Abstract
Testosterone deficiency (TD) is common in men with obesity. The association between TD and obesity is bidirectional; low testosterone (T) is a contributing cause to obesity, and obesity is a contributing cause to low T, creating a vicious circle. Most guidelines recommend weight loss by diet/exercise as the first point of intervention to stop this vicious cycle. However, it requires a large amount of weight loss that is maintained over time. In clinical practice, this is rarely achieved by lifestyle interventions. Bariatric surgery is currently the main obesity treatment modality that results in a large amount of weight loss with decent weight loss maintenance. However, bariatric surgery is an invasive and expensive procedure, with risk for complications. Considering the high prevalence of TD in men with obesity, a more practical and sustainable obesity treatment for men is testosterone therapy (TTh). Thanks to the metabolic effects of T, TTh results in more fat loss and preservation of fat-free mass, compared with diet/exercise interventions alone. In contrast to weight loss achieved by diet/exercise and bariatric surgery, TTh significantly preserves both muscle and bone mass. Further, TTh has psychological effects that may increase the motivation and ability of men to adhere to diet/exercise programs. Real-world evidence studies of long-term TTh for up to 11 years provide compelling evidence that TTh holds tremendous potential as a new treatment modality for obesity in men, with long-term weight loss maintenance and health benefits far exceeding those achieved by lifestyle interventions, approved obesity drugs, as well as bariatric surgery.
Introduction
The prevalence and disease burden of obesity continues to rise worldwide.1–4 Among men, 40% have obesity. 5 Importantly, waist circumference (WC) in men is increasing more rapidly than body weight and body mass index (BMI). 6 Although both general and abdominal obesity are associated with an increased risk of premature mortality, 7 it has been shown that abdominal obesity may be a stronger risk factor for mortality than BMI.8–11
Cardiovascular disease (CVD), diabetes, and kidney disease are among the leading causes of obesity-related death and disability-adjusted life years (DALYs, a measure of overall disease burden, expressed as the number of years lost due to ill health or disability). 2 Developed countries are facing a public health crisis related to overweight and obesity, which has likely been a key driver of the reversal of the decline in CVD mortality rates in these countries. 12 In particular, the finding that overweight and obesity are affecting CVD mortality in younger age cohorts where lifetime exposure to obesity has been much shorter than in older cohorts are very concerning and are likely to adversely impact CVD mortality trends and hence life expectancy in future. 12
A large meta-analysis of prospective studies in four continents showed that men with obesity had three times the death rate than women with obesity. 1 This is consistent with previous observations that, at equivalent BMI levels, men have greater insulin resistance, ectopic (e.g., liver) fat mass, and prevalence of type 2 diabetes mellitus (T2DM). 13 The greater obesity-related disease burden in men was confirmed in a recent analysis of the Global Burden of Disease Study, which showed that global deaths and DALYs attributable to obesity are significantly higher in men than women (aged <70 years old). 14
The aim of this review is to summarize evidence regarding the need for sex-specific interventions to treat obesity in men, focusing on the link between obesity and testosterone deficiency (TD) and the clinical utility of long-term testosterone therapy (TTh) as a treatment for obesity in men.
The Need for Sex-Specific Interventions to Treat Obesity in Men
The influence of sex hormones on metabolism and regulation of energy balance, body composition, body fat distribution, and appetite is well established. 15 Sex has emerged as a significant predictive factor in the development of CVD associated with metabolic dysregulation, such as obesity. 16 Even though the prevalence of obesity (BMI ≥30 kg/m2) is slightly higher in women than men, men suffer from more obesity-related comorbidities and are at a higher risk of obesity-related premature death.1,17–19
A study that investigated patterns of weight regain among men and women during a long-term follow-up found that men have poorer weight loss maintenance than women. 20 The Look Action for Health in Diabetes (AHEAD) trial likewise found sex differences in response to an intensive lifestyle intervention (ILI); although both men and women lost body fat and lean mass compared with the control group at year 1, by year 8, men in the ILI group had regained all body fat but not the lost lean mass, so that there was no difference in body fat but a significantly reduced lean mass compared with the control group at study end. In contrast, women in the ILI group had a significantly reduced amount of body fat but no difference in lean body mass compared with the control group at year 8. 21
Sex-specific activation of the androgen receptor in the hypothalamus, skeletal muscle, liver, adipose tissue, and pancreatic islet β cells accounts for maintenance or disruption (in the case of TD) in energy metabolism and glucose homeostasis in men. 22 Therefore, the androgen receptor is a logical target for obesity interventions in men. However, there is a paucity of clinical trials examining sex-specific interventions and outcomes of obesity treatments (lifestyle-based or pharmacological). 15 The study of sex differences in obesity and regulation of energy expenditure, appetite, and body composition is an area rich with clinical research opportunities. As countries around the world struggle to deal with increasing population obesity, it will be important to focus on sex differences when designing obesity interventions to achieve improved health and economic outcomes. 15
There are also psychological differences between men and women, which have implications for obesity treatments. For instance, it seems that men have more difficulty perceiving they have obesity, because of the desire to be muscular and have the masculinity of a large body size. 23 Unsurprisingly, a key motivator for men to start a weight loss treatment is an actual diagnosis of obesity, as opposed to simply being overweight. 23 Men from various cultures reported that being overweight did not concern them, and in some cases represented an ideal weight, as they did not want to be “too thin.” 23 Further, men are more reluctant than women to undergo strict diets, and prefer to attend “men-only” treatments. 23 Effective obesity interventions for men, therefore, need to frame messages that are suited to the male mindset and appeal to the masculinity ideal. One male-specific benefit of weight loss is reduction in erectile dysfunction (ED). 24 The ability to perform sexually can, in turn, boost men's self-confidence and motivation to adhere to weight loss programs. Therefore, obesity interventions in men should not only focus on weight loss; they should also place greater emphasis on boosting sexual function/performance, and pleasure will likely provide far better adherence and sustained health outcomes.
As explained in the remaining sections of this review, long-term TTh qualifies as a highly effective treatment for obesity, resulting in marked weight loss/body fat reduction even without dietary restriction, and preservation of lean (muscle) mass. Being well known as the “male sex hormone,” TTh is obviously a “men only” treatment. With its well-documented effects on muscle gain and sex drive/function, TTh is uniquely positioned to appeal to the masculine desires of men, while curbing an epidemic public health threat.
Obesity (BMI ≥30 kg/m2) Versus Abdominal Obesity (WC ≥94 cm)
Clinical guidelines diagnose obesity as BMI ≥30 kg/m2.25–27 This is problematic for several reasons. BMI does not distinguish between body fat mass and muscle mass, which have an opposite impact on health outcomes. In addition, BMI does not provide information about abdominal obesity, which is a greater risk factor for premature atherosclerosis, heart disease, and mortality than overall obesity. 28 It is, therefore, alarming that the prevalence of abdominal obesity in the NHANES III survey is exceeding that of BMI-defined obesity (≥30 kg/m2). 29
The WC is the most common measure of abdominal obesity. For Caucasian/Europid populations, medical organizations recommend the WC thresholds of ≥94 cm (37 inches) in men and ≥80 cm (31 inches) in women for diagnosing abdominal obesity. 30 Waist size typically increases with increasing body weight, whereas abdominal obesity can occur even in normal weight (BMI 18.5 to 24.9) people. 31 In fact, the prevalence of normal weight abdominal obesity is increasing, now affecting at least one in three adults. 32
The WC is more strongly associated with all-cause7,9,11,33 and heart disease mortality33,34 than BMI. Importantly, abdominal obesity is associated with higher mortality risk, independent of BMI.8–11 For any given BMI category, apparently healthy individuals with a larger WC have an increased risk of premature mortality compared with those with a smaller WC.8–11 Even among individuals with normal weight, abdominal obesity is associated with higher mortality than BMI-defined obesity. 31 Therefore, when interpreting outcomes of TTh studies, changes in WC are more clinically meaningful than changes in body weight (BMI).
