Abstract
Introduction:
Because of the rising popularity of novel tobacco products, there has been an increasing interest in the biological effects of these products. This study was performed to investigate the biological effects of vapor from a novel tobacco vapor product (NTV) in comparison with cigarette smoke (CS) from a 3R4F reference cigarette by using two types of in vitro model.
Materials and Methods:
A two-dimensional (2D) culture system composed of a submerged monolayer of BEAS-2B cells was used to detect cellular oxidative stress responses. A three-dimensional (3D) culture system composed of a pseudostratified air–liquid interface culture of normal human bronchial epithelial cells was used to detect tissue inflammation. In both culture systems, exposures were conducted with an aqueous extract (AqE) of NTV vapor or CS.
Results:
In the 2D culture system, exposure to CS AqE induced glutathione oxidization and antioxidant response element reporter gene activation. NTV vapor AqE also induced reporter gene activity, but the concentration required for activation was higher than that of CS AqE. In the 3D culture system, exposure to CS AqE increased the secretion of various inflammatory mediators. In contrast, the effect of NTV vapor AqE on tissue inflammation was limited, and only interferon-γ showed a slight increase in expression.
Conclusions:
Overall, these results demonstrate that NTV vapor has fewer effects on cellular oxidative stress and tissue inflammatory responses as compared with CS in in vitro models using human cells.
Introduction
Various types of novel tobacco products have been introduced into the market worldwide.1,2 The novel tobacco vapor product (NTV), which we developed, is one of these products. The NTV is distinct from most existing tobacco products because it does not heat tobacco leaves directly, and the carrier liquid does not contain nicotine.3,4 A previous study demonstrated that the amounts of several harmful and potentially harmful chemicals present in cigarette smoke (CS) were markedly lower in NTV vapor. 5 Moreover, the results of a clinical study showed lower levels of biomarkers of exposure (e.g., exhaled CO and urinary metabolites) in NTV users compared with those in smokers. 6 However, because there is insufficient epidemiological data on long-term NTV use and associated health outcomes, it is still debatable whether using NTV instead of cigarettes has any benefit to public health by reducing the incidence of smoking-related diseases.
Smoking is a well-known risk factor for lung diseases, 7 indicating that the respiratory system is a primary target organ of CS. In the field of inhalation toxicology, in vivo experiments are performed as the gold standard, and the Organization for Economic Co-operation and Development has produced test guidelines (TGs) for acute (TG 403), sub-acute (TG 412), and sub-chronic (TG 413) inhalation toxicity tests. 8 Until epidemiological data can be obtained for NTV, in vivo experiments may be useful for investigating whether NTV vapor has fewer biological effects on animals in comparison with CS. Although the usefulness of in vivo studies for investigating the biological effects of aerosols from various tobacco products is clear,9–11 interspecies differences, such as those in lung geometry and metabolic capacity, exist between humans and animals.12,13 These interspecies differences can be problematic for research, especially regarding drug development; for example, unexpected toxicologic effects that were not found in animal testing were observed in human clinical trials. 14
In this context, the development of an in vitro test system with human cells is anticipated to avoid the difficulties caused by interspecies differences in the field of inhalation toxicology. 15 Such an in vitro test system will also reduce the time, cost, and animal use currently necessary for the assessment of test substances. Although most of the in vitro test systems using human cells have not been completely proven to respond similarly to their in situ setting, these systems will aid the mechanistic investigations of the toxicologic effects of test substances. 16 The induction of oxidative stress and inflammation are well-known mechanisms related to the adverse effects of CS. 17 For example, CS produces oxidative stress within epithelial cells in the lungs of smokers. Cellular oxidative stress initiates the production of chemokines and cytokines, which lead to tissue inflammation throughout the whole lung. Cumulative cellular oxidative stress and tissue inflammatory responses induced by chronic exposure to CS disrupt lung homeostasis and contribute to disease development.18,19 Therefore, when assessing the biological effects of NTV vapor using an in vitro test system, both the cellular oxidative stress and tissue inflammatory responses should be examined in comparison with CS.
For the in vitro analysis of cellular responses, a lung cell line can be useful because it is easy to handle and produces highly reproducible data. 20 Consequently, lung cell lines are often applied in high-content screening approaches. 21 In contrast, primary cells can be used to investigate tissue responses because primary normal human bronchial epithelial cells differentiate into pseudostratified mucociliary epithelia, which resemble in vivo tissues under air–liquid interface culture conditions. 22 In the experiments presented here, we used both of these types of cell culture system to investigate the biological effects of aqueous extract (AqE) of NTV vapor in comparison with AqE of 3R4F reference CS: a two-dimensional (2D)-culture system composed of a submerged monolayer of BEAS-2B cells, which were established by the immortalization of normal human bronchial epithelial cells, and a commercially available three-dimensional (3D) culture system (MucilAir) composed of a pseudostratified air–liquid interface culture of primary normal human bronchial epithelial cells. Each exposure was performed by using an AqE of 3R4F CS or NTV vapor; the resulting cellular oxidative stress responses were detected in the 2D-culture system (Fig. 1A), and the resulting tissue inflammation was detected in the 3D-culture system (Fig. 1B).
