Abstract
Cancer care often involves making complex medical decisions within a challenging environment: a balkanized medical system of many specialists, information overload and obsolescence, limited time with doctors, and siloed data. New AI tools can enable patients and caregivers to more actively participate in treatment decisions. For example, consider receiving a complex scan report in a patient portal. While today, a patient may face confusion in interpreting a complex diagnostic report, a personalized generative AI agent could help by translating the scan report into language a patient can understand, and even contextualizing it within a patient’s personal health history and clinical evidence/guidelines. By providing an understandable version of the report and the clinical context of their test results, patients and their caregivers can engage more fully in decision-making with their oncology team. As described by Clayton Christensen’s “The Innovator’s Dilemma,” industry incumbents typically do not adopt disruptive technologies for fear of cannibalizing existing revenue streams (such as the case of Kodak and digital photography). This leads incumbents to serve their existing customers with the same value proposition, while ignoring “disruptive innovations” that offer a new value proposition to underserved customers (such as the transistor radio, which enabled teenagers to take their music with them). This theory predicts that institutional health care will focus on adopting AI for incremental operational improvements (e.g., patient scheduling, scan interpretation, claims processing). In this review, we argue that a positive disruptive impact of AI in oncology can come if AI-enabled software agents are used to support patients and caregivers in seeking better outcomes through personalized care. We review existing gaps and challenges that patients face as they go through receiving a cancer diagnosis, selecting a treatment plan, and then living with cancer. We use this process view to map uses of novel AI technologies that can assist patients and caregivers. This allows us to describe how current AI technologies/research and policies need to evolve to support patients and caregivers.
Introduction: Engaged Patients and Caregivers Are Critical to Achieve the Transition to Personalized Medicine
Precision medicine has transformed oncology practice, especially in the advanced setting. In precision medicine, advanced diagnostic testing is used to stratify characteristics of tumors at a population level, which are used to match patients to optimized care guidelines (e.g., matching all patients with advanced Non-Small Cell Lung Cancer [NSCLC] and an EGFR mutation to an EGFR tyrosine kinase inhibitor [TKI]). While precision oncology has given us many new therapeutic options that benefit subsets of patients, these therapeutic options may provide patients with limited survival improvement, 1 while clinical implementation challenges have limited the number of patients who successfully start a medication that is matched to their tumor’s molecular signature.2,3 Implementing existing guideline-based genomically driven care has exponentially increased clinician workload, 4 and contributed to gaps in patient education that inhibit empowered and informed patient-driven decision-making. 5
This combinatorial complexity of precision medicine contributes to the daunting mental load that cancer patients and caregivers face. The empowerment of patients and caregivers with software agents and information can create a sea change in the current medical system, placing patients and caregivers first and protecting them from supply-side failures, such as the following:
By focusing on a tumor’s molecular profile instead of a patient’s holistic health and personal treatment goals, health care providers run the risk of placing patients on medicines that provide marginal improvements in response but that have high out-of-pocket costs and provide negligible overall survival or quality-of-life improvements. 2 Escalating out-of-pocket health care costs pose a financial burden to patients, especially those patients who are managing complex or chronic diseases and conditions: when patients do not understand the role of their medications or are unable to finance their own care,9,10 they are unlikely to adhere to their medication plans. Similarly, adding complex precision medicine decision-making tasks (such as considering how therapy order or combination may improve overall survival) can help patients to preserve treatment options or avoid patients’ developing treatment resistance 11 at the cost of increased physician workload and strain on the system. While some molecular tumor boards and clinical trials have attempted these approaches, 12 they may be economically challenging to support at the scale of the entire American health care system.
We see the imperative in moving from population-level precision medicine to patient-centric personalized medicine as a critical step in closing trust and access barriers that patients face, and that severely impact the day-to-day lives of patients and clinicians. While new reimbursement pathways aimed at improving access to care navigation could help to make this level of care financially viable within well-resourced health systems, 13 successfully addressing patient needs across the cancer care continuum would necessitate growing a large de novo workforce of navigators with specialized skills to assist with financial 9 or clinical trial counseling. 14 Rigorously constructed AI systems can be deployed at scale with a lower cost than human workers. 15 If applied to care navigation tasks, agentic AI has the potential to bridge long-standing informational gaps that contribute to poor outcomes for patients. Examples of this include improving adherence to treatment protocols and the management of side effects by educating patients about their disease and treatment, 5 and helping to address information gaps that degrade patient trust in the clinical trial system 16 by helping patients to seek care that is aligned to their personal risk tolerance and survival and quality-of-life goals.
In this review article, we review how patients experience cancer care today, with a lens toward biomarker-driven precision medicine. By reviewing the research literature on clinical implementation gaps and patient/caregiver sentiment, we identify four areas where patients and caregivers routinely experience navigational gaps. We use these gaps to identify key navigational workflows where an AI-powered agent could support empowered patients and caregivers, and review existing computational tools (both AI and non-AI) that provide solutions to these workflow steps. These workflows include logistical care navigation (finding the best health care experts, coordinating communication between patients, caregivers, their medical team, and friends and family), clinical monitoring and educational support (test report interpretation, education about their condition and testing and treatment options, monitoring and guiding the patient through therapy to help avoid care gaps), and addressing care access barriers (supporting reimbursement, cross-specialty care, and trial access).