Bidirectional Link Between Obesity and TD
It is well documented that the association between obesity and TD is bidirectional.35,36 Obesity is the most common and strongest risk factor for TD, more so than aging itself (as explained below).37–40
Meta-analysis evidence that obesity is a cause of TD comes from studies of weight loss (induced by either low-calorie dieting or bariatric surgery) that show that increases in T levels are proportional to the amount of weight lost. 41 However, it should be pointed out that a large degree of sustained weight loss is required to elevate T levels that are enough to be of clinical significance. In men with TD, diet-associated weight loss leads to modest increases in T, 2.87 nmol/L (83 mg/dL) with 10% loss of body weight. 41 Bariatric surgery (30% loss of body weight) is associated with a more marked increase in total T of 8.73 nmol/L (252 mg/dL), 42 and it has been suggested that this may contribute to the health benefits seen in men after bariatric surgery. 41
A Mendelian randomization analysis confirmed the causal effect of BMI on serum T in men; weight loss resulting in a reduction of BMI from 30 to 25 kg/m2 was estimated to correspond to a 13% increase in serum T. 43 Similar results were found in the prospective Massachusetts Male Aging Study (MMAS). 37 Although normalization of T levels is a possible mechanism contributing to the beneficial health effect of bariatric surgery in excessive obesity, the increase in endogenous T levels with nonsurgical weight loss interventions is minor and of questionable clinical significance. Support for this comes from a randomized controlled trial (RCT) of TTh plus diet in men with obesity; a rigorous diet program leading to 11 kg (24 lb) weight loss in the placebo group increased serum T by only 2.9 nmol/L (84 mg/dL). 44 Not surprisingly, there was no improvement of hypogonadal symptoms, 45 and this effect was not sustained 18 months after the trial. 46
These data suggest that although obesity is a strong cause of TD, efforts to treat TD by typical diet/exercise weight loss interventions are futile.
There is also solid evidence supporting the reverse direction that low T is a causal contributor to development of obesity. Experimental induction of hypogonadism in healthy men aged 20–50 years significantly increases body fat mass already after 12 weeks 47 and 16 weeks, 48 and men with prostate cancer (PCa) receiving androgen deprivation therapy show marked increases in total body fat mass and abdominal visceral fat within 6 months. 49 A prospective study of 3351 community-dwelling men showed that men with low T had higher BMI, WC, and risk of metabolic syndrome (MetS) after a 10-year follow-up. 50 Interestingly, men with higher baseline T levels not only had the lowest BMI, WC, and risk of MetS, but they also had the lowest risk of incident CVD events—irrespective of physical activity level—and had the lowest risk of dying from CVD. 50 Low T is a particularly strong predictor of the development of central adiposity 51 with visceral fat accumulation. 52 Another prospective study specifically examined the association between baseline T levels and visceral fat accumulation (measured by computed tomography) after a follow-up of 7.5 years. 52 Men with low T at baseline had a significantly greater increase in visceral fat, even after adjustment for baseline visceral and subcutaneous fat mass, BMI, age, T2DM status (oral glucose tolerance test by the World Health Organization diagnostic criteria), and fasting C-peptide. 52 A real-world evidence (RWE) study of men with TD and various body weight categories investigated the effect of long-term TTh for up to 11 years. 42 All men in the control group, regardless of baseline weight status, experienced a marked increase in body weight and WC. 42 Importantly, the greatest increases in body weight and WC occurred after 6 years in men with normal weight and overweight at baseline. As expected, T levels significantly declined over time, from 9.5 to 8.5 nmol/L in the normal weight, from 9.6 to 8.2 nmol/L in the overweight, and from 9.8 to 7.7 in the obese groups, respectively. 42 This suggests that the causal role of TD in the development of obesity takes multiple years to fully manifest.
Further proof of the causal role of TD in the pathogenesis of obesity comes from a growing number of studies showing that TTh significantly reduces markers of obesity (including body weight, WC, waist-to-height ratio, and BMI),42,53–55 total body fat mass,47,48,56–61 and intra-abdominal fat mass.61–64 The remaining sections in this review will summarize important studies regarding the potential use of TTh as a treatment for obesity in men with TD.
Obesity Is More Strongly Linked to Low Testosterone than Age
A common misperception is that advancing age is the main cause of TD. Although the prevalence of TD increases with advancing age, this is mainly due to comorbidities—particularly obesity and T2DM—rather than aging per se.37,65–72 As a consequence of the growing obesity epidemic among all age groups, the prevalence of TD in young men is increasing. 73 A meta-analysis of 18 studies (comprising a total of 4546 men with obesity, age ranging from 27.9 to 61.9 years, and BMI ranging from 24.8 to 50.3 kg/m2) found a TD prevalence as high as 50–80%. 74 Studies using total T and free T showed the same trend of increasing prevalence of TD with increasing BMI. 74
The Coronary Artery Risk Development in Young Adults (CARDIA) study showed that increasing obesity, particularly abdominal obesity, is associated with decreasing T levels in young men aged 18–30 years.75,76
A report from the Centers for Disease Control showed that among men, the prevalence of obesity was 40.3% in those aged 20–39, 46.4% among those aged 40–59, and 42.2% among those aged 60 and older. 73 In other words, obesity is nearly as common in younger men as in older men. Not surprisingly, up to 33–58% of young men (age 35 years or younger) with obesity have TD.77–79 Another study found that among young men (aged 33–45 years) with obesity attending primary care, the prevalence of TD was as high as up to 75%. 80 This is similar to the TD prevalence seen in older men with obesity.81–84
The MMAS showed that an increase in BMI of 4–5 kg/m2 was associated with a reduction in T levels comparable to that seen during ∼10 years of aging in men whose body weight remained unchanged. 37 In the European Male Aging Study (EMAS), obese men (BMI ≥30 kg/m2) had a 30% lower T level—equivalent to almost three decades of aging—and a 13-fold increase in TD prevalence compared with men with a BMI of <25 kg/m2.38,39 Prospective data from EMAS showed that a 10% weight gain (+12.3 kg) and 10% weight loss (−13.7 kg) was associated with a proportional decrease (−2.4 nmol/L) and increase (+2.9 nmol/L) in T levels during a 4-year follow-up, respectively. 40 Obesity is associated with low T levels even in young men aged 24–41 years, a finding that supports the notion that obesity is a stronger risk factor for TD than age itself. 75
Facing the current epidemic of obesity, it is critical for health care professionals to know that body fatness is more strongly associated with low T levels than age.29,72,85 Therefore, T levels should be measured in men with excess body fat and/or large waist size, regardless of age, and TTh should be offered to all men who have no contraindications. 26
Clinical Utility of TTh as a Treatment for Obesity: Long-Term RWE
The RWE from long-term observational studies have provided compelling support for the use of TTh as a sustainable and feasible treatment for obesity.42,53–55,86–93 These observational studies comprise men with TD and various degrees of obesity and weight classes, ranging from normal weight to obesity class III, who are receiving long-term treatment with T undecanoate injections in real-life urology practices.
Because the health benefits of TTh are time dependent, these RWE studies provide valuable data about the true clinical significance of TTh, which cannot be derived from RCTs due to their short-term nature. The longest duration TTh RCTs are 3 years. In contrast, RWE data on metabolic/sexual, skeletal, and prostate outcomes have been continuously collected for 12 years, 93 6 years, 94 and 17 years, 95 respectively.
One RWE study examined the effects of long-term TTh on anthropometric and metabolic parameters in 411 men with TD (mean age 60 years) and different degrees of obesity: class I (BMI 30–34.9 kg/m2; n = 214), class II (BMI 35–39.9 kg/m2; n = 150), and class III (BMI ≥40 kg/m2; n = 47). TTh was given as T undecanoate injections in 12 weeks intervals for up to 8 years. 55 In all three classes of obesity, TTh resulted in significant weight loss and a decrease in WC and BMI (Table 1). In men with class I and class II obesity, BMI dropped to below 30 kg/m2, hence moving men from the obese state to the overweight state (Table 1). 55
Changes in Obesity Parameters After Testosterone Therapy for up to 8 Years
Data from: Saad et al. 55
When interpreting elevations in T levels (total and free), one has to keep in mind that these reported levels are trough levels, i.e. the lowest level measured immediately before the next injection. Trough levels can serve as an indicator of whether therapeutic T levels have been achieved..
