Abstract
Abstract
To begin development of a mechanistically relevant humane alternative platform for safety assessment of dog food ingredients, comparative in vitro cytotoxicity of 20 ingredients was assessed in four canine cell types relevant for toxicity assessments. Previously, we described the toxicity of 13 compounds (clove leaf oil, eugenol, guanosine monophosphate [GMP], GMP plus inosine monophosphate, sorbose, ginger root extract, cinnamon bark oil, cinnamaldehyde, thyme oil, thymol, lemon grass oil, xylitol, and citric acid) using in vitro primary canine cell culture models for liver, kidney, bone marrow-derived mesenchymal stem cells (BMSC), and enterocyte-like cells (ELC). In this report, dose–response cytotoxicity studies and LC50 using alamar blue assays are reported for seven additional compounds: denatonium benzoate, eucalyptol, hexahydroisohumulone, tetrahydroisohumulone, green tea catechin extract, epigallocatechin gallate, and sodium copper chlorophyllin. Data across 20 compounds were compared between different cell types and responses were not parallel, precluding the use of a single cell line for in vitro ingredient hazard assessment. Hepatocytes were most resistant to all compounds, consistent with their xenobiotic detoxification functions. BMSC and ELC showed an increase in sensitivity to the essential oils eucalyptol, eugenol, and thymol compared to renal cells and hepatocytes. These studies provide a baseline of acute cytotoxicity of 4 canine cell types to 20 different food components that begin to illustrate how such an in vitro panel could be used for hazard assessment.
Introduction
T
Assessment of in vivo safety cannot be accomplished alone using in vitro cell-based assays because the pharmacokinetic processes involving absorption and metabolism are not accounted for, integrated systemic toxicological responses are not detected, immunological hypersensitivity responses cannot be easily determined, and the effects seen after chronic exposure are not amenable to study. Therefore, in vitro studies are useful to assess if unique chemical- and species-specific toxicological responses occur and provide some assessment of organ-specific responses. In vitro studies provide a basis for comparing cytotoxicity data across species when only in vitro data are available. They can be used to predict in vivo toxicity only if independent exposure and pharmacokinetic disposition and metabolism studies are also available.1,5
In an effort to begin development of an alternative platform for safety assessment of dog food ingredients, we developed an in vitro canine cell culture for liver, 6 kidney, 7 bone marrow-derived mesenchymal stem cells (BMSC), and enterocyte-like cells (ELC). 8 These primary cells were selected because they represent major excretory and target organs for any systemically absorbed chemical (liver and kidney) and, in the case of enterocytes, are the initial cell type encountered by food ingredients in the gastrointestinal tract, making them the proximate target cell for direct contact with food ingredients. Use of this diverse cell panel thus provides a range of different cells with widely different biological functions (transporters, metabolizing enzymes, etc.) to assess potential adverse effects after chemical exposure. Initial published studies involved optimization of harvest and cell culture conditions. Cytotoxicity was expressed, using the alamar blue viability assay, as the lethal concentration of the test substance that killed 50% of exposed cells (LC50). Thirteen compounds were initially assessed: clove leaf oil (CLO), eugenol (EUG), guanosine monophosphate (GMP), GMP plus inosine monophosphate (IMP), sorbose (SORB), ginger root extract (GRE), cinnamon bark oil (CBO), cinnamaldehyde (CINA), thyme oil (TO), thymol (THYM), lemon grass oil (LGO), xylitol (XYL), and citric acid (CA). These data, along with studies validating specific positive cellular toxicity markers and other mechanistic endpoints, were published previously for the canine liver, 6 kidney, 7 BMSC, and ELC. 8
The research described herein, and in previous work, represents the first steps to building such an integrated system focused on the dog. The present study used these previously described cell culture systems and assessed the cytotoxicity of seven additional compounds: denatonium benzoate (DB), eucalyptol (EUC), hexahydroisohumulone (HEX), tetrahydroisohumulone (TRA), green tea catechin extract (GTE), epigallocatechin gallate (EPI), and sodium copper chlorophillin (SCC). The previous articles should be consulted for more specific details on the background of the original ingredients tested.6–8
DB is a bittering agent used to enhance bitter flavor, as an aversive additive in household products to discourage consumption of poisonous substances, and also in cosmetics as a denaturant. The human bitterness threshold is 0.05 ppm for the benzoate form of denatonium. 9 DB has been used to discourage consumption of rat poison, ethylene glycol, and methanol-containing products such as antifreeze and wiper fluids. In mice and rats, the LD50 was 865 mg/kg 9 and 485–740 mg/kg, 10 respectively. The G protein-coupled bitter taste receptors (TAS2R) respond to DB stimulation, 11 triggering both metabolic and immunologic responses. DB (10 μM) can promote fat and protein digestion through the stimulation of cholecystokinin secretion in enteroendocrine cells 12 and in stimulating innate immunity as it activates T2R-mediated antimicrobial peptide secretion in primary human sinonasal epithelial cells. 13
EUC (1,8-cineole) is naturally found in thyme (58%), cardamom (73%), cranberry (15%), laurel, rosemary, marjoram citric fruit peel oils, and ginger.14–16 EUC is used as a flavoring agent in baked goods, meat products, and beverages. 15 EUC has antimicrobial activity at IC50 35–62 μg/ml and is not toxic to mouse macrophages or bovine aortic endothelial cells. 16 At an IC50 of 80–94 μg/ml, EUC inhibits HSV-2 in RC-37 cells (IC50 0.3 mg/ml) and infectious bronchitis virus hosted in Vero cells (IC50 10 mg/ml).17,18 It exerts analgesic and anti-inflammatory activity19,20 and inhibits proinflammatory cytokines in lymphocytes and monocytes. 21 EUC is considered weakly genotoxic, causing oxidative DNA damage (771 ng/ml) in cells. 22 A dose of 381 mg/kg of EUC showed subacute renal toxicity in rats based on the accumulation of the eosinophilic protein droplets in the proximal tubule cell cytoplasm. 23