We conclude by reviewing the trust/safety, regulatory, and workflow integration challenges that patient-centric AI must address for successful deployment. The success of AI-enabled software agents will depend on both technical solutions to current weaknesses (such as bias, knowledge cutoff where large language models [LLMs] cannot accurately answer questions that depend on knowledge that has changed since they were trained, 17 and hallucinations where LLMs generate unsupported “factual statements” 18 ) and the thoughtful integration of software agents into existing patient support workflows (navigation, care coordination, and patient education) through patient-centered design and workforce education. The concerted action of patient advocates and policy lobbyists has driven disruptive changes in the American health care through actions such as the National Cancer Act in 1971, the Americans with Disabilities Act of 1990, the National Cancer Survivorship Act of 1996, ACT UP/AIDS Coalition to Unleash Power, and the Affordable Care Act. As health care recovers from the COVID pandemic and starts to invest in AI technologies, we have the opportunity to use lessons learned from past patient advocacy movements and the COVID pandemic to address the key gaps that patients encounter with precision medicine.
A Significant Opportunity for AI in Medicine is to Address the Challenges That Patients and Caregivers Face
Flaws in the structure of health care markets lead to financial returns (and thus power) accruing to large enterprises (such as health insurance companies, pharmaceutical companies, and hospital systems), at the expense of patients and caregivers.
19
This creates powerful systemic incentives that can conflict with patients’ best interests. An example of this is the increasing cost of cancer medications. Surveys of patients indicate that patients are skeptical of the value of extremely high-priced oncology medications that provide marginal survival benefits.
20
However, the approval and pricing of these medications have direct and indirect systemic effects on patients:
Existing oncology AI tools have predominantly been developed by researchers working within the health care enterprise, who experience organization pressures to develop innovations that “sustain” their existing market. While the term “disruption” is used colloquially to describe new technical innovations and business models, Clayton Christensen’s Theory of Disruptive Innovation provides a more formal definition. 27 This theory distinguishes between sustaining and disruptive innovations, as shown in Figure 1: a sustaining innovation is a technological innovation that allows an incumbent technology to sustain its advantage in an existing market (with the same processes, value proposition, and business model), while a disruptive innovation is a technological innovation that allows a technology to enter new, previously inaccessible markets for an underserved customer (typically with a new value proposition and business model). Disruptive innovations typically reduce the cost of providing a service/technology by more than an order of magnitude (enabling new market entry), but may not have the performance at introduction necessary to be used in existing “high-end” applications.
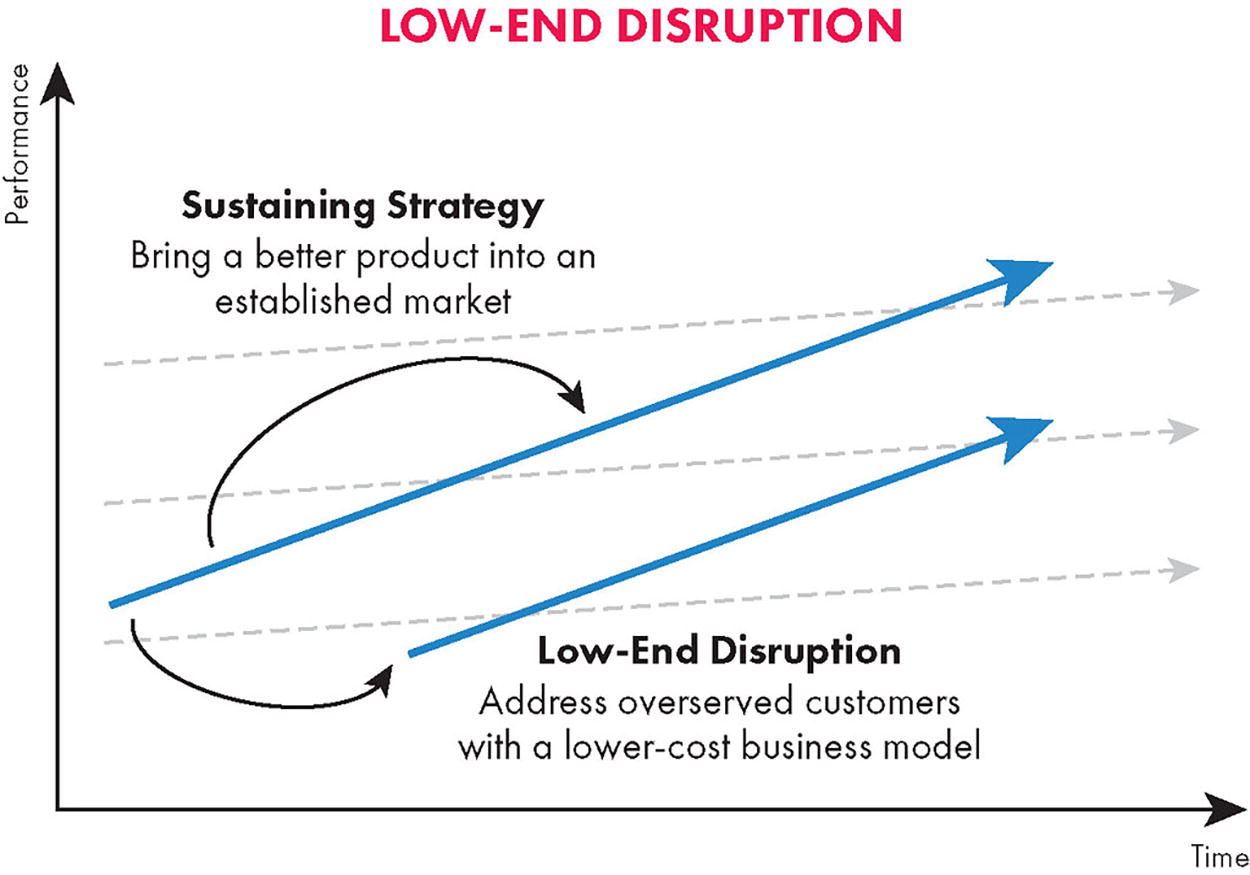
While the term “disruption” is often used colloquially to describe new market entrants, Christensen’s theory of disruptive innovation 27 distinguishes between “sustaining innovations” (which drive improvements in existing markets, e.g., improving the efficiency of working in an EHR) and “disruptive innovations,” which create new markets through innovations that reduce the cost of delivering a service to currently un- or underserved consumers. (Fig. is reproduced from Kalbach, 2020, 28 under a Creative Commons CC-BY 2.0 license.).