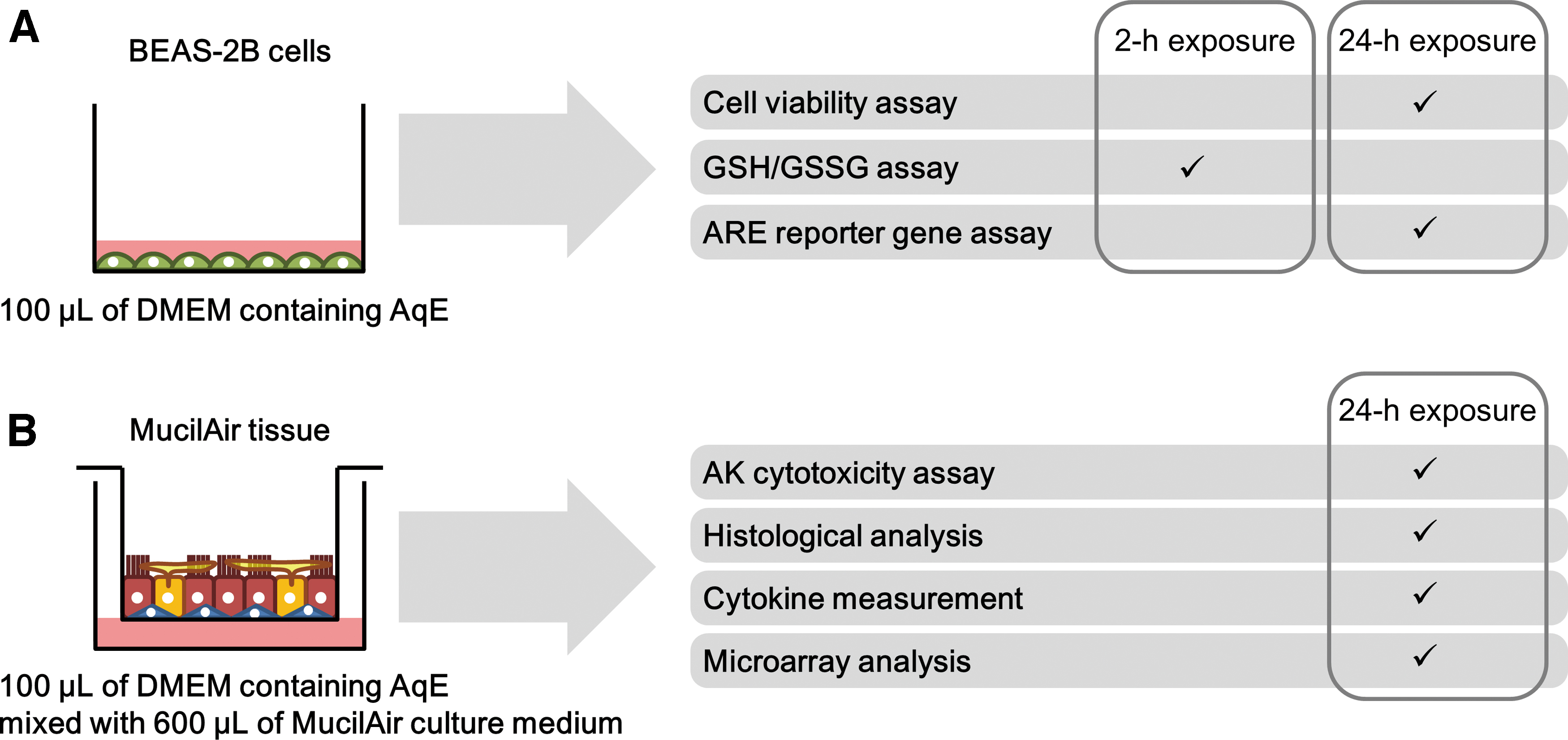
Study design for 2D- and 3D-culture assays.
Materials and Methods
AqE preparation
AqEs were prepared from NTVs (a prototype of Ploom TECH; manufactured by Japan Tobacco, Inc., Tokyo, Japan) and from 3R4F reference cigarettes (University of Kentucky, Lexington, KY). 3R4F is a previous generation reference cigarette. The latest reference cigarette is 1R6F. A previous study demonstrated no distinct differences between 3R4F and 1R6F when used for in vitro toxicity testing or chemical analyses. 23 The NTVs used in this study were prepared specifically for experimental purposes. The NTVs are each composed of a battery, cartridge, and tobacco capsule. 5 The 3R4F cigarettes and NTVs (tobacco capsules and cartridges) were conditioned at 22°C ± 1°C and 60% ± 3% relative humidity at least 48 hours before use. An RM20H smoking machine (Borgwaldt KC GmbH, Hamburg, Germany) was used for smoke/vapor generation. The 3R4F cigarettes were smoked under the International Organization for Standardization (ISO) intensive smoking conditions (55-mL puff volume, 30-second puff interval, 2-second puff duration, bell-shaped puff profile, and 100% blocking of ventilation holes), 24 until a point 3 mm from the tipping paper was reached. The NTVs were vaped with the ISO standard conditions for vapor products (55-mL puff volume, 30-second puff interval, 3-second puff duration, and square-shaped puff profile), 25 until the puff number reached 50 per capsule. The generated whole smoke/vapor was bubbled into 15 mL of ice-cold Dulbecco's modified Eagle's medium (DMEM; Thermo Fisher Scientific, Waltham, MA) in the impinger. The number of puffs bubbled into the AqE was ∼2 puffs/mL for 3R4F cigarettes (three cigarettes were smoked) and 16.7 puffs/mL for NTV (five capsules were vaped). The AqE of each test product was freshly prepared before every exposure.
AqE nicotine concentration analysis
For nicotine analysis sample preparation, each AqE was first diluted 200-fold with purified water; then, 50 μL of diluted AqE was mixed with 200 μL of 0.028% ammonia water, 20 μL of purified water, and 20 μL of internal standard solution (100 ng/mL nicotine-methyl-d3). Extraction was then performed by using an OASIS HLB μElution Plate (Waters, Milford, MA) without prior conditioning. The prepared sample was applied to the plate and washed with 200 μL of purified water; then, the analyte was eluted with 100 μL of methanol. The resulting elution was mixed with 100 μL of 10 mmol/L ammonium acetate aqueous solution (pH 9.0) and subjected to ultra-performance liquid chromatography (UPLC; ACQUITY Ultra Performance LC, Waters) coupled with tandem mass spectrometry (MS/MS; API4000; AB Sciex, Framingham, MA). UPLC separation was performed in a gradient elution by using an Inertsil Ph-3 column (2.1 × 50 mm, 2 μm; GL Sciences, Inc., Tokyo, Japan). The mobile phase contained: Solvent A, 10 mmol/L ammonium acetate aqueous solution with 0.01 vol% acetic acid; and Solvent B, methanol/acetonitrile (50/50, v/v). Solvent B was maintained at 40% for an initial 0.8 minutes, then increased to 90% over the next 0.01 minutes, maintained at 90% until 1.30 minutes, decreased to 40% over the following 0.01 minutes, and maintained at 40% until 1.40 minutes. The flow rate was set at 0.5 mL/minutes, the column temperature was set at 50°C, and the injection volume was 10 μL. MS/MS detection was performed in electrospray ionization mode, and the ion source polarity was set to positive ionization mode. Precursor-to-product ion transitions for multiple reaction monitoring were m/z 163–130 for nicotine and m/z 166–130 for nicotine-methyl-d3.