BMI, body mass index.
In a 10-year follow-up of 115 patients who had been continuously treated with T undecanoate injections, BMI decreased from 31 to 27 kg/m2. The decrease was significant versus baseline (p < 0.0001) and significant versus previous year for the first 8 years. 87 Body weight decreased from 97.3 to 84.6 kg. The weight loss was progressive and reached 18.5% (range 6.19% to 31.97%) after 10 years. At baseline, nearly all men (97.4%) had an elevated WC (≥94 cm), out of which 67% had a substantially elevated WC (≥102 cm). Mean WC decreased progressively from 106.5 to 92.3 cm (12%). The decrease was significant versus baseline (p < 0.0001) and significant versus previous year for the first 7 years. 87
Interestingly, men with moderate-to-severe ED or lower urinary tract symptoms have a more severe cardiovascular (CV) risk profile and benefit more from TTh than men with mild symptoms.91,93 In men with obesity and various degrees of ED, treatment with T undecanoate injections for up to 12 years reduced body weight by ∼18 kg (−16%), WC by 10 cm, and BMI by 6 kg/m2 (from 33 to 27 kg/m2). 93 Considering the high prevalence of ED in men with obesity, it is remarkable that erectile function continued to significantly improve for each successive year for 9 years. 93
A more recent analysis of RWE data examined the effect of TTh for up to 11 years in men (n = 823) with various weight classes. 42 Of these men, 474 (57.6%) had obesity, 286 (34.8%) overweight, and 63 (7.7%) normal weight. In 428 men (281 with obesity, 121 with overweight, and 26 with normal weight), T undecanoate 1000 mg injections were administered every 12 weeks after an initial 6-week interval, for up to 11 years. The remaining 395 men served as a control group. Anthropometric and metabolic parameters were measured at least twice yearly, and changes were adjusted for confounding factors to account for baseline differences between groups. In the normal weight group, T treated men had a weight loss of −3.4 kg (−4.8%), whereas untreated men had a weight gain of 6.1 kg (+8%). In the overweight group, T-treated men had a weight loss of −8.5 kg (−9.6%), whereas untreated men had a weight gain of 6.0 kg (+6.9%). In the obese group, T-treated men had a weight loss of −23.2 kg (−20.6%), whereas untreated men had a weight gain of 4.2 kg (+5.1%). Corresponding changes were seen in WC and BMI. The WC decreased in T-treated men (increased in untreated men) by −3.4 cm (+5.6 cm), −4.7 cm (+5.5 cm), and −12.9 cm (+5.6 cm) in the normal weight, overweight, and obese groups, respectively. The differences in percent body weight change between groups, after adjustments for confounding factors, became significant in the normal weight group after year 1, and in the overweight and obese groups after year 2 (Fig. 1A). The differences in WC change between groups, after adjustments for confounding factors, became significant in the normal weight group after year 5, in the overweight group after year 2, and in the obese group after year 1 (Fig. 1B). The changes in body weight and WC were accompanied by corresponding changes in lipids and glucose control (improvement in T-treated men and worsening in untreated men).

In another RWE analysis, 316 men with prediabetes (defined as hemoglobin A1c [HbA1c] 5.7–6.4%) and TD (defined as total T levels ≤12.1 nmol/L combined with symptoms) were included. 54 Of these, 229 men received T undecanoate injections for 8 years, and 87 men served as untreated control subjects. Metabolic and anthropometric parameters were measured twice yearly. At baseline, in both groups, BMI, HbA1c, and International Index of Erectile Function, Erectile Function Domain (IIEF-EF) were 30 kg/m2, 5.9%, and 11, respectively. TTh led to substantial improvement in glycemic parameters, with significant reductions in fasting blood glucose and HbA1c. In contrast, in the untreated group, glycemic parameters worsened over time. At the last observation, all 229 patients (100%) in the TTh group had an HbA1c of <6.5% (48 mmol/mol), and 205 of these 229 patients (90%) achieved normal glucose regulation with an HbA1c <5.7% (39 mmol/mol). In the untreated group, only 1 patient (of 87, i.e., 1%) had HbA1c <5.7% (39 mmol/mol) whereas 35 men (40.2%) had progressed to frank T2DM with HbA1c >6.5% (48 mmol/mol). 54 Men in the TTh group achieved a weight loss of 8% at 8 years, whereas untreated men experienced a weight gain of 9%. This corresponded to a weight loss of 9.2 kg in T-treated patients and a weight gain of 8 kg in the untreated group. The WC decreased by 6.8 cm in the TTh group whereas it increased by 7.4 cm in the untreated group. 54 This is the first study to show that long-term TTh not only completely prevents progression of prediabetes to frank T2DM but also restores normal glucose regulation in most men.
Another RWE analysis of 356 men with T2DM and TD examined whether long-term TTh can result in remission of T2DM. 92 Remission of T2DM was defined as HbA1c <6.5% and discontinuation of all diabetes drugs, including metformin. All patients received standard T2DM treatment. One hundred seventy-eight men additionally received treatment with T undecanoate injections 1000 mg every 12 weeks after an initial 6-week interval. The remaining 178 men did not receive TTh and served as a control group. 92 The T-treated men had a progressive and sustained reduction in body weight of −22.1 kg (−19.3%) and WC of −13.3 cm. In contrast, the control group had an increase in body weight and WC of +6.8 kg (+7.4%) and +7.1 cm, respectively. In parallel with the reduction in body weight and WC, T-treated men had significant progressive and sustained reductions in fasting glucose, HbA1c, and fasting insulin throughout the entire treatment period. In contrast, the control group had progressive elevations in fasting glucose, HbA1c, and fasting insulin. The T-treated men who had been on insulin at baseline had a reduction in insulin dose requirement compared with the control group, suggesting improved β cell function. In contrast, 20 patients in the control group were started on insulin treatment during the observation time, suggesting deteriorated β cell function. Remarkably, among T-treated men, 34.3% (61 out of 178 patients) achieved remission of T2DM (22 of these patients had been on insulin at baseline). The average time to discontinuation of diabetes medications was 8.6 years; the average time in remission was 2.5 years. There were no relapses. Overall, 46.6% of patients achieved normal glucose regulation (these patients, if not yet in remission, are eligible for discontinuation of diabetes drugs, which will be explored in an upcoming update of this ongoing study), 83.1% reached the HbA1c target of 6.5%, and 90% reached the HbA1c target of 7.0%. In contrast, no remission of T2DM or reduction in glucose or HbA1c levels was noted in the control group. 92
Importance of Uninterrupted TTh and Achievement of Therapeutic Testosterone Levels
A prerequisite for achievement of maximal metabolic and body composition benefits (loss of excess body fat with preserved or increased fat-free mass [FFM]) of TTh is long-term uninterrupted treatment. Multiple studies have shown that the beneficial effects of TTh are not maintained if treatment is discontinued.46,53,96–100 This applies not only to improvements in body composition but also to improvements in muscle strength, erectile function, HbA1c, total cholesterol, low-density lipoprotein cholesterol (LDL), high-density lipoprotein cholesterol (HDL), triglycerides, AMS, IPSS, IIEF-EF, residual voiding volume and bladder wall thickness, quality of life, and likely most—if not all—other T-related outcomes.46,53,96–100 Nevertheless, beneficial effects return when TTh is resumed. 98 As pointed out in the British Society for Sexual Medicine guidelines on Adult Testosterone Deficiency, cessation of TTh results in progressive reappearance of symptoms and reversal of benefits (commonly within 6 months), so TTh is likely to be required lifelong for persistent symptom resolution and maintenance of health benefits. 65
A related issue occurs when TTh is not properly dosed to achieve a high enough elevation of T levels (within the physiological range) for therapeutic efficacy. There are different T threshold levels for different responses to TTh; hence, TTh needs to achieve sustained therapeutic T levels to be effective. 101
The Hormonal Regulators of Muscle and Metabolism in Aging (HORMA) Trial found that increases in total T of 1046 ng/dL (95% confidence interval [CI] = 1040–1051) and 898 ng/dL (95% CI = 892–904) were necessary to achieve median increases in lean body mass of 1.5 kg and appendicular skeletal muscle mass of 0.8 kg, respectively, which, in turn, were required to significantly enhance one-repetition maximum strength. 102 Therefore, for maximal benefits the dose of TTh should be titrated to achieve target T blood levels. 102
The correlation between declining T levels during TTh and suboptimal response can be seen in several studies. For instance, in a long-term TTh study, body fat % dropped from 31.5% at baseline to 26.5% at month 24. 103 This was accompanied by an increase in total and free T levels from 9.1 to 19.1 nmol/L (263 to 551 ng/dL) and from 6.2 to 14.3 ng/dL, respectively. However, at month 96, total and free T levels had dropped to 372 and 8.68 ng/dL respectively, with body fat % no longer being significantly different from baseline, 103 suggesting inadequate dosing/adherence to TTh. Similarly, other studies have found that men who receive TTh that fails to normalize (restore) T levels (due to inadequate dose or adherence) do not experience a reduction in myocardial infarction (MI), stroke, atrial fibrillation or all-cause mortality, which is seen in men who receive TTh that does normalize (restore) T levels.104–106 Suboptimal TTh also occurs in randomized controlled trials. In the Testosterone's Effects on Atherosclerosis Progression in Aging Men (TEAAM) trial, 156 men were randomized to receive TTh and 152 were randomized to receive placebo for 3 years. Despite aiming at achieving T levels between 500 and 900 ng/dL, at 6, 18, and 36 months total T levels were ∼640, 600, and 460 ng/dL, respectively, and free T levels were 12, 11, and 8.5 ng/dL, respectively.
