Abstract
HIV-associated wasting (HIVAW) is an underappreciated AIDS-defining illness, despite highly effective antiretroviral therapy (ART). We (a) assessed the association between incident HIVAW/low weight and all-cause mortality and (b) described virologic outcomes after people with HIV (PWH) experienced HIVAW/low weight while on ART. In the Observational Pharmaco-Epidemiology Research & Analysis (OPERA®) cohort, PWH without prior HIVAW/low weight who were active in care in 2016–2020 were followed through the first of the following censoring events: death, loss to follow-up, or study end (October 31, 2021). HIVAW/low weight was a diagnosis of wasting or low body mass index (BMI)/underweight or a BMI measurement <20 kg/m2. Hazard ratios (HRs) and 95% confidence intervals (CIs) for the association between time-dependent HIVAW/low weight and mortality were estimated with extended Cox regression models. Over a median follow-up of 45 months (interquartile range: 27, 65), there were 4,755 (8%) cases of HIVAW/low weight and 1,354 (2%) deaths among 62,314 PWH. PWH who experienced HIVAW/low weight had a significantly higher risk of death than those who did not (HR: 1.96; 95% CI: 1.68, 2.27) after adjusting for age, race, ethnicity, and changes in viral load (VL) and Veterans Aging Cohort Study Mortality Index scores over follow-up. Among 4,572 PWH on ART at HIVAW/low weight, 68% were suppressed (VL of <200 copies/mL); subsequent virologic failure was uncommon (7%). Among viremic PWH, 70% and 60% achieved suppression and undetectability (VL of <50 copies/mL), respectively, over follow-up. HIVAW remains a challenge for some PWH. Particular attention needs to be paid to HIVAW/low weight and virologic control to restore health and potentially reduce the risk of death.
Introduction
Human immunodeficiency virus (HIV), which was previously a life-threatening illness, has now become a manageable chronic disease. As a result of developments in antiretroviral therapy (ART), people with HIV (PWH) have experienced improvements in quality of life and prolonged survival. 1,2 In the North American AIDS Cohort Collaboration on Research and Design, the average life expectancy for a 20-year-old PWH on ART increased from 36.1 years in 2000–2002 to 51.4 years in 2006–2007. 3 A recent meta-analysis of four cohort studies in high-income countries reported life expectancies of an additional 43 years (95% confidence interval [CI]: 42, 44) and 32 years (95% CI: 31, 33) for PWH on ART at ages 20 and 35 years, respectively; life expectancies increased over time between 1996 and 2011. 4 However, as PWH live longer, they remain at risk of various comorbidities, some related to their HIV infection or use of ART. Indeed, an analysis of Kaiser Permanente California data from 2000 to 2016 showed that PWH lived almost 10 fewer years without major comorbidities than people without HIV infection. 5 An analysis of 18 European and North American cohort studies in the Antiretroviral Therapy Cohort Collaboration reported that PWH who initiated ART in 1996–1999 and survived ≥10 years most commonly died due to non-AIDS cancer, AIDS, cardiovascular disease, and liver disease. 6
HIV-associated wasting (HIVAW) is defined as progressive, involuntary weight loss among PWH, 7 characterized by a loss of both fat and metabolically active lean tissue. 8 It was a frequently occurring syndrome prior to the development of highly effective ART and was included as an AIDS-defining illness by the Centers for Disease Control and Prevention (CDC) in 1987. 9 It was defined by the CDC as an involuntary and unexplained loss of >10% of baseline body weight plus diarrhea or weakness with fever for ≥30 days. 9 However, numerous criteria related to diagnoses, weight loss, and treatment for HIVAW have been utilized in the absence of a standard definition or objective measure for identifying HIVAW. In addition, there have been few observational studies on the burden of HIVAW among PWH over time. In the United States, two claims studies and one observational cohort study have reported period prevalence estimates of HIVAW in the past 20 years (i.e., modern era of highly effective ART): 8% in 2005–2007 10 and 18% in 2012–2018 11 in the claims studies and 12% in both 2012–2015 and 2016–2020 in the cohort study. 12 HIVAW, weight loss, and low muscle mass have all been associated with increased mortality risk among PWH. 13 –15
As part of a comprehensive evaluation of HIVAW in the Observational Pharmaco-Epidemiology Research & Analysis (OPERA®) cohort, 12 we aimed to evaluate the association between incident HIVAW and all-cause mortality and to describe the achievement or loss of viral suppression after incident HIVAW while on ART.