2D-culture system
The normal human bronchial epithelial cell line BEAS-2B was purchased from American Type Culture Collection (Manassas, VA). BEAS-2B cells containing an antioxidant response element (ARE) reporter gene (no. E3641; Promega, Madison, WI) were prepared by using a previously described protocol. 26 The cells were seeded in a 96-well plate at a density of 5.0 × 103 cells/well and cultivated with 100 μL/well of DMEM containing 10% fetal bovine serum (2916554, lot 1608A; MP Biomedicals, Carlsbad, CA) for 24 hours before exposure. Fetal bovine serum was inactivated before use by heating for 30 minutes in a 56°C water bath. The cells were then cultivated with 100 μL/well of exposure medium containing test product AqE (Fig. 1A). An exposure–medium dilution series was prepared by initially combining 1 mL of fetal bovine serum with 9 mL of AqE, then performing serial dilutions of this solution with DMEM containing 10% fetal bovine serum. The maximum exposure–medium concentration was 0.2 puffs/mL for 3R4F CS and 15 puffs/mL for NTV vapor (Table 1). Cells were subjected to three different assays after exposure: cell viability assay, reduced glutathione/oxidized glutathione (GSH/GSSG) assay, and ARE reporter gene assay (Fig. 1A). The three assays were performed by using the same batch of AqE of each test product in a single experiment. The experiment was repeated three times with three different batches of AqE.
Number of Puffs Bubbled into and the Nicotine Concentration in the Highest Exposure-Medium Concentration Used for Two-Dimensional-Culture Assays
Values are the mean and SD (n = 8).
AqE, aqueous extract; NTV, novel tobacco vapor product; SD, standard deviation.
Cell viability assay
After 24 hours of exposure to test product AqE, the cell viability was analyzed by using a Cell Titer-Fluor Cell Viability Assay (Promega) in accordance with the manufacturer's instructions. Fluorescence was measured with an Infinite 200 Pro plate reader (Tecan Group Ltd, Männedorf, Switzerland) at excitation/emission wavelengths of 400/505 nm.
GSH/GSSG assay
After 2 hours of exposure to test product AqE, the reduced GSH/GSSG ratio of the remaining adherent cells was analyzed by using a GSH/GSSG-Glo Assay (Promega) in accordance with the manufacturer's instructions. The resulting luminescence was detected with an Infinite 200 Pro plate reader. The GSH/GSSG ratio was calculated by using the formula provided in the manufacturer's instructions.
ARE reporter gene assay
After 24 hours of exposure to test product AqE, the ARE reporter gene activity of the cells was analyzed by using a Luciferase Assay System (Promega). The luminescence was measured with an Infinite 200 Pro plate reader immediately after the addition of a substrate solution by an injector.
Statistical analysis of 2D-culture assay results
Each 2D-culture assay was performed three times, and the obtained experimental data were expressed as a percentage of the control values. Statistical analyses were performed on these data by using EXSUS (Ver. 8.1.0; CAC Croit Corporation, Tokyo, Japan). Bartlett's test was used to confirm the homogeneity of variance of each group. A one-way analysis of variance followed by Dunnett's multiple-comparison test was then performed against the control condition values. The differences were considered significant if the p-value was <0.05. The data obtained from the ARE reporter gene assay were log transformed before being subjected to statistical analysis.
3D-culture system
MucilAir tissues from a healthy non-smoker (Epithelix Sàrl, Geneve, Switzerland) were used as the basis of the 3D-culture system. The tissues used in all experiments consisted of bronchial epithelial cells from a 41-year-old male non-smoker, with no pathology reported (batch no.: MD072001) as described in the certificate of analysis provided by Epithelix Sàrl. Before their use in experiments, the MucilAir tissues were first acclimatized by incubation with 700 μL/well of MucilAir culture medium (Epithelix Sàrl) for more than 1 week, with medium replacement every 2–3 days. The tissues were then cultured with 700 μL/well of exposure medium containing test product AqE (Fig. 1B). An exposure-medium dilution series was prepared by first diluting AqE with DMEM, then performing additional 1:6 dilutions with MucilAir culture medium. Unlike in the 2D-culture assays, the diluted AqEs were mixed with MucilAir culture medium before they were used for exposure in the 3D-culture assays (Fig. 1B). This method was employed, because it was not possible to directly bubble the generated smoke or vapor into MucilAir culture medium in the impinger; the overflow of the excessive foaming from the impinger that occurred with direct bubbling may have been caused by the proteins with which the MucilAir culture medium was supplemented. Consequently, the maximum exposure concentrations in the NTV AqE differed between that used for the 2D-culture assays (Table 1) and that used for the 3D-culture assays (Table 2). The maximum exposure-medium concentration was 0.2 puffs/mL for 3R4F CS and 2 puffs/mL for NTV vapor. A single batch of AqE of each test product was prepared, and exposure was performed by using four tissues.
Number of Puffs Bubbled into and the Nicotine Concentration in the Highest Exposure-Medium Concentration Used for Three-Dimensional-Culture Assays
Values are the mean and SD (n = 8).