Because target T blood levels (T thresholds) likely vary between individuals, it is important to regularly monitor T levels during TTh, and in cases of suboptimal or lack of response, increase the TTh dose for a sufficient length of time and then re-evaluate.107,108 In a secondary analysis of the BLAST study, examining T levels associated with clinical and biochemical improvements, it was pointed out that men with severe obesity may need shorter dosing intervals of T undecanoate—around 10 weeks between injections as opposed to 12 weeks—to achieve ideal therapeutic T levels, especially if the target levels of >15 nmol/L recommended in clinical guidelines are to be reached within 3–6 months. 101
TTh in Comparison to Traditional Obesity Treatments
To fully appreciate the potential of TTh as an obesity treatment modality, it is informative to compare the weight loss and changes in body composition achieved with TTh with those achieved in traditional obesity interventions. A meta-analysis of weight loss trials reported a mean weight loss of 5 to 8.5 kg (5% to 9%) during the first 6 months from interventions with reduced-energy diets and/or weight-loss medications, with weight loss plateaus commonly occurring at ∼6 months. 109 A systematic review of long-term weight loss studies found that diet/lifestyle interventions result in <5 kg weight loss after 2–4 years and obesity drugs result in 5–10 kg weight loss after 1–2 years. 110 After completing structured weight-loss programs, subjects regained half of the lost weight after 1 year and nearly three quarter during the first three years; less than 3% of subjects maintained their weight loss at all annual visits for 4–5 years after completion of a weight-loss program.20,111,112 Meta-analysis shows that people on average maintain a weight loss of 3 kg and a reduced weight of 3% of initial body weight after 5 years. 113 Obesity medications approved for long-term use, when prescribed with lifestyle interventions, produce additional weight loss relative to placebo ranging from ∼3% to 9% of initial weight at 1 year. 114 However, a major limitation of diet/exercise and obesity drug interventions is high dropout and a large proportion of subjects lost to follow-up.110,115
The Look AHEAD trial showed that an ILI resulted in a weight loss of −6% after 4 years. 116 In the RWE study of TTh in men with various severity of obesity (summarized above), weight loss at year 4 ranged from −10% to −15%, depending on obesity grade. 55 An 8-year analysis of The Look AHEAD trial (baseline BMI of 35 kg/m2) reported a weight loss of 7.9, 3.7, and 4.0 kg for year 1, 4, and 8, respectively. 21 The corresponding weight loss in men on long-term TTh was 3, 10.7, and 17.4 kg in class I obese men, 3.8, 16.6, and 25.3 kg in class II obese men, and 3.5, 18.5, and 30.5 kg in class III obese men (Table 2). 55
Long-Term Weight Loss with Testosterone Therapy Compared with Intensive Lifestyle Intervention a
AHEAD, Action for Health in Diabetes.
As shown in Table 2, there was a marked weight regain after the first year in the ILI. In contrast, long-term TTh resulted in a sustained and progressive weight loss, with no plateau or weight regain. After 8 years, weight loss was four- to eight-fold greater with TTh than ILI.
The Look AHEAD trial, which is the longest duration ILI with body composition data, clearly shows a marked loss of FFM. 21 In a sex-specific analysis, differences in fat mass among men in the intervention and control groups were not significant at year 8; however, men in the intervention group had a significantly greater loss of FFM at all time points. 21 From baseline to year 1, fat mass and FFM did not change in men in the control group, whereas men in the intervention group had a weight loss of 9.4 kg, of which nearly 3 kg (30%) was FFM. From year 1–8, there was little change in the fat mass and FFM in the control group, and FFM in the intervention group. However, the year 1–8 weight gain in men (as well as women) in the intervention group was ∼100% fat mass. 21
As of this writing, there is no long-term TTh trial with body composition data that included a control group. Nevertheless, body composition trials of TTh (summarized below) show that TTh results in a greater reduction in fat mass per unit weight loss (in other words, preservation of FFM) compared with diet/exercise interventions. The large reduction in WC seen in the RWE studies outlined earlier confirms the beneficial effect of TTh on body composition and body fat distribution.
Weight loss is only one aspect of obesity treatment. Perhaps more important is the issue of weight loss maintenance. Weight-loss programs based on diet, exercise, and obesity drugs can help people lose weight, whereas maintaining weight loss seems to be the greatest challenge.117–119 Even when obesity drugs are used during the maintenance phase, the vast majority regain most of the lost weight. 120 Considering that long-term TTh results in a progressive sustained reduction in obesity markers, combined with data showing that men may be more likely to regain weight after weight loss programs,20,121 TTh holds tremendous potential as a unique obesity treatment for men.
TTh in Comparison to Bariatric Surgery
Among approved obesity treatments, bariatric surgery results in the greatest long-term weight loss maintenance,122,123 with an average reduction in body weight after 2, 10, 15, and 20 years in the magnitude of −23%, −17%, −16%, and −18%, respectively. 122 A meta-analysis showed a weight-loss maintenance of 22.2% (30.1 kg) 20 years after bariatric surgery. 123 In a multicenter longitudinal study of 2348 bariatric surgery patients, after roux-en-Y gastric bypass (RYGB) and laparoscopic adjustable gastric banding (LAGB) the 7-year mean weight loss was 28.4% (−38.2 kg) and 14.9% (−18.8 kg), respectively. 124 Between years 3 and 7, there was a mean weight regain of 3.9% (RYGB) and 1.4% (LAGB). Among RYGB patients, 75% maintained at least 20% weight loss, and 50% of LAGB patients maintained at least 16% weight loss through 7 years. The remission of T2DM at year 7 was 60% for RYGB and 20.3% for LABG. 124
Compared with usual care, bariatric surgery is associated with a long-term reduction in overall mortality [adjusted hazard ratio (HR) = 0.71, 95% CI 0.54–0.92; p = 0.01] and decreased incidence of T2DM (adjusted HR = 0.17; p < 0.001), MI (adjusted HR = 0.71; p = 0.02), and stroke (adjusted HR = 0.66; p = 0.008). 122 One study found that remission of T2DM after bariatric surgery was achieved in 58.2% (n = 2090) of patients at 2 years, and in 46.6% of patients at 5 years. 125 Although T2DM remission can be as high as 74% at 1 year postsurgery, T2DM relapse increased over time with a longer follow-up duration.125,126
As summarized earlier, TTh for 8 years results in weight loss corresponding to 16.8% to 23.6%, depending on baseline obesity grade (Table 1). 55 An 11-year analysis of RWE data showed that men who had been receiving TTh had a weight loss of −23.2 kg (−20.6%), whereas untreated men had a weight gain of 4.2 kg (+5.1%). 127 The marked weight loss seen during long-term TTh is accompanied by a reduced incidence of T2DM 54 and remission of T2DM in 34.3% of patients. 92 In an RWE analysis, the average time to discontinuation of diabetes medications (i.e., achievement of T2DM remission) was 8.6 years. 92 During a follow-up time of 2.5 years, there were no relapses. 92 Longer-term follow-up of this RWE cohort will tell whether T2DM relapse will occur. Currently, data suggest that with increasing the duration of TTh, the greater the rate of T2DM remission, that is, opposite to that seen with bariatric surgery.