Humulus lupulus L. (Cannabaceae), commonly known as hops, is a preserving and bittering ingredient in the brewing industry as well as used for medicinal purposes. 24 Hops α-acids (humulone) and β-acids (lupulones) constitute the essential bitter resin. Boiling of hops α-acid, humulone, causes isomerization; further hydrogenation yields TRA, with borohydride treatment yielding HEX.25,26 These reduced isohumulones were fed to rats for 90 days and did not cause adverse effects at 150 mg/kg. TRA at 50 mg/kg and HEX at 100 mg/kg fed to dogs for 90 days showed an increase in serum alkaline phosphatase levels that were not attributed to liver toxicity and therefore considered not to cause adverse effects in dogs. 26 C57BL/6N mice fed a diet with 1% isohumulone showed hepatomegaly accompanied by reduced plasma triglyceride levels. 27 Isohumulones also possess anti-inflammatory and anticancer activities. 28 They can inhibit LPS-induced iNOS activity in RAW 264.7 macrophages at 6–18 μg/ml and induce NAD(P)H:quinone reductase activity at 1–10 μg/ml in murine hepatoma cells with cytotoxicity seen at an LD50 > 20 μg/ml. 29
Green tea derived from Camelis sinensis consists of 30% polyphenols termed catechins. 30 The major catechins are (−) epigallocatechin-3-gallate (EPI), (−) epicathin-3-gallate, (−) epigallocatechin, and (−) epicatechin. EPI accounts for 50–80% of catechins, representing 200–300 mg in a brewed cup of green tea. 31 Catechins from green tea have been purported to possess many health benefits, including prevention and/or control of cancer, diabetes, kidney/liver injury, vascular diseases, obesity,32,33 and oxidative stress,34,35 as well as antibacterial and antiviral properties.36,37 However, there are case reports regarding human hepatic failure associated with GTE in Europe.38–42 High concentration of GTE showed pathological changes and mortality in laboratory animals.43–47 High doses of GTE and EPI (>1000 mg/kg) caused hepatotoxicity in mice. 48
Unlike some of the other compounds studied, in vivo catechin studies were reported in dogs. A study showed that 1000 mg purified GTE/kg caused a decrease in body weight and reduced food consumption and death starting at day 9 in a 9-month chronic study in beagle dogs. Subsequently, this study was terminated on day 17 because of morbidity and mortality. A follow-up study for 13 weeks of 200 mg/kg of GTE in fasted dogs resulted in gastrointestinal distress and greater toxicity than dogs in a fed state. 45 In another 13-week study in dogs dosed with 50, 150, and 500 mg GTE/kg/day, the NOAEL in dogs for the fasted state was estimated as 50 mg/kg. 45 Finally, a NOAEL in prefed beagle dogs and in a fasted state were 500 and 40 mg EPI/kg/day, respectively. 44
SCC is water-soluble sodium copper salt derived from chlorophyll widely used at 0.2% as an additive for controlling odor and imparting color to dry beverage mixes. 49 SCC binds to DNA-intercalating mutagenic compounds, potentially preventing chemically induced mutagenesis and carcinogenesis. 50 SCC is a potent antioxidant and effective in scavenging reactive oxygen species. In human peripheral blood lymphocytes, SCC at 0.1–100 μg/ml did not decrease cell viability. 51 SCC at 10 and 100 μg/ml protected cell and DNA damage induced by mycotoxins, ochratoxin A at 4 μM, and fumonisin B1 at 20 μg/ml. 51 Additionally, SCC injected into the abdominal wall at 40 mg/kg/day for 5 days in mice reverted titanium dioxide nanoparticle (500, 1000, or 2000 mg/kg/day)-induced DNA damage in hepatocytes 52 and in mice cardiac cells. 53 SSC also demonstrated antioxidant activity by an increase in glutathione levels, hepatic superoxide dismutase, catalase, and glutathione peroxidase activities. 52 SSC at 4 mg/kg reduced TGF-β signaling, blocked cell proliferation, angiogenesis, invasion, and metastasis, and induced mitochondria-mediated cell death, inhibiting the development of n-methyl-n′-nitro-n-nitrosoguanidine-induced forestomach carcinomas. 54 In aplastic anemic mice fed 25, 50, or 100 mg/kg/day of SCC, mesenchymal stem cell proliferation, osteo differentiation, and immunomodulation were promoted. 55
The aims of this article are to report on the cytotoxicity of these additional compounds across four canine cell systems, to compare chemical ingredient response from all studies conducted to date to begin compiling a multicellular canine in vitro cytotoxicity database, to determine the necessity of conducting such analyses in multiple in vitro cell systems, and to assess if specific compounds exhibited unique responses in specific organ cell types.
Materials and Methods
Chemicals
Lemon grass oil East-Indian type (LGO) CAS 8007-02-1, ginger root extract (GRE) CAS 84696-15-1, and CLO CAS 8000-34-8 were purchased from Spectrum Chemical (Gardena, CA). Guanosine 5′-monophosphate disodium salt hydrate (GMP) CAS 5550-12-9, inosine 5′-monophosphate disodium salt hydrate (IMP) CAS 352195-40-5, cinnamon bark oil (CBO) CAS 8015-91-6, cinnamaldehyde (CINA) CAS 104-55-2, thyme oil (TO) CAS 8007-46-3, thymol (THYM) CAS 89-83-8, citric acid (CA) CAS 77-92-9, eugenol (EUG) CAS 97-53-0, L-sorbose (SORB) CAS 87-79-6, xylitol (XYL) CAS 87-89-0, GTE Cat #G-016, EPI CAS 989-51-5, DB CAS 3734-33-6, sodium copper chlorophyllin (SCC) CAS 11006-34-1, and EUC CAS 470-82-6 were purchased from Sigma Aldrich (St. Louis, MO). TRA CAS 28815-20-5 and HEX CAS 685110-34-3 were donated by Kalsec, Inc. (Kalamazoo, MI).
Animals
The dogs used in these studies were male and female mongrel dogs of indeterminate breed ranging from 1 to 9 years of age (n = 6/liver, 6/kidney, and 6/BMSC and ELC), averaging 26 kg. Dogs were housed individually under normal environmental conditions at 20–23°C, 40–60% relative humidity, and 12 hr light/12 hr dark cycle, and provided with food and water ad libitum. All dogs were anesthetized with a 1:1 mixture of ketamine and xylazine (i.v., 55–74 μl/kg) and within 10 min euthanized with sodium pentobarbital (i.v., 222 μl/kg) after owner consent. Dogs were not bred specifically for this research. The use of these tissues for research purposes was approved by the University's Institutional Animal Care and Use Committee Protocol 3214.