Materials and Methods
Study design and population
The OPERA cohort’s database contained prospectively captured, routine clinical data from the electronic health records (EHRs) of 140,817 PWH from over 100 clinic locations across 22 U.S. states and territories at the time of this study, representing approximately 13% of people living with diagnosed infection in the United States. 16 OPERA complies with all Health Insurance Portability and Accountability Act and Health Information Technology for Economic and Clinical Health Act requirements and has received annual institutional review board (IRB) approval by Advarra IRB, including a waiver of informed consent and authorization for use of protected health information.
This study included PWH, 18 years of age or older, who had at least one visit in OPERA from January 01, 2016 through December 31, 2020. Eligible PWH also had no malignancy (except basal or squamous cell carcinoma or in situ cancer) within 3 years prior to the date of eligibility (baseline), no AIDS-defining opportunistic infection (OI) within 12 months prior to baseline, and no HIVAW/low weight prior to baseline. The baseline date was defined as the first OPERA visit in 2016–2020 at which an individual met all eligibility criteria.
Key study measures
HIV-associated wasting/low weight
Because there is no standard definition of HIVAW and our criteria differ from the CDC’s original definition, we refer to HIVAW as HIVAW/low weight in this study. HIVAW/low weight included a wasting or low BMI/underweight diagnosis (via International Classification of Diseases [ICD] codes or diagnosis title search) or a BMI vitals measurement <20 kg/m2. To ensure that HIVAW/low weight was not attributable to an underlying condition known to result in wasting or excessive weight loss, PWH who experienced malignancy within 3 years or an AIDS-defining OI within 12 months prior to the date of HIVAW/low weight were not classified as having experienced HIVAW/low weight.
Veterans Aging Cohort Study Mortality Index
The Veterans Aging Cohort Study (VACS) investigators developed a composite score 17,18 to predict mortality among PWH, including those who experience HIVAW/low weight. Although the score includes typical predictors of mortality risk among PWH (age, CD4 cell count, and HIV viral load), it also includes markers of organ system injury that are routinely monitored in PWH: hemoglobin, Fibrosis-4 index for liver fibrosis (platelets, aspartate, and alanine transaminase), estimated glomerular filtration rate for kidney function (creatinine), and co-infection with hepatitis C virus. Of note, this index predicts mortality independent of ART use and can be utilized among PWH with an undetectable HIV VL. The VACS score has been shown to be associated with inflammatory biomarkers 19,20 and is a measure of physiologic frailty. 21
Statistical analysis
All-cause mortality
Eligible PWH were followed through the first of the following censoring events: death from any cause, loss to follow-up (12 months after last clinical contact), or study end (October 31, 2021). The median and interquartile range (IQR) for the time from baseline to death and from incident HIVAW/low weight to death were computed. Hazard ratios (HRs) and 95% CIs for the association between HIVAW/low weight and all-cause mortality were estimated with extended Cox regression models, which allow for time-dependent variables. The adjusted model included age at baseline, race, ethnicity, and time-dependent covariates (log10 VL, VACS Mortality Index score); covariates were identified a priori using directed acyclic graphs. 22 Linear and quadratic terms of age, log10 VL, and VACS score were included. Incident HIVAW/low weight was a time-dependent exposure. Thus, someone diagnosed with incident HIVAW/low weight 8 months after baseline contributed 8 months of person-time to the unexposed group (no HIVAW/low weight), and the remainder of their follow-up time was attributed to the exposed group (incident HIVAW/low weight). This helps to prevent immortal time bias, where individuals in the exposed group would be obliged to survive longer than those in the unexposed group by definition. 23 Similarly, changes in VL and VACS Mortality Index scores measured over follow-up were incorporated in the extended Cox regression model; each PWH included a row of data that represented a period of time in which the exposure and covariates were constant, adding up to the full amount of time they were followed.