Adenylate kinase cytotoxicity assay
Medium collected from cultured cells after 24 hours of exposure to test product AqE was subjected to an adenylate kinase (AK) cytotoxicity assay with ToxiLight (Lonza, Basel, Switzerland). The resulting luminescence was detected with an Infinite 200 Pro plate reader. Cytotoxicity is expressed as a percentage of the level measured for the positive control (samples obtained from tissues exposed to 1% Triton X-100).
Histological analysis
Tissues collected after 24 hours of exposure to test product AqE were fixed with 4% paraformaldehyde, and they were then embedded in paraffin. Sections prepared from these embedded tissues were subjected to hematoxylin and eosin staining.
Cytokine measurement
Medium collected from cultured cells after 24 hours of exposure to test product AqE was subjected to cytokine measurement by a V-PLEX Human Cytokine 30-Plex Kit (Meso Scale Diagnostics, Rockville, MD) in accordance with the manufacturer's instructions, using a Sector Imager 6000 (Meso Scale Diagnostics). Cytokines with concentrations higher than the lower limit of detection in all samples at each exposure condition were subjected to statistical analysis.
Microarray analysis
Tissues collected after 24 hours of exposure to test product AqE were subjected to mRNA extraction with RNeasy (Qiagen, Hilden, Germany). The resulting mRNAs were sent to Takara Bio, Inc. (Shiga, Japan), where a microarray analysis was performed by using the GeneChip Human Genome U133 Plus 2.0 Array. Microarray data were processed with GeneSpring Version 14.0 (Agilent Technologies) as described in a previous article. 27 Genes were considered differentially expressed if they had an absolute log2-fold change of >1.0 and a p-value of <0.05 (false-discovery-rate corrected). The differentially expressed genes (DEGs) were subjected to Ingenuity Pathway Analysis (IPA; Qiagen), and pathways showing significant perturbations (absolute z-score >2 and p-value <0.05) were identified. The resulting transcriptomic data are available in ArrayExpress under accession number E-MTAB-8920.
Statistical analysis of 3D-culture assay results
3D assay data were obtained from four tissue samples. AK assay and cytokine data were subjected to Dunnett-type Bonferroni's multiple-comparison test by using EXSUS (Ver. 8.1.0). The results were considered significant if p < 0.05. Data obtained from cytokine measurements were subjected to statistical analysis after log transformation.
Results
Exposure conditions for the 2D-culture system
A normal human bronchial epithelial cell line (BEAS-2B) cultivated under submerged conditions was used as a 2D-culture system of human bronchial epithelia and experimentally exposed to 3R4F CS or NTV vapor AqE (Fig. 1A). The test product AqE concentration to which the 2D-culture system was exposed was determined by the number of puffs bubbled into the medium. When the exposure concentration was expressed in terms of the puff number, the maximum concentration of NTV vapor AqE was 75 times higher than that of 3R4F CS AqE (Table 1). Because nicotine is present in both test products, we also analyzed the nicotine concentration in the test product AqEs collected under the same conditions as the biological test. When the exposure concentration was expressed in terms of the nicotine level in the AqE, the maximum concentration used for NTV vapor AqE exposure was approximately six times higher than that used for 3R4F CS AqE exposure (Table 1).
Post-exposure cell viability in the 2D-culture system
After a 24-hour exposure to test product AqE, the viability of BEAS-2B cells in the 2D-culture system was analyzed. There was a clear concentration-dependent decrease in cell viability after exposure to 3R4F CS AqE; at the maximum exposure concentration, the cell viability was ∼20% (Fig. 2A). The BEAS-2B cell viability also decreased in a concentration-dependent manner after NTV vapor AqE exposure (Fig. 2A). However, in contrast with the 3R4F CS AqE-exposed cells, more than 60% of BEAS-2B cells were still viable after exposure to the maximum NTV vapor AqE concentration.
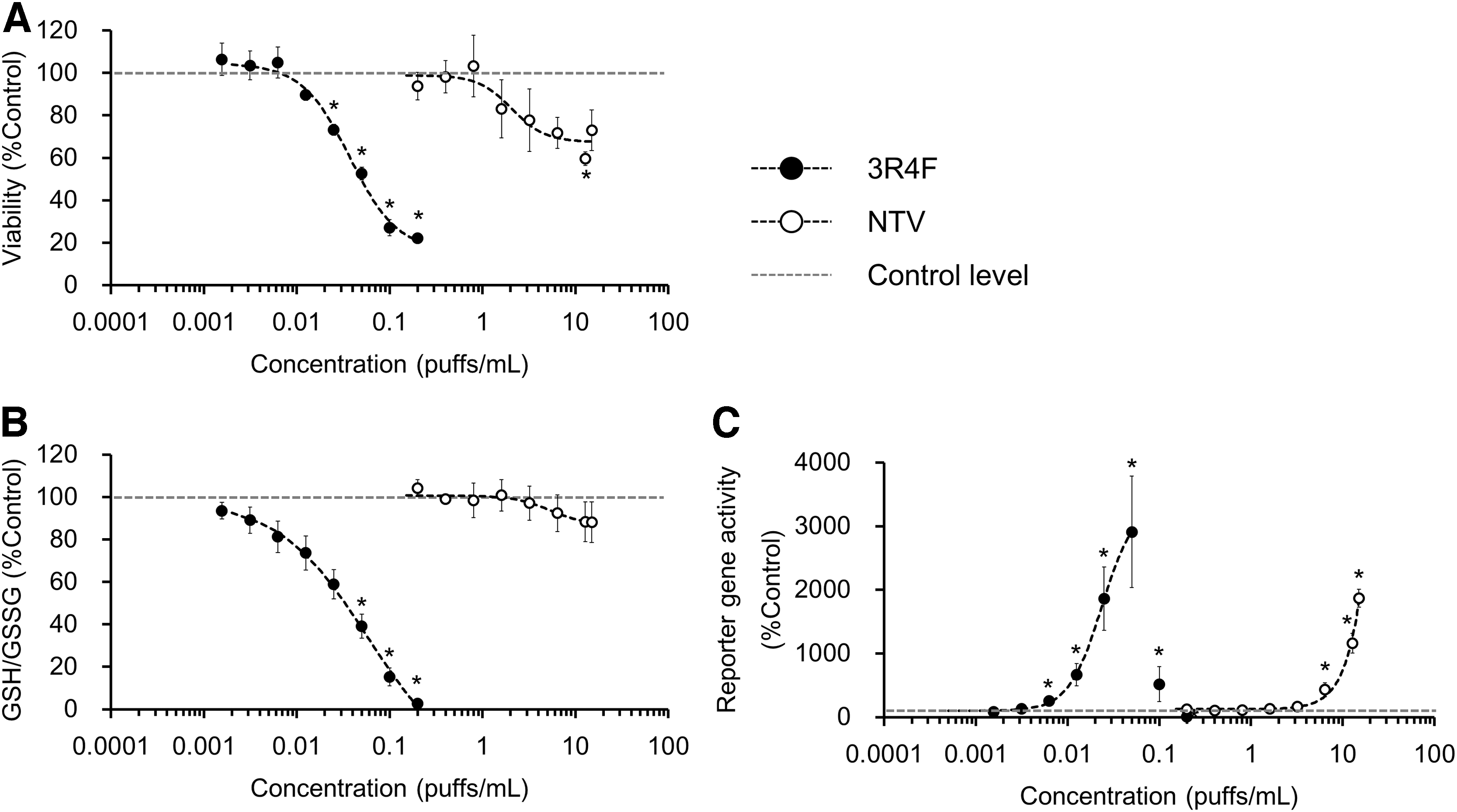
Assay results from a 2D-culture system exposed 3R4F reference CS or NTV vapor AqE.