Cell culture methods
The methods used to harvest and culture the cells have been described in great detail in the previous publications for canine primary renal proximal tubule cells of the kidney, 7 canine hepatocytes for the liver, 6 and BMSC and ELC differentiation. 8 For all systems, cells were seeded on 96-well plates and test chemicals dosed at concentrations limited by specific chemical solubility in medium (n = 3 for hepatocytes and n = 6 for proximal tubule cells, BMSC, and ELC wells/concentration) for 24 hr. Cell passaging ranged from 9 to 12 for BMSC and ELC, and 4–5 for proximal tubule cells, and hepatocytes were isolated and used directly for these experiments. The alamar blue (aB) viability assay (Invitrogen, Carlsbad, CA) was used to assess the cytotoxicity of all ingredients. Cell viability was assessed by adding a 1:10 dilution of the aB reagent to the cell culture medium followed by 3 hr of incubation. Fluorescence was detected using a synergy H1 hybrid multimode microplate reader at Ex555/Em585nm (BioTek Instruments Inc., Winooski, VT). Cell viability, which is proportional to an increase in fluorescent intensity, was normalized to controls and expressed as percent viability at mg/ml of the test compound.
Data analysis
LC50 was determined using statistical regression in OriginPro 8 SRP software (OrigeLab Corp., Northampton, MA) with resulting data expressed as mean ± SD for n = 3 dogs per compound treatment. In some cases, cell viability was not reduced at the solubility-limited highest concentration tested, which precluded determination of an LC50. In these cases, data are denoted as nonlethal (NL) and were not included in the radial compass or 3D plots. Radial compass plots were generated using Microsoft Excel 2010 (Microsoft, Redmond, WA) and 3D plots were generated using SAS 9.3 (SAS Institute Inc., Cary, NC). Correlation coefficients were determined between LC50 of liver (used as point of reference) versus the three cell types for all chemicals with LC50 values using GraphPad Prism 6 (GraphPad Software Inc., La Jolla, CA).
Results