Virologic outcomes
In a separate analysis of the same eligible PWH at baseline, we identified a subset who were on ART at the time they experienced incident HIVAW/low weight over follow-up. They were characterized as viremic (HIV VL of ≥200 copies/mL) or suppressed (HIV VL of <200 copies/mL) based on the VL measurement on or within <12 months prior to the date of incident HIVAW/low weight. These PWH with incident HIVAW/low weight were then followed through the first of the following censoring events: virologic outcome of interest, discontinuation of ART (>45 days without any ART), death, loss to follow-up (12 months after last clinical contact), or study end (October 31, 2021).
Among viremic PWH, achievement of virologic suppression was defined as the first HIV VL <200 copies/mL over follow-up, and achievement of virologic undetectability was defined as the first HIV VL <50 copies/mL over follow-up. Among virologically suppressed PWH, virologic failure was defined as two consecutive VLs ≥200 copies/mL or an ART regimen switch/discontinuation following a VL ≥200 copies/mL. The date of the confirmatory event (second VL of ≥200 copies/mL or ART regimen switch/discontinuation) was used as the date of virologic failure. The proportions of PWH experiencing virologic undetectability, suppression, and failure were computed, as were the medians and IQRs for time from incident HIVAW/low weight to the virologic outcome. Univariate Poisson regression was used to obtain the incidence rates and 95% CIs of the virologic outcomes of interest.
Results
Study population
There were 67,119 PWH with no prior HIVAW/low weight who were eligible for the study. The study population was young (median age: 41 years, IQR: 31, 52) and predominantly male (82%); a substantial proportion of PWH were Black (47%) or Hispanic (22%). At baseline, the median number of months since HIV diagnosis was 57 (IQR: 2, 156) and a majority of PWH (78%) were ART-experienced (Table 1).
Baseline date = First OPERA® visit in 2016–2020 at which person with HIV was ≥18 years of age, had no malignancy within 3 years prior to the visit (except basal or squamous cell carcinoma of the skin or in situ cancer), and had no AIDS-defining opportunistic infection within 12 months prior to the visit.
Wasting diagnosis (ICD codes or title search), low BMI or underweight diagnosis (ICD codes, title search) or BMI <20 kg/m2 (vitals measurement).
4,805 (7%) of the full study population were not included in the models because of missing data in the included covariates (age, race, ethnicity, viral load, and VACS Mortality Index score).
ART, antiretroviral therapy; HIV, human immunodeficiency virus; HIVAW, HIV-associated wasting; IQR, interquartile range; mL, milliliter; n, number; µL, microliter; VACS, Veterans Aging Cohort Study.
All-cause mortality
Over a median of 44 months of follow-up (IQR: 26, 65), 5,052 (8%) PWH experienced incident HIVAW/low weight (Table 2). Only 2% of eligible PWH died (n = 1,421); most PWH were followed through the end of the study (n = 47,216; 70%) (Table 2). A greater proportion of PWH who experienced incident HIVAW/low weight died over follow-up (5%; 228 of 5,052 PWH) than PWH who had not experienced incident HIVAW/low weight (2%; 1,193 of 62,067 PWH). There were 4,805 (7%) PWH who were not included in the extended Cox regression models because of missing covariate data. We evaluated whether the subset of the study population included in the modeling (n = 62,314) was different from the overall study population (n = 67,119); baseline demographic and clinical characteristics were nearly identical (Table 1).
HIVAW/Low Weight a and All-Cause Mortality in the OPERA® Cohort, January 2016 Through October 2021
Wasting diagnosis (ICD codes or title search), low BMI or underweight diagnosis (ICD codes, title search) or BMI <20 kg/m2 (vitals measurement).
4,805 (7%) of the full study population were not included in the models because of missing data in the included covariates (age, race, ethnicity, viral load, and VACS Mortality Index score).
Date of loss to follow-up is 12 months after last clinical contact.
Hazard ratios and 95% confidence intervals for the association between incident HIVAW/low weight (time-dependent exposure) and all-cause mortality (outcome) were estimated with extended Cox regression models.
