.982.
Depression was commonly found in the LOH group and had the strongest relationship despite the severity of androgen deficiency (15.2% of severe depression cases in the LOH group vs. 0.0% of cases in the non-LOH group, p = 0.000). The relationship of good strength had been found between LOH and depression severity (Cramer's V criterion 0.518, p = 0.000).
CVD risk was assessed in all participants by using the Systematic Coronary Risk Evaluation (SCORE) scale to determine the associations between a decline in TT level and total CVD risk. Participants with moderate to high risk prevailed in the study population. There were a few high-risk participants; however, very high-risk participants were not reported. Importantly, high-risk participants were found only among men with LOH (p = 0.003; Fig. 2).
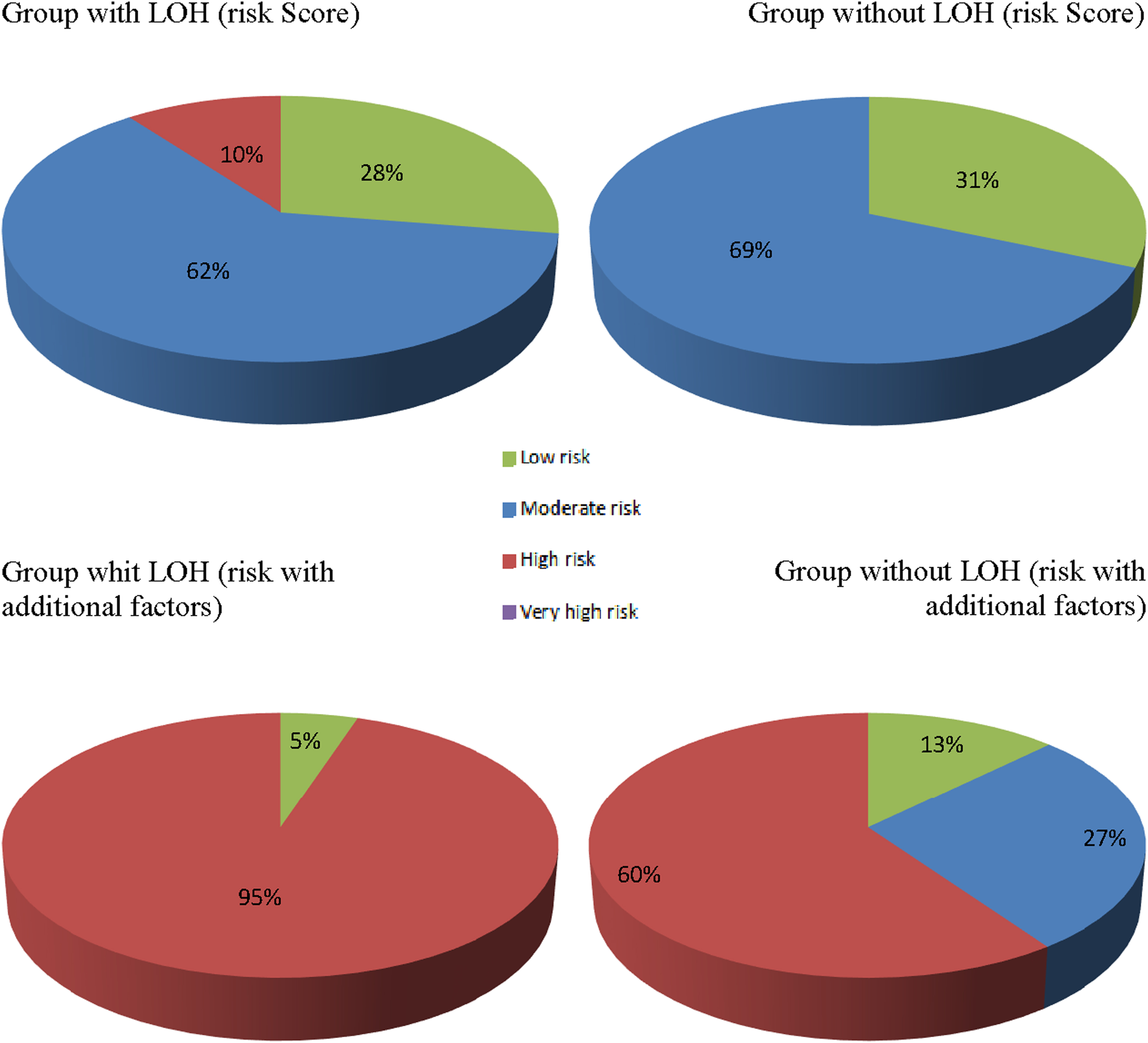
Cardiovascular disease risk prevalence among the study population according to the Systematic Coronary Risk Evaluation model and after the adjustment to additional risk factors (p = 0.001). LOH, late-onset hypogonadism.
However, recent evidence has suggested that the SCORE model does not fully predict CVD risk, since the impact of certain risk factors may be underestimated. These factors include high cholesterol, high blood pressure, obesity, metabolic syndrome, and markers of subclinical organ damage (decreased glomerular filtration rate [GFR], left ventricular hypertrophy, microalbuminuria, and signs of peripheral atherosclerosis). 31
After adjusting the resultant model to additional CVD risk factors, risk stratification in both groups changed toward men with high risk. Differences found between the groups became more pronounced in the LOH group, where the highest proportion of participants (94.9%) had a high risk of CVD (Fig. 2).