The idea of using artificial intelligence to “disrupt” health care is not new; many oncology clinicians and researchers remember both the promises of how the IBM Watson platform could change medicine 29 and the limited reality of what it achieved. 30 While IBM Watson never achieved the accuracy and usability necessary to be a clinical companion, market forces also limit how institutional health care can adopt disruptive innovations. This can be seen in the ongoing dialogue about how health care will pay for AI, which focuses on fitting ambient AI applications into the existing “fee-for-service” reimbursement model.31,32 In this model, the profitability of a hospital or health care practice is effectively determined by the productivity of billable procedures (the mix of procedure quantity and value) that a hospital/practice completes, and the overhead costs of the hospital/practice (what is the cost of running shared services, such as the electronic health record [EHR] or hospital call center). This model inhibits the adoption of technologies that improve patient experiences and outcomes, 33 unless the technologies are attached to reimbursement codes or governmental (dis)incentives.
Early experiences with generative AI have encountered this issue, since existing reimbursement pathways typically mean that an AI tool has to look similar to a “procedure” or “medical device.”32,34 As such, early adoption has primarily accrued to diagnostic AI, or to “sustaining technologies” that provide a clear productivity-based financial benefit to hospitals such as AI scribes that improve physician productivity. While value-based care (VBC) reimbursement models, such as the Center for Medicare and Medicaid Services’ “Enhancing Oncology Model” can provide additional leverage for the adoption of technologies that improve patient outcomes, initial patient experiences have been mixed to slightly negative, 35 as VBC models are still centered on institutional health care and have not historically comprehensively incorporated patient’s experienced outcomes. 36 Ultimately, the current reimbursement model externalizes these costs to patients, in both high out-of-pocket costs and poor care experiences.
Given the challenges of health care reimbursement and health IT incumbency, is the most viable path for disruptive innovation to use generative AI to address patient’s information needs? If AI continues to improve and it becomes cost effective for patients to have on-demand 24/7 access to a doctor with generalist knowledge across all medical specialties, we can enable a number of patient education tasks that go underserved due to health care staffing pressures that have reduced the amount of time that oncology care providers can allocate to patient education. 37 The information needs of a person living with cancer change dramatically over the course of treatment, and vary given their personal and family health histories. Several studies38,39 have used patient interviews to identify common phases that patients experience; Figure 2, reproduced from Hall et al., 39 shows how patients’ support and information needs change over time. Patients with advanced or recurrent/refractory cancer will pass through these phases multiple times, with differing needs over time: if their cancer is staying well managed, they may seek education that can help them mitigate quality-of-life issues related to treatment, 40 while patients with progressive disease may be seeking education about treatment beyond the standard of care or clinical trials. 14
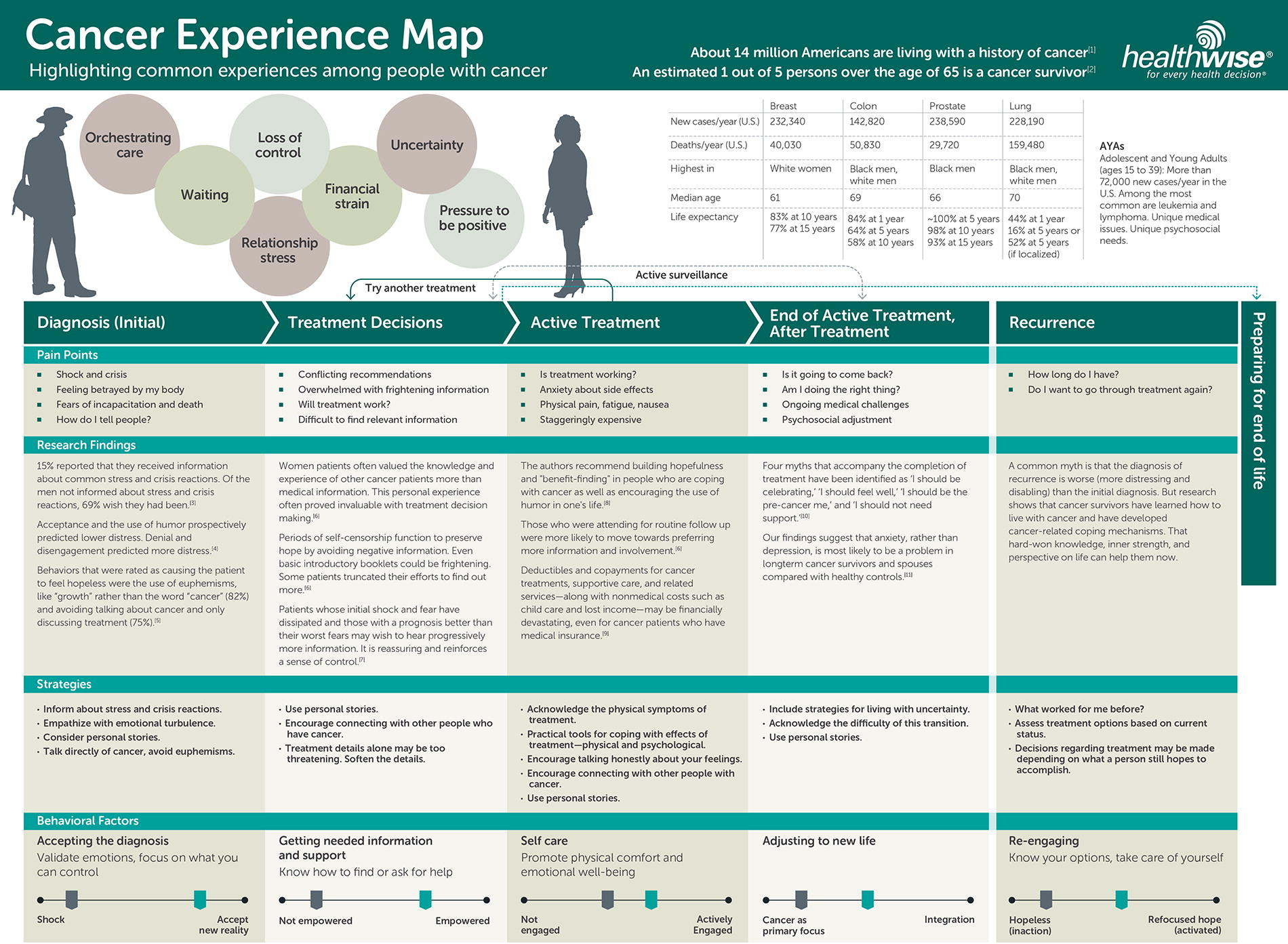
Cancer experience map, reproduced from Hall et al., 39 under a Creative Commons CC-BY 2.0 license. This map shows how cancer patient’s day-to-day experience, needs, and level of knowledge change over time. After an initial diagnosis, patients face a challenging learning process driven by the complexity of oncology, and the shock of receiving a diagnosis. Patient engagement typically increases over time, as patients’ knowledge grows, but can be hampered by factors such as adverse events or other life stressors.