Dose–response plots and LC50 results for DB, EPI, EUC, GTE, HEX, SCC, and TRA are depicted for canine renal proximal tubule cells (Fig. 1), hepatocytes (Fig. 2), ELC (Fig. 3), and BMSC (Fig. 4). Table 1 is a tabulation of the LC50 for the compounds reported, as well as those previously reported for the canine renal proximal tubule cells, 7 hepatocytes, 6 BMSC, and ELC. 8 Correlation coefficients for regression analyses between the liver (as reference for all compounds for which statistically valid LC50 values could be obtained) and kidney cells, between the liver cells and ELC, and between the liver cells and BMSC were 0.934, 0.991, and 0.990, respectively (all p < 0.0001). This complete data set for all compounds with estimated LC50 and the cell types is graphically compared using a radial compass plot (Fig. 5) and a 3D plot (compound vs. cell type vs. LC50) (Fig. 6). In Figures 5 and 6, compounds are ordered by decrease in toxicity of the hepatocytes as the reference cell type.
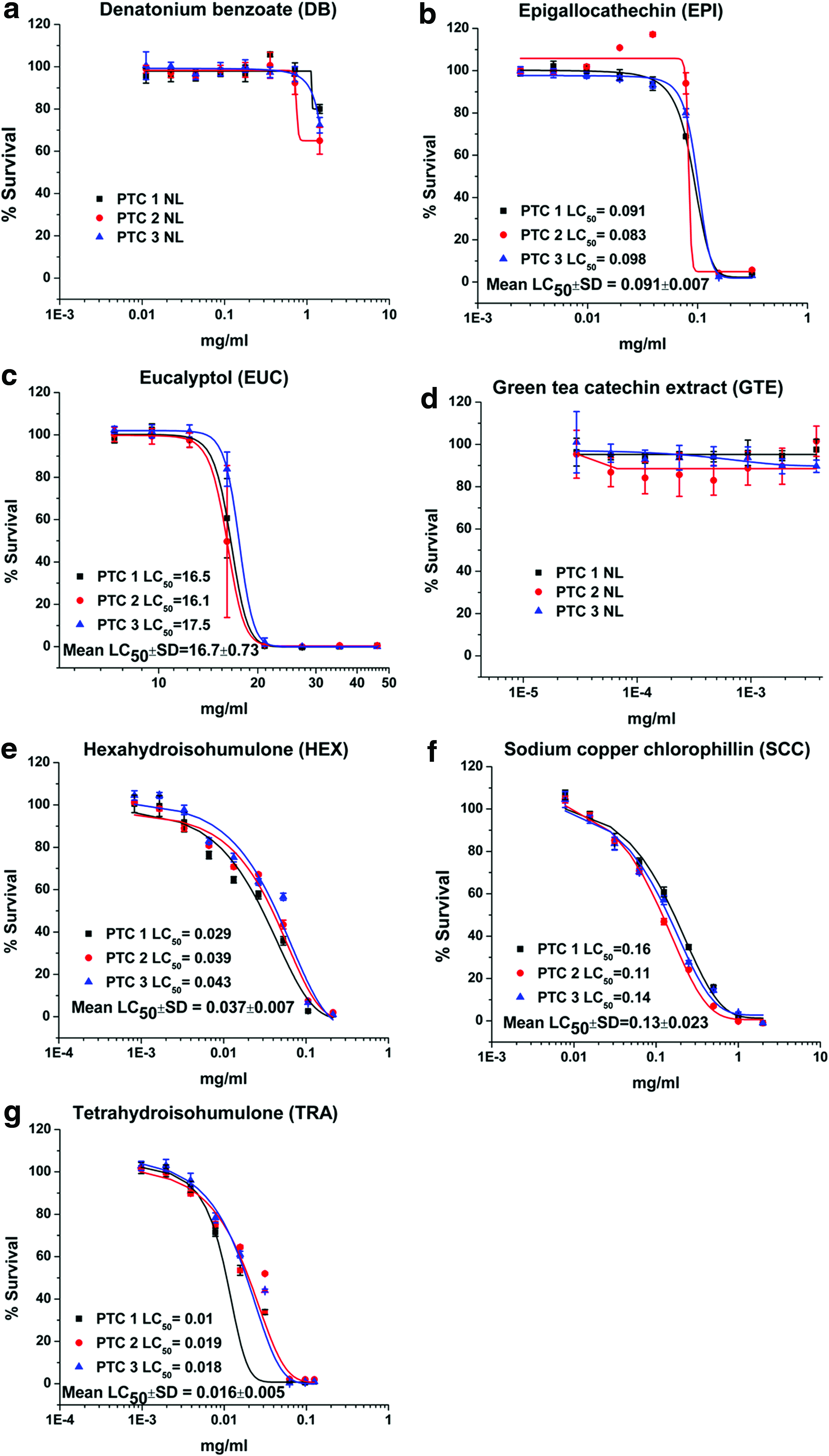
Viability and LC50 (mg/ml) for
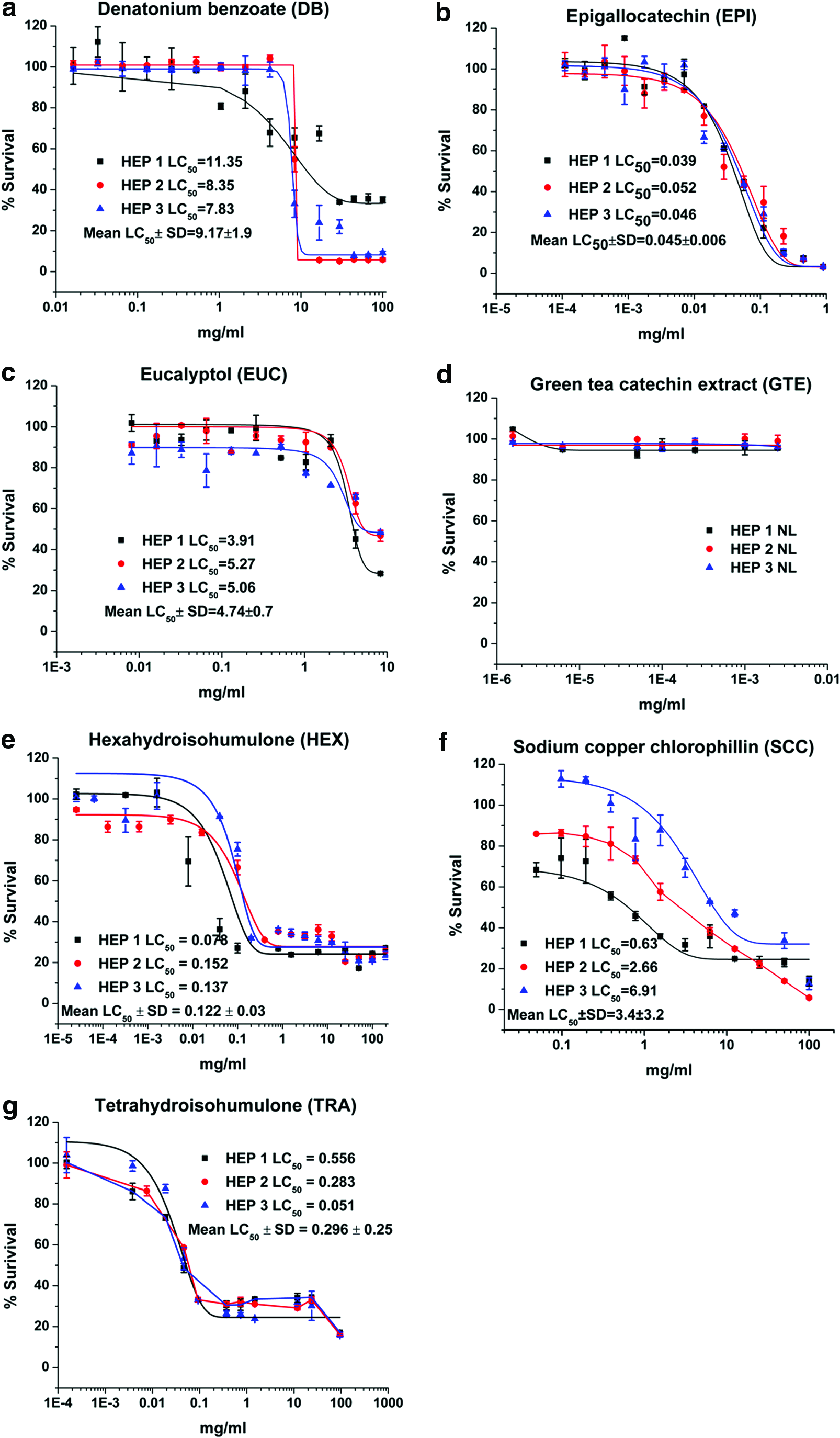
Viability and LC50 (mg/ml) for
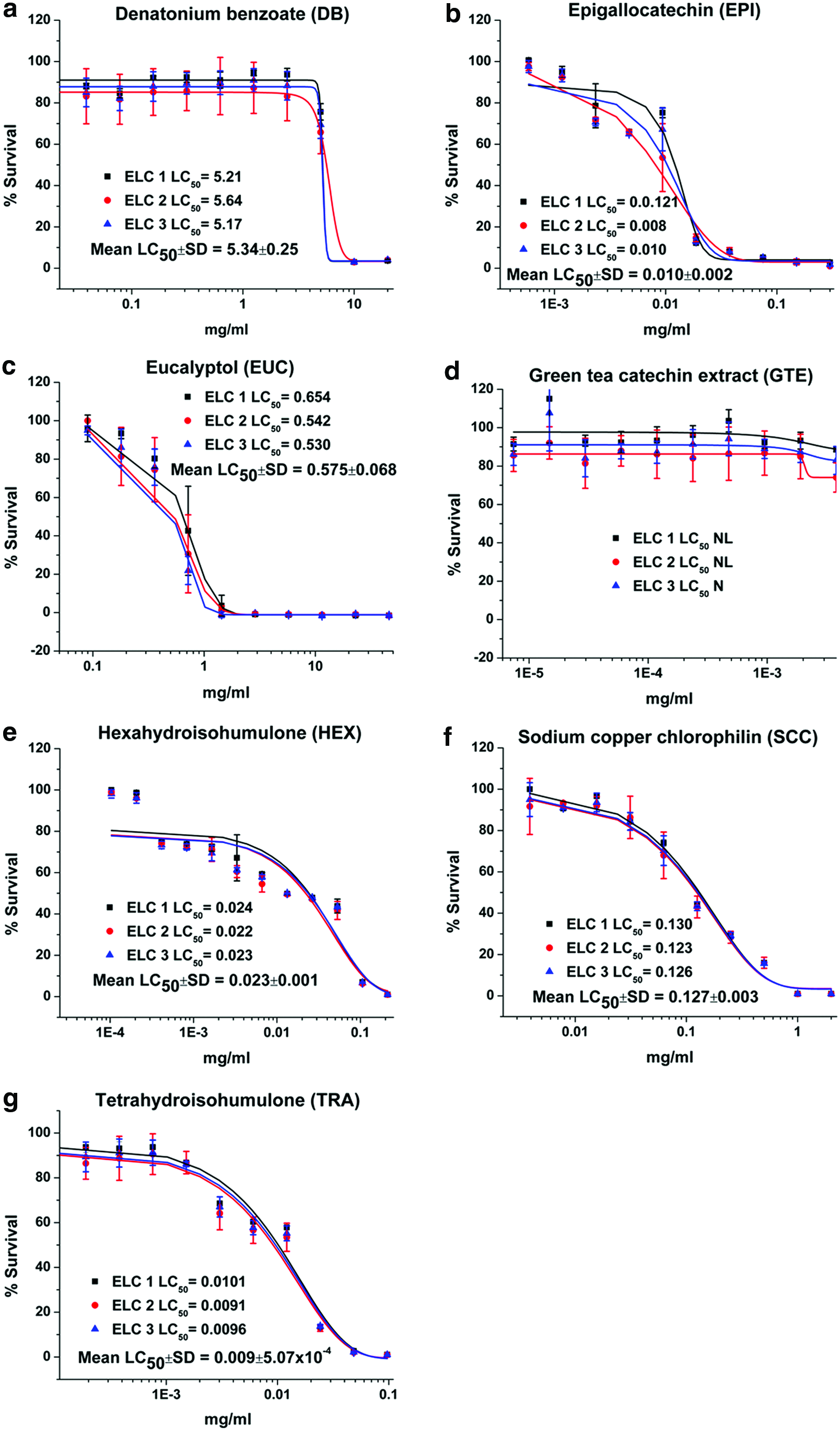
Viability and LC50 (mg/ml) for