Discussion
The study on hypogonadism in males reported a prevalence of hypogonadism of 38% in men aged ≥45 years. 9 Recent studies have demonstrated a similar prevalence of LOH of varying severity in men aged 40–70 years, ranging from 30% to 40%. Our results have confirmed a high prevalence of LOH among healthy middle-aged men, which is generally consistent with the medical literature.8,9,15 Therefore, diagnosis and treatment of LOH remain an issue of concern for middle-aged men. Due to its multifactorial etiology, the clinical symptoms of androgen deficiency are nonspecific and can be even found in men with normal testosterone levels. 5 It seems reasonable that the diagnostic process should rely on the laboratory measurements of testosterone levels and require the exclusion of other possible causes of hypogonadism.5,7
We found that the mean levels of sex hormones are significantly lower in men with LOH than in men without it. Importantly, low testosterone levels may be found even in those participants who do not have any clinical signs of LOH. 7 Therefore, particular concern should be paid to timely identify low testosterone levels in middle-aged men even in the absence of clinical symptoms. The mean testosterone level that is even lower in the LOH subgroup than in the asymptomatic low testosterone subgroup has stated the relationship between the clinical severity of LOH and sex hormone levels. The etiopathogenesis of LOH includes the mechanisms underlying the development of both, primary and secondary hypogonadism.7–9 The absence of significant differences in gonadotropin levels in the study population has suggested the mixed etiopathogenesis of LOH. In addition, compensated androgen deficiency in the non-LOH group with normal levels of sex hormones that are maintained due to high levels of gonadotropins may also contribute to leveling the differences out between the groups in the mean gonadotropin levels.
There is no universal questionnaire for assessing the symptoms of LOH. The AMS questionnaire, being a widely used method for detecting clinical manifestations of LOH, has a sensitivity of 50–54% and a specificity of 40–41.2%. 32 However, this survey allowed us to detect 63% of participants with LOH. In addition, the relationship of good strength between the severity of symptoms and the presence of LOH was reliable.
One of the most specific symptoms of LOH is ED.4,5,33 However, we found that in some cases LOH was manifested only by ED, determined by the IIEF-5. The prevalence of ED in the LOH group was significantly higher, suggesting a significant contribution of decreased testosterone levels to the onset of ED. The detection of ED in both groups allows us to assume the presence of other causes, in addition to androgen deficiency. Psychological, neurological, vascular, and endocrine diseases as well as certain medications can cause ED. This argument may explain its detection among men who did not suffer from LOH. 34 Since ED has been recently considered as a marker of atherosclerosis, we may suggest that there are common pathogenetic mechanisms underlying the development of ED and LOH.
As ED and other clinical symptoms of LOH can be detected even without the decreased testosterone levels confirmed by the laboratory testing, none of them can be used independently for the diagnosis of LOH. On the other hand, a relatively high prevalence of subclinical LOH does not allow recommending the existing questionnaires for LOH screening.
Recent studies have reported the presence of associations between low testosterone levels and CVD, including hypertension and coronary artery disease. The effects of testosterone on heart and vascular functions and its association with CVD risk factors such as lipid and carbohydrate metabolism disorders, obesity, and depression have been also reported.10–12,14 We found rather weak and moderate signs of LOH that may explain the absence of the differences between the groups in lipid parameters. However, the literature data have stated the negative effects of LOH on lipid profile. 22 In addition, we have not found higher prevalence of dyslipidemia and hypertension in men with LOH.11,15 However, depression, obesity, and metabolic syndrome were associated with the presence of LOH, which could contribute to a higher cardiovascular risk in men with LOH. We did not measure the level of insulin and calculate the homeostasis model assessment of insulin resistance index, though the relationship between FT levels, SHBG levels, the risk of developing insulin resistance, and type 2 diabetes mellitus has been already reported. The Massachusetts Male Aging Study found odds ratios for incident diabetes of 1.58 for a 1 standard deviation decrease in free (ubound) T (4 ng/dL) and 1.89 for SHBG (16 nmol/L). 2 Thus, androgen deficiency in men is closely correlated with insulin resistance and hyperinsulinemia, and the administration of testosterone may improve it.
Whether low testosterone level may be considered as a risk factor or a marker for developing diseases associated with androgen deficiency remains unknown.11,18,22 The reported relationships between LOH and CVD risk factors may suggest their mutual potentiation with the formation of a so-called “vicious circle,” which requires active detection of LOH, as well as strict control of these factors in men with androgen deficiency.
The prospective cycle of this study will be devoted to the analysis of major adverse cardiac events in the study population after a 3-year follow-up.
Conclusion
The relevance of early diagnosis and management of LOH in middle-aged men may ensure the improvement of the quality of life, an increase of work capacity, and the ability to manage risk factors, resulting in a more efficient reduction of CVD morbidity and mortality. Therefore, timely diagnosis of testosterone deficiency by using TT levels is preferable, especially when patients suffer from ED, obesity, metabolic syndrome, and depression. If LOH is diagnosed, it will require medical therapy and a more aggressive approach to the modification of CVD risk factors should be adopted.
Footnotes
Authors' Contributions
N.B.L. has conceptualized, written, and revised the article. V.V.G. has performed data acquisition and analysis.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
No funding was received for this article.