Post-exposure oxidative stress responses in the 2D-culture system
Reducing cellular oxidative stress is an important response to CS exposure. GSH functions as a primary defense mechanism by eliminating reactive oxygen species (ROS) through its conversion to GSSG. In the 2D-culture system, 3R4F CS AqE exposure induced a concentration-dependent decrease in the GSH/GSSG ratio, and GSH was nearly depleted at the maximum exposure concentration (Fig. 2B). In contrast, exposure to the maximum NTV vapor AqE concentration produced a statistically insignificant decrease (to ∼90% of control levels) in the GSH/GSSG ratio (Fig. 2B).
The cellular oxidative stress response was also assessed by using an ARE reporter gene assay, which detects nuclear factor-erythroid 2-related factor-2 (NRF2)-mediated oxidative stress responses. A concentration-dependent increase in reporter gene activity was detected after exposure to 3R4F CS AqE (Fig. 2C). A significant increase in reporter gene activity was detected at concentrations higher than 0.00625 puffs/mL of 3R4F CS AqE, and the maximum response was detected at 0.05 puffs/mL of 3R4F CS AqE (Fig. 2C). Higher concentrations of 3R4F CS AqE exposure (i.e., 0.1 and 0.2 puffs/mL) decreased reporter gene activity because of the increased cytotoxicity at these concentrations (Fig. 2A). In contrast with the GSH/GSSG assay results, the ARE reporter gene assay results show a clear concentration-dependent increase in reporter gene activity after exposure to NTV vapor AqE (Fig. 2C). A significant increase in reporter gene activity was induced by exposure to NTV vapor AqE concentrations higher than 6.4 puffs/mL (Fig. 2C).
Exposure conditions for the 3D-culture system
In addition to conducting experiments with the 2D-culture system, experiments using a 3D-culture system of human bronchial epithelia (MucilAir tissues) were also performed (Fig. 1B). The AqEs used for 3D-culture assays were prepared with the same protocol used for the 2D-culture assays, that is, by bubbling 3R4F CS or NTV vapor into DMEM. When the exposure concentration was expressed in terms of the number of puffs bubbled into the medium, the maximum concentration used for NTV vapor AqE exposure was 10 times higher than that used for 3R4F CS AqE exposure (Table 2). However, when the exposure concentration was expressed in terms of the amount of nicotine in the AqE, the maximum concentration used for NTV vapor AqE exposure was comparable to that used for 3R4F CS AqE exposure (Table 2).
Post-exposure AK cytotoxicity and histological analysis of 3D-culture tissues
The cytotoxic effect of the AqE of each test product on 3D-culture tissues was analyzed after 24 hours of exposure. There were no significant increases in cytotoxicity after exposure to either 3R4F CS or NTV vapor AqE (Fig. 3A). The results of a histological analysis on the 3D-culture tissues indicate that the pseudostratified-differentiated epithelium was maintained after exposure to either 3R4F CS AqE or NTV vapor AqE (Fig. 3B), which is consistent with the AK cytotoxicity results.
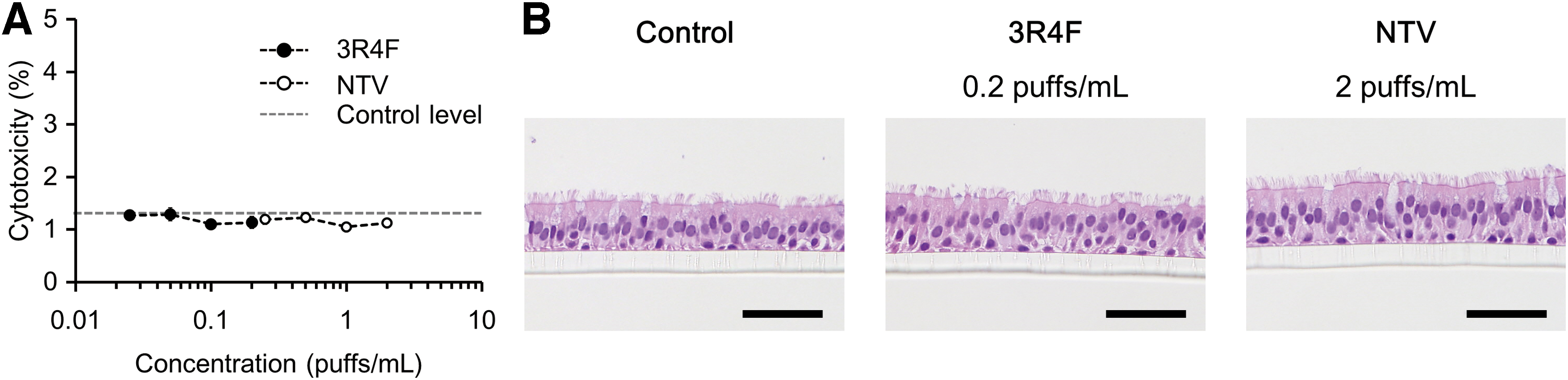
Cytotoxicity assay and histological analysis results from a 3D-culture system exposed to 3R4F reference CS or NTV vapor AqE for 24 hours.