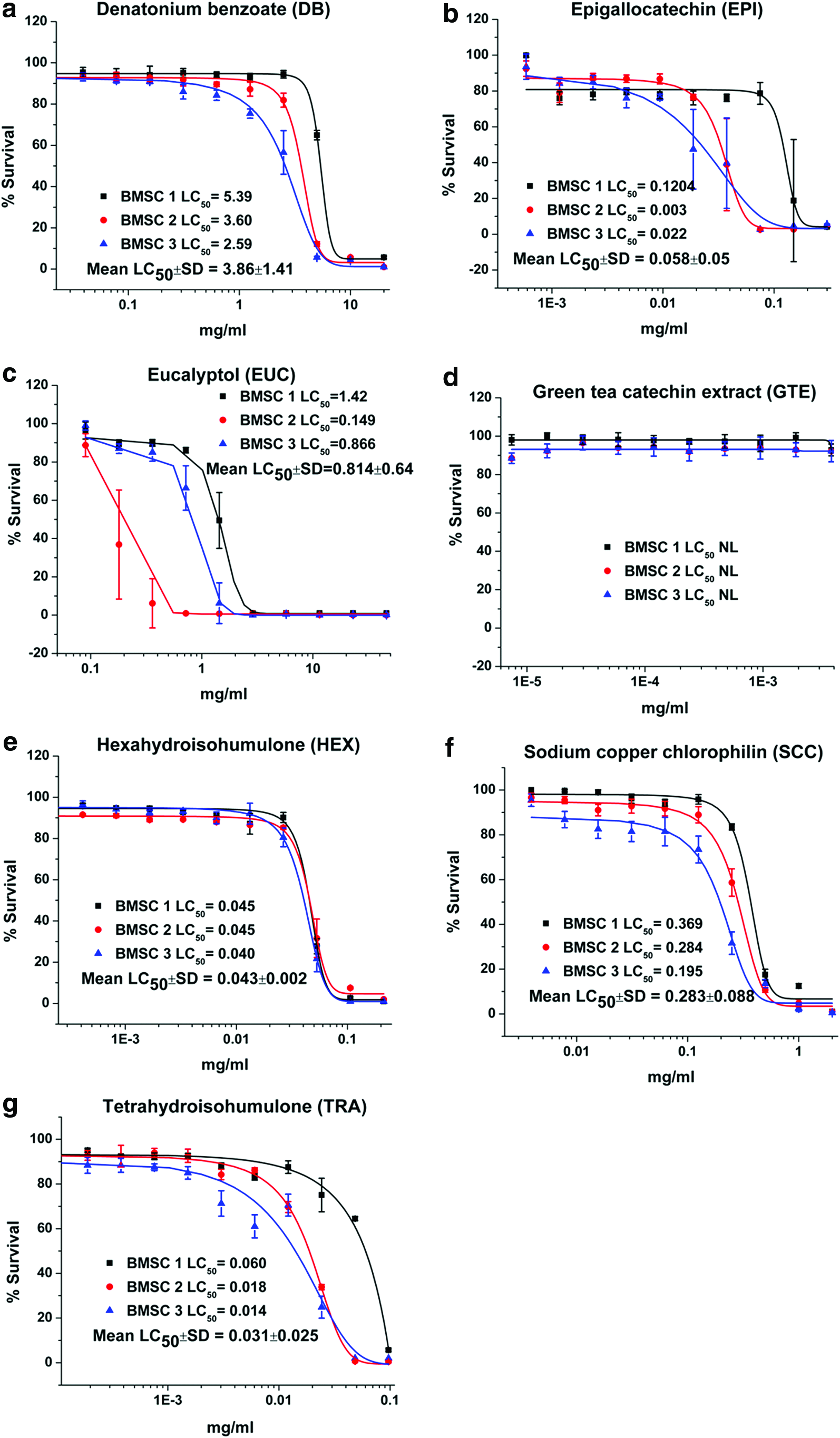
Viability and LC50 (mg/ml) for
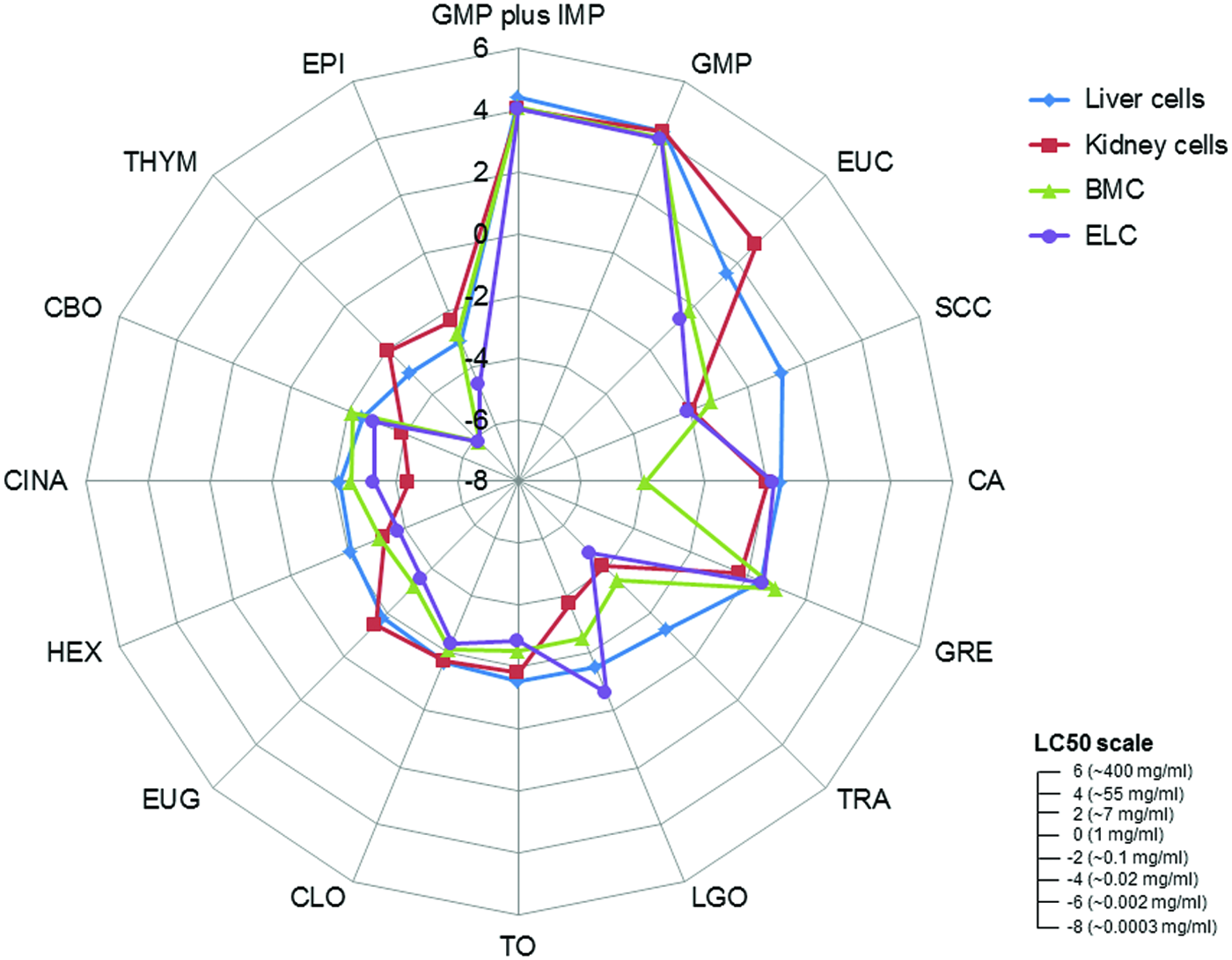
Radial compass plot comparing 16 compounds (compounds designated as nonlethal were not included) calculated as ln LC50 (mg/ml) across all cell lines. LC50 are plotted from the lowest value at the center of the circle outward in ln units. Ruler insert depicts approximate comparison between ordinal and logarithmic LC50 values. Compounds are plotted clockwise starting at noon in order of decreasing hepatocyte LC50. Color images available online at www.liebertpub.com/aivt
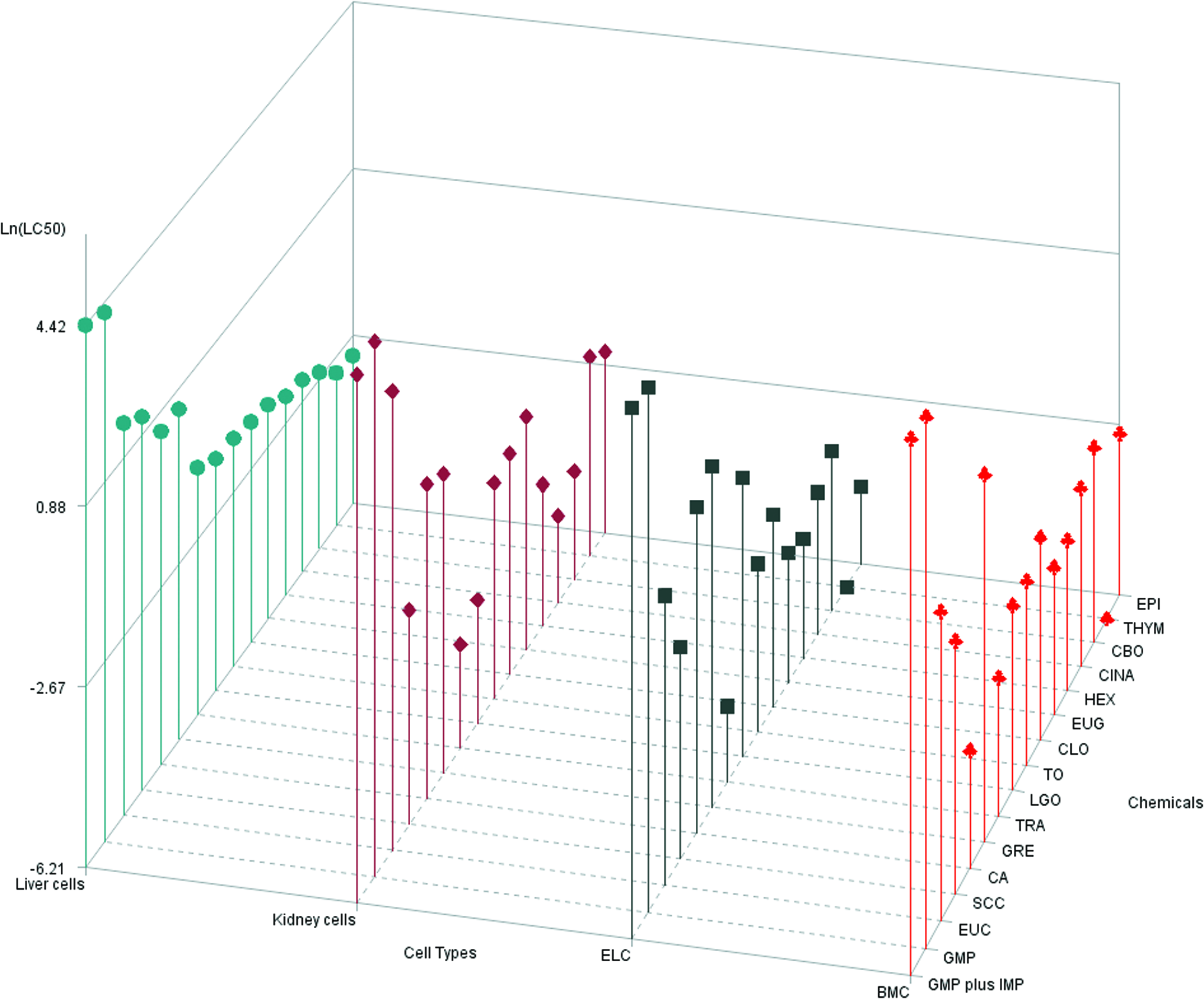
Comparison of ln LC50 (mg/ml) across all 16 compounds (compounds designated as nonlethal were not included) and 4 cell types. Compound LC50 are plotted in decreasing order of hepatotoxicity (left side of graph) to demonstrate different rank orders of toxicity across cell types. Color images available online at www.liebertpub.com/aivt
NL indicates that no toxicity was observed or that LC50 values could not be calculated at the highest concentrations tested.