As outlined later, TTh also compares favorably regarding long-term reduction in overall mortality, MI, and stroke. Although bariatric surgery results in greater short-term (<2 years) weight loss and T2DM, it is an invasive and very expensive procedure. Due to its risk for complications and high cost, it is only an option for a small minority of men with obesity and will hence not help curb the global obesity epidemic.
Effect of Traditional Obesity Treatments and Bariatric Surgery on Bone Mineral Density and Fractures
In addition to loss of FFM (discussed in the next section), another negative effect of diet-based obesity intervention128,129 and bariatric surgery is significant loss of bone mineral density (BMD). Studies of both diet-induced weight loss and bariatric surgery show changes in bone markers, reflecting an increased bone turnover, evidenced by increased urinary excretion of deoxypyridinoline and serum levels of osteocalcin. 130 A 30-month follow-up study of a 1-year lifestyle intervention trial found a significant progressive reduction in total hip BMD despite lack of change in FFM or appendicular lean mass. 131 Weight loss of 10% or more beginning at age 50 years increases the risk of hip fracture in older men. 132 Among patients with T2DM, a weight loss of ≥20% is a significant risk factor for fractures, especially for men. 133 The Osteoporotic Fractures in Men (MrOS) study demonstrated that the impact of weight loss in older men on rates of bone loss is increased in the presence of low T levels. 134 Support for this comes from another MrOS analysis showing that low free T is an independent predictor of low BMD and increased prevalence of fractures. 135
Bariatric surgery appears to be a particularly strong risk factor for BMD loss136,137 and fractures.138–140 A 7-year prospective study after RYGB (baseline mean age 43 ± 8 years, BMI 42 ± 6 kg/m2) found continuous decline in BMD and deterioration of bone microarchitecture, as well as reduced estimated bone strength compared with baseline and 2 years postsurgery. 137 A meta-analysis concluded that bariatric surgery significantly increases fracture risk. 138
In contrast, long-term TTh for 6 years results in a significant progressive improvement of BMD and T-scores, while simultaneously reducing obesity markers (body weight and WC). 94 Another long-term study found that TTh for 8 years significantly increased vertebral and femoral BMD, despite sub-optimal TTh dosing. 103 The anabolic effect of TTh on the bone was unequivocally demonstrated in the Testosterone Trials (T-Trials). 141 These findings underscore that full realization of the osteoanabolic effects of TTh can only be realized in the long term.
When given in conjunction with a diet program, TTh modulates bone remodeling markers in a way indicative of a favorable long-term effect on BMD. 142 TTh is the only available treatment that results in long-term sustained weight loss combined with increased BMD. Further study is needed to find out whether this translates into a reduced incidence of fractures.
Weight Loss Versus Fat Loss
Obesity treatment interventions commonly target weight loss alone,26,115,143 without attention to effects on body composition and health outcomes.144–148 In fact, regulatory agencies require that obesity drug trials have change in body weight as the primary efficacy end-point. 149 This is a problem, as a myopic focus on weight loss alone can mask important body composition improvements, such as reduction in total and/or visceral fat mass accompanied by preservation, 44 or even gain in FFM. 150 Further, it can lead to frustration and dropout among patients with obesity who think that weight loss is the only desirable result.
The goal of obesity treatment is to lose excess body fat mass while preserving FFM, which consists of 40–68% muscle mass.127,151,152 The preservation of FFM is critical, because loss of FFM may increase risk for weight regain and increased fatness, by lowering maintenance energy requirement and triggering increased hunger/appetite. 153 Further, FFM is important for maintenance of metabolic health, 151 bone mass/skeletal integrity, 154 and quality of life. 155 Higher FFM (muscle mass) is associated with a significantly reduced risk of the MetS, 156 nonalcoholic fatty liver disease, 157 T2DM, 158 as well as lower mortality risk, independently of body fat mass, CV, and metabolic risk factors.159–162 In older age, ≥65 years, loss of FFM is a particularly strong risk factor for mortality, independently of body fat. 163 Hence, the preservation of FFM during weight loss is important for both successful weight loss maintenance as well as reduction of comorbidities and mortality risk.
A concern with traditional obesity treatment modalities is that they cause a significant loss of FFM. 127 Currently, no guidelines are available that define how much FFM loss is excessive during or after weight loss interventions. According to the “Quarter FFM Rule,” in healthy weight loss, the fraction of weight loss that can be attributed to FFM should not exceed 25%. 164 Although the percentage of weight loss that constitutes FFM is influenced by variables such as physical activity, diet composition, degree of caloric restriction, etc., 25% is a reasonable reference point for examination of the degree to which different weight loss interventions achieve preservation of FFM and a healthy weight loss.
A meta-analysis of lifestyle interventions for weight loss (minimum intervention period including follow-up of ≥12 months) found that 25% of weight loss comprises FFM. 165 A systematic review found that the mean FFM loss as a percentage of weight loss after dietary and drug-based weight-loss interventions resulting in a weight loss of >10 kg is 27% and 31%, respectively. 166 In a sex-specific body composition analysis at 1, 2, and 5 years after bariatric surgery, baseline body weight and FFM in men was 137.4 kg and 77.8 kg, respectively. 167 At 1, 2, and 5 years postsurgery, body weight (weight loss) was 94.4 kg (−43 kg), 98.1 kg (−39.3 kg), and 105.4 kg (−32 kg), and FFM (FFM loss) was 69.1 kg (−8.7 kg), 70.2 (−7.6 kg), and 69.9 kg (−8.7 kg). The percentage of weight lost as FFM was, hence, 20% at year 1, 19% at year 2, and 25% at year 5. 167 However, bariatric surgery can cause FFM loss as high as 53% of weight loss 2 years postsurgery. 168 A head-to-head comparison showed that bariatric surgery results in a significantly greater reduction in muscle mass than diet/exercise programs after 1 year. 169
Effect of TTh on Body Composition During Diet-Induced Weight Loss
Currently, there are only a few RCTs with body composition data that specifically investigated the effect of TTh in the context of weight loss in men with obesity. A notable diet RCT by Ng Tang Fui et al. investigated whether TTh has beneficial effects on weight loss and body composition over and above caloric restriction alone. 44 Men with obesity (BMI ≥30 kg/m2) and low T ≤ 12 nmol/L (346 ng/dL) were randomized to receive TTh with T undecanoate injections every 10 weeks (n = 49, baseline weight 118.3 kg) or matching placebo (n = 51, baseline weight 120.7 kg) for 56 weeks. Both groups underwent a very-low calorie diet during week 1 to 10, which was followed by a weight maintenance period of 46 weeks. Subjects were advised to perform at least 30 min of moderate-intensity exercise each day and completed exercise questionnaires and accelerometer testing (at weeks 0, 10, and 56) to reinforce and encourage participation in exercise. After the very-low calorie diet (phase 1, week 0 to 10), subjects were put on a less stringent calorie-restricted diet for the remaining 46 weeks (phase 2, week 11 to 56). At the end of the 10-week diet phase, weight loss was 12 kg and 13.5 kg in the T-group and placebo group, respectively (difference between groups not significant). There was a trend toward a 0.4 kg greater reduction in body fat mass and a 0.9 kg smaller reduction in lean mass in the T-group. At the end of phase 2 (week 56), weight loss remained largely stable in the T-group (+0.6 kg, p = 0.62), whereas there was weight regain in the placebo group (+2.6 kg, p = 0.06). At study end, men on TTh had, compared with placebo, lost significantly more body fat mass (−2.9 kg vs. −0.4 kg, p = 0.04) and visceral fat (−2678 mm2 vs. −1099 mm2, p = 0.04), and regained the diet-induced loss of lean mass (+3.4 kg vs. +0.9 kg, p = 0.002). As the combined loss of body fat and lean mass in men on placebo was similar to the amount of fat mass lost in men on TTh, there was no difference in body weight at study end. It is remarkable that men who had received TTh had a 7.3-fold greater reduction in total body fat and a 2.4-fold greater reduction in visceral fat mass than men on placebo.