Currently, the vast majority of patients turn to the internet to educate themselves about their conditions and care options: a 2021 survey that was conducted by the American Health Information Management Association Foundation 41 found that up to 94% of patients use Google to answer health-related questions. As their condition changes and their learning needs evolve, patients and caregivers may move from generalist resources such as Google or WebMD’s Cancer Resource Center to more specialized web resources that educate them about genomics (including general resources such as MyHeritage’s Promethease or the Wellcome Genome Center’s YourGenome.org, or gene/biomarker-focused resources such as the Global Alliance for Genomics and Health's [GA4GH] BRCA Exchange 42 or International Cancer Advocacy Network's [ICAN] Biomarker Collaborative), their medication options (such as the National Comprehensive Cancer Network's [NCCN] Guidelines for Patients), or potential clinical trial options.
However, searching for information about oncology care on the internet is time-intensive, prone to error and misinformation, 43 and is often inaccessible to patients and caregivers with limited English proficiency 44 or disabilities. 45 In addition, common oncology terminology such as “salvage chemotherapy” or “palliative treatment intent” can have negative connotations that may dissuade patients from pursuing care that they may benefit from 46 and can contribute to worse outcomes through the “nocebo” effect.47,48
Early research applying LLMs to patient communication has shown that LLMs have the potential to communicate with patients in a highly empathetic manner.
49
However, specialization is needed for LLMs to perform in a trustworthy manner when interacting with patients. Moor et al.
50
proposed three necessary capabilities for “generalist” medical foundation models; here, we reframe these capabilities to focus on patient-facing navigation workflows:
In the rest of this section, we use the patient journey map as a guide and identify four thematic areas where patients and caregivers commonly encounter challenges in making fully informed decisions. While generative foundation models face challenges with hallucinations that must be resolved (which we discuss later in this article), generative AI technologies may provide an opportunity to meet navigation needs in a way that improves patient trust and reduces the complexity of navigating oncology care.
Treatment volatility and complexity
The rate of oncology drug development has accelerated dramatically in the precision medicine era. In the decade following the approval of the initial “precision oncology” medicine imatinib (2002–2012), the FDA approved 65 new oncology medicines. 24 In 2018 alone, the FDA issued 46 new oncology medicine approvals, which applied to more than 400,000 cancer patients in the United States. 54 There are now over 180 precision oncology treatments with more than 90 companion biomarkers. 55
While having more treatment choices benefits patients and oncologists, it also increases the complexity of decision-making. As a proxy, we see evidence of growing complexity in oncology practice guidelines: from 1996 to 2019, the mean length of site-specific NCCN guidelines grew by more than 700%, from 26 to 198 pages. 8 A survey of physicians across specialties identified that 60% of physicians identify the length and complexity of practice guidelines as an inhibitor to both using guidelines in care 56 and patient education. A study by Kaupp et al. 57 identified that fewer than 50% of oncology patients are satisfied with the education they receive about their medication options, and a related study outlined how educational gaps contribute to reduced patient empowerment in the community hospital setting. 5
Similar issues inhibit enrollment in clinical trials. While cancer patients who participate in a clinical trial are likely to have improved outcomes,
58
recent estimates show that only 7% of U.S. cancer patients participated in a trial
59
and 20% of oncology clinical trials fail due to low enrollment.
60
Some of these problems are because structural issues in trial design contribute to access disparities that bias trials toward enrolling patients healthier than average
61
while excluding women
62
and ethnically underrepresented patients.
63
While approaches that use AI and real-world data can help to design trials that are more likely to enroll,63,64 patients encounter information and workflow gaps that contribute to them not enrolling in a trial:
Figure 3 depicts the decision-making process that patients go through when they are considering a clinical trial. At each step of the flowchart, we outline typical questions that a patient is likely to consider, and give an example reason that a patient may not proceed to enroll after that step.