Not surprisingly, the toxicity of these very different chemicals has an LC50 ranging from 0.0003 mg/ml to greater than 400 mg/ml—a million fold difference. Some compounds are obviously potent in certain cell types, while others are essentially nontoxic. An interesting comparison is the difference between a complete extract versus one of its components, such as GTE vs. EPI or CLO vs. EUG. In the case of green tea, EPI is significantly more toxic than the GTE. In contrast, with clove leaf oil both CLO and EUG are similar. TRA and HEX provide another interesting comparison. As indicated in the Introduction, the α-acid humulone HEX is a derivative of TRA. In our studies, there are cell-specific differences in potency across cell lines for TRA/HEX LC50 [ratio]: hepatocytes 0.296/0.122 [2.42], proximal tubule cells 0.016/0.037 [0.43], ELC 0.009/0.023 [0.39], and BMSC 0.031/0.043 [0.72]. Hepatocytes are both less sensitive to TRA (Fig. 1g) and HEX (Fig. 1e) compared to other cell types, and have less sensitivity to TRA [ratio >1] compared to the other cell types [ratio <1]. Hepatocytes appear to be relatively resistant to SCC (Fig. 2f) compared to the other three cell types. Finally, BMSC are much more sensitive to CA exposure than the other three cell types.
There are a number of interesting trends that can be seen from these data. The first is that the rank order of compound toxicity between different cell types is different, an observation supported by the different correlations (using hepatocytes as the reference cell line) seen in Table 1. The kidney represented by proximal tubule cells is prominent in this regard. The 3D plot (Fig. 6) allows for easy visualization of the rank order of all compounds across all cell types. In this case, the compounds are ordered by their rank of toxicity in the liver (leftmost pattern), which clearly shows that different order is needed for kidney, ELC, and BMSC. In most cases, hepatocytes are relatively resistant. This suggests that knowing the toxicity in one cell type does not necessarily predict toxicity in another cell type.
The compass plot (Fig. 5) clearly depicts that individual compounds have different responses across all four cell types. These data are again ordered by liver toxicity. Some compounds have very similar responses (e.g., GMP, GRE, TO, and CLO) in all four cell types, while others (e.g., EUC, LGO, CA, and THYM) differ across all cell types or are only different in one cell type (e.g., CA and TRA). THYM, EUG, and EUC show greater toxicity in BMSC and ELC compared to kidney or liver cells.
Discussion
This work presents the first compilation of comparative toxicity data on a wide variety of chemicals in four different defined primary canine cells that have relevancy for toxicity assessments. ELC represents gastrointestinal-like cells that are in direct contact with food ingredients and would be expected to reflect responses such as acute irritation leading to gastrointestinal distress. Hepatocytes and renal proximal tubule cells represent elimination and biotransformation functions that are responsible for compound elimination and potentially reflective of in vivo liver or kidney toxicity. BMSC are relatively nonspecialized young differentiating stem cells that express ABC transporters 56 but do not have the full complement of cellular functions associated with fully differentiated cell types, such as expression of intestinal alkaline phosphatase 57 and fructose transporters. 58 Our previous study 8 should be consulted for a full description of the differences between these cell types.
The first observation is that responses between different cell types do not parallel one another, thereby precluding the use of a single cell line for in vitro assessment of ingredient hazards. However, there are some general patterns that can be noted. Proximal tubule cells have the weakest correlation to hepatocytes (Table 1), a finding potentially consistent with the numerous transmembrane transport systems present in these excretory cells, which the other cell types do not possess. Hepatocytes tended to be more resistant to all compounds studied, a finding consistent with their xenobiotic detoxification functions. An interesting finding was the unique response of hepatocytes to the hop-derived humulones, HEX (Fig. 2e), and its hydrogenated precursor TRA (Fig. 2g). Hepatocytes were relatively resistant and showed an inverse ratio of sensitivities to TRA versus its reduction product HEX, compared to all cell types. This suggests a unique hepatic response to these hop-derived humulones that would not be predicted from studying the other cell types, again most probably secondary to the specialization of hepatocytes for xenobiotic biotransformation. 5 The most sensitive cell type to these humulones is ELC, especially to TRA, which would be the first cells encountered after oral exposure.