Post-exposure cytokine analysis of 3D-culture tissues
Although the AK cytotoxicity assay and histological analysis results suggest that the test product exposure concentrations were noncytotoxic, cytokine secretion in the 3D-culture tissues was significantly affected by exposure to the AqE of CS. Levels of 30 inflammatory mediators in the medium of the tissues were analyzed with the V-PLEX Human Cytokine 30-Plex Kit. Several cytokines in the kit, including tumor necrosis factor (TNF)-α, interferon (IFN)-γ, interleukin (IL)-1β, IL-8, and vascular endothelial growth factor-A (VEGF-A), had significantly upregulated expression after tissue exposure to 3R4F CS AqE (Fig. 4A). In addition, the expression levels of IL-7 and TARC were significantly downregulated after tissue exposure to 3R4F CS AqE (Fig. 4A). In contrast, only IFN-γ had significantly upregulated expression levels in 3D-culture tissues at the maximum exposure concentration of NTV vapor AqE (Fig. 4B).
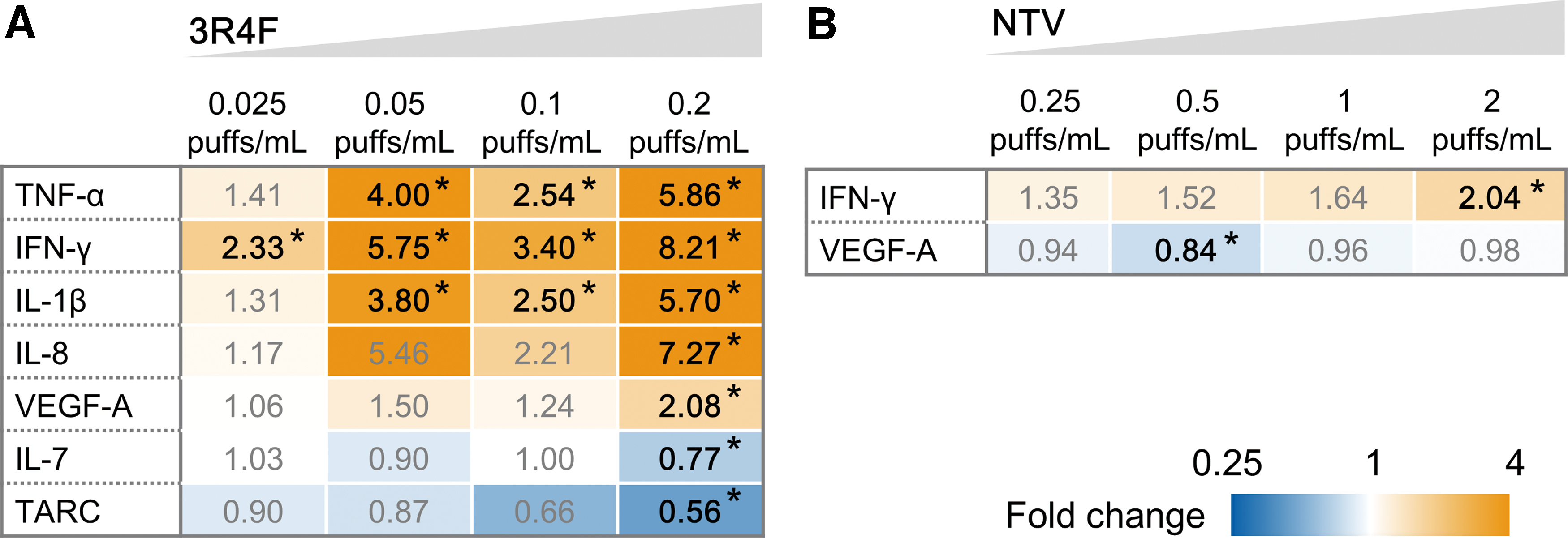
Cytokine levels in a 3D-culture system exposed to 3R4F reference CS or NTV vapor AqE for 24 hours.
Post-exposure microarray analysis of 3D-culture tissues
To obtain a comprehensive understanding of the conditions in 3D-culture tissues exposed to the AqE of 3R4F CS or NTV vapor, global gene expression profiles were analyzed by using microarray technology. The number of DEGs concentration-dependently increased in tissues exposed to test product AqE (Fig. 5A). There were 434 DEGs in tissues exposed to 0.2 puffs/mL of 3R4F CS AqE, whereas there were only 25 DEGs in tissues exposed to 2 puffs/mL of NTV vapor AqE (Fig. 5A).