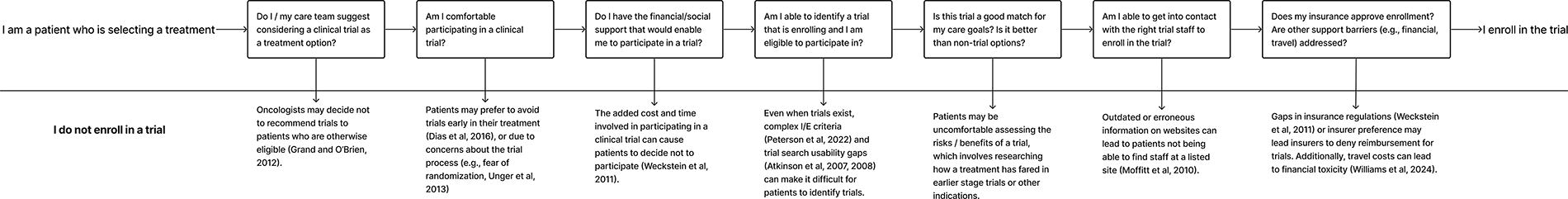
Even when patients have trials that are a match, they face numerous barriers that can lead to them not enrolling. While technology solutions often focus on the problem of matching patients to trials,52,71 it is critical to also focus on financial gaps and technology measures that can help patients to better understand the clinical research process and the pros/cons of participating in a trial.
Testing access and test interpretation
While the rapid growth in the number of therapeutics available gives new choices to patients and clinicians, taking advantage of these choices is far more difficult. Deciding on the correct therapy has caused an exponential increase in clinician workload, as it requires the integration of insights about both molecular biology and a patient’s treatment and overall health history.4,73 As a consequence, real-world usage of precision medicines remains low, with estimates showing that up to 60% of eligible patients fail to receive optimized treatment due to the failure to both test biomarkers and to interpret those biomarker results correctly. 2
While the suboptimal usage of precision medicines is partially driven by workflow issues that can be addressed through clinical informatic enhancements, 74 interpretation gaps cause approximately 25% of patients who receive biomarker testing to fail to start an optimized treatment. 2 Even if testing is properly ordered and returned on time, the correct understanding of biomarker testing results depends on having a comprehensive understanding of both pathology and molecular biology. 75 Biomarker testing results are typically explained to patients by their oncologist, who is unlikely to have received training in genetics/genomics.76,77 This training gap is complicated further by the design of typical clinical genomic reports: these reports are written to give diagnosticians accurate technical information while complying with applicable regulation. While this is critically important, conventional diagnostic report formats are hard for patients to understand 78 and create workflow problems for oncologists, as reports are often not integrated into the EHR. 79
Even when correct biomarkers are tested, discordant test results can result when biomarker test results are generated by orthogonal methods (e.g., PD-L1 quantification via FISH vs. bulk RNA-seq
80
) or next-generation sequencing (NGS) tissue sources.
81
While the FDA’s companion diagnostic (CDx) pathway is used to jointly approve a biomarker test alongside a medication,
82
there may be multiple—potentially discordant—CDxs for a single biomarker, or non-CDx laboratory developed tests (LDTs) that diagnosticians may choose. For an example of how this can cause biomarker status discordance that can impact decision-making, we can look at the biomarkers associated with the two pan-solid tumor approvals of pembrolizumab,83,84 which can be tested with orthogonal diagnostic tests:
Continuous biomarkers such as TMB or PD-L1 are likely to pose a greater concordance challenge than categorical variants. Biomarker testing concordance is likely to be high in well-characterized genes (such as BRCA1/2 92 ) and in genes where there are single-locus changes that have therapy associations (e.g., KRAS G12C 93 ). However, biological factors (such as clonal hematopoiesis of indeterminate potential, CHIP 94 ), sample collection differences (such as sequencing a solid tissue biopsy vs. a “liquid biopsy”81,95), and technical factors (such as bioinformatic filtering 96 ) can lead to discordance in the determination of categorical biomarker status.
Care fragmentation and time pressures
Patients are likely to see multiple clinical practitioners within their treatment, either within a single institution or across institutions. Cancer patients regularly need cross-specialty care for comorbidities, 97 and are likely to work with both oncologists and oncology-specialized advanced practice providers. 98 Coordination errors within a patient’s care team are a primary reason that treatment gets delayed, 99 and can contribute to worse survival outcomes for cancer patients with multiple chronic conditions. 100 While the broader adoption of EHRs has made it easier to share health data between practices, patients encounter integration gaps driven by emerging health information exchange technologies 101 and conflicting data standards.102–104
Process and clinician-to-clinician communication gaps also present an issue, especially during transitions in care between primary care providers (PCPs) and oncologists at the beginning and end of treatment. Gaps at the start of care can contribute to worse outcomes for patients with chronic conditions. Managing drug–drug and drug–comorbidity interactions becomes both more complex and more important as the therapeutic landscape in oncology shifts to therapies targeted at specific molecular pathways and immune signatures, requiring increased review by pharmacists and primary care practitioners. 105 Gaps at the end of active treatment can delay transitioning to survivorship 40 or palliative 106 care.
Time pressure exacerbates this problem. Cancer patients, especially those with advanced tumors, face an unusual paradox: while they spend approximately three hours every four days obtaining care, 107 they typically have only an average of 23 minutes for office visits with their oncologist in the United States. 108 While dedicated navigation supports can help to mitigate these problems and improve patient outcomes, 109 tools for shared decision-making are essential to help patients, caregivers, and all of the clinicians involved in their care—which may include oncologists, oncology nurses, surgeons, pathologists, psychiatrists—make informed decisions together.
Reimbursement and paying for care
Uncertainties about the cost of care influence patients’ choices throughout their journey. While American regulators have issued regulations in support of health care price transparency, their impact on cancer care has been limited because cancer centers are often not in compliance with these statutes 6 and many common oncology therapies are not included in pricing lists. 7 In addition, price transparency laws cover hospitals but not care given in ambulatory settings, or diagnostic tests processed by nonhospital laboratories (such as commercial genomic panels).