A unique benefit of TTh in the context of weight loss is preservation of FFM (muscle mass). At the end of phase 1 (after 10 weeks on a very-low calorie diet), the fraction of weight loss comprising FFM was 32.5% and 35.5% in men on TTh and placebo, respectively. At study end (week 56), the fraction of weight loss comprising FFM was 5.3% in men on TTh and 36.7% in men on placebo. To the best of our knowledge, 5.3% is the smallest reduction in FFM ever reported in weight-loss trials. Support for the conclusion that TTh preserves muscle mass comes from the finding that men on TTh had a significantly higher appendicular lean mass and increased handgrip strength than men on placebo.
At the end of phase 1 (week 10), both groups had increased daily step count (p < 0.01) and activity levels (p < 0.05). However, at study end (week 56), daily step count was increased only in men on TTh, as was percentage of daily nonsedentary time (due to spending less time in sedentary activities and more time in light activities. Notably, the dropout rate was higher among men assigned to placebo compared with TTh. This suggests that TTh may make it easier for men with obesity to adhere to diet/exercise programs.
A likely explanation for the lack of preservation of lean mass during the initial diet phase may be the short duration of 10 weeks, as T-induced changes in lean mass or FFM typically require several months to manifest. 170 It should be pointed out that the greater reduction in body fat and preservation of lean mass in the T-group occurred despite a modest increase in endogenous total and free T levels (+2.9 nmol/L [+84 ng/dL] and +30.3 pmol/L [+10.5 pg/mL], respectively) in the placebo group. 44 This diet-induced elevation in T levels is similar to what has been seen in previous weight-loss studies,41,171 and it suggests that the endogenous rise in T subsequent to dieting is not sufficient to prevent diet-related loss of lean mass.
The results from Ng Tang Fui et al. confirm findings from previous smaller studies showing that TTh combined with lifestyle modification results in better outcomes than lifestyle modification alone. In 2009, a small RCT with 16 subjects who were newly diagnosed with T2DM and MetS showed for the first time that the combination of TTh and lifestyle intervention leads to greater therapeutic improvement in glycemic control, and reverses the MetS, after 52 weeks of treatment. 172 An 18-week (4.5 months) RCT in men with obesity and obstructive sleep apnea showed that the combination of TTh and lifestyle modification improved insulin sensitivity, ameliorated fatty liver, and increased muscle mass, in comparison to placebo and lifestyle modification alone. 173 In elderly men (age 65–85 years), TTh for 12 weeks combined with a resistance exercise program did not have any effect on body composition compared with placebo. 174 This is likely due to the short 3-month duration.
An RCT of 167 generally healthy community-dwelling older men (age 66 years) found that TTh for 12 months combined with resistance exercise training resulted in a greater reduction in fat mass by 1.2 kg, a greater increase in FFM by 1.7 kg, and a 0.3 kg greater increase in arm FFM than resistance exercise training alone. 175
An observational, parallel-arm, open-label 54-week study investigated the effect of hypocaloric diet plus exercise (DPE; n = 12) or DPE plus treatment with T undecanoate injections (DPE + T; n = 12), followed by 24 weeks of DPE alone in 24 men with obesity (BMI 42 kg/kg 2 ; age 54 years). 97 After 54 weeks, the DPE + T group had improvement in epicardial fat thickness, ejection fraction, diastolic function, carotid intima-media thickness (CIMT), and endothelial function (p < 0.01 vs. controls). Also, hormonal (T, p < 0.0001; GH, p < 0.01), metabolic (Homeostatic Model Assessment of Insulin Resistance [HOMA-IR], p < 0.01; microalbuminuria, p < 0.01), lipid (total cholesterol, p < 0.05), and inflammatory (fibrinogen, p < 0.05) parameters improved. After 24 weeks from T withdrawal, most parameters returned to baseline. A 33% dropout rate was reported in the DPE group, whereas there was no dropout among men receiving TTh. 97
Effect of TTh on Body Composition Without Dieting
Studies with body composition outcomes highlight the importance of looking beyond weight change when evaluating the potential clinical utility of TTh for prevention of obesity. It should be pointed out that shorter-term studies of TTh in the absence of dieting may show lack of change in body weight or even an increased body weight, but they still have beneficial effects on body composition by reducing body fat and increasing FFM.
For example, in men with normal weight and low T (13.6 nmol/L or 392 ng/dL), TTh for 1 year selectively reduced visceral fat accumulation without change in total body FM and increased total body FFM and total body and thigh skeletal muscle mass. 64 In another study of men with obesity (BMI 40 kg/m2) and hypogonadism (231 ng/dL), TTh for 24 weeks reduced subcutaneous fat mass by −3.3 kg and increased lean mass by 3.4 kg. 150 Despite a lack of change in body weight, there was a significant improvement in insulin sensitivity. 150 In men with overweight, TTh for 6 months increased lean body mass by 3.6 kg while simultaneously decreasing total fat mass by −1.2 kg, resulting in a net gain in body weight despite a significant reduction in percent body fat. 176
One RCT aimed at investigating the effect of TTh with T undecanoate injections versus placebo on CV risk factors, atherosclerosis progression, and body composition in a population of men with hypogonadism and MetS and T2DM. 177 After 12 months, men in the T-group had a reduction in body fat percent and WC by −18.5% and −8.5 cm, and an increase in FFM by 4.8 kg, despite no change in body weight. This was accompanied by a marked improvement in insulin sensitivity (HOMA-IR, p < 0.001) and reduction in HbA1c (p < 0.01), high-sensitivity C-reactive protein (p < 0.001), as well as CIMT (p < 0.0001). In contrast, no changes were seen in the placebo group. 177
In 161 men with TD and baseline BMI of 26 kg/m2, TTh for 52 weeks reduced body fat from 28.8% to 24.5%. 178 In a long-term follow-up of this study, body fat declined from 31.5% to 26.5% at month 24, whereas T levels increased from 263 to 551 ng/dL. Thereafter, there was a reduction in T levels, so that at month 96 the mean T level was 372 ng/dL. As the T level declined, body fat increased, so that at the end of the study there was no difference in body fat compared with baseline. 103 This underscores the importance of maintaining high enough elevations of T levels throughout the entire treatment period.