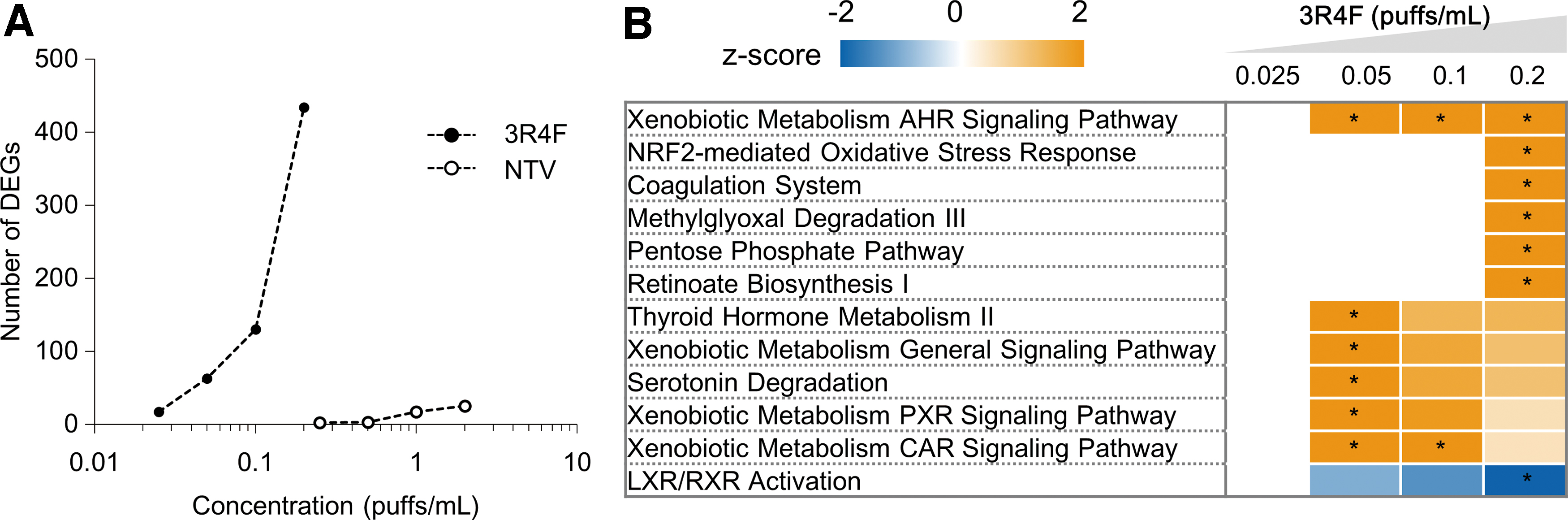
Microarray analysis of a 3D-culture system exposed to 3R4F reference CS or NTV vapor AqE for 24 hours. The mRNAs extracted from four tissue samples were used for analysis.
To identify toxicologic pathways significantly altered by 3R4F CS or NTV vapor AqE exposure, the detected DEGs were subjected to IPA. The DEGs detected after exposure to 3R4F CS AqE were significantly related to the oxidative stress response pathway (the NRF2-mediated oxidative stress response and pentose phosphate pathways) and pathways related to the metabolism of external chemicals (xenobiotic metabolism pathways) (Fig. 5B). In contrast, no pathways were significantly altered by NTV vapor AqE exposure.
Discussion
The use of various novel tobacco products is increasing worldwide. Consequently, there is great interest in the development of in vitro methods to assess the biological effects of vapor from these products.15,16 Here, we employed two types of cell culture systems to investigate the biological effects of NTV vapor AqE in comparison with 3R4F CS AqE: a 2D-culture system (submerged culture of BEAS-2B cells), which was used to detect cellular oxidative stress responses, and a 3D-culture system (air–liquid interface culture of MucilAir tissues), which was used to detect tissue inflammatory responses. Detecting toxicologic effects at various biological levels (e.g., molecular, cellular, tissue, organ, and organism) is important for the prediction of adverse outcomes to test substances. 28 Moreover, the integration of multiple assays for testing and assessment is expected to increase prediction accuracy. 29 Although no accepted adverse outcome pathway or validated in vitro assays for inhalation toxicology are currently available, a combination of 2D- and 3D-culture assays may allow the accurate prediction of inhalation toxicity for test substances in the future. 30
The usefulness of a 2D-culture system for detecting CS-induced oxidative stress responses was demonstrated by a previous interlaboratory study in which the exposure of 2D-culture systems using different lung cell lines (i.e., H292 and BEAS-2B cells) to total particulate matter from CS induced intracellular oxidative stress responses with interlaboratory reproducibility. 31 That study also exposed 2D-culture systems to the aerosol-collected masses of two tobacco heating products and an e-cigarette, and the results indicated that no oxidative stress responses were induced. Unlike this previous study, we used AqE of vapor from our proprietary novel tobacco product (i.e., NTV) for exposure instead of aerosol-collected mass because the use of AqE for exposure enables the application of higher concentrations of vapor exposure compared with the use of aerosol-collected mass. The AqE of aerosol from tobacco products has been applied to a variety of toxicological assays. 32 Previous work demonstrated the usefulness of AqE for comparing the biological effects between novel tobacco products and CS. 33
To detect the effects of 3R4F CS or NTV vapor AqE on cellular oxidative stress responses, assays were performed on BEAS-2B cells grown in a 2D-culture system. Oxidative stress responses initiated by ROS generation in cells exposed to CS have been shown to occur in the respiratory system of smokers.34,35 The generated ROS can be eliminated by GSH through its conversion to GSSG. Consequently, the GSH/GSSG ratio decreases under oxidative stress conditions, and this decrease can trigger the nuclear translocation of NRF2, which binds to ARE to activate the transcription of antioxidant genes.18,36,37 The clear concentration-dependent decrease in the GSH/GSSG ratio and increase in the ARE reporter gene activity observed in 2D-culture cells after exposure to the AqE of CS indicates that oxidative stress responses were induced under these conditions (Fig. 2B, C). The decrease in GSH/GSSG observed in this study reflects a decrease in GSH in the BEAS-2B cells. Further, the concentration of 3R4F CS AqE that decreased GSH/GSSG at 2 hours after exposure (Fig. 2B) also decreased cell viability at 24 hours after exposure (Fig. 2A). Therefore, the decrease in GSH/GSSG may occur in BEAS-2B cells via various mechanisms (e.g., oxidation, conjugation with reactive metabolites, or leakage from dead cells) during the process of cell death. A significant increase in ARE reporter gene activity was also detected in the BEAS-2B cells after exposure to NTV vapor AqE. However, the minimum concentration required to induce a significant level of activation was ∼100 times higher for NTV vapor AqE (6.4 puffs/mL) compared with 3R4F CS AqE (0.00625 puffs/mL) (Fig. 2C). Therefore, NTV vapor AqE has less of an effect on the cellular oxidative stress response compared with CS AqE.