Adjusted for age at baseline, race, ethnicity, and time-dependent covariates (log10 viral load and VACS Mortality Index score); age, log10 viral load, and VACS score were modeled as continuous variables with linear and quadratic terms.
CI, confidence interval; HIVAW, HIV-associated wasting; HR, hazard ratio; IQR, interquartile range; n, number; N/A, not applicable.
Among the 62,314 eligible PWH with complete covariate data (model study population), a similar median follow-up time of 45 months (IQR: 27, 65) and the same proportion (8%) experiencing incident HIVAW/low weight over follow-up were observed as in the full study population. A slightly higher proportion of the model study population (72%) was followed through the end of the study (Table 2). The proportions of PWH who died over follow-up were the same as in the full study population (5% of PWH with incident HIVAW/low weight and 2% of PWH without). After adjusting for baseline age, race, and ethnicity, as well as time-updated VL and VACS Mortality Index scores, PWH who experienced incident HIVAW/low weight were nearly twice as likely to die, compared with PWH who did not experience incident HIVAW/low weight, during follow-up (HR: 1.96, 95% CI: 1.68, 2.27) (Table 2).
Virologic outcomes
In a separate analysis, 4,962 PWH experienced incident HIVAW/low weight over follow-up, and most (n = 4,572; 92%) were on ART at the time of incident HIVAW/low weight identification. The most common core agent class was integrase strand transfer inhibitors (62%), with 23% on dolutegravir, 20% on elvitegravir, and 17% on bictegravir/tenofovir alafenamide/emtricitabine; 13% were taking multiple core agents (Fig. 1). At the time of incident HIVAW/low weight, PWH had been on their ART regimen for a median of 6 months (IQR: 1, 17). Of the 4,065 (89%) PWH with a known date of ART initiation, the median time between ART initiation and incident HIVAW/low weight was 15 months (IQR: 1, 49).
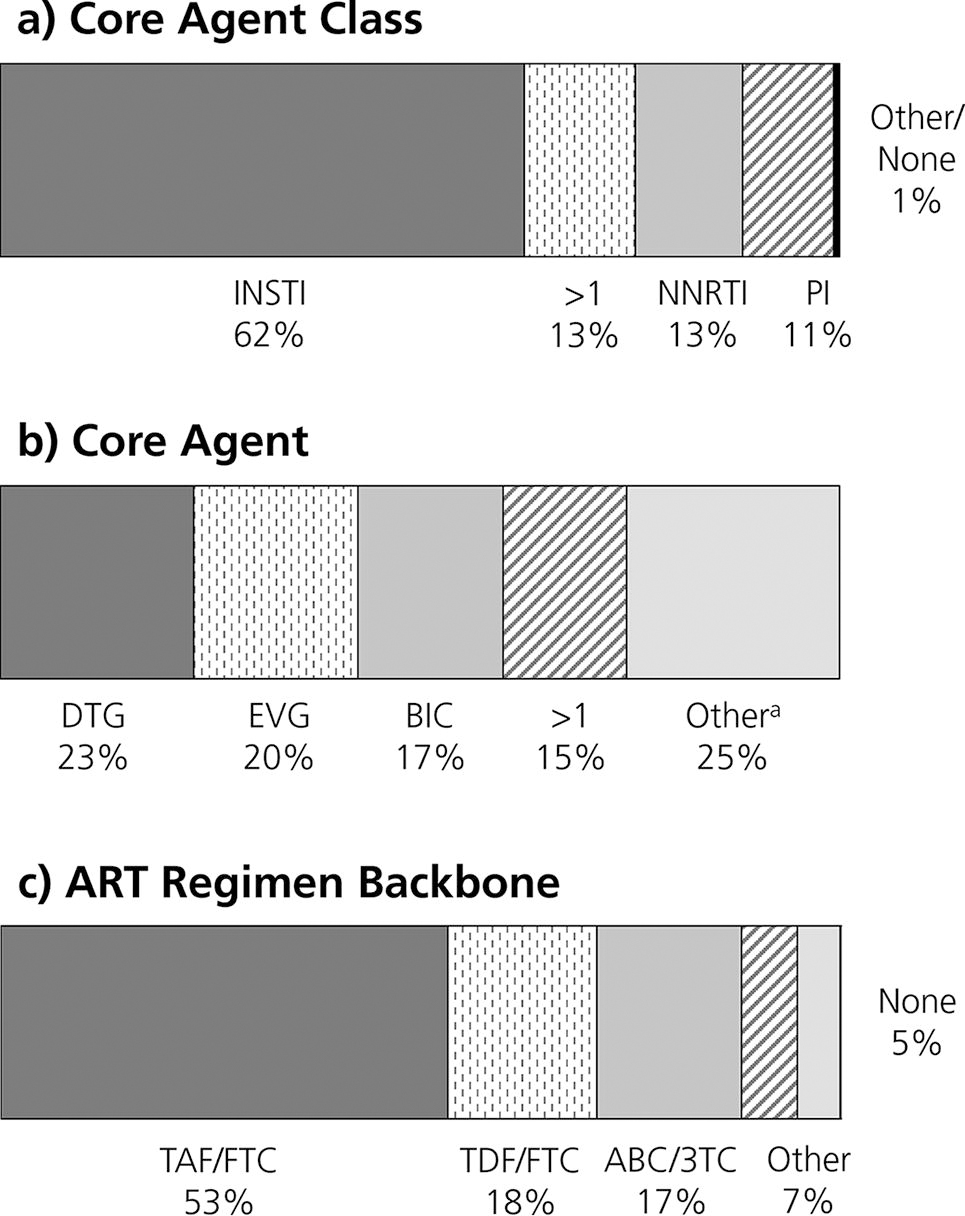
Distribution of core agent classes