Patients and oncologists may choose to forgo diagnostic testing due to concerns about reimbursement 110 or overall cost, 91 as the cost of large clinical NGS panels can approach $5,000. 111 This cost becomes a significant consideration to a patient who is choosing between an array of diagnostic testing panels (e.g., selecting between multiple providers of a tumor/normal matched sequencing panel). Patients and oncologists may struggle to accurately assess the value of a test, given that the value depends on both the sensitivity of the test for detecting key biomarkers coupled with the likelihood that a patient will be matched to optimized treatment after testing. 112
The cost of care also has a significant impact on the treatments that patients pursue. Recent estimates place the monthly out-of-pocket costs of an American cancer patient at $180–2,600 per month. 113 These costs can rise significantly if a patient is on a medication with a high copay; the FDA’s Project Optimus has recently drawn attention to sotorasib114,115: sotorasib costs approximately $21,000 per month, with patients reporting out-of-pocket costs of up to $3,000 per month. While patients may be able to access programs that reduce out-of-pocket costs, such as free medication and medication copay assistance programs, these programs require additional administrative work, which some hospitals support through financial navigation and counseling programs. 9 However, these programs are most likely to be deployed in well-resourced academic hospitals, which are less likely to serve patients facing financial distress. 116 Patients considering clinical trials face similar concerns, driven by uncertainty over insurance coverage of clinical trials. 14
The Solution Concept: Applying AI Through Software Agents to Enable Patients and Caregivers to Be Copilots
If we apply Clayton Christensen’s theory of disruptive innovation 27 and Moor et al.’s 50 three common patterns for generalist medical AIs to the patient challenges articulated earlier, we can envision an AI-enabled patient-centric oncology model where well-informed personal software agents will work on behalf of patients and caregivers to improve patients’ and caregivers’ experiences and patients’ outcomes. Low-cost software agents could help patients and caregivers to both set and achieve well-informed care goals (including outcome, quality of life, and financial factors) by providing the information and workflow assistance needed to help patients choose and integrate health care and supportive services. In this way, AI-powered consumer-facing information and advisory services, can augment and support a patient’s existing medical team.
It is useful and important work to pursue AI-enabled solutions that will try to fix the many systemic breakdowns and opportunities for process improvement within the incumbent health care industry institutions. Since the health care system is composed of many humans working at a vast scale, human factors contribute to the challenge of reforming health care. Beyond workload and education issues, factors such as preference/attitude asymmetry,16,40 bias, 117 provider training gaps and discomfort, 118 and relationship comfort, 119 make it difficult to redesign the clinical trial, care delivery, and reimbursement processes in a patient-centric manner. The careful design of health care information technology that centers patients’ and caregivers’ needs can improve care outcomes. 120
To date, the development of benchmarks for medical question/answer tasks121–123 has driven rapid improvement in the accuracy of knowledge of generalist medical AIs. However, these benchmarks do not necessarily translate to clinical workflows, which may involve multiple steps and involve reasoning over private data stored in an EHR. 124 To maximize the utility and safety of a generalist AI that patients might use, we need to develop a good understanding of the tasks the generalist AI might be used for, and characterize what a good answer might look like. To further flesh out this solution, we use the “jobs to be done” framework, 125 which was developed by Christensen as a complement to the theory of disruptive innovation. 27 Roughly, this framework describes situations where a customer “hires” a product to complete a task.
As an example, consider a cancer patient getting a scan to determine the progression of the disease. Patients commonly experience “scanxiety,” which is distress or anxiety occurring before, during, and after cancer-related imaging or scans. 126 Since the “information blocking” prohibitions enacted by 2021's 21st Century Cures Act require that patient notes (including imaging reports) are released promptly to patients via a patient portal, there is evidence of a significant increase in the number of patients who review their imaging reports. 127 While current guidelines for imaging reports focus extensively on the technical detail and structure of the reports, they typically do not focus on how patients may interpret the report, which can lead to additional patient anxiety. 128
Earlier this year, one of the authors received a pathology report that he struggled to interpret due to the complex anatomical terms and cancer-specific shorthand. He wanted to test whether generative AI could help. He “hired an LLM” by copying and pasting his full pathology report into ChatGPT with a prompt to “translate into seventh-grade English,” as has been described in the literature. 129 The LLM helped to make the report understandable, and a review of the transliteration by several doctor friends confirmed that the AI translation was accurate. The author was put at ease: he understood what the report said. That said, he still needed more expert interpretation from his oncologist to know how good or bad he should feel about the results, and the process involved a manual step of copying his medical data into ChatGPT.
While large technology companies are developing high performing general purpose generative AI models such as Microsoft/OpenAI GPT family, Google’s Gemini, the Anthropic Claude model, and Meta’s Llama, their business models will steer them to offer services that are viable as products for a billion or more customers. Thus, cancer care is likely to be too narrow of a market for them to develop specific tools, limiting accuracy and thus utility. 130 Health care-specific LLMs have advanced rapidly in both capability and accuracy, through the progression from early text-focused models such as ClinicalBERT, 131 BioGPT, 132 and GatorTron, 133 to recent multimodal models such as MedFlamingo, 51 Polaris, 15 and MedGemini. 134 However, they will still require customization to converse with patients of disparate knowledge levels, and integrate with medical systems of record (e.g., the EHR).
In the remainder of this section, we map the challenges that patients face to “jobs to be done,” and highlight existing point solutions that map to these tasks. We aim to augment and extend other recent reviews (e.g., Bitterman et al. 135 ) by highlighting the potential benefits to patients of using AI to help automate these tasks, the prior art (both models, services, and evaluations), the data sources that these algorithms might be trained on, the health care technology integrations that these tools may need, and the task-specific risks that AI operating in these areas may encounter. A more general discussion of the regulation and validation is included in the following section of this article.