In addition to employing a 2D-culture system, the present study used a 3D-culture system to investigate the effects of CS and NTV vapor AqEs on tissue inflammation. Intracellular oxidative stress induces the secretion of inflammatory mediators, and these mediators affect other surrounding cells, leading to inflammation of the whole tissue. 17 The 3D-culture system, which has been successfully applied to previous tobacco research, shows pseudostratified mucociliary epithelia that resemble in vivo tissues and is suitable for studying tissue inflammation.38–42 Although the concentration of CS AqE used for exposure was determined to be noncytotoxic (Fig. 3), the expression levels of various inflammatory mediators (i.e., the cytokines TNF-α, IFN-γ, and IL-1β, the chemokine IL-8, and growth factor VEGF-A) were significantly upregulated after exposure to CS AqE (Fig. 4A). Secretion of these inflammatory mediators was reported to be important for lung defense to external stimuli, including CS.43,44 Therefore, the tissue inflammation observed in this 3D bronchial tissue culture resembles that of actual in vivo tissue. Notably, when the 3D-culture tissues were exposed to NTV vapor AqE, only IFN-γ was found to have a significant increase in expression. Therefore, compared with CS AqE exposure, NTV vapor AqE exposure has less of a biological effect on the secretion of mediators of inflammation in vitro.
In addition to measuring cytokine levels, global gene expression profiles were analyzed to characterize the condition of 3D bronchial tissue culture after test product AqE exposure. The IPA analysis of DEGs predicted the significant activation of pathways related to oxidative stress responses, that is, NRF2 and its related pentose phosphate pathway. 45 These results are consistent with the 2D-culture assay results, showing a significant activation of NRF2-induced ARE reporter genes after exposure to CS AqE. Prior reports on the activation of these pathways in the bronchial epithelia of smokers46,47 support the idea that 3D-culture tissues react similarly to in vivo tissues after exposure to CS. Notably, the microarray results indicate that the number of DEGs was lower in tissues exposed to NTV vapor AqE than that in tissues exposed to 3R4F CS AqE. When the DEGs detected after NTV vapor AqE exposure were subjected to an IPA analysis, a significant activation of the affected pathways observed in 3R4F CS AqE-exposed tissues was not detected. Because NTV generate vapor without combusting tobacco leaves, the chemical composition of the produced vapor is different from that of 3R4F CS. A previous study showed that the emission levels of constituents, so-called “Hoffmann analytes,” in NTV vapor were reduced compared with those in the 3R4F reference CS. 5 The emission levels for most of the analytes in NTV vapor were below the detection limit. Although formaldehyde, acetone, and ammonia can be detected in NTV vapor, their emission levels were confirmed to be reduced by 94%, 98%, and 58% compared with 3R4F CS. 5 The IPA analysis results reflect this difference and support the idea that the levels of specific biologically active chemicals present in 3R4F CS AqE are lower in NTV vapor AqE.
In this study, a 3D bronchial tissue culture was exposed to test product AqE to investigate biological responses. Because this study was conducted with a single batch of 3D bronchial tissue culture composed of cells from a single donor, it is desirable to confirm whether the same results can be obtained by using multiple batches of 3D bronchial tissue culture composed of cells from different donors in future studies. However, some of the responses observed in this study, such as the upregulation of IL-8 after exposure to CS AqE, have been reported in previous studies using 3D bronchial tissue cultures exposed to CS.38–42 Therefore, this endpoint is likely to be robust and useful for comparing the biological effect of CS and NTV vapor AqEs.
In this study, exposure was performed by using AqEs of 3R4F CS or NTV vapor. When the exposure concentration was expressed in terms of the number of puffs bubbled into the medium, the concentration of NTV vapor AqE used for exposure was higher than that of 3R4F CS AqE (Tables 1 and 2); the exposure concentration was also higher or comparable when it was expressed in terms of the amount of nicotine (Tables 1 and 2). The observed results from both 2D- and 3D-culture systems exposed to NTV vapor AqE indicate that NTV vapor AqE has fewer biological effects compared with CS AqE. However, because several reactions to NTV vapor AqE, such as increased ARE reporter gene activity, were detected, further study is required to determine whether these responses are observed in NTV users with approaches with higher sensitivity than what is required for CSs. For that purpose, it is necessary to compare the exposure concentration used in this in vitro study with the exposure concentration in the tissues of actual NTV users. The development of an appropriate computational model that can calculate the actual human tissue doses of exposure for smokers and NTV users will enable this important comparison.48–50
Conclusions
Overall, our results demonstrate that NTV vapor AqE has fewer effects, compared with 3R4F CS AqE, on the cellular oxidative stress response in a 2D-culture system and on tissue inflammatory responses in a 3D-culture system. Further studies, including in vivo studies and clinical trials, will confirm whether the data obtained using these two culture systems can be used to predict the biological effects of NTV vapor on an organ and/or a whole organism.
Footnotes
Acknowledgments
The authors are grateful to Dr. Tomoki Nishino for his support in conducting this study. They thank Hiroshi Ito for his technical input on the smoke/vapor generation for the preparation of AqEs. They also thank Dr. Yuichiro Takanami and Dr. Nobumasa Kitamura for their technical input on the nicotine analysis. They are grateful to Katie Oakley, PhD, from Edanz Group for editing a draft of this article.
Author Disclosure Statement
The authors are employees of Japan Tobacco, Inc. All experimental work was funded by Japan Tobacco, Inc. LSI Medience Corporation, Tokyo, Japan, conducted all experimental work except the microarray analyses.
Funding Information
Japan Tobacco, Inc. is the sole source of funding of this project. No external sources of funding were used for this project.