In contrast to RCTs of TTh in men with normal weight and overweight, which show improvement in body composition with no change or increase in body weight, 179 RCTs that included mostly men with obesity show significant reduction in body weight and WC.53,180,181 The first double-blind, placebo-controlled study conducted exclusively in men with T2DM and obesity was published in 2014. Patients (n = 211) were recruited from general practices for a 30-week RCT of TTh with T undecanoate injections, followed by 52 weeks of open-label treatment. 180 After 30 weeks, there was a significant reduction in WC by −2.5 cm and a small nonsignificant reduction in body weight. After 82 weeks, there was a significant weight loss of −2.7 kg and reduction in WC by −4.2 cm. At both time points, there were significant improvements in HbA1c and total cholesterol. Hypogonadism symptoms had significantly improved at week 82, but not week 30. 180 This suggests that 30 weeks (7.5 months) is not enough for weight loss and symptomatic improvement. Further support for the importance of long-term TTh comes from a long-term follow-up of this study, which found that WC and erectile function continued to improve for 4 years with ongoing uninterrupted TTh. 53 Remarkably, the progressive improvement in erectile function for nearly 4 years was independent of PDE5i use. 53
Another RCT of TTh in 55 men with obesity and T2DM showed that treatment with T undecanoate injections versus placebo injections for 1 year significantly reduced HOMA-IR by −4.64 versus −0.52, HbA1c (%) by −0.94 versus −0.24, fasting glucose (mmol/L) by −1.23 versus −0.13, fasting insulin (mE/L) by −8.52 versus −0.51 and CIMT (mm), a surrogate measure of atherosclerosis, by −0.10 versus −0.05. 182 Endothelial function, measured by flow mediated dilatation (FMD, %), improved by 2.40 versus −0.08. Although the between-group difference in body weight and WC was not significant after 1 year, in a follow-up of this study, where the placebo group was switched over to treatment with T undecanoate injections and the T group continued receiving T undecanoate injections for a second year, there was a significant reduction in body weight and. 181 In men who had been receiving T undecanoate injections for 2 years (which increased total T to 23.5 nmol/L and free T (calculated) to 594 pmol/L), there was a significant reduction in WC by 3.51 cm and BMI by −1.6 kg/m2. 181 This was accompanied by further improvement in insulin resistance/glucose control, lipids, atherosclerosis, and endothelial function; HOMA-IR (−5.94, final reading 5.51), fasting glucose (−1.83 mmol/L, final reading 8.23 mmol/L), HbA1c (−1.51%, final reading 6.6%), total cholesterol (−0.97 nmol/L, final reading 4.34 nmol/L), LDL (−0.40 nmol/L, final reading 2.39 nmol/L), HDL (+0.15 nmol/L, final reading 1.15 nmol/L), triglycerides (−0.99 nmol/L, final reading 1.86 nmol/L), non-HDL (1.12 nmol/L, final reading 3.19 nmol/L), FMD (+2.46%, final reading 7.14), and CIMT (−0.14 mm, final reading 0.73). It should be pointed out that these beneficial effects of TTh were achieved without any diet or exercise intervention. This RCT with follow-up shows that TTh for at least 2 years may be needed in some populations of men to achieve significant improvements in body weight, WC, insulin resistance/glucose control, lipids, atherosclerosis, and endothelial function.
More studies of TTh with long-term body composition data are needed to elucidate how the effects of T treatment on fat loss, FFM gain, and metabolic outcomes are influenced by TTh dose, treatment duration, as well as diet and exercise (or lack thereof), in men with obesity, the MetS and/or T2DM.
Reduction in CV Events and Mortality with Long-Term TTh in Daily Clinical Practice
Considering that obesity is a strong CV risk factor, and the widespread misperception about TTh and CV risk, it is important to examine long-term RWE data of TTh in men with obesity. In one RWE study, 77 men with obesity, hypogonadism, and a history of CVD were treated with T undecanoate injections for up to 8 years. 88 No patient suffered a major adverse CV event during the entire 8 year-long observation time. It was concluded that TTh in men with a history of CVD may be effective as an add-on treatment for secondary prevention of CV events. 88
In the first RWE study analysis that included a control group, 360 men with obesity and TD received T undecanoate injections for up to 10 years. 89 This TTh group was compared with a control group of 296 men who had opted to not receive TTh. There were 26 cases of nonfatal MI and 30 cases of nonfatal stroke in the control group and none in the TTh group. In the nontreated control group, there were 21 deaths, 19 of which were due to CV events (5 MI, 4 stroke, 7 heart failure, 2 thromboembolisms, 1 lung embolism). In the TTh group, there were only 2 deaths, and none was related to CV events (one was due to postsurgical thromboembolism and the other due to traffic accident). The estimated reduction in mortality for the TTh compared with the control group was between 66% and 92%. 89 This is the first large RWE study showing a significant reduction in hard clinical end-points with long-term TTh.
In the prediabetes RWE study described earlier, the incidence of nonfatal MI and mortality was 0.4% and 7.4% in the TTh group, and 5.7% and 16.1% in the control group, respectively. 54 Similarly, in the T2DM remission RWE study, there was no MI or stroke in T-treated men. Among untreated men, 31% had MI and 25% stroke. Mortality rate was 7% in T-treated men and 29% in untreated men. 92
The effect on hard clinical end-points after treatment with T undecanoate injections for up to 11 years in men with different weight classes, compared with untreated men, is illustrated in Figure 2. 42 Remarkably, there were no cases of MI nor stroke in any of the TTh groups.

Incidence of death, nonfatal myocardial infarction, nonfatal stroke, and prostate cancer in men with TD and normal weight, overweight, and obesity after treatment with T undecanoate injections (T), compared with untreated men (non-T). MI, myocardial infarction. Data from: Saad et al. 42
Adverse events in men with different weight classes during treatment with T undecanoate injections for up to 11 years could be compared with untreated (control) patients. 42
In men with various degrees of ED, TTh was given to 412 patients for up to 12 years, whereas 393 served as a control group. 93 In the TTh groups, no MI nor stroke occurred during the entire observation period (Fig. 3). In the untreated groups, MI and stroke occurred in 18.9% and 14.9% of patients with moderate/severe ED, respectively, and 16% and 15.3% in patients with no/mild ED. In the TTh groups, death from all causes occurred in 6.5% and 2.3% of men with no/mild ED and moderate/severe ED, respectively. In the untreated groups, death occurred in 14.6% and 21.3% of men with no/mild ED and moderate/severe ED, respectively. In the T-treated groups, PCa occurred in 0% and 2.3% of men with no/mild ED and moderate/severe ED, respectively. In the untreated groups, PCa occurred in 6.9% and 6.8% of men with no/mild ED and moderate/severe ED, respectively.