Approximately a third of PWH on ART at the time of incident HIVAW/low weight were not virally suppressed (VL of ≥200 copies/mL); 70% achieved viral suppression (VL of <200 copies/mL) a median of 2 months (IQR: 1, 5) after incident HIVAW/low weight, and 60% achieved undetectability (VL of <50 copies/mL) after a median of 4 months (IQR: 2, 8). Of the 68% of PWH who were virally suppressed, only 7% experienced virologic failure after a median of 16 months (IQR: 8, 26) of follow-up (Table 3).
Virologic Outcomes after Incident HIVAW/Low Weight a While on ART in the OPERA® Cohort, January 2016 Through October 2021
Wasting diagnosis (ICD codes or title search), low BMI or underweight diagnosis (ICD codes, title search) or BMI <20 kg/m2 (vitals measurement).
Viral load measurement on or within <12 months prior to the date of incident HIVAW/low weight.
Viral load ≥200 copies/mL.
Viral load <200 copies/mL.
Viral load <50 copies/mL.
Two consecutive viral loads ≥200 copies/mL or a regimen switch/discontinuation following a viral load ≥200 copies/mL.
ART, antiretroviral therapy; CI, confidence interval; HIVAW, HIV-associated wasting; IQR, interquartile range; mL, milliliter; n, number; PWH, people with HIV; VL, viral load.
Discussion
We evaluated all-cause mortality and virologic outcomes after incident HIVAW/low weight in the U.S.-based OPERA observational clinical cohort from January 01, 2016 through October 31, 2021. In the analysis of 62,314 PWH in care without missing covariate data, death due to any cause was an uncommon event (2%). After adjusting for covariates that included time-dependent VL measurements and VACS Mortality Index scores over follow-up, incident HIVAW/low weight was associated with nearly twice the risk for all-cause mortality (HR: 1.96, 95% CI: 1.68, 2.27) compared with remaining free of HIVAW/low weight. In the analysis of 4,572 PWH who were on ART at the time of incident HIVAW/low weight, 32% were viremic (VL of ≥200 copies/mL), of whom 70% achieved suppression (VL of <200 copies/mL) and 60% achieved undetectability (VL of <50 copies/mL) over follow-up. Of the remaining 68% who were already suppressed at the time of incident HIVAW/low weight, only 7% experienced virologic failure over follow-up.