Enable 24/7 support for patients through AI-supplemented care navigation services
Cancer patients need multidisciplinary care: a core care team will likely include a surgeon, a radiation oncologist, a medical oncologist, a pathologist, and a radiologist. Patients can also benefit from working with a social worker, 136 a mental health specialist, 137 and a pharmacist. 138 Working with a large multidisciplinary team can cause care fragmentation, so software agents and AI tools can help patients to set treatment goals, share insights about their care with their medical team, and advocate for services that help improve their psychosocial state and minimize time toxicity (e.g., access telemedicine services wherever appropriate 139 ). Table 1 describes example jobs-to-be-done that assist patients with care navigation.
Patient Navigation “Jobs-to-Be-Done” That Can Benefit from Agentic AI
We expect that tools in this space are most likely to have the “form factor” of a personalized software agent, a la ChatGPT. While tools in this category will rely on curated medical knowledge, we defer a detailed discussion of this to the following subsection. The successful implementation of these tools is likely to depend on the following integration points:
Real-time patient support tools face interesting safety and accuracy challenges. While some workloads are unlikely to embody significant safety risks (e.g., searching for doctors, health proxy planning), patients and caregivers may use care monitoring and care-team communications tools during emergent situations, such as a patient experiencing a treatment-related adverse event. While there is evidence that digital platforms can improve the management of critical health events such as adverse events, 151 if an AI tool provides incorrect guidance or neglects to inform a health care practitioner of the patient’s adverse event concerns, this would cause clear risk to patients. Tool development and assessment are starting to place a deeper emphasis on safety and trust that goes beyond model accuracy, 15 but a further characterization of failure modes for AI-enabled patient communication platforms is necessary.
Empower patients in evidence-based treatment decision-making through the use of AI and health care interoperability technologies
Some of the most lauded example use cases for AI in medicine are minimizing diagnostic errors 152 and improving treatment selection performance. 12 AI can examine a patient’s tumor DNA and pinpoint the genetic mutations causing the cancer with the goal of suggesting a personalized, evidence-based treatment strategy directed at the patient’s molecular profile. 153 Furthermore, generative AI can help doctors and health care providers to accurately predict patient outcomes. 154 Learning from large data sets of patients, it can uncover patterns linked to specific outcomes and help make informed decisions. Table 2 describes example “jobs-to-be-done” that support patients in understanding their diagnostic and treatment options.
Clinical Decision and Evidence Interpretation “Jobs-to-Be-Done” That Can Benefit from Agentic AI
Use cases in this category rely heavily on the ability to accurately reason about complex medical tasks. This drives a strong dependence on having up-to-date medical data: Patients and caregivers will access continuously updated medical knowledge bases (such as WebMD, but curated to meet the specific needs of the rapidly evolving field of oncology) to make it easy to answer questions (including quality-of-life side effects), and provide the sources and experts to bring to their clinicians. These use cases are likely to harness integrations focused on:
Applications in this category face the strongest safety risk, as the decisions surfaced by an AI tool are likely to directly impact the treatment that a patient receives. Multiple different regulatory pathways cover AI algorithms in this area. The integration of the AI tool into health care will influence whether it is likely to be considered an AI Clinical Decision Support tool, 174 AI-powered software-as-a-medical-device, 175 or be covered under non-software pathways, such as the LDT regulations. While these three pathways are all regulated by the FDA in the United States, they are seen as presenting different risk levels and thus have differing validation requirements before clinical use.
Streamline reimbursement, care coordination, and trial access by leveraging agent technologies
Access to financial assistance programs and clinical trials often involves complex processes and additional paperwork. By using agents to help automate these processes, 176 we can reduce the time and financial burden that patients face, while also closing understanding gaps that stand in the way of truly informed consent. 16 This will help to reduce financial and time toxicity, while also enabling more patients to access clinical trial. 14 Table 3 shows example “jobs-to-be-done” that can support patients with reimbursement and care access workflows.
Reimbursement and Access “Jobs-to-Be-Done” That Can Benefit from Agentic AI
Since these use cases are primarily nontreatment and aim to reduce time/financial toxicity to patients, these use cases generally present a low patient safety risk (with the notable exception of the clinical trial matching use case, which is similar in risk to the diagnostic/treatment selection workloads). We expect that use cases in this category will place an emphasis on data normalization (e.g., helping to avoid the overhead of repeated patient data entry into consent forms) and cross-system integration. Given that many of these workflows may lead to health data being sent to a third party (e.g., records may be sent to a patient assistance program, a clinical trial site, or a third-party patient health record), they face a unique risk of inadvertent release of a patient’s health data. As such, it is critical that these tools manage patient data in a way that both complies with HIPAA’s Privacy Rule by ensuring that patients are adequately informed and consent to health data sharing, and also ensuring that patients can access their health data in an unencumbered manner under the scope of both HIPAA law and the 21st Century Cures Act’s “Information Blocking” prohibitions. 185
Call to Action: A Roadmap for Using Data and AI to Enable Democratized, Patient-Led Care
Health care AI is at a critical movement. Using AI as an automation tool has the potential to dramatically reduce the cost of providing accurate, well-contextualized health care information and advice. As the cost of providing personalized health care services drops, new business and engagement models become feasible and sustainable. In health care, this can shift power to consumers (patients and caregivers), who can receive better outcomes as a result of care that is personalized to their goals, values, and history, while creating a learning health system where care improvement opportunities are identified proactively and patients and doctors can learn from the experiences of “patients like me.”
However, previous solutions focused on using data and AI to bring forward personalized care have encountered obstacles. These obstacles still need to be addressed for new health care AI:
Given the magnitude of the benefits of personalized care—especially better patient outcomes, a health care revolution leveraging AI-enabled software agents to personalize care is possible. We believe that advocacy along two avenues is necessary to accelerate this revolution: (1) developing collaborations that leverage the unique strengths of health care providers, new technology entrants, and technology platforms; and (2) a political movement that ensures that health AI regulation meets both patients’ safety and access needs.