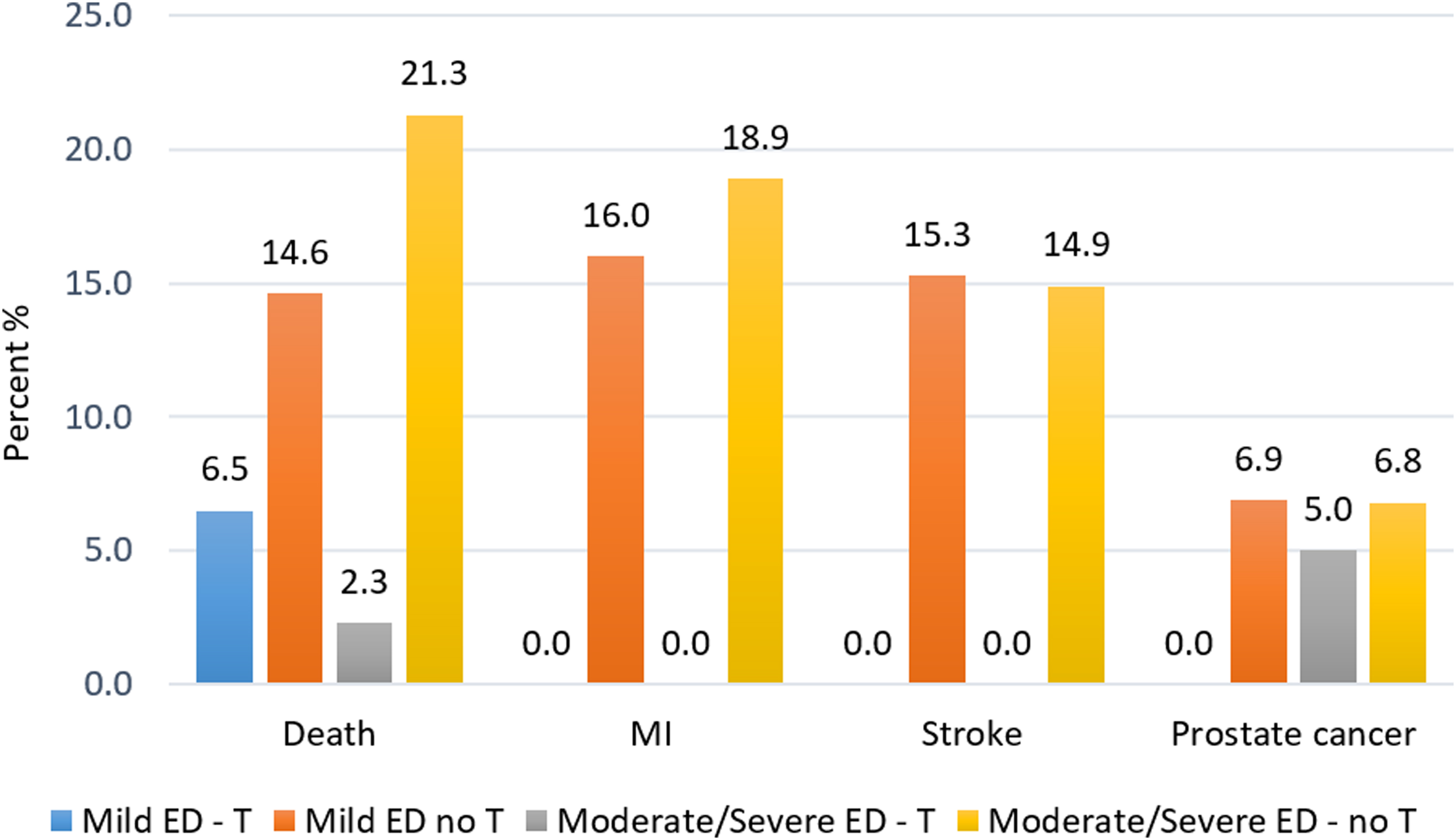
Adverse events (%) in patients with no/mild and moderate/severe ED during treatment with T undecanoate injections for up to 12 years (T), compared with untreated (control) patients (no T). ED, erectile dysfunction; T, testosterone. Data from: Saad et al. 93
Discussion and Conclusion
T is a key metabolic hormone with well-documented effects on body fat, muscle mass, and the skeleton.36,141,170,179,183,184 TD is particularly common in men with obesity. Because T is a key metabolic hormone, achieving and maintaining weight loss in men who have TD is extremely difficult. Numerous studies have shown that adding TTh to lifestyle interventions increases fat loss while preserving or increasing FFM.97,172,173,175
Loss of FFM and regain of body fat over time is a major drawback of diet and/or drug-based obesity treatment interventions. 127 Preservation of FFM during weight loss is critical, as reduction in FFM may counteract the beneficial health effects of weight loss, 185 as well as increase risk for weight regain. 153 In studies of diet/exercise interventions, around 25–35% of weight loss comprises FFM. After bariatric surgery, 20–50% of weight loss comprises FFM.167,168 The RCT data show that dieting men who receive placebo lose both body fat and lean mass, whereas weight loss in dieting men receiving TTh was almost exclusively body fat. 44 After 1 year of reduced caloric intake, the fraction of weight loss comprising FFM was 36.7% in men on placebo but only 5.3% in men on TTh. This suggests that TTh augments diet-induced loss of body fat and prevents loss of FFM. The mechanisms underlying the anabolic effect of TTh on muscle during diet interventions are starting to get elucidated, and it has been concluded that TTh is a viable strategy for preserving muscle mass during caloric restriction. 186
Although RCTs are considered to provide the highest quality evidence, efficacy of an intervention under controlled conditions is not synonymous with effectiveness, that is, intervention outcomes in “real-world” conditions.187–189 Because obesity is a chronic condition that is greatly influenced by lifestyle, evidence for how weight loss/maintenance interventions work in daily living conditions in the long term is essential.
Long-term RWE studies of men with TD and obesity who receive TTh in routine clinical practice show large amounts of weight loss that is progressive and sustained over time, with no plateau reported for up to 11 years. 42 This is opposite to the experience with obesity drugs, which have declining drug efficacy/effectiveness over time, often culminating in a weight-loss plateau within ∼1 year. 190 Further, because T is a key metabolic hormone, by correcting TD, TTh creates a metabolic environment that facilitates fat loss and long-term maintenance of an improved body composition.
The prevalence of severe obesity (BMI ≥35 kg/m2) is rising at an alarming pace; by 2030, it is projected to affect one in four adults.191,192 Considering that conventional weight-loss interventions (diet, exercise, drugs) have minimal impact in patients with severe obesity, 193 it is particularly noteworthy that long-term TTh for up to 8 years reduced BMI from 37.3 to 29.5 in men with grade II obesity (BMI 35–39.9) and from 41.9 to 32.4 in men with grade III obesity. 55 This suggests that TTh also offers significant clinical value for men with more severe obesity.
The RWE studies have additionally provided valuable insight about the consequences of untreated TD. 42 Men with TD who do not receive TTh have an increase in body weight and WC over time, regardless of baseline bodyweight. It is remarkable that in men with obesity, long-term TTh results in a weight loss of similar magnitude as that achieved with bariatric surgery. Change in body weight and BMI are the most frequently used measures to evaluate bariatric surgery outcomes. However, body weight-based metrics do not provide information about change in body composition (body fat and lean mass), which is what ultimately matters in terms of chronic disease risk, particularly T2DM and CVD. Bariatric surgery results in a large amount of weight loss and better weight loss maintenance compared with diet/exercise interventions; however, this is followed by a disproportional regain of body fat during the second and third year after bariatric surgery.194,195 A sex-specific analysis showed that men have increases in all adipose tissue depots between 12 and 24 months postsurgery with weight regain. 196 In parallel with the significant postsurgery body fat rebound, there is progressive worsening of lipid parameters, suggesting that the adverse switch in the change in body composition between the first and third year may underlie the observed recurrence of CV risk factors over time.194,195 In stark contrast, the weight loss and reduction in WC observed in RWE studies of long-term TTh is progressive over time and accompanied by marked improvement in glycemic control and lipid profile. Support for the beneficial effects of TTh on body composition comes from 36-month RCTs showing progressive reduction in body fat percent with TTh versus placebo.56,197 However, a prerequisite for achievement of body fat loss with TTh is adequate dosing of TTh and adherence to treatment, so that a sufficient T level elevation is reached and maintained in the long term. 103
In addition to increased fat loss and preservation of FFM, TTh may make it easier for men to adhere to diet/exercise programs. 44 Support for this comes from experimental studies showing that TTh increases spontaneous physical activity by enhancing CNS sensitivity to dopamine. 198 Previous studies have shown that TTh consistently improves mood and feelings of energy, and it reduces fatigue199–204 ; this, in turn, may bolster motivation and the ability to adhere to diet/exercise programs.172,205,206
Effective intervention to curb the rapidly growing obesity epidemic, which is affecting men of all ages, is urgently needed. Long-term “real-life” studies provide compelling evidence that TTh offers significant clinical utility in the treatment of men with obesity, as well as in the prevention of obesity development, in men with TD.42,55,87,93 This is accompanied by T2DM remission 92 and a significant reduction in T2DM incidence, 54 MI, stroke, PCa, and mortality.54,88,89,92,93 Because obesity is a gateway to ill health, 207 especially T2DM and CVD, a large-scale initiative for widespread implementation of TTh, as part of medical care for men with obesity, is highly warranted.
Footnotes
Author Disclosure Statement
F. Saad is a former employee of Bayer AG and now a consultant to Bayer AG. M. Caliber is an independent medical writer.
Funding Information
No funding was received for this article.