Few studies have specifically evaluated the association between HIVAW and mortality, and most have examined individual aspects of the syndrome such as weight loss. In an analysis of 678 PWH in the Nutrition for Healthy Living Study in 1995–2000, weight loss was the strongest predictor of mortality. In fact, there was a four- to sixfold increase in the risk of mortality with weight loss of ≥10% since baseline/last visit compared with weight maintenance or gain. 14 As with our study, this relationship was independent of ART use. In the Multicenter AIDS Cohort Study, HIVAW among male PWH was associated with shorter survival (9 years, 95% CI: 8, 10) than among male PWH without HIVAW (12 years, 95% CI: 11, 12) or males without HIV infection. Survival was lowest among male PWH who continued to lose weight (5 years, 95% CI: 5, 6), 13 suggesting that interventions to stop continued weight loss over time have the potential to impact mortality risk. Although our study evaluated the hazard of all-cause mortality and did not include weight measurements over time, our findings are still consistent with the finding of a survival difference between PWH with and without HIVAW. Finally, the Fat Redistribution and Metabolic Change in HIV Infection (FRAM) study examined the relationship between skeletal muscle and adipose tissue with 5-year mortality risk among 922 PWH identified in 2000–2002. There was a dose–response relationship with lower arm muscle and increased mortality rates (23%, 11%, and 8% for the lower, middle, and higher tertiles, respectively). Visceral adipose tissue (VAT) was also independently associated with mortality; PWH in the highest tertile of VAT were twice as likely to die than those in the lowest tertile of VAT (odds ratio: 2.1, 95% CI: 1.1, 4.0). 15 Although our study did not evaluate muscle mass and adipose tissue (and these data are not generally available in the EHR), the FRAM study findings are consistent with an association between HIVAW and mortality and offer some insight into the complicated mechanisms through which the increased risk may be operating.
Although there was a median follow-up of almost 4 years, our study population was young (median age: 41 years), making mortality a rare outcome; only 2% of the PWH in this study died over follow-up. In addition, the relationships among HIVAW/low weight, virologic control, ART use, and mortality are complex, and the use of EHR data to investigate these relationships can be challenging. In the all-cause mortality analysis, we adjusted for time-updated VL measurements, both as an adjustment for virologic control as well as a proxy for ART use (i.e., PWH on effective ART tend to have very low VLs). Because many comorbidities can impact mortality risk in various ways, we chose to adjust for the time-updated VACS Mortality Index score; characteristics related to liver and renal function are particularly important for PWH. However, our model did not consider other comorbidities (e.g., cardiovascular disease) or their treatments (including HIVAW/low weight treatment). In addition, the model did not include behaviors that may contribute to the risk of comorbidities and mortality, such as smoking or alcohol use, diet, exercise, or information related to food insecurity, because these data are not consistently available in the EHR. Although residual confounding remains a consideration, it is important to note the nearly doubled risk of mortality with HIVAW/low weight even after adjusting for the most common confounders of the relationship between HIVAW/low weight and mortality. Residual confounding is unlikely to explain the entire association observed. The EHR data, though extensive, is not always complete; 7% of our full study population was excluded from our model population because of missing covariate data, most commonly VL and VACS score. Although the full and model study populations were nearly identical across all characteristics, it is important to consider who was excluded. In this study, a greater proportion of PWH who were lost to follow-up had missing covariate data; it is unclear if these are healthier PWH with stable VLs on a stable ART regimen who are seen less frequently by their healthcare provider or sicker PWH who may have been more likely to die over follow-up. Finally, wasting can be difficult to identify, especially because it can occur among all body sizes. Indeed, a majority of PWH met our criteria for HIVAW/low weight by having a single BMI measurement <20 kg/m2; our identification of HIVAW/low weight may overrepresent younger PWH who are naturally thinner and does not specifically consider weight loss trajectories, which may contribute to mortality risk in a different manner. Treating a syndrome for which there is no objective measure of identification may be a hurdle in clinical settings, especially when HIV primary care is often focused on ART use and virologic control, usually in the presence of other concerning comorbidities.