It is not appropriate to conduct detailed comparisons across compounds because chemicals were selected for various reasons, and not to define a balanced and representative chemical property space needed for a balanced quantitative structural activity analysis. In addition, the properties of these compounds were very different, ranging from plant extracts such as GTE, to complex natural aromatic oils like LGO, to specific individual components and chemicals. Many of these natural compounds are complex mixtures obtained from plant extracts, and thus contain multiple potentially active ingredients. For some plant-derived compounds, geographical location and time of harvest can effect a mixture's composition. Methods of extract production (water vs. alcoholic reduction) would yield different constituents, making any interstudy comparison difficult unless the same preparation was used.1,59
In one specific case in this present study, an extract (EPI) was much more potent than the material from which it was derived (GTE), while for another pair (CLO vs. EUG), potencies were similar. As discussed in the Introduction, GTE has been reported to be toxic to dogs, causing lesions in the intestinal mucosa, liver, and kidney after chronic exposure, and more pronounced in the fasted versus the fed state. When these dogs were exposed in the fasted state for 13 weeks, gastrointestinal distress was observed in the absence of severe liver or kidney toxicity. 45 In our studies, GTE was nonlethal for all cell types, but its component EPI was very toxic across all cell types, especially ELC (Fig. 3b), suggesting that any in vivo effects observed would be related to this component. These findings are consistent with the gastrointestinal toxicity seen in vivo as ELC was the most sensitive cell.
Another finding was the increase in sensitivity of BMSC and ELC compared to renal or hepatic cells to the essential oils EUC, EUG, and THYM. EUC is a cineol compound, whereas both EUG and THYM are phenolic derivatives. It is likely that the lower cytotoxicity observed for these compounds in renal and hepatic cells is because of these cell's increased capacity for biotransformation of these readily metabolized compounds. Our finding of increased ELC toxicity is consistent with essential oil toxicity reported in humans with manifestations including vomiting and diarrhea, consistent with gastrointestinal irritation.60,61 Guidelines for use of most essential oils in pets recommend against internal exposure, though all reports are anecdotal.
Hepatocytes were resistant to the water-soluble sodium copper chlorophyllin (Fig. 2f) compared to other cell types, consistent with some observations of gastroenteritis as an early sign of acute copper ingestion. 62 It is possible that the increased enzymatic capacity of the hepatocyte may offer protection not seen in the other cell types. The BMSC was uniquely sensitive to the CA exposure compared to the other three cell types (Table 1). Because BMSC are young differentiating cells, they may be more sensitive because of lack of developed detoxification systems. The compound CA might also lower the pH, and so it makes it unsuitable for the BMSC to survive because they may not be able to handle acid stress as could more differentiated cells.
The kidney proximal tubule cells were the most sensitive cell type compared to other cells only for LGO exposure (Table 1). As previously reported, gene expression arrays suggested oxidative stress as the primary mechanism in renal cells, in contrast to hepatocytes and BMSC, which showed upregulation of cytochrome P450 enzymes, and ELC showed upregulation of genes involved in heat shock response, beta oxidation, and mitochondrial energy metabolism.6–8
The relevance of single-compound cytotoxicity data to effects observed when included in food is also difficult to predict because interactions with other food constituents (e.g., binding and chemical reactions) may occur. 63 Food served as a protective mechanism in the GTE exposures discussed above. 45 High-temperature or high-pressure treatments aimed to process and preserve food may decompose some of these chemicals. Compounds in essential oils may volatilize if heat extrusion processes are used in manufacturing. These issues are common to all test systems that do not assess safety of finished products, including in vivo dosing of the pure compound.
The use of alternative toxicity systems such as described herein reduces the use of animals to conduct initial hazard assessments of novel food ingredients. They are useful for probing mechanisms of toxicity in specific cell types when in vivo exposure is known to produce poisoning. In fact, this is consistent with our use of aflatoxin B1 and 4-aminophenol as positive controls when we developed these assays in hepatocytes 6 and in proximal tubule cells, 7 respectively. There is an ethical concern in conducting a hazard assessment in dogs for products destined for use in dogs. Use of in vitro cell panels begins to assess this concern.
In vitro cytotoxicity assays presented here are useful for many purposes, but also have inherent limitations relative to the ability to extrapolate these results to in vivo responses. When comparative in vitro data are available across multiple species, relative sensitivities can begin to define interspecies comparisons. Such data are also very helpful to design experiments to define (1) mechanisms of compound action, (2) pathways of compound entry into cells, and (3) interactions with other compounds. The primary missing pieces for directly using in vitro data alone for in vivo hazard assessment include (1) absence of knowledge of the delivered target dose to tissues that requires in vivo biodistribution or pharmacokinetic analyses; (2) assessing only parent chemical toxicity, which precludes predicting the toxicological responses if either the parent drug is deactivated to nontoxic metabolites or if the parent compound is bioactivated to toxic intermediates; (3) nonabsorbance of the compound, preventing it from having any systemic effect; and (4) finding toxicological effects only after chronic administration. A consistent finding across all compounds studied was the relative resistance of hepatocytes to cytotoxicity for the compounds evaluated—a finding phenomenologically consistent with their function as the cells responsible for xenobiotic detoxification through biotransformation. However, should a specific compound exert its toxicity through metabolic bioactivation, hepatocytes would be most sensitive.
The ELC model does not have these pharmacokinetic limitations because the concentration presented is within the range of that encountered in the gastrointestinal tract in vivo, and direct exposure occurs in both cases. Even in this scenario, intestinal bacterial metabolism of a compound could alter the response. In some cases, in vivo effects would be immunological in origin or mediated from the central nervous system (e.g., emesis through activation of vomiting center) and not amenable to in vitro detection in a single cell type. However, it has been suggested that the primary effects of most xenobiotics are manifested within the gastrointestinal tract and expressed as protective emesis or diarrhea. 64 We speculate that, if such compounds are acutely toxic to ELC, gastrointestinal distress evidenced by emesis or diarrhea would be observed.
In conclusion, these studies provide a baseline of cytotoxicity responses of 4 canine cell types to a wide variety of 20 different food components that begins to illustrate how such an in vitro panel could be used for hazard assessment. They clearly show the potential strengths of alternative model systems and limitations of such in vitro systems and shed light on interesting activities of some select compounds.
Footnotes
Acknowledgments
This study was funded by Mars Inc. Global Quality and Food Safety, McLean, VA. Parts of this research were presented at the 54th annual meeting of the National Society of Toxicology in San Diego, CA.
Author Disclosure Statement
No competing interest exists for any of the authors except B. Jeffery, who is employed by Mars Inc.