Achieving ubiquitous distribution of AI-enabled consumer health services through cooperation between health care, technology startups, and technology platforms
Much investment in the development of novel AI technologies is driven through the “hyperscaler” technology platform companies, such as Google/Alphabet, Amazon, Apple, Meta, and Microsoft. The products of these companies are data and services, which they sell through their various platforms. In health care, the platform services developed by a technology platform provider are typically not used at the point-of-care: a health care institution must build its own clinical informatics offering using the hyperscaler’s platform. 189 Therefore, the technology giants are likely to develop generalist tools for health care that serve a billion or more customers. Given their existing investments in AI, coupled with their key resource advantages (capital, technical staff, and computational), they can develop state-of-the-art health care foundation models, both in specific modalities (e.g., GigaPath 190 ) and for generalist tasks (e.g., MedGemini, Saab et al. 134 ). Emerging startups will build focused AI-enabled tools using the generalist AI platforms.
While platform companies can benefit from scale, the converse is also true: developing a patient/clinician facing tool to provide information and advice on a specific cancer will be too small of a market for them to want to serve themselves, 189 especially if most of the value accrues to the organization delivering the health care service, instead of the technology. 191 As a consequence, integrating AI into the point-of-care will need to be done by other parties. For instance, implementing LLM-based trial matching has been done by hospital-affiliated academics 71 and technology startups (e.g., Triomics OncoLLM for trial matching 52 ). This exposes a safety/access dichotomy that is similar to the discretion provided to hospital laboratories under the new FDA final rule regarding the regulation of “LDTs” 192 : while analyses developed within a specific hospital may be safer because they are developed with greater understanding of the hospital environment, this also can restrict access to necessary developments, especially for patients with rare diseases, where there may not be local clinical expertise. 193
To allow all patients to benefit from health AI, regardless of resources or clinical setting, we will need to identify approaches that allow these tools to become ubiquitous. This could take numerous forms, between open science collaborations with the goal of openly developing and disseminating best practice methods standards (e.g., GA4GH 194 ) reforming health care reimbursement to lower the barrier-to-entry for health care technology startups,31,32 or coming up with novel partnership models between hospitals, technology companies, and potentially third parties such as pharmaceutical or diagnostic companies. Another form could be building on the consumer services of the technology hyperscalers: they are likely to support new service providers who build on top of their platforms, for example, for supporting patients and caregivers with a particular cancer, such as lymphoma. Since these new service providers will use the hyperscaler’s cloud services, the hyperscalers may want to invest in these emerging service providers to increase use of their IT services, a dynamic seen in the 2010s with bioinformatic service providers building on the major clouds.
Ensuring robust and rapid adoption of AI-enabled software tools that touch patients directly through sensible regulation
The adoption of personal AI-enabled software agents brings the opportunity to dramatically improve the accessibility of high-quality health care, especially in lower resource settings, where there are fewer human experts and more language and health literacy barriers. 195 However, the current regulatory environment appears to favor the creation of regulatory regimes that may slow the adoption of patient-facing AI applications, 196 by either favoring “lower risk” back office applications 197 or by enacting practice guidelines that may create barriers that may limit the adoption of AI technologies. 198
It is worth drawing comparisons between the current moment for AI in medicine and the recent history of the adoption of biomarker testing in oncology. A recent survey of oncologist attitudes toward AI conducted by Hantel et al. 187 indicated that 53% of oncologists surveyed had not received training on the use of AI in health care and that 93% felt that they needed additional education. These numbers are remarkably close to the results of a 2023 survey covering UK oncologists’ attitudes toward NGS 77 that indicated that 39% of oncologists had not received training in interpreting NGS results and that 93% felt that they needed additional training in genomics and genetic counseling.
Ultimately, the health care industry and regulators have an ethical imperative to proceed wisely while integrating the perspectives of the patients and caregivers who consume the health care system’s services. Current regulatory proposals put a very strong emphasis on safety.
199
While validating the safety of novel AI tools is critical, regulation should balance the harm of inaccuracy with the harms of delaying patient access to helpful tools,
200
and be mindful of how regulation can create barriers to market entry at the cost of consumers, such as the Epic/Cerner EHR duopoly resulting from the HITECH act.
188
To maximally deploy health AI in a way that improves patient-centricity through personalization, the health care system needs to chart a pragmatic course to deploying AI:
The political and regulatory environment will focus on the needs of patients and caregivers if institutional health care puts in the will to work closely with patient-centric groups. For example, the National Cancer Act in 1971, the Americans with Disabilities Act of 1990, the National Cancer Survivorship Act of 1996, ACT UP (
Conclusion
In this review article, we have identified key use cases where AI has the potential to dramatically improve how patients and caregivers experience cancer care. AI-enabled services have the potential to address challenges patients and caregivers face as they go through receiving and understanding a cancer diagnosis, 5 selecting a treatment plan,4,73 and then living with cancer, enabling them to become copilots with their medical team. As a contrast to “sustaining” innovations that make “back office” services of health care operationally more efficient, using AI-enabled software agents to support personalized care and the patient and caregiver experience has the potential to disruptively impact oncology care by allowing all patients and caregivers to access personalized care and navigation services, regardless of health literacy or resource barriers.
Footnotes
Authors’ Contributions
F.N.: Conceptualization, Writing—original draft, Writing—review & editing. B.P.: Conceptualization, Writing—original draft, Writing—review & editing.
Author Disclosure Statement
Brad is a founding member of ennov1; an advisor to 4DPath, Alva10, Cancer Commons, Consuli, Rabble Health, and Travera; and is an active contributor to the Personalized Medicine Coalition. Frank: Employment and stock ownership:
Funding Information
No funding was received for this article.