This study also has several strengths. It is one of the only recent observational studies evaluating HIVAW among PWH and the only study, to our knowledge, to evaluate the association between HIVAW/low weight and all-cause mortality in recent years using EHR data. The OPERA HIV cohort represents approximately 13% of PWH in the United States who are living with diagnosed HIV infection. 16 The large study population of 67,119 PWH without prior HIVAW/low weight in 2016–2020 allowed for a more in-depth look at clinical outcomes that follow incident HIVAW/low weight, which, though relatively uncommon, remains a concern for some PWH. The prospectively collected clinical data from the EHR of these PWH allowed for the adjustment of clinical changes over follow-up that are likely to impact the risk of mortality, namely viral loads which may reflect use of effective ART regimens, and the VACS Mortality Index, which includes several comorbidities that can impact the long-term health of PWH and other markers for mortality.
Between January 01, 2016 and October 31, 2021, both incident HIVAW/low weight and death from any cause were uncommon (7% and 2%, respectively) in a large population of PWH in the United States. However, there was a strong association between HIVAW/low weight and all-cause mortality. Particular attention needs to be paid to incident HIVAW/low weight among PWH, regardless of ART use and the presence of additional comorbidities, to restore health and potentially reduce the risk of death. In addition, it remains important to monitor the virologic status of PWH with incident HIVAW/low weight who are on ART. Although virologic failure was rare among virologically suppressed PWH and both virologic suppression and undetectability were common among viremic PWH, 30% remained viremic throughout follow-up; thus, there is room for improvement. Continuing to monitor and improve upon the virologic control of PWH on ART, especially among those with HIVAW/low weight, should remain a priority. Future studies should further explore the complex relationships among HIVAW/low weight, ART use, virologic changes, and mortality.
Footnotes
Acknowledgments
This research would not be possible without the kind participation of people with HIV and their OPERA® caregivers. The authors are grateful to the following individuals: Kelly Oh (SAS Programming), Robin Beckerman (Quality Assurance), Bernie Stooks and Stephen Connellee (Data Architecture), Lisa Lutzi and Nicole Shaw (Data Management/Quality), and Judy Johnson (Clinical Data Categorization) at Epividian, and Jesus Felizzola (Clinical Expertise) at EMD Serono.
Authors’ Contributions
MBW, RPW, LB, JSF, RKH, and GPF share the responsibility for the conceptualization and design of this study. JSF and GPF helped secure funding for the study. MBW, JSF, RKH, and GPF contributed to the acquisition of data. RPW is responsible for all the analyses. All authors have contributed to the interpretation of results, have critically reviewed and approved the article, and have participated sufficiently in the work to take public responsibility for its content.
Data Availability
The datasets used in this study are not publicly available owing to privacy concerns and the proprietary nature of the database but can be accessed upon reasonable request through the corresponding author to the OPERA® Epidemiology and Clinical Advisory Board. Access to codes may be granted upon request, with parties agreeing to privacy restrictions and technological specifications and requirements.
Author Disclosure Statement
MBW has participated in post-conference advisory boards for the Conference on Retroviruses and Opportunistic Infections (CROI) and International AIDS Conference (IAC) and serves as a principal investigator on ViiV Healthcare clinical trials but does not receive personal compensation for this work, which goes directly to the AIDS Healthcare Foundation. MBW is also a member of the Epidemiology and Clinical Advisory Board for Epividian, Inc. RPW, LB, JSF, and GPF are employed by Epividian, Inc.; Epividian has had research funded by the AIDS Healthcare Foundation, EMD Serono, Gilead Sciences, Janssen, Merck & Co., Theratechnologies Inc., and ViiV Healthcare. JS is a consultant for Merck & Co., AbbVie, and Shionogi. MH, ALP, and BH were employed by EMD Serono at the time of this analysis. RKH has received a research grant from Gilead Sciences; received speaker’s honoraria and is on the advisory boards for ViiV Healthcare, Bristol Myers Squibb, Merck & Co., Gilead Sciences, and Janssen; and is a member of the Epidemiology and Clinical Advisory Board for Epividian, Inc.
Funding Information
This research was funded by EMD Serono (CrossRef Funder ID: 10.13039/100004755).
